Abstract
Understanding the processes that shape biodiversity is essential for effective environmental management. Across the world’s coral reefs, algal farming damselfish (Stegastes sp.) modify the surrounding benthic community through their creation of algae “farms”. Using a long-term monitoring dataset (2005–2019) from Moorea Island, French Polynesia, we investigated whether the density of dusky damselfish (Stegastes nigricans) is associated with benthic habitat composition, the density of predators and/or competitors, and whether the survey area was inside or outside of a Marine Protected Area (MPA). We found no evidence that benthic cover or number of competitors were associated with dusky damselfish densities, both inside and outside MPAs. In contrast, fluctuations in dusky damselfish densities were negatively associated with the density of predators (e.g. Serranidae, Muraenidae and Scorpaenidae) in the preceding year in non-MPA areas, and both within and outside of MPAs when predator densities were high (2005–2010). These results suggest that healthy predator populations may be important for regulating the abundances of keystone species, such as algal farming damselfish, especially when predator densities are high.
Similar content being viewed by others
Introduction
Coral reefs are among the most biodiverse ecosystems on the planet, supporting roughly three million species and approximately 25% of all marine life. Unfortunately, the world’s coral reefs are under increasing pressure from local and global stressors (e.g. acidification, agricultural pesticides, plastics, temperature rise, and wastewater) that degrade reef condition and function1,2,3,4,5. These pressures risk causing drastic changes to coral reefs, such as “algal phase-shifts”, where scleractinian coral-dominated communities “shift” to less productive macroalgae- or algal turf-dominated communities3,4,6,7. Understanding the forces that affect community dynamics8,9 and identifying the processes that confer resilience to coral reefs in the face of change10,11, is thus a important challenge for marine ecologists.
Species interactions are important ecological processes that shape and regulate biodiversity. However, species do not affect surrounding biodiversity equally, and keystone species refer to those that disproportionately large effect compared to their abundance12. They influence ecosystem processes, such as by regulating prey populations (predators), regulating predator populations (prey), supporting other species through cooperative interactions (mutualists), linking mutualistic species (hosts, clients or partners), and by creating habitats that affect surrounding species (modifiers)13. Across the world’s coral reefs, some herbivorous fish are keystone species and the diverse array of species within this guild provides various ecosystem services, including those that may aid in the recovery of live hard coral11,14,15,16,17. By contrast, other keystone species, such as farming damselfish, can favor the development of algal turfs and defend them against other herbivorous fish18,19,20.
Damselfish of the genus Stegastes (Pomacentridae, herein ‘Stegastes’) are highly territorial species that develop dense turf algae patches (i.e. farms) which act as a food source18,19. Reliance on algae for sustenance varies across Stegastes species, spanning from facultative to obligate21,22,23. They actively control the algal species composition within their farms, and defend them against other herbivorous fish, invertebrate micro-herbivorous grazers and sea-urchins17,19,20. In order to provide substratum for their algal field, they can actively removed scleractinian corals, such as by biting the living tissue and cultivating dense algal lawns on the coral skeletons24. Correspondingly, the farms of several Stegastes species (e.g. S. apicalis and S. nigricans) can act as reservoirs of pathogens that cause coral diseases25. While these species can modify habitats around them, other species can regulate their density and distribution. For example, Precht et al.24 found that Stegastes densities were strongly regulated by predation risk, and Randazzo-Eisemann et al.26 found that predators are likely to play an important role in regulating the distribution of Stegastes and reducing the stress that they impose on the coral system. Overall, research conducted on farming damselfish over the last four decades has highlighted the influence of Stegastes population dynamics on the benthic cover of coral reefs and thus their potential usefulness as an ecological indicator, but also the potential effects some competitors and predators have on their density18,19.
