Abstract
Deep endometriosis (DE) occurs in 15–30% of patients with endometriosis and is associated with concomitant adenomyosis in around 25–49% of cases. There are no data about the effect of the presence of adenomyosis in terms of surgical outcomes and complications. Thus, the aim of the present study was to evaluate the impact of adenomyosis on surgical complications in women with deep endometriosis undergoing laparoscopic surgery. A retrospective cohort study including women referred to the endometriosis unit of a referral teaching hospital. Two expert sonographers preoperatively diagnosed DE and adenomyosis. DE was defined according to the criteria of the International Deep Endometriosis Analysis group. Adenomyosis was considered when 3 or more ultrasound criteria of the Morphological Uterus Sonographic Assessment group were present. Demographical variables, current medical treatment, symptoms, DE location, surgical time, hospital stay and difference in pre and post hemoglobin levels were collected. The Clavien–Dindo classification was used to assess surgical complications, and multivariate analysis was performed to compare patients with and without adenomyosis. 157 DE patients were included into the study; 77 (49.05%) had adenomyosis according to transvaginal ultrasound (TVS) and were classified in the A group, and 80 (50.95%) had no adenomyosis and were classified in the noA group. Adenomyosis was associated with a higher rate of surgical complications: 33.76% (A group) vs. 12.50% (noA group) (p < 0.001). Multivariate analysis showed a 4.56-fold increased risk of presenting complications in women with adenomyosis (CI 1.90–11.30; p = 0.001) independently of undergoing hysterectomy. There was a statistically significant association between the number of criteria of adenomyosis present in each patient and the proportion of patients presenting surgical complications (p < 0.001). Adenomyosis is an independent preoperative risk factor for surgical complications in DE surgery after adjustment for known demographic, clinical and surgical risk factors.
Similar content being viewed by others
Introduction
Adenomyosis is a benign uterine condition defined as the presence of endometrial glands and stroma within the myometrium1. Deep endometriosis (DE) was defined as endometriosis infiltrating the peritoneum by > 5 mm2. For years endometriosis and adenomyosis were related, but they are now mainly considered as separate entities3,4. Deep endometriosis (DE) occurs in 15–30% of patients with endometriosis and is associated with concomitant adenomyosis in around 25–50% of cases5,6.
Advances in pelvic ultrasound provide a high accuracy in the diagnosis of the different forms of endometriosis7 and adenomyosis1,8,9 and allow topographical planning prior to surgery10,11.
Hormone therapy is the first line treatment for DE and adenomyosis in patients not seeking pregnancy12,13. When severe pain persists following medical treatment, or in cases of intestinal or ureteral obstruction, conservative or radical surgical approaches may be necessary depending on the need for fertility preservation14,15. Surgery has been widely demonstrated to improve endometriosis-related symptoms16. Some previous studies have shown that pelvic pain was significantly associated with concomitant adenomyosis in patients with DE17. Moreover, after surgical treatment of DE, pelvic pain and abnormal uterine bleeding (AUB) were significantly more likely to persist with the presence of adenomyosis”18.
Resection of endometriotic lesions is often challenging. Most DE interventions are highly complex and are associated with a significant risk of complications that must be preoperatively taken into account19. Moreover, there is no a reliable preoperative marker to determine the severity of endometriosis for extrapolation to surgical difficulty20. To the best of our knowledge, there are no data about the effect of the presence of adenomyosis in terms of surgical outcomes and complications.
Thus, the aim of the present study was to assess the impact of adenomyosis on the presentation of surgical complications in patients with DE undergoing laparoscopic surgery.
Materials and methods
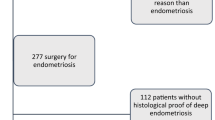
A retrospective cohort study including women referred to the Endometriosis Unit of the Hospital Clinic of Barcelona, who underwent DE surgery from July 2018 to December 2019 was designed.
The study was approved by the Ethical Committee of the Hospital Clinic (Reg: HCB/2019/1152), all research was performed in accordance with relevant guidelines and regulations and informed consent was obtained from all patients.
