Abstract
To flexibly regulate their behavior, children’s ability to inhibit prepotent responses arises from cognitive and motor mechanisms that have an intertwined developmental trajectory. Subtle differences in planning and control can contribute to impulsive behaviors, which are common in Attention Deficit and Hyperactivity Disorder (ADHD) and difficult to be assessed and trained. We adapted a Go/No-Go task and employed a portable, low-cost kinematic sensor to explore the different strategies used by children with ADHD or typical development to provide a prepotent response (dominant condition) or inhibit the prepotent and select an alternative one (non-dominant condition). Although no group difference emerged on accuracy levels, the kinematic analysis of correct responses revealed that, unlike neurotypical children, those with ADHD did not show increased motor planning in non-dominant compared to dominant trials. Future studies should investigate whether motor control could help children with ADHD compensate for planning difficulties. This strategy might make inhibition harder in naturalistic situations that involve complex actions. Combining cognitive and kinematic measures is a potential innovative method for assessment and intervention of subtle differences in executive processes such as inhibition, going deeper than is possible based on accuracy outcomes alone.
Similar content being viewed by others
Introduction
Performing cognitive operations and motor actions can be considered two faces of the same coin, as they vastly rely on shared mechanisms that allow us to produce appropriate responses with respect to goals and context1. All relevant processes specialize with age, with motor and cognitive development being closely connected and inter-related in a dynamic process of exploring and adjusting to the demands of the external physical and social environment2. Although cognitive and motor difficulties often co-occur in neurodevelopmental conditions and have been extensively studied as separate processes3,4, their common underlaying mechanisms are still to be furthered. We strongly believe that an integrated approach will provide a more complete understanding of the interplay between low-level sensorimotor processes and high-level executive functioning. Indeed, executive functions are those top-down processes (i.e., working memory, inhibition, shifting) that enable people to plan, monitor and control sensorimotor, socio-affective and cognitive processes, being fundamental to mental and physical wellbeing5. Among these functions, the ability to inhibit automatic and highly probable responses, and let less probable alternatives successfully compete for control of cognition and behaviors, ensures that we are flexible and open to learning from the surrounding environment6.
Different paradigms are commonly used to measure the inhibition of prepotent responses (e.g., Stroop, Stop-signal and Go/No-Go tasks), with diverse versions that rely on mainly cognitive processes or entail varying degrees of motor components, and activate both distinct and shared neural areas7,8. For instance, cognitive inhibition of prepotent responses is conceived as the ability to focus on the task and ignore irrelevant distractors, as in the case of reading the word “blue” written in red ink. The motor component comes into play when the response requires some sort of movement (from pressing a button to reaching a target), which sometimes has to be voluntarily stopped before or during its execution7. Usually, these motor executions are not main targets of study, as they are considered only a way to obtain from individuals a response that is believed to reflect certain cognitive mechanisms. However, the very planning of this motor response could reveal important information about the processes at play. Thus, a deeper understanding of motor responses in cognitive tasks needs an improved consideration, leading to a new perspective on the shared mechanisms that underpin adaptive behaviors.
Inhibition of prepotent responses is a well-studied process being affected by disorders such as Attention Deficit and Hyperactivity Disorder (ADHD)9, which is diagnosed based on inattentiveness, impulsiveness and hyperactivity symptoms10. At the cognitive level, it is established that people with ADHD, despite the wide variability that characterizes developmental trajectories, are overall impaired in executive functions11. ADHD can entail several aspects of cognitive and motor impulsivity, that consists of nonreflective stimulus-driven processes and manifests itself through inhibitory difficulties, distractibility, faster and less accurate responses to neuropsychological tests12. At the motor level, it is still debated whether motor signs of atypical development can be detected from infancy and interpreted as early risk factors for the following development of ADHD cognitive and behavioral symptoms13. Some co-occurrent difficulties in motor skills (e.g., fine motor precision, manual dexterity, bilateral coordination, balance, and postural control, running speed and agility, limb coordination, strength) can be found in about 50% of individuals with ADHD14. However, those are not a diagnostic criterion and there is no evidence so far supporting the link between motor impairments and ADHD-specific symptoms such as inhibitory deficiencies14. To shed light on this, an approach that studies these two aspects in an integrated manner could provide an innovative perspective on difficulties with inhibition and behavioral hyperactivity. Potential underlying mechanisms of inhibition difficulties relate to motor planning, which is responsible for selecting the action target and the timing of movements (e.g., reaction times, movement times, and acceleration/velocity parameters)15.
Adults with ADHD have been found to show atypical motor profiles, with longer reaction times to start moving after a “Go” cue and higher variability in the velocity shape along time, suggesting impaired motor planning capacities16. It is interesting to note that there is a kind of slowness in sensorimotor and cognitive processes that underlie behavioral manifestations of impulsivity, hyperactivity, and inattention. A developmental perspective is needed to understand how these atypicalities have emerged and are maintained from childhood to adulthood. This would help us design targeted and age-appropriate interventions to promote a change on the mechanisms underlying the cognitive and behavioral difficulties of ADHD. Notably, purely cognitive training specifically targeting executive functions such as working memory, attention, inhibition, and shifting rarely result in cognitive nor behavioral or academic improvements, with scarce effect on ADHD core symptoms17,18. It has been speculated that leveraging embodied cognition and cognitive-motor approaches could boost training efficacy19. This multidimensional perspective would eventually chart the way to define and test both motor and cognitive interventions to strengthen inhibition by passing through multidimensional doorways. Despite their presence and impact, motor difficulties of people with ADHD often end up being overlooked by research and clinical practice.
