Abstract
Considering the dynamic nature of body mass index (BMI) and its importance in determining cardiovascular risks, this study aimed to investigate the life-course trajectory pattern of women’s BMI and its association with cardiovascular risk factors. A total of 1356 couples with 2976 children were recruited and followed up for an average period of 20 years. Latent growth curve modeling was applied to determine women's BMI trajectories; logistic regression was used to investigate the associations between trajectory patterns and cardiovascular risk factors, including hypertension (HTN), dyslipidemia, diabetes mellitus (DM), and obesity. Women were classified into three trajectories, including normal, stage 1 obesity, and stage 2 obesity. Compared to women’s in the normal trajectory group, those in obesity trajectories had higher odds ratios for HTN, DM, and dyslipidemia. Men with obese spouses showed a higher rate of HTN 1.54 (95% CI 1.05–2.25) and DM 1.55; (95% CI 1.00–2.44). The odds of men’s obesity were higher in obese spouses (OR 1.70; 95% CI 1.10–2.62). Offspring of stage 2 obese (OR 2.39; 95% CI 1.67–3.44) and stage 1 obese (OR 4.81; 95% CI 3.16–7.34) mothers were more likely to be obese. Our findings emphasized paying more attention to women with excessive weight to promote familial cardiovascular health in the communities.
Similar content being viewed by others
Introduction
Cardiovascular diseases (CVDs) are the leading causes of death in developed and developing countries1,2. The worldwide mortality rate from CDVs showed a rising trend, up to 15%, between 2006 and 20161. According to existing evidence, CVDs are also lead to 12% of global disability during life years3. In Iran, around 50% of deaths are caused by CVDs4. Obesity, diabetes mellitus (DM)5, dyslipidemia, and hypertension (HTN)6 as well-documented risk factors for CVDs, could significantly threaten individuals' health. It is predicted that by 2030, a total of 2.16 and 1.12 billion individuals will be overweight and obese, respectively, and 30% and 9.3% of adults worldwide have hypertension and DM, respectively7,8,9. In Iran, the rate of excessive weight is considerably high; recent data showed 12.8–76.4 and 2.4–35.4% of Iranian adults suffer from overweight and obesity, respectively8. Moreover, the prevalence of hypertension and DM were 29.9% and 9.94%, respectively10,11.
The familial environment has been proposed as an essential contributor to CVD risk factors, including obesity12. Socioeconomic status (SES) and parental modeling of eating behaviors are examples of familial factors that have been related to developing CVD risk factors13,14,15. A growing number of studies showed the association between parental obesity and their offsprings’ weight status16,17,18. Studies indicated parental body mass index (BMI), particularly mother’s BMI, as the primary determinant of excessive weight gain in children during the life span from childhood to adulthood. It is significantly contributed to other cardio-metabolic risk factors19. However, the relationships between mothers’ weight status and metabolic complications in offspring are under debate. This issue of whether offspring with obese mothers are at higher risk for cardiometabolic impairment needs to be determined20.
In addition to offspring, weight status in women may affect their spouses’ cardiovascular risk factors. A study assessed the reciprocal effects of couple obesity and indicated that excessive weight in wives increased the risk of type 2 diabetes in their husbands; however, this effect was not observed in women21. This finding indicates the potential role of women’s weight status in association with their husbands’ cardiovascular risk factors. The spousal concordance, defined as consistency in health status between a husband and a wife, has been well known22. Several cross-sectional studies on spousal concordance have been conducted in chronic diseases, including hypertension23,24, cardiovascular diseases25,26, cancers, and type 2 diabetes27, which may be explained by the shared environment22. In Iran, the spousal concordance for type 2 diabetes and hypertension has been previously shown in cross-sectional studies28,29.
Several studies investigated the cross-sectional association between BMI and CVD risk factors30. Due to the changes in BMI patterns in recent years, the growth trajectories have been used to assess the life-course prevention of CVDs. A few studies are available on the long-term life-course change of BMI; however, no findings are revealed on the cardiovascular risk factors associated with the life-long change of women’s BMI in familial association28,31,32,33. In addition, one cannot extend these findings due to the different socio-environmental factors among populations. To the best of our knowledge, this is the first longitudinal study in the world that aimed to determine the life-course trajectory patterns of women’s BMI and identify which trajectory pattern was associated with a higher cardiovascular risk in whole family members as well as women’s cardiovascular risk factors.