A preoperative diagnosis of DE and adenomyosis was made by two expert sonographers within 6 months prior to surgery. DE was described according to the fourth step method suggested by the International Deep Endometriosis Analysis (IDEA) group7 with a 2-dimensional and 3-dimensional transvaginal sonography (TVS) using an endovaginal probe (type RIC5-9, Voluson V730 Expert; GE Healthcare, Milwaukee, WI) with previous bowel preparation21. The location and extent of DE was described within the pelvis: rectovaginal septum, torus uterinus, uterosacral ligaments, vaginal fornix, bladder, ureteral and bowel involvement and the mean maximum size for each DE nodule was evaluated22. Adenomyosis ultrasound features according to the criteria of the Morphological Uterus Sonographic Assessment (MUSA) group8, were: asymmetrical thickening, cysts, hyperechoic islands, fan-shaped shadowing, echogenic subendometrial lines and buds, translesional vascularity, irregular junctional zone and interrupted junctional zone. Adenomyosis was diagnosed when at least 3 of the above-mentioned ultrasound features were present according to our hospital protocol.
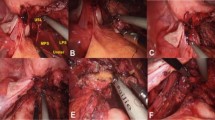
DE surgery was always performed by the same team of skilled endometriosis surgeons and a colorectal surgeon or urologist when needed. In order to standardize all the surgical procedures, they were classified into: adnexal (including salpingectomy, ovarian cystectomy or CO2 laser vaporization and adnexectomy), pelvic (vagina, uterosacral ligaments, torus uterinus and rectovaginal septum), bowel (shaving, discoid or segmental resection), urinary (ureterolysis, bladder nodule excision, nephrectomy) and hysterectomy. Some patients underwent more than one of these procedures.
After preoperative assessment, surgery was performed as follows: first, in cases of hysterectomy, a uterine manipulator (Hohl, Karl Storz, Tuttlingen, Germany) was placed. Surgery was begun with the dissection of rectosigmoid adhesions through the pelvic rim with the opening of the retroperitoneum bilaterally in order to control the entire pathway of the ureters. After that, we proceeded to open and dissect bilateral pararectal spaces to individualize DE nodules, avoiding hypogastric plexus injury; the medial pararectal space was dissected between the mesorectum space, the rectal pillars and the uterosacral ligaments and laterally to the mesoureter creating the lateral para rectal fossa. Dissection of the rectovaginal septum (up to recognizable healthy areolar tissue) allowed vaginal and rectal nodule excision. Then, bowel, ureteral, parametrial, uterine (torus, USL) or vaginal nodules were identified and subsequently removed according to the specific surgical technique. In patients undergoing hysterectomy we then performed the transection of the uterovarian or infundibulopelvic ligaments (depending on bilateral adnexectomy or not), the transection of round ligaments, which had previously been cut to better access to pararectal spaces; caudally, both ureteral tunnels were identified. The uterine vessels were dissected and cut at the cross-point of the ureter. The spaces between the rectum and the endometriotic lesions were previously identified and dissected, which allowed skeletonization and transection of the posterior parametrium. Finally, the cardinal ligaments were transected and colpotomy was made through the uterine manipulator.
If the ovaries were affected by ovarian endometriomas they were drained as a first surgical step (to better prepare space or visualization) and at the end of surgery, after removal of the DE nodules, OMAS were treated by stripping or ablation with CO2 laser (in order to preserve ovarian reserve).
In addition, endometriosis was staged according to the revised-American Society of Reproductive Medicine (r-ASRM) classification score23.
The following demographic data were collected: age, body mass index (BMI), previous endometriosis surgery, infertility, parity, preoperative hormonal treatment and surgical indication. The endometriosis-related symptoms considered were: dysmenorrhea, dyspareunia, dyschezia, dysuria and non-cyclic pelvic pain, using a numerical rating scale (NRS) in which 0 was no pain and 10 unbearable pain. Abnormal uterine bleeding (AUB) was also registered.
Patients were divided into two groups according to the preoperative TVS features: with adenomyosis (A) or without adenomyosis (noA). The main goal of the study was to compare postoperative complications in the two groups according to the Clavien-Dindo (CD) classification24. Differences in demographic characteristics, medical treatment, symptoms, DE location, types of DE surgery, surgical time (minutes), hospital stay (days) and pre and post hemoglobin levels (gr/dl) were also assessed.
