Abstract
Serology provides tools for epidemiologic studies, and may have a role in vaccine prioritization and selection. Automated serologic testing of saliva, especially specimens that are self-collected at home and sent to a laboratory via the mail without refrigeration, could be a highly-scalable strategy for population-wide testing. In this prospective study, non-vaccinated patients were recruited after PCR testing to self-collect saliva and return their specimens via mail. Longitudinal specimens were analyzed in order to monitor seroconversion in the weeks after a diagnostic PCR test for SARS-CoV-2. Diverse users self-collected saliva and returned specimens via mail in compliance with shipping regulations. At our pre-established threshold (0.963 AU/mL), salivary IgG reactivity to full-length spike protein achieved 95.8% sensitivity and 92.4% specificity at 2–4 weeks after diagnostic testing, which is comparable to the typical sensitivity and specificity achieved for serum testing. Reactivity to N antigen also was detected with 92.6% sensitivity and 90.7% specificity at 4–8 weeks after diagnostic testing. Moreover, serologic testing for endemic coronaviruses performed in multiplex with SARS-CoV-2 antigens has the potential to identify samples that may require retesting due to effects of pre-analytical factors. The easy-to-use saliva collection kit, coupled with thresholds for positivity and methods of flagging samples for retest, provides a framework for large-scale serosurveillance of SARS-CoV-2.
Similar content being viewed by others
Introduction
Serology assays measuring the development of host antibodies against SARS-CoV-2 antigens are important tools in the public health response to COVID-19. At the population level, serology can be used for disease surveillance and the evaluation of public health countermeasures to decrease transmission rates, and is a critical component of clinical studies to qualify new vaccines1,2,3,4. Serosurveillance can be invaluable for determining the case fatality rate, tracking incidence and prevalence, and investigating medical sequelae following recovery5,6. Serology has also been used in population studies to evaluate approaches to vaccine prioritization7 and dosing8 and to assess the durability of antibody production as a correlate of immunity after vaccination9 or natural infection10. At the individual level, there is some debate about the utility of serology assays for diagnosing acute COVID-19 infection11; however, the assays provide a tool for diagnosing post-acute or chronic COVID-19 symptoms in individuals who may have had asymptomatic or undiagnosed acute infections12. Serology assays are also likely to become increasingly important, as the correlation of antibody levels and protection from infection becomes better defined, for identifying individuals in need of vaccine boosters1,3.
The use of serology assays in some applications, especially for serosurveillance, is limited by the use of serum as the most common sample matrix and the need to collect blood from the target population. To take advantage of blood that is being collected for other purposes, many serosurveillance studies have relied on testing of blood at dialysis or blood donation centers13,14,15,16,17. Expanding serosurveillance to a diverse population presents several logistical challenges to obtain, process, and transfer serum. Self-collected specimens reduce some of these logistical challenges18,19,20,21. Saliva is especially easy to self-collect without pain or the use of needles or lancets. The bulk of salivary IgG is thought to originate from blood due to transudation or bleeding from the gingival tissue22,23,24. The potential of saliva as a sample for SARS-CoV-2 serosurveillance is supported by the strong correlation that has been observed in the antibody response in serum and saliva during acute infection and recovery25,26,27,28,29. Although the FDA has cleared serology tests for HIV30,31, saliva-based serology tests for SARS-CoV-2 are not yet commercially available.
Considerations in developing a saliva-based serology test include ease of use, regulatory requirements, and demands of downstream processing. A prior study found that patients can confidently and correctly self-collect specimens at home19. Other prior studies29,32 supervised saliva collection in research settings using devices that have not been FDA approved and/or need secondary shipping containers to meet UN3373 category B requirements. Numerous methods and devices have been designed to collect oral fluids33,34. Some use swabs or sponges to collect saliva from under the tongue, or the fluid at the interface of teeth and gums (gingival crevicular fluid). Swab-based methods are especially convenient for young children and people who find handling or viewing liquid saliva unpleasant. Extraction of saliva from swabs, however, requires additional processing steps. A FDA registered device from Salimetrics is commercially available that enables a clean and simple protocol to collect liquid saliva into a tube by drooling through a fit-for-purpose straw34.
Previously, we developed and pilot tested an at-home saliva-collection kit using the Salimetrics device35. Specimens were returned via mail without refrigeration or preservatives. Moreover, streamlined processing techniques were developed for assaying saliva using quantitative, multiplexed immunoassays that can be automated to achieve the high throughput required for population-level testing.
Here, we evaluated the sensitivity and specificity of a “spit and mail” serology test using prospective samples and pre-established thresholds set by our pilot study. By testing samples provided from non-vaccinated individuals in the weeks following nasal PCR testing for SARS-CoV-2, our study addressed questions relevant for performing a large-scale serological survey. We verified the ability of a diverse group of participants to return specimens in compliance with shipping regulations using only written instructions without supervision or in-person training. We measured the dynamics of salivary anti-CoV-2 antibodies and computed the sensitivity and specificity at various time points after diagnostic PCR testing. Additionally, we compared strategies of identifying samples that may have been affected by pre-analytical issues inherent in shipping self-collected saliva without preservatives or refrigeration.
