Abstract
Bombesin is a putative antibacterial peptide isolated from the skin of the frog, Bombina bombina. Two related (bombesin-like) peptides, gastrin-releasing peptide (GRP) and neuromedin B (NMB) have been found in mammals. The history of GRP/bombesin discovery has caused little attention to be paid to the evolutionary relationship of GRP/bombesin and their receptors in vertebrates. We have classified the peptides and their receptors from the phylogenetic viewpoint using a newly established genetic database and bioinformatics. Here we show, by using a clawed frog (Xenopus tropicalis), that GRP is not a mammalian counterpart of bombesin and also that, whereas the GRP system is widely conserved among vertebrates, the NMB/bombesin system has diversified in certain lineages, in particular in frog species. To understand the derivation of GRP system in the ancestor of mammals, we have focused on the GRP system in Xenopus. Gene expression analyses combined with immunohistochemistry and Western blotting experiments demonstrated that GRP peptides and their receptors are distributed in the brain and stomach of Xenopus. We conclude that GRP peptides and their receptors have evolved from ancestral (GRP-like peptide) homologues to play multiple roles in both the gut and the brain as one of the ‘gut-brain peptide’ systems.
Similar content being viewed by others
Introduction
The fourteen-amino acid peptide, bombesin, was initially described as a possible antibacterial peptide isolated from the skin of the European fire-bellied toad, Bombina bombina, and was shown to have potent bioactivity in the mammalian nervous system1,2. Subsequently, the mammalian bombesin-like peptides, gastrin-releasing peptide (GRP)2 and neuromedin B (NMB)3, were isolated. GRP is a 27-amino acid peptide (29-amino acids in rodents) originally isolated from the porcine stomach as the mammalian equivalent of bombesin2. Many studies have indicated that GRP is widely expressed in the central nervous system (CNS) in addition to the gastrointestinal tract in mammals4,5. Because GRP could reproduce most of the biological effects of bombesin in many mammals, GRP had long been considered as the mammalian equivalent of amphibian bombesin5,6,7. Bombesin-like peptides appear to function via a family of three G protein-coupled receptors (GPCRs)8, namely the GRP-preferring receptor (GRPR or BB2 receptor)9, the NMB-preferring receptor (NMBR or BB1 receptor)10, and the potential orphan receptor, bombesin receptor subtype-3 (BRS-3 or BB3 receptor) in mammals11. To date, in the mammalian CNS, it has been reported that the GRP system might be integral, through GRPR-mediated mechanisms9, in a variety of autonomic-related functions, including food intake12,13, circadian rhythms14,15,16, fear memory consolidation17,18,19, male reproductive function20, control of sighing21, and itch sensation22,23.
In birds, the expression of orthologous genes for GRP and GRPR has been reported in the chicken CNS24,25. To our knowledge, the only report of a central role of GRP in avian behavior is decreased feeding after the intracerebroventricular administration of GRP in chickens26. In amphibians, however, little information is currently available on the function of the GRP/GRPR system in the CNS, because GRP has long been considered as the mammalian equivalent of bombesin5,6,7. Thus, the central role of GRP in non-mammalian species remains unclear. However, it has been reported that frogs synthesize both GRP and bombesin, which are genetically distinct peptides, suggesting that GRP is not mammalian bombesin27. In addition, molecular cloning analyses revealed that 3 classes of receptor subtypes were identified in the frog brain28. Based on amino acid sequence, two of the classes were clearly the amphibian orthologous genes of the GRPR and NMBR, but not the BRS-328. Moreover, a fourth class (BB4) of receptor from Bombina was identified in amphibians and, interestingly, this had a higher binding affinity for bombesin than for either GRP or NMB28.
As described above, understanding of the orthologous relationships among GRP/NMB/bombesin and their receptors in vertebrates is still in its infancy. Taking advantage of the recent availability of various non-model vertebrate genome information, in this study, we classified the peptides and their receptors from the viewpoint of phylogeny. In particular, to understand its derivation of GRP system from the ancestor of amniotes, we have focused on the GRP system that is widely conserved among vertebrates, and examined its expression in the anamniote clawed frog (Xenopus tropicalis) and contrasted it with bombesin, as amphibians represent an important place in the evolution of tetrapods and our understanding of bombesin and GRP.
Results
Sequence and structure of GRP and GRPR in Xenopus
To verify sequences of GRP and GRPR in X. tropicalis, we cloned cDNA encoding GRP and the GRPR in X. tropicalis and confirmed they are identical to the sequences registered in the GenBank (GRP: XM_018090834.1, GRPR: XP_002938295.1). The deduced amino acid sequence of Xenopus prepro-GRP reveals three major components: a signal peptide (31aa in Xenopus; 31aa in medaka fish; 27aa in chicks; 23aa in rats; 23aa in humans) (Supplementary Fig. S1, gray box); this is followed by the bioactive GRP1−29 (mature GRP, GRP1–24 in medaka fish; GRP1–27 in chicks; GRP1–29 in rats; GRP1–27 in humans) (Supplementary Fig. S1, highlighted in pink), including a motif encoding a 10-amino acid-peptide called neuromedin C (NMC or GRP-10; GRP15–24 in medaka fish; GRP20–29 in chicks; GRP20–29 in rats; GRP18–27 in humans) at the carboxyl-terminus of mature GRP (Supplementary Fig. S1, magenta box). The motif is highly conserved in vertebrates; and finally a carboxyl-terminal extension peptide termed pro-GRP33–121 in Xenopus (pro-GRP28–99 in medaka fish; pro-GRP31–124 in chicks; pro-GRP33–124 in rats; pro-GRP31–125 in humans) (Supplementary Fig. S1, black bar). The mature GRP in Xenopus shared high similarity, particularly at the identical [Ser2] form-NMC (GRP-10) region, with that in rodents and birds but not with the [Asn2] form-NMC in humans and fishes (Supplementary Fig. S1). Peptide sequences of NMB are highly conserved among vertebrates, particularly 11 amino acids in the carboxyl-terminus are identical between human, rat, chick, medaka, and zebrafish (Supplementary Fig. S1). Bombesin-like peptides have previously been identified in some frogs; Rana catesbeiana, R. pipiens (ranatensin)29, Alytes maurus (alytesin)30, Phyllomedusa sauvagii ([Leu8] form-phyllolitorin and [Phe8] form-phyllolitorin)31, Bombina orientalis ([Ser3, Arg9,Phe13] form-bombesin, [Phe13] form-bombesin, [Leu13] form-bombesin)32,33 and Bombina variegate ([Phe13] form-bombesin, [Phe13] form-bombesin-like peptide, bombesin, and [His6] form-bombesin)34 (Supplementary Fig. S1, highlighted in blue). Sequences of carboxyl-terminus regions of GRP, NMB and bombesin peptides are quite similar, 4 amino acids (W, A, G, M) are common between these peptides. On the other hands, sequences of signal peptide region and carboxyl-terminal extension peptide region were diversified between GRP, NMB, and bombesin, and also between the animals.
In terms of receptors for bombesin-like peptides, hydrophobicity analysis showed that Xenopus GRPR, NMBR, and BRS-3 contain seven transmembrane domains (TM1–7) as well as those in other vertebrates (e.g. human, rat, chicken, and zebrafish) (Supplementary Fig. S2). Homology was highest in the hydrophobic domains, and the [Asp85] in the second hydrophobic domain that has possibly been linked to ligand binding is well conserved in vertebrates35 (Supplementary Fig. S2).
