Abstract
The transtheoretical model (TTM) is a commonly used model of health-related behavioral change. However, the practical effect of using this model for chronic kidney disease (CKD) self-management remains unclear. This study aimed to investigate the association between stages of change for lifestyle behavior and the incidence of CKD in the general Japanese population. A retrospective cohort study was conducted among 178,780 non-CKD participants aged 40–74 years who underwent annual health check-ups for two consecutive years between 2008 and 2009. Health behavior change was determined using questionnaires based on the TTM, which consists of five stages of change (precontemplation, contemplation, preparation, action, and maintenance). The exposure of interest was the change in stages between two years. Participants were categorized into 3 groups ‘improved’, ‘unchanged’, or ‘deteriorated’. The association between the change in stages and the incidence of CKD was examined using logistic regression analysis. After one year of follow-up, 20.0% of participants developed CKD. Participants in the deteriorated group showed a significantly higher risk of CKD incidence than in the improved group. Promoting the stage of change for healthy lifestyle behaviors evaluated by the TTM was associated with a risk reduction for the incidence of CKD.
Similar content being viewed by others
Introduction
The incidence and prevalence of chronic kidney disease (CKD) continue to increase worldwide1. CKD is a progressive disease that leads to end-stage kidney disease, and is associated with an increased risk of cardiovascular disease (CVD) and mortality2. Because nearly one in eight adults have CKD in Japan3, the Japanese government assigned CKD as a national target disease for strategic medical research in 20074,5. Recent investigations of research priority settings for kidney disease identified the need to establish optimal strategies to enable patients to manage their CKD and related comorbidities, and highlighted self-management as a top priority to prevent progression of CKD6,7. Nevertheless, effective self-management strategies for patients with CKD remain unclear.
Behavioral change toward a healthy lifestyle is a key component of self-management of CKD. The transtheoretical model (TTM), which includes five stages of change (precontemplation, contemplation, preparation, action, and maintenance), is a behavioral change model that can guide health care providers to understand individuals’ motivation toward establishing and maintaining health-related behavioral changes8. The TTM has been useful in several studies on smoking cessation9,10, improving eating habits, and promoting physical activity11,12. Furthermore, a recent systematic review suggested that the TTM can be applied to prevent chronic diseases13. However, a limited number of studies have investigated the practical use of the TTM in the field of CKD.
The purpose of this study was to examine the association between change in stage of the TTM and the incidence of CKD in a Japanese nationwide population that underwent health check-ups over two consecutive years between 2008 and 2009.
Results
Participants characteristics
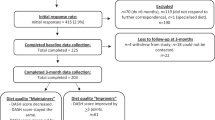
Of the 658,782 individuals who participated in health check-ups in 2008, we excluded those who had missing information on serum creatinine (n = 109,222), urinalysis (n = 14,094), and the stage of change for health behavior (n = 171,883). Then, we excluded participants with CKD at baseline (i.e., 2008) (n = 63,084). Finally, we excluded those who had not participated in health check-ups in 2009 (n = 121,719). Thus, a total of 178,780 were included in this study (Fig. 1). Compared to the 121,719 excluded participants without CKD who underwent a health check-up only in 2008, the included participants were older and less likely to be smokers (Supplemental Table 1).
Among the 178,780 included participants, 54,870 (30.7%), 55,034 (30.8%), 20,630 (11.5%), 14,059 (7.9%), and 34,187 (19.1%) were in the precontemplation, contemplation, preparation, action, and maintenance stages of change, respectively. Participants in the precontemplation and maintenance stages were older, more likely to be male, and had lower BMIs, lower waist circumferences, and lower low-density lipoprotein cholesterol levels than those in other stages. The frequency of smoking decreased in the more active stages of change, and daily drinkers showed a similar trend. The prevalence of hypertension and dyslipidemia increased between the precontemplation and action stages but decreased in the maintenance stage, while the prevalence of diabetes increased between the precontemplation and maintenance stages. After one year of follow-up, more than half of the participants in the precontemplation and maintenance stages had the same stages as those at baseline. However, participants in the contemplation, preparation, and action stages changed to different stages; in particular, participants in the preparation stage tended to change to contemplation, and participants in action stage tended to move into the maintenance stage (Table 1).