While long-term studies have found that the density of Stegastes-associated turf can increase significantly as the damselfish take advantage of coral mortality26,27,28,29, few monitoring studies have tracked whether their abundance is associated with, and affected by, predator and competitor populations as well as benthic habitat composition30. Notably, Naim et al.28 showed that algal turf abundance increased significantly between 1993 and 2002 due to the expansion of Stegastes territories over time in some coral reefs at Reunion Island. During their monitoring across 64 Mesoamerican reefs, Randazzo-Eisemann et al.26 showed that the density of algal-gardening damselfish increased from 1.5 individuals per 100m2 in 2006 to 4.7 individuals per 100m2 in 2016, and that their density was strongly correlated with fleshy macroalgae cover. It is also recognised that some herbivorous fish and invertebrate micro-herbivorous grazers are competitors of farming damselfish by regulating the height of the algal canopy and inhibiting algal dominance on the reef17,30. However, sediment-rich algal turfs, that farming damselfish cultivate, may inhibit herbivory due to their high carbonate content as that may interfere with herbivorous fish digestion 31. Thus, a reduction in herbivory will favor first denser turf algae patches, and subsequently the farming damselfish population. Conversely, if predator populations are reduced due to overfishing, the farming damselfish population may increase26.
In the present study, we utilized an extensive long-term monitoring dataset (2005–2019) to investigate how the dusky gregory (Stegastes nigricans, Tahitian name: 'atoti) abundances, the abundance of predators and algal competitors, and benthic cover vary through time inside and outside Marine Protected Areas at Moorea Island (French Polynesia). Given this species’ close association with turf algae, we predicted that variations in the density of S. nigricans would correlate with variations in the proportion of living coral and algal covers. We also predicted a negative association between predators and/or competitors populations and S. nigricans density.
Results
Changes in S. nigricans density since 2005
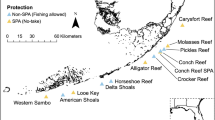
Stegastes nigricans are common on the shallow reef flats around Moorea32. Thus, the shallow reef fish surveys from eight MPA and five non-MPA sites as part of the Fisheries Service of French Polynesia and the CRIOBE’s long-term monitoring efforts (2005–2019, Fig. 1)33 were used in this study (see Methods for more details.
Map of Moorea Island, French Polynesia (drawn by the authors using PhotoFiltre 7 software [version 7.1.2—www.photofiltre.com]) and location of the 13 shallow reef flats (MPA sites—black rectangles; non-MPA sites—white rectangles) used in this study.
From 2005 to 2019, the density of S. nigricans increased significantly inside and outside MPAs (Mann–Kendall tests, τ14 = 0.52, P = 0.008) (Fig. 2A and B). In 2005, the density was 2.64 ± 0.25 fish per 50m2 (mean ± SE) on reef flats throughout all sites. In 2019, the density was 12.06 ± 2.19 fish per 50m2. A significant increase in fish density was observed both inside and outside of the MPAs from 2005 to 2010, before cyclone Oli hit Moorea. Then, a stabilization was observed between 2010 and 2013. Following this, the density of S. nigricans increased again, but only within MPA areas, until 2019 (Fig. 2A). Outside of the MPAs, the density of S. nigricans showed more variability and remained at the same level from 2016 onwards (Fig. 2B).
Number of fish per 50 m2 of Stegastes nigricans and predators (A, B), competitors (C, D) and proportions of live coral, algal turf, and macroalgae (E, F) from 2005 to 2019 at Moorea on reef flats within no-take marine protected areas (MPA) and on non-protected sites (non-MPA), where fishing is allowed. Values are mean ± S.E. A COTS outbreak is indicated in grey (2006–2009), the 2010 hollow arrow indicates a cyclonic event (Cyclone Oli), and the filled arrows indicate weak bleaching events on the reef flats in 2016 and 2019.