Statistical analysis
Statistical analysis was performed using SPSS v 21.0 software (IBM, Armonk, NY, USA). Patient characteristics were described using frequency tables for nominal variables and measures of central tendency and dispersion for continuous variables.
To compare outcomes between the two groups, the Chi-squared or Fisher exact tests was used for categorical data, and continuous variables were compared using independent t-tests or the Mann–Whitney test as appropriate. Bivariate logistic regression analysis was used to determine the factors related to the incidence of surgical complications. All bivariate statistical tests were performed at a significance level of p < 0.05 (two-sided). In order to identify a logistic predictive regression model, clinical and statistical judgment led to the assessment of the following independent variables that were related to complications in the bivariate analysis with a p < 0.15: adenomyosis, surgical time, bowel resection and hysterectomy.
This model explained the probability of the presentation of surgical complications with respect to non-presentation of complications as a function of the variables included. For the multivariate analysis, up to p < 0.10 was illustrated.
Furthermore, we also conducted a subanalysis of the complications between groups considering patients without hysterectomy.
Results
A total of 157 patients undergoing DE surgery were included during the study period. According to TVS, 77 (49.05%) patients had 3 or more adenomyosis criteria and were classified in the A group and 80 (50.95%) patients had less than 3 adenomyosis criteria and were classified in the noA group.
No differences were observed between groups regarding age, BMI and parity (Table 1). A total of 52.86% of women had one or more previous endometriosis surgeries, with no significant differences between groups. Among the 157 patients studied, 130 (82.81%) received preoperative continuous hormonal medical treatment, mainly with oral combined contraceptives, with no significant differences between groups.
Table 1 shows the endometriosis-related symptoms of both study groups. The mean of all NRS scores and the presence of AUB, although not significant, were higher (except for dyschezia) in the group of patients with adenomyosis (group A). The main indication for surgery was pain in both groups (96%) despite receiving medical treatment, with 46.75% (group A) and 35% (group no A) having associated infertility (p 0.495). The main indications for patients undergoing hysterectomy without adenomyosis were: uterine fibroids (9), persistence of abnormal uterine bleeding despite hormonal treatment (6) and patient's preference not to preserve the uterus (3).
The different interventions performed in the two groups are shown in Table 2. All the procedures were performed by laparoscopy with 56 hysterectomies being registered: 38 in the A group and 18 in the noA group (p 0.002). We found parametrial involvement in 5.09% (8/157) of patients (5 A i 3 noA), with no differences between groups and almost 17% of patients were diagnosed with ureteral DE during surgery.
DE was confirmed histologically in all cases as well as the presence of adenomyosis when hysterectomy or uterus-sparing adenomyosis surgery was performed. In 2 out of 18 cases (11.11%) we found incidental small foci of adenomyosis in the histopathologic post-operative analysis of patients in the no A group.
The presence of adenomyosis showed significant differences in the CD complications rates: A group 33.76%, noA group 12.50% (p = 0.001), mean surgical time: A group 231 ± 101 min, noA group 181.08 ± 91.61 min (p = 0.011) and difference in pre-post hemoglobin levels: A group 2.17 ± 1.89 g/dl, and noA group 2.05 ± 1.22 g/dl (p = 0.049). There were no significant differences in length of hospital stay: A group 3.32 ± 3.70 days, noA group 2.75 ± 1.85 days (p = 0.09). Most of the complications were minor (88.8% CD I and II). Sixteen patients presented CD II complications including: anemia requiring blood transfusion (5), urinary infection (3), hematoma of the vaginal vault (4), wound trocar infection (2) and intestinal subocclusion (2) (Table 3). Four patients in the A group, presented major complications (CD III): one rectovaginal fistula, two anastomotic leakages and one bowel obstruction. All patients were recovered at 3–6 months of post-operative follow-up.
