Abstract
Faunal provincialism between the North and South parts of Eastern Asia is shown to have been in place since the late Eocene. This provincialism structured the mammalian dispersals across Eurasia for millions of years and provides insights into both palaeonvironments and palaeoclimate zonation. In addition, this study reveals the oldest record of a crown ruminant (Iberomeryx from Shinao, China). Ecologically, as well as economically, ruminant artiodactyls are one of the most important large mammal groups today. The revision of the ruminants from the Shinao Formation, from the Caijiachong marls and Xiaerhete, resulted in two new taxa and shows that the different provinces were populated by distinct taxa living in different environments, dominated by the monsoon in the South and drier conditions in the North. Evaluating this result in a Eurasian context demonstrates that the dispersals from Asia to Europe was complex. These results confirm that there were at least two dispersal events, distinct in space and time: the Grande-Coupure from Northern and Central Asia along the North ca. 34 Mya and the Bachitherium dispersal event from the Southern province along a southerly route ca. 31 Mya.
Similar content being viewed by others
Introduction
The fossil record of Asia holds most of the spectacular findings in vertebrate palaeontology of the several last decades and many questions in mammal evolution can be tackled now thanks to these discoveries. The Ruminantia (crown group comprising the Pecora: pronghorns, giraffes, deer, cows, and musk deer; the Tragulidae: mouse deer; and all fossil representatives until their last shared ancestor) is an ecologically and economically important group of artiodactyls. Like for other mammals, the origin of that clade is hidden as well somewhere in the long Asian geological history. Most likely their roots go back to the late middle Eocene, at ca. 45 Mya1,2. Traditionally, the Eocene was considered as rather homogenous in climate and faunal composition at a large scale in Asia3. However, while the South-eastern part of Asia has shown very few changes in a warm and humid climate and environment since the Eocene4, Northern Asia faced a progressive but complex transition from warm and humid subtropical environments during the Eocene to steppe environments in the Pliocene, e.g.3,4,5. The aridification in Northern and Central Asia (recorded from the early Bartonian, ca. 40–37 Mya, onwards in Central Asia6,7,8,9) was triggered by a variety of factors, e.g.9,10,11,12, which led to a strong latitudinal environmental and climatic zonation during the middle and the late Eocene, e.g.6,13. Despite this environmental heterogeneity, a relative homogenous fauna is described in Asia during that time.
All of the earliest ruminants are grouped into the Asian family Archaeomerycidae sensu2 (including Archaeomeryx, Xinjiangmeryx, Indomeryx, Miomeryx, and Notomeryx) that did not survive the Eocene/Oligocene transition2. Lophiomeryicidae are another clade of stem ruminants. The oldest lophiomerycid appeared during the late middle Eocene (Zhailimeryx hetii14) and did not diversify until the late Eocene and Oligocene when the family was distributed across the whole of Eurasia before it disappeared during the latest Oligocene14,15,16. Besides the co-occurence of Archaeomerycidae and Lophimomerycidae, the late Eocene also shows the first large scale diversification of ruminants with the appearance of Tragulidae in Southeast Asia17, “Gelocidae” and Praetragulidae (sensu2) in Central Asia and Bachitheriidae in the Southeast Europe18,19. The Tragulidae are the sole still surviving ruminant from all the above-mentioned families known during the Eocene.
Key localities for a better understanding of the Asian terrestrial ecosystem modifications during the Paleogene are the Shinao Formation and the Caijiachong (= Tsaichiachung) marls, as well as the Xiaerhete locality, where a diversified fauna has been discovered. The Shinao Formation (Southwestern Guizhou and adjoining Eastern Yunnan, China) yielded a relatively diverse and unique large mammal fauna includig the genus Lophiomeryx20. For a long time the Formation was dated to the early Oligocene, as Lophiomeryx was considered diagnostic for a post-Eocene age. New and revised data on the stratigraphic range of Lophiomerycidae (including the reappraisals of other Asian localities) and the faunal content of the Shinao Formation now proves a late Eocene age for the locality2,14,20,21,22,23,24,25.
Like the Shinao Formation, the Caijiachong marls (Qujing Yunnan, China) have long been considered as early Oligocene of age26. However, considering the great similarity of this fauna with the one of the Lunan Formation and of Ardyn Obo26,27, we agree with Vislobokova21 that the Caijiachong fauna should be assigned to the late Eocene, as well as Xiaerhete based on preliminary analyses28.
Here we reassess the late Eocene ruminants from the Shinao Formation and the specimens attributed to cf. Miomeryx sp. from the Caijiachong marls and we describe a new ruminant specimen from the late Eocene from Xiaerhete. The re-description of these specimens leads to the erection of a new genus and two new species and reveals the by far oldest crown ruminant, the tragulid Iberomeryx. This study shows for the first time that two latitudinal palaeoenvironmental provinces shaped the Eurasian dispersal patterns for more than 10 million years and were already present during the Eocene. It aids in a better understanding of Cenozoic Eurasian palaeobiogeography and the structure of palaeoclimate zonation, as well as the spreading of mammals across the continent during their evolutionary history. Indeed, the Northern and Central Asiatic Lophiomeryx, living in more arid environments, is known in Western Europe from the Grande-Coupure dispersal event (34 Mya), while the Southern Asiatic Iberomeryx, living in more humid environments, is known in Western Europe from the Bachitherium dispersal event (31 Mya).
Systematic palaeontology
Mammalia Linnaeus, 175829.
Artiodactyla Owen, 184830.
Ruminantia Scopoli, 177731.
Infraorder Tragulina Flower, 188332.
Family Lophiomerycidae Janis, 198733.
Included genera
Lophiomeryx, Zhailimeryx, Krabimeryx, Chiyoumeryx nov. gen.
Genus Krabimeryx Métais, Chaimanee, Jaeger, and Ducroq, 200117.
Etymology
Krabi—from Krabi Basin, where the fossils were found, and—meryx is the Greek word for ruminant.
Diagnosis [modified after Métais et al.17]
Small primitive ruminant with lower molars morphologically close to those of Zhailimeryx. Krabimeryx differs from Zhailimeryx in: more laterally compressed lingual cuspids in the lower molars; an entoconid displaced to anterior with respect to the hypoconid; the lack of both a paraconid and a hypoconulid in m1 and m2; a p4 with a mesolingual conid that is located more posterior and less individualized; a p4 without a distinct posterolingual conid. Krabimeryx differs from Lophiomeryx by less selenodont labial cuspids in the lower molars, the presence of a developed external postmetacristid, and by a distinct groove on the anterior side of the entoconid, the entoconidian groove. Krabimeryx can be distinguished from Iberomeryx in having a well-marked entoconidian groove; the lack of a clear external postprotocristid; the third lobe of m3 not forming a complete buckle; and a more transversely compressed hypoconulid in the m3. Krabimeryx possesses a huge notch in lingual view between the entoconid and the third lobe in the m3.
Type species
Krabimeryx primitivus Métais, Chaimanne, Jaeger, and Ducroq, 200117.
Included species
Krabimeryx gracilis nov. comb. (Miao, 198220).
Krabimeryx gracilis nov. comb. (Miao, 198220).
