Abstract
Soluble urokinase plasminogen activator receptor (suPAR) is an inflammatory biomarker and risk factor for kidney diseases, with a potential prognostic value in critically ill patients. In this monocentric prospective study, we measured plasma suPAR levels immediately after ICU admission in unselected 237 consecutive patients using a turbidimetric assay. Primary objective was the prognostic value for ICU- and 28-day mortality. Secondary objectives were association with sequential organ failure assessment (SOFA) score, coagulation and inflammation markers, AKI-3 and differences in prespecified subgroups. Median suPAR levels were 8.0 ng/mL [25th-75th percentile 4.3–14.4], with lower levels in ICU survivors than non-survivors (6.7 vs. 11.6 ng/mL, p < 0.001). SuPAR levels were higher in COVID-19, kidney disease, moderate-to-severe liver disease, and sepsis. ICU mortality increased by an odds ratio (OR) of 4.7 in patients with the highest compared to lowest quartile suPAR. Kaplan–Meier overall survival estimates at 3 months were 63% and 49%, in patients with suPAR below/above 12 ng/mL (log-rank p = 0.027). Due to an observed interaction between SOFA score and suPAR, we performed a random forest method identifying cutoffs. ICU mortality was 53%, 17% and 2% in patients with a SOFA score > 7, SOFA ≤ 7 & suPAR > 8 ng/mL, and SOFA score ≤ 7 & suPAR ≤ 8 ng/mL, respectively. suPAR was a significant predictor for AKI-3 occurrence (OR per doubling 1.89, 95% CI: 1.20–2.98; p = 0.006). suPAR levels at ICU admission may offer additional value for risk stratification especially in ICU patients with moderate organ dysfunction as reflected by a SOFA score ≤ 7.
Similar content being viewed by others
Introduction
The collective of patients admitted to the intensive care unit (ICU) is fairly heterogeneous. Sepsis, acute kidney injury (AKI) or respiratory failure account for a large proportion of patients, and many patients have pre-existing medical conditions1,2,3,4. Soluble urokinase plasminogen activator receptor (suPAR) is the circulating form of a membrane-bound protein, named uPAR, which can be found on endothelial cells and immune cells such as neutrophils, activated T-cells and macrophages5,6. Usually, suPAR levels are low but increase upon inflammatory stimulation during immune system activation. Increased levels of suPAR emerge in numerous conditions as surrogate of inflammation and can therefore serve as a prognostic marker7,8,9,10,11. Higher levels were shown to be associated with worse outcomes in various diseases, and suPAR was able to predict mortality in patients with bacteremia and systemic inflammatory response syndrome12,13,14,15. Previous studies show that elevated levels of suPAR predict AKI and CKD, for which it was shown to be a risk factor16,17. Furthermore, AKI can be mitigated using monoclonal antibodies against uPAR17. Nevertheless, the prognostic role of suPAR in patients in an ICU real-world setting has yet to be determined as earlier studies retrospectively assessed the values of suPAR at the end of the study period in selected patient cohorts. Additionally, previous published studies used enzyme-linked immunosorbent assays (ELISA) to determine suPAR levels, which are costly, labor-intensive, and slow. To solve these issues, an automated turbidimetric immunoassay was introduced recently. This method not only enables high throughput testing in routine laboratories but also comes along with significantly reduced time to results at approximately one hour. In this prospective cohort study, we aimed to delineate the role and feasibility of plasma suPAR assessed by an immunoturbidimetric test as a prognostic marker and risk stratification tool in an unselected cohort of consecutive patients admitted to a medical ICU.
Methods
Study population and study design
We prospectively recruited all consecutive patients older than 18 years admitted to the ICU of the Department of Internal Medicine at the Medical University of Graz, Austria from July until November 2020. Exclusion criteria were a therapy objective order (“comfort terminal care”) given directly at the time of admission, or a decline to participate. The study protocol was approved by the Institutional Review Board (IRB, Ethics committee Graz) of the Medical University of Graz, Austria (32–472 ex 19/20), registered in the WHO approved German study registry (DRKS00022458) and complied with the Declaration of Helsinki. Written informed consent was obtained from all conscious participants. In comatose non-surviving patients, the local IRB acted as legal guardian and gave consent for participation within the study.