When we divided participants into three groups as their change in the stage of lifestyle behaviors between two years, 49,202 (27.5%), 84,121 (47.1%), and 45,457 (25.4%) were in the improved, unchanged, and deteriorated group, respectively. Participants in the unchanged group had lower BMI and waist circumferences, and tended to show lower proportion of comorbidities than other groups (Table 2).
Incidence of CKD and proteinuria with stage of change
Of the 178,780 participants, 35,734 (20.0%) developed CKD in 2009. Participants in the deteriorated group showed significantly higher odds for the incidence of CKD. In an unadjusted model, unchanged and deteriorated groups had graded associations with the incidence of CKD using the improved group as reference. These associations remained significant after additional adjustments in Model 1 and Model 2. The adjusted odds ratios for incidence of CKD in Model 2 were 1.02 (95% confidence interval: 0.99–1.05) and 1.07 (95% confidence interval: 1.04–1.11) (Table 3).
The incidence of proteinuria was observed in 4,986 (2.8%) participants in 2009. Similar to the incidence of CKD, graded associations were observed in the unchanged and deteriorated group when we used the improved group as a reference. The adjusted odds ratios for incidence of proteinuria in Model 2 were 1.05 (95% confidence interval: 0.98–1.12) and 1.10 (95% confidence interval: 1.01–1.18), respectively.
Discussion
This nationwide retrospective cohort study revealed that about 30% of health check-up participants in Japan without CKD were not interested in changing toward a healthy lifestyle. In addition, another 30% had an interest but had not changed their lifestyle behaviors. Although more than half of participants in the precontemplation and maintenance stages did not change or maintained their stages after one year of follow-up, those in the middle stages (i.e., contemplation, preparation, action stage) shifted to various stages, suggesting that deterioration and improvement of stages of change were common in the general population without CKD. Furthermore, participants in the deteriorated group showed significantly higher odds for the incidence of CKD and proteinuria. Our current findings support the view that a better stage of change in lifestyle behaviors might be important to prevent the development of CKD and proteinuria.
Previous observational studies have shown that several unhealthy lifestyle factors, such as obesity, weight gain after maturity, poor diet quality, smoking, and heavy alcohol consumption, were independently associated with CKD and proteinuria14,15,16,17. It has been reported that a combination of unhealthy lifestyles or not changing unhealthy lifestyle behaviors was associated with an increased risk of incidence of CKD and proteinuria18,19,20,21. Moreover, the accumulation of healthy lifestyle behaviors, especially those related to habitual moderate exercise and no bedtime snacking, was suggested to reduce the risk of CKD22. The present study, with a sufficient number of participants, provided evidence that deterioration of stage of change for a healthy lifestyle between a year was associated with the development of CKD and proteinuria.
Although health behavior changes toward a healthy lifestyle are a key component of prevention for CKD, the optimal method for promoting changes in health behaviors for CKD self-management has not been clarified. The TTM is one of the most commonly used methods in behavioral change modeling and has been used for prevention interventions for chronic diseases like diabetes, hypertension, and cancer13. With respect to CKD, a randomized controlled trial of 160 patients with early-stage CKD showed that targeted interventions matched to the TTM stages of change promoted adherence to proper diet, exercise behavior, and positive lifestyle modifications23. The findings of our study support interventions for healthy lifestyle behaviors matched to the TTM might reduce the incidence of CKD and proteinuria.
We observed that 20% of participants developed CKD in this study. Surprisingly, participants in the promoted stages (i.e., preparation, action, maintain stage) at baseline showed a higher incidence rate of CKD. We have speculated that participants with promoted stages had a higher prevalence of comorbidities, resulting in a higher risk of CKD development. Approximately 30% of participants were in the precontemplation stage, and this group had lower BMIs as well as better glycemic and lipid control than those in the contemplation, preparation, and action stages. From these observations, we speculated that many participants in the precontemplation stage were healthier with no health problems. Therefore, they were not interested in making changes to healthier lifestyles. On the other hand, participants in the maintenance stage had similar BMI and lipid control to those in the precontemplation stage. Although they had higher rates of prevalence of diabetes and dyslipidemia, they showed significantly lower risk of proteinuria relative to those in other groups. These findings further support the argument that maintaining a healthy lifestyle reduces the risk of proteinuria18.