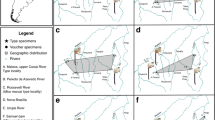
Dentition of Krabimeryx gracilis nov. comb. (Miao, 1982)20 (A, B, G, H), Chiyoumeryx nov. gen. shinaoensis (Miao, 1982)20 (C, D), Chiyoumeryx nov. gen. flavimperatoris nov. sp. (E) and Iberomeryx miaoi nov. sp. (F–I). Krabimeryx gracilis nov. comb. (Miao, 1982)20: (A) IVPP V 6546-1 (holotype), partial skull with right and left M1–M3; (B) IVPP V 6546-2 (holotype), right fragmented mandible with m2–m3. Chiyoumeryx nov. gen. shinaoensis (Miao, 1982)20: (C) IVPP V 6531 (holotype), right mandible with p2–m3 and tooth socket of p1; (D) IVPP V 6532 (paratype), right fragmented maxillary with P4-M3. Chiyoumeryx nov. gen. flavimperatoris nov. sp.: (E) IVPP V 6547 (holotype), right mandible with p4–m3; Iberomeryx miaoi nov. sp.: (F) IVPP V 6551 (holotype), left mandible with m1–m3 (mirrored); (G) lower molar Lophiomerycidae dental nomenclature (based on the m3 of IVPP 6546-2): 1 internal postmetacristid, 2 metaconid, 3 external postmetacristid, 4 internal preentocristid, 5 entoconidian groove, 6 external preentocristid, 7 entoconid, 8 posthypoconulidcristid, 9 hypoconulid, 10 prehypoconuldicristid, 11 posthypocristid, 12 hypoconid, 13 prehypocristid, 14 ectostylid, 15 postprotocristid, 16 protoconid, 17 preprotocristid, 18 anterior cingulid; (H) upper molar Lophiomerycidae dental nomenclature (based on the M2 of IVPP 6546-1): 1 postmetacrista, 2 metacone, 3 premetacrista, 4 mesostyle, 5 postparacrista, 6 paracone, 7 paraconid labial groove, 8 preparacrista, 9 parastyle, 10 preprotocrista, 11 anterolingual cingulum, 12 protocone, 13 postprotocrista, 14 entostyle, 15 additional cone, 16 premetaconulecrista, 17 metaconule, 18 postmetaconulecrista; (I) lower molar Tragulidae dental nomenclature (based on the m2 of IVPP V 6551, reversed): 1 metaconid, 2 external postmetacristid, 3 Dorcatherium fold, 4 internal postmetacristid, 5 preentocristid, 6 entoconid, 7 postentocristid, 8 posterior cingulid, 9 posthypocristid, 10 hypoconid, 11 prehypocristid, 12 ectostylid, 13 external postprotocristid, 14 Tragulus fold, 15 internal postprotocristid, 16 protoconid, 17 preprotocristid, 18 paraconid, 19 preparacristid. (J) phylogenetic position and stratigraphie of the Shinao/Yangjiachong/Xiaerhete ruminants (topology2). a stem Ruminantia, b Archaeomeryx, c Chiyoumeryx nov. gen. and Krabimeryx gracilis, d crown Ruminantia, e Iberomeryx miaoi nov. sp.; 1 lingual view, 2 occlusal view. Scale bare is 1 cm.
*v pars1982 Lophiomeryx gracilis—Miao: 532, Table 3, Figs. 6 and 720.
v non1982 Lophiomeryx gracilis?—Miao: 536, Fig. 820.
v pars1987 L. gracilis—Janis: 21133.
v pars1997 L. gracilis—Vislobokova: Fig. 321.
v pars2000 L. gracilis—Guo, Dawson, and Beard: 247, Table 214.
v pars2001 L. gracilis—Métais, Chaimanee, Jaeger, and Ducroq: 239, 24117.
v pars2012 L. gracilis—Mennecart: 6234.
Neodiagnosis
Krabimeryx gracilis has an m2 that is wider than the m3; this is the other way round in K. primitivus. Moreover, the entoconid is less anterior relative to the hypoconid in K. gracilis than it is in K primitivus. The ectostylid is large in K. gracilis, while it is absent in K. primitivus. The cingulum on the upper molars in K. gracilis is more developed than in K. primitivus.
Holotype
IVPP V 6546, partial skull with right and left M1–M3 (IVPP V 6546-1) and an associated right fragmented mandible with m2–m3 (IVPP V 6546-2) found in occlusion with the skull.
Additional material
IVPP V 6549, right m3 on fragmented mandible; IVPP V 6550 left fragmented mandible with m1–m2; IVPP V 26638, right m1. Measurements are given in Table S1.
Localities
Shinao Basin, Panxian County, Southwestern Guizhou, China; Xiaerhete locality, Jiminay County, Xingjiang, China. Late Eocene.
Taxonomical attribution
The herein described specimens were first attributed to the genus Lophiomeryx20. However, the thorough reassessment of the specimens now leads to the conclusion that Lophiomeryx gracilis sensu Miao20 contains three different species and genera, but none of them can be assigned to Lophiomeryx.
Based on the presence of a strong lingual cingulum in upper molars and a short anteroposteriorly oriented postprotocrista, as well as the absence of a premetacristid and an anterior fossa widely open in the lower molars, we can conclude that the specimens, IVPP V 6546-1, IVPP V 6546-2, IVPP V 6549, and IVPP V 6550, belong to Lophiomerycidae or Tragulidae35,36. However, the absence of a large paraconid and the absence of an elongated external postmetacristid distinguish the specimens from primitive Tragulidae17,36. In Zhailimeryx jingweni, the cuspids are more slender than in the herein described specimens14, a feature the taxon shares with K. primitivus. In Z. jingweni, m1 and m2 are of relative similar width14, while in K. primitivus and the herein described specimens from Shinao the m2 is clearly bigger than the m117. Similarly to K. primitivus, the herein described specimens differ from Z. jingweni in its lower molar lingual cusps being more laterally compressed, and in an entoconid that is slightly shifted to anterior with respect to the hypoconid, while it is more posterior in Z. jingweni14,17. Furthermore, K. primitivus and the herein described specimens from Shinao both lack the rudimentary paraconid present in Z. jingweni14,17.
Like K. primitivus, the here-described specimens differ from Chiyoumeryx nov. gen. (described below) and the Lophiomeryx species L. mouchelini, L. chalaniati and L. angarae by having more massive and more bunomorph lower molars16,17,24,34,37. Furthermore, Zhailimeryx jingweni, K. primitivus, and the herein described specimens differ from Lophiomeryx by the presence of a developed external postmetacristid and by a distinct entoconidian groove on the anterior side of the compressed entoconid14,17. In Lophiomeryx, the back fossa of m3 is widely open due to the strong reduction of the posthypoconulidcristid34,37. In contrast to this, Krabimeryx primitivus possesses a clearly developed posthypoconulidcristid forming a buckle on the m3 back basin17, similarly to the specimens from Shinao described here.
Summing up, the general morphology of the teeth in the herein described specimens is most similar to the one observed in K. primitivus. They both share a similar huge notch in lateral view between the third lobe of m3 and the entoconid and the entoconidian groove, features that clearly distinguishing them both from Lophiomeryx and Zhailimeryx. Thus, we attribute the specimens IVPP V 6546-1, IVPP V 6546-2, IVPP V 6549, and IVPP V 6550 to the genus Krabimeryx. However, significant differences occur with the type species, ruling out the synonymisation of K. gracilis nov. comb. and Krabimeryx primitivus. While both species are very similar in size, K. primitivus has an m3 wider than m2, while it is the converse for K. gracilis nov. comb. Moreover, the entoconid is less shifted to the anterior with respect to the hypoconid in K. gracilis nov. comb. than in K primitivus. There is no ectostylid in K. primitivus, while it is large in K. gracilis nov. comb., forming a transverse cristid between the protoconid and the hypoconid. The cingulum on the upper molars is more developed in K. gracilis nov. comb. than in K. primitivus.
