Abstract
In vitro whole blood infection models are used for elucidating the immune response to Mycobacterium tuberculosis (Mtb). They exhibit commonalities but also differences, to the in vivo blood transcriptional response during natural human Mtb disease. Here, we present a description of concordant and discordant components of the immune response in blood, quantified through transcriptional profiling in an in vitro whole blood infection model compared to whole blood from patients with tuberculosis disease. We identified concordantly and discordantly expressed gene modules and performed in silico cell deconvolution. A high degree of concordance of gene expression between both adult and paediatric in vivo–in vitro tuberculosis infection was identified. Concordance in paediatric in vivo vs in vitro comparison is largely characterised by immune suppression, while in adults the comparison is marked by concordant immune activation, particularly that of inflammation, chemokine, and interferon signalling. Discordance between in vitro and in vivo increases over time and is driven by T-cell regulation and monocyte-related gene expression, likely due to apoptotic depletion of monocytes and increasing relative fraction of longer-lived cell types, such as T and B cells. Our approach facilitates a more informed use of the whole blood in vitro model, while also accounting for its limitations.
Similar content being viewed by others
Introduction
According to the World Health organisation (WHO) Global Tuberculosis Report 20211, there was an estimated 1.3 million deaths caused by tuberculosis (TB) in 2020. Despite being curable and preventable, TB has lingered at the top of the killer communicable disease list for many decades. Limited access to healthcare in poorer countries, an absence of diagnostic tests with sufficient sensitivity, robustness and affordability, and the lack of a universal vaccine capable of conferring immunity to both adults and children are contributing to the strain. Progress in the latter two is hindered by the elusive host immune response to TB, and the incomplete understanding of immunological mechanisms modulating an individual’s ability to fight the infection.
TB is caused by the airborne pathogen, Mycobacterium tuberculosis (Mtb). Upon inhalation Mtb is faced with the first line of defence in the form of “professional” phagocytes (macrophages, neutrophils, and dendritic cells). If successful, Mtb infects these cells and rapidly proliferates within them. Once TB infection is established it may stay dormant for years in a delicate interplay between the host immune system and the bacilli, with one quarter of the world’s population2,3 estimated to be infected with Mtb. An individual is at highest risk of developing TB disease within the first two years after infection but can remain at risk for their lifetime4. Younger children are more likely to progress to primary infection. In older children and adults, progression from latent TB infection (LTBI) to TB disease may occur as a consequence of a weakened host immune response, through factors such as co-infections, other diseases or ageing, as well as pathogen immune-escaping mechanisms. These include evading degradation within the phagolysosomes, delaying the activation of the adaptive immune response, and mutating its surface antigens to evade T-cell recognition5,6,7.
Whole blood transcriptomics can improve our understanding of the host immune response to Mtb infection and progression to disease. Blood gene expression profiling of patients with TB has highlighted Mtb specific transcriptional changes associated with cellular pathways involving interferon gamma (IFN-γ) and T cell receptor signalling and proliferation8. As the patterns of expression in the transcriptional profiles are specific to different pathogens, the last decade has focussed much attention to exploring the role of gene expression signatures in disease diagnosis and prognosis in paediatric and adult patient cohorts with pulmonary TB or extrapulmonary TB 9,10,11,12. Gene signatures identified between active TB and other disease control cohorts are currently being investigated for use as biomarkers in order to form the basis of more sensitive diagnostics tests13.
In vitro infection models provide a highly controlled way of studying host–pathogen interaction and allow for robust, reproducible, and translatable research. Infection models have played an important role in current understanding, treatment, and prevention of many infectious diseases, including TB disease14,15,16,17. In comparison to studying natural infection in patient cohorts, they can be less confounded by various factors including pathogen strain and dose, can be broader in scope, cost-efficient, less time-consuming, and can provide longitudinal measurements. However, the relevance of any given model to the human infection and disease needs to be evaluated, as there is the risk that it may not recapitulate biological processes underpinning natural infection. Understanding how the in vitro model resembles in vivo infection is essential for extrapolating the model’s experimental findings to in vivo natural infection and inform vaccine development and host-directed therapies.
Whole blood infection assays (WBA) are a reliable in vitro method used to assess human cellular immune responses to Mtb or Mtb antigens. They have been shown to better model the diverse cellular interactions that manifest during the immune response to infection compared to peripheral blood mononuclear cells (PBMCs), with higher cell viability and encompassing all immune cell types and non-cellular components of the human blood17. WBAs have also been used to evaluate Mtb-specific T cell responses, their breadth and specificity15. More recently, bulk human RNA transcriptional profiles from an in vitro Mtb infection WBA were employed to assess longitudinal host responses to Mtb, revealing a widespread immune suppression over time6. Whilst this model provided important new data in the study of the host immune response to Mtb with implications for the discovery of new vaccines and therapeutics, its similarity to natural host Mtb infection has not been previously assessed.
Recently a comparative transcriptomics approach for human and murine transcriptional responses to Mtb infection identified the drivers of concordance and discordance between the two systems14. This method evaluates the degree of similarity based on the direction of gene regulation (up or down) weighted by the magnitude of the effect size and associated significance.
Here, we aim to identify the similarities and differences at the transcriptional level of an Mtb infection WBA and natural in vivo host response in adults and children with TB disease. Identifying biological pathways that drive concordance and discordance in these datasets is of great importance to facilitate a better understanding of the in vitro WBA system, and the components of the in vivo infection that are recapitulated over time. Lastly, we employ in silico cell deconvolution prediction to elucidate the role of possible differences in immune cell populations in driving concordance and discordance between the in vivo and in vitro systems.