A major strength of this study was its large-scale longitudinal nature, with participants from throughout Japan. However, several limitations of this study should be mentioned. First, the stages of change were determined based on information obtained through a self-report questionnaire, thus their responses may not be accurate. Second, we were not able to assess which lifestyle modification affected to reduce the risk of the incidence of CKD. Healthy lifestyle behaviors, such as exercising and healthy eating habits, might reduce the risk of CKD. However, we could not obtain this information in this study. Third, there may have been selection bias because the included subjects received annual health check-ups for two consecutive years, and thus these subjects may have a higher interest in their health. In fact, excluded subjects who did not have CKD and underwent health check-ups only in 2008 had a higher proportion of smokers. Therefore, included participants might be a relatively health-conscious, and the stages of change for lifestyle behaviors may be underestimated in this study population. Fourth, we could not assess the kind of antihypertensive medication, such as angiotensin receptor blockers or angiotensin-converting enzyme inhibitors, which may affect both renal function and proteinuria, due to not including in this database. Finally, unmeasured or residual confounding factors may still exist even though we adjusted for potential confounding factors.
In conclusion, the improved stages of change for healthy lifestyle behaviors were associated with reduced risk of incidence of CKD and proteinuria. Our results indicate that appropriate intervention strategies for promoting healthy lifestyle behaviors could substantially reduce the incidence of CKD. Further studies are needed to investigate whether an improved lifestyle minimizes the incidence of cardiovascular disease or mortality.
Methods
Study population
This retrospective cohort study used data from the Specific Health Check and Guidance, which was initiated by the Japanese government to promote the early diagnosis of metabolic syndrome and establish prompt intervention in 2008. Clinical details of this cohort have been described previously24,25,26. We obtained data from 15 prefectures (Hokkaido, Miyagi, Fukushima, Niigata, Tokyo, Kanagawa, Ishikawa, Nagano, Osaka, Tokushima, Fukuoka, Saga, Nagasaki, Miyazaki, and Kumamoto) whose local governments agreed to participate. We used participants without CKD, aged 40–74 who received their periodic health check–up in both 2008 and 2009.
The study was performed in accordance with the Declaration of Helsinki and Ethical Guidelines for Epidemiological Studies published by the Ministry of Education, Science and Culture and the Ministry of Health, Labour and Welfare of Japan. The requirement for informed consent was waived because the data were anonymous. The study protocol was granted ethical approval by Fukushima Medical University (IRB Approval Number #1485, #2771).
Measurement and definition
All participants answered a self-administered questionnaire that covers medical history, smoking habits, alcohol intake, exercise habits, and eating patterns. The stage of change for lifestyle behaviors was assessed by a questionnaire consisting of a 5-item algorithm, based on the TTM. The first item asked if respondents intended to change their lifestyle in the next six months; patients who responded “No” were considered in the pre-contemplation stage, while those who responded “Yes” were in the contemplation stage. Respondents who answered that they were going to change their lifestyle in the next few months were in the preparation stage. Respondents who had already started appropriate actions, such as reducing dietary fat and starting exercise within the past six months, were in the action stage. Respondents who had been taking action for more than six months were in the maintenance stage.
Participants were considered to have a history of CVD if they responded “yes” to either of the questions, “Have you ever been told that you have had a stroke or have you received treatment for stroke?” or “Have you ever been told that you have heart disease or have you received treatment for heart disease?”.
The physicians involved in this study performed a physical examination of each participant and rechecked their medical history to verify the precision of the information. Trained staff measured participants’ height, weight, waist circumference, and blood pressure. The body mass index (BMI) was calculated as the ratio of body weight (kg) to height squared (m2). Blood pressure was measured using a standard sphygmomanometer or an automated device while in a seated position after the subject had rested for 5 min. Blood and urine samples were collected after an overnight fast and assayed within 24 h.