Previous studies mainly based on correlational analysis of motor skills and purely cognitive performance at inhibition tasks, and failed to find clear relationships14. On the other hand, investigating inhibition without dissociating motor and cognitive aspects that are deeply interrelated offers further insights on the underlying processes. The compelling possibility of integrating a kinematic measure to the traditional neuropsychological evaluation is strongly limited by the need of sophisticated motion capture systems. Those used for research purposes are often expensive and bulky, thus being hardly affordable for most clinical centers. In order to use low-cost portable solutions and boost the applicability of motion analysis, inertial sensors have been recently recommended for their good measurement reliability and validity20. Adopting this technology in clinical practice would allow for a more detailed analysis of the mechanisms underlying the child's performance on tests of interest. It could be used during assessment for setting specific intervention goals, for monitoring treatment effects, and as a treatment tool itself when used as biofeedback.
The present study aims at investigating children’s ability to inhibit prepotent motor responses, through an adapted version of the Go/No-Go paradigm. The task embedded a reaching movement and kinematic measures to surface motor planning characteristics of inhibition21. Children with ADHD or typical development were recruited. A commercially available, low-cost, easy to use, wearable accelerometer sensor was employed to capture movement features. Previous studies highlighted that portable inertial sensors, compared to gold standard motion capture systems, offer reliable measures on some kinematic indices20. Our previous study suggested that the apparatus we employ here is effective in measuring the velocity shape across movement time (“when”)21.
Distinct kinematic indices were considered to study the progress of motor planning and control in the various phases of action. Researchers often use Reaction Time (RT) to indicate the time from the appearance of the Go stimulus to the moment when the person gives the response (which corresponds to the end of the movement). This is the index of choice for studying variability in the inhibitory abilities of people with ADHD22. However, using RT as the total response time does not consider the two underlying processes separately: the preparatory activities that take place before the start of the movement, and the actual motor execution23. We hereby calculate RT as the time from the appearance of the Go stimulus to the beginning of the movement, so that it gives us a measure of pure motor planning. Higher need for motor planning is expected to result in higher RTs24. We then calculate movement execution time from when the response movement begins to when it ends (as measured by the Movement Duration—MD).
Across MD, motor planning gradually gives way to control and monitoring of the ongoing movement, which involve distinct processes15. Therefore, the percent Time to Peak Velocity (TPV) may represent a useful index to disentangle how much of the movement time is devoted to planning or control. Theoretical (e.g., in robotics) reaching trajectories starting and ending at full rest will show a bell-shaped velocity path, with the first half of MD spent accelerating and the second one decelerating, resulting in a 50% TPV25,26. The more cognitive load is required in a given task, the more human reaching movements have a greater need for motor planning, thus resulting in longer acceleration phase and TPV21. A higher percentage of time before the peak velocity (acceleration) inevitably means a lower percentage of time following the peak velocity (deceleration). Consequently, the TPV takes both aspects into account simultaneously, and the percentage of time after the peak velocity can be intuitively calculated as 100 (total)—TPV.
We have previously found that, to correctly inhibit a prepotent response and select the instructed alternative one, neurotypical adults show longer RT and MD, as well as increased TPV overall dedicating more resources to motor planning than monitoring and control of ongoing movements21. Assuming that this is the motor strategy that has been established as most effective in adults, a developmental perspective is needed to understand how it specializes during childhood and is potentially subject to deviation in cases of atypical development. We therefore expect age-related differences in the kinematic profile of motor planning and control necessary to inhibit prepotent responses. Moreover, we hypothesize that children with ADHD, compared to neurotypical controls, would show greater difficulties inhibiting the prepotent response, which the literature also refers to as motor impulsivity27. We expect children with ADHD to make more errors than controls in the non-dominant condition, and show an atypical motor profile, with reduced or less effective motor planning. As markers of motor impulsivity, we particularly expect reduced RT and TPV in the group of children with ADHD27.
Materials and methods
Participants
We recruited 17 children with ADHD (4 female children) from 6 to 15 years of age (M = 9.4, SD = 2.2), and 26 children with Typical Development (TD control group; 10 female children), from 6 to 13 years of age (M = 9.2, SD = 2.1). Three additional participants (2 in the ADHD and 1 in the TD group) were excluded due to technical issues that prevented them from completing at least 50% of the trials.
Data collection was planned to take place between December 2019 and April 2020, as part of a collaborative project with a clinical centre in northern Italy, which is specialised in ADHD diagnosis and intervention. Data collection was interrupted at the beginning of the Covid-19 pandemic and resumed when the centre was authorised to reopen to external operators (i.e., the investigators). Thus, a further phase of data collection was carried out between October and December 2021. The partner centre had an average intake of 60 children, and all were offered voluntary participation in the study. The final sample of children with ADHD was determined by the number of parents and children who joined and participated. Since ADHD is an inherently heterogeneous condition28,29, we have not established inclusion or exclusion criteria based on IQ, level of support needed, or possible presence of co-occurring medical or neuropsychological conditions. Thus, we aimed to include participants from the heterogeneous ADHD population. Psychologists confirmed children’s diagnosis and provided IQ assessments through the WISC-IV scale. Moreover, we collected parent-reported questionnaires on the child’s executive (Executive Functions Questionnaire-Q.FE30) and sensory profile (Short Sensory Profile-SSP31), as well as the presence and severity of restricted and repetitive behaviors (Repetitive Behavior Scale-Revised-RBS-R)32. A convenient control group of children with typical development in the same age range was tested at the University of Padova. According to parents’ reports, typically developing children had no medical nor neuropsychological conditions.
