Abstract
Our aim was to assess effects of breast-feeding (BF) in the association between large-for-gestational age (LGA) and body mass index (BMI) trajectories on childhood overweight from 1 to 4 years old. A total of 1649 healthcare records of mother–child pairs had detailed records of feeding practices and were included in this retrospective cohort study. Data were available in Medical Birth Registry of Xiamen between January 2011 and March 2018. Linear and logistic regression models were used to access the difference between BF and no-BF group. For offspring were LGA and BF was significantly associated with a lower BMI Z-score from 1 to 4 years old after adjustment confounders in Model 1 to 3 [difference in BMI Z-score in Model 1: estimated β: −0.07 [95%CI: −0.13 to −0.01]; Model 2: estimated β: −0.07 (−0.13 to −0.004); Model 3: estimated β: −0.06 (−0.12 to −0.001); P = 0.0221, 0.0371, 0.0471]. A significantly lower risk of childhood overweight was observed in Model 1 [odd ratio (OR): 0.85 (95%CI, 0.73 to 1.00)], P = 0.0475) with adjustment for maternal pre-pregnancy BMI. Furthermore, Model 2 and Model 3 showed LGA-BF infants had a lower risk for childhood overweight then LGA-no-BF infants [OR: 0.87 and 0.87 (95%CI, 0.73 to 1.03; 0.74 to 1.03)], however, there was no statistical significance (P = 0.1099, and 0.1125)]. BF is inversely related to BMI Z-score and risk for overweight in children were LGA from 1 to 4 years old. Adjustment for maternal pre-pregnancy BMI, the protective association between BF and childhood overweight was more significant.
Similar content being viewed by others
Introduction
The prevalence of obesity in children have markedly increased in most countries over the past four decades1. Children overweight and obesity prevalence has increased from less than 3% in 1985 to 20.5% in 20142. The causes of childhood obesity are multifactorial, such as sedentary behavior, changes in lifestyle, excessive energy intake3. Studies indicate that pre-pregnancy body mass index (BMI) is established risk factors for having large-for-gestational age (LGA) infants4,5,6. Meanwhile, LGA infants are more likely to be overweight or obese in childhood7.
On the other hand, some studies have shown that obesogenic environment from birth to 2 years old can affect infant development resulting in a higher risk of obesity8,9. Obesity developed during this period can enhance risk of excess weight gain over one’s lifetime10. Additionally, a study revealed that 11.7% of the children were obese by formula-feeding before 2 years of age compared to 5.6% of children were breast-feeding (BF)11. As well, a meta-analysis demonstrated that the children taken in BF were 15% less likely to become overweight as compared to non-BF, like formula-feeding and mixed-feeding12.
Furthermore, studies detecting above associations have evaluated child’s BMI, but single BMI cannot account for changes in BMI. A study showed that BMI Z-score trajectory overcomes this limitation, and can be compared throughout ages13. Besides, the National Institutes of Health highlighted the exploration of these associations as a major research direction14. Thus, the aim of this study was to further confirm if children’s BMI Z-score trajectory and childhood overweight from 1 to 4 years of age is associated with infant feeding practices (BF), based on all children were LGA status, in a longitudinal study of Chinese children.
Materials and methods
Study design and study population
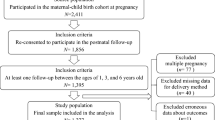
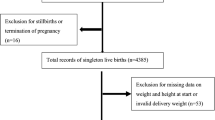
We conducted a population-based retrospective study using the healthcare records data were from the Medical Birth Registry in Xiamen system, China, between January 2011 and March 2018. This was a registration system established in 2007 in Xiamen based on a compulsory notification of all live and stillbirths from 12 weeks’ gestation onward. This study was approved by the ethics committee of the First Affiliated Hospital of Xiamen University (KYH2018-007) and conducted in accordance with the rules of the Declaration of Helsinki of 1975, revised in 2013.
