Abstract
Rheumatoid arthritis (RA) is a chronic systemic autoimmune disease. The present study reported the burden of RA in the Middle East and North Africa (MENA) region from 1990 to 2019 by age, sex, and socio-demographic index (SDI). Publicly available data from the Global Burden of Disease (GBD) 2019 study was used to report the modelled point prevalence, annual incidence, and disability-adjusted life-years (DALYs) of RA, as counts and age-standardised rates with their corresponding 95% uncertainty intervals (UIs). In 2019, RA had an age-standardised point prevalence of 120.6 per 100,000 population (107.0–135.7) and an annual incidence rate of 5.9 (5.2–6.6) in MENA, which have increased 28.3% and 25.2%, respectively, since 1990. In 2019, the number of DALYs due to RA in the region was 103.6 thousand (74.2–136.7), with an age-standardised rate of 19.0 (13.9–24.9) DALYs per 100,000 population, which has increased by 18.6% since 1990 (6.7–28.2). The highest point prevalence was found in females aged 50–54, and in males aged 45–49. The highest number of DALYs was observed in the 50–54 age group. The MENA DALY rate was lower than the global rate (19.0 vs. 39.6 per 100,000), but the rate was higher in all age groups in 2019, when compared with 1990. In addition, from 1990 to 2019 an increased burden from RA was associated with an increase in SDI. In line with global trends, the burden of RA in the MENA region showed a steady increase from 1990 to 2019. This highlights the increasing need for updating the available health data to design more accurate guidelines to enable the early detection and treatment of RA in the MENA countries.
Similar content being viewed by others
Introduction
Rheumatoid arthritis (RA) is a chronic systemic autoimmune disease that is characterized by inflammatory changes in the synovial membranes. The main clinical manifestation of the disease is symmetrical polyarthritis of the small synovial joints of the hands and the feet, which spreads to larger joints as the disease progresses1. The exact etiology of RA remains unknown, although genetic, immunological, and environmental factors all appear to have a role in the pathophysiology of RA2. Multiple risk factors have been proposed for RA, including familial and genetic factors3,4, smoking and alcohol consumption5,6, insufficient physical activity7, high-fat diets8, lower socioeconomic status9, and occupational exposures10.
The Global Burden of Disease (GBD) study 2017 reported the global age-standardised prevalence of RA to be 246.6 (per 100,000 population) and the annual incidence rate to be 14.9 (per 100,000 population), demonstrating increases of 7.4% and 8.2%, respectively, since 1990. Globally, the point prevalence showed an increase with advancing age and these rates were highest in females aged 70–74 and among males aged 75–79 years old11.
The Middle East and North Africa (MENA) region encompasses a wide range of diverse ethnicities, cultures, lifestyles, and weather conditions, all of which may affect the burden of RA12. Therefore, the epidemiological pattern of RA needs to be explored for the individual countries that comprise the MENA region. In 2017, the age-standardised disability-adjusted life-years (DALYs) and annual incidence rates of RA were 36.7 and 15.0 per 100,000 population, respectively, in the MENA region11.
A study using World Health Organization (WHO) data reported the global burden of musculoskeletal diseases in 2000, 2010, and 2015, but did not specifically focus on RA and the estimates contained therein are now outdated13. Similarly, a study using GBD 2017 data reported the global, regional, and national burden of RA from 1990 to 2017, which also needs updating. Furthermore, most of the epidemiological patterns were presented at the global-level and so age and sex patterns of RA, and its attributable risk factors, have not been reported for several regions, including MENA11. Thus, there is a dearth of comprehensive studies specifically aimed at determining the burden of RA and its trends in the MENA region. Also, the rapidly changing economic, social and cultural landscape of the MENA region further underlines the importance of having a more detailed understanding of the disease. Therefore, the present study reported the most up-to-date data on the point prevalence, annual incidence, and DALYs of RA in the MENA region by age, sex and Socio-demographic Index (SDI), from 1990 to 2019.
Methods
Overview
The Institute of Health Metrics and Evaluation (IHME) conducts the GBD studies, which has collected data on the burden of 369 diseases and injuries from 1990 to 2019 in 204 countries and territories, seven super-regions and 21 regions14. The IHME is an independent global health research organisation that is based in the School of Medicine at the University of Washington. The IHME collaborates with partners across the world to produce timely, pertinent, and valid evidence about the global state of health. We used the publicly available GBD data, which were provided by the IHME and are available from the following website: https://vizhub.healthdata.org/gbd-results/. The countries located in the MENA region include: Afghanistan, Algeria, Bahrain, Egypt, Iran (Islamic Republic of), Iraq, Jordan, Kuwait, Lebanon, Libya, Morocco, Oman, Palestine, Qatar, Saudi Arabia, Sudan, Syrian Arab Republic, Tunisia, Turkey, United Arab Emirates and Yemen. A detailed description of the GBD 2019 methodology and the improvements made since 2017 are provided elsewhere14,15. Furthermore, more details on the fatal and non-fatal estimates can be found at https://vizhub.healthdata.org/gbd-compare/ and http://ghdx.healthdata.org/gbd-results-tool.