Due to these differences we decided to create the new combination Krabimeryx gracilis nov. comb.
Chiyoumeryx nov. gen.
ZooBank LSID
urn:lsid:zoobank.org:act:464C46E0-5A69-4AC1-A9DD-8A7DF76D5CC0.
Etymology
Chiyou is a tribe leader of the ancient China, about 5–4 k years ago. Chiyou's tribe was believed to be in relation with the peoples in southern China; -meryx means ruminant in Greek.
Diagnosis
Chiyoumeryx nov. gen. differs from Zhailimeryx and Krabimeryx notably by the absence of the entoconidian groove. The lower teeth are more laterally compressed in Chiyoumeryx nov. gen. and the metaconid is linguo-labiallly more central than in the two other genera. The posthypoconulidcristid in the lower molars of Chiyoumeryx nov. gen. is longer than in Krabimeryx and its p4 is posteriorly extended, while this part is reduced in Krabimeryx. Chiyoumeryx nov. gen. differs from Lophiomeryx by the shape of the mandible. In Chiyoumeryx nov. gen. there is no diastema between p1 and p2 and the diastema between c and p1 is extremely reduced. The outline of the mandible in occlusal view is relatively straight in this species. Lophiomeryx possesses a long diastema between c and p1 and a small one between p1 and p2, as well as a regularly curved occlusal outline of the corpus. The lower premolars of Chiyoumeryx nov. gen. are laterally compressed giving a more elongated aspect to these teeth than in Lophiomeryx. The trigonid is smaller than the talonid in m1 and m2 in Chiyoumeryx nov. gen. and the preprotocristid terminates centrally and does not reach the lingual side. In Lophiomeryx the trigonid and talonid are of similar size and the preprotocristid is longer and reaches the lingual side. Moreover, in Chiyoumeryx nov. gen., the posthypoconulidcristid is longer than in Lophiomeryx. The shape of the P4 in Chiyoumeryx nov. gen. differs from the one in Lophiomeryx: the posterolingual crista does not meet the posterolabial crista.
Type species
Chiyoumeryx nov. gen. shinaoensis (Miao, 198220).
Included species
Chiyoumeryx nov. gen. flavimperatoris nov. sp.; ?Chiyoumeryx nov. gen. turgaicus (Flerow 193838).
Chiyoumeryx nov. gen. shinaoensis (Miao, 198220).
*v1982 Lophiomeryx shinaoensis—Miao: 530, Table 3, Figs. 3–520.
v1987 Lophiomeryx shinaoensis—Janis: 203, 204, 211, 212, Fig. 8B33.
v1997 Lophiomeryx shinaoensis—Vislobokova: Fig. 321.
v2000 L. shinaoensis—Guo, Dawson, and Beard: 247, Table 214.
v2001 L. shinaoensis—Métais, Chaimanee, Jaeger, and Ducroq: 239–241, 24117.
v2012 L. shinaoensis—Mennecart: 6234.
Neodiagnosis
Chiyoumeryx nov. gen. shinaoensis is bigger than Chiyoumeryx nov. gen. flavimperatoris nov. sp. but smaller than ?Chiyoumeryx turagicus. The transversely oriented anterior conid in the p4 in Chiyoumeryx nov. gen. shinaoensis differs from the obliquely oriented one in Chiyoumeryx nov. gen. flavimperatoris nov. sp. In Chiyoumeryx nov. gen. shinaoensis, the posterolingual conid is vestigial on p4. Chiyoumeryx nov. gen. shinaoensis has no anterior cingulid, while in Chiyoumeryx nov. gen. flavimperatoris nov. sp. there is a tiny anterior cingulid. Chiyoumeryx nov. gen. shinaoensis possesses lower crowns than ?Chiyoumeryx nov. gen. turgaicus. Chiyoumeryx nov. gen. flavimperatoris nov. sp. possesses an ectostylid, which is absent in ?Chiyoumeryx nov. gen. turgaicus.
Holotype
IVPP V 6531, right mandible with p2–m3 and tooth socket of p1.
Paratype
IVPP V 6532, right fragmented maxillary with P4–M3.
Additional material
IVPP V 6533, right mandible with p2–m3 and tooth socket of i1–p1; IVPP V 6534, left fragments mandible with m1–m3; IVPP V 6535, right fragmented mandible with m1–m3; IVPP V 6536, left fragmented mandible with p4–m3; IVPP V 6537, right fragmented mandible with p4–m2; IVPP V 6538, left p4; IVPP V 6539, right maxillary with P3–M3; IVPP V 6540, right maxillary with P4–M2; IVPP V 6541, right maxillary with M2–M3; IVPP V 6542, left maxillary with P3–M1; IVPP V 6543, right maxillary with M1–M3; IVPP V 6544, Left M3; IVPP V 6545, left maxillary with P4–M3. Measurements are given in Table S1.
Locality
Shinao Basin, Panxian County, Southwestern Guizhou, China. Late Eocene.
Taxonomical attribution
Miao20 attributed the here described specimens to the genus Lophiomeryx assuming that these fossils belong to a traguloid. “Lophiomeryx” shinaoensis clearly is a Lophiomerycidae: anterior and posterior fossae are open on the lower molars due to the absence of a premetacristid and the extreme reduction or absence of a postentocristid, there is no external postprotocristid, there is a mesolingual conid on the p4, the symphysis of the mandible extends backward up to the p12,36. It also shares with undisputable Lophiomerycidae a reduced posthypoconulidcristid that does not enclose the third lobe lingually.
“Lophiomeryx” shinaoensis differs from Zhailimeryx and Krabimeryx in the absence of the entoconidian groove14,17. Moreover, the teeth are more laterally compressed in “Lophiomeryx” shinaoensis and the metaconid is linguo-labially more centeral14,17. The posthypoconulidcristid in “Lophiomeryx” shinaoensis is more elongated than in Krabimeryx and its p4 has an extended posterior part, while it is reduced in Krabimeryx17.
Contrary to what was suggested by Métais and Vislobokova2, Miomeryx altaicus24 is currently known only by its holotype, which is an upper tooth row (AMNH 20383, see Matthew and Granger24). Comparable to M. altaicus, the postprotocrista reaches the premetaconulecrista on the M2 in “Lophiomeryx” shinaoensis. These two cristae fuse totally on the M3 in the here described specimens. However, even if both genera also bear a very strong cingulum, “Lophiomeryx” shinaoensis clearly differs from M. altaicus in having broader and squarer molars and straighter lingual cristae in the P4.