A total of 1649 mother–child pair healthcare records were available. All women over the age of 18 performed a 75-g oral glucose tolerance test (OGTT) between 24 and 28 weeks of gestation. We excluded the mother–child pairs with lacking mother’s weight or height data, missing children’s birth weight data, multiple births, premature infants, gestational age < 37 weeks, medical history of diabetes, and fasting glucose level ≥ 7.0 mmol/L before 12 gestational weeks due to the likelihood of having underdiagnosed diabetes pre-pregnancy. The inclusion criteria were as follows: an OGTT was performed between 24 and 28 weeks of gestation; gestational age ate delivery ≥ 37 weeks, with no major neonatal malformations or fetal/neonatal death; and the offspring was followed-up through 4 years of age.
Data source and linkage
The data source and linkage were referred as our previous published article4. All women in Xiamen are registered at their community health centers when they get pregnant, and were then referred to a secondary hospital or a tertiary hospital for healthcare from the 32nd gestational week till delivery. All children were given health examinations at birth (< 3 days afterbirth), and annual examination starting at age 1 until age 4. The data was available for 4 years. Women and children were linked by individual record linkages to the Xiamen citizenhealth information system using the person-unique identification number assigned to each Xiamen citizen. Every child was also linked to his/her biological mother’s maternal identification number.
Child feeding practices
BF was referred as exclusive breastfeeding in this study. The definitions referred to World Health Organization15 as follows: BF while giving no other food or liquid, not even water, with the exception of drops or syrups consisting of vitamins, mineral supplements or medicines. It is measured by following open-ended questions to the parents: ‘For how many months did you breast-feed him/her?’ and ‘How old was your child when you began feeding him/her formula?’16.
Variables definition
Large for gestational age
LGA was defined as birth weight was above 90 percentile for gestational age, according to gestational age and gender-specific intergrowth-21st curves17.
Gestational diabetes mellitus
Gestational diabetes mellitus (GDM) cases were identified by conducting OGTT at 24–28 weeks’ gestation. According to the 2014 National Health and Family Planning Commission of the People’s Republic of China criteria, after a 75 g glucose load, pregnant women would be considered to have GDM if one of the following plasma glucose values was met or exceeded: 0 h, 5.1 mmol/L; 1 h, 10.0 mmol/L; or 2 h, 8.5 mmol/L18.
Maternal pre-pregnancy BMI classification
Mother’s height and weight were measured in light indoor clothing and without shoes by trained practitioners in community health centers using standardized protocol during pregnancy. Maternal pre-pregnancy BMI was calculated by medical records of self-reported height and weight. BMI was calculated as weight (kg)/height2 (m2). Referring to WHO criteria19, the pre-pregnancy BMI was classified into four groups as follows: underweight (< 18.5 kg/m2), normal (18.5–24.9 kg/m2), overweight (25.0–29.9 kg/m2), and obese (≥ 30.0 kg/m2).
Other maternal and offspring covariates
The maternal and offspring covariates quote from our previous study4. Maternal and offspring information were from the Medical Birth Registry in Xiamen system, which included maternal information such as maternal age, education, weight of pre-pregnancy, occupation, first visit date, numbers of pregnancy/infants, last menstrual period, expected delivery date, smoking habits, alcohol use, medical history of diseases, family history of diseases, hypertension history, pregnancy reactions, and labour status. Furthermore, GDM, gestational weight gain, gestational age at delivery, height, weight, blood pressure, fasting glucose, gynaecological examinations, ultrasonography, gestational diabetes screening results, other lab tests results, complications during pregnancy, and pregnancy outcomes were also included in the Medical Birth Registry in Xiamen system. Medical Birth Registry in Xiamen system also included information from newborns to preschool-aged children on date of birth, sex, gestational age at birth, weight, Apgar score, names of the child and his/her parents, family history of diseases, date of examination, weight, height, number of teeth, and blood pressure. Meanwhile, this study is a longitudinal study, attrition were inevitable as more waves of data were contained.