Hypertension was defined as the use of antihypertensive drugs, a systolic blood pressure ≥ 140 mmHg and/or a diastolic blood pressure ≥ 90 mmHg, or both. Diabetes was defined in accordance with American Diabetes Association Guidelines, and was identified by a fasting plasma glucose concentration ≥ 126 mg/dL, a glycated hemoglobin (HbA1c) value ≥ 6.5%, or the use of an antidiabetic drug. The HbA1c value was estimated using the National Glycohemoglobin Standardization Program equivalent value calculated using the following equation: HbA1c (%) = HbA1c (Japan Diabetes Society) + 0.4%27. Dyslipidemia was defined as the use of antilipidemic drugs, a low-density lipoprotein cholesterol concentration ≥ 140 mg/dL, or both28. We defined CKD as estimated glomerular filtration rate (eGFR) < 60 mL/min/1.73 m2, or proteinuria on urinalysis, or both. The eGFR was calculated using the formula for Japanese29. The results of urinalysis were recorded as (−), ( ±), (1 +), (2 +), and (3 +), and the presence of proteinuria was defined as a dipstick urinalysis score ≥ (1 +).
Exposure and outcomes
The exposure of interest for this study was stage of change for lifestyle behaviors between two years. Participants were categorized into three groups ‘improved’ (changed from worse to the better stage), ‘unchanged’ (the same stage), or ‘deteriorated’ (changed from better to the worse stage). The primary outcome was the incidence of CKD, and the secondary outcome was the incidence of proteinuria.
Statistical analysis
All variables were reported as medians with interquartile ranges, or frequency (percent) as appropriate. Differences in baseline characteristics between included versus excluded participants were compared by standardized differences because of the large sample size of this study30,31. Nonparametric trend tests (Jonckheere–Terpstra trend test or a Cochran–Armitage trend test32,33,34,35) evaluated differences in baseline characteristics across the stage of change and change in stage categories. Associations between the incidence of CKD and the change in stage categories were examined by logistic regression analysis. We used hierarchical adjustment with two models as follows: (1) Model 1, which adjusted for age, sex, BMI, and serum creatinine; (2) Model 2, which included the above variables plus smoking status, drinking status, and comorbidities (hypertension, diabetes, and dyslipidemia), and history of CVD. The frequency of missing data was low (< 1% for most variables, except for hypertension [12%] and CVD [9%]), and the multiple imputation method with 20 datasets was used in all regression analyses. All analyses were conducted using STATA MP, version 15.1 (Stata Corp, College Station, TX).
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Mills, K. T. et al. A systematic analysis of worldwide population-based data on the global burden of chronic kidney disease in 2010. Kidney Int. 88, 950–957. https://doi.org/10.1038/ki.2015.230 (2015).
Go, A. S., Chertow, G. M., Fan, D., McCulloch, C. E. & Hsu, C. Y. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N. Engl. J. Med. 351, 1296–1305. https://doi.org/10.1056/NEJMoa041031 (2004).
Imai, E. et al. Prevalence of chronic kidney disease in the Japanese general population. Clin. Exp. Nephrol. 13, 621–630. https://doi.org/10.1007/s10157-009-0199-x (2009).
Iseki, K. Chronic kidney disease in Japan from early predictions to current facts. Nephron Clin. Pract. 110, c268-272. https://doi.org/10.1159/000170094 (2008).
Yamagata, K. et al. Design and methods of a strategic outcome study for chronic kidney disease: Frontier of Renal Outcome Modifications in Japan. Clin. Exp. Nephrol. 14, 144–151. https://doi.org/10.1007/s10157-009-0249-4 (2010).
Hemmelgarn, B. R. et al. Determining the research priorities for patients with chronic kidney disease not on dialysis. Nephrol. Dialysis Transplant. 32, 847–854. https://doi.org/10.1093/ndt/gfw065 (2017).
Tong, A. et al. Research priority setting in kidney disease: A systematic review. Am. J. Kidney Dis. 65, 674–683. https://doi.org/10.1053/j.ajkd.2014.11.011 (2015).