Phylogenetic analyses of GRP/NMB/bombesin and GRPR/NMBR/BRS-3
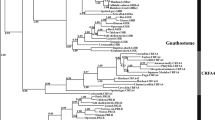
To clarify the relation among GRP/NMB/bombesin family, phylogenetic analysis of GRP/NMB/bombesin precursors (preprohormone) was performed (Fig. 1). Homologous genes for GRP/NMB/bombesin family peptides were found in gnathostomes (vertebrates except cyclostomes/agnathans). For the analysis, we used the deduced amino acid sequence in Mammalia Homo sapiens and Rattus norvegicus, Aves Gallus gallus, Reptilia Gekko japonicus, Amphibia X. laevis, X. tropicalis, Nanorana parkeri (frogs), Rhinatrema bivittatum and Microcaecilia unicolor (Caecilians), teleost fish Danio rerio and Oryzias latipes, cartilaginous fish Callorhinchus milii and Rhincodon typus, because their genomes have been decoded. In addition, the sequences of prepro-bombesin-like peptides, which have been reported for other frog species were also included (Fig. 1).
Molecular phylogeny of precursors of gastrin-releasing peptide (GRP)/neuromedin B (NMB)/bombesin family peptides. GRP/NMB/bombesin family peptides are classified into a GRP clade (pink box) and an NMB/bombesin clade (gray frame). While GRP was found in all the animals examined, genes included in NMB clade were not found in frogs (yellow box). All bombesin-like peptides in frogs appeared to form a single bombesin clade (blue box). Species name in magenta: amphibians; bold: frogs. IDs for the protein sequences used in this analysis are shown in Supplementary Table S1.
The prepro-GRP/NMB/bombesin sequences were divided into two major clades: GRP and NMB/bombesin clades (Fig. 1). A single GRP gene was found in almost all animals examined, although X. laevis which is allotetraploid has one gene on each of chromosome L and S (Fig. 1, pink box). The NMB/bombesin clade was further divided into the sub-clades: the NMB clade (Fig. 1, yellow box) and the bombesin clade (Fig. 1, blue box); the bombesin clade and NMB clade were found only in frogs; and all the other animals including caecilians, respectively. X. tropicalis and Nanorana parkeri have single bombesin gene, and X. laevis has the gene on each of chromosome L and S, but these frogs do not possess any genes of the NMB group (Fig. 1). In addition, all the precursors of bombesin-like peptides which have previously been identified in other frogs: one in Rana catesbeiana, R. pipiens, and Alytes maurus; two in Phyllomedusa sauvagii; three in Bombina orientalis; and four in Bombina variegate34 were also included in the bombesin clade (Fig. 1).
These results indicate two possibilities for the evolution of NMB/bombesin: one is the specialization of NMB into bombesin in the frog lineage; the other is the divergence into NMB and bombesin clades resulting, respectively, in the undetectability of frog NMB and the disappearance of bombesin in vertebrates other than frogs. We therefore examined synteny in genes surrounding the vertebrate NMB/bombesin locus (Fig. 2). Comparison of the genome of X. tropicalis, Nanorana parkeri, Microcaecilia unicolor, Rhinatrema bivittatum, Danio rerio and Homo sapiens, indicates that the order of genes around the frog bombesin genes and the NMB genes in other vertebrates were highly conserved, although an inversion of the ZNF592—SEC11A—bombesin—KTI12 region of X. tropicalis genome has occurred. Thus, it can be concluded that bombesin and NMB are respective orthologues and that specialization of the NMB sequence in the frog lineage resulted in bombesin (Fig. 2). In addition, synteny of the genes surrounding GRP genes was highly conserved among Danio rerio, Xenopus tropicalis, and Homo sapiens, suggesting that the ortholog of vertebrates GPR in Xenopus is GPR, but not bombesin. In 5' upstream region of GPR gene and NMB/bombesin gene in the vertebrates, synteny of closely related genes was observed. This supports the results of phylogenetic analysis (Fig. 1) that the bombesin-like peptides have diverged from an ancestral gene36.
Schematic representation of gene synteny around the neuromedin B (NMB)/bombesin gene and gastrin-releasing peptide (GRP) genes in vertebrates. Horizontal lines (gray) indicate chromosome fragments. Genes are represented as arrows (yellow: NMB gene, blue: bombesin genes, pink: GRP genes, white: other genes) according to their transcriptional orientation. Protein IDs are shown below the genes. Broken lines indicate correspondences of orthologous genes. Gene synteny around the NMB/bombesin gene in vertebrates is highly conserved, suggesting an orthologous relationship between bombesin in frogs and NMB in other vertebrates.
We also performed phylogenetic analysis of the receptors for the GRP/NMB/bombesin family; GRPR, NMBR and BRS-3 (Fig. 3). The results suggest that Gnathostomata basically have orthologues in each of the three groups with no specialization in the frog lineage as seen in NMB/bombesin. The BB4 receptor in Bombina also belongs to the mammalian BRS-3 group. In addition, the conservation of GRPR and NMB, BRS3 was not found in teleosts (e.g. Danio rerio and Oryzias latipes) and cartilaginous fish (e.g. Callorhinchus milii and Rhincodon typus), but only in archaic fish such as Latimeria chalumnae (coelacanth), Erpetoichthys calabaricus (reedfish), Lepisosteus oculatus (gar), and Acipenser ruthenus (sturgeon) (Supplementary Table S1).
Molecular phylogeny of gastrin-releasing peptide-preferring receptor (GRPR)/neuromedin B-preferring receptor (NMB)/bombesin receptor subtype-3 (BRS-3). The GRPR/NMBR/BRS-3 family gene diverged into GRPR (pink box), NMBR (yellow box) and BRS-3 (blue box) in the ancestor of vertebrates. All the three receptors are widely conserved among vertebrates including frogs, although BRS-3 genes are not found in teleost and cartilaginous fish. Species name in magenta: amphibians; bold: frogs; cyan: cartilaginous fish. IDs for the protein sequences used in this analysis are shown in Supplementary Table S1.
Reverse transcription (RT)-PCR of GRP and GRPR mRNA in Xenopus
In contrast to the diversification of NMB/bombesin in the frog lineage and the loss of the BRS-3 gene in some fish lineages, GRP and the GRPR are widely conserved in vertebrates. In this study, we used frogs to investigate the principal (conserved) role of these bombesin-family systems. We confirmed the expression of GRP, Bombesin and GRPR mRNA in a variety of Xenopus tissues (brain, spinal cord, heart, lung, and stomach) by RT-PCR. Bands were detected at the expected sizes for GRP, bombesin, GRPR and GAPDH genes (Fig. 4a). As the internal control in Xenopus, nearly equivalent amounts of GAPDH cDNA were amplified from RNA preparations among these tissues, which showed that no significant RNA degradation had occurred and a proper RT was obtained (Fig. 4a). GRP mRNA was highly expressed in the brain, spinal cord, stomach, and weakly expressed in the lung (Fig. 4a). GRPR mRNA was detected in all the tissues, though the expression level was low in the heart (Fig. 4a). In addition, bombesin mRNA was highly expressed in the brain and stomach and weakly expressed in the spinal cord (Fig. 4a). On the other hand, we did not detect expression of GRP, bombesin or GRPR in the skin.
The expression of gastrin-releasing peptide (GRP) and GRP receptor (GRPR) mRNA in Xenopus tropicalis. (a) Reverse transcription (RT)-PCR analysis of GRP and GRPR mRNA expression. Gene expression of GRP (upper panel) and GRPR (middle panel) in the brain, spinal cord, heart, lung and stomach were examine by RT-PCR. Glyceraldehyde-3-phosphate dehydrogenase gene (GAPDH) was used as the internal control (bottom panel). Cropped gel images of electrophoresis are shown. Original images are shown in Supplementary Fig. S4. (b, c) The expression levels of GRP and GRPR mRNA in Xenopus central nervous system measured by the real-time quantitative PCR. Relative expression levels of GRP (b) and GRPR (c) in the telencephalon, the diencephalon/mesencephalon/pons/cerebellum, the medulla oblongata, and the spinal cord of males and females were analyzed. P values indicate statistical tests using linear mixed model with Bonferroni correction for multiple comparisons (vs. telencephalon, *P < 0.01; vs. diencephalon/mesencephalon/pons/cerebellum, †P < 0.01; vs. medulla oblongata, ‡P < 0.01; vs. spinal cord, §P < 0.05). No sex differences and no interactions between sex and tissue were detected. Dots and bars indicate values of each sample and means of the samples, respectively. Black: males, magenta: females.