Offspring measurements and data transformations
During health examination, children’s height and weight was measured by a trained clinician. Before 2 years old, we measured children’s height as length and as standing height to the nearest 0.1 cm after age 2 measured a wall-mounted stadiometer. Body weight was measured in kilograms using regularly calibrated electronic scales. Children were classified into two groups according to LGA and feeding practices status: children born to LGA-BF and children born to LGA-no-BF. BMI Z-score for age was used to present the trajectory tracking of offspring BMI. We calculated age-adjusted z-score of childhood BMI referred to Chinese reference growth charts20. Childhood overweight was defined as a BMI at or above the 85th percentile and below the 95th percentile, and obesity was defined as a BMI at or above the 95th percentile20. Birth weight was transformed into a Z-score21.
Informed consent
The ethics committee of the First Affiliated Hospital of Xiamen University (KYH2018-007) approved of the waiver for the informed consent because this was a retrospective study.
Statistical analysis
Mean [standard deviation (SD)] are showed for continuous variables, n (%) were presented as and categorical variables. To evaluate our hypothesis that LGA and BF were associated with offspring BMI growth trajectories, we performed several analyses. Mean BMI Z-score were visually compared in yearly time intervals between LGA-BF and LGA-no-BF. We used linear and logistic regression models to assess the differences in offspring’s anthropometric outcome (BMI Z-score, and overweight/obese) in yearly time intervals between infants that were breastfed as compared to those who were not breastfed. Between BF exposure and anthropometric outcomes measured from 1 to 4 years of age were performed through mixed-effects regression models with random intercepts, accounting for the correlation between repeated observations within subjects. Three multivariable-adjusted models were performed: Model 1 adjusted for maternal pre-pregnancy BMI (continuous); Model 2 was further adjusted for Model 1 + maternal age before pregnancy (continuous), sex (discontinuous), education (high school and above, middle school and below); and Model 3 was adjusted for Model 2 + GDM (yes/no). In addition, we evaluated potential effect modification by modeling the cross-product term of the stratifying variable with LGA-BF. Statistical significance was two-tailed with a P-value < 0.05. All calculations were carried out using SAS 9.4 (SAS Institute Inc, Cary, North Carolina, USA).
Statement of ethics
This study was approved by the ethics committee of the First Affiliated Hospital of Xiamen University (KYH2018-007) and conducted in accordance with the rules of the Declaration of Helsinki of 1975, revised in 2013. Informed consent was not required because this was a retrospective study.
Results
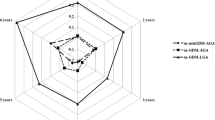
A total of 1649 mother–child pair healthcare records were available, including 676 children exposed to LGA-BF and 973 offspring exposed to LGA-no-BF. 1613 children were detailedly recorded food practices status at 1 year of age, 646 offspring with BF, 967 children with formula-feeding (FF) or mixed-feeding. Characteristics of participants based on BF status are shown in Table 1. Furthermore, we found that no-BF offspring had a higher BMI Z-score at 3 years of age; however, this relationship was not statistically significant (P > 0.05). Additionally, women were in the LGA-no-BF group had slightly higher 1-h and 2-h plasma glucose (P < 0.01). The characteristics of mothers and their offspring were similar between the two groups (Table 1). In order to assess BMI Z-score trajectory of offspring who were LGA-BF from 1 to 4 years of age, we adjusted some variates, as showed in Table 2. In model 1, 2, and 3, BMI Z-score trajectory of offspring were LGA-BF without significant difference at 1 year, 2 years, and 4 years of age (all P > 0.05). Interestingly, only at 3 years of age, a higher BMI Z-score trajectory presented to LGA-no-BF group in any model (all P < 0.05). The specific tendency chart shown in Fig. 1.
BMI Z-score trajectories of children were LGA-BF and LGA-no-BF from 1 to 4 years in different models. LGA large-for-gestation age, BMI body mass index, BF breast-feeding. Model 1, adjusted for maternal pre-pregnancy BMI; Model 2, adjusted for covariates in Model 1 + maternal age, education, and child sex; Model 3, adjusted for covariates in Model 2 + gestational diabetes melitus.