Changes in fish predators and competitors since 2005
In 2005, the predator density inside the MPAs was 2.00 ± 1.00 fish per 50m2. Following this, there was an increase to 6.67 ± 2.19 fish per 50m2 in 2006, before stabilizing between 3.00 ± 1.00 and 3.67 ± 1.20 fish per 50m2 from 2007 to 2010. From 2011 to 2019, predator density was similar to 2005 levels, ranging between 1.00 ± 0.00 and 2.00 ± 0.82 fish per 50m2 (Fig. 2A). The density of predators inside the MPAs showed a significant decreasing trend (Mann–Kendall test, τ14 = -0.43, P = 0.008). The same pattern was observed outside the MPAs; however, this trend was not significant (Mann–Kendall test, τ14 = −0.36, P = 0.056). Taken together, while predator densities in 2005 and 2019 were both similar, the larger increase in recruitment within the MPA sites in 2006 appears responsible for the statistical decrease in abundance within the MPA versus the non-significant result outside of the MPA sites. For competitors, no significant trend was detected either inside or outside MPAs throughout the years (densities ranged from 110 to 260 fish per 50m2), except for two peaks in abundance in 2006 and 2009, with densities higher than 500 fish per 50m2 (MPAs: Mann–Kendall tests, τ14 = −0.16, P = 0.30; non-MPAs: Mann–Kendall test, τ14 = 0.12, P = 0.55) (Fig. 2C and D).
Substrate cover
From 2005 to 2019, no significant trend was found for live hard corals (MPAs: Mann–Kendall tests, τ14 = −0.05, P = 0.86; non-MPAs: Mann–Kendall test, τ14 = −0.19, P = 0.36), algal turf (MPAs: Mann–Kendall tests, τ14 = −0.04, P = 0.88; non-MPAs: Mann–Kendall test, τ14 = −0.24, P = 0.29) and macroalgae (MPAs: Mann–Kendall tests, τ14 = −0.08, P = 0.74; non-MPAs: Mann–Kendall test, τ14 = 0.19, P = 0.25) (Fig. 2E and F).
Relationship between predator and S. nigricans density
Overall, the density of fish predators was not correlated with the density of damselfish within the same year inside (ρ = −0.23, S = 694, P = 0.39) or outside of the MPAs (ρ = −0.14, S = 640, P = 0.61) (Fig. 3A and B). However, there was a significant negative relationship between predator density and S. nigricans density in the following year outside of MPAs (ρ = −0.61, S = 734, P = 0.019) but not inside of MPAs (ρ = −0.18, S = 536, P = 0.54) (Fig. 3C and D). A correlation analysis performed for data from 2005 to 2010 (before predators density stabilized, see above) revealed that over that period, the same trend existed with the density of fish predators being negatively correlated with the density of S. nigricans in the following year inside the MPAs as well (ρ = −0.80, S = 61, P = 0.05).
Relationship between competitors and S. nigricans density
The density of fish competitors was not correlated with the density of damselfish within the same year inside (ρ = 0.14, S = 482, P = 0.62) or outside of the MPAs (ρ = −0.04, S = 584, P = 0.88). The same was observed between competitor density and S. nigricans density in the following year (inside of MPAs: ρ = −0.13, S = 514, P = 0.66; outside of MPAs: ρ = −0.44, S = 654, P = 0.12).
Relationship between habitat composition and S. nigricans density
The proportions of live coral, algal turf, and macroalgae were not correlated with the density of damselfish within the same year inside (live coral: ρ = −0.15, S = 646, P = 0.58; algal turf: ρ = 0.02, S = 547, P = 0.93; macroalgae: ρ = −0.13, S = 634, P = 0.64) or outside of the MPAs (live coral: R = 0.27, t = 1.00, P = 0.33; algal turf: R = 0.38, t = 1.48, P = 0.16; macroalgae: R = 0.25, t = 0.93, P = 0.37). The same was observed for live coral, algal turf, macroalgae and S. nigricans density in the following year (live coral: ρ = −0.17, S = 534, P = 0.41; algal turf: R = −0.23, t = −0.84, P = 0.42; macroalgae: ρ = −0.17, S = 535, P = 0.55 for inside the MPAs ; live coral: R = −0.20, t = −0.72, P = 0.48; algal turf: R = 0.04, t = 0.13, P = 0.90; macroalgae: R = −0.09, t = −0.30, P = 0.77 for outside the MPAs).
