Abstract
There is growing evidence that environmental noise exposure could increase the risk of atherothrombotic events, including acute myocardial infarction (MI). We analysed the burden of environmental noise on atherothrombotic risk in MI patients. From the RICO survey, 879 consecutive MI patients included from 2004 to 2008 and living in an urban unit of > 237,000 inhabitants were analysed. Atherothrombotic risk was calculated using the TRS-2P score. TRS-2P categories were split into low (TRS-2P = 0/1) (40.8%), medium–low (TRS-2P = 2) (25.7%), medium–high (TRS-2P = 3) (21.8%) and high risk (TRS-2P ≥ 4) (11.6%). Noise exposure was associated with atherothrombotic risk, with the LAeq,24 h (OR (95% CI): 1.165 (1.026–1.324)) and Lnight (OR (95CI): 1.157 (1.031–1.298)), for each 10 dB(A) increase. After adjustment, noise exposure remained a predictor of atherothrombotic risk, with LAeq,24 h (OR (95% CI): 1.162 (1.011–1.337)) and with Lnight (OR (95% CI): 1.159 (1.019–1.317)). The relationship with transportation Lnight was significant for men (OR (95% CI): 1.260 (1.078–1.472)) but not for women (OR (95% CI): 0.959 (0.763–1.205)). We found a significant association between residential traffic noise exposure and atherothrombotic risk in men but not in women. These results could have major consequences for secondary prevention.
Similar content being viewed by others
Introduction
Environmental noise has a major impact on health, especially in urban areas where people are continuously exposed to noise from road traffic, railways, and aircrafts. A growing body of evidence has led the World Health Organization (WHO) to declare that exposure to environmental noise can exert non-auditory effects including reduced well-being and quality of life, annoyance, adverse birth outcomes, cognitive impairments, sleep disturbance, metabolic abnormalities, and poorer mental health1. In addition, the WHO estimates that the number of DALYs (disability-adjusted life-years) lost as a result of environmental noise in western Europe ranges from 1 to 1.6 million2.
Epidemiological studies have also suggested that noise exposure could be associated with an increased risk of cardiovascular (CV) disease including acute myocardial infarction (MI)3, and that this association could be even more pronounced in patients with coronary artery disease (CAD). A recent meta-analysis commissioned by the WHO concluded that, from a threshold of 50 dB, road-traffic noise increases the incidence of CAD by 8% per 10 dB(A) (Lden) (A). This degree of risk, obtained from 7 longitudinal studies, was rated as a high level of evidence4. However, only few studies have investigated the specific burden of environmental noise exposure on atherothrombotic risk. Moreover, it remains unclear whether exposure to transportation noise at home could have sex-specific effects since few studies have assessed whether there is a gender-based difference in the effect of noise exposure. In an experimental study, Beheshti et al. found that women experience more annoyance than men when exposed to low-intensity noise5. Conversely, men might be more disturbed by traffic noise than women during sleep6. An association between hypertension and residential aircraft noise exposure was observed only in men7,8. On the contrary, Babisch et al. found no sex-based differences in the association between traffic noise and CV risk9.
In the present study, using the database of a regional registry for acute MI and supported by environmental prediction models, we aimed to analyse the burden of residential environmental noise on atherothrombotic risk and to assess the potential differences between men and women.
Methods
Study population
The population of this study came from the RICO (obseRvatoire des Infarctus de Côte-d'Or). This ongoing French regional survey prospectively collects data from consecutive patients hospitalized for acute MI in 6 hospitals of one eastern region of France, since 1 January 2001, as previously described10,11. Each new episode of MI leads to a new entry in the RICO, so a patient can be registered several times in case of recurrence. Research protocol was performed in accordance with the Declaration of Helsinki. Experimental protocol was approved by ethics committee from CHU Dijon Bourgogne and informed consent was obtained from all subjects.