Methods
Study design and population
This study used data from the Tehran Lipid and Glucose Study (TLGS), an ongoing population-based cohort with every-three-year measurements to determine the prevalence of non-communicable diseases (NCDs) risk factors in a representative sample of residents of district no.13 of Tehran. The first survey, including 15,005 participants (women and men, aged ≥ three years), was initiated in 1999–2001. Details of the TLGS have previously been published34.
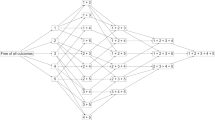
For the present study, 2556 couples were selected from the first phase and followed for 20 years (till phase 6); after the exclusion of 330 couples with reported widowhood or divorce, 2226 couples remained. Afterward, mothers with at least four missing measurements on BMI were excluded (n = 870). The final analytic sample for latent class growth analysis (LCGA) comprises 1356 mothers with 2976 children (51.8% girls), aged ≤ 18 years at baseline. The study design was shown in Fig. 1. Written informed consent was obtained from all participants. The study protocol was approved by the ethics committee of the Research Institute for Endocrine Sciences (RIES) of the Shahid Beheshti University of Medical Sciences.
Measurements
Trained interviewers collected data on age, sex, educational level, marital status, smoking status, physical activity, and medical and drug history using a pretested questionnaire. Blood pressure was recorded in a sitting position using a standard mercury sphygmomanometer with the cuff placed on the right arm. Two measurements were taken twice at the interval of 5 min each, and mean values were considered for systolic and diastolic blood pressure (SBP and DBP)34. Blood glucose and lipid levels, including triglycerides (TGs) and high-density lipoprotein (HDL-C), total cholesterol (TC), fasting plasma glucose (FPG), and 2‐hour post‐load plasma glucose (2 h‐PLPG), were measured using a morning blood sample which was obtained from participants in each phase at the TLGS research laboratory on the day of blood collection34. Weight of individuals was measured using digital scales, in light clothing and without shoes, and recorded to the nearest 100 g. Height was measured using a tape meter. BMI was calculated as weight in kilograms divided by square of height in meters. Reliable and validated the Modifiable Activity Questionnaire (MAQ) was used to measure three forms of activities, including leisure time, job, and household activities in the past year based on MET-minutes/week35.
Definition of terms
Hypertension was defined as mean SBP ≥ 140 mm Hg or mean DBP ≥ 90 mm Hg or taking antihypertensive drugs36, and DM was defined as FPG ≥ 7 mmol/L or two hour‐PLPG ≥ 11.1 mmol/L37 or using glucose‐lowering medications. Dyslipidemia was defined as TG ≥ 1.69 mmol/L or HDL‐C levels < 1.04 mmol/L in men and < 1.29 mmol/L in women or TC ≥ 5.2 mmol/L or using lipid-lowering medications38. A BMI equal to or more than 30 kg/m2 in adults is defined as obesity39. Education level was classified into three categories: primary, secondary, and higher. Smoking status was classified as a smoker (current smokers) and nonsmoker (past or never smokers). A person who smokes cigarettes or uses other tobacco products daily or occasionally was considered as current smoker.
Statistical analysis
Baseline characteristics of the mothers, fathers, and their offspring were summarized as means ± SD and frequencies (percentages) for continuous and categorical variables, respectively. Results were stratified based on mother trajectories. Continuous and categorical variables were compared between mother trajectories using the one-way ANOVA and the Chi-square tests.
LCGA is a semi‐parametric technique used to identify distinct subgroups of individuals following a similar pattern of change over time on a given variable. In the current analysis, this approach identifies patterns of BMI, which determines mothers with similar behavioral trajectories. We applied the PROC TRAJ procedure extension for SAS to build group-based multi-trajectory models via a particular application of finite mixture modeling. Model selection was made in two steps. The number of trajectory groups was determined based on the Bayesian information criterion (BIC) and substantive significance40. In the second step, we tested the various shapes of each latent class to identify the pattern of change over time (linear, quadratic, or cubic). In all analyses, the minimal class sizes of at least 5% of the sample were considered. The models' goodness of fit was assessed using the average posterior probability (APP) of group membership ≥ 70% and that the odds of correct classification (OCC) ≥ 5 for each group and similarities between the estimated probability of the trajectory group and the proportion assigned to the group as suggested by Nagin40. Following the identification of the trajectory groups, each group was assigned a label regarding their pattern of BMI during the follow-up phases.