Discussion
Our long-term monitoring data add to the ongoing debate regarding the role that Stegastes play as macro-scale ecosystem engineers on coral reefs, and provides insights into the factors that regulate their abundances. Over the 15 year study period (2005–2019) the density of Stegastes increased from 0.05 to 0.24 fish per m2. This increase was not associated with changes in the cover of live hard corals, macroalgae, or algal turf inside or outside the MPAs (Fig. 2). However, while we found no relationship between predator density and S. nigricans densities in the same year, a significant negative relationship was found across the whole study period outside of MPAs when considering predator density from the previous year, and this negative relationship was significant both inside and outside of MPAs during periods where predator density was relatively high (e.g. 2005–2010) (Fig. 3). These data suggest that predators may play a role in regulating the density of this species on coral reefs, especially when predator densities are high.
While correlative, our results provide insights into some of the ecological factors that may regulate the abundance of S. nigricans. Most notably, our data suggest that high predator densities may suppress the abundances of S. nigricans in the following year. This result was expected and is consistent with the findings of several comparable studies29,34; however, the density of predators was only high for part of our study period (2005–2011). Predator densities were consistently low between the years 2012–2015, after which it rose again slightly towards the final years of the study. While it is difficult to discern whether predator densities were unusually low between 2012 and 2015, or whether they were unusually high between 2005 and 2011, it may be worth noting that Moorea Island was hit by Cyclone Oli in 2010 and the observed decrease in predator abundance appears to align with that. Furthermore, while predator abundances decreased and stabilized at low levels in the years following Cyclone Oli, the abundance of S. nigricans increased both within and outside of the MPA areas. In addition to predator densities being generally low during this period 35, the increase in abundance of S. nigricans is also likely a product of increased coral damage by the cyclone, which can support the development of Stegastes’ farms25.
In contrast to past studies19,36,37, we observed no significant relationships between habitat substrates (hard corals, algal turf and macroalgae) and S. nigricans abundances. Interestingly, as similar to our finding that only high predation density affected S. nigricans density, the lack of an effect of S. nigricans on substrate composition may be due to their generally low densities around Moorea Island. For instance, we found that S. nigricans abundance increased to 0.24 fish per m2 over the course of this study, which is substantially less than the 4.2 fish per m2 reported by the study by Wilkes et al.37 in Florida, which found that Stegastes may have substantial effects on benthic community dynamics. Additionally, Ceccarelli et al.18 reviewed the role of territorial damselfishes as determinants of benthic communities, and concluded that most observations and experiments have been undertaken at the scale of individual territories, and found a strong influence of Stegastes on benthic communities24,26,37,38. As our work is a long-term study over a large geographic area, it differs from most of these past studies. We are cautious about drawing major conclusions on the role that S. nigricans play as a habitat modifier, given our results and their relatively low density around Moorea Island.
Overall, our long-term study conducted over a large spatial scale suggests that the density of S. nigricans around Moorea Island was not associated with variations in substrate composition or the density of their competitors. Instead, we found a negatively association with the density of their predators, but only when predator densities were relatively high. While correlative, our results complement the results of past work on this topic, which have tended to be much more targeted in their investigations. As a correlative study, we were unable to investigate the direct/indirect effects of additional factors such as the bleaching events that occurred in 2016 and 2019, ocean acidification, or temperature rise. Nonetheless, our results would suggest that predators may play a role in regulating the populations of keystone species, such as algae-farming damselfishes, in order to avoid “algal phase-shifts” in coral reef ecosystem.
