Abstract
Ocular cranial nerve palsy of presumed ischemic origin (OCNPi) is the most common type of ocular cranial nerve palsy (OCNP) in patients aged ≥ 50 years; however, no definite diagnostic test exists. As diagnostic criteria include clinical improvement, diagnoses are often delayed. Diagnostic biomarkers for OCNPi are required; we hypothesized that cerebral small vessel disease is associated with OCNPi. We analyzed 646 consecutive patients aged ≥ 50 years with isolated unilateral OCNP who underwent work-ups at two referral hospitals. White matter hyperintensities (WMHs), silent infarctions, and cerebral microbleeds (CMBs) were assessed. In multivariate analyses, mild (grades 1–3) and moderate to severe (grades 4–6) WMHs were significantly associated with OCNPi compared to OCNP of other origins (odds ratio [OR] 3.51, 95% confidence interval [CI] 1.91–6.43, P < 0.001; OR 3.47, 95% CI 1.42–8.48, P = 0.006, respectively). Silent infarction and CMBs did not remain associated (OR 0.96, 95% CI 0.54–1.70, P = 0.870; OR 0.55, 95% CI 0.28–1.08, P = 0.080, respectively). Associations between WMH and OCNPi remained after excluding patients with vascular risk factors. In conclusion, the presence of WMH could independently predict ischemic origin in patients with isolated unilateral OCNP at early stage of admission.
Similar content being viewed by others
Introduction
Ocular cranial nerve palsy (OCNP) is a neurological deficit that occurs due to dysfunction of one or more oculomotor, trochlear, and abducens nerves. It is important to clarify the cause of OCNP because the management plan and prognosis will be completely different depending on its cause. The possible causes of OCNP include microvascular ischemia, tumor, infection, aneurysm, trauma, and vascular malformation1. Careful history-taking, physical examination, and neuroimaging studies are helpful in revealing the causes of OCNP2.
OCNP of presumed ischemic origin (OCNPi) is the most common cause of OCNP among patients aged ≥ 50 years3, 4. However, it has no definitive diagnostic test, and other possible causes should be ruled out before diagnosis5. Vascular risk factors, such as diabetes, hypertension, and hyperlipidemia, could increase the probability of OCNPi, but it can occur in patients without any history of vascular risk factors5. OCNPi can be diagnosed when (i) no other cause is found on clinical examination or neuroimaging, (ii) there is no other accompanying neurological deficit, and (iii) symptoms improve over 3–6 months6, 7. In other words, a confirmatory diagnosis cannot be made until symptoms improve. This uncertainty in diagnosis could worry patients during the follow-up. Magnetic resonance imaging (MRI) findings predicting OCNPi could help reduce this.
Ischemic OCNP occurs due to endothelial deformation, loss of tight junctions, basement membrane hypertrophy, and loss of pericytes, leading to endoneurial space edema and hypoxia in ocular cranial nerves8. Arteriolar hyalinization has been observed in post-mortem studies of OCNPi patients9. In brains of patients with vascular risk factors, the arteriolar and capillary levels of the vasculature can be affected, resulting in cerebral small vessel disease (SVD)10. SVD is well visualized on MRI and is characterized by heterogeneous findings, such as white matter hyperintensities (WMH), silent infarction, cerebral microbleeds (CMBs), and enlarged perivascular space10. Thus, we hypothesized that cerebral SVD could be associated with OCNPi. In this study, the imaging markers of cerebral SVD, WMHs, silent infarction, and CMBs on MRI of patients with acute-onset isolated OCNP were evaluated.