Patients who were admitted for MI for the first time between 1 January 2004 and 31 December 2008 were eligible for the present study (Fig. 1). Inclusion criteria were: living in the urban unit of Dijon (15 towns and 237,000 inhabitants), a known and precise home address at the time of the MI episode. Patients living in an area exposed to the aircraft noise from the military air base of Dijon (2 towns, 200 inhabitants) were excluded because noise data were not available. According to the guidelines of the EU directive, a 5-year period (2004–2008) was selected for the environmental prediction models used for noise exposure.
Medical data
Clinical, biological and demographic data were collected at hospital admission, including risk factors, comorbidities, chronic medications, heart failure (HF) assessed by Killip class, and hemodynamic parameters. An echocardiography was performed < 12 h after admission to measure left ventricular ejection fraction, as previously reported10,11.
The Thrombolysis in Myocardial Infarction Risk Score for Secondary Prevention (TRS-2P) was used to quantify atherothrombotic risk12. TRS-2P is calculated by the arithmetic sum of 9 variables associated with atherothrombotic risk: age ≥ 75 years, diabetes mellitus, hypertension, current smoking, peripheral artery disease (PAD), prior stroke, prior coronary artery bypass grafting (CABG), history of HF, and renal failure (estimated glomerular filtration rate < 60 mL/min) (supplementary Fig. 1). TRS-2P was developed in a large population with atherothrombosis to predict recurrent CV events12 and has been validated in various populations to characterize long-term risk after recent acute MI13,14. The TRS-2P score ranges from 0 to 9 and was split for more risk classification relevance, into 4 classes of atherothrombotic risk namely: low-risk (TRS-2P = 0/1), medium–low-risk (TRS-2P = 2), medium–high-risk (TRS-2P = 3) and high-risk (TRS-2P ≥ 4).
Environmental noise exposure
According to the European directive 2002/49/EC15, environmental noise prediction models were used to assess the outdoor residential exposure of each study patient; these emission-propagation models were elaborated with MITHRA-SIG v3.7 (Geomod/CSTB) software. The models were validated by means of several measurement campaigns16,17. Noise levels were calculated for the residential building, in front of each façade and at each floor. They were then linked to the patient through their self-declared home address at the time of their MI. The address of each patient at the time of their MI was carefully gathered and verified to ensure that the assigned building corresponded to the right exposure. For each patient, the average building noise levels in front of the entire façade were calculated. Multiple noises sources were considered: rail traffic, road traffic, and pedestrian traffic. Four indices were computed: road traffic noise and the railway noise (considering only one source, i.e. road or railway respectively), transportation noise (combining road and railway sources), and global noise (combining all the sources). Noise levels were calculated for two time periods, as defined in ISO 1996-2:201718, and expressed as the daily equivalent A-weighted noise level LAeq,24 h and the night equivalent A-weighted noise level Lnight (22:00–6:00).
Air pollution exposure: nitrogen dioxide (NO2) and particulate matter (PM10)
The NO2 and PM10 outdoor concentrations were calculated using a two-step emission and diffusion modeling19,20 First, the daily averaged annual road-traffic emissions were calculated using Circul’Air, software developed and used by the French air quality monitoring network on the basis of the COPERT4 European standard methodology. Second, air pollutant sources from the pollutant emission inventory related to all activity sectors21, especially heating, industries or agriculture, were introduced in ADMS-Urban©. This pollution diffusion software relies on state of the art algorithms based on traffic flow, meteorological data, background concentrations and buildings morphology. It has been developed in accordance with the WHO guidelines by the Cambridge Environmental Research Consultants company22 and is widely used in Europe for modeling air quality on scales ranging from large urban areas to the street level. The validity of the NO2 and PM10 models were verified using both sampling seasonal measurement campaigns and/or the city's fixed air-quality monitoring network23. These two air pollutants concentrations were expressed in µg/m3 considering the 3-months period before the occurrence of the MI.
Socioeconomic level
The neighbourhood socioeconomic level was estimated with a deprivation index based on the French sub-municipal census block groups (called IRIS). As defined by the National Institute of Statistic and Economics Studies, an IRIS (acronym of aggregated units for statistical information) has homogeneous type of habitat and about 2000 inhabitants20,24. This index was split into three classes: affluent, mixed and deprived IRIS. The socioeconomic level of each patient was calculated with the R package SesIndexCreator24.