Methods
Sampling sites
In 2004, the Fisheries Service of French Polynesia and the CRIOBE a monitoring program surveying eight Marine Protected Area sites (MPA—no-take zones) and five non Marine Protected Area sites (non-MPA—no fishing restriction) was set up around Moorea Island, French Polynesia33. A total of 13 sites were selected to monitor coral reef biodiversity around Moorea: eight Marine Protected Areas (MPA sites) and five non-Marine Protected Areas (non-MPA sites). Each MPA or non-MPA site extends from the shore to beyond the reef crest and out to the 70 m isobath on the outer reef slope. Three distinct reef habitats are thus surveyed at each site: the shallow reef flat (or fringing reef), the barrier reef, and the outer slope. On the shallow reef flats of the 13 sites, three permanent 25 m linear transects placed at the center of a 2 m-wide belt were located using a handheld global positioning system (GPS) receiver and some stakes fixed onto the reef. Since 2004, the 13 shallow reef flat sites had the same disturbance histories: a crown-of-thorns starfish (COTS) Acanthaster cf. solaris outbreak from 2006 to 2009, a cyclone in 2010, and weak bleaching events in 2016 and 2019. Stegastes nigricans are mainly found on shallow reef flats (i.e., reefs contiguous to the coast—less than 400 m from the coastline) around Moorea32,39,40,41. Therefore, within each site (eight MPA and five non-MPA sites), only shallow reef flats were considered this study (Fig. 1). The surveys were done between 8:00 and 11:00am, once per year (in February—warm and wet season).
Fish surveys
For the fish surveys, a belt transect with an underwater visual censuses sampling technique (snorkeling) was used. A 25 m linear transect was placed at the center of a 2 m-wide belt. All fish seen along the transect were identified to the species level. On the first transect pass, the observer recorded highly mobile fish that entered the transect but usually fled as a snorkeler approached. On the second pass, less mobile, cryptic, and site-attached species were targeted with more detailed examinations of crevices. For substrate surveys, the cover proportions of live hard corals, dead corals with algal turf, macroalgae, sand, rubble, and others (e.g. anemones, shells, soft corals) were sampled using the Point Intercept Transect method every 50 cm over 25 m of the belt transect used for the fish survey. Sampling was conducted along three transects (three replicates) separated by 25 m at each site. The three belt transects were set up in the middle of each shallow reef flat (depth: 1 m).
A total of 221 fish species were identified on the fringing reef of all MPA or non-MPA sites at Moorea35. The classification of these species as predators or competitors of S. nigricans was based on direct observations made during previous research at these locations11,39,40,41,42,43. When research on a particular species was not available in the literature, we relied on expertise provided by Prof. Galzin, who has worked on coral reef fishes at these locations continuously since 197439,40,41,42,43. Among the 221 recorded species, those within the families Serranidae (Epinephelus hexagonatus, E. merra and Cephalopholis argus), Muraenidae (Gymnothorax javanicus, G. meleagris, G. undulates and Echidna nebulosa) and Scorpaenidae (Scorpaenopsis diabolus, Pterois antennata and P. radiata) were considered to be predators of S. nigricans. Likewise, the species identified as competitors of S. nigricans (i.e., able to eat the algal turf farmed by S. nigricans) were Ctenochaetus striatus, Acanthurus nigrofuscus, A. triostegus and some Scaridae species (Chlorurus spilurus, Scarus frenatus, S. globiceps, S. oviceps, S. rubroviolaceus, and S. schlegeli). Moreover, some other territorial damselfish such as Chromis viridis, Dascyllus aruanus, and Pomacentrus pavo may compete with S. nigricans for space. As only adult fish are likely to be competitors/predators of S. nigricans, we only considered S. nigricans, predators, and competitors that reached their adult sizes in our analyses44.
Statistical analyses
The normality of the distributions of the density of S. nigricans, predators, and competitors was tested with Shapiro–Wilk tests (W = 0.53 – 0.61; P < 10−3), like the distributions of the proportion of hard live corals, algal turf, and macroalgae (W = 0.64 – 0.87; P < 10−3). Temporal trends for these variables were then evaluated from 2005 to 2019 both inside and outside MPAs with modified Mann–Kendall tests for serially correlated data using the approach proposed by Hamed and Rao45 with variance correction to address potential autocorrelation, using the R package "modifiedmk”46.