Miao20 compared the here revised fossils with the seven Lophiomeryx species considered valid at that time. Unfortunately, very few specimens document most of these species and there is considerable doubt considering the genus attribution of most of them34,36,37,38,39. In any case, we agree with Miao20 (p. 535) that “L. [= Praetragulus] gobiae is readily distinguished from other known Lophiomeryx species as well as from L. shinaoensis by the absence of p1, the anterior flange of metaconid joining protoconid crescent.”. Miao20 (p. 535) already noticed that “Lophiomeryx chalaniati, Lophiomeryx gaudry [= Iberomeryx minor], and Lophiomeryx benarensis are radically different from the present specimens in the anterior branches of the protoconid crescent [= preprotocristid], of m1 and m2 not reaching the lingual border while the posterior branches of hypoconid crescent [= posthypocristid], doing so”. “Lophiomeryx” shinaoensis shares this condition with the Mongolian Lophiomeryx angarae24. However, the trigonid is smaller than the talonid on m1 and m2 in “Lophiomeryx” shinaoensis and the preprotocristid ends in the labio-lingual axis of the molars, while trigonid and talonid are of more similar width combined with a longer preprotocristid in the European Lophiomeryx species and L. angarae16,34,37. The shape of the P4 in “Lophiomeryx” shinaoensis is very different from Lophiomeryx (see Brunet and Sudre37, Figs. 4 and 6). In Lophiomeryx, the posterolingual crista fuses with the posterolabial crista. In “Lophiomeryx” shinaoensis, the curved posterolingual crista does not join the distal end of the posterolabial crista but reaches the labial side. Furthermore, “Lophiomeryx” shinaoensis clearly differs from L. angarae L. mouchelini, and L. chalaniati in the shape of the mandible. These three species of Lophiomeryx possess a very elongated diastema between c and p1 and a small one between p1 and p224,36,37. As part of the genus diagnosis, Mennecart34 (p. 62 and p. 67), adapted from Brunet and Sudre37 and Métais and Vislobokova2, noticed that “the corpus mandibulae presents [in Lophiomeryx: L. angarae, L. mouchelini, and L. chalaniati24,34,37] a concave ventral profile just behind the mandible symphysis, then it becomes regularly convex until the beginning of the ramus, where there is a rounded incisura vasorum. […] On the anterior part of the mandible there are two foramen mentale.” Moreover he wrote that the “p1 is always reduced and leaf-like, separated from c and p2 by diastemata.” (Mennecart34, p. 67). In “Lophiomeryx” shinaoensis there is no diastema between p1 and p2 and the diastema between c and p1 is extremely reduced. The p1 is relatively big considering the root size. The lower outline of the mandible in lateral view is relatively straight. “Lophiomeryx” shinaoensis shares these characteristics with “Lophiomeryx” turgaicus40. Miao20 (p. 535) already noticed strong similarities between “Lophiomeryx” turgaicus and “Lophiomeryx” shinaoensis. The lower premolars of “Lophiomeryx” turgaicus and “Lophiomeryx” shinaoensis are strongly laterally compressed and the p4 is rectangular, giving the lower premolar toothrow an more elongated aspect than in L. angarae, L. mouchelini, and L. chalaniati20,24,30,38,40. Moreover, in these two species, the posthypoconulidcristid is of similar length, longer than in L. angarae, L. mouchelini, and L. chalaniati.
Based on these observations, we can assume that “Lophiomeryx” shinaoensis and “Lophiomeryx” turagicus cannot be assigned to the genus Lophiomeryx and may both belong to the same new Lophiomerycidae genus that we here name Chiyoumeryx nov. gen. Chiyoumeryx nov. gen. shinaoensis differs from ?Chiyoumeryx nov. gen. turgaicus nov. comb. in being lower crowned, smaller, possessing an ectostylid, having the symphysis starting under p1, and a shorter diastema.
Chiyoumeryx nov. gen. flavimperatoris nov. sp.
v1961 cf. Miomeryx sp.—Xu: 316, 323, 32426.
v pars1982 Lophiomeryx gracilis—Miao: 532, Table 3, Fig. 9a,b20.
v non1982 Lophiomeryx gracilis?—Miao: 536, Fig. 820.
1983 Lophiomeryx sp.—Wang & Zhang: 122, 12741.
v1983 cf. Miomeryx sp.—Wang & Zhang: 12341.
v1997 Miomeryx sp.—Vislobokova: Fig. 321.
v pars1997 L. gracilis—Vislobokova: Fig. 321.
v1999 cf. Miomeryx sp.—Zhang, Long, Ji, & Ding: 7, Table 527.
v pars2000 L. gracilis—Guo, Dawson, and Beard: 247, Table 214.
v pars2001 L. gracilis—Métais, Chaimanee, Jaeger, and Ducrocq: 239, 24117.
v2007 Miomeryx sp.—Métais and Vislobokova: 1942.
v pars2012 L. gracilis—Mennecart: 6234.
ZooBank LSID
urn:lsid:zoobank.org:act:1DF6F58C-F08B-4657-BD4A-7C597653926F.
Etymology
meaning yellow (flavor-) emperor (imperatoris) in latin. Chiyou fought with the Yellow Emperor, the ancestor of Chinese, but was defeated.
Diagnosis
Chiyoumeryx nov. gen. flavimperatoris nov. sp. shows the above-mentioned characteristics of the genus. Chiyoumeryx nov. gen. flavimperatoris nov. sp. is smaller than Chiyoumeryx nov. gen. shinaoensis and ?Chiyoumeryx nov. gen. turgaicus. The p4 of Chiyoumeryx nov. gen. flavimperatoris nov. sp. differs from Chiyoumeryx nov. gen. shinaoensis by an oblique anterior conid, which is labio-lingually oriented in the larger species. A very short posterolingual conid is located between the posterolabial cristid and the transverse cristid in the p4 of Chiyoumeryx nov. gen. flavimperatoris nov. sp., while it is absent on Chiyoumeryx nov. gen. shinaoensis. In Chiyoumeryx nov. gen. flavimperatoris nov. sp., there is a tiny anterior cingulid, while it is absent in Chiyoumeryx nov. gen. shinaoensis.
Holotype
IVPP V 6547, right mandible with p4–m3 (previously attributed to Lophiomeryx gracilis20).
Paratype
IVPP V 6548, left mandible with p4–m3 (previously attributed to Lophiomeryx gracilis20).
Additional material
IVPP V 2600, left p4–m2 (previously attributed to cf. Miomeryx sp.26). Measurements are given in Table S1.
Localities
Yangjiachong locality lying in the Caijiachong marls, Qujing, Yunnan, China; Shinao Basin, Panxian County, Southwestern Guizhou, China. Late Eocene.
Taxonomical attribution
IVPP V 6547 and IVPP V 6548 from Shinao were previously attributed to Lophiomeryx gracilis20, while IVPP V 2600 from Caijiachong marls was first described as cf. Miomeryx sp.26. All these specimens share the same size and dental morphology, and originate from a similar stratigraphic position. That is why we attribute them to the same species.
None of these specimens can be attributed to Krabimeryx or Zhailymeryx, as the entoconidian groove is absent14,17. Furthermore, the external postmetacristid is more marked in the considered specimens than in Krabimeryx and Zhailymeryx, forming a deep groove. The third basin is also very different in the here-described specimens from Krabimeryx and Zhailymeryx: the third lobe is a little tilted parallel with the prehypoconulidcristid and posthypoconulidcristid. The back fossa of m3 is very narrow.
Furthermore, the here-described specimens can be distinguished from K. gracilis (previously attributed to the same species), by a smaller size and a slenderer shape. The ectostylid is smaller than in K. gracilis. The anterior cingulid in the lower molars is stronger in K. gracilis than in the here-considered specimens. The small postentocristid (especially on m3) of the here-described specimens is absent in K. gracilis.