Real-time quantitative PCR (qPCR) of GRP and GRPR mRNA in Xenopus CNS
To quantify the GRP and GRPR expression at the transcription level, we performed real-time qPCR analyses for four parts of the CNS of males and females: the telencephalon; the diencephalon/mesencephalon/pons/cerebellum; the medulla oblongata; and the spinal cord. Although GRP and GRPR mRNA expression was detectable in these all tissues of both sexes, no sex differences and no interactions between sex and tissue were detected in any of the tissues we examined (black bars indicate means of males, magenta bars indicate means of females) (Fig. 4b, c). Thus, in the comparisons of expression level between tissues, sexes were combined. The expression of GRP mRNA was higher in the diencephalon/mesencephalon/pons/cerebellum and the medulla oblongata, than in the telencephalon and the spinal cord (Fig. 4b). In contrast, the expression of GRPR mRNA was the highest in the spinal cord, and the lowest in the telencephalon (Fig. 4c).
Distribution of GRP in Xenopus CNS
The localization of GRP in Xenopus CNS was next examined (Fig. 5a–j). The transverse (from rostral to caudal) brain and spinal cord sections (Fig. 5k) were shown to contain many cell bodies and fibers of GRP-immunoreactive (+) neurons in Xenopus CNS. The overall neuroanatomical distribution of GRP+ neuronal cell bodies and their fiber projections is schematically summarized in Table 1. The specificity of the GRP antiserum reactivity was confirmed by control absorption experiments in which the primary rabbit antiserum against Xenopus GRP20–29 was preabsorbed with an excess of Xenopus GRP20–29 antigen peptide ([Ser2] form-NMC); in these experiments no immunostaining was seen (Supplementary Fig. S3). In the spinal cord, GRP+ fibers and numerous varicosities were found throughout the spinal grey matter area (Supplementary Fig. S3a, b, d, and e). A cluster of GRP+ cell bodies was located mainly in the dorsal field of spinal grey in the cervical spinal cord (df; Supplementary Fig. S3a, b, and e). In the thoracic and lumbosacral spinal cord, similar GRP+ fibers were frequently observed, but few GRP+ cell bodies could be detected.
Cell bodies and fibers in Xenopus brain immunohistochemically labeled with the anti-gastrin-releasing peptide (GRP) antiserum. Schematic cross sections illustrating the distribution of GRP-like immunoreactivity (left panels; a–j) and representative photomicrographs (right panels; a″–j″) of transverse sections are shown. (k) Letters (a–j) in the schematic lateral profile of the brain indicate the rostrocaudal level of each transverse section. In the schematic sections, immunoreactive cell bodies and fibers in Xenopus brain are shown by magenta circles and magenta lines, respectively. The density of the symbols shown in the right hemispheres is roughly proportional to the relative density of the immunoreactive elements. The anatomical structures are indicated on the left hemispheres according to the nomenclature of ten Donkelaar (1998)70,71,72. Dorsal is up and lateral is right. The histology of the brain regions where cell bodies are densely distributed was shown by the Nissl staining (middle panels; a′–c′, e′, f′, i′). In the micrographs of immunohistochemical staining for GRP, relative weakly immunoreactive cell bodies and fibers are observed throughout the ventral telencephalic area, e.g. in the nucleus accumbens (Acc; a″ − 1) and in the striatum, ventral part (Strv; a″ − 2). Immunoreactive cell bodies are abundant in the amygdala, e.g. in the amygdala pars medialis (Apm; b″), and in the amygdala pars lateralis (Apl; c″ − 1). GRP-immunoreactive fibers are also observed in the preoptic area (Poa; c″ − 2) and in the nucleus preopticus magnocellularis (Mg; d″). In the diencephalon, GRP-immunoreactive somata are detected in the nucleus ventromedialis thalami (VM; e″) and in the tuberculum posterius (TP; f″). A weak distribution of GRP-immunoreactive fibers is also found in the area near the tractus opticus basalis (optb; g″), and the nucleus reticularis isthmi (Ris; h″). In the brainstem, GRP-immunoreactive cell bodies and fibers are observed in the nucleus raphes (Ra; I″). In the posterior part of the medulla oblongata, GRP-immunoreactive fibers and cell bodies are observed in the nucleus descendens nervi trigemini (Vds), the tractus descendens nervi trigemini (trVds; j″ − 1), the nucleus motorius nervi vagi (Xm; j” − 2), and the nucleus reticularis inferior (Ri; j″ − 3). Scale bars = 50 μm, 10 μm in enlarged images. Arrows indicate representative GRP-immunoreactive cell bodies. Arrowheads indicate representative GRP-immunoreactive fibers. For abbreviations, see Table 1.
In the telencephalon, abundant but weakly GRP-immunoreactive cell bodies and fibers were located throughout the ventral telencephalic area, e.g. cell bodies and fibers in the nucleus accumbens (Acc) and thin fibers in the tractus olfactorius accessorius (ola) (Fig. 5a). Immunoreactive cell bodies were abundant in the amygdala pars medialis (Apm; Fig. 5b), and in the amygdala pars lateralis (Apl; Fig. 5c). In the diencephalon, a small number of labeled cells were located in the nucleus ventromedialis thalami (VM; Fig. 5e). In the hypothalamus, many intensely labeled cell bodies and fibers were found in the tuberculum posterius (TP; Fig. 5f). In the brainstem, large but only weakly immunoreactive cell bodies and fibers were found in the nucleus raphes (Ra; Fig. 5i). In the posterior part of the medulla oblongata, GRP-immunoreactive cell bodies are observed in the nucleus motorius nervi vagi (Xm; Fig. 5j), and the nucleus reticularis inferior (Ri; Fig. 5j).
GRP+ fibers were observed in many parts of the CNS (Table 1). A fine network of GRP+ fibers was also observed in the Apm, in the Apl, and prominently in the striatum, pars ventralis (Strv; Fig. 5a). GRP+ fibers were also found in some diencephalic nuclei surrounding the third ventricle; e.g. VM (Fig. 5e), nucleus preopticus anterior (Poa; Fig. 5c), and nucleus preopticus magnocellularis (Mg; Fig. 5d). A weak distribution of GRP+ fibers was also found in the area near the tractus opticus lateralis (optl), the tractus opticus basalis (optb; Fig. 5g), and the nucleus reticularis isthmi (Ris; Fig. 5h). In the posterior part of the medulla oblongata, the network of GRP+ fibers spreading in the nucleus descendens nervi trigemini (Vds) was intensely immunoreactive and such fibers were also scattered in the tractus descendens nervi trigemini (trVds; Fig. 5j).
Expression of GRPR protein in Xenopus CNS
Western immunoblot analysis with the polyclonal antiserum against Xenopus GRPR was performed to determine the presence of GRPR protein in homogenates derived from the brain and spinal cord of adult male Xenopus. An intense protein band was observed in the brain and spinal cord extracts, and its electrophoretic mobility was located at ~ 43 kDa, which is the expected molecular weight of Xenopus GRPR (Fig. 6). Preabsorption of the antiserum with an excess of antigen peptides (50 μg/mL) prevented the immunostaining of the ~ 43-kDa protein band in the brain and spinal cord (Fig. 6). Immunoblot analyses were repeated independently three times by using different three frogs and gave similar results. We also tried immunohistochemical localization of GRPR in Xenopus CNS using our antibody generated in this study, but unfortunately the antibody did not work in immunohistochemistry.