Methods
Data analysis was performed in R version R-4.0.318, and a script including all analytical steps is available upon request.
Data acquisition
Previously published whole blood gene expression microarray datasets were downloaded from Gene Expression Omnibus database (accession number for in vitro dataset: GSE1083636, for adult in vivo dataset: GSE3725019, for paediatric in vivo dataset: GSE3994110). The gene expression microarray datasets had been generated using Illumina HumanHT-12 v4 Expression BeadChip microarrays.
The in vitro dataset derives from a study of healthy adult donor peripheral whole blood infected with a bioluminescent strain of Mtb (H37Rv Mtb lux) using a WBA. This assay incubates infected and uninfected whole blood over a time-course allowing for analyses, including bulk gene expression microarray analysis in this instance, to be performed at different time-points. In this dataset, whole blood samples included 44 infected and 52 uninfected controls at 6 h, 24 h, 48 h, 72 h, and 96 h in the discovery and validation cohorts (n = 4 and 6, respectively).
In vivo datasets were probed for gene expression changes in adults and children diagnosed with active TB, relative to LTBI. The paediatric dataset consisted of samples from patients from South African, Kenyan, and Malawian cohorts, with 82 active TB samples and 57 LTBI samples (mean age 5.1 years, median age 3.3 years, min = 3 months, max = 15.4 years). The adult dataset consisted of a South African and Malawian cohort, with 97 active TB samples and 82 LTBI samples (mean age 34.8 years, median age 29.7 years, min = 17.8 years, max = 78.9 years). The high burden of tuberculosis in sub-Saharan Africa ensures that the results obtained in these studies, and therefore this current study, are clinically relevant. The heterogeneity of these cohorts allows for extrapolation of the data beyond the three countries.
Data preprocessing
The datasets were quantile normalised across arrays and transformed to a logarithmic scale (base 2). Batch correction was performed using ComBat function from the sva package20.The in vivo datasets were corrected for the study sites and the in vitro dataset was corrected for discovery and validation cohorts. For in vivo datasets, only the samples from Human Immunodeficiency Virus (HIV)-uninfected participants with culture-confirmed TB were included in the analysis. HIV-infected participants were excluded from the analysis as the WBA study participants were all HIV-uninfected. LTBI individuals were used as a proxy for controls. Sample GSM914578 from the adult dataset (GSE37250) was excluded due to uncertain HIV status. For the in vitro dataset, only Mtb-infected and uninfected samples were used. Principal component analysis was performed using the PCA tools package21. Other packages used for data processing included: pheatmap22, ggplot223, ggrepel24, magrittr25, RColorBrewer26, mgsub27, data.table28, biomaRt29, taRifx30, reshape31, edgeR32.
Differential gene expression analysis and pathway analysis
Limma R package33 was used for differential gene expression analysis. The linear models accounted for disease group, study site, sex, and age, in the in vivo datasets and for treatment condition (Mtb-infected and uninfected whole blood) and treatment time in the in vitro dataset. Gene mapping was performed using biomaRt29 and differentially expressed genes were visualised using the Enhanced Volcano package34. Log2 fold change (FC) threshold was set at 0.5 and − 0.5 and Benjamini–Hochberg adjusted p-value was used at a cut-off at 0.05. The transcript identifiers were mapped to Human Genome Organisation (HUGO) gene symbols which were used for annotation. Pathway analysis on the individual datasets was performed using TopGO package35, and significance threshold (adjusted p-value of < 0.01, Benjamini-Hochberg) of identified pathways was tested using Kolmogorov–Smirnov test and elim method. Identified GO terms were reduced and summarised using rrvgo package36.
Discordance- concordance analysis
For a transcript to be considered for each in vitro/in vivo contrast, it had to be significantly differentially expressed in at least one of the two comparisons. All transcripts with adjusted p-value below 0.05 were considered in the downstream analysis, regardless of their Log2FC values. A significance metric, “disco.score”, was then calculated, which accounted for both the adjusted p-values and log2 FC values associated with each transcript. This metric adjusted the effect of low-expressing genes in the downstream analysis. The metric was calculated as follows 14:
Each transcript was associated with two log2FC values as well as a disco.score. These were plotted on a disco plot as x and y values and colour intensity, respectively. Discoplots were divided into four quadrants which designated their concordance or discordance status. Tmod package37 was used to apply Gene Set Enrichment Analysis38 to each of the four quadrants. The resulting gene modules were summarised into parent terms and visualised on dotplots. Gene module count was visualised using heatmaps.
In silico cell deconvolution
Cell fraction proportions of 22 leucocyte cell types were estimated using the online tool CibersortX39. The cell types under investigation were B cells naïve, B cells memory, Plasma cells, T cells CD8, T cells CD4 naïve, T cells CD4 memory resting, T cells CD4 memory activated, T cells follicular helper, T cells regulatory (Tregs), T cells gamma delta, NK cells resting, NK cells activated, Monocytes, Macrophages M0, Macrophages M1, Macrophages M2, Dendritic cells resting, Dendritic cells activated, Mast cells resting, Mast cells activated, Eosinophils and Neutrophils. The proportions were used to calculate a FC between the control and disease groups for all cell types. Cell type pairs were found between each in vitro–in vivo comparison and analogue to a disco.score was calculated using the in vitro and in vivo log2FC and their associated p-values. These metrics were plotted on cell type discoplots.