The annual average density of S. nigricans was correlated to the annual density of its predators and competitors with Spearman’s correlations tests. The annual average density of S. nigricans was correlated to the annual proportions of live coral, algal turf, and macroalgae with Spearman’s or Pearson’s correlations tests. As a latency in the response may occur, the annual average density of S. nigricans in year n + 1 was also correlated to the annual density of predators, competitors and the proportions of live coral, algal turf, and macroalgae in year n. In our survey protocol, S. nigricans juveniles become adult fish after one year on the reef40. Moreover, predators (Serranidae, Muraenidae and Scorpaenidae) may eat juvenile S. nigricans32. Therefore, we correlated the annual average density of S. nigricans in year n and n + 1 to the annual density of predators in year n. The statistical analysis was conducted using R-Studio and R version 3.5.147,48 at the significance level α = 0.05.
Ethical approval
This study did not involve endangered or protected species and was carried out in accordance with the guidelines of the French Polynesia Code de l’Environnement for animal ethics and scientific research (https://www.service-public.pf/diren/partager/code/). Moreover, the visual surveys (no experiment conducted on fish) were approved by SNO CORAIL (licensing committee: PGEM 2004—http://observatoire.criobe.pf/wiki/tiki-index.php). Lastly, the study was carried out in compliance with the ARRIVE guidelines (http://www.nc3rs.org.uk/page.asp?id=1357) to improve the reporting of research involving animals.
Data availability
All data generated and analysed during this study are available upon reasonable request to the corresponding author (DL).
References
Spalding, M. D., Ravilious, C. & Green, E. P. World atlas of coral reefs, University of California Press, Berkeley, CA, USA (2001).
Hoegh-Guldberg, O. et al. Coral reefs under rapid climate change and ocean acidification. Science 318, 1737–1742 (2007).
Hughes, T. P. et al. Phase shifts, herbivory, and the resilience of coral reefs to climate change. Curr. Biol. 17, 360–365 (2007).
Moritz, C., Vii, J., Lee Long, W., Tamelander, J., Thomassin, A. & Planes, S. Status and Trends of Coral Reefs of the Pacific. Global Coral Reef Monitoring Network, 114 pp (2018).
Morrison, T. H., Hughes, T. P., Adger, W. N. & Brown, K. Save reefs to rescue all ecosystems. Nature 573, 334–336 (2019).
Bellwood, D. R., Hughes, T. P. & Hoey, A. S. Sleeping functional group drives coral-reef recovery. Curr. Biol. 16, 2434–2439 (2006).
Graham, N. A. et al. Managing resilience to reverse phase shifts in coral reefs. Front. Ecol. Environ. 11, 541–548 (2013).
Cavender-Bares, J., Keen, A. & Miles, B. Phylogenetic structure of Floridian plant communities depends on taxonomic and spatial scale. Ecology 87, 109–122 (2006).
Gajdzik, L. et al. Similar levels of trophic and functional diversity within damselfish assemblages across Indo- Pacific coral reefs. Function. Ecol. 32, 1358–1369 (2018).
Holling, C. S. Resilience and stability of ecological systems. Ann. Rev. Ecol. Syst. 4, 1–23 (1973).
Viviani, J. et al. Synchrony patterns reveal different degrees of trophic guild vulnerability after disturbances in a coral reef fish community. Divers. Distrib. 5, 1–12 (2019).
Paine, R. T. A note on trophic complexity and community stability. Am. Nat. 103, 91–93 (1969).
Mills, L. C., Soule, M. E. & Doak, D. F. The keystone-species concept in ecology and conservation. Bioscience 4, 219–224 (1993).
Bouchon, C. et al. Status of the coral reefs of the Lesser Antilles after 2005 coral bleaching event in Status of Caribbean coral reefs after bleaching and hurricanes in 2005 (eds. Wilkinson C et al.) 85–104 (Global Coral Reef Monitoring Network and Reef and Rainforest Research Center, Townsville, 2008).
