Abstract
Currently, the Galapagos sea lion (GSL, Zalophus wollebaeki) and Galapagos fur seal (GFS, Arctocephalus galapagoensis) are among the most important endemic species for conservation in the Galapagos Archipelago. Both are classified as “Endangered” since their populations have undergone drastic declines over the last several decades. In this study we estimated the abundance of both otariids, and their population trends based using counts conducted between 2014 and 2018 in all their rookeries, and we analyzed the influence of environmental variability on pup production. The GSL population size in 2018 in the archipelago was estimated to be between 17,000 to 24,000 individuals and has increased at an average annual rate of 1% over the last five years after applying correction factors. The highest number of GSL counted in the archipelago was in 2014 followed by a population decline of 23.8% in 2015 that was associated with the El Niño event that occurred during that year. Following this event, the population increased mainly in the northern, central and southeastern rookeries. The GSL pup abundance showed a decreasing trend with the increase in intensity of the El Niño. The GFS population in 2018 was counted in 3,093 individuals and has increased at an annual rate of 3% from 2014 to 2018. A high number of GFS counted in 2014 was followed by a population decrease of 38% in 2015, mainly in the western rookeries. There was interannual population fluctuations and different growth trends among regions of the archipelago. GSL and GFS pup abundance has a strong decreasing tendency with the increase in the subthermocline temperature (ST) and the El Niño 1 + 2 index. Our results provide evidence that both species are highly vulnerable to periodic oceanographic-atmospheric events in the Galapagos Archipelago which impact prey abundance and the flow of energy in the unique Galapagos ecosystem.
Similar content being viewed by others
Introduction
Population assessments for marine mammals are challenging but necessary as they provide information for management success1,2. Generally, these evaluations are more accurate for species that breed on land or near the coast, such as pinnipeds and sea otters3,4. However, the proportion of the population that is ashore can be challenging to determine, since breeding rookeries can be difficult to access and, in some cases, are geographically dispersed5,6. Estimating the abundance of otariids presents other challenges, since there are an unknown proportion of adults at sea that are not counted during a count7,8. However, pups are confined to land in breeding rookeries for the first few months of life, so their numbers are more constant6,9. Therefore, pups can be used as an indicator of abundance and a basis for estimating population trends10,11.
Density-dependent (ecological conditions) and independent factors (environmental and anthropogenic conditions) are expected to influence population dynamics of pinnipeds12,13. Environmental variability influences the distribution and abundance of otariids14,15, as these species use specific areas during their sea foraging trips16,17. Oceanographic variability, among other factors, also influences pup production rates18,19. For example, environmental perturbations such as El Niño–Southern Oscillation (ENSO) decrease the productivity levels in marine ecosystems reducing the availability of main prey of marine predators20,21. This results in nutritional stress and increase in population mortality rates, especially in species that inhabit tropical systems14,22.
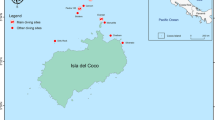
The Galapagos sea lion (GSL, Zalophus wollebaeki) and Galapagos fur seal (GFS, Arctocephalus galapagoensis) are otariids endemic to the Galapagos Archipelago (Fig. 1), adapted to this hotspot of local productivity in the midst of a tropical environment in the equatorial Pacific23. This creates an ecological challenge for these species, as they depend on a surrounding ocean with generally poorer conditions than other otariids living at higher latitudes24,25. The GSL is the most abundant of the two otariids and it is distributed throughout the archipelago, with larger rookeries on islands of the southeastern region10, while the GFS breeds on eight main rookeries in the western and northern islands, with its largest population on Fernandina island26. Currently, both species are classified as Endangered in the Red List of the International Union for Conservation of Nature (IUCN) as their populations have undergone a drastic decline in the last few decades27,28. Consequently, these otariids are among the species with the highest priority for conservation in the Galapagos Marine Reserve29.
Breeding rookeries of the Galapagos sea lion (orange symbols) throughout the Galapagos Archipelago and Galapagos fur seal (green symbols) in western and northern islands of the archipelago. The symbol sizes are according to the individuals’ abundance. The regionalization scheme of the archipelago is shown. (Map courtesy of Byron X. Delgado, GIS Research / Knowledge Management, Charles Darwin Foundation).