The here-described specimens possess all characteristics in the lower molars that are typical for Chiyoumeryx nov. gen. and distinguish this genus from Lophiomeryx24,34,37. Furthermore, as in Chiyoumeryx nov. gen. shinaoensis, the p4 is laterally compressed giving it a more elongated aspect than in Lophiomeryx24,34,37. Therefore, we consider it justified assigning the here-described specimens to Chiyoumeryx nov. gen. However, they differ from Chiyoumeryx nov. gen. shinaoensis in as smaller size and the morphology of the p4: (1) the anterior conid is oblique while it is labio-lingually oriented in Chiyoumeryx nov. gen. shinaoensis. (2) There is a tiny anterior cingulid that is absent in Chiyoumeryx nov. gen. shinaoensis. (3) There is no additional cristid on the mesolingual conid, which is a well-rounded conid, while in Chiyoumeryx nov. gen. shinaoensis, there is a short posterolingual cristid. (4) The posterolingual conid stands between the posterolabial cristid and the transverse cristid, while in Chiyoumeryx nov. gen. shinaoensis, the posterolingual conid is very small and oblique between the transverse cristid and the posterior stylid and does not join the posterolabial cristid. Due to these distinct differences we erect a new species: Chiyoumeryx nov. gen. flavimperatoris nov. sp.
Family Tragulidae Milne-Edwards, 186442.
Genus Iberomeryx Gabunia, 196443.
Diagnosis (modified from Mennecart et al.36)
Small-sized ruminant with upper molars possessing the following combination of characters: well-marked parastyle and mesostyle in small-column shape; strong paracone rib; metacone rib absent; metastyle absent; unaligned external walls of metacone and paracone; strong postprotocrista stopping against the anterior side of the premetaconulecrista; continuous lingual cingulum, stronger under the protocone. Lower dental formula is primitive (3–1–4–3) with non-molarized premolars. Tooth c is adjacent to i3. Tooth p1 is single-rooted, reduced and separated from c and p2 by a short diastema. The premolars have a well-developed anterior conid. Teeth p2–p3 display a distally bifurcated mesolabial conid. Tooth p3 is the largest premolar. Tooth p4 displays no mesolingual conid and a large posterior valley. Regarding the lower molars, the trigonid and talonid are lingually open with a trigonid more tapered than the talonid. The anterior fossa is open, due to a forward orientation of the preprotocristid and the presence of a paraconid. The internal postprotocristid is oblique and the external postprotocristid reaches the prehypocristid. The internal postprotocristid, postmetacristid and preentocristid are fused and Y-shaped. Protoconid and metaconid display a weak Tragulus fold and a well-developed Dorcatherium fold, respectively. The mandible displays a regularly concave ventral profile in lateral view, a marked incisura vasorum, a strong mandibular angular process, a vertical ramus, and a stout condylar process.
Type species
Iberomeryx parvus Gabunia, 196443 from Benara (Georgia), late Oligocene44.
Included species
I. minor45, Iberomeryx miaoi nov. sp.
Iberomeryx miaoi nov. sp.
v 1982 Lophiomeryx gracilis?—Miao: 536, Fig. 820.
ZooBank LSID
urn:lsid:zoobank.org:act:EE3F88E9-0EAF-4EC6-A46F-8623241E614B.
Diagnosis
Iberomeryx with a very large paraconid, which is smaller in Iberomeryx minor and Iberomeryx parvus. The metastylid is not strong but is more developed than in the other species. The ectostylid is big on m1, smaller on m2 and absent on m3, while I. minor displays an ectostylid on all molars and I. parvus none at all. Iberomeryx miaoi nov. sp. is of similar size to I. minor and its m2 is smaller than the one of I. parvus. It differs from I. minor by a thin anterior cingulid. Moreover, its protoconid is positioned slightly more anterior than in I. parvus. The molars appear to be more massive and bulkier in this species than in I. minor and I. parvus.
Holotype
IVPP V 6551, left mandible with m1–m3 (only specimen known). m1 5.1 × 3.5, m2 5.2 × 4.1, m3 8.0 × 4.0.
Etymology
We dedicate this species to Prof. Miao Desui who was the first to describe the Shinao fauna.
Locality and horizon
Shinao Basin, Panxian County, Southwestern Guizhou, China. Late Eocene.
Taxonomical attribution
This minute ruminant was referred to Lophiomeryx gracilis? by Miao20. However, he already noticed that the size of this individual was smaller than in the other specimens attributed to Lophiomeryx gracilis. Miao20 excluded an attribution of IVPP V 6551 to "Lophiomeryx" gaudryi due to a closed posterior section of the posterior fossa on the m3. However, in both teeth, the posterior fossa is still open by the reduction of the postentocristid.
The here-described specimen clearly differs from Lophiomeryx by the presence of an external postmetacristid forming a slight Dorcatherium fold, a developed external postprotocristid (clearly visible at least on m2), and a large paraconid36. Furthermore the external postprotocristid and prehypocristid are connected on their distal ends and the third basin of m3 forms a well-formed buckle, unlike the condition in Lophiomerycidae14,16,33,36,37. The combination of these characters is typical for Tragulidae36.
Very few taxa are so far known in the early evolution of the Tragulidae. Only Archaeotragulus, Iberomeryx, and Nalameryx are recognized as potential Paleogene Tragulidae17,36,46, of which Archaeotragulus is currently the oldest representative described17,47. Archaeotragulus possesses lower molars with a broadened talonid in comparison to the trigonid and displays an entoconidian groove36. In the case of IVPP V 6551, the trigonid and talonid are of similar size and no specific entoconidian groove can be observed. Mennecart et al.36 considered Nalameryx a Tragulidae notably based on the presence of the M structure (the external postmetacristid, the internal postmetacristid, the internal postprotocristid, and the external postprotocristid are interconnected forming a M in occlusal view), including the Tragulus fold and Dorcatherium fold, and the absence of a rounded mesolingual conid in the p435. IVPP V 6551 differs from Nalameryx in having an m3 wider than m1 and similar m1 and m2 widths17. In size proportions and molar morphology, IVPP V 6551 resembles the genus Iberomeryx. In IVPP V 6551, the relative size of the m2 is more similar to I. minor. In Iberomeryx minor, the anterior cingulid is big36,46, while in Iberomeryx parvus the cingulid is thin48 like in IVPP V 6551. The teeth of IVPP V 6551 appear to be more massive and bulkier than in I. minor and I. parvus36,48. Similarly to I. minor, the protoconid of IVPP V 6551 is a little more anterior than in I. parvus36,48. IVPP V 6551 clearly differs from I. parvus and I. minor by the presence of a very large paraconid, which is smaller in the two other species36,48. Moreover, the metastylid in IVPP V 6551 is slightly more developed than in I. minor and not present in I. parvus43,48. Iberomeryx minor displays an ectostylid on all molars36, while this structure is absent from I. parvus48. The ectostylid in IVPP V 6551 is large on m1 to absent on m3. Based on these differences we decided to erect the new species Iberomeryx miaoi nov. sp.