Characteristics of the ADHD group are provided in Table S3 of SI Appendix, which includes IQs, and scores from the parent reported assessment. Diagnostic subtypes and comorbidities are also described in SI Appendix. All children's parents signed a written consent form. All experimental methods received ethical approval from the Research Ethics Committee of the School of Psychology, University of Padova (protocol no. 3251). The experiment was carried out in accordance with the approved guidelines and regulations.
Procedure and task
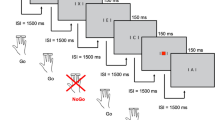
Children sat on a desk and wore an accelerometer sensor on their dominant wrist. They were instructed to place the dominant hand at a specific starting position, monitored by a presence sensor, and completely extend their arm to tap on the response touchscreen. A Go/No-Go paradigm was adapted to assess the inhibition of a prepotent response and tested with neurotypical adults in a previous work21. Upon comparison of a central stimulus (red/green, upwards/downwards arrow), participants were asked to select, reach, and press one of two response keys (either a red or green circle) placed one on the left and one on the right side of the central stimulus, following specific instructions. Before the start of the next trial, participants had to return their hand on the sensor. As soon as the hand was in place, the next trial started after a random delay (range = 0:2 s), which prevented children from anticipating the onset of the next trial. The set-up and procedure are illustrated in Fig. 1.
More in detail, participants were told to select the response key of the same color of the central stimulus when it was an upwards/downwards (counterbalanced between participants) arrow (dominant condition). On the other side, they were told to select the response key of the different color when the central stimulus was an averted (either upwards or downwards, counterbalanced between participants) arrow (non-dominant condition). We elicited a prepotent response for the same-color action (occurring the 75% of times), and an inhibitory response for the alternative different-color action (occurring the 25% of times). Two blocks were administered, with the red/green response keys being located once on the right and once on the left side of the touchscreen. To maintain participants’ engagement during the task, a short (30 s on average) video from well-known movies appeared every 40 trials. The task lasted about 15 min. Technical features of the apparatus (e.g., programming language, devices for conducting the experiment) are described in detail in our previous work21.
Participants were instructed to reply as quickly and accurately as possible. Failure to press any keys within 2,000 ms was marked as “omission”. Movements starting before the cue stimulus onset were tagged as “anticipation” (the program aborted the trial by showing no cue stimulus). Omissions and anticipations were considered invalid trials, therefore excluded from the analysis. The task ended upon completion of 160 valid trials (i.e., trial with correct/incorrect answer) or a maximum of 180 total trials (in case omissions and anticipations occurred). We then planned to exclude those responses whereby either RT or MD was less than 100 ms, being them ascribable to anticipation. We only included responses whereby the TPV was within the 5–95% range, thus considering extremes as due to extra-task movements.
Ethics declarations
All experimental methods received ethical approval from the Research Ethics Committee of the School of Psychology, University of Padova (protocol no. 3251).
Informed consent
All children's parents signed a written informed consent form.
Results
To analyze children’s performance, we considered 4 dependent variables. Accuracy indicates the percentage of correct responses out of the total number of valid responses (after discarding anticipations and omissions). RT measures the time from the appearance of the central stimulus to the onset of movement (the time when the hand is raised by the presence sensor). MD measures the duration of the movement (from when the sensor is released to when a response is given). We then computed the percent Time to Peak Velocity (TPV), which is the percentage of MD spent from movement onset to maximum peak velocity. Our previous work describes the rationale behind the selection of these kinematic variables and their reliability, the steps for data calibration and pre-processing of raw acceleration data, as well as the computation of TPV values21.
TD children provided 4,104 valid responses out of 4,526 total trials (91%). Children with ADHD provided 2472 valid responses out of 3,023 total trials (82%). This demonstrates both successful task competition (with our task being adequate for both groups), and low rate of discarded data. From valid trials performed by both groups, we excluded 217 out of 6,576 responses (3.3%), whereby either RT or MD was less than 100 ms, or the TPV was out of the 5–95% range. Excluded responses were not further analyzed. Final dataset comprehended 6,359 observations.
An exploratory approach was elected to test different potential hypotheses linking each dependent variable to the predictors of interest. Through separated sets of model comparisons, different research hypotheses were specified as statistical models, and their statistical evidence was evaluated using information criteria33. Generalized mixed-effects models were employed to account for the repeated measures design of the experiment (i.e., trials nested within participants, which has been included as a random effect in the analyses) and specify the distribution of each dependent variable. For each dependent variable, a set of models were compared through the Akaike weights (AICWt) (i.e., the probability of each model, given the data and the set of considered models)33, using the AICcmodavg33 R package. Then, likelihood ratio tests were used to compare the chosen models, and test the effects predicted by the best model.
As an index of goodness of prediction, conditional R2 (the ratio of variance explained by fixed and random effects over total variance) and marginal R2 (the ratio of variance explained by fixed effects over total variance) were calculated to quantify the variance explained by the whole model (including the contribution of individual variability) or the fixed effects only (excluding the contribution of individual variability)34. Higher percentages of explained variance indicates a stronger strength of association between the dependent variable and the predictors, with the selected model making better predictions. The analyses have been run with R, version 4.0.235.
We separately investigated whether each dependent variable (Accuracy, RT, MD, TPV) was influenced by the fixed effects of Condition (within-subjects, two levels categorical factor: dominant versus non-dominant), Group (between-subjects, two levels categorical factor: ADHD versus TD), and Age (continuous numeric variable). All models accounted for the random effect of participants (i.e., interpersonal variability). We considered the five models that follow.
-
m0 (null model) specified the hypothesis of no difference due to the independent variables and only accounted for individual variability
-
m1 specified the hypothesis of a Condition effect
-
m2 specified the hypothesis of additive Condition and Group effects
-
m3 specified the hypothesis of additive Condition, Group and Age effects
-
m4 specified the hypothesis of a two-way interaction effect between Condition and Group, with the additive Age effect.