Western immunoblotting of gastrin-releasing peptide receptor (GRPR). The number on the left indicates the molecular weight (kDa). Extracts of protein from Xenopus brain and spinal cord were transferred onto polyvinylidene difluoride membranes and probed with the rabbit polyclonal antiserum against Xenopus GRPR (1:100,000). The antiserum recognized a single band at the expected molecular weight of GRPR (~ 43 kDa) on a Western blot of the brain and spinal cord. Preabsorption of the antiserum with an excess of antigen peptides (50 μg/ml) eliminated the staining of the ~ 43-kDa protein band. The gel image was processed by cropping to remove the irrelevant area. Original images are shown in Supplementary Fig. S5.
Discussion
GRP was first identified from the porcine stomach as a mammalian orthologue of the anuran bombesin2. Thus, GRP has long been considered as the mammalian equivalents of bombesin5,6,7. It has also been reported that frogs have independent genes for both GRP and bombesin, and this raises the possibility that mammals have an as yet uncharacterized gene encoding a true mammalian bombesin27. Bombina was shown to express the genes for bombesin in the brain, stomach and skin, and for GRP in the brain and stomach only, respectively27. This suggests that bombesin and GRP are distinct peptides which could have different physiological functions in amphibians. However, due to the historical background of the GRP discovery1,2, little attention has yet been paid to the evolutionary relationship of GRP/bombesin and of its receptors in vertebrates. Our phylogenetic analysis indicates that GRP/NMB/bombesin can be divided into two clades; GRP and NMB/bombesin clades (Fig. 1). We further found by using synteny analysis that bombesin and NMB are relative orthologues and that specialization of the NMB sequence only in the frog lineage resulted in bombesin (Fig. 2). In Bombina, it has been suggested that bombesin functions as an antibacterial peptide or toxin secreted from the skin1. Frogs may have started to produce bombesin in the skin as exocrine secretions to protect themselves from bacterial infection and/or predators in the terrestrial adaptation. To our knowledge, this is the first demonstration of the gene divergence of bombesin and bombesin-like peptides in vertebrates, which appeared to be divided into the two families (i.e. GRP and NMB/bombesin) from the vertebrate ancestor (Fig. 7).
The evolutionary lineage of the gastrin-releasing peptide (GRP)/bombesin. (a) Previous studies suggested that GRP (pink box) and neuromedin B (NMB, yellow box) should be considered as the mammalian equivalents of bombesin (blue box). The peptide like bombesin (black box) was thought to be an ancestral form of the bombesin-like family peptides. (b) Our current study demonstrates that GRP is not a mammalian orthologue of bombesin and also that, whereas the GRP system is widely conserved among vertebrates, the NMB/bombesin system has diversified in certain lineages, in particular in frog species. The ancestral peptide (black box) is considered to be similar to GRP from the research of the basal chordate amphioxus36.
For bombesin-like peptide receptors, we found that most gnathostomes have an orthologous gene in each of the three groups (GRPR/NMBR/BRS-3), although BRS-3 was not found in Teleostei and Chondrichthyes (Fig. 3). This suggests that GRPR/NMBR/BRS-3 diverged into three branches in the ancestor of gnathostomes but that the BRS-3 genes have been lost in Teleostei and Chondrichthyes lineages. Otherwise, it is possible that there were two genes for GRPR and NMBR respectively at the divergence of cartilaginous fish, and that the BRS-3 gene then appeared by duplication of the GRPR gene or NMBR gene in the ancestor of Sarcopterygii while the BRS-3 gene was lost in teleost fish. If this is the case, the amino acid sequence of BRS-3 appears to be specialized, considered from the standpoint of the phylogenetic tree. In addition, no specialization for the receptors was observed in the frog lineage as seen in bombesin-like peptides. In mammals, despite the molecular characterization of BRS-3, determination of its function has been difficult as a result of its low affinity for GRP and NMB and its lack of an identified natural ligand37,38,39. Because the natural ligand (corresponding to bombesin) for this receptor has never been identified in mammals, birds, or reptiles, BRS-3 is currently considered to be an orphan receptor37,38,39. BRS-3-deficient mice develop a mild obesity, associated with hypertension and impairment of glucose metabolism40. These results indicate that BRS-3 is required for the regulation of endocrine processes responsible for energy balance in mammals40. Knockout mouse studies have also demonstrated that neither GRPR nor NMBR is on its own essential9, suggesting that these three bombesin-like peptide receptors can compensate for each other’s function in mammals. It is often the case that a number of GPCRs with different affinities can be coupled to just one neuropeptide. Recently, it was reported that, in placental mammals, BRS-3 has lost its binding affinity for NMB/GRP and is constitutively active in a ligand-independent manner, in contrast to BRS-3 in non-placental vertebrates including Xenopus, which has significant affinity for NMB/GRP38. Particularly in Bombina, BRS-3 was suggested to be a ‘bombesin-preferring receptor’28. This rather promiscuous relationship between GPCR and ligand could be important in the diversity of cellular functions controlling different life phenomena.
Despite the diversification of NMB/bombesin in the frog lineage and the loss of the BRS-3 gene in some fish lineages, GRP and GRPR are conserved through vertebrates. Peptides synthesized in endocrine cells of the gastrointestinal tract and in neurons are traditionally considered not only as modulators of metabolism, energy balance, appetite, etc., but also as neuromodulators (neuropeptides); so called ‘gut-brain peptides’ in mammals41. GRP appears to be one of the gut-brain peptides, and the GRP system might play multiple roles in both the gut and the brain5. Indeed, Holmgren et al.42 reported immunohistochemical evidence that the myenteric plexus and circular muscle layer of the stomach of the mudpuppy (Necturus maculosus), a salamander, are richly innervated by GRP+ fibers, and Kim et al.43 reported that endogenous GRP potently stimulates the contraction of longitudinal muscle and relaxes circular muscle in Xenopus stomach, suggesting that GRP is important in the regulation of gastric motility in Xenopus. Furthermore, in Xenopus, we found expression of the mRNA for both GRP and GRPR in the brain and stomach, suggesting that Xenopus GRP systems play physiological roles locally in the CNS as well as in the stomach. In addition, our qPCR analysis suggests that, in Xenopus CNS, the expression of GRP mRNA is highest in the brain, whereas GRPR mRNA expression is highest in the spinal cord. In mammals, it has been demonstrated that GRPR plays an important role in itch sensation in the spinal dorsal horn22,23, suggesting a similar action of GRPR in Xenopus spinal cord. Our immunohistochemical analysis shows that GRP+ cell bodies are distributed in several telencephalic, diencephalic, and rhombencephalic regions and spinal cord of Xenopus (see Table 1). In particular, we observed many GRP+ cell bodies in the hypothalamus and putative limbic system in Xenopus, which corresponds well with the mammalian case5. GRP+ fibers were also observed in many parts of the CNS of Xenopus (Table 1). Thus, GRP might play an important role in multiple physiological functions in Xenopus CNS. In mammals, it is reported that orofacial pruriceptive inputs are processed mainly in the superficial layers of the trigeminal sensory nucleus caudalis in the medulla oblongata, which is similar to the spinal dorsal horn44. Therefore, GRP/GRPR signaling in the trigeminal ganglion-trigeminal sensory nuclei of the medulla oblongata appears to play an important role in orofacial itch sensation in mammals45,46. We also found that, in Xenopus, abundant GRP+ fibers are distributed in the trVds and Vds areas, which appear to correspond to the trigeminal somatosensory system in mammals. Thus, GRP may modulate neurotransmission and integration of somatosensory information in Xenopus. Taken together, these results indicate that GRP functions not only as a gastrointestinal bioactive peptide but also as a neuropeptide in Xenopus CNS, and that GRP functions as a gut-brain peptide in both amphibians and mammals.