We examined the associations of each lifetime BMI trajectory group with cardiometabolic risk factors by logistic regression models. BMI trajectory group as a predictor was included in models, and the normal group was considered as a reference category. The first model was unadjusted; the second model was adjusted for age, education, employment, and marital status, and the third model was further adjusted for smoking status and physical activity. Also, in assessing the association between mother's trajectory groups and her offspring, sex was also considered in all mentioned models. Statistical analyses were performed using SAS (version 9.4; SAS Institute Inc) and IBM SPSS Statistics version 22. P < 0.05 was considered statistically significant.
Ethics approval and consent to participate
This study was approved by the research ethics committee of the Research Institute for Endocrine Sciences (RIES), Shahid Beheshti University of Medical Sciences. All procedures were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study. Before data collection, both children and parents were informed about the study procedure and its aims, and if the child and parent agreed to participate in the study, parents were asked to sign a written consent form.
Results
BMI trajectory patterns
BMI of women for each predicted trajectory group across follow-up measurements was presented in Fig. 2. The LCGM identified three trajectory groups for BMI in 1356 mothers. Based on the goodness of fit criteria in Table 1-supplementary material, although four and five trajectory groups were statistically appropriate, a three-trajectory group was the best fitting model according to our basis hypothesis. The BIC for this model was − 18,208.71, and the APP ranged from 0.95 to 0.96. The OCC was 33.69, 26.70, and 169.72 for the first to third groups, respectively, and 41.6%, 43.70%, and 14.7% were grouped into class 1, class 2, and class 3, respectively. The three patterns were labeled as class 1/Normal, class 2/ Stage 1 obesity, class 3/ Stage 2 obesity. The normal trajectory pattern showed a steady normal BMI during baseline to the last follow-up assessments. The stage 1 obesity trajectory group showed a higher BMI increase during the follow-up period, whereas the BMI level was always within the stage 1 obesity range. The stage 2 obesity trajectory group was characterized by a steep rise in BMI over follow-ups, including those obese at baseline and remained in the same weight status in all follow-up measurements. Table 2-supplementary material shows BMI changes in each group over the follow-up period. As expected, in all trajectory groups, BMI increases through the follow-up period. The highest and lowest mean BMI were about 39 and 24 at the fourth and baseline measurements.
Participants' characteristics based on the BMI trajectory groups in women
The characteristics of all participants (women, husbands, and offspring) based on women’s trajectories are shown in Table 1. The fathers ranged in age from 57 to 61 years old, and the average age of mothers varied between 51 to 55 years old in different groups, and the corresponding values for their offspring ranged from 27 to 28 years old. Comparing men’s characteristics in various women’s BMI trajectories showed a significant statistical difference for all examined variables except for dyslipidemia and smoking. Furthermore, apart from employment status, all women’s variables were significant among their BMI trajectory groups. In the last follow-up, compared to those in other BMI trajectory patterns, women in the stage 2 obesity trajectory group and their spouses were the oldest (p < 0.001). Most of them had a low level of education and were less physically active, and more smokers. In addition, hypertension and diabetes type 2 were more prevalent in steady obese mothers and their spouses. In terms of offspring, obesity, marital status, dyslipidemia, and age were statistically different among their mothers’ BMI trajectory groups. Children whose mothers were in the stage 2 obesity trajectory group had the highest prevalence of obesity (31.9%) and dyslipidemia (64.9%). In comparison, the corresponding values for children with mothers in the normal weight group were 9.1% and 52.3%, respectively. In addition, the prevalence of obesity and dyslipidemia were reported to be 18% and 58.7%, respectively, in children whose mothers were in the stage 1 obesity group.