Accuracy
After excluding omissions and anticipations, children with ADHD provided 2,234 correct and 137 incorrect (i.e., the wrong answer was provided) responses. TD children provided 3,777 correct and 211 incorrect responses (percentages of correct responses according to Group and Condition are reported in Table S1, SI Appendix). Model comparison was run with the glmmTMB36 R package. The binomial distribution was specified to account for the binary nature of the dependent variable (1 = correct; 0 = incorrect). According to AIC weights (AICWt_m0 < 0.01; AICWt_m1 = 0.39; AICWt_m2 = 0.14; AICWt_m3 = 0.15; AICWt_m4 = 0.14), the best model was m1 (39% probability of being the best model; χ2 = 369.3; P < 0.001), which revealed a significant effect of Condition (P < 0.001). As visualized in Fig. 2, accuracy was reduced in the non-dominant condition.
Conditional R2 (the ratio of variance explained by fixed and random effects over total variance) indicates that m1 explains 33% of variance, whereas marginal R2 (the ratio of variance explained by fixed effects over total variance) indicates that Condition explains 19% of variance. Therefore, 14% of variance was explained by individual variability (i.e., the random effect of participants).
Kinematics
We further explored kinematic features of correct responses to investigate whether, beyond accuracy, children with ADHD would show subtle motor atypicalities. Means and standard deviations of RT, MD, and TPV of correct responses in each condition and group are reported in Table S2, SI Appendix. In SI Appendix, we have also conducted a visual inspection of the velocity shape and trend across movement time, describing group and individual differences. At the group level, children with ADHD show a flatter velocity profile over the time course of the movement, with a less evident peak velocity at the beginning of the movement.
RT
Model comparison was run with the glmer function of lme437 R package. The gamma distribution was specified to account for the positively skewed nature of the dependent variable. According to AIC weights (AICWt_m0 < 0.001; AICWt_m1 < 0.01; AICWt_m2 < 0.01; AICWt_m3 = 0.19; AICWt_m4 = 0.80), the best model is m4 (80% probability of being the best model; χ2 = 4.9; P = 0.03), which reveals a significant interaction between Condition and Group (P = 0.03), and a significant effect of Age (P < 0.001). As visualized in Fig. 3, TD children showed increased RT in the non-dominant compared to the dominant condition, thus devoting more time to motor planning when the response required inhibition. This pattern was not present in children with ADHD, who did not differentiate RT depending on Condition. Moreover, there is a negative association between RT and Age, with RT decreasing at older ages, regardless of group.
Conditional R2 (the ratio of variance explained by fixed and random effects over total variance) indicates that m4 explains 37% of variance, whereas marginal R2 (the ratio of variance explained by fixed effects over total variance) indicates that Condition*Group and Age explain 28% of variance. Therefore, 9% of variance is explained by individual variability (i.e., the random effect of participants).
MD
Model comparison was run with the glmer function of lme437 R package. The gamma distribution was specified to account for the positively skewed nature of the dependent variable. According to AIC weights (AICWt_m0 < 0.001; AICWt_m1 = 0.29; AICWt_m2 = 0.41; AICWt_m3 = 0.22; AICWt_m4 = 0.08), the best model is m2 (41% probability of being the best model; χ2 = 2.7; P = 0.1), which reveals a significant effect of Condition (P < 0.001), and a non-significant effect of Group (P = 0.09). As visualized in Fig. 4, MD increased in the non-dominant condition compared to the dominant condition.
Conditional R2 (the ratio of variance explained by fixed and random effects over total variance) indicates that m4 explains 38% of variance, whereas marginal R2 (the ratio of variance explained by fixed effects over total variance) indicates that Condition and Group explain 20% of variance. Therefore, 18% of variance is explained by individual variability (i.e., the random effect of participants).
TPV
Model comparison was run with the glmmTMB36 R package. The beta distribution was specified to account for the nature of the dependent variable (continuous proportions on the interval 0:1). According to AIC weights (AICWt_m0 < 0.01; AICWt_m1 = 0.08; AICWt_m2 = 0.06; AICWt_m3 = 0.04; AICWt_m4 = 0.83), the best model is m4 (83% probability of being the best model; χ2 = 8.3; P = 0.004), which reveals a significant interaction between Condition and Group (P = 0.004), and a non-significant effect of Age (P = 0.3). As visualized in Fig. 5, TD children showed increased TPV in the non-dominant compared to the dominant condition, thus devoting more time to motor planning when the response required inhibition. This pattern was not present in children with ADHD, who did not differentiate TPV depending on Condition. At both the group and individual level, further graphical inspection of velocity shape across time is described in SI Appendix.
Conditional R2 (the ratio of variance explained by fixed and random effects over total variance) indicates that m4 explains 71% of variance, whereas marginal R2 (the ratio of variance explained by fixed effects over total variance) indicates that Condition*Group and Age explain 9% of variance. Therefore, 62% of variance is explained by individual variability (i.e., the random effect of participants).
Discussion
The present study explored the mechanisms underlying the inhibition of a prepotent motor response, which is frequently reported to be affected in children with ADHD. The performance of the ADHD and TD groups at our motor adaptation of the Go/No-Go task, showed both similarities and differences.