Jackson, J. B. C., Donovan, M. K., Cramer, K. L., Lam, V. V. Status and Trends of Caribbean Coral Reefs: 1970–2012. Global Coral Reef Monitoring Network, IUCN, Gland, Switzerland (2014)
Cernohorsky, N. H., McClanahan, T. R., Babu, I. & Horsa, M. Small herbivores suppress algal accumulation on Agatti atoll, Indian Ocean. Coral Reefs 34, 1023–1035 (2015).
Altman-Kurosaki, N. T., Priest, M. A., Golbuu, Y., Mumby, P. J. & Marshell, J. Microherbivores are significant grazers on Palau’s forereefs. Mar. Biol. 165, 74–86 (2018).
Ceccarelli, D. M., Jones, G. P. & McCook, L. J. Territorial damselfishes as determinants of the structure of benthic communities on coral reefs. Oceanog. Mar. Biol. Annu. Rev. 39, 355–389 (2001).
Hata, H. & Ceccarelli, D. M. Farming behaviour of territorial Damselfishes in Biology of Damselfishes (eds. Parmentier, E. & Frederich B.) 122–152 (CRC Press, 2016).
Brooker, R. M. et al. Niche construction and the natural selection of domestication. Nat. Commun. 11, 6253 (2020).
Hata, H. & Kato, M. Monoculture and mixed-species algal farms on a coral reef are maintained through intensive and extensive management by damselfishes. J. Exp. Mar. Biol. Ecol. 313, 285–296 (2004).
Ceccarelli, D. M. Modification of benthic communities by territorial damselfish: a multi-species comparison. Coral Reefs 26, 853–866 (2007).
Emslie, M. J. et al. Regional-scale variation in the distribution and abundance of farming damselfishes on Australia’s Great Barrier Reef. Mar. Biol. 159, 1293–1304 (2012).
Precht, W. F., Aronson, R. B. & Moody, R. M. Changing patterns of microhabitat utilization by the threespot damselfish, Stegastes planifrons, on Caribbean reefs. PloS ONE 5, e10835 (2010).
Casey, J. M., Ainsworth, T. D., Choat, J. H. & Connolly, S. R. Farming behaviour of reef fishes increases the prevalence of coral disease associated microbes and black band disease. Proc. R. Soc. B. 281, 20141032 (2014).
Randazzo-Eisemann, Á., Montero Muñoz, J. L., McField, M., Myton, J. & Arias-González, J. E. The effect of Algal-gardening damselfish on the resilience of the mesoamerican Reef. Front. Mar. Sci. 6, 414–421 (2019).
Sandin, S. A. et al. Baselines and degradation of coral reefs in the Northern Line Islands. PLoS ONE 3, e1548 (2008).
Naim, O. et al. Fringing reefs of Reunion Island and eutrophication effects: long term monitoring of primary producers. Atoll Res. Bull. 597, 1–14 (2013).
Figueira, W. F., Lyman, S. J., Crowder, L. B. & Rilov, G. Small-scale demographic variability of the biocolor damselfish, Stegastes partitus, in the Florida Keys USA. Environ. Biol. Fish 81, 297–311 (2008).
Carpenter, R. C. Partitioning herbivory and its effects on coral reef algal communities. Ecol. Monogr. 56, 345 (1986).
Bellwood, D. R. & Fulton, C. J. Sediment-mediated suppression of herbivory on coral reefs: decreasing resilience to rising sea-levels and climate change?. Limnol. Oceanogr. 53(6), 2695–2701 (2008).
Galzin, R. Biomasse ichtyologique dans les écosystèmes récifaux. Etude préliminaire de la dynamique d'une population de Stegastes nigricans dans le lagon de Moorea (Société, Polynésie française). Rev. Trav. Inst. Pêches Marit. 40, 575–578 (1977).
Lison de Loma, T., Galzin, R. & Planes, S. A framework for assessing impacts of marine protected areas in Moorea (French Polynesia). Pacif. Sci. 62, 431–441(2008).