Women's BMI trajectory patterns and familial cardiovascular risk factors
As presented in Table 2, in all models, compared to women's in the normal trajectory group, those in stage 1 and 2 of obesity trajectory groups had higher odds ratios for HTN, DM, and dyslipidemia (p < 0.05). The second model showed a higher rate of DM in men with obese (stage 2) spouses (OR 1.56; 95% CI 1.01–2.39) as well as a higher rate of HTN 1.54 (95% CI 1.05–2.25) even after adjusting for potential confounders. The odds ratio for DM remained significant after adjusting behavioral factors in model 3 (adjusted OR 1.55; 95% CI 1.00–2.44). In the second and third models, the odds of men's obesity were higher in their obese (stage 2) spouses compared to normal weight ones, and the odds ratios were (OR 1.65; 95% CI 1.09–2.51) and (OR 1.70; 95% CI 1.10–2.62) respectively. In the full adjusted model, offspring of obese (stage 1 and 2) mothers were more likely to be obese than those whose mothers were normal weight. The odds ratios were (OR 2.39; 95% CI 1.67–3.44) and (OR 4.81; 95% CI 3.16–7.34) respectively. The corresponding values for dyslipidemia in offspring with obese (stage 2) mothers were 1.42 (95% CI 1.01–1.98) and 1.39 (95% CI 1.00–1.94) in the second and third models, respectively. No significant associations were found between the trajectory groups of mothers and their offspring’s outcome, including HTN and DM.
Discussion
The current study aimed to evaluate the trajectory patterns of women’s BMI and to determine the association between these patterns with cardiovascular risk factors at both individual and familial levels. Using six measurements of women's BMI during a 20 years follow-up period, three rising patterns of BMI were identified, including normal, stage 1 obesity, and stage 2 obesity. Our findings indicated that women in obese trajectories were more likely to have HTN, DM, and dyslipidemia. In addition, men with obese (stage 2) spouses showed a higher rate of HTN and DM. In terms of the mother–offspring relationship, the odds of obesity in offspring with obese (stage 1 and 2) mothers were higher than those whose mothers were normal weight.
Few studies investigated developmental trajectories of BMI in particular among women. In this novel longitudinal study, we assessed trajectory patterns of BMI among Iranian women. The model classified individuals in three rising BMI patterns. The current findings were consistent with world health organization (WHO) cut-off points for BMI categorization41. Although there is no comparable data, we compared with other studies on BMI patterns in specific life periods. A study conducted among the Chinese population aged 6–60 years identified a continuous increase of BMI in all groups of BMI42. Some studies indicated less variability in the BMI trajectories in midlife in the total population and women43,44,45, whereas another study reported different patterns during middle age46. A survey conducted among Australian women showed significantly three distinct BMI patterns, similar to our findings47. There is only one study in Iran that determined two patterns of BMI; however, their sample included adolescents28.
In addition, we linked the trajectory pattern of women’s BMI to several cardiovascular risk factors, including high blood pressure, high blood glucose, obesity, and dyslipidemia. Our findings indicated that women in both obesity (stage 1 and 2) trajectories were more likely to have HTN, DM, and dyslipidemia, suggesting the importance of early intervention. Previous studies have illustrated that an elevated BMI in women contributes to a high risk for DM, HTN, and high-risk HDL cholesterol, consistent with our results48,49,50. In contrast, another study indicated that a rising trend of BMI was not associated with high blood pressure. This finding may have been due to other related factors with blood pressure, such as smoking51. Compared with the current study, different statistical methods applied among these studies and the limited time points would also be considered. Additionally, differences among populations such as ethnicity, culture, and socioeconomic status (SES) may also explain this inconsistency52.
Our study showed that men with obese spouses showed a higher rate of HTN and DM in terms of spousal association. There was no comparable data regarding the effect of one spouse’s weight trajectories on cardio-metabolic risk factors in the other spouse. However, in line with our results, some cross-sectional studies considered the spousal concordance in chronic diseases. Studies showed that husbands' health status is influenced by their wives who get chronic diseases53, and husbands' weight status were linked to their spouses’ DM. Moreover, another study showed this association in the other direction in which the weight gain in wives increased their husbands' risk of type 2 diabetes21. The reasons may be explained by the predominant role of wives as caregivers in the families54. Regarding lifestyles and health management, husbands may be more dependent on their spouses. Women with obese spouses are more likely to have chronic diseases such as obesity, HTN, and DM55.
Regarding the association between women’s BMI and CVD risk factors in their offspring, the current study reported that mothers with high BMI values are predicted to have obese children, consistent with previous studies56. Moreover, some cross-sectional studies in Iran and other countries reported that children with obese parents had significantly higher odds of obesity and elevated BP18,57,58. This association is more attributed to the mother–offspring relationship59. This may be due to genetic/epigenetic factors, shared family environment, and parents' health-related attitudes, particularly mothers, because of their leading roles in the family, which influence their children's behaviors60. Although some studies reported the association between parental obesity and HTN61, DM62 of their offspring, an investigation did not find any significant association which was in line with the current results62,63.