Case definition and data sources
We provide a summary of the GBD methodology for RA. Whilst RA is a systemic autoimmune disease with articular and extra articular manifestations, the disability weights (DWs) used in GBD 2019 do not include extra articular disabilities. The 1987 American College of Rheumatology (ACR 1987) was used to define RA in GBD 201916. ACR 1987 contains seven diagnostic criteria, which are described elsewhere14,16. Evidence collected from a systematic review was used in GBD 2019 to calculate the prevalence, incidence and mortality of RA. The exclusion criteria included: (1) studies with samples that were not nationally representative, (2) non-population-based studies, (3) those providing inadequate primary data on epidemiological parameters, (4) studies on a specific type of RA (e.g., seropositive RA) and (5) and review articles. Furthermore, studies using inpatient data were also excluded, since they would not be representative of the true prevalence14. GBD 2019 also included USA claims data for 2000, 2010–2012, and 2014–2016 (by state) and Taiwanese claims for 2016 that have not been included in the previous iterations of GBD14. RA mortality was estimated using data from vital registration, verbal autopsy and surveillance sources17. Data were excluded as outliers when they were implausibly high or low (relative to global or regional patterns), varied widely from established age or temporal patterns, or conflicted with other similar data sources from the same locations or locations17. The sources used to estimate the fatal and non-fatal burden of RA are available at: http://ghdx.healthdata.org/gbd-2019/data-input-sources.
Disease model
Premature mortality from RA was estimated using the standard Cause of Death Ensemble model (CODEm), with the covariates included being shown in the appendix section of a recent GBD 2019 publication17. The prevalence, incidence and mortality data concerning RA were entered into DisMod-MR 2.1, which is a Bayesian meta-regression tool used for modeling and calculating estimates by pooling all available heterogeneous data in order to adjust for methodological differences and to examine internal consistency.
It was also assumed that there were no incidence or prevalence numbers in those younger than five years of age14. In order to improve the estimation of the range of case definitions, data from all sources were re-extracted by IHME. The ACR 1987 criteria16 was used as the reference for the other definitions, such as Rome 196118, the American Rheumatology Association 195819 or the European League against Rheumatism criteria20, which were identified using a single study covariate (non‐ACR_1987).
Further study covariates were added using data from administrative health systems, studies which covered regional instead of nationally/sub-nationally representative populations, and also for claims data.
Severity and years lived with disability
The International Classification of Disease 10 (ICD-10) codes for RA (i.e. M05-M06 and M08) were used with three sequelae (severity levels), each of which had a specific DW, which ranged from 0.117 to 0.581 (Table S1). The DW values were calculated using the GBD 2013 European Disability Weights Measurement Study and the GBD 2010 Disability Weights Measurement Study21,22. The proportion of RA patients with each severity level was estimated using the Medical Expenditure Panel Survey (MEPS)21 and these proportions were used to divide the overall prevalence of RA into the three severity categories. Lastly, the severity-specific DWs were multiplied by the point prevalence of each severity category in order to produce years lived with disability (YLDs).
Compilation of results
The years of life lost (YLLs) were estimated by multiplying the number of deaths in each age group by the remaining life expectancy in that age group, using information from the GBD standard life table. DALYs were calculated by adding together the YLDs and YLLs. Furthermore, 95% uncertainty intervals (UIs) were reported for each estimate, which were based upon the 25th and 975th values of 1000 ordered draws. All estimates were standardised using the GBD standard population, which is the non-weighted mean of the 2019 age-specific proportional distributions from the GBD 2019 population estimates for all national locations, except for populations lower than five million. The association between SDI and the burden of RA, in terms of DALYs, was produced using Smoothing Splines models23. SDI is a composite indicator that is based upon average income per capita, average years of schooling for the population older than 15 years of age and total fertility rate under the age of 25. SDI ranges from 0 (less developed) to 1 (most developed). The figures for the final estimates of the point prevalence and annual incidence rates were generated by R Software (V.3.5.2) using data available from http://ghdx.healthdata.org/gbd-results-tool.
Ethics approval and consent to participate
The ethics committee of the Tabriz University of Medical Sciences approved the study (ID: IR.TBZMED.REC.1400.193). All methods were performed in accordance with the national guidelines and regulations.
Results
The Middle East and North Africa region
In 2019, there were 18.6 million (95% UI: 17.0–20.4) prevalent cases of RA globally, with an age-standardised point prevalence of 224.2 per 100,000 population (95% UI: 204.9–246.0), which represents an 8.1% increase since 1990 (95% UI: 7.5–8.6). Also in 2019, globally RA had an age-standardised incidence rate of 13.0 (95% UI: 11.8–14.3) per 100,000 and a DALY rate of 39.6 (95% UI: 30.5–49.5) per 100,000.
In 2019, there were 672.8 thousand (95% UI: 588.7–768.0) prevalent cases of RA in the MENA region, with an age-standardised point prevalence of 120.6 per 100,000 population (95% UI: 107.0–135.7), which represents a 28.3% increase since 1990 (95% UI: 25.5–30.9) (Tables 1 and S2). RA accounted for 36.5 thousand (95% UI: 32.1–41.6) incident cases in 2019, with an age-standardised rate of 5.9 (95% UI: 5.2–6.6), an increase of 25.2% since 1990 (95% UI: 22.4–27.7) (Tables 1 and S3). In 2019, the number of regional DALYs due to RA was 103.6 thousand (95% UI: 74.2–136.7), with an age-standardised rate of 19.0 (95% UI: 13.9–24.9) DALYs per 100,000 population, an increase of 18.6% since 1990 (95% UI: 6.7–28.2) (Tables 1 and S4).