Multiple logistic regression showed the following factors to be independently related to the development of surgical complications: adenomyosis, surgical time, bowel resection and hysterectomy. The attributed risk of each factor is shown in Table 4. The risk of presenting complications increased 4.56 times in the A group (CI 1.90–11.30; p = 0.001). Moreover, there was a statistically significant association between the number of criteria of adenomyosis and the proportion of patients presenting a surgical complication (p < 0.001) (Fig. 1).
A higher CD complication rate was also observed in the A group when excluding patients with hysterectomy in both groups (Table 5).
Discussion
The possibility of identifying predictable preoperative features that could modify surgical results would be of great interest25. Previous studies26,27 have already described a higher rate of complications depending on DE severity, although in our patients, the nodule size was not related. The results of our study suggest, for the first time, that the presence of adenomyosis may contribute to increasing the surgical complication rate in DE patients. Furthermore, this increased risk is related to the number of TVS adenomyosis criteria.
While the impact of adenomyosis on surgical results has been assessed in other benign conditions, to our knowledge no previous study has evaluated the impact of adenomyosis on surgical complications in women with DE undergoing laparoscopic surgery28. Previous studies in benign pathology reported an increased rate of bladder and ureteral complications for vaginal hysterectomy in patients with only adenomyosis29 but this was not observed with the laparoscopic approach30. Accurate preoperative imaging assessment has been described in cases of bowel DE, and several previous studies have reported a high accuracy of up to 89.90% and 98.10% in correlating TVS findings with surgical difficulties11,20. These studies are primarily based on ultrasound findings of ovarian mobility and the presence or not of bowel endometriosis but did not specifically included the presence of adenomyosis.
It has been shown that several 2D and 3D TVUS features are associated with adenomyosis; however the importance of each item, the minimum number of items or their combination in the diagnosis of adenomyosis has yet to be defined1,31. Indeed, there is no current consensus on the criteria for the diagnosis of adenomyosis by TVUS. In order to improve accuracy, in our study we considered adenomyosis when 3 of the MUSA criteria were present. Some authors32 also evaluated the diagnostic accuracy of the question mark sign and TVUS uterine tenderness in the diagnosis of adenomyosis, concluding that they are also useful tools.
Due to the surgical complexity of DE procedures, even in expert hands, the rate of complications is still considerable especially when colorectal resection is involved, ranging from 3.40 to 25%25,33,34. The use of endovenous ICG during surgery for rectosigmoid or ureteral endometriosis has recently been proposed to assess blood perfusion of the bowel during anastomosis and the ureter blood supply, in order to guide surgical decisions and avoid complications35,36. In our study, clinically relevant complications (CD type III) were only recorded in 2.54% of cases. These complications were also related to bowel surgeries, similar to previously published data15,33,34,37.
Hysterectomy is considered the surgical treatment of choice for most women with adenomyosis who do not wish to preserve fertility after medical treatment has failed38. Concomitant hysterectomy in the A group was performed in 49.35% vs. 22.50% in the noA group (p = 0.002). An explanation for this high proportion of hysterectomy may be that our hospital is a referral center which receives complex patients, with a high percentage of previous pelvic surgeries (52.86%), and thus, a high number of women are elective for a definitive surgery undergo elective surgery. In the context of DE hysterectomy has been shown to be associated with a longer operative time39 and per se may be an important confounder for the presence of postoperative complications. However, the application of a multivariate model demonstrated that hysterectomy was an independent factor with or without adenomyosis. This was also confirmed in the subanalysis of surgical outcomes between groups performed considering only patients without hysterectomy. Interestingly, the history of previous surgery was not related to a higher risk of complications likely due to our small sample size.
Arena et al.40 reported that factors such as previous endometriosis surgery, the presence of adenomyosis and parametrial location may increase the risk of intraoperative or postoperative complications in cases of ureteral endometriosis surgery. In our study all the factors that could contribute to a higher risk of complications (bowel and urinary surgery, previous endometriosis surgeries and operative time) were adjusted in the multivariate analysis in order to prevent bias.
