Abstract
Although aplastic anemia (AA) does not come under the category of blood malignant diseases, the infection that frequently occurs in this bone marrow failure can make it worse. Pulmonary infection is the most prevalent but limiting clinical diagnosis. To find biomarkers predicting bacterial or bacterial-combined fungal infections in the lungs, we reviewed 287 AA medical records including 151 without any infection, 87 with pure pulmonary bacterial infection, and 49 with bacterial and fungal infection were reviewed. There were substantial changes in IL-17F, IL-17A, IFN-γ, IL-6, IL-8, and IL-10 levels between the non-infected and lung bacterial infection groups (P < 0.05). Further, a significant variation in IL-17A, TNF-β, IL-1β, IL-2, IL-4, IL-6, IL-8, IL-10, IL-22, and IL-12p70, between the uninfected group and the pulmonary bacterial and fungal infection group (P < 0.05) was observed. The results further revealed significant differences in TNF-β, IL-12p70, IL-6, IL-8, and IL-10 between the pulmonary bacterial infection group and the fungal infection group (P < 0.05). Moreover, by calculating ROC and cut-off values, we determined that IL-6 (AUC = 0.98, Cut-off = 14.28 pg/ml, P = 0.0000) had a significant advantage than other cytokines, body temperature (AUC = 0.61, P = 0.0050), PCT (AUC = 0.57, P = 0.0592), and CRP (AUC = 0.60, P = 0.0147) in the detection of lungs bacterial infections. In addition, IL-6 (AUC = 1.00, Cut-off = 51.50 pg/ml, P = 0.000) and IL-8 (AUC = 0.87, Cut-off = 60.53 pg/ml, P = 0.0000) showed stronger advantages than other cytokines, body temperature (AUC = 0.60, P = 0.0324), PCT (AUC = 0.72, Cut-off = 0.63 ng/ml, P = 0.0000) and CRP (AUC = 0.79, Cut-off = 5.79 mg/l, P = 0.0000) in distinguishing bacteria from fungi. This may suggest that IL-8 may play a role in differentiating co-infected bacteria and fungi. Such advantages are repeated in severe aplastic anemia (SAA) and very severe aplastic anemia (VSAA).In conclusion, aberrant IL-6 elevations in AA patients may predict the likelihood of bacterial lung infection. The concurrent increase of IL-6 and IL-8, on the other hand, should signal bacterial and fungal infections in patients.These findings may help to suggest bacterial or fungal co-infection in patients with AA (Focus on VSAA and SAA).
Similar content being viewed by others
Introduction
“Aplastic” refers to the inability of bone marrow to form blood, the end-organ effect of diverse pathophysiologic mechanisms. Mostly, Aplastic anemia (AA) patients have pancytopenia, with decreased levels of platelets and white blood cells1,2. The immune function of AA patients is always low, which makes AA patients susceptible to infection, and infection is also a significant cause of death in AA patients3,4. Infection, especially early pulmonary infection, has always been a fundamental problem for hematologists, mainly because pulmonary infection, especially early infection, is not easy to diagnose. Body temperature, PCT, and CRP were less-sensitive in multiple infection studies on hematological malignancies5,6. The main problem with commonly used sputum culture is the low positive rate 7.Computerized tomography (CT) is still the most important tool for lung infection clinical diagnosis. However, it may not well diagnose the disease at the early stages, and may be restricted by the health center.
None of the laboratory tests for lung infections are perfect. In addition to the above examinations, lung lavage fluid culture and lung lavage fluid microbial next-generation sequencing(mNGS) have a wider and more accurate identification of pathogenic microorganisms in basic research and clinical experiments, and are also subject to many restrictions in clinical use (such as price, long sampling process and time to obtain results)8, especially for some blood system diseases. Like most developing countries or economically backward regions, the biggest problem we have is that because of economic reasons, the lung lavage fluid culture and lung lavage fluid micro next generation sequencing (mNGS), especially mNGS, can only be developed among a small number of people. Therefore, more auxiliary examinations are needed to strengthen the identification of pulmonary infection.