In conclusion, GRP has long been considered as the mammalian equivalents of bombesin6,7 (Fig. 7a). We now demonstrate, by phylogenetic and synteny analyses (Fig. 1, 2), that GRP is not a mammalian orthologue of bombesin, and that the GRP system is widely conserved throughout vertebrates, whereas the NMB/bombesin system diversified in some lineages (Fig. 7b). Furthermore, we demonstrate that the GRP system might play multiple roles both in the gut and in the brain of amphibians as one of the ‘gut-brain peptide’ systems.
Indeed, it has recently been reported that the expression of the common ancestral genes for GRP/NMB/bombesin (possibly GRP as an ancestral gene) in amphioxus (which belongs to the subphylum Cephalochordata, an extant representative of the most basal chordates) is abundant in the gut, and is also observed in the cerebral vesicle that has been proposed as the homologue of the vertebrate brain36. While GRP is known to be involved in autonomic regulations such as metabolism, reproductive function, food intake and itch, it was suggested that bombesin has antibacterial functions in frogs, indicating that subfunctionalization of bombesin-like peptides has proceeded especially in the frog lineage. In Xenopus, GRP and bombesin are expressed in the brain, spinal cord and stomach, and GPR/GRPR but not bombesin is expressed in the lung and/or the heart (Fig. 4a). The different expression patterns/physiological functions might also emphasize a subfunctionalization of these distinct peptides. However, the original roles of GRP as an ancestral gene remain unclear at the moment and further functional studies by using lower vertebrates are needed to draw a firm conclusion.
Methods
Animals
Male and female adult clawed frogs (X. tropicalis), the Nigerian BH (Golden) strain, were provided by Hiroshima University Amphibian Research Center through the National BioResource Project (NBRP) of the Japan Agency for Medical Research and Development (AMED). Frogs were maintained in same sex groups according to well established protocols. Sexually matured males (average ~ 4 cm in body length) and females (average ~ 5 cm in body length) (0.5–2 years old in both sexes) were used in this study (Igawa et al., in preparation).
Phylogenetic analysis and gene synteny analysis
For a molecular phylogenetic analysis of precursors of GRP/NMB/bombesin family peptides and GRPR/NMBR/BRS3, protein sequences were obtained from NCBI protein database. Multiple alignments were produced with CLUSTALX (2.1) with gap trimming47. Sequences of poor quality that did not well align were deleted using BioEdit48. Phylogenetic analyses were performed using the Neighbor-Joining method49 by CLUSTALX with the default parameters (1000 bootstrap tests and 111 seeds). Representative phylogenetic trees were drawn by using NJ plot50. Signal peptide site in GRP/NMB/bombesin and transmembrane domain in GRPR/NMB/BRS-3 were predicted by SignalP-5.051 and TMHMM2.0 program52.
For comparison of gene synteny around the NMB/bombesin gene in the amphibian genome, the genome regions upstream and downstream of NMB/bombesin genes were examined using assembled genome sequences of aMicUni1.1 (GCF_901765095.1) for Microcaecilia unicolor, aRhiBiv1.1 (GCF_901001135.1) for Rhinatrema bivittatum, ASM93562v1 (GCF_000935625.1) for Nanorana parkeri, X. tropicalis_v9.1 (GCF_000004195.3) for X. tropicalis, GRCz11 (GCF_000002035.6) for Danio rerio, and GRCh37.p13 (GCF_000001405.25) for Homo sapiens.
Genome and amino acid sequences of Homo sapiens, Rattus norvegicus53, Gallus gallus54, Gekko japonicus55, X. laevis56, X. tropicalis57, Nanorana parkeri58, Rhinatrema bivittatum59,60, Microcaecilia unicolor, Danio rerio61, Oryzias latipes62, Callorhinchus milii63, Rhincodon typus64, Rana catesbeiana, Rana pipiens29, Alytes maurus30, Phyllomedusa sauvagii31, Bombina orientalis32,33, Bombina variegate34, Latimeria chalumnae65,66, Lepisosteus oculatus67, Branchiostoma floridae68, Octopus bimaculoides69 and Drosophila melanogaster70 used for the analysis were obtained from GenBank. Accession IDs were shown in Supplementary Table S1.
cDNA cloning of GRP and GRPR in Xenopus
Adult male frogs (n = 2) were anesthetized with 1% MS-222 (tricaine, Sigma-Aldrich, St. Louis, MO) and sacrificed by decapitation. Immediately, dissected tissues (hypothalamus, spinal cord, and stomach) from frogs were fixed with RNAlater solution (Ambion, Austin, TX) and stored at − 30 °C until RNA extraction. Total RNA was extracted from samples using the illustra RNAspin Mini RNA Isolation kit (GE Healthcare, Buckinghamshire, UK) according to the manufacture’s protocol. The concentration of total RNA was measured using a Qubit RNA assay kit (ThermoFisher Scientific, Waltham, MA). In order to identify genomic sequences of Xenopus GRP and GRPR, the 1st cDNA was synthesized from 200 ng of total RNA of stomach origin by using oligo-dT primers and an iScript cDNA Synthesis Kit (Bio-Rad Laboratories, Hercules, CA). The sequences of primers for cDNA cloning were designed based on the gene sequences in GenBank (see Supplementary Table S1 and Supplementary Table S2). The resulting RT-PCR amplicons of the stomach sample (full open reading frame sequence for GRP or partial sequence for GRPR) were subcloned into the pGEM-T easy vector (Promega, Madison, WI) followed by transfection into Escherichia coli DH5α competent cells (Takara Bio, Shiga, Japan). Positive clones were identified by blue-white screening and at least three positive clones were sequenced. The primer sequences used in this study are shown in Supplementary Table S2.
RT-PCR
To determine the tissue distribution of GRP, bombesin and GRPR mRNA in X. tropicalis (n = 3 of each sex), RT-PCR analysis was performed. Total RNA was extracted from various tissues (brain, spinal cord, heart, lung, stomach and skin), using illustra RNAspin Mini RNA Isolation Kit (GE Healthcare). First-strand cDNA was synthesized from 200 ng of total RNA in a 20-μL reaction volume using oligo-dT primers and iScript RT (Bio-Rad Laboratories). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as the internal control. Primer pairs are shown in Supplementary Table S2. The resultant PCR amplicons were electrophoresed on 2% agarose gels. RT-PCR studies were repeated four times using independently extracted RNA samples from different animals. Consistent results were obtained from each run.
Real-time qPCR
The quantification of GRP and GRPR in Xenopus CNS was performed using real-time qPCR. In brief, whole brains and spinal cords of males or females were quickly removed and placed on ice (n = 3 of each sex). Brains were divided into 3 parts: 1 the telencephalon; 2 the diencephalon/mesencephalon/pons/cerebellum; and 3 the medulla oblongata. The preparations were carefully dissected under a dissecting microscope (Olympus, Tokyo, Japan). Total RNA was isolated from samples using illustra RNAspin Mini RNA Isolation Kit with RNase-free DNase I (GE Healthcare) according to the manufacture’s protocol. Samples were reverse transcribed from 500 ng of total RNA in a 20 μL volume using iScript cDNA synthesis kit (Bio-Rad Laboratories). Real-time qPCR was carried out on a C1000 Thermal Cycler (Bio-Rad Laboratories). Reactions were performed in 20 μL solution, with 200 nM of each primers, 1 μL of 5 ng cDNA samples and SYBER-green master mix (KAPA SYBER FAST qPCR kit, KAPA Bio-systems, Boston, MA, USA) according to the manufacture’s instructions. Assays (in triplicate) were repeated at least twice with the constitutive Drosha as a normalizing control. Primer pairs are shown in Supplementary Table S2. The data relative changes in mRNA expression were determined using the 2−∆∆CT method. Statistical tests were performed using linear mixed model with Bonferroni correction for multiple comparisons by IBM SPSS ver. 24 for Windows (IBM, Armonk, NY). The fixed effects factors are sex and tissue, and the random effects factor is individual.