On the other hand, Van den Bosch et al.9 proposed a new classification system which includes different adenomyosis subtypes, including intrinsic/extrinsic adenomyosis, adenomyosis external and focal adenomyosis in the outer myometrium (FAOM)5,41. Focal adenomyosis commonly affects the external part of the myometrium and may attach the uterus to other structures, hindering surgery and increasing the risk of presenting surgical complications and impairing other surgical outcomes and may be a possible explanation for our study results although a definite reason is unknown. Moreover, the chronic inflammation, proliferation and fibrosis that supports the pathogenesis of adenomyosis1 may be another feasible explanation, creating an inflammatory environment more likely to present an increased risk of surgical complications. Further prospective studies are needed to support this correlation with the type of adenomyosis (since this classification was published after performing the present study) and to find a pathogenic mechanism explaining the clinical presentation of our surgical results.
This study has several strengths: it is the first to describe the impact of adenomyosis on surgical outcomes by conditioning the presentation of a surgical complication. Second, the TVS evaluation was performed by two expert sonographers who have previously demonstrated a high diagnostic TVS accuracy for determining the presence of DE (sensitivity 100%, specificity 96%)21. Moreover, all surgeries were performed by the same team of experienced surgeons in advanced laparoscopy surgery focused on endometriosis.
However, some limitations need to be acknowledged. This was a retrospective, single-center study. In the absence of previous studies, no formal power calculation was performed for sample size determination. The limitation of our small sample size can probably explain why previous surgeries and bowel surgery were not determinant factors in the rate of complications in our study. Secondly, a patient selection bias may be present. The patients were probably more complex since they were referred to the Endometriosis Unit of a tertiary university center and were, therefore, associated with a higher degree of surgical difficulty and may not be comparable with the general population. Another limitation of our study, is that patients were staged according to the r-ASRM classification score that correlates poorly with pain and surgical complexity. Recently, an anatomy-based and user-friendly scoring system that correlates with surgical complexity has been developed by the American Association of Gynecologic Laparoscopists42, and its use should be considered in future studies to predict surgical difficulty and avoid complications.
Conclusion
According to our findings, adenomyosis is an independent preoperative risk factor for surgical complications after adjustment for known demographic, clinical and surgical risk factors. Thus, patients with a preoperative diagnosis of DE and adenomyosis programmed to undergo surgery should be informed about the possible development of surgical complications and ideally attended in a referral center by an expert multidisciplinary endometriosis team. However, further well-designed prospective studies should be carried out in order to confirm these findings taking into account different adenomyosis phenotypes and DE severity, and including patients without hysterectomy.
References
Chapron, C. et al. Diagnosing adenomyosis: An integrated clinical and imaging approach. Hum. Reprod. Update. 26(3), 392–411 (2020).
Koninckx, P. R., Ussia, A., Adamyan, L., Wattiez, A. & Donnez, J. Deep endometriosis: Definition, diagnosis, and treatment. Fertil. Steril. 98(3), 564–571 (2012).
Vannuccini, S. et al. Pathogenesis of adenomyosis: An update on molecular mechanisms. Reprod. Biomed. Online. 35(5), 592–601 (2017).
Maruyama, S., Imanaka, S., Nagayasu, M., Kimura, M. & Kobayashi, H. Relationship between adenomyosis and endometriosis; different phenotypes of a single disease?. Eur. J. Obstet. Gynecol. Reprod. Biol. 253, 191–197 (2020).
Chapron, C. et al. Relationship between the magnetic resonance imaging appearance of adenomyosis and endometriosis phenotypes. Hum. Reprod. 32(7), 1393–1401 (2017).
Lazzeri, L. et al. Preoperative and postoperative clinical and transvaginal ultrasound findings of adenomyosis in patients with deep infiltrating endometriosis. Reprod. Sci. 21(8), 1027–1033 (2014).
Guerriero, S. et al. Systematic approach to sonographic evaluation of the pelvis in women with suspected endometriosis, including terms, definitions and measurements: A consensus opinion from the International Deep Endometriosis Analysis (IDEA) group. Ultrasound Obstet. Gynecol. 48(3), 318–332 (2016).
Van Den Bosch, T. et al. Terms, definitions and measurements to describe sonographic features of myometrium and uterine masses: A consensus opinion from the morphological uterus sonographic assessment (MUSA) group. Ultrasound. Obstet. Gynecol. 46(3), 284–298 (2015).