Definitions and pre-specified subgroups
Subgroups as predefined by the study protocol were patients with active hematological malignant disease, active cancer (non-hematological), moderate to severe liver disease, CKD 3–5, pre-existing chronic obstructive pulmonary disease (COPD), acute myocardial infarction, elective valve surgery or percutaneous intervention, and continuous RRT. Sepsis was defined according to sepsis-3 criteria with a suspected infection by the treating physician and a sequential organ failure assessment (SOFA) score increase by ≥ 2 points18. Baseline SOFA score was deduced from previous medical records. Septic shock was specified as vasopressor therapy to maintain a mean arterial pressure (MAP) ≥ 65 mmHg in the absence of hypovolemia, and a lactate level > 2 mmol/L18. Acute kidney injury 3 (AKI-3) was defined as by KDIGO guidelines including a creatinine level ≥ 4 mg/dL, an increase of creatinine levels by three times from baseline, urine output ≤ 0.3 ml/kg/h for ≥ 24 h OR anuria for ≥ 12 h, or need for renal replacement therapy (RRT)19. AKI-3 was assessed separately on admission and during the ICU stay. Chronic kidney disease (CKD) definition was based on previous creatinine levels available from electronic medical records. Estimated glomerular filtration rate (eGFR) was calculated using the CKD-EPI formula20.
suPAR measurement
We determined suPAR levels in plasma samples anticoagulated with lithium heparin using a turbidimetric immunoassay (suPARnostic® TurbiLatex, ViroGates A/S, Copenhagen, Denmark). The test was applied and used according to the manufacturer’s instructions on a Cobas® 8000 c502 analyzer (Roche Diagnostics, Mannheim, Germany). The lower limit of quantification is 1.8 ng/mL, and the upper limit of quantification is 16 ng/mL. Dilution with human serum albumin 5% solution (1:1 ratio, Baxter) was performed in case levels exceeded the upper limit of detection and quantification. For other laboratory measurements see supplementary data.
Objectives
The primary objective was to assess the prognostic value of baseline plasma suPAR at ICU admission with regard to ICU- and 28-day mortality. Secondary objectives as prespecified by the study protocol included the association of suPAR with coagulation and inflammatory markers, as well as SOFA score. Furthermore, suPAR-level differences in prespecified subgroups and the diagnostic value of suPAR with regards to AKI-3 were investigated.
Statistical analyses
All statistical analyses were performed with SPSS 26 (SPSS Inc, Chicago, Illinois, USA) and Stata 15.0 (Stata Corp., Houston, TX, USA). Continuous variables were summarized as medians [25th-75th percentile], and categorical variables as absolute frequencies (%). Associations between variables were computed with cross-tabulations, Mann–Whitney-U-tests, χ2-tests, and Fisher’s exact tests, as appropriate. Spearman’s rank-based correlation coefficient was used for correlations analyses. The prognostic associations between 28-day / ICU mortality and other potential baseline predictors were computed with univariable and multivariable logistic regression. Formal adjustment for multiple testing was not performed. Significance level was determined at 0.05.
Ethics approval and consent to participate
The study protocol was approved by the Institutional Review Board (IRB) of the Medical University of Graz, Austria (32–472 ex 19/20), registered in the WHO approved German study registry (DRKS00022458) and complied with the Declaration of Helsinki. Written informed consent was obtained from all conscious participants. In comatose non-surviving patients, the local IRB acted as legal guardian and gave consent for participation within the study.
Results
Baseline characteristics and laboratory results of the study population
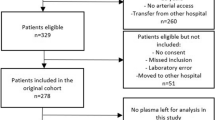
We screened 242 patients, of whom 237 were prospectively enrolled (Supplementary Fig. 1). Forty percent of the cohort were female and the median age at enrollment was 65 years [25th-75th percentile: 55–74] (Table 1). The median SOFA score was 7 points3,4,5,6,7,8,9,10. The median duration of ICU stay was three days, and the overall ICU and 28-day mortality rates were 27% (63 patients), and 38% (90 patients), respectively.