The limitations of these common examination items can easily lead to early lung infection not being detected in time, which further develops into serious infection, affecting the treatment of patients, and even affecting the survival of patients. In other words, the initial pulmonary infection is often found and treated after the infection is aggravated. Therefore, it is important to provide evidence of infection, which may help to accelerate the diagnosis of pulmonary infection.Cytokines are now gaining more attention, and previous studies have found cytokines to be more sensitive to lung infection than PCT, CRP, and body temperature in the absence of other diseases9,10. Furthermore, cytokines have the advantages of requiring less quantity of samples for detection, eliminating the need to consider secondary pollution, and allowing for faster flow cytometry detection. Although cytokines have been shown to have a strong relationship with inflammation, they have also been used to predict inflammation.
Our team has also been working on studying cytokines indicating pulmonary infection in patients with hematological diseases. Previous research results suggest that cytokines predicting pulmonary infection also apply to hematological tumors. But we also found differences in cytokine expression between different types of blood diseases5,11: considering the difference in disease, especially the difference between non-tumor disease and tumor disease. The primary focus of the current study is to investigate AA patients(Focus on VSAA and SAA) and explore the expression pattern of cytokines in the lungs of AA patients with bacterial infection and find the best potential biomarkers.
Material and methods
Declaration
All methods were performed in accordance with the relevant guidelines and regulations.The “Declaration of Helsinki” and the “International Code of Ethics for Human Biomedical Research” jointly formulated by the International Council of Medical Science Organizations and the World Health Organization were strictly followed throughout this study. At the same time, the project was approved by the Ethics Committee of the First People's Hospital of Yunnan Province, number: KHLL2022-KY075. All study participants provided informed consent simultaneously.
Study participants and data collection
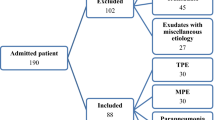
A total of 287 non-children AA medical records from patients who visited Yunnan First People’s Hospital’s Department of Hematology from June 2017 to May 2022 were evaluated for this study. It consists of 70 VSAA cases and 217 SAA cases. At the initial examination of the visit factor, we recorded the body temperature, Th1/Th2/Th17, CRP, and PCT cytokines. Refer to the following diagnostic criteria for bacterial lung infection12,13,14; bacterial lung infection, suspected or confirmed, irrespective of the presence or absence of bacteria in respiratory specimens, and is classified based on clinical symptoms and diagnostic standards. Cough that is becoming worse or new, shortness of breath, hypoxia, or dyspnea are all clinical criteria. Chest X-rays to detect inflammatory changes and sputum culture to detect bacterial infection or other auxiliary tests. In certain patients with suspected pulmonary disease, chest CT scans were used to confirm bacterial infection. For patients with bacterial and fungal infection, we only included patients with positive bronchoalveolar lavage fluid (BAL)1,3- β- D glucan test (G test) or/and galactomannan test (GM test) culture.
Statistical analysis
The data were initially examined for normality to see whether they have a normal distribution. Then, a t test or Mann–Whitney U test was conducted on independent samples. Continuous data are represented by the median and interquartile range (IQR), whereas percentages and frequencies describe categorical variables. We calculated the area under the curve (AUC) using the ROC curve to evaluate the diagnostic value of each parameter. Using the highest Youden index, the optimal Cut-off was determined. The GraphPad Prism 7 software was utilized to generate distribution plots, and the IBM SPSS 21.0 (Armonk, NY, USA) was used for statistical analysis. P-values of < 0.05 on (two-sided) were considered statistically significant.