National level
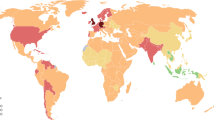
In 2019, the national age-standardised point prevalence of RA among the countries located in the MENA region ranged from 73.9 to 217.6 cases per 100,000 population. Turkey [217.6 (95% UI: 197.0–238.8)], Bahrain [154.8 (95% UI: 135.9–177.8)] and Kuwait [113.3 (95% UI: 96.6–133.9)] had the three highest estimates of age-standardised point prevalence of RA in 2019. In contrast, Yemen [73.9 (95% UI: 63.5–86.5)], Sudan [83.0 (95% UI: 71.6–97.1)] and Afghanistan [87.3 (95% UI: 75.9–100.9)] had the three lowest rates (Fig. 1A and Table S2).
Age-standardised point prevalence (A), incidence (B), and DALYs (C) for rheumatoid arthritis (per 100,000 population) in the Middle East and North Africa region in 2019, by sex and country. DALY disability-adjusted-life-years. (Generated from data available from http://ghdx.healthdata.org/gbd-results-tool).
In 2019, the national age-standardised annual incidence rates of RA among the countries that comprise the MENA region varied from 3.7 to 11.4 cases per 100,000 population. The highest rates were observed in Turkey [11.4 (95% UI: 10.3–12.5)], Bahrain [8.1 (95% UI: 7.1–9.2)] and Kuwait [5.7 (95% UI: 4.9–6.7)]. In contrast, the lowest rates were found in Yemen [3.7 (95% UI: 3.2–4.4)], Sudan [4.2 (95% UI: 3.6–4.8)] and Afghanistan [4.4 (95% UI: 3.8–5.1)] (Fig. 1B and Table S3).
In 2019, the national age-standardised DALY rate of RA among the countries located in the MENA region ranged from 12.5 to 35.4 cases per 100,000 population. The highest rates were observed in Turkey [35.4 (95% UI: 26.2–45.9)], Bahrain [27.6 (95% UI: 20.4–35.7)] and Iran [17.3 (95% UI: 12.4–22.7)]. Conversely, the lowest rates were seen in Yemen [12.5 (95% UI: 8.9–16.4)], Sudan [13.4 (95% UI: 9.6–17.9)] and the United Arab Emirates [14.0 (95% UI: 9.6–18.5)] (Fig. 1C and Table S4).
Increases in the age-standardised point prevalence, from 1990 to 2019, was observed in all the MENA countries, with Saudi Arabia [43.5% (95% UI: 37.5–50.1)], Turkey [41.8% (95% UI: 33.7–49.6)] and Lebanon [40.3% (95% UI: 33.8–48.1)] showing the largest increases over the measurement period (Table S2 and Fig. S1). There were no countries in the MENA region that showed a decrease in the age-standardised incidence or DALY rates of RA from 1990 to 2019. Turkey [43.1% (95% UI: 34.9–51.4)], Saudi Arabia [42.3% (36.9–48.6)] and Bahrain [40.6% (95% UI: 32.4–48.9)] showed the largest increases in the age-standardised incidence rates of RA over the measurement period (Table S3 and Fig. S2). In addition, Bahrain [48.3% (95% UI: 7.6–83.0)], Kuwait [40.8% (95% UI: 23.1–60.9)] and Lebanon [37.4% (95% UI: 14.8–61.7)] showed the largest increases in the age-standardised DALY rate over the same period (Table S4 and Figure S3).
Age and sex patterns
In 2019, the regional point prevalence of RA was highest in the 60–64 age group in females and the 65–69 age group in males, but decreased from there to the oldest age group (95+). Similarly, the number of prevalent cases was highest in the 50–54 and 45–49 age groups in females and males, respectively, and then decreased with increasing age. Furthermore, the global point prevalence and number of prevalent cases of RA were higher in females of all ages (Fig. 2A).
Number of prevalent cases and prevalence (A), number of incident cases and incidence rate (B), and the number of DALYs and DALY rate (C) for rheumatoid arthritis (per 100,000 population) in the Middle East and North Africa region, by age and sex in 2019; Dotted and dashed lines indicate 95% upper and lower uncertainty intervals, respectively. DALY disability-adjusted-life-years. (Generated from data available from http://ghdx.healthdata.org/gbd-results-tool).
In 2019, the regional incidence rate of RA was highest in females aged 45–49 and in males 50–54 years old. The number of incident cases peaked in the 40–44 and 35–39 age groups for males and females, respectively, after which there was a decline with increasing age (Fig. 2B).
There was a clear increase in the regional DALY rate of RA up to the 65–69 age group, followed by a decrease in the remaining age groups up to the oldest age group (95+). Females had a substantially higher DALY rate, with the highest number of DALYs being in the 50–54 years age group. The number of DALYs was higher in females, than males, in all age groups (Fig. 2C).
The rate ratio comparing the age-standardised DALY rates in MENA to global rates for different age groups, in 1990 and 2019, showed an increase up to the 25–39 age group in males and up to 20–24 age group in females, while the rate decreased thereafter in all age groups except for increases for females in the 90–94 and 95+ age groups. Although the MENA DALY rate was lower than the global rate, the DALY ratio for the MENA/Global rate of RA was higher in 2019 in all age groups than in 1990, meaning that the RA burden in the MENA region is heading towards the global level (Fig. 3).
Ratio of the Middle East and North Africa region to global rheumatoid arthritis DALY rate according to age group and sex, 1990–2019. DALY disability-adjusted-life-years. MENA Middle East and North Africa. (Generated from data available from http://ghdx.healthdata.org/gbd-results-tool).