There is limited published data to estimate the actual population sizes and growth trends of these species, despite their year-round presence in the archipelago and their high fidelity to their breeding rookeries10,26,30. However, performing simultaneous counts is complex due to the large distance between the breeding sites29. Overall populations of GSL and GFS were estimated at approximately 16,000 and 8,000 individuals, respectively, according to a census in 200126. Therefore, to date, it is known with certainty that populations have decreased by 50% since the last global census of 1978, when the populations were estimated at 40,000 and 16,000 individuals of GSL and GFS respectively30. These population decreases are related to the effects of oceanographic-atmospheric disturbances such as the ENSO, that occur in the Pacific Ocean and affect the Galapagos Archipelago, leading to a lack of food resources in the marine environment24. These effects are exacerbated when combined with anthropogenic stressors that contribute to the deterioration of their habitat10.
Oceanographic variability in the Galapagos Archipelago causes regional changes in marine productivity that are reflected in the ecological conditions of GSL populations throughout the archipelago31. These changes are influenced by the major ocean features that make the western region highly productive through the strong upwelling caused by the Equatorial Undercurrent, the central and southeast regions less productive through localized upwelling, while the north region has the lowest productivity levels due to its proximity to the Equatorial Front32,33. Although the oceanic waters around the archipelago are considered an upwelling system in the equatorial zone34, this area is vulnerable to oceanographic disturbances like El Niño events that cause negative fluctuations in marine productivity with resulting demographic impacts on Galapagos otariids10,22.
The GSL and GFS are non-migratory species that maintain small rookeries throughout the year, contrary to other pinnipeds in cold zones which aggregate on land mainly during short, highly synchronized breeding seasons35,36. It has been shown that in small otariids populations (< 1,300 individuals), all age/sex categories can be identified and counted on land10. Since a proportion of the population will be out at sea during the census, estimates of the total population size usually require a correction factor with the count data8,37. Given the relevance to conservation efforts, the Galapagos National Park Directorate (GNPD) launched a management plan for the conservation of the GSL in 2012, including a standardized method of counting populations (direct counts in the rookeries during the annual census in the entire archipelago) to determine their population size and to propose appropriate management strategies for this species29. All management measures and counting methods proposed for GSL also apply to the GFS populations.
There is a need to generate substantial information to establish the population size and status of the Galapagos otariids in the face of the high environmental variability and growing frequency of ENSO events in the archipelago. Here, we assess the current status of GSL and GFS by analyzing their abundance and population trend based on the counts performed from 2014 to 2018 throughout the archipelago. We estimate the population size of GSL using correction factors applied to the annual censuses8,10. We also analyzed the relationships between pup production of both species and oceanographic variables and discuss the possible causes of the variability in these populations.
Methods
Study area and data collection
The Galapagos Archipelago is located at 960 km from mainland Ecuador in the Tropical Eastern Pacific. Waters associated with this archipelago make up the Galapagos Marine Reserve (GMR), an area of about 138,000 km2 that has protected endemic and native marine species for over 20 years38. The region’s oceanographic setting is largely responsible for sporadic colonization of the islands, leading to the evolution and presence of the diverse species such as the Galapagos pinnipeds31. The Equatorial Undercurrent and the South Equatorial Current carry high-nutrient waters to the GMR and affect the marine ecosystem dynamics32,33. This results in different regions within the archipelago with differentiated marine productivity, sea temperature patterns and biodiversity levels39,40. The presence and intensity of these currents in the archipelago determine two seasons throughout the year: a warm season (January to May), and a cold season (June to December), that influence the sea surface temperature (SST), ranging up to eight °C between seasons41.
This research was performed as part of the pinnipeds population monitoring program conducted by the GNPD and the Universidad San Francisco de Quito (USFQ). Most of the data were derived from the annual research cruises conducted by the Galapagos Science Center (GSC) in collaboration with the GNPD to assess the impact of climate change on the emblematic fauna of the Galapagos Archipelago. From 2014 to 2018, all breeding rookeries of GSL and GFS were monitored during October, corresponding to the peak in pup births of both species. A total of 43 censuses per year (32 rookeries of GSL and 11 of GFS) were carried out on 13 islands of the archipelago (Fig. 1). Censuses were performed on land using a direct count method commonly employed in otariid surveys10,42. Each census started at 6 am and required approximately two to three hours to complete, depending on the size of the rookery. Two trained observers, situated in opposite boundaries of the rookery, walked along the coastline, simultaneously counting animals and identifying them by age/sex categories. The census was completed when the observers encountered each other in the middle of the rookery10.