Determination of cytokines
The kit used to detect serum cytokines is Aimplex Cytokine (QuantoBio, Tianjin, China) with a detection range of 1–2500 pg/ml, and the detected cytokines include Th (1/2/17), namely: interferon (IFN) -γ, IL -(1β, 2, 4, 5, 6, 8, 10, 12p70, 17A, 17F, 22) and tumor necrosis factor (TNF-α and β) using flow cytometry (NovoCyte D3000).
Results
Patients characteristics
1.4/5Among the 287 medical records, there were 125 females and 162 males, the median age was 29 (22–49) years, 151 patients without any infection, the median body temperature was 36.8 (36.6–37.0) °C (the mean value is 36.86 ± 0.46 °C), and the median PCT was 0.06 (0.04–0.2) ng/ml, median CRP 3.00 (1.55–8.78). Eighty-seven patients had a simple pulmonary bacterial infection, the median body temperature was 36.90 (36.70–37.60) °C(the mean value is 37.07 ± 0.60 °C), the median PCT was 0.12 (0.04–0.32) ng/ml, and the median CRP was 5.00 (2.48–29.00). Forty-nine patients had bacterial and fungal infections, with a median body temperature of 36.8 (36.7–37.6) °C (the mean value is 37.08 ± 0.63 °C), median PCT of 0.26 (0.08–1.10) ng/ml, and median CRP of 19.10 (10.04–39.15) (Table 1 and Fig. 1A–C).
The Mann–Whitney U test was used to test the differences among patients with respiratory tract bacterial infection, patients with respiratory tract bacterial complicated with fungal infection and patients with no bacterial infection. (A)Body temperature, (B)PCT, (C)CRP, (D) IL-6, (E) IL-8, (F) IL-10. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, ns no statistical difference.
Cytokine expression
In our study cohort IL-4, IL-5, IL-6, IL-8, IL-10, IL-12p70, IL-1β, IL-2, IFN-γ, TNF-α, TNF-β, IL-17A, IL-17F and IL-22 The median expression levels in non-infected patients were 2.72 (1.82–3.95) pg/ml, 2.15 (1.59–2.98) pg/ml, 8.19 (6.10–10.41) pg/ml, 14.13 (6.75–35.46) pg/ml, 4.38 (3.3–6.44) pg/ml, 3.94 (3.27–5.71) pg/ml, 1.64 (1.16–2.16) pg/ml, 3.08 (2.23–4.80) pg/ml, 2.87 (1.89–3.82) pg/ml, 2.84 (2.02–3.93) pg/ml, 3.16 (2.57–3.90) pg/ml, 2.29 (1.37–3.33) pg/ml, 3.36 (2.47–4.67) pg/ml, 1.01 (0.58–1.80) pg/ml (Table 2).
The median expression levels in patients with pulmonary bacterial infections were 2.84 (1.80–5.00) pg/ml, 2.29 (1.71–3.64) pg/ml, 26.43 (17.40–37.48) pg/ml, 26.62 (14.19–60.58) pg/ml, 4.96 (3.95–8.20) pg/ml, 4.29 (3.44–6.52) pg/ml, 1.75 (1.41–2.32) pg/ml, 3.49 (2.60–5.28) pg/ml, 3.07 (2.43–4.10) pg/ml, 3.07 (2.05–4.42) pg/ml, 3.20 (2.66–3.75) pg/ml, 2.69 (1.59–4.23) pg/ml, 4.23 (13.06–6.50) pg/ml, 1.24 (0.62–2.59) pg/ml. The median expression levels in patients with pulmonary comorbid bacteria and fungi were 3.55 (2.08–6.67) pg/ml, 2.19 (1.45–3.71) pg/ml, 104.73 (75.80–260.86) pg/ml, 103.15 (46.06–209.31) pg/ml, 7.08 (4.18–15.61) pg/ml, 5.80 (4.32–6.83) pg/ml, 1.93 (1.38–2.45) pg/ml, 4.44 (3.04–6.03) pg/ml, 3.56 (2.10–4.56) pg/ml, 3.17 (2.42–4.36) pg/ml, 3.73 (2.99–4.09) pg/ml, 3.40 (1.93–4.69) pg/ml, 3.99 (2.48–5.69) pg/ml, 1.39 (0.92–1.98) pg/ml (Table 2). Among the three groups of patients, there were significant differences in IL-6, IL-8, IL-10, IFN-γ, IL-17A, and IL-17F between the non-infection group and the pulmonary bacterial infection group (P < 0.05) as given in Table 2, Fig. 1D–F, and Supplementary Fig. 1D–F.