Results
Distinct patterns of differential transcript expression across the three datasets
Differential expression analysis for the patient microarray data (Fig. 1a), was conducted at the transcript level using the limma package33. After preprocessing and visualisation using principal component analysis (Supplementary Fig. 1), transcript expression data was analysed using a linear model approach33, for the in vitro infected versus uninfected control groups and the natural infection TB disease vs LTBI groups. The uninfected control and the LTBI groups served as the references. It was previously reported that there were no observable differences in whole blood transcriptome between uninfected healthy controls and those with LTBI (unless stimulated)40.
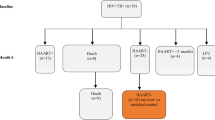
Dataset cohorts and concordance analysis design. (a) Three publicly available microarray gene expression datasets deposited in the Gene Expression Omnibus were mined for the analysis. In vitro data derives from an adult whole blood cell infection assay; ‘Ctrl’ and ‘inf’ denote control samples (uninfected) and samples infected with Mtb, respectively. RNA was extracted at five time points (6–96 h) post-infection in the infected and uninfected samples and at 0 h in the control/uninfected group. For in vivo dataset, only Human Immunodeficiency Virus (HIV)-negative and culture confirmed TB patients were included in the analysis. LTBI individuals were used as a proxy for healthy control patients. Sample size is shown for each treatment and cohort group. (b) Workflow used to identify concordance and discordance between in vitro and in vivo datasets. Corresponding gene transcripts were mapped to each other to form transcript unions, composed of transcripts significant in vivo mapped to their non-significant counterparts in vitro denoted by X; transcripts significant in both datasets mapped to each other denoted by Y; transcripts significant in vitro mapped their non-significant counterpart in vivo denoted by Z. A transcript pair is said to be concordant if both of its associated log2FC values have the same sign. This can be further segmented into concordantly upregulated transcript pairs (both log2FC values are positive) and concordantly downregulated transcript pairs (both log2FC values are negative). A transcript pair is said to be discordant if its associated log2FC values have differing signs, also resulting in two final groups (positive log2FC in vitro & negative log2FC in vivo; and negative log2FC in vitro & positive log2FC in vivo). Disco.score was calculated for each corresponding transcript pair, and its value was proportional to the magnitude of concordance or discordance of said transcript pairs. Concordant transcript pairs were sorted by decreasing disco.score while discordant transcript pairs were sorted by increasing disco.score. Gene Set Enrichment Analysis (GSEA) was performed on each of the four groups of transcript pairs.
Firstly, for the in vitro dataset we conducted differential expression analysis for the 6 h, 24 h, 48 h, 72 h and 96 h Mtb infected timepoints against their respective uninfected control (Supplementary Fig. 2). There were 45 significantly differentially expressed (SDE) genes at 6 h, 1639 at 24 h, 1409 at 48 h, 1210 at 72 h and 1623 at 96 h, with adjusted p-value < 0.5 and log2FC threshold of 0.5. For the human natural infection datasets, we compared TB disease vs LTBI for the adult and paediatric sets separately. 2268 genes were found SDE in the paediatric comparison, and 1950 were found SDE in the adult TB comparison (Supplementary Fig. 2a). Downregulation dominated the in vitro response at 24 h-post infection, with 65.2% (1068/1639) of all SDE transcripts under-expressed (Supplementary Fig. 2b). Reduced expression remained consistent across the later time points, with 63.6% (896/1409) of SDE transcripts under-expressed at 48 h (Supplementary Fig. 2c) and 69.2% (837/1210) of SDE transcripts under-expressed at 72 h (Supplementary Fig. 2d). In contrast, most transcripts in in vivo TB disease were over-expressed, with 66.0% (1499/2268) SDE transcripts upregulated in paediatric TB (Supplementary Fig. 2e) and 62.6% (1221/1950) of SDE transcripts upregulated in adult TB (Supplementary Fig. 2f) (exact binomial test p-value < 2.2 × 10−16).
Genes that were significantly upregulated and downregulated in infected patients compared to the controls were subjected to Gene Ontology (GO) pathway analysis using the topGO package35. Pathways enriched in downregulated genes in vitro (24–96 h) included immune activation (elim KS = 0.0052), antigen processing (elim KS = 0.0132) and defence response to bacteria (elim KS = 7.2 × 10−05), whereas pathways enriched in over-expressed genes were implicated in lymphocyte chemotaxis (elim KS = 4.2 × 10−07), neutrophil chemotaxis (elim KS = 2.3 × 10−06), and cellular response to IL-1 (elim KS = 4.0 × 10−05).
Pathways enriched in genes downregulated in natural infection included negative regulation of cytokine production (elim KS = 0.0051), cytoskeleton organisation (elim KS = 0.00606), Fc receptor signalling pathway (elim KS = 0.00580). Pathways enriched in genes upregulated in natural infection included defence response to bacteria (elim KS = 0.00022), innate immune response in mucosa (elim KS = 3.9 × 10−5) and positive regulation of IL-1B (elim KS = 0.00091).