This study has several strengths. This is the first report in the world, which addressed the association of BMI trajectories with cardiovascular risk factors at both individual and familial levels. The current study was conducted among the Iranian population with a relatively large sample and a long follow-up period. From an analytical perspective, LCGA was applied as a precise longitudinal method to characterize distinct BMI trajectory patterns. However, several limitations of this study need to be considered. First, the sample was limited to a metropolitan city and cannot be generalized to a rural population. Secondly, some potential predictors of cardiovascular risk factors such as genetic susceptibility, diet data, and environmental conditions such as air pollution64,65 were not available in the current study.
Conclusion
In conclusion, to our knowledge, for the first time in a relatively large community-based sample of Iranian families, our data identified three developmental trajectories of women's BMI and showed significant associations between these trajectories with obesity, dyslipidemia, HTN, and DM in their spouses and children. These findings indicated the critical role of a shared environment in determining cardiovascular risk factors. Accordingly, family-based prevention strategies should be considered for controlling cardiovascular risk factors in whole family members.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Vos, T. et al. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396(10258), 1204–1222 (2020).
Kelly, B. B. & Fuster, V. Promoting cardiovascular health in the developing world: A critical challenge to achieve global health. (2010).
Murray, C. J. et al. Disability-adjusted life years (DALYs) for 291 diseases and injuries in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 380(9859), 2197–2223 (2012).
Hatmi, Z. et al. Prevalence of coronary artery disease risk factors in Iran: a population based survey. BMC Cardiovasc. Disord. 7(1), 32 (2007).
Grundy, S. M. et al. Diabetes and cardiovascular disease executive summary conference proceeding for healthcare professionals from a special writing group of the American Heart Association. Circulation 105(18), 2231–2239 (2002).
Kjeldsen, S. E. Hypertension and cardiovascular risk: General aspects. Pharmacol. Res. 129, 95–99 (2018).
Omboni, S. et al. Hypertension types defined by clinic and ambulatory blood pressure in 14 143 patients referred to hypertension clinics worldwide. Data from the ARTEMIS study. J. Hypertens. 34(11), 2187–2198 (2016).
Jafari-Adli, S. et al. Prevalence of obesity and overweight in adults and children in Iran; a systematic review. J. Diabetes Metab. Disord. 13(1), 121 (2014).
Saeedi, P. et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas. Diabetes Res. Clin. Pract. 157, 107843 (2019).
Mahdavi, M. et al. Insight into blood pressure targets for universal coverage of hypertension services in Iran: The 2017 ACC/AHA versus JNC 8 hypertension guidelines. BMC Public Health 20(1), 1–9 (2020).
Majeed, A. et al. Diabetes in the Middle-East and North Africa: An update. Diabetes Res. Clin. Pract. 103(2), 218–222 (2014).
Davison, K. K. & Birch, L. L. Childhood overweight: A contextual model and recommendations for future research. Obes. Rev. 2(3), 159–171 (2001).
Moore, C. J. & Cunningham, S. A. Social position, psychological stress, and obesity: a systematic review. J. Acad. Nutr. Diet. 112(4), 518–526 (2012).
Cutting, T. M. et al. Like mother, like daughter: familial patterns of overweight are mediated by mothers’ dietary disinhibition. Am. J. Clin. Nutr. 69(4), 608–613 (1999).
Pike, K. M. & Rodin, J. Mothers, daughters, and disordered eating. J. Abnorm. Psychol. 100(2), 198 (1991).
Vik, K. L. et al. Comparison of father-offspring and mother-offspring associations of cardiovascular risk factors: family linkage within the population-based HUNT Study, Norway. Int. J. Epidemiol. 43(3), 760–771 (2014).
Danielzik, S. et al. Impact of parental BMI on the manifestation of overweight 5–7 year old children. Eur. J. Nutr. 41(3), 132–138 (2002).
Gibson, L. Y. et al. The role of family and maternal factors in childhood obesity. Med. J. Aust. 186(11), 591–595 (2007).
Drake, A. J. & Reynolds, R. M. Focus on obesity: Impact of maternal obesity on offspring obesity and cardiometabolic disease risk. Reproduction 140, 387–398 (2010).
Lee, C.-Y. et al. Association of parental overweight and cardiometabolic diseases and pediatric adiposity and lifestyle factors with cardiovascular risk factor clustering in adolescents. Nutrients 8(9), 567 (2016).