Statistical analysis
Patient characteristics for each sex are summarized as numbers (percentages) for categorical variables and as medians (interquartile range) for continuous variables. Atherothrombotic risk was categorized on 4 classes named: low-risk, medium–low-risk, medium–high-risk, high-risk. The choice of 4 TRS-2P score division was made to balance the number in each risk category, as patients in the highest risk groups were rare (TRS-2P = 6, N = 7; TRS-2P = 7, N = 2), in the absence of consensual discretisation13,14,25. Ordinal regressions with adjacent category models were used to assess the relationship between atherothrombotic risk and noise levels. The Brant test was used to test the proportional odds assumption. Variables associated with atherothrombotic risk at a P-value ≤ 0.20 in the analysis were included using a bidirectional step-by-step elimination procedure in the multivariable analysis. Multivariable models were then adjusted for sex, socio-economic level and heart rate; in the analysis stratified on sex, the multivariable model was only adjusted for socio-economic level and heart rate. Sensitivity analyses were performed with the TRS-2P score in 8 levels, with cumulative logit models and by stratifying the age into two groups (threshold 60 y for men, 65 y for women). Odds ratios associated to noise exposure were expressed for an increase of 10 dB(A). Statistical analysis was performed using R version 4.0.2 statistical software26. Sensitivity analyses were further performed using polytomous logistic regression and cumulative ordinal logistic regression.
Results
Among the 1,350 eligible patients, 879 patients were included in the study (Fig. 1). Baseline characteristics of the population are shown in Table 1. Most patients were men (n = 567, 64.5%). The highest risk group of the TRS-2P score did not include any women. Women were on average 10 years older than men (74 vs 64 y). Hypertension and renal failure were more frequent among women (p < 0.001). There were more male current smokers (p < 0.001), and men had higher diastolic blood pressure on admission than women (p < 0.001).
Nearly half of men (N = 282, 49.7%) were in the low-risk category, while the majority of women (N = 104, 33.3%) were in medium–high-risk category (Table 1, Fig. 2). The levels of environmental exposure are presented in supplementary Table 1. Not surprisingly, the LAeq,24 h were higher than the Lnight (Fig. 3 and supplementary Table 1). An environmental noise map (global LAeq,24 h) and the spatial distribution of the observed cases of myocardial infarction are presented in Fig. 4. The score criteria and the distribution of each of the items used in the TRS-2P according to the four atherothrombotic risk classes are shown in supplementary Figs. 2 and 3. Among the components of the risk score, hypertension was the main driver of atherothrombotic risk, except in the low-risk group, where smoking was the most frequent (supplementary Figs. 2–3).
Noise exposure was significantly associated with atherothrombotic risk for all the noise sources and the LAeq,24 h and Lnight in univariable analysis. The odds ratio associated with a 10 dB(A) increase in road-related noise and railway-related noise were 1.161 (95% CI 1.021–1.320, P = 0.024) and 1.028 (95% CI 1.000–1.057, P = 0.049), respectively. In multivariable analysis (model 1), the association was only weakly modified after adjustment for global and traffic noise over 24 h and at night, for an increase of 10 dB(A), and most associations remained significant or nearly significant (Table 2). After adjustment on air pollutant exposure (model 1 + PM10 or model 1 + NO2), the observed OR increased and the associations remained significant.
The analyses of the interactions between noise exposure and sex are presented in supplementary Table 2. In stratified analysis, the associations remained significant in men (Table 3), but were no longer found in women (Table 4). Global noise, and the transportation and railway noise levels were significantly associated with an increase in atherothrombotic risk among men for both periods. For men, the odds associated with an increase of 10 dBA in transportation Lnight was 1.260 (95% CI 1.078–1.472, P = 0.004) (model 1). After adjustment on air pollutant exposure (model 1 + PM10 or model 1 + NO2), the observed OR weakly decreased and the associations remained significant in model 1 + PM10. For women, for an increase of 10 dBA in transportation Lnight,, the OR was 0.959 (95% CI 0.763–1.205, P = 0.721) Adjustment on air pollutants lead to the same results.