Results
Received specimens
We received self-collected saliva from 121 unique participants: 40 participants (33%) who tested positive for SARS-CoV-2 infection by RT-PCR and 81 participants who tested negative (67%). As shown in Table 1, the participants were diverse in age, gender, ethnicity, and socioeconomic status (SES). The samples were collected from non-vaccinated individuals from July 2020 to March 2021 within the Washington, DC metropolitan area, prior to the broad availability of SARS-CoV-2 vaccines. Participants who tested positive had similar demographics to participants who tested negative. The groups were well-balanced by gender and SES, as indicated by the area disadvantage index (ADI)36. Participants whose PCR test did not detect SARS-CoV-2 (PCR- participants) tended to be slightly older and more likely to be insured through Medicare than participants whose PCR test detected SARS-CoV-2 (PCR + participants). This trend may result from a lower positivity rate among older asymptomatic patients undergoing pre-surgical clearance for outpatient surgery than among patients tested due to symptoms of COVID-19. Additionally, PCR + participants were less likely to identify as non-Hispanic White, reflecting the rates of COVID-19 infection among different ethnic groups37.
Participants were instructed to return specimens immediately upon receiving an enrollment kit and then again at 10 and 30 days after their PCR test. Among participants who provided a specimen, the majority provided three specimens as instructed; however, the timing of collection and mailing in some cases varied considerably from the specified times. Samples were mailed by participants as early as 1 day and as late as 102 days after their RT-PCR test. The mode and median of days between PCR test and mailing the first sample were 9 and 11 days, respectively. To create groups with roughly equal numbers of samples, samples were divided into three time categories depending on whether they were mailed by participants < 2 weeks, 2–4 weeks, or 4–8 weeks after the PCR test (Table 2).
The transit time of specimens from participants to the laboratory ranged from 1 to 31 days (1st quartile = 1.4 days, median = 1.9 days, 3rd quartile = 3.5 days) in the mail with 91% arriving in less than 5 days, which was our target window based on prior testing showing stability for 5 days35. Two specimens that were in the mail over 20 days were excluded from analysis of test performance, but were included in an analysis investigating potential indicators of sample degradation.
IgG reactivity to SARS-CoV-2 antigens
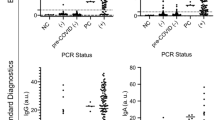
Concentrations of salivary IgG reactive to coronavirus antigens are shown in Fig. 1. Antibody positivity for SARS-CoV-2 antigens was determined based on pre-established thresholds set at the 98th percentile for saliva self-collected from presumed naive participants (no PCR confirmed diagnosis, no household exposure, and no symptoms of COVID-19) in a previous study35. Pre-established IgG thresholds for Spike, RBD, and N were 0.963, 0.244, and 3.18 AU/mL, respectively.
Salivary anti-SARS-CoV-2 IgG antibodies in the weeks after a PCR test. Reactivity to (a) full-length spike protein, (b) receptor binding domain (RBD) of the spike protein, and (c) N protein of SARS-CoV-2 were measured in saliva provided by 121 participants who had received a PCR test for SARS-CoV-2. Participants provided up to three samples. Matched samples provided by the same donor are connected with lines. 81 participants tested negative for SARS-CoV-2 (colored purple), and 40 participants tested positive (colored green). The dashed, red line indicates the pre-established cut-point for high versus low antibody levels. (d–f) Box and whisker plots of the same data shown in panels (a–c). The PCR + cohort was significantly different (p < 0.001; see Supplemental Tables) from the PCR- cohort at all timepoints. Asterisks indicate statistical significance (p < 0.05 by Mann–Whitney test) for difference between the < 2 weeks and 2–4 weeks timepoints.
Clinical performance of the serology assays was determined relative to the COVID-19 PCR test result at enrollment. Sensitivity and specificity were calculated, respectively, as (i) the proportion of saliva specimens from PCR-confirmed cases with antibody levels above the pre-established thresholds and (ii) the proportion of saliva specimens from PCR-negative cases with antibody levels at or below the pre-established thresholds. Measured sensitivity and specificity values are provided in Table 3. The SARS-CoV-2 Spike IgG assay provided the best overall accuracy. The sensitivity was only 40.7% within two weeks of PCR testing, but increased to 96.0% at 2–4 weeks and 92.6% at 4–8 weeks after PCR testing. The specificity was 92.4%. By comparison, when the same assay was evaluated with serum samples in an independent study, the sensitivity and specificity were reported as 90.8% and 97.4%, respectively38. The SARS-CoV-2 N IgG assay performed similarly to the SARS-CoV-2 Spike assay with point estimates for sensitivity and specificity that were not statistically different. The SARS-CoV-2 RBD IgG assay exhibited similar sensitivity; however, the specificity was significantly poorer (Table 3), which may indicate that the pre-set assay threshold was not optimal (see discussion of threshold verification below).