Association with the socio-demographic index (SDI)
In the period 1990 to 2019, the burden of RA generally increased with increasing socio-economic development, up to an SDI of around 0.7, but then decreased slightly as SDI increased. Countries such as Turkey, Bahrain, Iran, Palestine, and Afghanistan had much higher than expected burdens, whereas countries such as Sudan, the Syrian Arab Republic, Algeria, Lebanon, Egypt, Tunisia, and Libya had much lower than expected burdens (Fig. 4).
Age-standardised DALY rates of rheumatoid arthritis for 21 countries and territories in 2019, by SDI; Expected values based on the Socio-demographic Index and disease rates in all locations are shown as the black line. Each point shows the observed age-standardised DALY rate for each country in 2019. DALY disability-adjusted-life-years. SDI Socio-demographic Index (Generated from data available from http://ghdx.healthdata.org/gbd-results-tool).
Discussion
In the current study, the prevalence, incidence, and DALY counts and age-standardised rates were reported from 1990 to 2019 for RA in the 21 countries that comprise the MENA region, based on GBD 2019 data. Regionally, there were almost 672 thousand prevalent cases, 36 thousand incident cases and 103 thousand DALYs. Turkey, Bahrain and Kuwait had the highest national age-standardised prevalence and incidence rates, while the lowest national age-standardised prevalence and incidence rates were observed in Yemen, Sudan and Afghanistan. Turkey, Bahrain and Iran had the highest national age-standardised DALY rates, while Yemen, Sudan and the United Arab Emirates had the lowest national age-standardised DALY rates.
Previous studies have generally focused on describing global trends of RA and reporting the worldwide burden of the disease11,24, with the occasional small scale national report25,26,27,28,29,30,31,32. Therefore, there is a lack of comprehensive studies aimed specifically at analyzing the epidemiological patterns and burden of RA in MENA. In this study, we took an in depth look at the epidemiology and burden of RA in the countries that comprise the MENA region.
GBD 2019 shows that in the MENA region, RA was responsible for 103 thousand DALYs, which is different from the 187 thousand regional DALYs reported by GBD 201711. In addition, the number of prevalent and incident cases in this region, 673 thousand and 36 thousand, respectively, were much lower in our study than that found in GBD 2017, which reported 1.3 million prevalent cases and 83.7 thousand incident cases in MENA11. The age-standardised point prevalence, annual incidence and DALY rates observed in GBD 2019 were 120.6, 5.9 and 19.0 per 100,000 population, respectively, which were also much lower than those estimated in GBD 2017, which were 259.3, 15.0 and 36.7 (per 100,000 population, respectively) in MENA11.
As for the changes in the regional trends between 1990 and 2019, the age-standardised point prevalence, annual incidence and DALY rates have increased by 28.3%, 25.2% and 18.6%, respectively. These findings were in line with the GBD 2017 report of 13.3%, 12.8% and 6.0%, respectively, which did not show any substantial differences11. However, any disparities observed in the findings of the different GBD studies can mostly be attributed to the inclusion of additional data sources and the use of new methodologies in more recent GBD iterations. Furthermore, the elevated incidence and prevalence of RA may also be related to an increased awareness of RA12.
The present study also revealed that for both males and females, the point prevalence rates and the number of prevalent cases, as well as the annual incidence rates and the number of incident cases, increased steadily with increasing age, peaking between the 6th-7th and the 4th-5th decades, respectively. After the peak, the numbers declined steadily across the older age groups. Furthermore, the number of DALYs was higher in females (than males) across all age groups. The age and sex patterns observed in our study were consistent with the findings of GBD 2017, which reported that globally females and individuals aged 70–74, had the highest burden of RA11. These findings highlight the importance of targeting females and the elderly with primary and secondary preventive programs.
In the present study, we also examined the relationship between the development level and the burden of RA in the MENA region. GBD 2019 shows an overall positive association between the burden of RA and SDI, up to an SDI of 0.7, after which the association was reversed. Previously, GBD 2017 reported a linear association between the burden of RA and SDI in the MENA region11. It is important to note that the association between the burden of RA and SDI was not simple to interpret, since the constituent countries of MENA had lower or higher than expected burdens.
At the national level, a 2018 study conducted in Turkey, which used the 1987 ACR criteria for RA, reported the age-standardised point prevalence of RA for the general population to be 0.56%, which is higher than our estimates based on GBD 2019 findings (0.21% (95% CI: 0.21–0.25)25. In a 2012 study carried out in Iran, which used the COPCORD Core Questionnaire (CCQ) to screen 19,786 people, the crude prevalence of RA was observed to be 0.37% (95% CI: 0.29–0.46), which is again higher than our estimates based on GBD 2019 data (0.12% (95% CI: 0.11–0.14)27. In addition, a 2010 study conducted in Algeria, on 125,253 individuals using the 1987 ACR criteria, reported that a prevalence of RA of 0.13% (95% CI: 0.10–0.17) in Barika, Algeria31. This study also calculated the prevalence of RA to be 0.15% for the total population of Algeria31, which is not substantially different to our findings (0.11% [95% CI: 0.09- 0.13]). The reasons for the disparities between our findings and those from previous national studies are likely to be due to sample size restrictions and methodological differences.