Concordance and discordance analysis and accounting for the direction of gene regulation in the in vitro–in vivo comparisons
The disco.score was calculated for each pair of transcripts using the log2FC and p-values as described in the Methods (Fig. 1b). The discordance-concordance plots were segmented into four quadrants in each in vitro–in vivo comparison; those which were concordantly upregulated (quadrant I), concordantly downregulated (quadrant III), discordantly regulated such that gene pairs were either upregulated in vivo while downregulated in vitro (quadrant II) or downregulated in vivo while upregulated in vitro (quadrant IV) (Fig. 2). In this way, each in vitro time point was compared with both paediatric and adult patients (data for in vivo–in vitro 96 h comparison in Supplementary Fig. 3). As the number of transcripts at 6 h post-in vitro infection vs control was small in comparison to the other time point comparisons, fewer transcripts were identified as either concordant or discordant. The top concordantly up-regulated genes in the adult in vivo vs in vitro comparison included CARD17, involved in the negative regulation of IL-1β production, SOD2, which has an antiapoptotic role against inflammatory cytokines41 and CASP5, which, conversely, is implicated in apoptosis. Some of the downregulated genes included CCR3, a chemokine receptor highly expressed in eosinophils and basophils and DCANP1, specifically expressed in dendritic cells. Discordantly regulated genes included VpreB3, which is thought to play a role in B cell maturation, SLAMF1, involved in dendritic cell development, and CCR7, a gene coding for a receptor which activates B and T lymphocytes.
Disco plots. (a–d) Evaluation of concordance of gene expression changes in adult TB and in vitro time series (a = 6 h, b = 24 h, c = 49 h and d = 96 h). Disco.score shown by colour intensity, ranging from blue (low concordance) to dark red (high concordance). Transcripts found in each disco plot are segmented into four quadrants (Q) as follows; QI—concordantly upregulated, QII—discordant, upregulated in vivo & downregulated in vitro, QIII—concordantly downregulated and QIV—discordant, downregulated in vivo and upregulated in vitro. Numbers of identified transcripts shown in each quadrant. (e–h) Evaluation of concordance of gene expression changes in paediatric TB and in vitro time series (e = 6 h, f = 24 h, g = 48 h, h = 72 h).
For each in vitro–in vivo comparison, Gene Set Enrichment Analysis (GSEA) was performed on genes from each of the quadrants and counts of the identified gene sets (gene modules) were plotted as a heatmap (Fig. 3). Heatmaps revealed that there is a trend towards concordance in earlier time points (0–48 h), while discordance is more dominant from 48 h onwards. Paediatric in vivo–in vitro showed more concordantly downregulated modules (Fig. 3c), while adult in vivo–in vitro has a distinctly different pattern which showed many more concordantly upregulated modules (Fig. 3a). The largest number of modules were identified as discordantly regulated, upregulated in vitro and downregulated in vivo.
Counts of concordantly and discordantly regulated gene modules. Gene Set Enrichment Analysis (GSEA) was performed on transcripts from the four disco plot quadrants for each in vitro–in vivo comparison. Identified modules were counted and plotted as heatmaps in the same arrangement as quadrants of a discoplot from which the transcripts were taken. Colour intensity ranging from yellow to red for concordant modules, and yellow to blue for discordant modules, denotes increasing number of modules identified. (a) Counts of discordant modules, upregulated in vivo, and downregulated in vitro. (b) Counts of concordantly upregulated modules. (c) Counts of concordantly downregulated modules. (d) Counts of discordant modules, downregulated in vivo, and upregulated in vitro.
Concordance and discordance analysis of the adult in vivo–in vitro comparison
To explore the identified gene modules in a more comprehensive way, gene modules describing similar biological pathways were summarised, and thereafter visualised using dot plots (Figs. 4, 5). Remarkably, time signatures of concordantly and discordantly regulated modules can be clearly distinguished in these plots.
Dotplot of gene modules identified in adult in vivo–in vitro comparison. Gene Set Enrichment Analysis (GSEA) was performed on transcripts from the disco plot quadrants. Similar modules were summarised and plotted such that p-value of module enrichment is illustrated by the intensity of the colour (yellow–blue gradient denotes increasing significance of discordant pathways, yellow–red gradient denotes increasing significance of concordant pathways) and the effect size by the size of the dot. (a) Gene modules upregulated in vivo and downregulated in vitro. (b) Concordantly upregulated gene modules. (c) Concordantly downregulated gene modules. (d) Gene modules downregulated in vivo and upregulated in vitro.
Dotplot of gene modules identified in paediatric in vivo–in vitro comparison. Gene Set Enrichment Analysis (GSEA) was performed on transcripts from the disco plot quadrants. Similar modules were summarised and plotted such that p-value of module enrichment is illustrated by the intensity of the colour and the effect size by the size of the dot. (a) Gene modules upregulated in vivo and downregulated in vitro. (b) Concordantly upregulated gene modules. (c) Concordantly downregulated gene modules. (d) Gene modules downregulated in vivo and upregulated in vitro.
In agreement with our previous findings, concordance of the adult in vivo–in vitro comparison was primarily seen in upregulated modules (Fig. 4b, representing quadrant I), whereas very few concordantly downregulated modules were identified (Fig. 4c, representing quadrant III). Concordant upregulation (Fig. 4b) was driven by inflammation exhibiting the highest effect size (average cES = 12.8, average p val = 4.36 × 10−22) across all timepoints, as well as pathways relating to monocyte enrichment (average cES = 9.34, average p val = 4.57 × 10−11), chemokines (average cES = 6.67, average p val = 4.7 × 10−7), and cytokines (type 1 interferon response) (average cES = 2.98, average p val = 4.6 × 10−4). T cell and B cell modules were strongly concordantly downregulated in the comparisons with 6–24 h in vitro (cES = 14.5, p val = 6.1 × 10−13 and cES = 17.95, p val = 5.09 × 10−18 respectively) after which the abundance and significance of these pathways rapidly decreased. Concordant downregulation of monocyte-related pathways was also discernible, however the number of genes the modules were enriched in is sevenfold lower than T cell modules. Modules that are upregulated in vitro and downregulated in vivo (Fig. 4d, representing quadrant IV) show most apparent discordance in T cell and B cell modules, which increases over time (T cells at 6 h: cES = 17.54, p val = 8.27 × 10−25, at 96 h: cES = 46.87, p val = 1.67 × 10−94. B cells at 6 h: cES = 2.13, p val = 0.025, at 96 h: cES = 23.5, p val = 8.56 × 10−39). NK cells also have a slight time signature, with the strongest signal at 24–72 h (cES = 17, p val = 2.3 × 10−27). Other modules regulated in this way also relate to lymphocytes, their activation and function.