Sensitivity analyses were further performed using (i) polytomous logistic regression (ii) cumulative ordinal logistic regression and then (iii) groups stratified by age. The associations remained significant among men and non-significant among women.
Discussion
Our findings, obtained using data from a population of high-risk patients, show that exposure to environmental noise, in particular from transportation traffic, was associated with atherothrombotic risk. This relation was sex specific seeing as it was only observed in men.
The baseline characteristics of our study population, including sex ratio and rate of major risk factors, were similar to the general French MI population25, although our population was slightly younger (70.0 y vs 66.0 y, respectively). The distribution of low-/intermediate-/high-risk levels in our study was also similar (41%/26%/33% vs 42%/24%/34%)13. These data suggest the absence of a major selection bias in our study population and further reinforce the relevance of our findings. One strength of this study was the precise attribution of residential noise exposure, which was directly associated with geocoded patient addresses using a reproducible process completely unrelated to medical status. The acoustic levels in this city could be considered as moderate, The urban unit of Dijon is a ‘medium sized’ European city (i.e. city of 100,000 to 500,000 inhabitants)27. Over 44% of the European population is living in this category of cities28. The deprivation index was a collective measurement of neighbourhood socioeconomic status. It was defined using an accurate and reproducible procedure24, and it was thus possible to adjust for a potential influence of socio-economic factors in the studied relationships. Two pollutants related to road traffic were studied. NO2 is a gaseous pollutant known to be the main indicator of road traffic29. Particulate matters (PM10) are also generated by road traffic and residential heating and were chosen because of their significant impact on human30,31.
To the best of our knowledge, our study is the first to focus on the relationship between noise and atherothrombotic risk in high-risk patients. TRS-2P, a risk stratification tool, was developed to identify patients with a higher relative risk who are likely to benefit from the personalised administration of preventive measures. In real world patients, it was recently found to predict long-term outcomes, in particular for recurrent events14. Patients in elevated risk categories (TRS-2P score ≥ 5) had a dramatically increased risk of major cardiac events, in particular for recurrent MI (HR (95% CI) 10.03 (8.52–11.81)), when compared with lowest score (TRS-2P score = 0)32. Our data thus also suggests that chronic residential exposure could be associated with an increased risk of future recurrent ischemic events. However, further population-based studies are needed to support this hypothesis. Our decision to divide the TRS-2P score into 4 categories was made to balance the number in each risk category, since patients in the highest risk groups were rare (TRS-2P = 6, N = 7; TRS-2P = 7, N = 2). Furthermore, there is no consensus in the literature for any score threshold. Puymirat et al. chose to cut the score into 3 categories (0–1, 2, ≥ 3)13 whereas Bergmark et al. divided it into 4 groups (≤ 2, 3, 4, and ≥ 5)33 and Zafrir et al. into 6 (0, 1, 2, 3, 4, and ≥ 5)14.
Noise and CV diseases
Traffic noise is found to be associated with an increased risk of CV diseases, including MI and stroke34,35, and one meta-analysis has found that there is a linear relationship between exposure to transport noise and the incidence of ischemic heart disease. Nocturnal noise exposure has been shown to increase heart rate and blood pressure through activation of the sympathetic nervous system, reducing heart rate variability36,37. Moreover, associations between road traffic noise and CV diseases were found to be stronger among subjects sleeping with open windows38. Of course, we should assume that hypertension is the main cause of atherothrombotic risk, and a contributing factor for almost all patients in the high-risk group. However, the HYENA study also found an association between night-time aircraft noise and excess risk of hypertension (OR = 1.14, 95% CI 1.01–1.29)8. A meta-analysis also confirmed that there is a positive association between road traffic noise and hypertension39. Noise-induced annoyance could be one of the main contributors to the relationship between noise exposure and hypertension40.