Immunohistochemistry
Adult male frogs (n = 4) were killed by decapitation under deep anesthesia (see above). Brains and spinal cords were immediately dissected out and immersion-fixed in Bouin’s fixative solution [saturated picric acid:10% unbuffered formalin:acetic acid = 15:5:1 (v/v)] overnight at 4 °C. Subsequently, tissues were dehydrated and embedded in paraffin wax. Serial sections of each tissue were cut transversely on a microtome at 10-μm in thickness. We performed immunohistochemical analysis according to our established methods20. In brief, endogenous peroxidase activity was eliminated from the sections by incubation in a 0.1% H2O2 in absolute methanol solution for 20 min followed by three 5-min rinses with phosphate-buffered saline (PBS) (pH 7.4). After blocking nonspecific binding with 1% normal goat serum and 1% BSA in PBS containing 0.3% Triton X-100 for 30 min at room temperature, sections were then incubated in Can Get Signal A (TOYOBO) with a 1:2000 dilution of primary rabbit antiserum against rat GRP20–29, a 10-amino acid-peptide called NMC or GRP-10 (AssayPro, St. Charles, MO, RRID: AB_2571636) for 48 h at 4 °C. This GRP antiserum has previously been shown to be specific for rodent GRP20,46. The amino acid sequence of NMC (epitope residues) is identical between frogs and rodents. Immunoreactive-products were detected with a streptavidin–biotin kit (Nichirei, Tokyo, Japan), followed by diaminobenzidine (DAB) development according to our previous method20. The histology of Xenopus brain was studied by the Nissl (cresyl violet; Muto Pure Chemicals, Tokyo, Japan) staining. GRP-expressing cells in the CNS were observed under the Olympus FSX100 microscope. Nomenclature of brain areas and nuclei was based on the stereotaxic atlas of the CNS of anurans71.
Control procedures for the DAB method were performed using pre-absorption of the working dilution (1:2000) of the primary antiserum with saturating concentration of frog GRP20–29 antigen peptide (GSHWAVGHLM; 50 μg/mL: 2[Ser]-NMC, produced in AnaSpec; San Jose, CA) overnight at 4 °C before use. The information of GRP antiserum used in this study is shown in Supplementary Table S3.
Western immunoblotting
Western blotting was conducted according to our previously described methods72. In brief, adult male frogs (n = 3) were sacrificed by decapitation under deep anesthesia (see above). Brains and spinal cords were quickly removed and placed on ice, and were snap-frozen immediately in liquid nitrogen. The preparations (100 μg protein/lane) were boiled in 10 μL sample buffer containing 62.5 mM trishydroxymethyl-aminomethane-HCl (Tris–HCl; pH. 6.8), 2% SDS, 25% glycerol, 10% 2-mercaptoethanol, and a small amount of bromophenol blue. Samples were run on a 4–20% SDS-PAGE and electroblotted onto a polyvinylidene difluoride (PVDF) membrane (Bio-Rad Laboratories) using a semidry blotting apparatus (Bio-Rad Laboratories). Membranes were blocked with PVDF Blocking Reagent for Can Get Signal (TOYOBO, Tokyo, Japan) for 30 min at room temperature and incubated for 1 h at room temperature in Can Get Signal Solution 1 (TOYOBO) with a 1:100,000 dilution of rabbit polyclonal antiserum against GRPR (RRID: AB_ 2832951). The polyclonal antiserum was produced in a rabbit using a Xenopus GRPR fragment peptide. The sequence of antigen peptide was 195–208 amino acid residues (CAPYPHSNGLHPRI) of Xenopus GRPR (GenBank accession No. XP_002938295.1). Blotted membranes were washed three times with 0.05% Tween 20 in Tris–HCl buffered saline (TBST) and incubated with horseradish peroxidase-conjugated goat polyclonal antibody against rabbit IgG (Bio-Rad Laboratories) at 1:10,000 dilution in Can Get Signal Solution 2 (TOYOBO) for 1 h at room temperature. After washing for three times with TBST, blots were visualized by Immun-Star WesternC Chemiluminescence Kit (Bio-Rad Laboratories). Images of the different immunoblots were slightly adjusted in brightness and contrast to provide a uniform background. Control procedures were performed by pre-absorption of the working dilution (1:100,000) of the primary antiserum with a saturating concentration of Xenopus GRPR195–208 antigen peptide, CAPYPHSNGLHPRI (50 μg/mL, produced in Sigma-Aldrich) overnight at 4 °C before use. The information of GRPR antiserum used in this study is shown in Supplementary Table S3.
Ethics statement
All experimental procedures were approved in accordance with the Guide for the Care and Use of Laboratory Animals prepared by Okayama University (Okayama, Japan. http://www.cc.okayama-u.ac.jp/~animal/committee.html), although an individual ID of approval was not provided because we used only amphibians for the experiments in this study. All efforts were made to minimize animal suffering and reduce the number of animals used in this study. The study design including experimental animals (strain, sex, age and sample size), experimental procedures and statistical methods is described in compliance with the ARRIVE guidelines.
Data availability
All relevant data are within the manuscript and the data that support the findings of this study are available from the corresponding author upon request.
References
Anastasi, A., Erspamer, V. & Bucci, M. Isolation and structure of bombesin and alytesin, 2 analogous active peptides from the skin of the European amphibians Bombina and Alytes. Experientia 27, 166–167 (1971).
McDonald, T. J. et al. Characterization of a gastrin releasing peptide from porcine non-antral gastric tissue. Biochem. Biophys. Res. Commun. 90, 227–233. https://doi.org/10.1016/0006-291X(79)91614-0 (1979).
Minamino, N., Kangawa, K. & Matsuo, H. Neuromedin B: A novel bombesin-like peptide identified in porcine spinal cord. Biochem. Biophys. Res. Commun. 114, 541–548. https://doi.org/10.1016/0006-291X(83)90814-8 (1983).
Panula, P., Nieminen, O., Falkenberg, M. & Auvinen, S. Localization and development of bombesin/GRP-like immunoreactivity in the rat central nervous system. Ann. N. Y. Acad. Sci. 547, 54–69 (1988).
Sakamoto, H. In Handbook of Hormones (eds Takei, Y. et al.) 191–192 (Academic Press, 2016).
Roesler, R., Kent, P., Schröder, N., Schwartsmann, G. & Merali, Z. Bombesin receptor regulation of emotional memory. Rev. Neurosci. 23, 571. https://doi.org/10.1515/revneuro-2012-0046 (2012).
Takanami, K. & Sakamoto, H. The Gastrin-Releasing Peptide Receptor (GRPR) in the spinal cord as a novel pharmacological target. Curr. Neuropharmacol. 12, 434–443. https://doi.org/10.2174/1570159X12666140923201432 (2014).
Kroog, G. S., Jensen, R. T. & Battey, J. F. Mammalian bombesin receptors. Med. Res. Rev. 15, 389–417. https://doi.org/10.1002/med.2610150502 (1995).
Battey, J. & Wada, E. Two distinct receptor subtypes for mammalian bombesin-like peptides. Trends Neurosci. 14, 524–528. https://doi.org/10.1016/0166-2236(91)90005-F (1991).
Wada, E. et al. cDNA cloning, characterization and brain region-specific expression of neuromedin B-preferring bombesin receptor. Neuron 6, 421–430 (1991).
Fathi, Z. et al. BRS-3: a novel bombesin receptor subtype selectively expressed in testis and lung carcinoma cells. J. Biol. Chem. 268, 5979–5984 (1993).
Ladenheim, E. E., Behles, R. R., Bi, S. & Moran, T. H. Gastrin-releasing peptide messenger ribonucleic acid expression in the hypothalamic paraventricular nucleus is altered by melanocortin receptor stimulation and food deprivation. Endocrinology 150, 672–678 (2009).
Ladenheim, E. E., Taylor, J. E., Coy, D. H., Moore, K. A. & Moran, T. H. Hindbrain GRP receptor blockade antagonizes feeding suppression by peripherally administered GRP. Am. J. Physiol. 271, R180-184 (1996).