suPAR levels in the study population and subgroups
The median baseline suPAR level of the total cohort was 8.0 ng/mL [4.3–14.4, range: 1.5–56.8], with lower levels in ICU survivors than ICU non-survivors (6.7 vs. 11.6 ng/mL, p = 0.0003; Table 1). suPAR levels were significantly lower levels in patients with acute myocardial infarction and intoxicated patients, and significantly higher levels in COVID-19 patients, CKD patients, patients with moderate-severe liver disease, and patients with sepsis including those with septic shock (Table 2). Levels of suPAR were also higher in patients with AKI-3 on admission compared to those without AKI-3 (11.2 vs 6.6 ng/mL; p = 0.0001).
suPAR levels positively correlated with SOFA score (rho = 0.47, p < 0.0001) and with the hemostasis parameters INR (rho = 0.36; p < 0.0001), aPTT (rho = 0.42; p < 0.0001), and fibrinogen (rho = 0.18; p = 0.012). CRP (rho = 0.50; p < 0.0001), PCT (rho = 0.62; p < 0.0001), and IL-6 (rho = 0.38; p < 0.0001) correlated significantly positively with suPAR levels, while white blood cell count (rho = 0.03; p = 0.607) did not.
Univariable logistic regression analyses – ICU mortality
In univariable logistic regression analyses treating ICU mortality as a binary outcome variable, higher suPAR, higher lactate, and higher SOFA were among others prognostic for higher risk of ICU mortality (Table 3). Next, we used z-standardization to compare the prognostic impact of CRP, PCT, IL-6, and suPAR on ICU mortality on a common scale (all four biomarkers standardized to mean of zero and standard deviation (SD) of 1). Only suPAR and IL-6 were significant predictors for ICU mortality, with 1SD increase in suPAR being associated with a 1.4-fold increase in the odds of ICU mortality, while Z-standardized CRP and PCT were not significantly associated with this endpoint (Table 3, Fig. 1). Furthermore, the highest compared to the lowest quartile of suPAR had an OR of 4.7 for ICU mortality (Fig. 2). The results for suPAR towards ICU mortality prediction were furthermore consistent among all pre-specified (Supplementary Fig. 2A) and exploratory (Supplementary Fig. 2B) subgroups.
Z-standardized biomarkers for prediction of ICU mortality. z-standardization = all four biomarkers standardized to a mean of zero and a standard deviation (SD) of 1. Note that the hollow green diamonds represent the odds ratio for the respective biomarker, and the associated bars represented the 95% confidence interval. The black vertical line represents an odds ratio of 1 (“line of unity”). ICU = intensive care unit; CI = confidence interval; suPAR = soluble urokinase plasminogen activator receptor; CRP = C-reactive protein; PCT = procalcitonin; IL-6 = interleukin-6.
Multivariable analysis for ICU mortality
In multivariable logistic regression analysis, a stepwise-backwards selection algorithm with p > 0.10 as the criterion for removal resulted in a multivariable model with higher SOFA score and higher lactate as independent predictors of increased ICU mortality, and higher SOFA score as independent predictor of increased 28-day mortality, respectively (Table 4).
Exploratory analysis of overall survival and suPAR levels
We observed 96 deaths (41%) from any cause during a median follow-up of 2.9 months. Using and validating a published suPAR cut-off at 12 ng/mL by Giamarellos-Bourboulis et al.21, Kaplan–Meier overall survival estimates at 3 months were 63% in the 160 patients with baseline suPAR levels below 12 ng/mL, and 49% in the 77 patients with baseline suPAR levels ≥ this cut-off (log-rank p = 0.027, Fig. 3).
Overall survival for suPAR levels above or below a 12 ng/mL cutoff. Median follow-up of the total cohort was 2.9 months with 96 observed deaths from any cause. Using a previously published suPAR cut-off at 12 ng/mL for validation, Kaplan–Meier overall survival estimates at 3 months were 63% in the 160 patients with baseline suPAR levels below 12 ng/mL, and 49% in the 77 patients with baseline suPAR levels ≥ this cut-off (log-rank p = 0.027).