In the longitudinal study (Table 3), we found a significant association between LGA-BF and offspring BMI Z-score. LGA-BF offspring had a significantly lower BMI Z-score across the 4 years after adjusted covariates in Model 2 and Model 3 (difference in BMI Z-score estimated β: −0.07 and −0.06 [95% conference interval (CI), −0.13 to −0.004; −0.12 to −0.001], P = 0.0371; 0.0471) and a lower trend risk of overweight but not statistically significant (odds ratio (OR):0.87 and 0.87 [95%CI, 0.73 to 1.03; 0.74 to 1.03], P = 0.1099; 0.1125). However, adjustment for pre-pregnancy BMI in Model 1, offspring were BF was related with a lower BMI Z-score at 1 year, 2 years, 3 years, and 4 years of age (difference in BMI Z-score: 0.07 [95%CI, −0.13 to −0.01]; P = 0.0221) and a lower risk trend for overweight with statistically significant (OR, 0.854 [95%CI, 0.73 to 1.00]; P = 0.0475).
Discussion
The aim of this population-based study was to assess the relative contribution of LGA and BF on the risk of being overweight in offspring. In a cohort of 1,649 children, the mean BMI Z-score was higher for offspring were LGA-no-BF aged 1 years, 3 years, and 4 years (unadjusted covariates). Additionally, the BMI Z-score of children were LGA-BF was only significant lower at 3 years old in adjusted models. Furthermore, the longitude study indicated that a significant lower risk for BMI Z-score difference of offspring who were LGA-BF from 1 to 4 years old. Offspring were LGA-BF had a significantly low risk trend for overweight in Model 1 with adjustment for maternal pre-pregnancy BMI. However, this association was no significance in Model 2 and Model 3 with adjustment for maternal pre-pregnancy BMI + other covariates.
Since 2007, China has adopted more breastfeeding interventions to promote breastfeeding. Maternity leave has been increased to 180 days in recent years. A meta-analysis22 showed that “any breastfeeding” rates are from 63.41 to 92.93% at six months among 2007 and 2018. Meanwhile, the “exclusive breastfeeding” rates at six months were between 17.87 and 58.50%. “any breastfeeding” and “exclusive breastfeeding” rate are all improved in the most recent years.
The estimates in the current study revealed that LGA-BF reduced the risk of childhood overweight by 15.0% compared with LGA-no-BF. A meta-analysis comparing BF with FF infants found a 15% decrease in the odds of childhood overweight12. The American Academy of Pediatrics (AAP) stated in 2012 that ‘although complex factors confound studies of obesity, there is a 15–30% reduction in adolescent and adult obesity rates if any BF occurred in infancy compared with non-BF’23. Meanwhile, some studies showed that BF is a significant protective factor against obesity in offspring24,25. However, this remains controversial. Some studies revealed there was no association between BF and childhood obesity 26,27. The reason why there were different results may be attributed to the limited number of included participants, neither this study nor above two studies did a subgroup analysis for feeding duration.
On the other hand, the AAP revealed any BF reduced the risk of otitis media by 23% and of gastroenteritis by 64% 23, further, exclusive BF for > 6 months reduced the risk of upper respiratory tract infection by 63%23. One study showed BF had higher cognitive function and better scores throughout childhood and adolescence compared with no-BF28. The Dutch medical institutions found a ‘protective effect of exclusive BF on asthma, atopic dermatitis, and wheezing in early childhood for at least 4 months29. Besides, independent studies have shown that FF also leads to higher morbidity and mortality in western countries30,31.
Possible mechanisms linking BF to lower risk for childhood overweight or obesity are not well demonstrated. Breast milk included some bioactive components, such as leptin, adiponectin, insulin-like growth factor-1, and ghrelin, may play a protective role in future offspring overweight or obesity32,33. In addition. The quantity of energy and protein in breast milk is lower than that in formula feeding, and the elevated protein intake of formula in infancy would result in rapid growth of adipose34,35. A longitudinal study indicated a significant relation between early protein intake and later BMI, showing that children have a higher protein intake early may enhance the risk of later overweight or obesity36. Meanwhile, animal researches were also found that protein intake played a key role in body composition and glucose metabolism in later life37,38. These findings might provide plausible explanations for a protective role of BF against overweight or obesity.