Gene modules upregulated in vivo and downregulated in vitro (Fig. 4a, representing quadrant II) include monocyte and inflammation modules can also be found (average cES = 9.09, p val = 2.87 × 10−44 and average cES = 11.08, p val = 6.38 × 10−26 respectively), alongside dendritic cell and antigen presentation modules.
Concordance analysis of the paediatric in vivo–in vitro comparison
GSEA was performed on genes from the individual quadrants of disco plot comparisons of paediatric in vivo–in vitro time series, as before. Concordantly upregulated modules (Fig. 5b, representing quadrant I) are dominated by a chemokine/cytokine module (average cES = 11.2, average p-value = 7.43 × 10−8), and secondary to it, inflammation, and monocyte related modules (cES = 5.09, p val = 3.01 × 10−8 and cES = 4.07, 1.28 × 10−3 respectively). Many more concordantly downregulated modules (Fig. 5c, representing quadrant III) were identified, compared to the adult in vivo–in vitro comparison. These include T cell activation, greatest at 6 h (cES = 25.4, p val = 3.1 × 10−16) and decreasing in both effect size and significance at 48 h (cES = 11.5, p val = 1.62 × 10−5). Modules relating to monocytes and antigen presentation also drove the concordance of downregulation in this comparison.
As in the adult in vivo–in vitro comparison, monocytes were also identified in quadrant II (Fig. 5a, representing quadrant II). Along with inflammation and dendritic cells, these modules have increasing signal towards later time points.
Modules that are upregulated in vitro and downregulated in vivo (Fig. 5d, representing quadrant IV) are primarily related to T cell signalling as in adult in vivo–in vitro comparison; however, B cell modules were identified but less pronounced (cES = 5.24, p val = 0.003). Other modules here included NK cells and those related to regulating chemokines and cytokines.
Comparison of in silico cell deconvolution corroborates the findings of the concordance analysis
Transcript expression data was also used for in silico cell deconvolution (CibersortX39) and the output of relative cell fractions of 22 immune cell types was split into adaptive and innate cells (Fig. 6). In adaptive cell fractions no significant changes over time were identified in vitro, with the exception of CD4 memory activated fraction, which increases from 24 h onwards post-infection compared to control (Fig. 6a). The differences between adult and paediatric TB disease compared to LTBI controls (in vivo data) are largely consistent across adaptive cells (Fig. 6b,c), apart from several T cell subpopulations. Namely, these are CD4 memory activated, CD4 memory resting and T cells regulatory fractions which are significantly elevated, reduced and reduced, respectively, in the adult TB and control comparison, but do not show any significant differences in the paediatric TB versus LTBI groups.
Cell deconvolution using CibersortX—adaptive cells. Changes in adaptive cell fractions (a) in vitro over time in the Mtb-infected and control group and in vivo in (b) paediatric TB patients and (c) adult TB patients and corresponding LTBI control patient groups. Error bars depict standard deviation from the mean, *p < 0.05, **p < 0.005, ***p < 0.001.
Several innate cell fractions (Fig. 7), including monocytes, neutrophils and activated mast cells, were reduced in vitro post Mtb-infection, while activated dendritic cells had a higher relative fraction post Mtb-infection (Fig. 7a). Both adult and paediatric patients with TB disease manifested with elevated monocyte, neutrophil and M0 macrophage fractions and reduced NK cell fractions compared to their respective LTBI controls (Fig. 7b,c).
Cell deconvolution using CibersortX—innate cells. Changes in innate cell fractions (a) in vitro over time in the Mtb-infected and control group and in vivo in (b) paediatric TB patients and (c) adult TB patients and corresponding LTBI control patient groups. Error bars depict standard deviation from the mean, *p < 0.05, **p < 0.005, ***p < 0.001.
Thus, agreement between in vitro–in vivo is most noticeable in innate cell fractions (Fig. 7), such as dendritic cell and non-activated (M0) macrophage fractions which are elevated from 24 and 48 h, respectively, and NK cells which are reduced from 24 h. In vitro–in vivo disagree in monocyte and neutrophil fractions which are increased in vivo, and diminished in vitro, and T cell fractions where CD8 and CD4 naïve increase in vivo while not showing any change in vitro (Fig. 6).
Discussion
Reproducible and clinically translatable TB research is crucial to address the urgent need for vaccines, novel therapeutics, and more sensitive diagnostic tests. In vitro studies combining WBAs with gene expression readouts have elucidated important facets of the interplay between Mtb and the host, particularly regarding immune cell interactions 6,16,42. WBAs contain most immune cell types and thus account for a much broader and a more physiological representation of the immune system than other in vitro cell type specific infection models. Interpreting and extrapolating in vitro observations from the WBA requires knowledge of the similarities and differences between the WBA and specific elements of natural TB infection and disease. For example, differences in cell populations resulting from longer culture and infection treatment times may drive divergence between in vitro and in vivo systems, if more short-lived cell types, such as neutrophils43, have undergone apoptosis and cannot participate in the full response to the pathogen. No studies have been performed to elucidate how representative the Mtb infection WBA transcriptomic profiles are in comparison to natural human infection and disease.