Origin of crown Ruminantia and dispersal pattern of Paleogene Eurasian ruminants
So far five families and 13 genera of Ruminantia are known during the middle and late Eocene in Eurasia2,18,19. Based on molecular data, the origin of crown ruminants should be searched for between the latest late Paleocene (56.5 Ma) and the latest early Oligocene (29 Ma)49,50. With the description of stem Tragulidae from the early Oligocene of Western Europe (Iberomeryx) and the late Eocene from Southern Thailand (Archaeotragulus)17, Mennecart et al.26 and Mennecart and Métais51 verified that the oldest crown ruminants date back at least to the latest Eocene (34 Mya). The presence of the tragulid genus Iberomeryx in Shinao, Southern China, further confirms this and may actually represent the oldest fossil of a Tragulidae known and thus of a crown Ruminantia (37–35 Mya, Fig. 1), since no Pecora is known during the Eocene so far51.
The here presented reassesment of the Shinao ruminants in combination with literature data reveals a clear pattern in the distribution of Eocene ruminants. Among Archaeomerycidae, Archaeomeryx and Miomeryx are found in Northern and Central Asia [Kazhakstan, Mongolia, and northern part of China2,21,53 (see Fig. 2)]. The lophiomerycid Lophiomeryx (as Lophiomeryx angarae) as well as the Asiatic Praetragulidae (Praetragulus) occupy the same area2. The Mongolian Lophiomeryx angarae is most likely closely related to the European species Lophiomeryx mouchelini. Due to the strong morphological similarities, some specimens of L. mouchelini were actually first described as Lophiomeryx cf. angarae54. Lophiomeryx mouchelini or its ancestors arrived in Europe with the Grande-Coupure dispersal event at the Eocene–Oligocene transition ca. 34 Mya ago (oldest European records: Calaf, Spain, MP22; Möhren 9, Germany, MP21-22; age comprised between the German localities Haag2 MP21 and Möhren 13 MP2234,37,53). The close relationship of these European and the Mongolian species confirms that the origin of the Grande-Coupure cohort may be deeply anchored in the Eocene of Central-Northern Asia (Fig. 2).
The Southern part of Asia presents a totally different ruminant community at the genus level and includes the Archaeomerycidae Indomeryx and Notomeryx, the Lophiomerycidae Krabimeryx and Chiyoumeryx nov. gen., the Bachitheriidae Bachitherium and the Tragulidae Archaetrogulus and Iberomeryx2,17,18,19,21,53 (see Fig. 2). The oldest Bachitherium is currently known from the Balkan area during the Eocene18,19. The Tethys Ocean separated this area from Western Europe until its progressive disappearance during the Oligocene, ca. 31 Mya55,56. Bachitherium and a cohort of rodents (Pseudocricetodon, Paracricetodon, and the Melissodontinae)19 did not reach Western Europe prior to the opening of this passage. Similarly to the genus Bachitherium, Iberomeryx arrived in Western Europe after the drying out of the Tethys Ocean ca. 31 Mya, during the Bachitherium dispersal event18,19,36. Iberomeryx is mainly known from the middle early Oligocene of Western Europe34,36,57 and the late Oligocene of Anatolia and Georgia43,48,58. Discovering Iberomeryx in the Eocene of Eastern Asia confirms an Asiatic origin of this genus. The close relationship between South-eastern Europe and South-eastern Asia is furthermore supported by anthracotheriids (extinct artiodactyls related to hippopotamids) and rhinocerotoids59,60.
Mennecart et al.18,19 proposed that mammals originating from Asia arrived in Western Europe during the early Oligocene in two faunal events: the Grande-Coupure, ca. 33.9 Mya and the Bachitherium dispersal event, ca. 31 Mya. These two faunal events imply two different and diachronous ways of dispersal. The fact that Eocene taxa from South-eastern Asia did not arrive in Western Europe prior to 31 Mya indicates that the Bachitherium dispersal Event cohort might be deeply anchored in the Eocene of Southern Asia (Fig. 2), while genera recorded from the Eocene of Central Asia are known to have arrived already during the Grande-Coupure and thus originated from a different palaeobiogeographic province. The Grande-Coupure was a dispersal event using a Northern way over the closed Turgai Strait and probably originating from Central Asia (Fig. 2). The Bachitherium dispersal event is a stepwise story with a first dispersion from Southern Asia to South-eastern Europe along the Southern path (Fig. 2) and then the dispersal throughout Europe thanks to the closure of the Tethyian Ocean18,19.
The south-eastern part of Asia has shown very few changes from a warm and humid climate and environment since the Eocene4, while Northern Asia underwent a transition from warm and humid subtropical environments during the Eocene to steppe environments in the Pliocene, e.g.3,4,5. In this light it is not surprising that an increasing number of paleontological and geological studies indicate that Asia had already experienced a strong latitudinal environmental zonation during the middle and the late Eocene, e.g.6,13.
These different climatic and environmental conditions in Central and South Asia led to two distinct palaeobiogeographical provinces clearly traceable in assemblages of herbivores like ruminants that was already apparent during the Eocene. The Central Asian ruminants were living in a more arid environment than the ones from South-eastern Asia (see Fig. 2). The tropical and wet environments from the South-eastern Asia led to the emergence of the Tragulidae (Iberomeryx and Archaeotragulus) and of the anthracotheriids.
Materials and methods
Materials
The fossils are housed at the Institute of Vertebrate Paleontology and Paleoanthropology of the Chinese Academy of Sciences in Beijing. Precise descriptions and measurements of the species can be found in Supplementary data 1.
Methods
Measurements have been realized thanks to a calliper (precision 0.2 mm) and can be found in Table S1. The dental nomenclature was modified after Bärmann and Rössner61 (see Fig. 1).
Conventional abbreviations used in front of the year in the synonymy list follow Matthews62: * = the work validates the species; v = the authors have seen the original material of the reference; pars = the reference applies only in part to the species under discussion; non = the reference actually does not belong to the species under discussion; no sign = the authors were unable to check the validity of the reference. Years in italics indicate a work without description or illustration.
Abbreviations
IVPP, Institute of Vertebrate Paleontology and Paleoanthropology of the Chinese Academy of Sciences in Beijing (China); AMNH, American Museum of Natural History (USA). p/P, lower/upper premolar’ m/M, Lower/upper molar.
Nomenclatural acts
This published work and the nomenclatural acts it contains, have been registered in ZooBank:
-
Chiyoumeryx nov. gen.: 464C46E0-5A69-4AC1-A9DD-8A7DF76D5CC0.
-
Chiyoumeryx nov. gen. flavimperatoris nov. sp.: 1DF6F58C-F08B-4657-BD4A-7C597653926F.
-
Iberomeryx miaoi nov. sp.: EE3F88E9-0EAF-4EC6-A46F-8623241E614B.
Change history
21 September 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41598-021-98946-1
References
Gentry, A. W., Rössner, G. E. & Heizmann, E. P. J. Suborder Ruminantia. In The Miocene Land Mammals of Europe (eds Rössner, G. E. & Heissig, K.) 225–258 (Verlag Dr. Friedrich Pfeil, 1999).
Métais, G. & Vislobokova, I. Basal ruminants. In The Evolution of Artiodactyls (eds Prothero, D. R. & Foss, S. C.) 189–212 (The Johns Hopkins University Press, 2007).
Qiu, Z. D. & Li, C. K. Evolution of Chinese mammalian faunal regions and elevation of the Qinghai-Xizang (Tibet) Plateau. Sci. China Ser. D Earth Sci. 48, 1246–1258 (2005).