The overall findings from the present study, as well as the findings from previous GBD reports, highlight the ever-increasing burden of RA in the MENA region. As a result, the need for early detection and treatment strategies, along with effective preventive measures, is even more important than ever. One approach for controlling RA is to focus on modifiable risk factors. Previous research has reported a number of risk factors for RA, with cigarette smoking being the most important modifiable risk factor by far, since smoking has a clear association with RA5. Smoking cessation has been shown to reduce the risk of RA over time, although it does not have an immediate effect. In other words, twenty to thirty years after quitting, ex-smokers still have modestly higher rates of RA when compared with lifetime nonsmokers33. Therefore, policies to control and decrease smoking are of particular interest in the prevention of RA. Globally, between 1990 and 2019, the age-standardised point prevalence of smoking decreased by 27.5% in males and 37.7% in females34. In the MENA region, during that same period, a decrease in smoking was most evident among the male population (11.2%), but was also seen to a lesser extent in females (2.9%)34. In 2019, Turkey, which had the highest point prevalence and annual incidence rates in our study, was among the top ten countries for the proportion of tobacco smokers34. Afghanistan, which is among the countries with the lowest point prevalence and annual incidence rates of RA in the region, had a low age-standardized point prevalence of smoking in 2019, although the rate has increased rapidly by 179% and 205% in females and males, respectively, since 1990, which is also in accordance with the increases in the point prevalence and annual incidence of RA in this country34. Conversely, Bahrain has the second highest national age-standardized point prevalence and annual incidence rates for RA in the region, but the age-standardised smoking prevalence were not among the highest rates in the region and smoking has declined substantially since 199034. Furthermore, research has shown the smoking attributable burden of RA to be 9.6%, with the remaining burden being comprised of other unknown factors15.
Depression has been found to be the most prevalent comorbidity of RA35,36,37. For example, a UK study of 4187 RA patients found that after five years 23.7% of men and 35.5% of women had been diagnosed with depression38. The predictive factors for depression among RA patients include high tension, low self-esteem, passive coping, fatigue, pain39, and female sex38. In addition, higher levels of rheumatoid factor, low income and low levels of mental health have been shown to be associated with depression in patients with RA40. Furthermore, serum interleukin-17 levels have been found to be significantly higher in RA patients with anxiety, compared with healthy controls and RA patients without anxiety41. Moreover, clinical depression raises the risk of mortality in RA patients, with a hazard ratio of 2.242, which highlights the substantial effect of depression on the burden of RA. Interestingly, it seems that the association between depression and RA is bidirectional43,44,45. Previous studies have found a higher incidence rate of RA among depressed patients (compared to non-depressed controls) and a higher rate of depression among RA patients, compared to those without RA46,47. Furthermore, it appears that antidepressants may protect against developing RA47. In conclusion, screening programs for the early diagnosis and treatment of depression among RA patients should be developed to control and reduce the burden of RA.
Another important, albeit non-modifiable risk factor for RA, is old age. The MENA region is one of the seven super-regions that have seen increased life-expectancy at birth over the past seven decades17. In addition, the countries in the MENA region are considered to be in the mid-transition stage, where the crude rate of birth is only just beginning to fall, meaning that MENA countries have large and still growing populations, and as their populations age they will have an increased burden of RA17.
Moreover, the burden of RA in the region is also affected by disease severity. For that reason, strategies aimed at reducing disease severity are another potential approach for lowering the burden of RA in the MENA region. Disease severity in RA is assessed objectively using a disease activity score, which is based on 28 joint assessments (i.e., Disease Activity Score—DAS28)48. Studies show that lower Gross Domestic Product (GDPs) per capita is associated with higher DAS28 scores in RA sufferers49,50. Also, in countries with lower GDPs, the level of access and use of biological disease-modifying antirheumatic drugs (DMARDs) is much lower than in high GDP countries (7.5% vs 25.0%)50. Biological DMARDs are believed to be more effective for controlling RA, compared with conventional synthetic DMARDs51,52,53. Over the years, many studies have suggested that the observed association between low socioeconomic welfare and high DAS28 (in poorer countries) was due to the low use of biological DMARDs54,55,56. However, a more in-depth study has demonstrated that the intake of biological DMARDs is only responsible for 6.7% of the association between low GDP and high DAS28 scores49. These findings clearly indicate that not all of the improvements in DAS28 in high GDP countries can be explained by their access to better medication, instead other factors related to healthcare systems might be responsible for the observed patterns, including higher coverage of specialised rheumatological care, early referral strategies and tight disease control49.
Another obstacle specific to the region and individual countries, is the poor implementation of standardised rheumatological care guidelines57. For example, the new American College of Rheumatology and European League Against Rheumatism (ACR/EULAR) criteria, is accepted by many regional rheumatological associations and has the added benefit of including early RA diagnostic criteria, which can lead to an improved prognosis by initiating early diagnosis and treatment24. However, despite the relevance and applicability of the EULAR guidelines, the use of these guidelines in clinical practice in MENA is low, due to the perceived cost of these strategies57. Therefore, it has been suggested that local guidelines be designed which are specific to the needs, resources and challenges in each county, which can help to ensure an evidence-based approach in rheumatological care and to improve patient outcomes by facilitating more timely access to medication and specialised care57. Several recommendations have been published in order to address this issue26,58. Additionally, it has been suggested that data needs to be collected about the prevalence and epidemiology of RA in these countries, and that healthcare professionals, insurance companies and service providers, as well as patients, need to be educated about the burden of RA and the indirect costs arising from the suboptimal management of the disease57.
The cultural background of many countries in the MENA region means that patients are more inclined to seek help from herbal remedies and alternative medicines, rather than referring to specialist medical care59,60. This means that even in some of the more wealthy MENA countries, where medication and specialist care is accessible for free or at very reasonable prices, the timely management of the disease is still hindered by patient preferences and health literacy59. Finally, the necessity of RA prevention and control strategies should not be judged solely based upon the observed rates, but also on the burden of the disease that are anticipated in the future in the MENA region.