In this study we characterised the concordance and discordance of gene expression profiles between an in vitro Mtb infection assay which uses peripheral blood from healthy adult donors and in vivo peripheral circulating blood samples from paediatric and adult TB patients. We compared the transcriptomic profiles and cell fractions between WBA and the patient data and identified conserved and divergent host immune gene expression. To better understand how the WBA relates to in vivo TB disease, and particularly how different experimental time points of the assay relate to specific elements of the host immune response, we implemented a concordance analysis of gene regulation accompanied by GSEA and in silico cell deconvolution39. The in vitro study compared TB infection vs uninfected controls, whereas the in vivo studies compared TB disease vs LTBI. Thus, the patient datasets have captured systemic disease aspects: lung pathology, tissue necrosis, unwellness in addition to the Mtb immune escape mechanisms which enabled TB disease to develop. This study has enabled us to assess which elements of the in vitro model recapitulate aspects of the in vivo anti-mycobacterial immune response.
We observed that concordance between in vivo–in vitro datasets is primarily associated with immune activation for adult TB disease and immune suppression for paediatric TB disease. Concordance in the adult in vivo versus WBA comparison was driven by upregulated modules including those involved in inflammation, pro-inflammatory signalling, neutrophil, and monocyte enrichment, which are important for the recognition of Mtb infection and the initiation of a large-scale immune response to the pathogen7. Fewer concordantly downregulated modules were observed, including those involved in T and B cell function, with the concordance identified at earlier time points (0 h and 24 h). We also identified the concordant and discordant gene modules in the WBA dataset that were consistent in both adult and paediatric in vivo TB disease datasets. We found that both comparisons had common concordance in modules related to inflammatory pathways expected during an infection with mycobacteria, such as cytokine (particularly IFN signatures) and chemokine signalling, and modules relating to T cell suppression and monocytes, most notably in earlier time points (6–24 h). Common discordant modules dominated both comparisons with the later time points of the WBA (72–96 h), especially those relating T cell signalling, dendritic cell activation and monocytes.
Apoptotic depletion of macrophages and dendritic cells in the in vitro model without the means to replenish them will lead to a substantial divergence between the WBA and in vivo studies. Therefore, the higher abundance of T cell signatures at later time points (48–96 h) in vitro relative to in vivo may be a result of a higher fraction in the bulk gene expression analysis following the loss of other cell types. Delay of the onset of the adaptive immune response in vivo as well as T cell exhaustion, manifested by reduced T-cell activation, may also explain the large contrast between the in vivo and in vitro systems. As mentioned previously, we also observed concordance in downregulated T cell modules in both paediatric and adult in vivo-in vitro comparisons at 6–24 h. It should be noted that, while the in silico cell deconvolution analysis has been shown to accurately predict the proportion of CD4 and CD8 T cell fractions in previous studies, estimations of other T cell fractions may be less accurate based on previous validation studies using this package44 and a more specific analysis of T cell fractions (e.g. Th1, Th2, and Th17 cells) may provide further insights towards understanding the concordance and discordance results observed here. Furthermore, the differences between paediatric and adult T cell populations and T cell priming are an important consideration when analysing these data. However, the prediction of the certain T cell populations which are known to differ between these groups (for example γδ T cells are highly relevant for paediatric TB responses45) may not be possible with many of the current in silico deconvolution tools.
The abundance of discordant modules increased at later time points, driven particularly by gene modules related to inflammation, T cells, monocytes, and dendritic cells. In both adult and paediatric in vivo vs in vitro comparisons, discordant modules based on genes up-regulated in vivo and down-regulated in vitro were associated with enrichment in monocytes, inflammation, and antigen presentation. On the other hand, modules based on genes down-regulated in vivo and up-regulated in vitro are primarily involved in T cell and NK cell regulation. These findings are largely supported by in silico cell deconvolution which revealed that the monocyte cell fraction significantly decreases in the infected group after 24 h in vitro, while T cell fractions stay constant (CD8) throughout the assay timeline or indeed increase in abundance (CD4) towards the later time points. In contrast, in the in vivo cell deconvolution results, the monocytes fraction is significantly elevated in both adult and paediatric TB patients. M0 macrophages, which derive from monocytes, were also found to be significantly elevated in both TB patient datasets. In contrast, CD8 and CD4 naïve T cells are significantly decreased in TB patients from both datasets. However, in the adult patient cohort, activated CD4 memory T cells are increased, while resting CD4 memory T cells were decreased.
When comparing the WBA versus the paediatric in vivo dataset, we identified gene modules involved in host immune signalling, such as those related to chemokines, cytokines, and other markers of inflammation, as concordantly upregulated, while modules associated with monocytes and dendritic cells, which harbour Mtb proliferation, were concordantly downregulated along with antigen presentation and immune activation signatures. This observed downregulation may reflect Mtb suppression of the host immune response, which has been previously described in paediatric TB disease46,47. By contrast, in the adult comparison, antigen presentation was discordantly upregulated in the adult in vivo patient dataset and downregulated in the WBA dataset. Differences in the whole blood transcriptome in children and adults with TB disease have been previously reported (e.g., inhibition of neutrophil degranulation and difficulty in T cell priming47,48). It is worth noting that the WBA utilises whole blood from naïve adult donors, and age difference between the WBA and the in vivo paediatric dataset could be driving some of the differences reported. In addition, the sample size for the groups in the paediatric in vivo dataset is lower compared to that in the adult in vivo dataset, which may be the cause of the slightly lower p-value significance levels of the differentially expressed transcripts in the paediatric cohort.