Sex-specific sensitivity to transportation noise
Our results suggest that men are particularly sensitive to environmental noise, and especially to transportation noise. Only few studies have analyzed the influence of sex on the health effects of noise41. In experimental contexts, women felt more annoyance than men when exposed to all frequencies of low-intensity noise5. However, Röösli et al. suggested that men could be more sensitive to traffic-noise-induced sleep disturbance6. Sleep duration in highly exposed men (> 55 dB) was reduced by 1.5 h compared with men who had low exposure (< 30 dB), while sleep duration in women was not affected6. Convergent data from the HYENA (Hypertension and Exposure to Noise near Airports) study on a large population of 4721 Swedish subjects followed for 8–10 years also suggest that the risk of hypertension associated with residential aircraft noise exposure was significantly increased in men but not in women (RR (95% CI) 1.21 (1.05–1.39), and 0.97 (0.83–1.13), respectively)7 . Moreover, in 4,861 persons 45–70 years of age, who had lived at least 5 years near 6 major European airports found significant relationship between daily road traffic noise exposure and risk of hypertension, which was stronger for men (OR of 1.54 (95% CI 0.99–2.40) in the highest exposure category (> 65 dB; p(trend) = 0.008)8. The recent DEBATS study also identified this association in men (OR = 1.34, 95% CI 1.00–1.97)42. Nonetheless, sex-specific sensitivity remains controversial9.
Underlying pathophysiological mechanisms
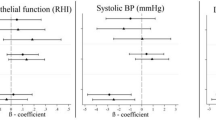
Experimental studies addressing the underlying mechanisms for noise-induced CV risk in animal or human are rare. Most works support the hypothesis of a causal relationship, through NADPH oxidase-induced vascular oxidative stress, exacerbation of hypertension, and promotion of pro-thrombotic and proinflammatory plasma phenotype43,44,45 and there is growing evidence from translational studies on how noise may trigger CV effects. In healthy adults, night-time exposure to aircraft noise was associated in a dose-dependent manner with impaired endothelial function46. In addition, night-time traffic noise has been shown to cause elevated levels of stress hormones such as cortisol and catecholamine, and sympathetic tone activation3. These factors could promote the development of vascular dysfunction (endothelial dysfunction) and high blood pressure, which, in turn, could elevate CV risk. Animal studies have also suggested that noise exposure during sleep causes a more pronounced inflammatory and oxidative stress response than wake-phase exposure47. However, some human studies are inconclusive48. Whether the extra-auditory effects of noise result directly from exposure or from sleep disturbance and related mental health problems, such as anxiety and depression, which are known CV risk factors, remains to be investigated.
Study limitations
Our study has some limitations. One is the use of TRS-2P, which includes the most important CV risk factors, as an index of atherothrombotic risk because it limited the relevance of adjustment on other variables. However, although the sample size was small, the odd ratios were only slightly modified by adjustments, highlighting the constancy of the associations between environmental noise and CV risk. We however acknowledge a weaker association between noise and the atherothrombotic risk when considering simultaneously the NO2 exposure. The lack of biological sampling to assess CV risk factors such as proinflammatory or prothrombotic markers, limited the conclusion on the link between the CV risk and noise. Furthermore, medical data collected at the time of admission does not represent baseline condition, whereas the noise levels assigned to each patient correspond to chronic exposure. Also, noise exposure levels were assigned to self-declared home address at the time of MI, although some individuals may have moved in the previous months. However, a recent study showed that the rate of moving in the French population was low (i.e. 11%)49, and older subjects, mainly retirees, were even less likely to move (9%). Therefore, considering the median age of our study population (70 y), it is likely that only few subjects moved and may thus have been assigned with erroneous levels of noise. People spend 80% of their time indoors50, and we did not take into account indoor noise. As this is a retrospective study, noise levels were not measured but modeled, so there is potential uncertainty regarding the validity environmental data. Differences in residential characteristics, such building insulation, were not recorded. Although subjects could spend part of their time out of their home, the noise exposure used was assigned at the home address. The potential errors of the Lnight estimate should therefore be smaller than for the LAeq,24 h, but our results were similar for LAeq,24 h and Lnight, which is in favor of a minor error of estimation. No questionnaire on sleeping habits, open or closed windows, sleeping room localization was available. The subject addresses contained only the street name and number. This could have limited the precision of the noise exposure assessment. Only global LAeq,24 h (dB(A)) map is presented, as noise map for nocturnal exposure could not be generated. However, LAeq,24 h usually closely correlates with Lnight.