There were significant differences in IL-4, IL-6, IL-8, IL-10, IL-12p70, IL-1β, IL-2, TNF-β, IL-17, and IL-22 between the uninfected group and the pulmonary bacterial and fungal infection group (P < 0.05) (Table 2, Fig. 1D–F, Supplementary Fig. 1A–E,G,H). There were significant differences in IL-6, IL-8, IL-10, IL-12p70, and TNF-β between the pulmonary bacterial infection group and the pulmonary bacterial, fungal infection group (P < 0.05) (Table 2, Fig. 1D–F, Supplementary Fig. 1A,G).
Determination of specific indicators
Cut-off values were calculated from ROC curves, retaining potentially valuable cytokines, and compared with body temperature, PCT, and CRP. Our results show that IL-6 (AUC = 0.98, Cut-off = 14.28 pg/ml, P = 0.0000) has better sensitivity than other cytokines, body temperature AUC = 0.61, P = 0.0050), PCT (AUC = 0.57, P = 0.0592) and CRP (AUC = 0.60, P = 0.0147) for predicting bacterial infection of the lung (Table 3 and Fig. 2A). Further, the AUC and Cut-off values between no infection and bacterial co-infection with fungal infection were further calculated. Surprisingly, IL-6 (AUC = 1.00, Cut-off = 51.50 pg/ml, P = 0.0000) also had a strong advantage, and IL-8 (AUC = 0.87, Cut-off = 60.53 pg/ml, P = 0.0000) also showed stronger than other cytokines, body temperature (AUC = 0.60, P = 0.0324, PCT (AUC = 0.72, Cut-off = 0.63 ng/ml, P = 0.0000) and CRP (AUC = 0.79, Cut-off = 5.79 mg/l, P = 0.0000) (Table 3 and Fig. 2B).
(A) the ROC curves of cytokine, PCT, Body temperature and CRP in patients with respiratory tract bacterial infection were compared with those without respiratory tract infection. (B) the ROC curves of cytokine, PCT, Body temperature and CRP in patients with respiratory tract bacterial and fungal infection compared with those without infection.
The common clinical types of AA are severe aplastic anemia and severe aplastic anemia. In our study cohort, there are 70 VSAA patients and 217 SAA patients. By comparison, we found that only IL-8 had statistical difference in patients without infection (P = 0.0004), only IL-6 had statistical difference in patients with pulmonary bacterial infection (P = 0.0120), and IL-12P70 (P = 0.0046) and INF- γ (P = 0.0279) in patients with pulmonary bacterial and fungal infection had statistical difference (Supplementary Table 1). Furthermore, we calculated the AUC of cytokines in SAA and VSAA respectively by ROC curve. Our results show that the infection sensitive indicators in SAA and VSAA are similar, and such results are consistent with those in AA (VSAA + SAA) (Supplementary Table 2, Supplementary Fig. 2A–D and Table 3). This may indicate that both IL-6 and IL-8 can indicate the lung bacterial infection of VSAA/SAA and the co infection of lung bacteria and fungi.