Van den Bosch, T. et al. Sonographic classification and reporting system for diagnosing adenomyosis. Ultrasound Obstet. Gynecol. 53(5), 576–582 (2019).
Exacoustos, C. et al. Ultrasound mapping system for the surgical management of deep infiltrating endometriosis. Fertil. Steril. 102(1), 143-150.e2 (2014).
Menakaya, U. et al. Performance of ultrasound-based endometriosis staging system (UBESS) for predicting level of complexity of laparoscopic surgery for endometriosis. Ultrasound. Obstet. Gynecol. 48(6), 786–795 (2016).
Taylor, H. S., Kotlyar, A. M. & Flores, V. A. Endometriosis is a chronic systemic disease: Clinical challenges and novel innovations. Lancet 397(10276), 839–852 (2021).
Mabrouk, M. et al. Combined oral contraceptive therapy in women with posterior deep infiltrating endometriosis. J. Minim. Invasive Gynecol. 18(4), 470–474 (2011).
Kho, R. M. et al. Surgical treatment of different types of endometriosis: Comparison of major society guidelines and preferred clinical algorithms. Best Pract. Res. Clin. Obstet. Gynaecol. 51, 102–110 (2018).
Singh, S. S., Gude, K., Perdeaux, E., Gattrell, W. T. & Becker, C. M. Surgical outcomes in patients with endometriosis: A systematic review. Obstet. Gynaecol. Can. 42(7), 881–888 (2020).
Chapron, C., Marcellin, L., Borghese, B. & Santulli, P. Rethinking mechanisms, diagnosis and management of endometriosis. Nat. Rev. Endocrinol. 15(11), 666–682 (2019).
Perelló, M. F. et al. Endometriotic pain is associated with adenomyosis but not with the compartments affected by deep infiltrating endometriosis. Gynecol. Obstet. Invest. 82(3), 240–246 (2017).
Parker, J. D. et al. Persistence of dysmenorrhea and nonmenstrual pain after optimal endometriosis surgery may indicate adenomyosis. Fertil. Steril. 86(3), 711–715 (2006).
Keckstein, J. et al. Recommendations for the surgical treatment of endometriosis. Part 2: Deep endometriosis. Hum. Reprod. Open. 2020(1), 1–25 (2020).
Tompsett, J. et al. Ultrasound-based endometriosis staging system: Validation study to predict complexity of laparoscopic surgery. J. Minim. Invasive Gynecol. 26(3), 477–483 (2019).
Ros, C. et al. Bowel preparation improves the accuracy of transvaginal ultrasound in the diagnosis of rectosigmoid deep infiltrating endometriosis: A prospective study. Minim. Invasive Gynecol. 24(7), 1145–1151. https://doi.org/10.1016/j.jmig.2017.06.024 (2017).
Di Giovanni, A. et al. Combined transvaginal/transabdominal pelvic ultrasonography accurately predicts the 3 dimensions of deep infiltrating bowel endometriosis measured after surgery: A prospective study in a specialized center. J. Minim. Invasive Gynecol. 25(7), 1231–1240. https://doi.org/10.1016/j.jmig.2018.03.003 (2018).
Nevalainen, A. Special contribution. Water Supply. 18(1–2), 298–299 (2000).
Dindo2004. Classification.pdf (2004).
Abrão, M. S., Andres, M. P., Barbosa, R. N., Bassi, M. A. & Kho, R. M. Optimizing perioperative outcomes with selective bowel resection following an algorithm based on preoperative imaging for bowel endometriosis. J. Minim. Invasive Gynecol. 27(4), 883–891 (2020).
Vallée, A. et al. Surgery for deep endometriosis without involvement of digestive or urinary tracts: Do not worry the patients!. Fertil. Steril. 109(6), 1079-1085.e1 (2018).
Gornes, H. et al. Identification of a group with high risk of postoperative complications after deep bowel endometriosis surgery: A retrospective study on 164 patients. Arch. Gynecol. Obstet. 302(2), 383–391. https://doi.org/10.1007/s00404-020-05604-4 (2020).