IgG reactivity to the SARS-CoV-2 spike protein was highly correlated with IgG reactivity to the N protein and RBD domain of spike protein, especially for samples from PCR-positive cases (Fig. 2). The correlation of the reactivities to RBD and Spike (Fig. 2a) shows that the concentrations of anti-RBD IgG antibodies tended to be about threefold lower than for full-length spike. As RBD is a fragment of the Spike protein, the difference in antibody activity is likely due to the reduced number of antigenic epitopes displayed for RBD relative to Spike. For PCR-negative individuals, measured reactivities of IgG to SARS-CoV-2 N tended to span a larger range than IgG to SARS-CoV-2 Spike, which was heavily skewed to the bottom of the assay range. This result may be a consequence of cross-reactive host antibodies from previous infections with other circulating coronaviruses, since the N protein has greater conservation across human coronaviruses than the Spike protein. However, the effect of any cross-reactivity on assay performance was small, with the N assay showing only a small and non-statistically significant decrease in specificity relative to the Spike assay.
We looked for evidence that elevated antibody levels in PCR-negative participants may have been due to undiagnosed infections. For the 67 PCR-negative participants who provided at least two samples, 6 of these 67 participants had at least one sample above the assay threshold for the SARS-CoV-2 Spike IgG assay. Of these 6 participants with at least one positive sample, 2 had salivary IgG levels above the threshold for SARS-CoV-2 Spike for all three of their samples. These participants also had salivary IgG levels above the threshold for SARS-CoV-2 N protein, which suggests an undiagnosed infection prior to enrollment. Of the 6 participants with at least one positive sample, 2 participants had an initial negative sample and then showed delayed sero-conversion after 30 days. The sero-conversion was also observed using the SARS-CoV-2 N IgG assay, which suggests that these participants may have been infected at enrollment and received a false negative PCR test39, or they may have become infected after enrollment.
Verification of pre-established thresholds
Receiver operator characteristic (ROC) curves were generated (Fig. 3), and the area under curve (AUC) values for the ROC curves were calculated (Supplemental Table 1) to compare the diagnostic performance of the serology assays at different times after nasal PCR testing and to confirm that the pre-determined thresholds were optimal for identifying infections. For all three SARS-CoV-2 antigens, the area under the curve (AUC) was significantly greater for samples collected more than two weeks after PCR testing relative to samples collected within 2 weeks of testing, largely reflecting the higher sensitivity that was observed for the later samples (Table 3). ROC curves for samples and the associated AUC values were not significantly different for samples collected 2–4 weeks and > 4 weeks after PCR testing, indicating that the assay achieved optimal diagnostic performance by the 2 week time point. The ROC curves for the SARS-CoV-2 Spike and N IgG assays were similar with AUC values of 0.926 and 0.916, respectively, for samples collected 4–8 weeks after PCR testing. The SARS-CoV-2 RBD assay provided poorer classification with an AUC value of 0.883 for the same samples.
Receiver-operator characteristic (ROC) curves for classifying SARS-CoV-2 PCR results based on salivary anti-SARS-CoV-2 IgG antibodies. Curves are drawn to show performance of assay at < 2 weeks, 2–4 weeks, and 4–8 weeks after a PCR test. The red dot indicates sensitivity and specificity achieved at the 4–8 week time point for the pre-established threshold. The black dot indicates, on the same curve, the threshold that maximizes sum of sensitivity and specificity.
To assess the validity of our pre-established thresholds, we computed the optimal thresholds using data only from this study by identifying thresholds that maximize the sum of sensitivity and specificity. The pre-established thresholds and the optimal thresholds for this study are compared graphically in Fig. 3. For the spike and N IgG assays, the pre-established thresholds were close to optimal and no significant improvement in sensitivity or specificity could be achieved by adjusting the threshold. In contrast, the pre-determined threshold for the RBD IgG assay was lower than optimal and increasing the threshold from 0.244 AU/mL to 0.684 AU/mL greatly improved the specificity for samples collected 4 weeks or later after PCR testing from 64 to 87%, while causing a much smaller loss in sensitivity from 93 to 89%.
Exploration of retest criteria
Although saliva collection is simple and intuitive, the potential for poor specimen quality should be addressed when specimens are self-collected without supervision and transported under uncontrolled conditions. We explored options for identifying specimens of high risk for providing inaccurate results including (i) the measurement of salivary antibodies that are expected to be universally abundant due to vaccinations or common natural infections, (ii) the measurement of total salivary immunoglobulin levels, and (iii) the measurement of background assay signals in the absence of an antigen target.
Prior infection with endemic coronaviruses is common40,41, so we expected that all donors would have high levels of antibodies to at least one of the four pre-COVID-19 endemic coronaviruses42. The multiplexed antigen panel used to measure antibodies to SARS-CoV-2 antigens also measured antibodies against the spike antigens for the four pre-COVID-19 endemic coronaviruses HKU1, NL63, OC43, and 229E (Fig. 4a). Nearly all specimens had readily detectable levels of antibodies to a spike protein of at least one endemic coronavirus. As an aggregate metric of reactivity to endemic coronaviruses, we computed the geometric mean of salivary IgG for HKU1, NL63, OC43, and 229E. We flagged eight outlier samples with geometric means below 0.17 AU/mL, which is the geometric mean of the 5th percentiles of the salivary IgG for these four antigens measured in a prior study35. These outliers appear to result from an issue with sample collection or sample deterioration, as opposed to the lack of immunity to an endemic coronavirus due to the absence of previous exposure or from general immunosuppression. In all cases where donors provided at least one other sample, normal levels of antibodies for endemic coronaviruses were measured at another time point. Sample deterioration due to delayed transit time in the mail could explain some of the flagged outliers, but not all of them. Two of the flagged samples were the samples with the longest transit times (> 20 days due to a general slowdown in mail during a period of this study), but the other 6 flagged samples were received within the target range of 5 days. For samples received within the target time range, there was no clear dependence of measured antibody levels with transit time (Fig. 4b). We note that for the 8 flagged samples, 6 were true negatives for SARS-CoV-2 infection by PCR testing, so excluding these samples did not significantly impact the reported sensitivity or specificity.