Methods
Study population, inclusion criteria, and exclusion criteria
We retrospectively reviewed the electronic and paper medical records of consecutive patients aged ≥ 50 years with isolated oculomotor, trochlear, or abducens nerve palsy diagnosed by neurologists and were admitted to the Seoul National University Hospital and the Seoul National University-Seoul Metropolitan Government Boramae Medical Center, South Korea between January 2000 and April 2020. We collected eligible cases using the following inclusion criteria: (i) isolated unilateral OCNP, (ii) OCNP unrelated to trauma or neurosurgery, (iii) no previous history of strabismus, and (iv) diagnostic workup including MRI. Isolated unilateral OCNP was defined as unilateral oculomotor, trochlear, or abducens nerve palsy with no other neurological symptoms or signs, except for headache and periorbital pain. The exclusion criteria were as follows: (i) poor MRI quality; (ii) incomplete work-up to support a final diagnosis; and (iii) follow-up loss within 6 months and inability to confirm symptom improvement. Because this study aimed to investigate OCNP induced by ischemia of the vasa nervorum, OCNP cases caused by stroke, which is a brain parenchymal ischemia due to occlusion of other intracranial arteries, were excluded. The study protocol was designed in accordance with the Declaration of Helsinki. The Institutional Review Boards (IRB) of the Seoul National University Hospital and Seoul National University-Seoul Metropolitan Government Boramae Medical Center approved this study (IRB No. J-2007-023-1139 and 10-2019-39, respectively) and waived the requirement to obtain informed consent because of its retrospective design.
Collection of clinical information
We collected the participants’ baseline demographic and clinical information, including age, sex, body mass index (BMI), hypertension, diabetes, hyperlipidemia, chronic kidney disease, history of ischemic heart disease, history of stroke, current cigarette smoking, and use of antithrombotics. Laboratory findings of hemoglobin A1c (HbA1c) and estimated glomerular filtration rate (eGFR) were collected.
The affected nerve, cause of OCNP, and presence of periorbital pain or headache were reviewed. OCNPi was diagnosed when symptoms spontaneously improved within 12 months of the follow-up period, with no other explainable diagnoses. Patients with no symptom improvement for > 1 year of follow-up and no explainable diagnosis were considered idiopathic.
Image analysis
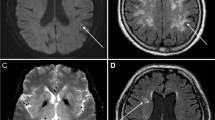
The patients were examined using either a 3 T or 1.5 T MRI unit. The types of scanner and imaging acquisition parameters are summarized in Supplementary Table S1 and Supplementary Fig. S1. For WMH, each periventricular WMH (PVWMH) and deep WMH (DWMH) was graded as previously described. PVWMH was graded as Grade 0 (absence), 1 (caps or pencil-thin lining), 2 (smooth halo), or 3 (irregular PVWMH extending into the deep white matter). DWMH was graded as 0 (absence), 1 (punctate foci), 2 (beginning confluence of foci), or 3 (large confluent areas)11. Total WMH was graded by summing the PVWMH and DWMH, which ranged from 0 to 6. The total WMH was divided into three groups as follows: no WMH, grade 0; mild WMH, grades 1–3; and moderate to severe WMH, grades 4–612,13,14. We defined silent infarction as abnormal low-signal intensity lesions of at least 3 mm in size on fluid-attenuated inversion recovery (FLAIR) with or without hyperintense rim15. CMBs were defined as dark blooming artifacts visible on T2*-weighted gradient echo (GRE) or susceptibility weighted images (SWI) surrounded by the brain parenchyma15. Two independent examiners evaluated MRI images without clinical information. The inter-rater agreement for the imaging parameters was assessed using the intraclass correlation coefficient (ICC) and 95% confidence interval (CI).
Statistical analysis
In the univariate analysis, Student’s t-tests and chi-square tests were used for continuous and categorical variables, respectively. Multivariate logistic regression analysis models were constructed using imaging markers as dependent variables with adjustment for relevant confounding variables. We also considered the MRI field strength (3 T vs. 1.5 T) and susceptibility sequence type (SWI vs. GRE) as possible confounding variables of the WMH and silent infarction and CMBs, respectively. Statistical significance was set at P < 0.05. All statistical analyses were performed using R (version 4.0.2; R Development Core Team, Vienna, Austria).