Struble, J., Reid, S. & Bedaiwy, M. A. Adenomyosis: A clinical review of a challenging gynecologic condition. J. Minim. Invasive Gynecol. 23(2), 164–185. https://doi.org/10.1016/j.jmig.2015.09.018 (2016).
Furuhashi, M., Miyabe, Y., Katsumata, Y., Oda, H. & Imai, N. Comparison of complications of vaginal hysterectomy in patients with leiomyomas and in patients with adenomyosis. Arch. Gynecol. Obstet. 262(1–2), 69–73 (1998).
Nugent, E. W., Orleans M. 304–11.
Harmsen, M. J. et al. Consensus on revised definitions of morphological uterus sonographic assessment (MUSA) features of adenomyosis: Results of a modified Delphi procedure. Ultrasound. Obstet. Gynecol. https://doi.org/10.1002/uog.24786 (2021).
Zannoni, L. et al. Question mark sign and transvaginal ultrasound uterine tenderness for the diagnosis of adenomyosis: A prospective validation. J. Ultrasound. Med. 39(7), 1405–1412 (2020).
Kondo, W. et al. Complications after surgery for deeply infiltrating pelvic endometriosis. BJOG An. Int. J. Obstet. Gynaecol. 118(3), 292–298 (2011).
Darai, E., Ackerman, G., Bazot, M., Rouzier, R. & Dubernard, G. Laparoscopic segmental colorectal resection for endometriosis: Limits and complications. Surg. Endosc. Other Interv. Tech. 21(9), 1572–1577 (2007).
Seracchioli, R., Raimondo, D., Arena, A., Zanello, M. & Mabrouk, M. Clinical use of endovenous indocyanine green during rectosigmoid segmental resection for endometriosis. Fertil. Steril. 109(6), 1135 (2018).
Raimondo, D. et al. Use of indocyanine green for intraoperative perfusion assessment in women with ureteral endometriosis: A preliminary study. J. Minim. Invasive Gynecol. 28(1), 42–49 (2021).
Nicolaus, K. et al. Extensive endometriosis surgery: rASRM and Enzian score independently relate to post-operative complication grade. Arch. Gynecol. Obstet. 301(3), 699–706 (2020).
Ajao, M. O. et al. Persistence of symptoms after total vs supracervical hysterectomy in women with histopathological diagnosis of adenomyosis. J. Minim. Invasive Gynecol. 26(5), 891–896 (2019).
Uccella, S. et al. Impact of endometriosis on surgical outcomes and complications of total laparoscopic hysterectomy. Arch. Gynecol. Obstet. 294(4), 771–778 (2016).
Arena, A. et al. Ureteral endometriosis, the hidden enemy: Multivariable fractional polynomial approach for evaluation of preoperative risk factors in the absence of ureteral dilation. Fertil. Steril. 116(2), 470–477 (2021).
Guo, S. W. The pathogenesis of adenomyosis vis-à-vis endometriosis. J. Clin. Med. 9(2), 485 (2020).
Abrao, M. S. et al. 2021 endometriosis classification: An anatomy-based surgical complexity score. AAGL 28(11), 1941-1950.e1 (2021).
Author information
Authors and Affiliations
Contributions
Author contributions statement: Conception and design of study: M.G., C.d.G. and F.C. Data collection: M.V.-B., I.M., M.T., M.A.M.-Z., L.Q. Data analysis and interpretation: M.G., C.d.G. and F.C. Responsible surgeon and imager: M.G., M.R., C.R. Statistical analysis: M.G., C.d.G. and F.C. Manuscript preparation: M.G., C.d.G., M.A.M.-Z. and F.C. Patient recruitment: M.V.-B., I.M. and M.T.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gracia, M., de Guirior, C., Valdés-Bango, M. et al. Adenomyosis is an independent risk factor for complications in deep endometriosis laparoscopic surgery. Sci Rep 12, 7086 (2022). https://doi.org/10.1038/s41598-022-11179-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-11179-8
This article is cited by
-
Understanding Ultrasound Features that Predict Symptom Severity in Patients with Adenomyosis: a Systematic Review
Reproductive Sciences (2024)
-
Bowel wall thickness measured by MRI is useful for early diagnosis of bowel endometriosis
European Radiology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.