Concentrations of anti-spike and anti-N IgG measured in saliva of patients tested for SARS-CoV-2. Saliva was self-collected and returned via mail to a laboratory for analysis. For indirect serology, recombinant proteins representing the receptor binding domain (RBD) and N-terminal domain (NTD) of spike along with full-length spike and N antigen were multiplexed in wells of a 96-well plate. (a) IgG antibodies to four endemic coronaviruses were readily detected in nearly all samples. Eight samples (highlighted in blue) showed consistently low levels of antibodies to all coronaviruses. (b) 91% of samples were in transit for five days or less, and transit time was not strongly correlated with the mean level of salivary IgG for the four endemic coronaviruses (CoVs). Dotted line indicates geometric mean for the 5th percentiles of endemic CoV salivary IgG measured in a prior study of presumed naïve controls without prior diagnosis, household exposure, or symptoms of COVID-1935. (c) Total levels of immunoglobulins (IgG, IgM, and IgA) were also measured in saliva.
We also measured the total concentration of salivary IgG, IgM and IgA using a separate assay panel run at a different dilution (Fig. 4c). Median concentrations of total IgG, IgA and IgM were 3.3 µg/mL, ≥ 200 µg/mL (the top of the assay dynamic range at the selected sample dilution) and 3.4 µg/mL, respectively, which are comparable to the values we measured previously35 (1.8 µg/mL for IgG, 124 µg/mL for IgA and 3.7 µg/mL for IgM). Moreover, the IgG, IgA and IgM concentrations align with published ranges measured using a different assay and collection method (IgG range = 0.4–93 µg/mL43; IgA = 50.2 ± 19.1 µg/mL44; IgM = 0.5–13.0 µg/mL45). Low observed levels for the endemic coronaviruses were generally associated with low levels of total immunoglobulin. Of the eight samples that were flagged for low antibody levels against the four endemic coronaviruses, six samples had undetectable total IgG levels at the sample dilution used for the total immunoglobulin measurement, and four samples provided the lowest measured levels of total IgA (Fig. 4c).
Non-specific binding is another potential source of measurement error. Bovine serum albumin (BSA) was included as an antigen in the multiplex as a negative control. Specific binding of anti-BSA antibodies in samples should not occur due to the high concentration of BSA present in the assay diluents, therefore, binding to the non-BSA element in the antigen array should be indicative of antibodies that are able to bind non-specifically to the array surface. Non-specific binding, as assessed by signal for the control spot coated with BSA, was generally low (average of 191 counts). Two specimens from the same PCR negative donor were noted to exceed 5000 counts on the BSA coated spot, whereas a third intermediate sample from the same donor showed low non-specific binding.
Discussion
We measured anti-SARS-CoV-2 antibodies in saliva self-collected at home by donors in the weeks following a nasal RT-PCR test. This is the first report to our knowledge of a “spit and mail” serology test for SARS-CoV-2. Importantly, we found that a diverse group of participants were able to self-collect saliva and send it for testing. Using only written instructions without in-person supervision or training, participants universally packaged their specimens according to UN3373 regulations. Specimens were of sufficient quality for analysis, and most participants provided multiple specimens. Multiplexing of antigens for SARS-CoV-2 alongside endemic coronaviruses identified approximately 3% of samples that appeared aberrant due to low levels of antibodies against the four pre-COVID-19 endemic coronaviruses, and those samples may require retesting due to poor sample quality. This study builds upon prior studies18,19 that found general acceptance for self-collected specimens by demonstrating feasibility of a “spit and mail” approach using a highly scalable kit.
Among participants whose PCR-tests were positive for SARS-CoV-2, the kinetics of changes in salivary antibodies closely paralleled the kinetics of serum antibodies. Antibodies appeared in saliva as early as the first week after PCR testing, and the concentration of salivary antibodies increased over the course of two months, which has also been reported for serum46. Our study indicates that measuring salivary antibodies can be an alternative to measurement using serum or finger-stick blood. Among PCR-positive participants who returned at least one sample 2–4 weeks after testing, 96% had detectable anti-SARS-CoV-2 spike IgG, which is notable since participants were recruited among patients with mild to moderate disease not requiring hospitalization. In a large study of individuals who recovered from SARS-CoV-2 infection, 91.1% (1,107 of 1,215) were seropositive based on serum testing using a different assay47. Testing among outpatients is more challenging since a systemic humoral response may be delayed or absent in some mild cases48, whereas seroconversion occurs in nearly 100% of hospitalized patients46,49. The sensitivity for detecting asymptomatic infection may be lower since individuals with mild cases tend to have less robust immune responses compared to individuals with severe disease50,51,52,53.