Mortality risk stratification in the ICU
We next assessed whether suPAR could improve mortality risk stratification towards ICU mortality beyond the SOFA score. In detail, the association between suPAR and higher risk of ICU mortality did not prevail after adjusting for SOFA score (adjusted OR for ICU mortality per doubling of suPAR = 1.08, 95% CI: 0.79–1.47, p = 0.636). However, we observed some evidence for an interaction between suPAR and SOFA (interaction p = 0.053), with higher SOFA scores reducing the prognostic impact of suPAR on ICU mortality, and vice versa (Supplementary Table 1). We thus employed a random forest to our ICU mortality data, which suggested SOFA score and suPAR cut-offs at 7 points and 8 ng/mL for ICU mortality risk stratification, respectively. In detail, the random forest suggested that prognosis is determined by the SOFA score in patients with > 7 points. However, in patients with a SOFA score ≤ 7 points, further prognosis is determined by suPAR levels at a cut-off at 8 ng/mL (Fig. 4A). Consequently, ICU mortality was 53% in the 99 patients with a SOFA score > 7 points, 17% in the 54 patients with a SOFA score ≤ 7 points and suPAR levels > 8 ng/mL, and 2% in the 84 patients with a SOFA score ≤ 7 points and suPAR levels ≤ 8 ng/mL. Furthermore, this association consists regarding overall survival (Fig. 4B, supplementary Fig. 3).
(A) Random forest analysis—overall survival according to risk stratification groups. For this analysis, we used the random forest machine learning method with the CHAID algorithm treating ICU mortality as a dependent variable and suPAR and SOFA score as independent variables, with tenfold cross validation. The random forest proposed cut-off values for SOFA score at 7 points (corrected p < 0.0001) and for suPAR at 8 ng/mL (corrected p = 0.022). Note that in patients with a SOFA score > 7 the prognosis was determined solely by the SOFA score. (B) Random forest analysis – Overall survival according to risk stratification groups. ICU mortality was 53% in patients with a SOFA score > 7 points (n = 99), 17% in patients with a SOFA score ≤ 7 points and suPAR levels > 8 ng/mL (n = 54), and 2% in patients with a SOFA score ≤ 7 points and suPAR levels ≤ 8 ng/mL (n = 84). Log-rank p < 0.0001.
suPAR prognostic performance regarding other clinical endpoints
Apart from mortality endpoints, we investigated other pre-specified and exploratory clinically relevant endpoints. In 12 patients, dialysis was performed before first sample of suPAR was drawn (median 7 h later), but there was no significant difference between suPAR levels before and after dialysis initiation (p = 0.959). We excluded patients with AKI-3 already being present on admission and investigated whether suPAR was able to predict the occurrence of AKI-3 during ICU stay. suPAR was a significant predictor for the occurrence of AKI-3 during ICU stay (suPAR per doubling OR 1.89, 95% CI: 1.20–2.98; p = 0.006). Furthermore, in the overall cohort, suPAR was, among others, also a predictor for the occurrence of the necessity of increasing catecholamine doses within the first 12 h after ICU admission (suPAR per doubling OR 1.48, 95% CI: 1.16–1.89; p = 0.002; Supplementary Table 2).
suPAR levels at hospital discharge
Fifty-four patients had suPAR samples obtained within 72 h before alive hospital discharge, either directly from ICU or from general ward. The median suPAR concentration on discharge from hospital was 5.8 ng/mL [3.4–10.3]. The values were higher in patients with (8.4 ng/mL [4.8–14.5]) compared to those without unplanned 90-day readmission (5.2 ng/mL [3.3–8.5], p = 0.084). The suPAR levels at hospital discharge showed a borderline significant association with the rate of unplanned 90-day readmission in our cohort (OR per 1 ng/mL suPAR increase 1.12 [1.00–1.24], p = 0.053). This fact might be driven by the small sample size of this subgroup and needs additional investigation.
Discussion
This study assessed suPAR levels on ICU admission in critically ill patients in a real-world setting. Levels of suPAR were significantly higher in ICU non-survivors compared to ICU survivors. Moreover, suPAR had the potential to further stratify and predict outcome in patients with moderate risk for ICU mortality according to SOFA score.