Prochaska, J. & Diclemente, C. Trans-theoretical therapy: Toward a more integrative model of change. Psychotherapy 19, 276–288. https://doi.org/10.1037/h0088437 (1982).
Koyun, A. & Eroglu, K. The effect of transtheoretical model-based individual counseling, training, and a 6-month follow-up on smoking cessation in adult women: A randomized controlled trial. Turk. J. Med. Sci. 46, 105–111. https://doi.org/10.3906/sag-1407-100 (2016).
Aveyard, P., Massey, L., Parsons, A., Manaseki, S. & Griffin, C. The effect of Transtheoretical Model based interventions on smoking cessation. Soc. Sci. Med. 1982(68), 397–403. https://doi.org/10.1016/j.socscimed.2008.10.036 (2009).
Carvalho de Menezes, M., Bedeschi, L. B., Santos, L. C. & Lopes, A. C. Interventions directed at eating habits and physical activity using the Transtheoretical Model: A systematic review. Nutr. Hosp. 33, 586. https://doi.org/10.20960/nh.586 (2016).
Hutchison, A. J., Breckon, J. D. & Johnston, L. H. Physical activity behavior change interventions based on the transtheoretical model: A systematic review. Health Educ. Behav. 36, 829–845. https://doi.org/10.1177/1090198108318491 (2009).
Hashemzadeh, M., Rahimi, A., Zare-Farashbandi, F., Alavi-Naeini, A. M. & Daei, A. Transtheoretical model of health behavioral change: A systematic review. Iran. J. Nurs. Midwifery Res. 24, 83–90. https://doi.org/10.4103/ijnmr.IJNMR_94_17 (2019).
Chang, A. et al. Lifestyle-related factors, obesity, and incident microalbuminuria: The CARDIA (Coronary Artery Risk Development in Young Adults) study. Am. J. Kidney Dis. 62, 267–275. https://doi.org/10.1053/j.ajkd.2013.02.363 (2013).
Wakasugi, M. et al. Weight gain after 20 years of age is associated with prevalence of chronic kidney disease. Clin. Exp. Nephrol. 16, 259–268. https://doi.org/10.1007/s10157-011-0565-3 (2011).
Yamagata, K. et al. Risk factors for chronic kidney disease in a community-based population: A 10-year follow-up study. Kidney Int. 71, 159–166. https://doi.org/10.1038/sj.ki.5002017 (2007).
Kurella, M., Lo, J. C. & Chertow, G. M. Metabolic syndrome and the risk for chronic kidney disease among nondiabetic adults. J. Am. Soc. Nephrol. 16, 2134–2140. https://doi.org/10.1681/asn.2005010106 (2005).
Wakasugi, M. et al. Association between overall lifestyle changes and the incidence of proteinuria: A population-based, cohort study. Intern. Med. 56, 1475–1484. https://doi.org/10.2169/internalmedicine.56.8006 (2017).
Michishita, R. et al. The association between changes in lifestyle behaviors and the incidence of chronic kidney disease (CKD) in middle-aged and older men. J. Epidemiol. 27, 389–397. https://doi.org/10.1016/j.je.2016.08.013 (2017).
Michishita, R. et al. The association between unhealthy lifestyle behaviors and the prevalence of chronic kidney disease (CKD) in middle-aged and older men. J. Epidemiol. 26, 378–385. https://doi.org/10.2188/jea.JE20150202 (2016).
Wakasugi, M., Kazama, J. J., Yamamoto, S., Kawamura, K. & Narita, I. A combination of healthy lifestyle factors is associated with a decreased incidence of chronic kidney disease: A population-based cohort study. Hypertens. Res. 36, 328–333. https://doi.org/10.1038/hr.2012.186 (2013).
Michishita, R. et al. The accumulation of healthy lifestyle behaviors prevents the incidence of chronic kidney disease (CKD) in middle-aged and older males. Environ. Health Prev. Med. 21, 129–137. https://doi.org/10.1007/s12199-016-0506-6 (2016).
Teng, H. L., Yen, M., Fetzer, S., Sung, J. M. & Hung, S. Y. Effects of targeted interventions on lifestyle modifications of chronic kidney disease patients: Randomized controlled trial. West. J. Nurs. Res. 35, 1107–1127. https://doi.org/10.1177/0193945913486202 (2013).