Samples were classified as antibody positive or negative based on thresholds determined in a previous study of self-collected saliva samples. Using these thresholds, we found that our assays for IgG against SARS-CoV-2 Spike and N antigens provided good sensitivity and specificity for classifying subjects based on COVID PCR test results. The ability to simultaneously measure IgG to both N and Spike proteins may potentially distinguish individuals who have been infected with SARS-CoV-2 (reactivity to both N antigen and Spike) from those that have been vaccinated without a prior infection (reactivity to Spike only). Poorer performance was observed for IgG to SARS-CoV-2 RBD. ROC analysis showed that the pre-determined threshold was not optimal. It is not clear why the optimal thresholds for the RBD IgG assay would be different for the earlier and current studies and why the difference would only affect one of the assays. The difference could be associated with differences in how self-collected samples were submitted for testing (collection in drop boxes vs. shipping by mail) or differences in the testing population (healthy individuals vs. individuals who presented for COVID testing and may have symptoms of a respiratory infection).
A limitation of this study is that we did not examine matched serum or finger-stick blood samples. Correlation of salivary antibody concentrations with finger-stick blood antibodies would help to determine the reason for the few cases when antibody levels were not as expected based on PCR testing. In particular, some participants may have previously been infected with SARS-CoV-2 prior to the nasal swab PCR test. Another limitation is that we did not measure reactivity of salivary IgA to SARS-CoV-2 antigens, which could potentially provide information that is different and complementary to IgG reactivity when assessing immune status.
Due to a lack of validated criteria to exclude samples based on atypical antibody testing, we excluded only two samples that were affected by prolonged postal delays. Measuring antibodies against endemic coronaviruses was identified as a potential approach, which can be easily multiplexed with the SARS-CoV-2 serology measurements, to flag specimens with suspected technical issues. Establishing retest criteria is recommended before the assay can be used for clinical testing. A geometric mean of antibody levels to four endemic coronaviruses is a promising metric for flagging suspect samples, especially because multiplex assays can obtain this metric without additional sample volume or processing. Additional studies with matched serum samples, or saliva samples collected under ideal conditions, are needed to refine and validate methods for identifying samples that may be affected by collection or handling issues.
Overall, we show the feasibility of a “spit and mail” test for large-scale serologic testing. The easy-to-use kit could be used by a diverse group of participants. The assays require minimal sample handling and can be performed rapidly using an automated analyzer capable of high-throughput testing. Potential uses include epidemiologic studies that require identifying people who have previously been infected or vaccinated. Also, saliva tests may find utility for monitoring durability of immunity.
Methods and materials
Participant recruitment and enrollment
Participants were recruited among adult patients tested for SARS-CoV-2 infection at Kaiser-Permanente clinics in Maryland via RT-PCR testing of nasal swabs. After the diagnostic test was performed, patients were invited to participate. All tested patients were eligible to participate regardless of symptoms, exposure risk, or reason for testing (e.g., pre-surgical evaluation, contact tracing). Participants who agreed to participate were provided with an enrollment box containing instructions, informed consent form, and three self-collection kits. All participants were recruited via a telephone call with the exception of one, who was recruited at a testing clinic and provided an enrollment box. Participants recruited via phone were shipped an enrollment box that arrived within 1–3 days after enrollment. Informed consent was obtained from all participants via a signed barcoded informed consent form and HIPAA authorization, which were mailed back to Kaiser-Permanente researchers. Participants were instructed to self-collect saliva on the day they received the kit and then at 10 and 30 days after their test. Saliva was returned directly to Meso Scale Diagnostics, LLC. (MSD) via the provided pre-paid mailers. MSD tracked participants through bar codes associated with the self-collection kits and had no access to identifiable personal information. Study procedures were approved by the IRB of Kaiser-Permanente. The KPMAS IRB reviewed, approved, and monitored the data collection, management, and analysis protocol in accordance with relevant guidelines/regulations.
PCR-confirmed diagnosis of SARS-CoV-2 infection
Nasal swabs were tested by RT-PCR using a SARS-CoV-2 NAAT and a PAN-SARS Coronavirus NAAT. SARS-CoV-2 NAAT amplified the unique ORF1ab non-structural region, and the PAN-SARS Coronavirus NAAT amplified the conserved pan-Sarbecovirus structural protein envelope E gene. The results were interpreted as positive if either the ORF1ab alone or both the ORF1ab and E gene resulted in a positive result for the detection of SARS-CoV-2. Detection of the pan-Sarbecovirus E gene alone resulted in a presumptive positive result. Results were deemed negative if neither target was detected.. Results were obtained from the participant’s electronic medical record by Kaiser-Permanente (KP) researchers. All participants tested either positive or negative, with no participants testing positive for the pan-Sarbecovirus E gene but negative for the ORF1ab alone.
Collection and transport of saliva
As described previously35, saliva was collected into a 2 mL screw-cap centrifuge tube (Sarstedt #72.609 with screw cap 65.716.xxx) using the Saliva Collection Aid (SCA; Salimetrics 5016.02), which is a straw-like device cleared by the FDA for collection of samples from adults and children. Centrifuge tubes were pre-labeled with barcodes that matched barcodes on informed consent forms in order to associate samples with participants in a deidentified manner. The screw cap contained an O-ring in order to be compliant with UN3373 Category B shipping requirements. Donors were instructed to wait 30 min after eating, drinking, or smoking before drooling into the tube according to the manufacturer’s directions. Donors were also instructed to provide saliva without assistance from others. The donors capped the tube, and placed it inside a biohazard bag (VWR 11215-684) containing an absorbent pad (ThermoSafe ZORB66). Samples were collected from July 2020 to March 2021 within the Washington, DC metropolitan area, prior to the broad availability of SARS-CoV-2 vaccines.