Results
We recruited 710 patients with acute-onset isolated unilateral OCNP who underwent diagnostic workup, including MRI, from January 2000 to April 2020. After excluding 64 cases, 646 were analyzed (61.6% male, mean age 67 ± 9 years) (Fig. 1). The baseline characteristics of the patients are shown in Table 1 and Supplementary Table S2. The proportions of oculomotor, trochlear, and abducens nerves were 39.5%, 21.5%, and 39.0%, respectively. The proportion of patients with OCNPi was 73.2% (473 patients). The detailed causes of OCNP and their respective proportions are listed in Table 2 and Supplementary Table S3. Three-tesla and 1.5 T MRI were used in 298 (46.1%) and 348 (53.9%), respectively, and SWI and GRE sequences were used in 244 (37.8%) and 327 (50.6%), respectively. Of the suggested MRI acquisition standards for neuroimaging for clinical research16, all MRIs met the in-plane resolution criteria of ≤ 1 mm × 1 mm, and 97.5% of MRIs met the slice thickness criteria of ≤ 5 mm. WMH, PVWMH, and DWMH were present in 512 (79.3%), 374 (57.9%), and 477 (73.8%) patients, respectively. Silent infarctions and CMBs were visualized in 152 (23.6%) and 86 (15.1%) cases, respectively. The inter-rater agreement of WMH, silent infarction, and CMBs was substantial for MRI parameters (Cohen’s kappa values of 0.78, 0.79, and 0.88, respectively; P < 0.001 for all parameters).
We analyzed factors associated with OCNPi compared to OCNP of other origin (OCNPo) using univariate analyses (Table 3). Of the 473 patients with OCNPi, 393 (83.1%) had WMH, and 119 (68.8%) of the 173 patients with OCNPo had WMH (P < 0.001). Both mild (grades 1–3) and moderate to severe (grades 4–6) WMH were more frequent in patients with OCNPi than in those with OCNPo. Both PVWMH and DWMH were also associated with OCNPi (PVWMH, 292 [61.7%] vs. 82 [47.4%]; DWMH, 363 [76.7%] vs. 114 [65.9%] in ischemic and OCNPo, respectively; both P = 0.007). Silent infarction was more prevalent in patients with OCNPi than those with OCNPo (OCNPi vs. the OCNPo, 122 [25.8%] vs. 30 [17.3%], P = 0.032). No association was observed between CMBs and OCNPi. Although the proportion of mild PVWMH was higher on 3 T MRI than on 1.5 T MRI, the proportion of WMH was comparable between 3 T and 1.5 T MRI (Supplementary Table S4). CMBs were more frequently visualized using SWI than using GRE (Supplementary Table S5). The proportions of 3 T MRI (vs. 1.5 T MRI) and SWI (vs. GRE) were also comparable between OCNPi and OCNPo. Among the clinical variables, male sex, old age, high BMI, hypertension, diabetes, history of ischemic heart disease, eGFR, and antithrombotic use were associated with OCNPi. The presence of at least one vascular risk factor among hypertension, diabetes, hyperlipidemia, and smoking was 83.9% (397/473) for OCNPi and 60.7% (105/173) for OCNPo (P < 0.001).
In the multivariate analysis, both mild (grades 1–3) and moderate to severe (grades 4–6) WMH were independent predictors of OCNPi (Table 4 and Fig. 2). The odds ratio (OR) and 95% CI of mild and moderate-to-severe WMH were 3.51 [1.91–6.43] and 3.47 [1.42–8.48], respectively (P < 0.001 and P < 0.006, respectively). Among the covariates, trochlear and abducens nerve involvement, male sex, high BMI, and diabetes were independent variables associated with OCNPi. Multivariate analyses were performed to investigate whether each MRI marker, silent infarction, and CMBs were independently associated with OCNPi (Table 5). There was no clear association between OCNPi and CMBs or silent infarctions. Neither PVWMH nor DWMH alone showed any association with the OCNPi (Supplementary Table S6).
Forest plots for odds ratios of the ocular cranial nerve palsy of presumed ischemic origin (OCNPi) compared with ocular cranial nerve palsy of other origin (OCNPo) based on multivariate logistic regression analysis. CN cranial nerve, BMI body mass index, WMH white matter hyperintensities, HTN hypertension, DM diabetes, IHD ischemic heart disease, eGFR estimated glomerular filtration rate. ***P < 0.001; **P < 0.01; *P < 0.05.
To exclude the confounding effects of vascular risk factors, we performed sensitivity analysis in patients without any vascular risk factors. A total of 144 patients were analyzed, and the baseline characteristics and factors associated with OCNPi are described in Supplementary Table S7 (76 patients with OCNPi vs. 68 patients with OCNPo). ORs for mild (grades 1–3) and moderate to severe (grades 4–6) WMH and OCNPi were 4.79 (95% CI 1.23–18.65, P = 0.024) and 11.58 (95% CI 1.29–104.18, P = 0.029), respectively (Table 6). There was no clear association between other SVD biomarkers and OCNPi.