Strengths and limitations of the study
To our knowledge, no previous study has comprehensively reported the burden of RA and the trends by demographic variables in the MENA region. Consequently, one strength of this study is that it used the most up-to-date evidence to estimate the burden of RA in the MENA region. However, our study also has the same limitations reported in previous GBD publications15,17. It should also be noted that due to the limited availability of population-based national data on the incidence and prevalence rates, in some countries the estimates presented here were based on models produced in DisMod-MR 2.1. Model performance may vary from country to country, depending on data availability, but the goal is to make as accurate an estimate as possible, by employing data from countries that are in a similar situation. Therefore, the national estimates presented here should be viewed with some degree of caution, especially those for countries like Afghanistan, Sudan and Yemen, which have been dealing with the devastating effects of war and social unrest. Also, owing to the effects of biological DMARDs in ameliorating the severity of RA and improving disease prognosis51, the availability of these treatment options should be taken into consideration in estimating the DWs and severity distribution within the different countries and regions24. Using the 1987 ACR criteria, instead of the new ACR/EULAR criteria which includes early symptoms of RA, may lead to an underestimation of the actual burden of RA, especially in regards to milder forms of RA. Therefore, the new ACR/EULAR criteria appears to be a better tool for estimating the actual burden of RA in future studies24,61. Also, GBD 2019 did not include the extra-articular symptoms of RA, which can affect the DW. Therefore, we suggest the modification of the DWs in further GBD iterations, based on extra-articular symptoms24.
Conclusions
The burden of RA in the MENA region shows a steady increase from 1990 to 2019, and is expected to reach the global average levels in the upcoming years. This highlights the increasingly urgent need to update the available health data in order to design more accurate guidelines for the early detection and treatment of RA in the countries that comprise the MENA region.
Data availability
The data used for these analyses are all publicly available at http://ghdx.healthdata.org/gbd-results-tool.
Abbreviations
- RA:
-
Rheumatoid arthritis
- DALYs:
-
Disability-adjusted life-years
- GBD:
-
Global burden of disease
- SDI:
-
Socio-demographic index
- MENA:
-
Middle East and North Africa
- UIs:
-
Uncertainty intervals
- ACR:
-
American College of Rheumatology
- EULAR:
-
European League Against Rheumatism
- CCQ:
-
COPCORD core questionnaire
- YLLs:
-
Years of life lost
- YLDs:
-
Years lived with disability
- MEPS:
-
Medical expenditure panel survey
- CODEm:
-
Cause of death ensemble model
- DWs:
-
Disability weights
- IHME:
-
Institute for Health Metrics and Evaluation
- WHO:
-
World Health Organization
- DMARDs:
-
Disease-modifying antirheumatic drugs
- DAS28:
-
Disease activity score, based on 28 joint assessments
References
Khurana, R. & Berney, S. M. Clinical aspects of rheumatoid arthritis. Pathophysiology 12(3), 153–165 (2005).
Scherer, H. U., Häupl, T. & Burmester, G. R. The etiology of rheumatoid arthritis. J. Autoimmun. 110, 102400 (2020).
Frisell, T. et al. Familial risks and heritability of rheumatoid arthritis: Role of rheumatoid factor/anti–citrullinated protein antibody status, number and type of affected relatives, sex, and age. Arthr. Rheum. 65(11), 2773–2782 (2013).
Okada, Y. et al. Genetics of rheumatoid arthritis contributes to biology and drug discovery. Nature 506(7488), 376–381 (2014).
Sugiyama, D. et al. Impact of smoking as a risk factor for developing rheumatoid arthritis: A meta-analysis of observational studies. Ann. Rheum. Dis. 69(01), 70 (2010).
Di Giuseppe, D., Alfredsson, L., Bottai, M., Askling, J. & Wolk, A. Long term alcohol intake and risk of rheumatoid arthritis in women: A population based cohort study. BMJ Brit. Med. J. 345, e4230 (2012).
Qin, B. et al. Body mass index and the risk of rheumatoid arthritis: a systematic review and dose-response meta-analysis. Arthritis Res. Ther. 17(1), 86 (2015).
Di Giuseppe, D., Wallin, A., Bottai, M., Askling, J. & Wolk, A. Long-term intake of dietary long-chain n-3 polyunsaturated fatty acids and risk of rheumatoid arthritis: a prospective cohort study of women. Ann. Rheum. Dis. 73(11), 1949 (2014).
Ghawi, H. et al. A novel measure of socioeconomic status using individual housing data to assess the association of SES with rheumatoid arthritis and its mortality: A population-based case–control study. BMJ Open 5(4), e006469 (2015).
Ilar, A., Alfredsson, L., Wiebert, P., Klareskog, L. & Bengtsson, C. Occupation and risk of developing rheumatoid arthritis: Results from a population-based case-control study. Arthr. Care Res. 70(4), 499–509 (2018).
Safiri, S. et al. Global, regional and national burden of rheumatoid arthritis 1990–2017: A systematic analysis of the Global Burden of Disease study 2017. Ann. Rheum. Dis. 78(11), 1463 (2019).
Almoallim, H. et al. A review of the prevalence and unmet needs in the management of rheumatoid arthritis in Africa and the Middle East. Rheumatol. Therapy. 8(1), 1–16 (2021).
Sebbag, E. et al. The world-wide burden of musculoskeletal diseases: a systematic analysis of the World Health Organization Burden of Diseases Database. Ann. Rheum. Dis. 78(6), 844 (2019).