Glaser, M. et al. Breaking resilience for a sustainable future: thoughts for the anthropocene. Front. Mar. Sci. 5, 34–40 (2018).
Siu, G. et al. Shore fishes of French Polynesia. Cybium 41, 245–278 (2017).
Tebbett, S. B., Chase, T. J. & Bellwood, D. R. Farming damselfishes shape algal turf sediment dynamics on coral reefs. Mar. Envirron. Res. 160, 104988 (2020).
Wilkes, A. A. et al. A comparison of damselfish densities on live staghorn coral (Acropora cervicornis) and coral rubble in Dry Tortugas National Park. Southeast. Nat. 7, 483–492 (2008).
Blanchette, A. et al. Damselfish Stegastes nigricans increase algal growth within their territories on shallow coral reefs via enhanced nutrient supplies. J. Exp. Mar. Biol. Ecol. 513, 21–26 (2019).
Galzin, R. Structure of fish communities of French Polynesian coral reefs. I. Spatial scales. Mar. Ecol. Prog. Ser. 41, 129–136 (1987a).
Galzin, R. Structure of fish communities of French Polynesian coral reefs. II. Temporal scales. Mar. Ecol. Prog. Ser. 41, 137–145 (1987b).
Lecchini, D. & Galzin, R. Spatial repartition and ontogenetic shifts in habitat use by coral reef fishes (Moorea, French Polynesia). Mar. Biol. 147, 47–58 (2005).
Galzin, R., Marfin, J. P. & Salvat, B. Long term coral reef monitoring program: heterogeneity of the Tiahura barrier reef (Moorea, French Polynesia). Galaxea 11, 73–91 (1993).
Galzin, R., Lecchini, D., Lison de Loma, T., Moritz, C. & Siu, G. Long term monitoring of coral and fish assemblages (1983–2014) in Tiahura reefs, Moorea, French Polynesia. Cybium 40, 31–41 (2016).
Froese, R. & Pauly, D. Eds. FishBase. World Wide Web electronic publication. www.fishbase.org (2018).
Hamed, K. H. & Rao, A. R. A modified Mann-Kendall trend test for autocorrelated data. J. Hydrol. 204, 182–196 (1998).
Patakamuri, S.K. & O’Brien, N. Modifiedmk: Modified versions of Mann Kendall and Spearman's Rho trend tests. R package version 1.5.0 (2020).
R Core Team. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna (2017).
RStudio Team. R Studio: Integrated Development for R. RStudio, PBC, Boston, MA (2020).
Acknowledgements
Service d'Observation CORAIL from CRIOBE kindly provided the data (http://observatoire.criobe.pf/). We thank Pauline Bosserelle, Vetea Liao and Marguerite Taiarui to have collected the substrate data. The authors has received several grants for this work: French Polynesia government (DRM & DIREN), Agence de l'eau – Rhone Méditérranée Corse (n°2018 1765); Fondation de France (2019-08602); LabEx CORAIL (Project 2018 Emul & project 2019 Plasti-perl); ANR-19-CE34-0006-Manini, ANR-19-CE14-0010-SENSO.
Author information
Authors and Affiliations
Contributions
DL, GS, TLDL, SP and RG designed the monitoring protocol. DL, GS, MA and RG collected the data. FB, VW, WEF and DL performed the statistical analyses. DL, FB, WEF, EG, GS, VW, MA, TLDL, SP and RG wrote the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Feeney, W.E., Bertucci, F., Gairin, E. et al. Long term relationship between farming damselfish, predators, competitors and benthic habitat on coral reefs of Moorea Island. Sci Rep 11, 14548 (2021). https://doi.org/10.1038/s41598-021-94010-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-94010-0
This article is cited by
-
Meta-analysis reveals weak associations between reef fishes and corals
Nature Ecology & Evolution (2024)
-
Effects of onshore development and damselfish (Stegastes nigricans) on coral richness in Opunohu Bay and Cook’s Bay in Moorea, French Polynesia
Coral Reefs (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.