Discussion
The clinical characteristics of 646 patients aged ≥ 50 years who were diagnosed with acute-onset unilateral isolated OCNP were investigated. We enrolled patients aged ≥ 50 years for this study because OCNPi is uncommon in younger patients17, and extrinsic causes such as trauma-related and surgery-related OCNP were not included to focus on the intrinsic factors associated with OCNPi. Because this study aimed to investigate ischemia of the peripheral cranial nerves, ischemia of the nuclear or fascicular lesions of the ocular cranial nerve caused by stroke were excluded. In this study, mild and moderate-to-severe WMHs were found to be independent predictors of OCNPi.
The positive finding we found on early MRI is meaningful as it might be used as a predictor of OCNPi. Since OCNPi is a diagnosis based on exclusion, the diagnosis can only be made by ensuring that there are no other explanatory causes. Currently, only a few studies have reported findings suggestive of OCNPi. One study group showed enhancement of the third cranial nerve on thin-section MRI but did not confirm the same findings of the fourth or sixth cranial nerves, and further validation is needed18, 19. WMH was associated with OCNPi regardless of the cranial nerve involved in this study.
The definition of the OCNPi varies between studies. One study defined presumed microvascular OCNP as a case in which symptoms improved within 6 months without other explainable causes. Another study defined an OCNPi as a case without other explainable causes and with at least one vascular risk factor. In this study, an OCNPi was defined as a case in which symptoms improved within 12 months without other explainable causes, regardless of the presence of vascular risk factors. These criteria were set because it is known that up to 85% of the OCNPi completely resolve within 12 months and typically resolve within 3 months6, 7. This reduced the number of patients with idiopathic OCNP (0.8%, 5/646) compared to those of other studies (2%–14%)3, 7.
In this study, 76 of 473 OCNPi patients (16.1%) and 68 of 173 OCNPo patients (39.3%) did not have vascular risk factors (i.e., hypertension, diabetes, dyslipidemia, or smoking). In a sensitivity analysis of this subgroup, both mild (grades 1–3) and moderate-to-severe (grades 4–6) WMH remained significant. This suggests that there is a common personal vulnerability for WMH and OCNPi other than the contribution of conventional vascular risk factors. Furthermore, we propose that a substantial proportion of patients previously diagnosed with idiopathic OCNP develop OCNP due to microvascular ischemia.
WMH, silent infarctions, and CMBs are representative biomarkers of cerebral SVD. These imaging findings refer to lesions mainly at the perforating arterial and arteriolar levels such as arteriosclerosis, lipohyalinosis, and fibrinoid necrosis. Cerebral SVDs can be observed in healthy elderly people as well as in individuals with vascular risk factors such as hypertension and smoking20. While silent infarction and CMBs are mainly due to a single perforating arterial or arteriolar occlusion or rupture, WMH is more diffusely distributed and observable from the early stage of SVD10. In addition to cerebral arteriosclerosis, blood–brain barrier dysfunction characterized by ischemic demyelination, gliosis, and selective neuronal necrosis at the capillary level increases the WMH burden10, 21. Similar to WMH, OCNPi shows pathological changes at both the arteriolar and capillary levels. Post-mortem pathology studies of the ischemic oculomotor nerve revealed arteriolar hyalinization and blood-nerve barrier dysfunction, characterized by edema and hypoxia of the endoneurial space9, 22, 23. Altogether, the association between WMH and OCNPi, not silent infarction or CMBs, among biomarkers of cerebral SVD suggests that a common pathophysiology of neurovascular dysfunction associated with both WMH and OCNPi might exist.
A greater proportion of OCNPi patients were taking antithrombotics at diagnosis compared with other OCNP patients in this study. This may be due to the more prevalent underlying diseases in patients with OCNPi. Whether antiplatelet agents could prevent OCNPi has not been elucidated. A study comparing OCNPi events between groups taking and not taking antiplatelet agents is needed to clarify this finding.