Discussion
Infection is prevalent in AA with bone marrow failure because immunosuppression is prevalent during the treatment of hematological diseases Among them, SAA and VSAA are the most frequently infected in clinic15,16. Due to the lack of corresponding clinical symptoms of early bacterial infection in the lungs, it is easy to be ignored or misdiagnosed at the time of diagnosis. This often leads to aggravation or even death of patients, so it is necessary to increase auxiliary diagnosis indicators. In this study, the abnormal expression of IL-6 was found suitable for the pulmonary bacterial infection of AA. When the expression level is high, it may indicate a pulmonary bacterial and fungal infection in AA patients. In addition, the abnormal expression of IL-8 may help distinguish whether AA patients develop a bacterial co-fungal infection, which is different from the previous report that the increase of cytokines contributes to the bacterial lung infection in hematological cancers. Simultaneous expression of IL-10, IL-8, and IL-6 is a decisive advantage in newly diagnosed hematological malignancies and pediatric patients5,6,17.
Consistent with lymphoma and other reports, IL-6 was the most dominant of all markers11,18,19; our findings provide insight into the availability of IL-6 in AA for predicting bacterial lung infection. Our results revealed that IL-10, IL-8, IL-6, IFN-, IL-17A, and IL-17F levels in patients with bacterial lung illness were abnormally higher than those without infection (Table 2). This is different from our study on non-Hodgkin's lymphoma, where only IL-6, IL-8, IL-10, and IFN-γ have intersection indicators, while non-Hodgkin’s lymphoma Abnormal TNF-β is also present11. These differences may be primarily due to the disease itself, e.g., TNF-β can be related to tumor burden20. In addition, TNF-β is also a non-Hodgkin’s poor prognostic factor21. Therefore, it is also necessary to study the cytokines of each blood system disease separately, which is beneficial to increase the reliability of the application of cytokines in different conditions.
Previous research has found that invasive pulmonary aspergillosis causes an increase in Th1/17 cytokines in individuals with hematological diseases and that IL-6 and IL-8 are strongly related to invasive pulmonary aspergillosis22,23. Aspergillus fumigatus protease induces IL-6 and IL-8 release from A549 lung epithelial cells and primary epithelial cells24.In addition, fungal spores also promote the secretion of IL-8 from A549 lung epithelial cells25,26.IL-6 expression plays a crucial role in protective immunity in mice infected with Aspergillus27. Further, the Aspergillus flavus can also induce the expression of IL-828.
Furthermore, abnormally high expression of IL-6 and IL-8 may indicate poor mortality in individuals with hematological malignancies who have pneumococcal infection29. These data suggest that IL-6 and IL-8 are overexpressed in fungal-infected patients, and numerous hematological malignancies have been documented. Our results complement that IL-6 and IL-8 are similarly aberrant in AA expression in hematological non-neoplastic diseases and may have a role in discriminating bacterial or co-fungal infections because only IL-6 had a solid sensitivity to bacterial lung infection in our results. But after bacterial infection with a fungus, the IL-6 has a strong sensitivity. IL-8 also possesses better sensitivity than PCT, CRP, and body temperature. (Table 3, Fig. 2A,B). However, we analyzed SAA and VSAA separately, and the results were repeated, which may indicate that this model is applicable to both SAA and VSAA(Supplementary Table 2, Supplementary Fig. 2A–D and Table 3).
Our study is a retrospective study, and the biggest regret is that the number of patients with accurate identification of pathogenic bacteria is small and we did not include it in the analysis. For lung infection, the technology widely used to identify pathogenic bacteria in most countries and regions is still sputum culture, but the positive rate of sputum culture is only about 30%7,30. The more popular studies are obtaining samples directly from affected lesions by bronchoalveolar lavage (BAL) or by safe brushing with bronchoscopy followed by culture or mNGS, which is an invasive procedure31. For the invasive examination of bone marrow failure disease AA in the state of immune deficiency, especially VSAA. People often worry about increasing microbial infection and causing trauma. Also similar to sputum cultures, routine phenotypic diagnosis of BAL sample for pathogenic microorganism detection (including G test and GM test) usually takes 48 to 72 h, and the sensitivity range is only 30% to 60%, leading to undesirable delays in pathogen detection and initiation of antimicrobial therapy, potentially leading patients to miss optimal timing of therapy32,33,34. Especially when invasive aspergillosis occurs, the mortality may exceed 50%35.