suPAR is a circulating protein and blood levels are low during physiological conditions6. suPAR is a sensitive but non-specific marker of acute disease, representing inflammation, the immune and coagulation system, and is also found on endothelial cells5,7. suPAR is not solely a representation of infection, but also mirrors comorbidities and organ dysfunctions22. suPAR originating from immature myeloid cells is furthermore part of the three-finger fold protein toxin superfamily with strong signaling capabilities23,24,25,26,27. It may come in different proteolytic fragments and isoforms, and can bind to integrins6,16. CRP, on the other hand, is a pentameric protein mainly synthesized in the liver with very low signaling properties28. Similarly, increased suPAR levels can be found in bronchoalveolar fluid during acute respiratory distress syndrome likely due to neutrophils present in lung tissue29. The normal blood levels for healthy individuals are reported to be 2–3 ng/ml9,30, and are clearly higher in ICU patients compared to patients outside the ICU15. In our study investigating critically ill ICU patients, suPAR levels were elevated at 8.0 [4.3–14.5] ng/mL. The previously reported mean difference of suPAR between sepsis survivors and non-survivors was 5.2 ng/ml31. We also found significantly lower suPAR levels in ICU survivors at 6.7 compared to non-survivors at 11.6 ng/mL. Furthermore, levels were elevated in patients with COVID-19 and sepsis likely due to the increased activity of immune cells. We can only speculate that the burden of inflammation and in parallel, the levels of suPAR are lower in patients with intoxication and acute myocardial infarction when compared to patients with sepsis32,33.
Higher suPAR levels have been associated with worse clinical outcome including higher 28-day mortality and rate of AKI in several selected patient cohorts12,15,34,35,36,37,38,39. In one study, suPAR was associated with clinical deterioration within 72 h in emergency department patients with bacterial sepsis40. In a study of ICU sepsis patients, suPAR results were also associated with hospital mortality but did not improve the area under the receiver operating characteristics (AUROC) of the SOFA or Acute Physiology And Chronic Health Evaluation (APACHE) score41. Also, the prognostic performance of suPAR was found to be worse than the Simplified Acute Physiology Score 2 (SAPS2) score34. A meta-analysis suggested the potential role of suPAR for the prediction of 28-day mortality in sepsis patients, but the reported AUROC (ranging from 0.670–0.788)31 were inferior to the SOFA score (0.773 [0.712–0.834]) and slightly higher than the AUROC of suPAR in our study (0.630 [0.559–0.702]). This difference may be explained by the fact that the aim of our study was to include a fairly heterogeneous (e.g., unselected) population of critically ill patients, while previous studies investigated a narrower patient cohort. Overall, we found that ICU non-survivors had higher levels of suPAR and that suPAR was a strong predictor of ICU mortality. Nevertheless, the prognostic value of the SOFA score for the overall cohort was not improved by adding suPAR to the model, and suPAR did not remain predictive in multivariable analysis. However, we found an interaction between the SOFA score and suPAR levels suggesting that the prognostic impact of suPAR decreased with increasing SOFA score. In the most severely ill patients with the highest rate of organ dysfunction, the prognostic performance of SOFA score outperformed suPAR and prognosis therefore was determined solely by the SOFA score. On the other hand, in moderately ill ICU patients as reflected by a SOFA score ≤ 7, suPAR was a strong marker to identify patients with an exceptionally good ICU prognosis independent of the underlying disease. Our findings are somewhat similar to another study that found that sepsis patients with an APACHE II score ≥ 17 and a suPAR level ≥ 12 ng/mL had the worst outcome, while patients with APACHE II < 17 and suPAR < 12 ng/mL had the best outcome21. We furthermore validated this previously reported cutoff of 12 ng/mL and found that overall survival estimates were significantly different between patients below or above this cutoff (63% vs. 49%).
The study by Hayek et al.also assessed critically ill patients and found that patients in the highest suPAR quartile had higher rates of AKI. In addition, in an animal model, the same group found that a monoclonal antibody therapy against suPAR attenuated the rate of AKI17. In another study, which involved adult hospitalized COVID-19 patients both inside and outside ICU, increasing suPAR tertiles were associated with AKI and dialysis42. Similarly, in our study, the rate of AKI-3 development during ICU stay in patients with AKI-3 not present on admission, was predicted by the suPAR level at the time of ICU admission. Therefore, future investigations should focus on suPAR as a potential therapeutic target to alter the clinical course on development of AKI-3. As an exploratory endpoint, we investigated whether suPAR levels on hospital discharge were able to predict unplanned 90-day readmission rate. Our data suggest that suPAR may be helpful in the decision process whether a patient can be safely discharged from the hospital; however, these results need to be validated in a separate larger trial.