Conclusion
Our results suggest, for the first time, that there is an association between transportation noise exposure and atherothrombotic risk, and they support the hypothesis that men are particularly sensitive to CV effects of chronic noise exposure. Further population-based prospective cohort studies are thus needed to better understand the interactions between sex and the CV effects of environmental noise, and to adapt, if necessary, public health messages.
References
WHO/Europe | Environmental Noise Guidelines. https://www.euro.who.int/en/health-topics/environment-and-health/noise/environmental-noise-guidelines-for-the-european-region (2021).
World Health Organization. Regional Office for Europe. Burden of disease from environmental noise: quantification of healthy life years lost in Europe. (World Health Organization. Regional Office for Europe, 2011).
Münzel, T. et al. Effects of noise on vascular function, oxidative stress, and inflammation: Mechanistic insight from studies in mice. Eur. Heart J. 38, 2838–2849 (2017).
van Kempen, E., Casas, M., Pershagen, G. & Foraster, M. WHO environmental noise guidelines for the European region: A systematic review on environmental noise and cardiovascular and metabolic effects: A summary. Int. J. Environ. Res. Public. Health 15, 379 (2018).
Beheshti, M. H. et al. The influence of personality traits and gender on noise annoyance in laboratory studies. Personal. Individ. Differ. 148, 95–100 (2019).
Röösli, M., Mohler, E., Frei, P. & Vienneau, D. Noise-related sleep disturbances: Does gender matter?. Noise Health 16, 197 (2014).
Eriksson, C., Bluhm, G., Hilding, A., Ostenson, C.-G. & Pershagen, G. Aircraft noise and incidence of hypertension–gender specific effects. Environ. Res. 110, 764–772 (2010).
Jarup, L. et al. Hypertension and exposure to noise near airports: The HYENA study. Environ. Health Perspect. 116, 329–333 (2008).
Babisch, W. Road traffic noise and cardiovascular risk. Noise Health 10, 27 (2008).
Zeller, M. et al. Impaired fasting glucose and cardiogenic shock in patients with acute myocardial infarction. Eur. Heart J. 25, 308–312 (2004).
Zeller, M. et al. Prevalence and impact of metabolic syndrome on hospital outcomes in acute myocardial infarction. Arch. Intern. Med. 165, 1192–1198 (2005).
Bohula, E. A. et al. Atherothrombotic risk stratification and the efficacy and safety of vorapaxar in patients with stable ischemic heart disease and previous myocardial infarction. Circulation 134, 304–313 (2016).
Puymirat, E. et al. Atherothrombotic risk stratification after acute myocardial infarction: The thrombolysis in myocardial infarction risk score for secondary prevention in the light of the French registry of acute ST elevation or non-ST elevation myocardial infarction registries. Clin. Cardiol. 42, 227–234 (2018).
Zafrir, B. et al. Long‐Term Risk Stratification of Patients Undergoing Coronary Angiography According to the Thrombolysis in Myocardial Infarction Risk Score for Secondary Prevention. J. Am. Heart Assoc. Cardiovasc. Cerebrovasc. Dis. 8, (2019)
Union, P. O. of the E. CELEX1, Directive 2002/49/EC of the European Parliament and of the Council of 25 June 2002 relating to the assessment and management of environmental noise - Declaration by the Commission in the Conciliation Committee on the Directive relating to the assessment and management of environmental noise. http://op.europa.eu/en/publication-detail/-/publication/27d1a64e-08f0-4665-a258-96f16c7af072/language-en (2002).