Vos, T. et al. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 396(10258), 1204–1222 (2020).
Murray, C. J. L. et al. Global burden of 87 risk factors in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 396(10258), 1223–1249 (2020).
Arnett, F. C. et al. The American rheumatism association 1987 revised criteria for the classification of rheumatoid arthritis. Arthr. Rheum. 31(3), 315–324 (1988).
Wang, H. et al. Global age-sex-specific fertility, mortality, healthy life expectancy (HALE), and population estimates in 204 countries and territories, 1950–2019: A comprehensive demographic analysis for the Global Burden of Disease Study 2019. Lancet 396(10258), 1160–1203 (2020).
Kellgren, J. H. Diagnostic criteria for population studies. Bull. Rheum. Dis. 13, 291–292 (1962).
Ropes, M. W., Bennett, G. A., Cobb, S., Jacox, R. & Jessar, R. A. Proposed diagnostic criteria for rheumatoid arthritis. Ann. Rheum. Dis. 16(1), 118 (1957).
Vonkeman, H. E. & van de Laar, M. A. F. J. The new European League Against Rheumatism/American College of Rheumatology diagnostic criteria for rheumatoid arthritis: how are they performing?. Curr. Opin. Rheumatol. 25(3), 354–359 (2013).
James, S. L. et al. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 392(10159), 1789–1858 (2018).
Salomon, J. A. et al. Disability weights for the global burden of disease 2013 study. Lancet Glob. Health 3(11), e712–e723 (2015).
Wang Y. Smoothing Splines: Methods and Applications. (Chapman and Hall/CRC, 2019).
Cross, M. et al. The global burden of rheumatoid arthritis: Estimates from the Global Burden of Disease 2010 study. Ann. Rheum. Dis. 73(7), 1316 (2014).
Tuncer, T. et al. Prevalence of rheumatoid arthritis and spondyloarthritis in Turkey: A nationwide study. Arch. Rheum. 33(2), 128–136 (2018).
Alhajeri, H. et al. Kuwait association of rheumatology 2018 treatment recommendations for patients with rheumatoid arthritis. Rheumatol. Int. 39(9), 1483–1497 (2019).
Davatchi, F. et al. Epidemiology of rheumatic diseases in Iran from analysis of four COPCORD studies. Int. J. Rheum. Dis. 19(11), 1056–1062 (2016).
Chaaya, M. et al. High burden of rheumatic diseases in Lebanon: A COPCORD study. Int. J. Rheum. Dis. 15(2), 136–143 (2012).
Alawneh, K. M., Madanat, W. Y., Alawneh, D. & Smadi, M. S. Prevalence of rheumatoid arthritis among hospital workers in the north of Jordan: Preliminary report of a hospital-based cohort study. Ann. Med. Surg. 60, 579–582 (2020).
Namas, R., Joshi, A., Ali, Z., Al Saleh, J. & Abuzakouk, M. Demographic and clinical patterns of rheumatoid arthritis in an emirati cohort from United Arab Emirates. Int. J. Rheumatol. 2019, 3057578 (2019).
Slimani, S. & Ladjouze-Rezig, A. Prevalence of rheumatoid arthritis in an urban population of Algeria: A prospective study. Rheumatology 53(3), 571–573 (2014).
Al-Dalaan, A. et al. The prevalence of rheumatoid arthritis in the qassim region of Saudi Arabia. Ann. Saudi Med. 18(5), 396–397 (1998).
Di Giuseppe, D., Orsini, N., Alfredsson, L., Askling, J. & Wolk, A. Cigarette smoking and smoking cessation in relation to risk of rheumatoid arthritis in women. Arthr. Res. Ther. 15(2), R56 (2013).
Reitsma, M. B. et al. Spatial, temporal, and demographic patterns in prevalence of smoking tobacco use and attributable disease burden in 204 countries and territories, 1990–2019: a systematic analysis from the Global Burden of Disease Study 2019. Lancet 397(10292), 2337–2360 (2021).
Dougados, M. et al. Prevalence of comorbidities in rheumatoid arthritis and evaluation of their monitoring: Results of an international, cross-sectional study (COMORA). Ann. Rheum. Dis. 73(1), 62–68 (2014).
Matcham, F., Rayner, L., Steer, S. & Hotopf, M. The prevalence of depression in rheumatoid arthritis: A systematic review and meta-analysis. Rheumatol. (Oxf.) 52(12), 2136–2148 (2013).
Fu, X. et al. The prevalence of depression in rheumatoid arthritis in China: A systematic review. Oncotarget 8(32), 53623–53630 (2017).
Jacob, L., Rockel, T. & Kostev, K. Depression risk in patients with rheumatoid arthritis in the United Kingdom. Rheumatol. Ther. 4(1), 195–200 (2017).
Covic, T., Tyson, G., Spencer, D. & Howe, G. Depression in rheumatoid arthritis patients: demographic, clinical, and psychological predictors. J Psychosom Res. 60(5), 469–476 (2006).
Ho, R. C. M., Fu, E. H. Y., Chua, A. N. C., Cheak, A. A. C. & Mak, A. Clinical and psychosocial factors associated with depression and anxiety in Singaporean patients with rheumatoid arthritis. Int. J. Rheum. Dis. 14(1), 37–47 (2011).
Liu, Y., Ho, R. C. M. & Mak, A. The role of interleukin (IL)-17 in anxiety and depression of patients with rheumatoid arthritis. Int. J. Rheum. Dis. 15(2), 183–187 (2012).