The study has certain limitations. As the WBA uses blood from healthy adult donors, some of the differences observed in the comparisons encompassing the paediatric dataset may be attributed to age. Conducting a WBA-based study that uses blood from children may be more relevant in elucidating paediatric-specific host response elements49. Secondly, although previous studies have reported the lack of differences between the transcriptomic profiles in unstimulated blood of Interferon-Gamma Release Assay (IGRA) positive individuals and healthy controls, discordance reported between the in vitro and in vivo datasets could be attributed to differences in the baseline comparator group (LTBI in vivo vs uninfected blood in vitro). WBA models that include donors with TB disease or LTBI and studied in relation to healthy donors, could provide us with further insights into the host response to Mtb. The analysis needs to be coupled with other readouts, such as experimental flow cytometry, in order to disentangle the causality of the relationships between the gene modules. Lastly, our analysis was conducted on bulk microarray gene expression data, which profiles transcripts included on the array, rather than the whole transcriptome. Future studies can be expanded to RNA-sequencing data and especially single-cell RNA-sequencing, to further examine the complexity of host immune response to Mtb, including multiple single cellular populations within a system.
In summary, we present an adaptation of concordance analysis for the comparison of host immune response in vivo and in vitro. Our findings suggest that earlier time points (24–48 h) of the whole blood assay are more concordant with patient disease, reflected by both the gene expression and in silico cell deconvolution at these time points. There are specific similarities of the assay with paediatric and adult in vivo disease, such as upregulation of inflammatory signatures, particularly relating to IFN signalling, providing a reference to tailor the assay to paediatric and adult TB disease in future studies. Our study also provides valuable information about immune cell components driving discordance between the datasets, which manifest especially at later time points (72–96 h). Understanding the limitations of in vitro assays is vital for their continued use and consequently for conducting more robust and translatable research. Identifying the magnitude and source of concordance and discordance of bulk RNA expression profiling in in vitro WBA compared to in vivo natural infection can help better understand and tailor the in vitro models to explore relevant biological questions of interest.
Data availability
Raw data is available at Gene Expression Omnibus database https://www.ncbi.nlm.nih.gov/geo/. Accession numbers for the datasets are as follows; in vitro dataset: GSE108363, adult in vivo dataset: GSE37250, paediatric in vivo dataset: GSE39941. Scripts are available on request.
References
GLOBAL TUBERCULOSIS REPORT 2021. (2021).
Cruz-Knight, W. & Blake-Gumbs, L. Tuberculosis: An overview. Primary Care Clin. Office Practice 40, 743–756 (2013).
Houben, R. M. G. J. & Dodd, P. J. The global burden of latent tuberculosis infection: A re-estimation using mathematical modelling. PLoS Med. 13, (2016).
Flynn, J. L. & Chan, J. Tuberculosis: Latency and reactivation. Infect. Immun. 69, 4195–4201 (2001).
Fogel, N. Tuberculosis: A disease without boundaries. Tuberculosis 95, 527–531 (2015).
von Both, U. et al. Mycobacterium tuberculosis exploits a molecular off switch of the immune system for intracellular survival. Sci. Rep. 8(1), 661 (2018).
de Martino, M., Lodi, L., Galli, L. & Chiappini, E. Immune response to mycobacterium tuberculosis: A narrative review. Front. Pediatr. 7, 350 (2019).
Gliddon, H. D. et al. Identification of reduced host transcriptomic signatures for tuberculosis disease and digital PCR-based validation and quantification. Front. Immunol. 12, 1664–3224 (2021).
Gliddon, H. D., Herberg, J. A., Levin, M. & Kaforou, M. Genome-wide host RNA signatures of infectious diseases: Discovery and clinical translation. Immunology 153, 171–178 (2017).
Anderson, S. T. et al. Diagnosis of childhood tuberculosis and host RNA expression in Africa. N. Engl. J. Med. 370, 1712–1723 (2014).
Hoang, L. T. et al. Transcriptomic signatures for diagnosing tuberculosis in clinical practice: A prospective, multicentre cohort study. Lancet Infect. Dis. 21, 366–375 (2021).
Mulenga, H. et al. Performance of host blood transcriptomic signatures for diagnosing and predicting progression to tuberculosis disease in HIV-negative adults and adolescents: A systematic review protocol. BMJ Open. 9, (2019).
Sweeney, T. E., Braviak, L., Tato, C. M. & Khatri, P. Genome-wide expression for diagnosis of pulmonary tuberculosis: A multicohort analysis. Lancet Respir. Med. 4, 213–224 (2016).
Domaszewska, T. et al. Concordant and discordant gene expression patterns in mouse strains identify best-fit animal model for human tuberculosis. Sci. Rep. 7, (2017).
Whatney, W. E. et al. A high throughput whole blood assay for analysis of multiple antigen-specific T cell responses in human mycobacterium tuberculosis infection. J. Immunol. 200, 3008–3019 (2018).
Newton, S., Martineau, A. & Kampmann, B. A functional whole blood assay to measure viability of mycobacteria, using reporter-gene tagged BCG or M.Tb (BCG lux/M.Tb lux). J. Visual. Exp. https://doi.org/10.3791/3332 (2011).
Silva, D., Ponte, C. G. G., Hacker, M. A. & Antas, P. R. Z. A whole blood assay as a simple, broad assessment of cytokines and chemokines to evaluate human immune responses to Mycobacterium tuberculosis antigens. Acta Trop. 127, 75–81 (2013).