Barba-Vasseur, M. et al. Does low to moderate environmental exposure to noise and air pollution influence preterm delivery in medium-sized cities?. Int. J. Epidemiol. 46, 2017–2027 (2017).
Pujol, S. et al. Urban ambient outdoor and indoor noise exposure at home: A population-based study on schoolchildren. Appl. Acoust. 73, 741–750 (2012).
ISO 1996-2:2017. ISO https://www.iso.org/cms/render/live/en/sites/isoorg/contents/data/standard/05/97/59766.html (2020).
Tenailleau, Q., Mauny, F., Joly, D., François, S. & Bernard, N. Air pollution in moderately polluted urban areas: How does the definition of ”neighborhood” impact exposure assessment?. Environ. Pollut. 206, 437–448 (2015).
Mariet, A.-S. et al. Multiple pregnancies and air pollution in moderately polluted cities: Is there an association between air pollution and fetal growth?. Environ. Int. 121, 890–897 (2018).
EMEP/EEA air pollutant emission inventory guidebook - 2009 — European Environment Agency. https://www.eea.europa.eu/publications/emep-eea-emission-inventory-guidebook-2009.
CERC > Environmental software > ADMS-Urban model. http://www.cerc.co.uk/environmental-software/ADMS-Urban-model.html.
Tenailleau, Q. et al. Do outdoor environmental noise and atmospheric NO2 levels spatially overlap in urban areas?. Environ. Pollut. 214, 767–775 (2016).
Lalloué, B. et al. A statistical procedure to create a neighborhood socioeconomic index for health inequalities analysis. Int. J. Equity Health 12, 21 (2013).
Puymirat, E. et al. Acute myocardial infarction: Changes in patient characteristics, management, and 6-month outcomes over a period of 20 years in the FAST-MI program (French registry of acute ST-elevation or non-ST-elevation myocardial infarction) 1995 to 2015. Circulation 136, 1908–1919 (2017).
R Core Team. R: A Language and Environment for Statistical Computing. (R Foundation for Statistical Computing, 2020).
Boddy, M. Geographical economics and urban competitiveness: A critique. Urban Stud. 36, 811–842 (1999).
Giffinger, R., Kramar, H., Fertner, C. & Meijers, E. City-ranking of European medium-sized cities. Futur. Cities 2, 1–12 (2007).
Air quality in Europe — 2011 report — European Environment Agency. https://www.eea.europa.eu/media/publications/air-quality-in-europe-2011/.
World Health Organization. Regional Office for Europe. Health aspects of air pollution with particulate matter, ozone and nitrogen dioxide : report on a WHO working group, Bonn, Germany 13–15 January 2003. https://apps.who.int/iris/handle/10665/107478 (2003).
The application of models under the European Union’s Air Quality Directive: A technical reference guide — European Environment Agency. https://www.eea.europa.eu/publications/fairmode.
Bohula, E. A. et al. Atherothrombotic risk stratification and ezetimibe for secondary prevention. J. Am. Coll. Cardiol. 69, 911–921 (2017).
Bergmark, B. A. et al. Risk assessment in patients with diabetes with the TIMI risk score for atherothrombotic disease. Diabetes Care 41, 577–585 (2018).
Münzel, T. et al. Environmental stressors and cardio-metabolic disease: part I-epidemiologic evidence supporting a role for noise and air pollution and effects of mitigation strategies. Eur. Heart J. 38, 550–556 (2017).
Münzel, T. et al. Environmental stressors and cardio-metabolic disease: Part II-mechanistic insights. Eur. Heart J. 38, 557–564 (2017).
Haralabidis, A. S. et al. Acute effects of night-time noise exposure on blood pressure in populations living near airports. Eur. Heart J. 29, 658–664 (2008).
Lusk, S. L., Gillespie, B., Hagerty, B. M. & Ziemba, R. A. Acute effects of noise on blood pressure and heart rate. Arch. Environ. Health 59, 392–399 (2004).