To return their samples, donors were instructed to place the biohazard bag containing the tube of saliva into a peel-and-seal cardboard mailer (Stephen Gould; MSD-CFM-SM) bearing a UN3373 label (LabelMaster L380B) and pre-paid shipping label as described previously35. The COVID-19 taskforce of the USPS reviewed the shipping materials and instructions for packing and mailing the specimens to ensure compliance with CDC guidelines and USPS regulations. A removable adhesive tag was attached to the back of each mailer alerting mail carriers to pick up the package.
Saliva receipt and storage
Samples were returned anonymously to MSD via USPS First Class Mail. Upon delivery to MSD, saliva was frozen promptly at ≤ − 70 ºC without further processing. The date that saliva was mailed was documented as the date that the sample was picked up as determined by the USPS tracking history. For the initial samples, the accuracy of the USPS tracking record was cross-verified against the date entered by the patient on the informed consent form.
Indirect serology
Indirect serology measurements were conducted using kits and reagents that are commercially available from MSD as described previously35. On the day of sample testing, saliva was thawed at room temperature. Saliva was centrifuged briefly to pull down any food particles or mucus. To assess sample quality, we visually verified that samples were saliva and not predominantly phlegm or mucus.
Prior to analysis, saliva samples were diluted five-fold by combining 20 µL of sample with 80 µL of a sample diluent (MSD® Diluent 2). Samples were assayed in a 96-well plate format using MSD V-PLEX® COVID-19 Coronavirus Panel 2 kits for measuring IgG (K15369U) antibody responses. Each well of the plates included an antigen array that enabled the multiplexed measurement of antibody responses against nine different coronavirus antigens as well as bovine serum albumin (BSA) as a negative control. These included four SARS-CoV-2 antigens (nucleocapsid protein, spike protein, spike receptor binding domain (RBD) and spike N-terminal domain (NTD)) and spike proteins from five other coronaviruses (SARS-CoV-1 and the four endemic coronaviruses 229E, HKU1, NL63, and OC43). Assay protocols were run according to the manufacturer’s recommended protocol for serum except for the use of sample diluents and dilution factors (as described above) that were optimized for saliva. Testing of saliva samples was carried out in an automated fashion using high-throughput automation developed at MSD. Time-to-result was approximately four hours.
For quantitation of antibody responses, an eight-point calibration curve was run in duplicate on all plates and the signals for each antigen were fit to a 1/Y2-weighted four parameter logistic (4PL) fit. Samples were run in duplicate and the antibody concentration against each antigen was calculated by back-fitting to the appropriate 4PL fit and correcting for dilution. The concentrations were presented in arbitrary units per mL (AU/mL) that were defined relative to the assigned values of the reference standard. Controls were also run in duplicate on each plate, including three serum-based controls (provided with the kit) and two saliva-based controls (pooled normal saliva sourced from Lee Biosolutions spiked with serum from COVID-19 patients).
Measurement of total salivary antibodies
Total levels of IgG, IgM, and IgA immunoglobulin were measured using MSD’s Isotyping Panel 1 Human/NHP Kit (K15203D) according to the manufacturer’s directions as described previously35. Saliva was run at a dilution of 1,000-fold. Calibration and quantitation were carried out as described above for the indirect serology measurements.
Data analysis
Data processing was performed in Excel. Graphing and statistical analysis were done in R. Concentrations below an assay’s limit of detection (LOD) were assigned concentration values equal to the LOD, which was calculated as the concentration corresponding to 2.5 standard deviations above the assay’s background signal. Concentrations exceeding the top calibrator were assigned the concentration of the top calibrator.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Gilbert, P. B. et al. Immune correlates analysis of the mRNA-1273 COVID-19 vaccine efficacy clinical trial. Science 375, 43-50. https://doi.org/10.1126/science.abm3425 (2021).
Earle, K. A. et al. Evidence for antibody as a protective correlate for COVID-19 vaccines. Vaccine 39, 4423–4428 (2021).
Feng, S. et al. Correlates of protection against symptomatic and asymptomatic SARS-CoV-2 infection. Nat. Med. 27, 2032-2040. https://doi.org/10.1038/s41591-021-01540-1 (2021).
Röltgen, K. et al. Immune imprinting, breadth of variant recognition, and germinal center response in human SARS-CoV-2 infection and vaccination. Cell 185, 1025-1040. https://doi.org/10.1016/j.cell.2022.01.018 (2022).
West, R., Kobokovich, A., Connell, N. & Gronvall, G. K. COVID-19 antibody tests: A valuable public health tool with limited relevance to individuals. Trends Microbiol. 29, 214–223 (2021).
Developing a National Strategy for SARS-CoV-2 Serosurveys in the US. Johns Hopkins Center for Health Security. https://www.centerforhealthsecurity.org/our-work/publications/developing-a-national-strategy-for-sars-cov-2-serosurveys-in-the-united-states.