Most previous studies used an ELISA to measure suPAR, which is time-consuming and often performed with some delay due to batch analyses. In our study, however, we used an automated immunoturbidometric assay, which can be run on routine laboratory analyzers and is therefore able to provide results within one hour. From this point of view, an implantation into clinical routine is more than promising. Intensive care scores, e.g. APACHE and SAPS, are time-consuming to calculate, and suPAR may therefore provide a quick stratification tool. Furthermore, suPAR outperformed other conventional inflammatory biomarkers such as CRP and PCT and was similar to IL-6 in prediction of ICU mortality in our cohort. Previous studies showed that suPAR levels remain stable over the first week39,43,44.
As a side note, like previous reports45, we were able to show that lipoproteins were significantly different between ICU survivors and non-survivors, with the latter having lower levels of HDL-C and total cholesterol. Most notably, HDL-C levels were significantly lower in patients with hemorrhagic shock or GI bleeding, moderate-severe liver disease and in sepsis patients (Supplementary Table 3).
Our study has several limitations. First, it was a single center study, therefore the external validity for other facilities may be reduced and results should be validated in larger multicentric trials. Second, our study was not designed for validation of an already approved turbidimetric immunoassay but to investigate its use in unselected critically ill patients for outcome prediction. However, based on our experience and the low workload for laboratory staff we assume that the turbidimetric test as used in our study provides much more clinical practicability. Third, currently there is no evidence that a prognostic biomarker improves outcomes even when the risk of a patient is clearly delineated. However, given the recent scientific data suPAR may not only have prognostic value but may also be a therapeutic target in the future. Fourth, our study was not powered for the 90-day readmission rate, so results must be interpreted as hypothesis-generating.
Conclusion
In summary, suPAR was associated with ICU mortality in univariable logistic regression, and may offer the highest prognostic potential for risk stratification in moderately ill ICU patients as reflected by a SOFA score ≤ 7. Furthermore, our study shows that suPAR level on ICU admission may be a useful biomarker for prediction of AKI-3 in ICU patients.
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Vincent, J. L., Jones, G., David, S., Olariu, E. & Cadwell, K. K. Frequency and mortality of septic shock in Europe and North America: A systematic review and meta-analysis. Crit. Care. 23(1), 196 (2019).
Pham, T. & Rubenfeld, G. D. Fifty years of research in ARDS. The epidemiology of acute respiratory distress syndrome. A 50th birthday review. Am. J. Respir. Crit. Care Med. 195(7), 860–870 (2017).
Bellani, G. et al. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA 315(8), 788–800 (2016).
Jiang, L. et al. Epidemiology of acute kidney injury in intensive care units in Beijing: The multi-center BAKIT study. BMC Nephrol. 20(1), 468 (2019).
Desmedt, S., Desmedt, V., Delanghe, J. R., Speeckaert, R. & Speeckaert, M. M. The intriguing role of soluble urokinase receptor in inflammatory diseases. Crit. Rev. Clin. Lab. Sci. 54(2), 117–133 (2017).
Thuno, M., Macho, B. & Eugen-Olsen, J. suPAR: The molecular crystal ball. Dis Markers. 27(3), 157–172 (2009).
Eugen-Olsen, J. & Giamarellos-Bourboulis, E. J. suPAR: The unspecific marker for disease presence, severity and prognosis. Int. J. Antimicrob. Agents. 46(Suppl 1), S33–S34 (2015).
Haupt, T. H. et al. Risk factors associated with serum levels of the inflammatory biomarker soluble urokinase plasminogen activator receptor in a general population. Biomark Insights. 9, 91–100 (2014).
Haupt, T. H. et al. Healthy lifestyles reduce suPAR and mortality in a Danish general population study. Immun. Ageing. 16, 1 (2019).
Reichsoellner, M., Raggam, R. B., Wagner, J., Krause, R. & Hoenigl, M. Clinical evaluation of multiple inflammation biomarkers for diagnosis and prognosis for patients with systemic inflammatory response syndrome. J. Clin. Microbiol. 52(11), 4063–4066 (2014).
Persson, M. et al. Soluble urokinase plasminogen activator receptor: A risk factor for carotid plaque, stroke, and coronary artery disease. Stroke 45(1), 18–23 (2014).