Nielsen, J., Hulman, A. & Witte, D. R. Spousal cardiometabolic risk factors and incidence of type 2 diabetes: A prospective analysis from the English Longitudinal Study of Ageing. Diabetologia 61(7), 1572–1580 (2018).
Meyler, D., Stimpson, J. P. & Peek, M. K. Health concordance within couples: a systematic review. Soc. Sci. Med. 64(11), 2297–2310 (2007).
Peek, M. K. & Markides, K. S. Blood pressure concordance in older married Mexican-American couples. J. Am. Geriatr. Soc. 51(11), 1655–1659 (2003).
Wang, Z. et al. Spousal concordance for hypertension: A meta-analysis of observational studies. J. Clin. Hypertens. 19(11), 1088–1095 (2017).
Di Castelnuovo, A. et al. Cardiovascular risk factors and global risk of fatal cardiovascular disease are positively correlated between partners of 802 married couples from different European countries. Thromb. Haemost. 98(09), 648–655 (2007).
Di Castelnuovo, A. et al. Spousal concordance for major coronary risk factors: a systematic review and meta-analysis. Am. J. Epidemiol. 169(1), 1–8 (2009).
Khan, A., Lasker, S. S. & Chowdhury, T. A. Are spouses of patients with type 2 diabetes at increased risk of developing diabetes?. Diabetes Care 26(3), 710–712 (2003).
Ahanchi, N. S. et al. Body mass index trajectories from adolescent to young adult for incident high blood pressure and high plasma glucose. PLoS ONE 14(5), e0213828 (2019).
Ramezankhani, A. et al. Spousal metabolic risk factors and incident hypertension: A longitudinal cohort study in Iran. J. Clin. Hypertens. 22(1), 95–102 (2020).
Babu, G. C. & Shantharajah, S. Optimal body mass index cutoff point for cardiovascular disease and high blood pressure. Neural Comput. Appl. 31(5), 1585–1594 (2019).
Lacey, R. et al. Work-family life courses and BMI trajectories in three British birth cohorts. Int. J. Obes. 41(2), 332–339 (2017).
VanWagner, L. B. et al. Body mass index trajectories in young adulthood predict non-alcoholic fatty liver disease in middle age: the CARDIA cohort study. Liver Int. 38(4), 706–714 (2018).
Hao, G. et al. Body mass index trajectories in childhood is predictive of cardiovascular risk: results from the 23-year longitudinal Georgia Stress and Heart study. Int. J. Obes. 42(4), 923–925 (2018).
Azizi, F. et al. Prevention of non-communicable disease in a population in nutrition transition: Tehran Lipid and Glucose Study phase II. Trials 10(1), 5 (2009).
Momenan, A. A. et al. Reliability and validity of the Modifiable Activity Questionnaire (MAQ) in an Iranian urban adult population. Arch. Iran. Med. 15(5), 279–282 (2012).
Chobanian, A. V. et al. The seventh report of the joint national committee on prevention, detection, evaluation, and treatment of high blood pressure: the JNC 7 report. JAMA 289(19), 2560–2571 (2003).
Association, A. D. 2. Classification and diagnosis of diabetes: standards of medical care in diabetes—2019. Diabetes Care 42(Supplement 1), S13–S28 (2019).
Expert Panel on Detection, E. Executive summary of the third report of the National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). JAMA 285(19), 2486 (2001).
Cole, T. J. et al. Establishing a standard definition for child overweight and obesity worldwide: International survey. BMJ 320(7244), 1240 (2000).
Nagin, D. S. & Nagin, D. Group-Based Modeling of Development (Harvard University Press, 2005).
Weir, C. B. & Jan, A. BMI classification percentile and cut off points. (2019).
Islam, M. T. et al. Life-course trajectories of body mass index and subsequent cardiovascular risk among Chinese population. PLoS ONE 14(10), e0223778 (2019).
Wang, M. et al. Trajectories of body mass index among Canadian seniors and associated mortality risk. BMC Public Health 17(1), 929 (2017).
Wang, M. et al. Body mass index trajectories among middle-aged and elderly Canadians and associated health outcomes. J. Environ. Public Health 2016, 7014857. https://doi.org/10.1155/2016/7014857 (2016).
Botoseneanu, A. & Liang, J. Latent heterogeneity in long-term trajectories of body mass index in older adults. J. Aging Health 25(2), 342–363 (2013).