Babisch, W., Ising, H., Gallacher, J. E., Sweetnam, P. M. & Elwood, P. C. Traffic noise and cardiovascular risk: The Caerphilly and Speedwell studies, third phase–10-year follow up. Arch. Environ. Health 54, 210–216 (1999).
van Kempen, E. & Babisch, W. The quantitative relationship between road traffic noise and hypertension: A meta-analysis. J. Hypertens. 30, 1075–1086 (2012).
Babisch, W. et al. Noise annoyance–a modifier of the association between noise level and cardiovascular health?. Sci. Total Environ. 452–453, 50–57 (2013).
Golmohammadi, R. & Darvishi, E. The combined effects of occupational exposure to noise and other risk factors—a systematic review. Noise Health 21, 125 (2019).
Evrard, A.-S., Lefèvre, M., Champelovier, P., Lambert, J. & Laumon, B. Does aircraft noise exposure increase the risk of hypertension in the population living near airports in France?. Occup. Environ. Med. 74, 123–129 (2017).
Eckrich, J. et al. Aircraft noise exposure drives the activation of white blood cells and induces microvascular dysfunction in mice. Redox Biol. 46, 102063 (2021).
Herzog, J. et al. Acute exposure to nocturnal train noise induces endothelial dysfunction and pro-thromboinflammatory changes of the plasma proteome in healthy subjects. Basic Res. Cardiol. 114, 2 (2019).
Steven, S. et al. Exacerbation of adverse cardiovascular effects of aircraft noise in an animal model of arterial hypertension. Redox Biol. 34, 101515 (2020).
Schmidt, F. P. et al. Effect of nighttime aircraft noise exposure on endothelial function and stress hormone release in healthy adults. Eur. Heart J. 34, 3508–3514a (2013).
Kröller-Schön, S. et al. Crucial role for Nox2 and sleep deprivation in aircraft noise-induced vascular and cerebral oxidative stress, inflammation, and gene regulation. Eur. Heart J. 39, 3528–3539 (2018).
Babisch, W., Gallacher, J. E., Elwood, P. C. & Ising, H. Traffic noise and cardiovascular risk. The Caerphilly study, first phase Outdoor noise levels and risk factors. Arch. Environ. Health 43, 407–414 (1988).
En 2014, un quart de la population qui déménage change de département - Insee Première - 1654. https://www.insee.fr/fr/statistiques/2882020 (2020).
How Europeans spend their time - Everyday life of women and men - Data 1998 - 2002. https://ec.europa.eu/eurostat/web/products-pocketbooks/-/KS-58-04-998 (2021).
Acknowledgements
The authors thank Suzanne Rankin for reviewing the English and Florence Bichat, Mailis Saint Jalmes, Etienne Baudouin and Jessica Massenot for their help with data collection and Thomas Morand for his editorial help.
Funding
This work (ENVI MI study) was supported by a grant from Fondation Coeur et Recherche. RICO is supported by the University Hospital of Dijon, the Association de Cardiologie de Bourgogne, Fédération Française de Cardiologie, and by grants from the Agence Régionale de Santé de Bourgogne Franche-Comté, and from the Conseil Régional de Bourgogne Franche-Comté.
Author information
Authors and Affiliations
Consortia
Contributions
M.K.: acquisition, analysis & interpretation of data. N.B.: substantial contributions to the conception & design of the work. F.M.: have drafted the work & substantively revised it. F.C.: acquisition of the data. S.P.: acquisition, analysis, or interpretation of data, mapping. M.M.: acquisition & analysis of the data. Y.C.: have drafted the work. M.Z.: conception & design of the work & substantively revised it.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Koczorowski, M., Bernard, N., Mauny, F. et al. Environmental noise exposure is associated with atherothrombotic risk. Sci Rep 12, 3151 (2022). https://doi.org/10.1038/s41598-022-06825-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-06825-0
This article is cited by
-
Towards extending the aircraft flight envelope by mitigating transonic airfoil buffet
Nature Communications (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.