Accuracy
Both ADHD and TD children made more errors in the non-dominant compared to the dominant condition. This indicates that the task was effective in inducing a prepotent response in the dominant condition, which was the more frequent one, and facilitated by the requirement to match the Go stimulus and the response option by color. Children with ADHD and typical development were equally accurate in selecting the correct response, so that no group difference was found on accuracy levels. This unexpected result could be due to the ease of the task, which required a rather simple motor response, as also evidenced by the high percentages of correct responses. In tasks with greater time pressure or greater complexity of the motor action required to answer, we could expect more marked differences between the two groups. Although the task was based on the central properties of Go/No-Go (i.e., more frequent administration of the dominant condition), some differences may have made our task easier than traditional ones at the level of inhibition of prepotent responses. In particular, responding by reaching rather than quickly pressing a button may have allowed participants more time to process the cue, recall the instructions, and redirect their response during movement. On the one hand, this may explain the high accuracy, and at the same time allowed us to study not only reaction time (movement pre-planning), but also what happens during movement (motor planning gradually gives way to control of the ongoing movement).
Motor planning
The main findings of this study revealed that, beyond accuracy, the ADHD group showed different motor patterns that possibly indicate reduced motor planning compared to the TD group. In the non-dominant condition compared to the dominant condition, TD children spent more time planning the movement, which resulted in longer Reaction Time (RT) and greater percent Time to Peak Velocity (TPV). Indeed, higher pre-planning (i.e., longer RT) and initial acceleration phase (i.e., greater time to peak velocity—TPV) is an efficient strategy of the motor system, that reduces the time and resources needed for online movement correction38. On the other hand, children with ADHD did not modulate RT and TPV according to condition, not dedicating more time to motor planning when needed to inhibit the prepotent response. This subtle lack of flexibility in adjusting the motor and cognitive strategies to the task demands can be interpreted as a marker of motor and cognitive impulsivity. Our findings are in line with previous literature showing that atypical activation of premotor systems may contribute to impaired response inhibition in children with ADHD39. There is an interesting debate in the literature on the link between motor preparation and spatial attention40,41, which could be further explored to understand the link between cognition and movement in ADHD.
Motor control
Across both groups, children showed increased Movement Duration (MD) in the non-dominant versus dominant condition. This indicates that inhibitory processes take place during movement execution. Throughout movement, motor planning gradually gives way to control and monitoring of the ongoing movement15. To better disentangle how much of the movement time is devoted to planning or control, we employed the percent Time to Peak Velocity (TPV) as a relative asymmetry index. Theoretical reaching trajectories starting and ending at full rest have a bell-shaped velocity path, with the first half of MD spent accelerating and the second one decelerating, resulting in a 50% TPV25,26. In actual reaching movements, distinct characteristics of the target differently affect the movement acceleration-deceleration symmetry. Whereas physical precision of the movement (e.g., to grasp small objects) requires more control and longer deceleration42, cognitive load affects the early stages of movements, thus requiring more planning43.
The smaller TPV captured across conditions in the ADHD compared to the TD group, indicates higher portion of movement being dedicated to the deceleration phase, that usually stands for motor control15,42. Increased movement variability in children with ADHD44 has often been interpreted as an indication of poor motor control, when instead it could be a compensatory strategy that, given a reduced planning, requires more online adjustments during movement execution. To better understand 'how' children with ADHD regulate movement in its final phase, future studies would benefit from the use of additional kinematic indices that capture online motor correction more precisely (e.g., number of direction changes and acceleration/deceleration units).
Although kinematic indices are widely used as a mirror of underlying cognitive mechanisms, it is even more informative to combine them with the study of neural correlates45. Previous evidence suggests that increased activation of prefrontal areas can help children with ADHD compensate for atypical activation of premotor areas in Go/No-Go tasks39. It is unclear whether this can be attributed to planning or control mechanisms. The study of EEG components and the timing of neural activities that precede and take place during responses to cognitive tasks can be coupled with kinematic indices to shed light on planning-control dynamics46,47. To the best of our knowledge, little is still known about the specifics of such mechanisms in ADHD.
Children with ADHD might employ compensatory strategies for planning difficulties, which may be sufficient to achieve good accuracy in very simple tasks as the one employed in our work. Indeed, they chose between two alternatives that differed only in one motor (i.e., the movement direction: reaching the key to the right or to the left of the central stimulus) and cognitive (i.e., the response key color) parameter. However, this might not be sufficient in more naturalistic situations, in which alternative choices differ in more complex kinematic parameters (e.g., using the right arm or the left arm to respond), or require finer cognitive processing (e.g., selecting the most appropriate behavior according to a specific social context).
Age
We can also see a progressive reduction in RT as the age of the participants increases, which is consistent with decades of findings from developmental studies48. This suggests that motor planning becomes globally more effective and rapid with age, and therefore requires fewer cognitive resources. Given the low sample size, the statistical models tested included the age variable as an additive effect (i.e., irrespective of experimental condition and group membership). Thus, we accounted for the differences attributable to the age of participants in the accuracy and overall kinematic profile. However, we did not specifically assess the role of age in interaction with the other predictors (i.e., experimental condition and group membership). Future studies with appropriate sample sizes and broader age ranges may further investigate developmental changes in inhibitory strategies, also exploring potential ADHD-related differences.
Limitations
It is worth mentioning that the present study has some limitations. As we were not interested in assessing gender differences, our sample is not balanced by participant gender, which reduces its representativeness of the general population. In addition, the sample size was determined by the number of families that agreed to participate in the study. Given the complexity of the experimental design (i.e., multiple dependent and independent measures are of interest), its exploratory nature, and the paucity of prior evidence on which to estimate expected effect sizes and appropriate sample sizes, our sample size may be insufficient to reveal further differences between groups. Further inferential research will be needed to confirm the considerations presented in this paper. Moreover, research on developmental populations with specific conditions frequently suffers from small sample sizes and even single-case studies. Replication of studies, meta-analyses and multi-lab projects would help deal with this issue in the long run of knowledge acquisition, whereby every study contributes to a piece of the puzzle.