Buscot, M.-J. et al. Distinct child-to-adult body mass index trajectories are associated with different levels of adult cardiometabolic risk. Eur. Heart J. 39(24), 2263–2270 (2018).
Kakoly, N. S. et al. Group-based developmental BMI trajectories, polycystic ovary syndrome, and gestational diabetes: a community-based longitudinal study. BMC Med. 15(1), 195 (2017).
Buscot, M.-J. et al. BMI trajectories associated with resolution of elevated youth BMI and incident adult obesity. Pediatrics 141(1), e20172003 (2018).
Yuan, Y. et al. Body mass index trajectories in early life is predictive of cardiometabolic risk. J. Pediatr. 219, 31–37 https://doi.org/10.1016/j.jpeds.2019.12.060 (2020).
Zheng, Y. et al. Associations of weight gain from early to middle adulthood with major health outcomes later in life. JAMA 318(3), 255–269 (2017).
Li, L. et al. Life-course body mass index trajectories and blood pressure in mid life in two British birth cohorts: stronger associations in the later-born generation. Int. J. Epidemiol. 44(3), 1018–1026 (2015).
Haines, J. et al. Family functioning and quality of parent-adolescent relationship: cross-sectional associations with adolescent weight-related behaviors and weight status. Int. J. Behav. Nutr. Phys. Act. 13(1), 68 (2016).
Valle, G. et al. Mental and physical health consequences of spousal health shocks among older adults. J. Aging Health 25(7), 1121–1142 (2013).
Maccoby, E. E. Gender and group process: A developmental perspective. Curr. Dir. Psychol. Sci. 11(2), 54–58 (2002).
Liao, J. et al. Gender specificity of spousal concordance in the development of chronic disease among middle-aged and older chinese couples: A prospective dyadic analysis. Int. J. Environ. Res. Public Health 18.6, 2886 (2021).
Chivers, P. et al. Parental and early childhood influences on adolescent obesity: a longitudinal study. Early Child Dev. Care 182(8), 1071–1087 (2012).
Hernández-Valero, M. A. et al. Maternal BMI and country of birth as indicators of childhood obesity in children of Mexican origin. Obesity 15(10), 2512–2519 (2007).
Ejtahed, H.-S. et al. Association of parental obesity with cardiometabolic risk factors in their children: The CASPIAN-V study. PLoS ONE 13(4), e0193978 (2018).
Muthuri, S. K. et al. Relationships between parental education and overweight with childhood overweight and physical activity in 9–11 year old children: Results from a 12-country study. PLoS ONE 11(8), e0147746 (2016).
Ventura, A. K. & Birch, L. L. Does parenting affect children’s eating and weight status?. Int. J. Behav. Nutr. Phys. Act. 5(1), 1–12 (2008).
Fraser, A. et al. Association of maternal weight gain in pregnancy with offspring obesity and metabolic and vascular traits in childhood. Circulation 121(23), 2557 (2010).
Veena, S. R. et al. Testing the fetal overnutrition hypothesis; the relationship of maternal and paternal adiposity to adiposity, insulin resistance and cardiovascular risk factors in Indian children. Public Health Nutr. 16(9), 1656–1666 (2013).
Labayen, I. et al. Intergenerational cardiovascular disease risk factors involve both maternal and paternal BMI. Diabetes Care 33(4), 894–900 (2010).
Sun, D. et al. Recent development of risk-prediction models for incident hypertension: An updated systematic review. PLoS ONE 12(10), e0187240 (2017).
Wu, L., Sun, D. & He, Y. Fruit and vegetables consumption and incident hypertension: dose–response meta-analysis of prospective cohort studies. J. Hum. Hypertens. 30(10), 573–580 (2016).
Acknowledgements
The authors are grateful to all families who participated in the current study.
Author information
Authors and Affiliations
Contributions
P.N. and P.A. designed the study. P.N. and A.-Z.S. carried out the statistical analysis. P.N., P.A., and A.-Z.S. contributed to interpretation of data. P.N., A.-Z.S. and P.A. drafted the manuscript. F.A. and P.A. supervised and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Naseri, P., Amiri, P., Zareie Shab-khaneh, A. et al. Predictive value of women’s weight trajectories in determining familial cardiovascular disorders: a family-based longitudinal study. Sci Rep 11, 17317 (2021). https://doi.org/10.1038/s41598-021-96154-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-96154-5
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.