Donadello, K. et al. Soluble urokinase-type plasminogen activator receptor as a prognostic biomarker in critically ill patients. J Crit. Care. 29(1), 144–149 (2014).
Hoenigl, M. et al. Diagnostic accuracy of soluble urokinase plasminogen activator receptor (suPAR) for prediction of bacteremia in patients with systemic inflammatory response syndrome. Clin. Biochem. 46(3), 225–229 (2013).
Molkanen, T., Ruotsalainen, E., Thorball, C. W. & Jarvinen, A. Elevated soluble urokinase plasminogen activator receptor (suPAR) predicts mortality in Staphylococcus aureus bacteremia. Eur. J. Clin. Microbiol. Infect. Dis. 30(11), 1417–1424 (2011).
Raggam, R. B. et al. Soluble urokinase plasminogen activator receptor predicts mortality in patients with systemic inflammatory response syndrome. J. Intern. Med. 276(6), 651–658 (2014).
Hayek, S. S. et al. Soluble Urokinase Receptor and Chronic Kidney Disease. N. Engl. J. Med. 373(20), 1916–1925 (2015).
Hayek, S. S. et al. Soluble urokinase receptor and acute kidney injury. N. Engl. J. Med. 382(5), 416–426 (2020).
Singer, M. et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA 315(8), 801–810 (2016).
Khwaja, A. KDIGO clinical practice guidelines for acute kidney injury. Nephron. Clin. Pract. 120(4), c179–c184 (2012).
Stevens, P. E., Levin, A. & Kidney Disease: Improving Global Outcomes Chronic Kidney Disease Guideline Development Work Group M. Evaluation and management of chronic kidney disease: Synopsis of the kidney disease: Improving global outcomes 2012 clinical practice guideline. Ann. Intern. Med. 158(11), 825–830 (2013).
Giamarellos-Bourboulis, E. J. et al. Risk assessment in sepsis: A new prognostication rule by APACHE II score and serum soluble urokinase plasminogen activator receptor. Crit. Care. 16(4), R149 (2012).
Koch, A. & Tacke, F. Risk stratification and triage in the emergency department: Has this become “suPAR” easy?. J. Intern. Med. 272(3), 243–246 (2012).
Galat, A. The three-fingered protein domain of the human genome. Cell Mol. Life Sci. 65(21), 3481–3493 (2008).
Kessler, P., Marchot, P., Silva, M. & Servent, D. The three-finger toxin fold: A multifunctional structural scaffold able to modulate cholinergic functions. J. Neurochem. 142(Suppl 2), 7–18 (2017).
Huai, Q. et al. Structure of human urokinase plasminogen activator in complex with its receptor. Science (New York, NY). 311(5761), 656–659 (2006).
Hahm, E. et al. Bone marrow-derived immature myeloid cells are a main source of circulating suPAR contributing to proteinuric kidney disease. Nat Med. 23(1), 100–106 (2017).
Gussen, H. et al. Neutrophils are a main source of circulating suPAR predicting outcome in critical illness. J. Intensive Care. 7, 26 (2019).
Nauta, A. J., Daha, M. R., van Kooten, C. & Roos, A. Recognition and clearance of apoptotic cells: A role for complement and pentraxins. Trends Immunol. 24(3), 148–154 (2003).
Reisinger, A. C. et al. SuPAR levels in BAL fluid from patients with acute respiratory distress syndrome-a pilot study. Crit. Care. 24(1), 576 (2020).
Chew-Harris, J., Appleby, S., Richards, A. M., Troughton, R. W. & Pemberton, C. J. Analytical, biochemical and clearance considerations of soluble urokinase plasminogen activator receptor (suPAR) in healthy individuals. Clin. Biochem. 69, 36–44 (2019).
Pregernig, A., Muller, M., Held, U. & Beck-Schimmer, B. Prediction of mortality in adult patients with sepsis using six biomarkers: A systematic review and meta-analysis. Ann. Intensive Care. 9(1), 125 (2019).
Peiro, O. M. et al. Soluble urokinase plasminogen activator receptor as a long-term prognostic biomarker in acute coronary syndromes. Biomarkers 25(5), 402–409 (2020).