Licht, A. et al. Asian monsoons in a late Eocene greenhouse world. Nature 513, 501–506 (2014).
Ruddiman, W. F. & Kutzbach, J. E. Forcing of late Cenozoic Northern Hemisphere climate by plateau uplift in Southern Asia and the American West. J. geophys. Res. 93, 409–427 (1989).
Gong, Y.-X. et al. Dietary reconstruction and palaeoecology of Eocene Lophialetidae (Mammalia: Tapiroidea) from the Erlian Basin of China: Evidence from dental microwear. Hist. Biol. https://doi.org/10.1080/08912963.2020.1722660 (2020).
Abels, H. A., Dupont-Nivet, G., Xiao, G., Bosboom, R. & Krijgsman, W. Step-wise change of Asian interior climate preceding the Eocene-Oligocene Transition (EOT). Palaeogeogr. Palaeoclimato. 299, 399–412 (2011).
Zhang, Z. et al. Early Eocene Asian climate dominated by desert and steppe with limited monsoons. J. Asian Earth Sci. 44, 24–35 (2012).
Kaya, M. Y. et al. Paleogene evolution and demise of the proto-Paratethys Sea in Central Asia (Tarim and Tajik basins): Role of intensified tectonic activity at ca. 41 Ma. Basin Res. 31, 461–486 (2019).
Bosboom, R. et al. Late Eocene palaeogeography of the proto-Paratethys Sea in Central Asia (NW China, southern Kyrgyzstan and SW Tajikistan). Geol. Soc. Spec. Publ. 427, 565–588 (2015).
Bosboom, R. E. et al. Aridification in continental Asia after the Middle Eocene Climatic Optimum (MECO). Earth Planet. Sci. Lett. 389, 34–42 (2014).
Ramstein, G., Fluteau, F., Besse, J. & Joussaume, S. Effect of orogeny, late motion and land-sea distribution on Eurasian climate change over the past 30 million years. Nature 386, 788–795 (1997).
Tsubamoto, T., Takai, M. & Egi, N. Quantitative analyses of biogeography and faunal evolution of middle to late Eocene mammals in East Asia. J. Vertebr. Paleontol. 24, 657–667 (2004).
Guo, J., Dawson, M. & Beard, K. C. Zhailimeryx, a new Lophiomerycid Artiodactyl (Mammalia) from the Late Middle Eocene of Central China and early evolution of ruminants. J. Mamm. Evol. 7, 239–258 (2000).
Scherler, L., Mennecart, B., Hiard, F. & Becker, D. Evolution of terrestrial hoofed-mammals during the Oligocene-Miocene transition in Europe. Swiss J. Geosci. 106, 349–369 (2013).
Mennecart, B. The European ruminants during the “Microbunodon Event” (MP28, latest Oligocene): Impact of climate changes and faunal event on the ruminant evolution. PLoS ONE 10, e0116830 (2015).
Métais, G., Chaimanee, Y., Jaeger, J.-J. & Ducrocq, S. New remains of primitive ruminants from Thailand: evidence of the early evolution of the Ruminantia in Asia. Zool. Scr. 30, 231–248 (2001).
Mennecart, B., Geraads, D., Spassov, N. & Zagorchev, I. Discovery of the oldest European ruminant in the late Eocene of Bulgaria: Did tectonics influence the diachronic development of the Grande Coupure. Palaeogeogr. Palaeoclimatol. Palaeoecol. 498, 1–8 (2018).
Mennecart, B., Predrag, R. & Zoran, M. New data on the earliest European ruminant (Mammalia, Artiodactyla): A revision of the fossil mandible from Rusce in the Pčinja basin (late Eocene, Southeastern Serbia). Palaeontol. Electron. 21, 1–12 (2018).
Miao, D. Early Tertiary fossil mammals from the Shinao Basin, Panxian County, Guizhou Province. Acta Palaeontol. Sinica 21, 526–536 (1982).
Vislobokova I. A. Eocene-early Miocene ruminants in Asia in Biochrom'M 1997 (eds Aguilar J.-P., Legendre S., Michaux J.) 215–223. (Ecole Pratique des Hautes Etudes Institut de Montpellier, 1997).
Averianov, A., Danilov, I., Chen, W. & Jin, J. A new brontothere from the Eocene of South China. Acta Palaeontol. Pol. 63, 189–196 (2018).
Bai, B. Eocene Pachynolophinae (Perissodactyla, Palaeotheriidae) from China, and their palaeobiogeographical implications. Palaeontology 60, 837–852 (2017).
Matthew, W. D. & Granger, W. New ungulates from the Ardyn Obo Formation of Mongolia. Am. Mus. Novit. 195, 1–12 (1925).
Zdansky, O. Die alttertiären Säugetiere Chinas nebst stratigraphischen Bemerkungen. Palaeontol. Sinica C. 6, 1–87 (1930).
Xu, Y.-X. Some Oligocene mammals from Chuching, Yunnan. Vert. PalAs. 4, 315–325 (1961).
Zhang, Y., Long, Y., Ji, H. & Ding, S. The Cenozoic deposits of the Yunnan region. Professional Papers Stratigr. Paleontol. 7, 1–21 (1999).
Stidham, T. A. & Ni, X.-J. Large anseriform (Aves: Anatidae: Romainvilliinae?) fossils from the Late Eocene of Xinjiang, China. Vert. PalAs. 52, 99–111 (2014).
von Linnaeus, C. Systema naturæ per regna tria naturæ, secundum classes, ordines, genera, species, cum characteribus, differentiis, synonymis, locis. (Editio decima reformat, 1758).
Owen, R. Description of teeth and proportion of jaws of two extinct Anthracotherioid quadrupeds (Hyopotamis vectianus and Hyopotamus bovinus) discovered by the Marchioness of Hastings in the Eocene deposits on the N.W. coast of the Isle of Wight: With an attempt to develop Cuvier’s idea of the classificiation of the pachyderms by the number of their toes. Quart. J. Geol. Soc. London 4, 103–141 (1848).
Scopoli, G. A. Introductio ad Historiam Naturalem Sistens Genera Lapidum, Plantarum, et Animalium: Hactenus Detecta, Caracteribus Essentialibus Donata. In Tribus Divisa, Subinde ad Leges Naturae (eds Scopoli, G. A. & Gerle, W.) 1–540 (Apud Wolfgangum Gerle, 1777).
Flower, W. H. On the arrangement of the orders and families of existing Mammalia. Proc. Zool. Soc. Lond. 39, 178–186 (1883).
Janis, C. M. Grades and clades in hornless ruminants evolution: The reality of the Gelocidae and the systematic position of Lophiomeryx and Bachitherium. J. Vertebr. Paleontol. 7, 200–216 (1987).
Mennecart, B. The Ruminantia (Mammalia, Cetartiodactyla) from the Oligocene to the Early Miocene of Western Europe: Systematics, palaeoecology and palaeobiogeography. GeoFocus 32, 1–263 (2012).
Métais, G., Welcomme, J.-L. & Ducrocq, S. New lophiomerycid ruminants from the Oligocene of the Bugti Hills (Balochistan, Pakistan). J. Vertebr. Paleontol. 29, 231–241 (2009).