Interestingly, we found that the BMI Z-score of children were LGA-BF was significant lower at 3 years old with adjustment for GDM. This phenomen may be contributed to maternal GDM. Our previous study4 eluciated that GDM have effect on child overweight or obestiy. Meanwhile, maternal pre-pregnancy BMI may play a key role in childhood overweight for children were LGA-BF. Many studies found that pre-pregnancy BMI was significant for later childhood overweight or obesity39,40,41. Furthermore, our another study was also indicated that the association between offspring were LGA and born to maternal GDM and childhood overweight was attenuated after adjusting for pre-pregnancy BMI4.
Our study is contemporary, population-based, and results are therefore generalizable. However, the current study still has several limitations. First, this study is a longitudinal study, attrition was inevitable as more waves of data was contained. Second, this study lacked of data on child BMI and BF for all children. Third, reference to measurement error, the outcomes measure, child BMI Z-score could not reflect body fat mass, particularly in young children. Fourth, the no-BF data were not classified as categories due to lacking detailed information of nutrition of breastfeeding such as formula and complementary food. Finally, Considering direct benefits of breastfeeding, the effect may be mainly mediated by higher prevalence of other beneficial lifestyle habits among the these families. Although there are some misclassifications, it is likely to be nondifferentiated and makes our outcomes towards to null. Additionally, adjustments were performed for covariates, thus figuring out the effects of other variables in the reported associations.
Conclusion
This population-based retrospective study shown that BF is inversely related to risk of overweight or obesity for offspring were LGA who aged 1 to 4 years. Additionally, the association was more significant after adjusting maternal pre-pregnancy BMI. These results suggest that it is important to promote and encourage pregnancy women to breastfeed children. Meanwhile, further research should analyze components of breast milk and feeding practices of mothers with LGA. Furthermore, pregnancy women should focus on pre-pregnancy BMI that might have long-term effects on childhood overweight or obesity. It is significant to explore and develop prenatal interventions that highlight childhood overweight or obesity prevention from an early stage.
Change history
19 April 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41598-022-10363-0
Abbreviations
- BF:
-
Breast-feeding
- FF:
-
Formula-feeding
- LGA:
-
Large-for-gestational age
- BMI:
-
Body mass index
- MBRX:
-
Medical Birth Registry in Xiamen
- OGTT:
-
Oral glucose tolerance test
- GDM:
-
Gestational diabetes mellitus
- AAP:
-
American Academy of Pediatrics
- CI:
-
Confidence interval
- OR:
-
Odd ratio
- SD:
-
Standard deviation
References
Collaboration NCDRF. Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: A pooled analysis of 2416 population-based measurement studies in 128.9 million children, adolescents, and adults. Lancet 390 (10113), 2627–2642 (2017).
Dong, Y. et al. Economic development and the nutritional status of Chinese school-aged children and adolescents from 1995 to 2014: An analysis of five successive national surveys. Lancet Diabetes Endocrinol. 7(4), 288–299 (2019).
Sun, X. et al. A 3-year longitudinal study of the association of physical activity and sedentary behaviours with childhood obesity in China: The childhood obesity study in China mega-cities. Pediatr. Obes. 2020, e12753 (2020).
Chen, Y. L. et al. Adverse pregnancy outcomes on the risk of overweight offspring: A population-based retrospective study in Xiamen, China. Sci. Rep. 10(1), 1549 (2020).
Tyrrell, J. et al. Genetic evidence for causal relationships between maternal obesity-related traits and birth weight. JAMA 315(11), 1129–1140 (2016).
Kc, K., Shakya, S. & Zhang, H. Gestational diabetes mellitus and macrosomia: A literature review. Ann. Nutr. Metab. 66(Suppl 2), 14–20 (2015).
Baird, J. et al. Being big or growing fast: Systematic review of size and growth in infancy and later obesity. BMJ 331(7522), 929 (2005).