Bubar, K. M. et al. Model-informed COVID-19 vaccine prioritization strategies by age and serostatus. Science 371, 916-921. https://doi.org/10.1126/science.abe6959 (2021).
Krammer, F. et al. Antibody responses in seropositive persons after a single dose of SARS-CoV-2 mRNA vaccine. N. Engl. J. Med. 384, 1372–1374 (2021).
Widge, A. T. et al. Durability of responses after SARS-CoV-2 mRNA-1273 vaccination. N. Engl. J. Med. 384, 80–82 (2021).
Dan, J. M. et al. Immunological memory to SARS-CoV-2 assessed for up to 8 months after infection. Science 371, 1–10 (2021).
Cota, G. et al. Diagnostic performance of commercially available COVID-19 serology tests in Brazil. Int. J. Infect. Dis. 101, 382–390 (2020).
Subramanian, R., He, Q. & Pascual, M. Quantifying asymptomatic infection and transmission of COVID-19 in New York City using observed cases, serology, and testing capacity. PNAS 118, 1–10 (2021).
Anand, S. et al. Prevalence of SARS-CoV-2 antibodies in a large nationwide sample of patients on dialysis in the USA: A cross-sectional study. The Lancet 396, 1335–1344 (2020).
Buss, L. F. et al. Three-quarters attack rate of SARS-CoV-2 in the Brazilian Amazon during a largely unmitigated epidemic. Science 371, 288–292 (2021).
Ng, D. L. et al. SARS-CoV-2 seroprevalence and neutralizing activity in donor and patient blood. Nat. Commun. 11, 4698 (2020).
Uyoga, S. et al. Seroprevalence of anti-SARS-CoV-2 IgG antibodies in Kenyan blood donors. Science 371, 79–82 (2021).
Bajema, K. L. et al. Estimated SARS-CoV-2 seroprevalence in the US as of September 2020. JAMA Intern. Med. 181, 450-460. https://doi.org/10.1001/jamainternmed.2020.7976 (2021).
Siegler, A. J. et al. Willingness to seek diagnostic testing for SARS-CoV-2 with home, drive-through, and clinic-based specimen collection locations. Open Forum Infect. Dis. 7, 269 (2020).
Valentine-Graves, M. et al. At-home self-collection of saliva, oropharyngeal swabs and dried blood spots for SARS-CoV-2 diagnosis and serology: Post-collection acceptability of specimen collection process and patient confidence in specimens. PLoS ONE 15, e0236775 (2020).
Sakhi, A. K. et al. Feasibility of self-sampled dried blood spot and saliva samples sent by mail in a population-based study. BMC Cancer 15, 265 (2015).
Etter, J. F., Perneger, T. V. & Ronchi, A. Collecting saliva samples by mail. Am. J. Epidemiol. 147, 141–146 (1998).
Brandtzaeg, P. Secretory immunity with special reference to the oral cavity. J. Oral Microbiol. 5, 20401 (2013).
Wu, X. & Jackson, S. Plasma and salivary IgG subclasses in HIV type 1 infection: Evidence of both transudation and local synthesis of IgG in parotid saliva. AIDS Res. Hum. Retroviruses 16, 1423–1431 (2000).
Strober, W., Blaese, R. M. & Waldmann, T. A. The origin of salivary IgA. J. Lab. Clin. Med. 75, 856–862 (1970).
Isho, B. et al. Persistence of serum and saliva antibody responses to SARS-CoV-2 spike antigens in COVID-19 patients. Sci. Immunol. 5, 52–54 (2020).
Pisanic, N. et al. COVID-19 serology at population scale: SARS-CoV-2-specific antibody responses in saliva. J. Clin. Microbiol. 59, 10 (2020).
Faustini, S. E. et al. Development of a high-sensitivity ELISA detecting IgG, IgA and IgM antibodies to the SARS-CoV-2 spike glycoprotein in serum and saliva. Immunology 164, 135-147. https://doi.org/10.1101/2020.06.16.20133025 (2021).
Heaney, C. D. et al. Comparative performance of multiplex salivary and commercially available serologic assays to detect SARS-CoV-2 IgG and neutralization titers. J. Clin. Virol. 145, 104997. https://doi.org/10.1016/j.jcv.2021.104997 (2021).
MacMullan, M. A. et al. ELISA detection of SARS-CoV-2 antibodies in saliva. Sci. Rep. 10, 20818 (2020).
Granade, T. C. et al. Detection of antibodies to human immunodeficiency virus type 1 in oral fluids: A large-scale evaluation of immunoassay performance. Clin. Diagn. Lab. Immunol. 5, 171–175 (1998).
Hodinka, R. L., Nagashunmugam, T. & Malamud, D. Detection of human immunodeficiency virus antibodies in oral fluids. Clin. Diagn. Lab. Immunol. 5, 419–426 (1998).
MacMullan, M. A. et al. Detection of SARS-CoV-2 antibodies in oral fluid obtained using a rapid collection device. J. Clin. Microbiol. 59, e0251020 (2020).
Khurshid, Z. et al. Human saliva collection devices for proteomics: An update. Int. J. Mol. Sci. 17, 846 (2016).