Ang, D. C., Choi, H., Kroenke, K. & Wolfe, F. Comorbid depression is an independent risk factor for mortality in patients with rheumatoid arthritis. J. Rheumatol. 32(6), 1013–1019 (2005).
Vallerand, I. A., Patten, S. B. & Barnabe, C. Depression and the risk of rheumatoid arthritis. Curr. Opin. Rheumatol. 31(3), 279–284 (2019).
Fakra, E. & Marotte, H. Rheumatoid arthritis and depression. Joint Bone Spine 88(5), 105200 (2021).
Ng, C. Y. H. et al. Elucidating a bidirectional association between rheumatoid arthritis and depression: A systematic review and meta-analysis. J. Affect. Disord. 311, 407–415 (2022).
Lu, M. C. et al. Bidirectional associations between rheumatoid arthritis and depression: A nationwide longitudinal study. Sci. Rep. 6, 20647 (2016).
Vallerand, I. A. et al. Depression as a risk factor for the development of rheumatoid arthritis: A population-based cohort study. RMD Open 4(2), e000670 (2018).
van Riel, P. L. & Renskers, L. The Disease Activity Score (DAS) and the disease activity score using 28 joint counts (DAS28) in the management of rheumatoid arthritis. Clin. Exp. Rheumatol. 34(5 Suppl 101), S40–S44 (2016).
Putrik, P. et al. Lower education and living in countries with lower wealth are associated with higher disease activity in rheumatoid arthritis: results from the multinational COMORA study. Ann. Rheum. Dis. 75(3), 540 (2016).
Sokka, T. et al. Disparities in rheumatoid arthritis disease activity according to gross domestic product in 25 countries in the QUEST–RA database. Ann. Rheum. Dis. 68(11), 1666 (2009).
Krishnan, E., Lingala, B., Bruce, B. & Fries, J. F. Disability in rheumatoid arthritis in the era of biological treatments. Ann. Rheum. Dis. 71(2), 213 (2012).
Aletaha, D. & Smolen, J. S. Diagnosis and management of rheumatoid arthritis: A review. JAMA 320(13), 1360–1372 (2018).
Moufarrej, M. N., Mahfoud, Z. & Badsha, H. Barriers to achieving controlled rheumatoid arthritis in the United Arab Emirates: A cross-sectional study. Rheumatol. Int. 35(4), 759–763 (2015).
Putrik, P. et al. Inequities in access to biologic and synthetic DMARDs across 46 European countries. Ann. Rheum. Dis. 73(1), 198 (2014).
Elshafie, A. I. et al. Active rheumatoid arthritis in Central Africa: A comparative study between Sudan and Sweden. J. Rheumatol. 43(10), 1777 (2016).
Putrik, P. et al. Variations in criteria regulating treatment with reimbursed biologic DMARDs across European countries. Are differences related to country’s wealth?. Ann. Rheum. Dis. 73(11), 2010 (2014).
El Zorkany, B. et al. Suboptimal management of rheumatoid arthritis in the Middle East and Africa: Could the EULAR recommendations be the start of a solution?. Clin. Rheumatol. 32(2), 151–159 (2013).
Omair, M. A., Omair, M. A. & Halabi, H. Survey on management strategies of rheumatoid arthritis in Saudi Arabia: A Saudi society for rheumatology initiative. Int. J. Rheum. Dis. 20(9), 1185–1192 (2017).
Tan, M., Uzun, O. & Akçay, F. Trends in complementary and alternative medicine in Eastern Turkey. J. Altern. Complement. Med. 10(5), 861–865 (2004).
Alrowais, N. A. & Alyousefi, N. A. The prevalence extent of complementary and alternative medicine (CAM) use among Saudis. Saudi Pharm. J. 25(3), 306–318 (2017).
Cornec, D. et al. Comparison of ACR 1987 and ACR/EULAR 2010 criteria for predicting a 10-year diagnosis of rheumatoid arthritis. Joint Bone Spine 79(6), 581–585 (2012).
Acknowledgements
We would like to thank the Institute for Health Metrics and Evaluation staff and its collaborators who prepared these publicly available data. This is a Master of Public Health (MPH) thesis and we would also like to acknowledge the support of the Connective Tissue Diseases Research Center at Tabriz University of Medical Sciences, Tabriz, Iran. This study is based on publicly available data and solely reflects the opinion of its authors and not that of the Institute for Health Metrics and Evaluation.
Funding
The Bill and Melinda Gates Foundation, who were not involved in any way in the preparation of this manuscript, funded the GBD study. The Tabriz University of Medical Sciences, Tabriz, Iran (Grant No. 66869) also supported the present report.
Author information
Authors and Affiliations
Contributions
S.S. designed the study. S.S., S.E.M. and S.A.N. analyzed the data and performed the statistical analyses. S.E.M., S.A.N., A.K., M.A., M.J.M.S., J.S.K., G.S.C. and S.S. drafted the initial manuscript. All authors reviewed the drafted manuscript for critical content. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mousavi, S., Nejadghaderi, S., Khabbazi, A. et al. The burden of rheumatoid arthritis in the Middle East and North Africa region, 1990–2019. Sci Rep 12, 19297 (2022). https://doi.org/10.1038/s41598-022-22310-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-22310-0
This article is cited by
-
Consensus-Based Overarching Principles and Recommendations on the Use of Biosimilars in the Treatment of Inflammatory Arthritis in the Gulf Region
BioDrugs (2024)
-
The rheumatology workforce in the Arab countries: current status, challenges, opportunities, and future needs from an ArLAR cross-sectional survey
Rheumatology International (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.