This study has some limitations. First, this was a medical record-based retrospective study conducted in two hospitals in South Korea. A prospective study is required to confirm the association between WMH and OCNPi. Second, inherent errors can exist in diagnosing OCNPi, because there is no definitive diagnostic test for OCNPi. All enrolled patients underwent MRI, which had a much wider range than the guidelines suggested: patients aged < 50 years, with history of any type of cancer at any time, with other neurologic signs or symptoms, with pupil-involving or partial third nerve palsy, and has no resolution 3 months after the initial visit24. Owing to the characteristics of the tertiary hospital, we performed additional workups, such as cerebrospinal fluid examination, if needed. Furthermore, the mean follow-up duration of 4.8 years after OCNP was sufficient to observe the patients’ symptoms and change the diagnosis as necessary. However, no direct pathological or neuroimaging evidence has been found. We cannot exclude other possible etiologies such as minor inflammation or demyelination. Since there are only three clinicopathological studies of OCNPi to date, more pathological or imaging studies are needed. Third, due to the heterogeneity of MRI scanners in our hospitals, MR images were obtained with heterogeneous acquisition parameters, although the proportion of 3 T MRI and SWI sequences was comparable between OCNPi and OCNPo.
In conclusion, we demonstrated that WMH is associated with ischemic isolated unilateral OCNP in patients aged ≥ 50 years. Owing to the lack of confirmatory diagnostic tests for OCNPi, the presence of WMH could predict OCNPi at early stage of admission.
Data availability
Data supporting the findings of this study are available from the corresponding author upon reasonable request.
References
Richards, B. W., Jones, F. R. Jr. & Younge, B. R. Causes and prognosis in 4,278 cases of paralysis of the oculomotor, trochlear, and abducens cranial nerves. Am. J. Ophthalmol. 113, 489–496. https://doi.org/10.1016/s0002-9394(14)74718-x (1992).
Danchaivijitr, C. & Kennard, C. Diplopia and eye movement disorders. J. Neurol. Neurosurg. Psychiatry 75(Suppl 4), iv24–iv31. https://doi.org/10.1136/jnnp.2004.053413 (2004).
Choi, K. D. et al. Acquired ocular motor nerve palsy in neurology clinics: A prospective multicenter study. J. Clin. Neurol. 15, 221–227. https://doi.org/10.3988/jcn.2019.15.2.221 (2019).
Chou, K. L. et al. Acute ocular motor mononeuropathies: Prospective study of the roles of neuroimaging and clinical assessment. J. Neurol. Sci. 219, 35–39. https://doi.org/10.1016/j.jns.2003.12.003 (2004).
Tamhankar, M. A. et al. Isolated third, fourth, and sixth cranial nerve palsies from presumed microvascular versus other causes: A prospective study. Ophthalmology 120, 2264–2269. https://doi.org/10.1016/j.ophtha.2013.04.009 (2013).
Akagi, T., Miyamoto, K., Kashii, S. & Yoshimura, N. Cause and prognosis of neurologically isolated third, fourth, or sixth cranial nerve dysfunction in cases of oculomotor palsy. Jpn. J. Ophthalmol. 52, 32–35. https://doi.org/10.1007/s10384-007-0489-3 (2008).
Comer, R. M., Dawson, E., Plant, G., Acheson, J. F. & Lee, J. P. Causes and outcomes for patients presenting with diplopia to an eye casualty department. Eye (Lond) 21, 413–418. https://doi.org/10.1038/sj.eye.6702415 (2007).
Galtrey, C. M., Schon, F. & Nitkunan, A. Microvascular non-arteritic ocular motor nerve palsies-what we know and how should we treat?. Neuroophthalmology 39, 1–11. https://doi.org/10.3109/01658107.2014.963252 (2015).
Weber, R. B., Daroff, R. B. & Mackey, E. A. Pathology of oculomotor nerve palsy in diabetics. Neurology 20, 835–838. https://doi.org/10.1212/wnl.20.8.835 (1970).
Wardlaw, J. M., Smith, C. & Dichgans, M. Small vessel disease: Mechanisms and clinical implications. Lancet Neurol. 18, 684–696. https://doi.org/10.1016/S1474-4422(19)30079-1 (2019).