Allicock, O. M. et al. Evaluation of saliva self-collection devices for SARS-CoV-2 diagnostics. BMC Infect. Dis. 22, 284. https://doi.org/10.1186/s12879-022-07285-7 (2022).
Campbell, C. et al. Quantitative serology for SARS-CoV-2 using self-collected saliva and finger-stick blood. Sci. Rep. 12, 6560. https://doi.org/10.1038/s41598-022-10484-6 (2022).
Singh, G. K. Area deprivation and widening inequalities in US mortality, 1969–1998. Am. J. Public Health 93, 1137–1143 (2003).
Kaufman, H. W., Niles, J. K. & Nash, D. B. Disparities in SARS-CoV-2 positivity rates: Associations with race and ethnicity. Popul. Health Manag. 24, 20–26 (2021).
Johnson, M. et al. Evaluation of a novel multiplexed assay for determining IgG levels and functional activity to SARS-CoV-2. J. Clin. Virol. 130, 104572 (2020).
Arevalo-Rodriguez, I. et al. False-negative results of initial RT-PCR assays for COVID-19: A systematic review. PLoS ONE 15, e0242958 (2020).
Gaunt, E. R., Hardie, A., Claas, E. C. J., Simmonds, P. & Templeton, K. E. Epidemiology and clinical presentations of the four human coronaviruses 229E, HKU1, NL63, and OC43 detected over 3 years using a novel multiplex real-time PCR method. J. Clin. Microbiol. 48, 2940–2947 (2010).
Killerby, M. E. et al. Human coronavirus circulation in the United States 2014–2017. J. Clin. Virol. 101, 52–56 (2018).
Westerhuis, B. M. et al. Homologous and heterologous antibodies to coronavirus 229E, NL63, OC43, HKU1, SARS, MERS and SARS-CoV-2 antigens in an age stratified cross-sectional serosurvey in a large tertiary hospital in The Netherlands. MedRxiv https://doi.org/10.1101/2020.08.21.20177857 (2020).
Riis, J. L., Bryce, C. I., Stebbins, J. L. & Granger, D. A. Salivary total immunoglobulin G as a surrogate marker of oral immune activity in salivary bioscience research. Brain Behav. Immunity Health 1, 100014 (2020).
Lehne, G. et al. Oral administration of a new soluble branched β-1,3-D-glucan is well tolerated and can lead to increased salivary concentrations of immunoglobulin A in healthy volunteers. Clin. Exp. Immunol. 143, 65–69 (2006).
Horsfall, A. C., Rose, L. M. & Maini, R. N. Autoantibody synthesis in salivary glands of Sjögren’s syndrome patients. J. Autoimmun. 2, 559–568 (1989).
Fu, Y. et al. Dynamics and correlation among viral positivity, seroconversion, and disease severity in COVID-19. Ann. Intern. Med. 174, 453-461. https://doi.org/10.7326/M20-3337 (2021).
Gudbjartsson, D. F. et al. Humoral immune response to SARS-CoV-2 in Iceland. N. Engl. J. Med. 383, 1724–1734 (2020).
Kellam, P. & Barclay, W. The dynamics of humoral immune responses following SARS-CoV-2 infection and the potential for reinfection. J. Gen. Virol. 101, 791–797 (2020).
Algaissi, A. et al. SARS-CoV-2 S1 and N-based serological assays reveal rapid seroconversion and induction of specific antibody response in COVID-19 patients. Sci. Rep. 10, 16561 (2020).
Choe, P. G. et al. Antibody responses to SARS-CoV-2 at 8 weeks postinfection in asymptomatic patients. Emerg. Infect. Dis. 26, 2484–2487 (2020).
Ko, J.-H. et al. Neutralizing antibody production in asymptomatic and mild COVID-19 patients, in comparison with pneumonic COVID-19 patients. J. Clin. Med. 9, 2268 (2020).
Long, Q.-X. et al. Antibody responses to SARS-CoV-2 in patients with COVID-19. Nat. Med. 26, 845–848 (2020).
Qu, J. et al. Profile of immunoglobulin G and IgM antibodies against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Clin. Infect. Dis. 71, 2255–2258 (2020).
Acknowledgements
This project has been funded in part with Federal funds from the National Institute of Allergy and Infectious Diseases, National Institutes of Health, Department of Health and Human Services (HHS), under Contract No. HHSN272201700013C.
Author information
Authors and Affiliations
Contributions
C.C., D.Roblin, L.G., and C.M. developed study protocols. L.F., J.W., A.D., C.M., L.G., and T.W. enrolled participants and/or compiled clinical information. N.P., D.Romero, and J.J. optimized assays for saliva and analyzed samples with advice from C.C. and G.S. C.C. and P.G. designed at-home self-collection kits. C.C. and D.Roblin wrote the manuscript. G.S. supervised the project and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
C.C., N.P., D. Romero, J.J., P.G., and G.S. are employees of Meso Scale Diagnostics, LLC. D. Roblin., L.F., T.W., J.W., C.M., A.D., and L.G. report no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Campbell, C., Roblin, D., Padmanabhan, N. et al. Saliva-based SARS-CoV-2 serology using at-home collection kits returned via mail. Sci Rep 12, 14061 (2022). https://doi.org/10.1038/s41598-022-17057-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-17057-7
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.