Gluckman, P. & Hanson, M. Developmental and epigenetic pathways to obesity: An evolutionary-developmental perspective. Int. J. Obes. (Lond) 32, S62–S71 (2008).
Anzman, S. L., Rollins, B. Y. & Birch, L. L. Parental influence on children’s early eating environments and obesity risk: Implications for prevention. Int. J. Obes. (Lond.) 34, 1116–1124 (2010).
Reilly, J. J. et al. Early life risk factors for obesity in childhood: Cohort study. BMJ 330(7504), 1357 (2005).
Gibbs, B. G. & Forste, R. Socioeconomic status, infant feeding practices and early childhood obesity. Pediatr. Obes. 9(2), 135–146 (2014).
Weng, S. F., Redsell, S. A., Swift, J. A., Yang, M. & Glazebrook, C. P. Systematic review and meta-analyses of risk factors for childhood overweight identifiable during infancy. Arch. Dis. Child 97(12), 1019–1026 (2012).
Ziyab, A. H., Karmaus, W., Kurukulaaratchy, R. J., Zhang, H. & Arshad, S. H. Developmental trajectories of Body Mass Index from infancy to 18 years of age: Prenatal determinants and health consequences. J. Epidemiol. Commun. Health 68(10), 934–941 (2014).
Lumeng, J. C., Taveras, E. M., Birch, L. & Yanovski, S. Z. Prevention of obesity in infancy and early childhood: A National Institutes of Health workshop. JAMA Pediatr. 169(5), 484–490 (2015).
WHO. Infant and Young Child Feeding: A Tool for Assessing National Practices, Policies and Programmes. https://www.who.int/nutrition/topics/global_strategy/en/. Accessed 5 July 2020.
Salahuddin, M., Perez, A., Ranjit, N., Hoelscher, D. M. & Kelder, S. H. The associations of large-for-gestational-age and infant feeding practices with children’s body mass index z-score trajectories: The Early Childhood Longitudinal Study, birth cohort. Clin. Obes. 7(5), 307–315 (2017).
Villar, J. et al. International standards for newborn weight, length, and head circumference by gestational age and sex: The Newborn Cross-Sectional Study of the INTERGROWTH-21st Project. Lancet 384(9946), 857–868 (2014).
Yan, B. et al. High, but stable, trend in the prevalence of gestational diabetes mellitus: A population-based study in Xiamen, China. J. Diabetes Invest. 10(5), 1358–1364 (2019).
Lim, Y. & Boster, J. Obesity and Comorbid Conditions (StatPearls, 2022).
Li, H., Ji, C. Y., Zong, X. N. & Zhang, Y. Q. Body mass index growth curves for Chinese children and adolescents aged 0 to 18 years. Zhonghua Er Ke Za Zhi 47(7), 493–498 (2009).
Yang, J. L. A., Uusitalo, U. M., Lynch, K. F., Veijola, R. & Winkler, C. E. A. Prevalence of obesity was related to HLA-DQ in 2–4-year-old children at genetic risk for type 1 diabetes. Int. J. Obes. 38, 1491–1496 (2014).
Li, Q., Tian, J., Xu, F. & Binns, C. Breastfeeding in China: A review of changes in the past decade. Int. J. Environ. Res. Public Health 17, 21 (2020).
Section on B. Breastfeeding and the use of human milk. Pediatrics 129 (3), e827–841 (2012).
Yan, J., Liu, L., Zhu, Y., Huang, G., & Wang, P.P. The association between breastfeeding and childhood obesity: A meta-analysis. BMC Public Health 14, 1267 (2014).
Arenz, S., Ruckerl, R., Koletzko, B. & von Kries, R. Breast-feeding and childhood obesity–A systematic review. Int. J. Obes. Relat. Metab. Disord. 28(10), 1247–1256 (2004).
Burdette, H. L., Whitaker, R. C., Hall, W. C. & Daniels, S. R. Breastfeeding, introduction of complementary foods, and adiposity at 5 y of age. Am. J. Clin. Nutr. 83(3), 550–558 (2006).