The detection of microorganisms by mNGS through BAL is the most accurate and extensive at present, and it is also a detection project carried out in developed areas and comprehensive hospitals with perfect conditions. In addition to concerns about the sampling risk and the long testing process (About 2–3 weeks), the core challenge of mNGS is to distinguish pathogens from colonized microorganisms, which also complicates the interpretation of results and is not conducive to clinician interpretation36,37. In contrast to mGNS, although the presence of non-specific cytokines, the colonized flora generally does not lead to cytokine disturbance38,39,40. In addition, the inspection fee is relatively high, which must be considered in developing countries and regions. But it must be acknowledged that mNGS can identify microorganisms more widely and more accurately, which has high value for both clinical testing and scientific research.
our research is carried out in developing countries, which may also reflect the common problems in most economically backward regions. Although IL-6 is generally elevated in infected patients, we still hope to elucidate the expression of cytokines in pulmonary bacterial or/and fungal infections of AA patients, which may provide clues for clinical practice in AA.The most urgent task for us as clinicians is to detect the presence of infection as early as possible and to intervene as soon as possible, especially in bone marrow failure diseases such as AA. All in all, we still hope to find more sensitivity indicators from more commonly used clinical tests to help clinicians detect pulmonary bacterial or co-fungal infection in patients as early as possible.
Conclusion
Cytokines are abnormally expressed in AA in pulmonary infection, and the abnormal increase in IL-6 suggests that the patient has a pulmonary bacterial infection. However, when IL-8 and IL-6 are elevated simultaneously, it may indicate that the patient has a bacterial and fungal infection. These findings may help suggest bacterial or concurrent bacterial and fungal infections in the lungs of SAA and VSAA patients.
Data availability
All data generated or analysed during this study are included in this published article [and its supplementary information files].
References
Young, N. S. Aplastic anemia. N. Engl. J. Med. 379(17), 1643–1656 (2018).
DeZern, A. E. & Churpek, J. E. Approach to the diagnosis of aplastic anemia. Blood Adv. 5(12), 2660–2671 (2021).
Valdez, J. M., Scheinberg, P., Young, N. S. & Walsh, T. J. Infections in patients with aplastic anemia. Semin. Hematol. 46(3), 269–276 (2009).
Atmar, K. et al. Functional and immune modulatory characteristics of bone marrow mesenchymal stromal cells in patients with aplastic anemia: A systematic review. Front. Immunol 13, 859668 (2022).
Li, Z. et al. Cytokine expression of lung bacterial infection in newly diagnosed adult hematological malignancies. Front. Immunol. 12, 748585 (2021).
Xia, T., Xu, X., Zhao, N., Luo, Z. & Tang, Y. Comparison of the diagnostic power of cytokine patterns and procalcitonin for predicting infection among paediatric haematology/oncology patients. Clin. Microbiol. Infect. 22(12), 996–1001 (2016).
Mandell, L. A. et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin. Infect. Dis. 44(Suppl 2), S27-72 (2007).
Diao, Z., Han, D., Zhang, R. & Li, J. Metagenomics next-generation sequencing tests take the stage in the diagnosis of lower respiratory tract infections. J. Adv. Res. 38, 201–212 (2022).
Nascimento-Carvalho, E. C. et al. Evolution of cytokines/chemokines in cases with community-acquired pneumonia and distinct etiologies. Pediatr. Pulmonol. 55(1), 169–176 (2020).
Branchett, W. J. & Lloyd, C. M. Regulatory cytokine function in the respiratory tract. Mucosal. Immunol. 12(3), 589–600 (2019).