Fazekas, F., Chawluk, J. B., Alavi, A., Hurtig, H. I. & Zimmerman, R. A. MR signal abnormalities at 1.5 T in Alzheimer’s dementia and normal aging. AJR Am. J. Roentgenol. 149, 351–356. https://doi.org/10.2214/ajr.149.2.351 (1987).
Cao, L., Guo, Y. & Zhu, Z. Effects of hyperhomocysteinemia on ischemic cerebral small vessel disease and analysis of inflammatory mechanisms. Int. J. Neurosci. https://doi.org/10.1080/00207454.2020.1744597 (2020).
Li, J., Zhao, Y. & Mao, J. Y. Association between the extent of white matter damage and early cognitive impairment following acute ischemic stroke. Exp. Ther. Med. 13, 909–912. https://doi.org/10.3892/etm.2017.4035 (2017).
Qu, M. et al. Retinal sublayer defect is independently associated with the severity of hypertensive white matter hyperintensity. Brain Behav. 10, e01521. https://doi.org/10.1002/brb3.1521 (2020).
Wardlaw, J. M. et al. Neuroimaging standards for research into small vessel disease and its contribution to ageing and neurodegeneration. Lancet Neurol. 12, 822–838. https://doi.org/10.1016/S1474-4422(13)70124-8 (2013).
Smith, E. E. et al. Prevention of stroke in patients with silent cerebrovascular disease: A scientific statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 48, e44–e71. https://doi.org/10.1161/STR.0000000000000116 (2017).
Kodsi, S. R. & Younge, B. R. Acquired oculomotor, trochlear, and abducent cranial nerve palsies in pediatric patients. Am. J. Ophthalmol. 114, 568–574. https://doi.org/10.1016/s0002-9394(14)74484-8 (1992).
Zhao, J., Li, Y., Chang, Q., Wang, J. & Sun, H. Diabetic oculomotor nerve palsy displaying enhancement of the oculomotor nerve in the orbit and cavernous sinus on MRI. Eur. Neurol. 84, 246–253. https://doi.org/10.1159/000514100 (2021).
Yang, Y., Lai, C., Yan, F. & Wang, J. Clinical significance of MRI contrast enhancement of the oculomotor nerve in ischemic isolated oculomotor nerve palsy. J. Clin. Neurol. 16, 653–658. https://doi.org/10.3988/jcn.2020.16.4.653 (2020).
Grinberg, L. T. & Thal, D. R. Vascular pathology in the aged human brain. Acta Neuropathol. 119, 277–290. https://doi.org/10.1007/s00401-010-0652-7 (2010).
Ostergaard, L. et al. Cerebral small vessel disease: Capillary pathways to stroke and cognitive decline. J. Cereb. Blood Flow Metab. 36, 302–325. https://doi.org/10.1177/0271678X15606723 (2016).
Asbury, A. K., Aldredge, H., Hershberg, R. & Fisher, C. M. Oculomotor palsy in diabetes mellitus: A clinico-pathological study. Brain 93, 555–566. https://doi.org/10.1093/brain/93.3.555 (1970).
Dreyfus, P. M., Hakim, S. & Adams, R. D. Diabetic ophthalmoplegia; Report of case, with postmortem study and comments on vascular supply of human oculomotor nerve. AMA Arch. Neurol. Psychiatry 77, 337–349 (1957).
Murchison, A. P., Gilbert, M. E. & Savino, P. J. Neuroimaging and acute ocular motor mononeuropathies: A prospective study. Arch. Ophthalmol. 129, 301–305. https://doi.org/10.1001/archophthalmol.2011.25 (2011).
Author information
Authors and Affiliations
Contributions
D.-W.K. and H.N. conceptualized and designed the study. D.-W.K. and S.Y.H. contributed to data preparation and acquisition. D.-W.K. wrote the first draft with the assistance of H.N. D.-W.K., S.Y.H., J.-J.S., and H.N. performed the data analysis and interpretation. D.-W.K., S.Y.H., J.-J.S., and H.N. contributed to critical revision of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kang, DW., Ha, S.Y., Sung, JJ. et al. Cerebral small vessel disease as imaging biomarker predicting ocular cranial nerve palsy of presumed ischemic origin at admission. Sci Rep 12, 12251 (2022). https://doi.org/10.1038/s41598-022-16413-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-16413-x
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.