Chew-Harris, J. et al. Soluble urokinase plasminogen activator receptor (suPAR) can predict long-term mortality in patients with acute chest pain. Eur. Heart J. 41(Issue Supplement_2), ehaa946-1695 (2020).
Backes, Y. et al. Usefulness of suPAR as a biological marker in patients with systemic inflammation or infection: A systematic review. Intensive Care Med. 38(9), 1418–1428 (2012).
Geboers, D. G. et al. Plasma suPAR as a prognostic biological marker for ICU mortality in ARDS patients. Intensive Care Med. 41(7), 1281–1290 (2015).
Rovina, N. et al. Soluble urokinase plasminogen activator receptor (suPAR) as an early predictor of severe respiratory failure in patients with COVID-19 pneumonia. Crit. Care. 24(1), 187 (2020).
Loonen, A. J. M. et al. High pneumococcal DNA load, procalcitonin and suPAR levels correlate to severe disease development in patients with pneumococcal pneumonia. Eur. J. Clin. Microbiol. Infect. Dis. 36(9), 1541–1547 (2017).
Jalkanen, V. et al. SuPAR and PAI-1 in critically ill, mechanically ventilated patients. Intensive Care Med. 39(3), 489–496 (2013).
Koch, A. et al. Circulating soluble urokinase plasminogen activator receptor is stably elevated during the first week of treatment in the intensive care unit and predicts mortality in critically ill patients. Crit. Care. 15(1), R63 (2011).
Lafon, T. et al. Prognostic performance of endothelial biomarkers to early predict clinical deterioration of patients with suspected bacterial infection and sepsis admitted to the emergency department. Ann. Intensive Care. 10(1), 113 (2020).
Suberviola, B., Castellanos-Ortega, A., Ruiz Ruiz, A., Lopez-Hoyos, M. & Santibanez, M. Hospital mortality prognostication in sepsis using the new biomarkers suPAR and proADM in a single determination on ICU admission. Intensive Care Med. 39(11), 1945–1952 (2013).
Azam, T. U. et al. Soluble Urokinase Receptor (SuPAR) in COVID-19-Related AKI. J. Am. Soc. Nephrol. 31(11), 2725–2735 (2020).
Hall, A., Crichton, S., Varrier, M., Bear, D. E. & Ostermann, M. suPAR as a marker of infection in acute kidney injury—A prospective observational study. BMC Nephrol. 19(1), 191 (2018).
Liu, X. et al. Prognostic significance of APACHE II score and plasma suPAR in Chinese patients with sepsis: A prospective observational study. BMC Anesthesiol. 16(1), 46 (2016).
Reisinger, A. C. et al. Arylesterase Activity of HDL Associated Paraoxonase as a Potential Prognostic Marker in Patients With Sepsis and Septic Shock-A Prospective Pilot Study. Front. Med. (Lausanne) 7, 579677 (2020).
Acknowledgements
We especially want to thank all colleagues and staff from the intensive care unit for helping with patient recruitment during the study.
Funding
The consumables for the turbilatex test were provided by the manufacturer Virogates A/S, Copenhagen, Denmark; and the distributor Biomedica Medical Products, Vienna, Austria. These companies had no influence on the study design and procedures. These funding parties did not have access to data and were not involved in the statistical analysis. Doctoral student ACR received funding from the Medical University of Graz through the Doctoral School Molecular Medicine and Inflammation for article publication fees and open access costs.
Author information
Authors and Affiliations
Contributions
A.C.R. and P.E. designed the study. G.H., R.B.R., J.P., T.N. and M.H. provided valuable input to the initial study protocol. A.C.R., P.E., G.S., S.H., and G.H. were responsible for patient recruitment. A.M.M., T.N. and G.S. provided valuable input for data acquisition and interpretation. A.C.R. and F.P. performed statistical analyses. A.C.R. and P.E. wrote the manuscript, and all other authors provided critical intellectual content to the manuscript. All authors approved the final version of this paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Reisinger, A.C., Niedrist, T., Posch, F. et al. Soluble urokinase plasminogen activator receptor (suPAR) predicts critical illness and kidney failure in patients admitted to the intensive care unit. Sci Rep 11, 17476 (2021). https://doi.org/10.1038/s41598-021-96352-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-96352-1
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.