Karatsoreos, I. N., Romeo, R. D., McEwen, B. S. & Silver, R. Diurnal regulation of the gastrin-releasing peptide receptor in the mouse circadian clock. Eur. J. Neurosci. 23, 1047–1053. https://doi.org/10.1111/j.1460-9568.2006.04633.x (2006).
Okamura, H. & Ibata, Y. GRP immunoreactivity shows a day-night difference in the suprachiasmatic nuclear soma and efferent fibers: Comparison to VIP immunoreactivity. Neurosci. Lett. 181, 165–168. https://doi.org/10.1016/0304-3940(94)90585-1 (1994).
Shinohara, K., Tominaga, K., Isobe, Y. & Inouye, S. T. Photic regulation of peptides located in the ventrolateral subdivision of the suprachiasmatic nucleus of the rat: daily variations of vasoactive intestinal polypeptide, gastrin-releasing peptide, and neuropeptide Y. J. Neurosci. 13, 793–800. https://doi.org/10.1523/JNEUROSCI.13-02-00793.1993 (1993).
Merali, Z. et al. Bombesin receptors as a novel anti-anxiety therapeutic target: BB1 receptor actions on anxiety through alterations of serotonin activity. J. Neurosci. 26, 10387–10396. https://doi.org/10.1523/JNEUROSCI.1219-06.2006 (2006).
Roesler, R. et al. Bombesin/gastrin-releasing peptide receptors in the basolateral amygdala regulate memory consolidation. Eur. J. Neurosci. 19, 1041–1045. https://doi.org/10.1111/j.0953-816X.2004.03175.x (2004).
Shumyatsky, G. P. et al. Identification of a signaling network in lateral nucleus of amygdala important for inhibiting memory specifically related to learned fear. Cell 111, 905–918 (2002).
Sakamoto, H. et al. Sexually dimorphic gastrin releasing peptide system in the spinal cord controls male reproductive functions. Nat. Neurosci. 11, 634–636. https://doi.org/10.1038/nn.2126 (2008).
Li, P. et al. The peptidergic control circuit for sighing. Nature 530, 293–297. https://doi.org/10.1038/nature16964 (2016).
Sun, Y. G. & Chen, Z. F. A gastrin-releasing peptide receptor mediates the itch sensation in the spinal cord. Nature 448, 700–703 (2007).
Sun, Y. G. et al. Cellular basis of itch sensation. Science 325, 1531–1534 (2009).
Maekawa, F., Tsukahara, S., Tanaka, K. & Ohki-Hamazaki, H. Distributions of two chicken bombesin receptors, bombesin receptor subtype-3.5 (chBRS-3.5) and gastrin-releasing peptide receptor (chGRP-R) mRNAS in the chicken telencephalon. Neuroscience 125, 569–582. https://doi.org/10.1016/j.neuroscience.2004.01.057 (2004).
Mo, C. et al. Characterization of NMB, GRP and their receptors (BRS3, NMBR and GRPR) in chickens. J. Mol. Endocrinol. 59, 61. https://doi.org/10.1530/jme-17-0020 (2017).
Tachibana, T., Matsuda, K., Khan, S. I., Ueda, H. & Cline, M. A. Feeding and drinking response following central administrations of bombesin-like peptides in chicks. Comp. Biochem. Physiol. A. Mol. Integr. Physiol. 156, 394–399. https://doi.org/10.1016/j.cbpa.2010.03.008 (2010).
Nagalla, S. R., Gibson, B. W., Tang, D., Reeve, J. R. & Spindel, E. R. Gastrin-releasing peptide (GRP) is not mammalian bombesin. Identification and molecular cloning of a true amphibian GRP distinct from amphibian bombesin in Bombina orientalis. J. Biol. Chem. 267, 6916–6922 (1992).
Nagalla, S. R. et al. Cloning of a receptor for amphibian [Phe13]bombesin distinct from the receptor for gastrin-releasing peptide: Identification of a fourth bombesin receptor subtype (BB4). Proc. Natl. Acad. Sci. 92, 6205–6209. https://doi.org/10.1073/pnas.92.13.6205 (1995).
Krane, I. M., Naylor, S. L., Helin-Davis, D., Chin, W. W. & Spindel, E. R. Molecular cloning of cDNAs encoding the human bombesin-like peptide neuromedin B. Chromosomal localization and comparison to cDNAs encoding its amphibian homolog ranatensin. J. Biol. Chem. 263, 13317–13323 (1988).
Konig, E. et al. Antimicrobial peptides and alytesin are co-secreted from the venom of the Midwife toad, Alytes maurus (Alytidae, Anura): Implications for the evolution of frog skin defensive secretions. Toxicon 60, 967–981. https://doi.org/10.1016/j.toxicon.2012.06.015 (2012).
Nagalla, S. R., Barry, B. J. & Spindel, E. R. Cloning of complementary DNAs encoding the amphibian bombesin-like peptides Phe8 and Leu8 phyllolitorin from Phyllomedusa sauvagei: Potential role of U to C RNA editing in generating neuropeptide diversity. Mol. Endocrinol. 8, 943–951. https://doi.org/10.1210/mend.8.8.7997236 (1994).
Nagalla, S. R. et al. There are three distinct forms of bombesin: Identification of [Leu]BOMBESIN, [Phe]BOMBESIN, and [Ser3, Arg, Phe]BOMBESIN in the frog bombina orientalis. J. Biol. Chem. 271, 7731–7737. https://doi.org/10.1074/jbc.271.13.7731 (1996).
Spindel, E. R., Gibson, B. W., Reeve, J. R. Jr. & Kelly, M. Cloning of cDNAs encoding amphibian bombesin: Evidence for the relationship between bombesin and gastrin-releasing peptide. Proc. Natl. Acad. Sci. USA 87, 9813–9817. https://doi.org/10.1073/pnas.87.24.9813 (1990).
Bai, B. et al. Parallel peptidome and transcriptome analyses of amphibian skin secretions using archived frozen acid-solvated samples. Mol. Biotechnol. 54, 187–197. https://doi.org/10.1007/s12033-012-9551-6 (2013).
Spindel, E. R., Giladi, E., Brehm, P., Goodman, R. H. & Segerson, T. P. Cloning and functional characterization of a complementary DNA encoding the murine fibroblast bombesin/gastrin-releasing peptide receptor. Mol. Endocrinol. 4, 1956–1963. https://doi.org/10.1210/mend-4-12-1956 (1990).
Wang, P. et al. Characterization of GRP as a functional neuropeptide in basal chordate amphioxus. Int. J. Biol. Macromol. 142, 384–394. https://doi.org/10.1016/j.ijbiomac.2019.09.109 (2020).
Ohki-Hamazaki, H., Iwabuchi, M. & Maekawa, F. Development and function of bombesin-like peptides and their receptors. Int. J. Dev. Biol. 49, 293–300. https://doi.org/10.1387/ijdb.041954ho (2005).
Tang, H. et al. Constitutively active BRS3 is a genuinely orphan GPCR in placental mammals. PLoS Biol. 17, e3000175. https://doi.org/10.1371/journal.pbio.3000175 (2019).
Xiao, C. & Reitman, M. L. Bombesin-like receptor 3: Physiology of a functional orphan. Trends Endocrinol. Metab. 27, 603–605. https://doi.org/10.1016/j.tem.2016.03.003 (2016).
Ohki-Hamazaki, H. et al. Mice lacking bombesin receptor subtype-3 develop metabolic defects and obesity. Nature 390, 165–169 (1997).
Vadnie, C. A. et al. Gut-brain peptides in corticostriatal-limbic circuitry and alcohol use disorders. Front. Neurosci. https://doi.org/10.3389/fnins.2014.00288 (2014).