Future perspectives
In everyday life, children constantly perform actions that require planning and control, as well as inhibition of automatic behaviors as the demands of their environment change. Further research is needed to investigate the implications of atypical motor and cognitive inhibition on the daily life, learning, and social skills of children with ADHD. For instance, some children with ADHD show stereotypies, which are involuntary, restricted and repetitive patterns of behaviors that limit the child’s resources to learn and practice various, appropriate and goal-directed actions49,50,51. Specifically, motor stereotypies are present in both neurodevelopmental conditions and typical development52, and might be related to ineffective motor planning53 and inhibitory difficulties54. Indeed, motor-related cortical potentials in premotor areas, which anticipate voluntary motor actions, are found to be absent before stereotypy onset in typical development53. Stereotypies are mostly studied in Autism Spectrum Disorder (ASD), as they are core symptoms of those conditions10. However, they are frequently found in ADHD, and show similar characteristics across ASD and ADHD55, which often co-occur, share clinical manifestations, and entail impairments in overlapping mechanisms56,57. Notably, stereotypies can be related to cross-diagnostic sensory, motor, and cognitive mechanisms. Atypical inhibition of prepotent responses is correlated with repetitive behaviors, with differences between higher-order (preoccupations, restricted interests, compulsive routines, ritualistic behaviors) and sensorimotor (repetitive movements and sensory preoccupations) stereotypies58,59. Moreover, stereotypies are associated with sensory difficulties60, that can be present in ADHD61,62,63, and are bounded to motor and cognitive processes through complex, dynamic, and multidirectional relationships. We can speculate that those children with greater stereotypies could have less effective sensory and executive profiles, as well as motor planning difficulties. They might need to devote more resources to motor control to effectively inhibit a prepotent response. Future studies may employ our paradigm to better understand whether atypical cognitive and motor inhibition may contribute to broader individual differences in everyday sensory, cognitive, and social functioning. Studies with more hypothesis-driven approaches and appropriate sample size would allow to draw clearer, more inferential conclusions on the complex relationships between these variables.
This study opens the door to important application challenges in bringing these methods and knowledge into clinical practice. It would be crucial to integrate the kinematic analysis to the classical neuropsychological tests that evaluate executive functions, to better understand how a response to a given test is planned and adjusted along the way. In this regard, the distinction between reaction time and movement duration is a promising perspective for neuropsychological research, as it allows a distinction to be made between two different mechanisms underlying a response (i.e., planning and control). These nuances are often obscured by the use of total response time in the literature. Moreover, this method would facilitate not only the identification of specific difficulties and the monitoring of the treatment effects, but also serve as an intervention tool itself. For instance, using kinematic measures as biofeedback could promote patients' awareness of their behaviors and facilitate learning strategies to modify them. Although the use of inexpensive and portable kinematic sensors removes one of the barriers to its use in the clinic, the difficulty of analyzing and interpreting the raw data obtained with such instruments remains. To overcome this obstacle, it will be necessary for researchers to develop and make available user-friendly software that process the raw kinematic data and calculate performance indices that are interpretable by clinicians. To this end, we first need large-scale validation studies that provide normative values and risk indices to evaluate an individual's performance.
Conclusions
Children with ADHD can exhibit similar accuracy than neurotypical controls in simple tasks tapping on the inhibition of prepotent motor responses. However, accurate inhibition appears to be achieved through different mechanisms, including less motor planning. Although alternative strategies may be sufficient to compensate for planning difficulties in simple experimental tasks, this could profoundly impact the behaviour of children with ADHD in everyday life contexts, which involve very complex choices among numerous possible alternatives. Moreover, motor, and cognitive impulsivity might be related to broader atypicalities, ranging from sensory atypia and stereotypies to executive difficulties in everyday tasks. For this reason, it is fundamental to understand the mechanisms underlying impulsivity, design interventions that are individualized on the child’s profile and synergistically target the motor and cognitive dimensions of executive functions. To this end, the use of portable, user-friendly, and low-cost kinematic sensors offers great possibilities for neuropsychological assessment and treatment, being also affordable for local clinical services. In sum, this study opens the door to further research that will help the scientific and clinical community understand and target impulsivity, leading to benefits on children's developmental trajectory and well-being.
Data availability
The original dataset and analysis script are available from the OSF public repository at the following URL: https://osf.io/gjha4/?view_only=6fa8fe8e2ee2442fb2df64e1dad06ac3.
References
Koziol, L. F., Budding, D. E. & Chidekel, D. From movement to thought: Executive function, embodied cognition, and the cerebellum. The Cerebellum 11, 505–525 (2012).
Thelen, E. Time-scale dynamics and the development of an embodied cognition. Mind Motion Explor. Dyn. Cogn. 2, 69–100 (1995).
Wilson, P. et al. Cognitive and motor function in developmental coordination disorder. Dev. Med. Child Neurol. 62, 1317–1323 (2020).
Sokhadze, E. M., Tasman, A., Sokhadze, G. E., El-Baz, A. S. & Casanova, M. F. Behavioral, cognitive, and motor preparation deficits in a visual cued spatial attention task in autism spectrum disorder. Appl. Psychophysiol. Biofeedback 41, 81–92 (2016).
Diamond, A. Executive functions. Annu. Rev. Psychol. 64, 135–168 (2013).
Leisman, G., Moustafa, A. A. & Shafir, T. Thinking, walking, talking: Integratory motor and cognitive brain function. Front. Public Health 4, 94 (2016).
Henry, M., Joyal, C. C. & Nolin, P. Development and initial assessment of a new paradigm for assessing cognitive and motor inhibition: The bimodal virtual-reality Stroop. J. Neurosci. Methods 210, 125–131 (2012).