Zhang, L. et al. Increased cytokine levels assist in the diagnosis of respiratory bacterial infections or concurrent bacteremia in patients with non-hodgkin’s lymphoma. Front. Cell. Infect. Microbiol. 12, 860526 (2022).
Lim, W. S. et al. BTS guidelines for the management of community acquired pneumonia in adults: Update 2009. Thorax 64(Suppl 3), 1–55 (2009).
Metlay, J. P. et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official clinical practice guideline of the American thoracic society and infectious diseases society of America. Am. J. Respir. Crit. Care Med. 200(7), e45–e67 (2019).
Donnelly, J. P. et al. Revision and update of the consensus definitions of invasive fungal disease from the european organization for research and treatment of cancer and the mycoses study group education and research consortium. Clin. Infect. Dis. 71(6), 1367–1376 (2020).
Peffault de Latour, R. et al. Burden of illness among patients with severe aplastic anemia who have had insufficient response to immunosuppressive therapy: A multicenter retrospective chart review study. Ann. Hematol. 99(4), 743–752 (2020).
Scheich, S. et al. Bloodstream infections with gram-negative organisms and the impact of multidrug resistance in patients with hematological malignancies. Ann. Hematol. 97(11), 2225–2234 (2018).
Tang, Y. et al. Evaluation of Th1/Th2 cytokines as a rapid diagnostic tool for severe infection in paediatric haematology/oncology patients by the use of cytometric bead array technology. Clin. Microbiol. Infect. 17(11), 1666–1673 (2011).
Iwase, S. et al. Interleukin-6 as a diagnostic marker for infection in critically ill patients: A systematic review and meta-analysis. Am. J. Emerg. Med. 37(2), 260–265 (2019).
Thompson, D. K., Huffman, K. M., Kraus, W. E. & Kraus, V. B. Critical appraisal of four IL-6 immunoassays. PLoS ONE 7(2), e30659 (2012).
Aggarwal, B. B. Signalling pathways of the TNF superfamily: A double-edged sword. Nat. Rev. Immunol. 3(9), 745–756 (2003).
Edlefsen, K. L. et al. Cytokines in serum in relation to future non-Hodgkin lymphoma risk: Evidence for associations by histologic subtype. Int. J. Cancer 135(4), 913–922 (2014).
Heldt, S. et al. Levels of interleukin (IL)-6 and IL-8 are elevated in serum and bronchoalveolar lavage fluid of haematological patients with invasive pulmonary aspergillosis. Mycoses 60(12), 818–825 (2017).
Heldt, S. et al. Diagnosis of invasive aspergillosis in hematological malignancy patients: Performance of cytokines, Asp LFD, and Aspergillus PCR in same day blood and bronchoalveolar lavage samples. J. Infect. 77(3), 235–241 (2018).
Borger, P. et al. Proteases from Aspergillus fumigatus induce interleukin (IL)-6 and IL-8 production in airway epithelial cell lines by transcriptional mechanisms. J. Infect. Dis. 180(4), 1267–1274 (1999).
Houser, J. et al. A soluble fucose-specific lectin from Aspergillus fumigatus conidia–structure, specificity and possible role in fungal pathogenicity. PLoS ONE 8(12), e83077 (2013).
Almeida, B. R., Barros, B., Araujo, A. C. L., Alcantara, C. & Suzuki, E. Paracoccidioides species present distinct fungal adherence to epithelial lung cells and promote different IL-8 secretion levels. Med. Microbiol. Immunol. 209(1), 59–67 (2020).
Cenci, E. et al. Impaired antifungal effector activity but not inflammatory cell recruitment in interleukin-6-deficient mice with invasive pulmonary aspergillosis. J. Infect. Dis. 184(5), 610–617 (2001).
Ghufran, M. S., Ghosh, K. & Kanade, S. R. A fucose specific lectin from Aspergillus flavus induced interleukin-8 expression is mediated by mitogen activated protein kinase p38. Med. Mycol. 55(3), 323–333 (2017).