Mennecart, B., Becker, D. & Berger, J.-P. Iberomeryx minor (Mammalia, Artiodactyla) from the Early Oligocene of Soulce (Canton Jura, NW Switzerland): Systematics and palaeodiet. Swiss J. Geosci. 104, S115–S132 (2011).
Brunet, M. & Sudre, J. Evolution et systématique du genre Lophiomeryx POMEL 1853 (Mammalia, Artiodactyla). Münch. Geowiss. Abh. 10, 225–242 (1987).
Flerow, K. K. On the remains of the Ungulata from Betpakdala. C. R. Acad. Sci. 21, 95–96 (1938).
Vislobokova, I. A. A new representative of the Hypertraguloidea (Tragulina, Ruminantia) from Khoer-Dzan Locality in Mongolia, with remarks on the relationships of the Hypertragulidae. Am. Mus. Novit. 3225, 1–24 (1998).
Trofimov, B. A. Nouvelles données sur les Ruminantia les plus anciens d’Asie. Cursillos y Conf. Inst. ‘Lucas Mallada’ 4, 137–141 (1957).
Wang, B. & Zhang, Y. New finds of fossils from Paleogene of Qujing, Yunnan. Vert. PalAs. 21, 119–128 (1983).
Milne-Edwards, A. Recherches anatomiques, zoologiques et paléontologiques sur la famille des chevrotains. Ann. Sci. Nat. Paris 5, 1–167 (1864).
Gabunia, L. The Oligocene Mammalian Fauna of Benara 268 (Academy of Sciences of URSS, 1964).
Lucas, S. G. & Emery, R. J. Taxonomy and biochronological significance of Paraentelodon, a giant Entelodon (Mammalia, Artiodactyle) from the Late Oligocene of Eurasia. J. Vertebr. Paleontol. 19, 160–168 (1999).
Filhol, H. Découverte de quelques nouveaux genres de mammifères fossiles dans les dépôts de phosphate de chaux de Quercy. C. R. Acad. Sci. 94, 138–139 (1882).
Sudre, J. Cryptomeryx Schlosser, 1886, tragulidé de l’Oligocène d’Europe relations du genre et considérations sur l’origine de ruminants. Palaeovertebrata 14, 1–31 (1984).
Rössner, G. E. Family Tragulidae. In The Evolution of Artiodactyls (eds Prothero, D. R. & Foss, S. C.) 213–220 (The Johns Hopkins University Press, 2007).
Métais, G. et al. Oligocene ruminants from the Kızılırmak Formation, Çankırı-Çorum Basin, Central Anatolia, Turkey. Palaeontol. Electron. 19, 1–23 (2016).
Bibi, F. A. Multi-calibrated mitochondrial phylogeny of extant Bovidae (Artiodactyla, Ruminantia) and the importance of the fossil record to systematics. BMC Evol. Biol. 13, 166 (2013).
Hassanin, A. et al. Pattern and timing of diversification of Cetartiodactyla (Mammalia, Laurasiatheria), as revealed by a comprehensive analysis of mitochondrial genomes. C. R. Biol. 335, 32–50 (2012).
Mennecart, B. & Métais, G. Mosaicomeryx gen. nov., a ruminant mammal from the Oligocene of Europe and the significance of ‘gelocids’. J. Syst. Palaeontol. 13, 581–600 (2015).
Scotese, C. R. Atlas of Paleogene Paleogeographic Maps (Mollweide Projection). Cenozoic PALEOMAP Atlas ArcGIS PALEOMAP Project Evanston IL 1, 8–15 (2014).
Russel, D. E. & Zhai, R.-J. The Paleogene of Asia: Mammals and stratigraphy. Mém. Mus. Natl Hist. Nat. 52, 1–488 (1987).
Heissig, K. Fossilführende Spaltenfüllungen Süddeutschlands und die Ökologie ihrer oligozänen Huftiere. Mitt. Bayer. Staatssamml. Paläontol Geol. 18, 237–288 (1978).
Berger, J.-P. et al. Paleogeography of the Upper Rhine Graben (URG) and the Swiss Molasse Basin (SMB) from Late Eocene to Pliocene. Int. J. Earth Sci. 94, 697–710 (2005).
Berger, J.-P. et al. Eocene-Pliocene time scale and stratigraphy of the Upper Rhine Graben (URG) and the Swiss Molasse Basin (SMB). Int. J. Earth Sci. 94, 711–731 (2005).
Weidmann, M. et al. Paléontologie et biostratigraphie de la Molasse de l’Oligocène et du Miocène basal du Talent et d’autres localités du Plateau vaudois (Suisse). Rev. Paléobiol. 33, 463–531 (2014).
Gabunia, L. Sur les Mammifères oligocènes du Caucase. Bull. Soc. Géol. Fr. 7, 857–869 (1966).
Böhme, M. et al. Na Duong (northern Vietnam)—An exceptional window into Eocene ecosystems from Southeast Asia. Zitteliana 53, 121–167 (2013).
Tissier, J. et al. New data on Amynodontidae (Mammalia, Perissodactyla) from Eastern Europe: Phylogenetic and palaeobiogeographic implications around the Eocene-Oligocene transition. PLoS ONE 13, e0193774 (2018).
Bärmann, E. V. & Rössner, G. E. Dental nomenclature in Ruminantia: Towards a standard terminological framework. Mamm. Biol. 76, 762–768 (2011).
Matthews, S. C. Notes on open nomenclature and on synonymy lists. Palaeontology 16, 713–719 (1973).
Acknowledgements
BM would like to thank Ursula Göhlich and Gudrun Daxner-Höck (Naturhistorisches Museum Wien, Austria), Ruth O’Leary, Judith Galkin, Jin Meng, Suzann Goldberg, Susan K. Bell, and Mark Norell (American Museum of Natural History, USA), and Loïc Costeur (Naturhistorisches Museum Basel, Switzerland) for providing access to comparative material and helpful discussions. WS would like to thank Wei Gao for photographying the specimenV26638. BM would like to thank the Swiss National Science Foundation is warmly thanked for granting the SNF project P300P2_161065 and P3P3P2_161066. WS would like to thank the Strategic Priority Cultivating Research Program, CAS (Grants No. XDB26000000, XDA 20070203). The authors are very grateful for the constructive and encouraging suggestions from the reviewer Christine Janis who greatly improved the manuscript with her comments and helped with the English, thank you very much, the editor Chenglong Deng for its encouraging feedback, and anonymous reviewer1.
Author information
Authors and Affiliations
Contributions
B.M. and S.Q. designed the study, B.M. wrote the paper, S.Q., M.A., Y.L., and C.L. provided critical comments. All authors gave final approval for publication.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: In the original version of this Article, ShiQi Wang was omitted as a corresponding author. Correspondence and requests for materials should also be addressed to wangshiqi@ivpp.ac.cn.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mennecart, B., Aiglstorfer, M., Li, Y. et al. Ruminants reveal Eocene Asiatic palaeobiogeographical provinces as the origin of diachronous mammalian Oligocene dispersals into Europe. Sci Rep 11, 17710 (2021). https://doi.org/10.1038/s41598-021-96221-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-96221-x
This article is cited by
-
New materials of Lophiomeryx (Artiodactyla: Lophiomerycidae) from the Oligocene of Nei Mongol, China
Journal of Mammalian Evolution (2023)
-
Ruminant inner ear shape records 35 million years of neutral evolution
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.