Gillman, M. W. et al. Developmental origins of childhood overweight: Potential public health impact. Obesity (Silver Spring) 16(7), 1651–1656 (2008).
Anderson, J. W., Johnstone, B. M. & Remley, D. T. Breast-feeding and cognitive development: a meta-analysis. Am. J. Clin. Nutr. 70(4), 525–535 (1999).
Nutrition ECo, Agostoni, C., Braegger, C. et al. Breast-feeding: A commentary by the ESPGHAN Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 49 (1), 112–125 (2009).
Bowatte, G. et al. Breastfeeding and childhood acute otitis media: A systematic review and meta-analysis. Acta Paediatr. 104(467), 85–95 (2015).
Kristensen, K. et al. Caesarean section and hospitalization for respiratory syncytial virus infection: A population-based study. Pediatr. Infect. Dis. J. 34(2), 145–148 (2015).
Zheng, J. S. et al. Exclusive breastfeeding is inversely associated with risk of childhood overweight in a large Chinese cohort. J. Nutr. 144(9), 1454–1459 (2014).
Assuncao, M. L., Ferreira, H. S., Coutinho, S. B., Santos, L. M. & Horta, B. L. Protective effect of breastfeeding against overweight can be detected as early as the second year of life: A study of children from one of the most socially-deprived areas of Brazil. J. Health Popul. Nutr. 33(1), 85–91 (2015).
Skledar, M. T. & Milosevic, M. Breastfeeding and time of complementary food introduction as predictors of obesity in children. Cent. Eur. J. Public Health 23(1), 26–31 (2015).
van Rossem, L. et al. Is the association of breastfeeding with child obesity explained by infant weight change?. Int. J. Pediatr. Obes. 6(2–2), e415–e422 (2011).
Rolland-Cachera, M. F., Deheeger, M., Akrout, M. & Bellisle, F. Influence of macronutrients on adiposity development: A follow up study of nutrition and growth from 10 months to 8 years of age. Int. J. Obes. Relat. Metab. Disord. 19(8), 573–578 (1995).
Desai, M. & Hales, C. N. Role of fetal and infant growth in programming metabolism in later life. Biol. Rev. Camb. Philos. Soc. 72(2), 329–348 (1997).
Burns, S. P. et al. Gluconeogenesis, glucose handling, and structural changes in livers of the adult offspring of rats partially deprived of protein during pregnancy and lactation. J. Clin. Invest. 100(7), 1768–1774 (1997).
Ruiz, M. et al. Impact of low maternal education on early childhood overweight and obesity in Europe. Paediatr. Perinat. Epidemiol. 30(3), 274–284 (2016).
Birbilis, M., Moschonis, G., Mougios, V., Manios, Y., Healthy Growth Study G. Obesity in adolescence is associated with perinatal risk factors, parental BMI and sociodemographic characteristics. Eur. J. Clin. Nutr. 67 (1), 115–121 (2013).
Rooney, B. L., Mathiason, M. A. & Schauberger, C. W. Predictors of obesity in childhood, adolescence, and adulthood in a birth cohort. Matern. Child Health J. 15(8), 1166–1175 (2011).
Acknowledgements
We would like to thank all the doctors, nurses, technicians, and patients involved in their dedication to the study.
Funding
This work was supported by the National Natural Science Foundation of China (grant numbers: 0070/K16A8035, 0070/K1615020).
Author information
Authors and Affiliations
Contributions
Y.L.C. and L.L.H. designed and performed the data analysis and drafted the paper. W.J.S., T.W., F.P.L., and Z.C. conceived this study. B.K.H., L.Y.W., and H.Q.S. collected data. X.L.S. and X.J.L. critically reviewed the paper for important intellectual content. All authors contributed significantly to the data analysis and reviewed and approved the final paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: The original version of this Article contained errors in the Title.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, Y., Han, L., Su, W. et al. The impact of breastfeeding on childhood obesity in children that were large-for-gestational age: retrospective study from birth to 4 years. Sci Rep 12, 4226 (2022). https://doi.org/10.1038/s41598-022-08275-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-08275-0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.