Rawlings, S. A. et al. Using interleukin 6 and 8 in blood and bronchoalveolar lavage fluid to predict survival in hematological malignancy patients with suspected pulmonary mold infection. Front. Immunol. 10, 1798 (2019).
Chalmers, J. D. et al. Epidemiology, antibiotic therapy and clinical outcomes in health care-associated pneumonia: A UK cohort study. Clin. Infect. Dis. 53(2), 107–113 (2011).
Chen, K. et al. Accuracy of molecular amplification assays for diagnosis of staphylococcal pneumonia: A systematic review and metaanalysis. J. Clin. Microbiol. 59(8), e0300320 (2021).
Souweine, B. et al. Diagnostic accuracy of protected specimen brush and bronchoalveolar lavage in nosocomial pneumonia: Impact of previous antimicrobial treatments. Crit. Care Med. 26(2), 236–244 (1998).
Craven, D. E. & Hjalmarson, K. I. Ventilator-associated tracheobronchitis and pneumonia: Thinking outside the box. Clin. Infect. Dis. 51(Suppl 1), S59-66 (2010).
Suaya, J. A. et al. Identification of Streptococcus pneumoniae in hospital-acquired pneumonia in adults. J. Hosp. Infect. 108, 146–157 (2021).
Nivoix, Y. et al. Factors associated with overall and attributable mortality in invasive aspergillosis. Clin. Infect. Dis. 47(9), 1176–1184 (2008).
Langelier, C. et al. Integrating host response and unbiased microbe detection for lower respiratory tract infection diagnosis in critically ill adults. Proc. Natl. Acad. Sci. U. S. A. 115(52), E12353–E12362 (2018).
Simner, P., Miller, S. & Carroll, K. Understanding the promises and hurdles of metagenomic next-generation sequencing as a diagnostic tool for infectious diseases. Clin. Infect. Dis. 66(5), 778–788 (2018).
Megha, K. B., Joseph, X., Akhil, V. & Mohanan, P. V. Cascade of immune mechanism and consequences of inflammatory disorders. Phytomedicine 91, 153712 (2021).
Razzuoli, E. et al. Yersinia enterocolitica-specific modulation of innate immune responses in jejunal epithelial cells. Vet. Microbiol. 242, 108596 (2020).
Gonzalez, J. F., Hahn, M. M. & Gunn, J. S. Chronic biofilm-based infections: Skewing of the immune response. Pathog. Dis. https://doi.org/10.1093/femspd/fty023 (2018).
Acknowledgements
The author would like to thank the patients who participated in the treatment at the First People’s Hospital and Kunming Junmai Technology Co. Ltd. for providing reagents and consumables.
Funding
This work was supported by the National Natural Science Foundation of China (82060810), Yunnan Provincial Department of Science and Technology- Kunming Medical University Joint Special Project on Applied Basic Research (202101AY070001-27) and Open Project of Yunnan Blood Clinical Medical Center (2021LCZXXF-XY08, 2020LCZXKF-XY08, 2021LCZXXF-XY05, 2020LCZXKF-XY07, 2021LCZXXF-XY09 and 2022LCZXKF-XY01).
Author information
Authors and Affiliations
Contributions
J.Z. wrote the manuscript and graphs, Z.Y. analyzed the data and prepared the tables. P.H., X.G., C.Z. and Y.Z. jointly counted the data. H.L. and T.Y. corrected the language and logic of the manuscript. Y.C. helped to check the data. R.Z. designed the research protocol and corrected the methodology. Z.L. participated in the design of the protocol, supporting the progress of the study and continuing the related research on cytokines in hematological diseases.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, J., Yang, Z., Hu, P. et al. Cytokines help suggest aplastic anemia with pulmonary bacterial or co-fungal infection. Sci Rep 12, 18373 (2022). https://doi.org/10.1038/s41598-022-22503-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-22503-7
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.