R Core Team. R: A Language and Environment for Statistical Computing. (2020).
Kaforou, M. et al. Detection of tuberculosis in HIV-infected and -uninfected african adults using whole blood RNA expression signatures: A case-control study. PLoS Med. 10, (2013).
Leek, J. T. et al. sva: Surrogate Variable Analysis. (2020).
Blighe, K. & Lun, A. PCAtools: PCAtools: Everything Principal Components Analysis. (2020).
Kolde, R. pheatmap: Pretty Heatmaps. (2019).
Wickham, H. ggplot2: Elegant Graphics for Data Analysis (Springer-Verlag, 2016).
Slowikowski, K. ggrepel: Automatically Position Non-Overlapping Text Labels with ‘ggplot2’. (2021).
Bache, S. M. & Wickham, H. magrittr: A Forward-Pipe Operator for R. (2020).
Neuwirth, E. RColorBrewer: ColorBrewer Palettes. (2014).
Ewing, M. mgsub: Safe, Multiple, Simultaneous String Substitution. (2020).
Dowle, M. & Srinivasan, A. data.table: Extension of `data.frame`. Preprint at (2021).
Durinck, S. et al. BioMart and bioconductor: A powerful link between biological databases and microarray data analysis. Bioinformatics 21, 3439–3440 (2005).
Friedman, A. B. taRifx: Collection of Utility and Convenience Functions. (2020).
Wickham, H. Reshaping data with the reshape package. J. Stat. Softw. 21, (2007).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: A bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Ritchie, M. E. et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43, e47 (2015).
Blighe, K., Rana, S. & Lewis, M. EnhancedVolcano: Publication-ready volcano plots with enhanced colouring and labeling. (2020).
Alexa, A. & Rahnenfuhrer, J. topGO: Enrichment Analysis for Gene Ontology. (2020).
Sayols, S. rrvgo: A Bioconductor package to reduce and visualize Gene Ontology terms. (2020).
Weiner, J. tmod: Feature Set Enrichment Analysis for Metabolomics and Transcriptomics. (2020).
Subramanian, A. et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. 102, 15545–15550 (2005).
Newman, A. M. et al. Determining cell type abundance and expression from bulk tissues with digital cytometry. Nat. Biotechnol. 37, (2019).
Broderick, C., Cliff, J. M., Lee, J. S., Kaforou, M. & Moore, D. A. Host transcriptional response to TB preventive therapy differentiates two sub-groups of IGRA-positive individuals. Tuberculosis 127, (2021).
Montano, M. A. E. et al. Inflammatory cytokines in vitro production are associated with Ala16Val superoxide dismutase gene polymorphism of peripheral blood mononuclear cells. Cytokine 60, 30–33 (2012).
Kampmann, B. et al. Novel human in vitro system for evaluating antimycobacterial vaccines. Infect. Immun. 72, 6401–6407 (2004).
Tak, T., Tesselaar, K., Pillay, J., Borghans, J. A. M. & Koenderman, L. What’s your age again? Determination of human neutrophil half-lives revisited. J. Leukoc. Biol. 94, 595–601 (2013).
Miao, Y. et al. ImmuCellAI: A unique method for comprehensive T-cell subsets abundance prediction and its application in cancer immunotherapy. Adv. Sci. 7, 1902880 (2020).
Whittaker, E., Nicol, M., Zar, H. J. & Kampmann, B. Regulatory T cells and pro-inflammatory responses predominate in children with tuberculosis. Front. Immunol. 8, (2017).
Hemingway, C. et al. Childhood tuberculosis is associated with decreased abundance of T cell gene transcripts and impaired T cell function. PLoS One 12, (2017).
Bah, S. Y., Forster, T., Dickinson, P., Kampmann, B. & Ghazal, P. Meta-analysis identification of highly robust and differential immune-metabolic signatures of systemic host response to acute and latent tuberculosis in children and adults. Front. Genet. 9, (2018).
Basu Roy, R., Whittaker, E. & Kampmann, B. Current understanding of the immune response to tuberculosis in children. Curr. Opin. Infect. Dis. 25, 250–257 (2012).
Roy, R. B. et al. An auto-luminescent fluorescent BCG whole blood assay to enable evaluation of paediatric mycobacterial responses using minimal blood volumes. Front. Pediatr. 7, (2019).
Acknowledgements
We would like to thank the authors of the manuscripts for making the datasets used in this study publicly available. A.C. acknowledges support from the Medical Research Council (1816898). C.B. acknowledges support from the NIHR Imperial College BRC (Imperial 4i fellowship-RDA02). M.K. acknowledges support from The Wellcome Trust and the Medical Research Foundation Grants (206508/Z/17/Z and MRF-160-0008-ELP-KAFO-C0801). P.B., A.C., C.B., S.M.N, M.L. and M.K. acknowledge support from the NIHR Imperial College BRC.
Author information
Authors and Affiliations
Contributions
A.C. and M.K.: conceptualization, P.B., A.C. and M.K.: methodology, P.B.: data curation, P.B., A.C. and M.K.: writing—original draft preparation. P.B., A.C. and M.K: visualization, A.C., S.M.N., M.L., M.K, supervision, All authors: writing, reviewing and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bachanová, P., Cheyne, A., Broderick, C. et al. Comparative transcriptomic analysis of whole blood mycobacterial growth assays and tuberculosis patients’ blood RNA profiles. Sci Rep 12, 17684 (2022). https://doi.org/10.1038/s41598-022-20409-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-20409-y
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.