Rubia, K. et al. Mapping motor inhibition: Conjunctive brain activations across different versions of Go/No-Go and stop tasks. Neuroimage 13, 250–261 (2001).
Barkley, R. A. Response inhibition in attention-deficit hyperactivity disorder. Ment. Retard. Dev. Disabil. Res. Rev. 5, 177–184 (1999).
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5®). (American Psychiatric Pub, 2013).
Doyle, A. E. Executive functions in attention-deficit/hyperactivity disorder. Science 6, 2666 (2006).
Nigg, J. T. Annual research review: On the relations among self-regulation, self-control, executive functioning, effortful control, cognitive control, impulsivity, risk-taking, and inhibition for developmental psychopathology. J. Child Psychol. Psychiatry 58, 361–383 (2017).
Athanasiadou, A. et al. Early motor signs of attention-deficit hyperactivity disorder: A systematic review. Eur. Child Adolesc. Psychiatry 29, 903–916 (2020).
Farran, E. K. et al. Is the motor impairment in attention deficit hyperactivity disorder (ADHD) a co-occurring deficit or a phenotypic characteristic? Adv. Neurodev. Disord. 4, 253–270 (2020).
Glover, S. Separate visual representations in the planning and control of action. Behav. Brain Sci. 27, 3–24 (2004).
Dahan, A. & Reiner, M. Evidence for deficient motor planning in ADHD. Sci. Rep. 7, 9631 (2017).
Rapport, M. D., Orban, S. A., Kofler, M. J. & Friedman, L. M. Do programs designed to train working memory, other executive functions, and attention benefit children with ADHD? A meta-analytic review of cognitive, academic, and behavioral outcomes. Clin. Psychol. Rev. 33, 1237–1252 (2013).
Cortese, S. et al. Cognitive training for attention-deficit/hyperactivity disorder: Meta-analysis of clinical and neuropsychological outcomes from randomized controlled trials. J. Am. Acad. Child Adolesc. Psychiatry 54, 164–174 (2015).
Moreau, D. Brains and brawn: Complex motor activities to maximize cognitive enhancement. Educ. Psychol. Rev. 27, 475–482 (2015).
Cahill-Rowley, K. & Rose, J. Temporal–spatial reach parameters derived from inertial sensors: Comparison to 3D marker-based motion capture. J. Biomech. 52, 11–16 (2017).
Angeli, A., Valori, I., Farroni, T. & Marfia, G. Reaching to inhibit a prepotent response: A wearable 3-axis accelerometer kinematic analysis. PLoS ONE 16, e0254514 (2021).
Vaurio, R. G., Simmonds, D. J. & Mostofsky, S. H. Increased intra-individual reaction time variability in attention-deficit/hyperactivity disorder across response inhibition tasks with different cognitive demands. Neuropsychologia 47, 2389–2396 (2009).
Miller, J. O. & Low, K. Motor processes in simple, go/no-go, and choice reaction time tasks: A psychophysiological analysis. J. Exp. Psychol. Hum. Percept. Perform. 27, 266–289 (2001).
Dahan, A., Bennet, R. & Reiner, M. How long is too long: An individual time-window for motor planning. Front. Hum. Neurosci. 13, 222 (2019).
Iuppariello, L. et al. A novel approach to estimate the upper limb reaching movement in three-dimensional space. Inform. Med. Unlocked 15, 100155 (2019).
Flash, T. & Hogan, N. The coordination of arm movements: an experimentally confirmed mathematical model. J. Neurosci. 5, 1688 (1985).
Lage, G. M., Malloy-Diniz, L. F., Neves, F. S., de Moraes, P. H. P. & Corrêa, H. A kinematic analysis of the association between impulsivity and manual aiming control. Hum. Mov. Sci. 31, 811–823 (2012).
Sonuga-Barke, E. & Thapar, A. The neurodiversity concept: Is it helpful for clinicians and scientists? Lancet Psychiatry 8, 559–561 (2021).
Wåhlstedt, C., Thorell, L. B. & Bohlin, G. Heterogeneity in ADHD: Neuropsychological pathways, comorbidity and symptom domains. J. Abnorm. Child Psychol. 37, 551–564 (2009).
Schweiger, M. & Marzocchi, G. M. Lo sviluppo delle Funzioni Esecutive: Uno studio su ragazzi dalla terza elementare alla terza media. G. Ital. Psicol. 35, 353–374 (2008).
McIntosh, D., Miller, L. & Shyu, V. Development and validation of the short sensory profile (SSP). Sens. Profile Exam. Man. Dunn W Ed 2, 59–73 (1999).
Fulceri, F. et al. Application of the repetitive behavior scale-revised–Italian version–in preschoolers with autism spectrum disorder. Res. Dev. Disabil. 48, 43–52 (2016).
Wagenmakers, E.-J. & Farrell, S. AIC model selection using Akaike weights. Psychon. Bull. Rev. 11, 192–196 (2004).
Nakagawa, S. & Schielzeth, H. A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol. Evol. 4, 133–142 (2013).
R Core Team. R: A language and environment for statistical computing. (2020).
Brooks, M. E. et al. glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J. 9, 378–400 (2017).
Bates, D., Mächler, M., Bolker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 67, 1–48 (2015).
Lage, G. M. et al. A kinematic analysis of manual aiming control on euthymic bipolar disorder. Psychiatry Res. 208, 140–144 (2013).
Suskauer, S. J. et al. fMRI of intrasubject variability in ADHD: Anomalous premotor activity with prefrontal compensation. J. Am. Acad. Child Adolesc. Psychiatry 47, 1141–1150 (2008).