Iseki, K. et al. Mortality risk among screened subjects of the specific health check and guidance program in Japan 2008–2012. Clin. Exp. Nephrol. 21, 978–985. https://doi.org/10.1007/s10157-017-1392-y (2017).
Yano, Y. et al. Association of high pulse pressure with proteinuria in subjects with diabetes, prediabetes, or normal glucose tolerance in a large Japanese general population sample. Diabetes Care 35, 1310–1315. https://doi.org/10.2337/dc11-2245 (2012).
Yano, Y. et al. Association between prehypertension and chronic kidney disease in the Japanese general population. Kidney Int. 81, 293–299. https://doi.org/10.1038/ki.2011.346 (2012).
Mellitus, T. et al. The Committee of the Japan Diabetes Society on the Diagnostic Criteria of Diabetes Mellitus. Report of the Committee on the Classification and Diagnostic Criteria of Diabetes Mellitus. J. Diabetes Invest. 2010(1), 212–228. https://doi.org/10.1111/j.2040-1124.2010.00074.x (2010).
Kinoshita, M. et al. Japan Atherosclerosis Society (JAS) guidelines for prevention of atherosclerotic cardiovascular diseases 2017. J. Atheroscler. Thromb. 25, 846–984. https://doi.org/10.5551/jat.GL2017 (2018).
Matsuo, S. et al. Revised equations for estimated GFR from serum creatinine in Japan. Am. J. Kidney Dis. 53, 982–992. https://doi.org/10.1053/j.ajkd.2008.12.034 (2009).
Austin, P. C. Balance diagnostics for comparing the distribution of baseline covariates between treatment groups in propensity-score matched samples. Stat. Med. 28, 3083–3107. https://doi.org/10.1002/sim.3697 (2009).
Schacht, A., Bogaerts, K., Bluhmki, E. & Lesaffre, E. A new nonparametric approach for baseline covariate adjustment for two-group comparative studies. Biometrics 64, 1110–1116. https://doi.org/10.1111/j.1541-0420.2008.00994.x (2008).
Sasieni, P. D. From genotypes to genes: Doubling the sample size. Biometrics 53, 1253–1261 (1997).
Cochran, W. G. Some methods for strengthening the common χ2 tests. Biometrics 10, 417–451. https://doi.org/10.2307/3001616 (1954).
Armitage, P. Tests for linear trends in proportions and frequencies. Biometrics 11, 375–386. https://doi.org/10.2307/3001775 (1955).
Zheng, G. & Gastwirth, J. L. On estimation of the variance in Cochran-Armitage trend tests for genetic association using case-control studies. Stat. Med. 25, 3150–3159. https://doi.org/10.1002/sim.2250 (2006).
Acknowledgements
This study was supported by a Health and Labor Sciences Research Grant for Study on the design of the comprehensive health care system for chronic kidney disease (CKD) based on the individual risk assessment by Specific Health Checkup from the Ministry of Health, Labor and Welfare of Japan, a Grant-in-Aid for Research on Advanced Chronic Kidney Disease (REACH-J), Practical Research Project for Renal Disease from Japan Agency for Medical Research and Development, AMED and JSPS KAKENHI Grant Number JP18K11131. We would like to thank Editage (www.editage.com) for English language editing.
Author information
Authors and Affiliations
Contributions
H.K. contributed to the conception and design of the study, conducted all statistical analyses, interpreted results, and wrote this manuscript. K.A. contributed to data collection and study measurements, conducted statistical analyses, interpreted results, and edited the manuscript. K.T., K.I., T.M., K.Y., K.T., S.F., I.N., T.K., M.K., M.K., Y.S., T.W., and J.J.K. interpreted results and edited the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kimura, H., Asahi, K., Tanaka, K. et al. Health-related behavioral changes and incidence of chronic kidney disease: The Japan Specific Health Checkups (J-SHC) Study. Sci Rep 12, 16319 (2022). https://doi.org/10.1038/s41598-022-20807-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-20807-2
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.