Holmgren, S., Jensen, J., Jönsson, A.-C., Lundin, K. & Nilsson, S. Neuropeptides in the gastrointestinal canal of Necturus maculosus. Cell Tissue Res. 241, 565–580. https://doi.org/10.1007/BF00214577 (1985).
Kim, J. B., Johansson, Å., Holmgren, S. & Conlon, J. M. Gastrin-releasing peptides from Xenopus laevis: Purification, characterization, and myotropic activity. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 281, R902–R908. https://doi.org/10.1152/ajpregu.2001.281.3.R902 (2001).
Olszewski, J. On the anatomical and functional organization of the spinal trigeminal nucleus. J. Comp. Neurol. 92, 401–413. https://doi.org/10.1002/cne.900920305 (1950).
Takanami, K. et al. Comparative anatomy of gastrin-releasing peptide pathways in the trigeminal sensory system of mouse and the Asian House Musk Shrew Suncus murinus. Acta Histochem. Cytochem. 49, 181–190. https://doi.org/10.1267/ahc.16030 (2016).
Takanami, K. et al. Distribution of gastrin-releasing peptide in the rat trigeminal and spinal somatosensory systems. J. Comp. Neurol. 522, 1858–1873. https://doi.org/10.1002/cne.23506 (2014).
Larkin, M. A. et al. Clustal W and Clustal X version 2.0. Bioinformatics 23, 2947–2948. https://doi.org/10.1093/bioinformatics/btm404 (2007).
Hall. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acid Symposium Series 41, 95–98 (1999).
Saitou, N. & Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 4, 406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454 (1987).
Perriere, G. & Gouy, M. WWW-query: An on-line retrieval system for biological sequence banks. Biochimie 78, 364–369 (1996).
Almagro Armenteros, J. J. et al. SignalP 5.0 improves signal peptide predictions using deep neural networks. Nat. Biotechnol. 37, 420–423. https://doi.org/10.1038/s41587-019-0036-z (2019).
Krogh, A., Larsson, B., von Heijne, G. & Sonnhammer, E. L. L. Predicting transmembrane protein topology with a hidden markov model: Application to complete genomes11Edited by F. Cohen. J. Mol. Biol. 305, 567–580. https://doi.org/10.1006/jmbi.2000.4315 (2001).
Gibbs, R. A. et al. Genome sequence of the Brown Norway rat yields insights into mammalian evolution. Nature 428, 493–521. https://doi.org/10.1038/nature02426 (2004).
International Chicken Genome Sequencing, C. Sequence and comparative analysis of the chicken genome provide unique perspectives on vertebrate evolution. Nature 432, 695–716. https://doi.org/10.1038/nature03154 (2004).
Liu, Y. et al. Gekko japonicus genome reveals evolution of adhesive toe pads and tail regeneration. Nat. Commun. 6, 10033. https://doi.org/10.1038/ncomms10033 (2015).
Session, A. M. et al. Genome evolution in the allotetraploid frog Xenopus laevis. Nature 538, 336–343. https://doi.org/10.1038/nature19840 (2016).
Hellsten, U. et al. The genome of the Western clawed frog Xenopus tropicalis. Science 328, 633–636. https://doi.org/10.1126/science.1183670 (2010).
Sun, Y. B. et al. Whole-genome sequence of the Tibetan frog Nanorana parkeri and the comparative evolution of tetrapod genomes. Proc. Natl. Acad. Sci. USA 112, E1257-1262. https://doi.org/10.1073/pnas.1501764112 (2015).
San Mauro, D. et al. Life-history evolution and mitogenomic phylogeny of caecilian amphibians. Mol. Phylogenet. Evol. 73, 177–189. https://doi.org/10.1016/j.ympev.2014.01.009 (2014).
Torres-Sanchez, M. et al. Multi-tissue transcriptomes of caecilian amphibians highlight incomplete knowledge of vertebrate gene families. DNA Res. 26, 13–20. https://doi.org/10.1093/dnares/dsy034 (2019).
Howe, K. et al. The zebrafish reference genome sequence and its relationship to the human genome. Nature 496, 498–503. https://doi.org/10.1038/nature12111 (2013).
Kasahara, M. et al. The medaka draft genome and insights into vertebrate genome evolution. Nature 447, 714–719. https://doi.org/10.1038/nature05846 (2007).
Venkatesh, B. et al. Survey sequencing and comparative analysis of the elephant shark (Callorhinchus milii) genome. PLoS Biol. 5, e101. https://doi.org/10.1371/journal.pbio.0050101 (2007).
Read, T. D. et al. Draft sequencing and assembly of the genome of the world’s largest fish, the whale shark: Rhincodon typus Smith 1828. BMC Genomics 18, 532. https://doi.org/10.1186/s12864-017-3926-9 (2017).
Amemiya, C. T. et al. The African coelacanth genome provides insights into tetrapod evolution. Nature 496, 311–316. https://doi.org/10.1038/nature12027 (2013).
Nikaido, M. et al. Coelacanth genomes reveal signatures for evolutionary transition from water to land. Genome Res. 23, 1740–1748. https://doi.org/10.1101/gr.158105.113 (2013).
Braasch, I. et al. The spotted gar genome illuminates vertebrate evolution and facilitates human-teleost comparisons. Nat. Genet. 48, 427–437. https://doi.org/10.1038/ng.3526 (2016).
Putnam, N. H. et al. The amphioxus genome and the evolution of the chordate karyotype. Nature 453, 1064–1071. https://doi.org/10.1038/nature06967 (2008).
Albertin, C. B. et al. The octopus genome and the evolution of cephalopod neural and morphological novelties. Nature 524, 220–224. https://doi.org/10.1038/nature14668 (2015).
Adams, M. D. et al. The genome sequence of Drosophila melanogaster. Science 287, 2185–2195. https://doi.org/10.1126/science.287.5461.2185 (2000).
ten Donkelaar, H. J. In The Central Nervous System of Vertebrates Volume 2 (eds Nieuwenhuys, R. et al.) 1151–1314 (Springer, 1998).
Satoh, K. et al. In vivo processing and release into the circulation of GFP fusion protein in arginine vasopressin enhanced GFP transgenic rats: response to osmotic stimulation. FEBS J. 282, 2488–2499. https://doi.org/10.1111/febs.13291 (2015).
Acknowledgements
We are grateful to the National BioResource Project (NBRP) of the Japan Agency for Medical Research and Development (AMED); ‘Xenopus tropicalis’ in the Amphibian Research Center (Hiroshima University, Japan) for providing X. tropicalis, the Nigerian BH (Golden) strain. We are grateful to Prof. John F. Morris (University of Oxford, UK) for valuable discussion and for reading this manuscript. This work was supported by Japan Society for the Promotion of Science (JSPS) KAKENHI (to H.S.; 15K15202, 15KK0257, 15H05724, 16H06280), the Japan Agency for Medical Research and Development (AMED) (to H.S.; 961149), a Grant-in-Aid for Scientific Research on Innovative Areas “Singularity Biology (No.8007)” of MEXT, Japan (to T. S.; 21H00428), and Okayama University Dispatch Project for Female Faculty members (to M.H.). K.T. and T.O. were supported by Research Fellowships of JSPS for Young Scientists.
Author information
Authors and Affiliations
Contributions
A.H., D.F., K.T., T.O. and Y.K. performed immunohistochemical experiments. A.H. and K.T. performed Western blotting. A.H., M.H., Y.K., and T.O. performed molecular biology, phylogenetic, and gene synteny analyses. T.S. interpreted the data and provided the advice. M.H. and H.S. wrote the paper. A.H. and M.H. contributed equally to this study. H.S. supervised the whole study. All authors had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hirooka, A., Hamada, M., Fujiyama, D. et al. The gastrin-releasing peptide/bombesin system revisited by a reverse-evolutionary study considering Xenopus. Sci Rep 11, 13315 (2021). https://doi.org/10.1038/s41598-021-92528-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-92528-x
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.