Craighero, L. & Rizzolatti, G. CHAPTER 31 - The Premotor Theory of Attention. in Neurobiology of Attention (eds. Itti, L., Rees, G. & Tsotsos, J. K.) 181–186 (Academic Press, 2005). https://doi.org/10.1016/B978-012375731-9/50035-5.
Smith, D. T. & Schenk, T. The premotor theory of attention: Time to move on? Neuropsychologia 50, 1104–1114 (2012).
Mari, M., Castiello, U., Marks, D., Marraffa, C. & Prior, M. The reach–to–grasp movement in children with autism spectrum disorder. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 358, 393–403 (2003).
Glover, S. & Dixon, P. Semantics affect the planning but not control of grasping. Exp. Brain Res. 146, 383–387 (2002).
Demers, M. M., McNevin, N. & Azar, N. R. ADHD and motor control: A review of the motor control deficiencies associated with attention deficit/hyperactivity disorder and current treatment options. Crit. Rev. Phys. Rehabil. Med. 25, 55558 (2013).
Castiello, U. The neuroscience of grasping. Nat. Rev. Neurosci. 6, 726–736 (2005).
Krigolson, O. E., Cheng, D. & Binsted, G. The role of visual processing in motor learning and control: Insights from electroencephalography. Vision Res. 110, 277–285 (2015).
Boulenger, V. et al. Subliminal display of action words interferes with motor planning: A combined EEG and kinematic study. J. Physiol. Paris 102, 130–136 (2008).
Eckert, H. M. & Eichorn, D. H. Developmental variability in reaction time. Child Dev. 48, 452–458 (1977).
Mahone, E. M., Bridges, D., Prahme, C. & Singer, H. S. Repetitive arm and hand movements (complex motor stereotypies) in children. J. Pediatr. 145, 391–395 (2004).
Mahone, E. M., Ryan, M., Ferenc, L., Morris-Berry, C. & Singer, H. S. Neuropsychological function in children with primary complex motor stereotypies. Dev. Med. Child Neurol. 56, 1001–1008 (2014).
Harris, K. M., Mahone, E. M. & Singer, H. S. Nonautistic motor stereotypies: Clinical features and longitudinal follow-up. Pediatr. Neurol. 38, 267–272 (2008).
Ghosh, D., Rajan, P. V. & Erenberg, G. A comparative study of primary and secondary stereotypies. J. Child Neurol. 28, 1562–1568 (2013).
Houdayer, E. et al. Absent movement-related cortical potentials in children with primary motor stereotypies: Premotor Potentials in Stereotypies. Mov. Disord. 29, 1134–1140 (2014).
Mirabella, G., Mancini, C., Valente, F. & Cardona, F. Children with primary complex motor stereotypies show impaired reactive but not proactive inhibition. Cortex 124, 250–259 (2020).
Brierley, N. J. et al. Factor structure of repetitive behaviors across autism spectrum disorder and attention-deficit/hyperactivity disorder. J. Autism Dev. Disord. 51, 3391–3400 (2021).
Jang, J. et al. Rates of comorbid symptoms in children with ASD, ADHD, and comorbid ASD and ADHD. Res. Dev. Disabil. 34, 2369–2378 (2013).
Sokolova, E. et al. A causal and mediation analysis of the comorbidity between attention deficit hyperactivity disorder (ADHD) and autism spectrum disorder (ASD). J. Autism Dev. Disord. 47, 1595–1604 (2017).
Mosconi, M. W. et al. Impaired inhibitory control is associated with higher-order repetitive behaviors in autism spectrum disorders. Psychol. Med. 39, 1559–1566 (2009).
Faja, S. & Nelson, D. L. Variation in restricted and repetitive behaviors and interests relates to inhibitory control and shifting in children with autism spectrum disorder. Autism 23, 1262–1272 (2019).
Fetta, A. et al. Relationship between sensory alterations and repetitive behaviours in children with autism spectrum disorders: A parents’ questionnaire based study. Brain Sci. 11, 484 (2021).
Shimizu, V. T., Bueno, O. F. A. & Miranda, M. C. Sensory processing abilities of children with ADHD. Braz. J. Phys. Ther. 18, 343–352 (2014).
Little, L. M., Dean, E., Tomchek, S. & Dunn, W. Sensory processing patterns in autism, attention deficit hyperactivity disorder, and typical development. Phys. Occup. Ther. Pediatr. 38, 243–254 (2018).
Fuermaier, A. B. M. et al. Perception in attention deficit hyperactivity disorder. ADHD Atten. Deficit Hyperact. Disord. 10, 21–47 (2018).
Acknowledgements
We sincerely thank the psychologists Alessandra Cagnin and Donatella Benetti for their enthusiastic collaboration on this project. Our gratitude to the students Irene Guglielminetti, Krizia Indri, and Giulia Mantovani, who contributed to data collection. Many thanks to Andrea Janna for his technical support.
Author information
Authors and Affiliations
Contributions
I.V., L.D.L and T.F. conceived the experiment, A.A. implemented the experiment, I.V. collected the data, I.V., A.A. and L.D.L. analyzed the results, I.V. wrote the original draft of the manuscript, T.F. and G. M. supervised the project and acquired fundings. All authors reviewed and edited the manuscript and approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Valori, I., Della Longa, L., Angeli, A. et al. Reduced motor planning underlying inhibition of prepotent responses in children with ADHD. Sci Rep 12, 18202 (2022). https://doi.org/10.1038/s41598-022-22318-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-22318-6
This article is cited by
-
Visuomotor tracking strategies in children: associations with neurodevelopmental symptoms
Experimental Brain Research (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.