Data analysis
In this study, the archipelago was divided into four regions (West, North, Central and Southeastern) (Fig. 1), based on the regional biogeography of the archipelago proposed by39,40 to identify potential influence of the environmental variability in the Galapagos pinnipeds populations. Six age/sex categories were distinguished in each census: adult males, subadult males, adult females, juveniles, pups and indeterminate (unidentified animals)42. Categorized data were organized in a matrix of total counted animals per rookery, bioregion and year (Supplementary Tables S1 and S2).
The GSL abundance in each rookery, island and region were estimated by correcting the number of counted animals using correction factors derived from the probability of observing individuals of different age categories ashore during the counts8. These correction factors were obtained by using the Lincoln-Petersen method, based on resights of marked animals, to estimate the population size of GSL in the Caamaño rookery in the central bioregion over 13 years of counts8. According to these authors, the probability of observing a given adult in the rookery (Pob-r) during the cold/reproductive season was 16% (95% confidence interval (CI) = 19%–12%), while for juveniles, the probability was 35% (95% CI = 37%–34%). Based on these values, we assumed that the proportion of animals at sea (ps) during the counts was 1/(Pob-r/100). Thus, the numbers of adults and juveniles counted ashore (Ca) were corrected by multiplying Ca * ps. For adults, the ps values used were 6.25 (95% CI = 5.26–8.33), and for juveniles, the ps values were 2.86 (95% CI = 2.78–2.94). Individuals categorized as indeterminate in the censuses were considered juveniles for the analyses, as categorization uncertainties mostly concerned the immature category36. The sum of the corrected counts of adults and juveniles and the raw counts of pups corresponded to the population estimates in each rookery in a given year.
A regression between years and total individuals counted between 2014 and 2018 was used to estimate the population trends of GSL and GFS in the archipelago. The regression between years and total pups counted was used to estimate the pup abundance trends for the same sites. Census data used in the regressions were natural-logarithm transformed. The value of the slope (r) in the regression analysis was converted to the finite rate of increase (ʎ) as exp (r). The average annual growth rates of the population and of pup abundance, expressed in percentages, were calculated as 100*(ʎ-1). The variability in pup abundance in each rookery throughout the years was determined using the coefficient of variation (CV). Differences in pup abundance per rookery in each region were tested using a Kruskal Wallis test, along with a multiple comparison of the mean ranks for all groups.
Abundance and environmental variables
The effects on the GSL and GFS pups abundance of oceanographic variables, such as the anomalies in SST (°C) linked to El Niño and La Niña events (measured from the El Niño 1 + 2 index), the subthermocline temperature (ST, °C; between approximately 60 m and 100 m of depth) and the depth-integrated chlorophyll-a concentration (mg m−2), a proxy of phytoplankton biomass, were examined per region in the archipelago. For this, census data were natural-logarithm transformed. The values of the El Niño 1 + 2 index were taken from the NOAA website (http://www.cpc.ncep.noaa.gov/data/indices/sstoi.indices). This index is the three months running mean SST anomalies in the region and is commonly used to indicate the status of the equatorial Pacific coasts, including the Galapagos Archipelago. The criterion often used to classify El Niño or La Niña events are five consecutive 3-month running mean SST anomalies exceeding the threshold of ± 0.5 °C.
Temperature and salinity profiles to ~ 100 m depth were obtained using a Seabird SBE 19plus V2 SeaCAT Profiler, collected within two kms offshore of the rookeries. All CTD casts were corrected using SeaBird’s SeaSoft software. Profiles typically exhibited a surface mixed layer and a deep layer of nearly constant properties, separated by a thermocline of varying thickness. The subthermocline temperature (ST) was determined as the average temperature from the bottom of the cast (approximately 100 m) to the depth where the potential density was 0.2 kg/m3 less than the density at the bottom of the profile43. The upper depth bound averaged from 45–65 m across the years surveyed.
Chlorophyll-a samples (chl a) were collected in triplicate by gravity filtering 400 ml of seawater through Isopore 5 µm polycarbonate filters (47 mm) to obtain the large cell size fraction (> 5 µm). The filtrate was then filtered onto a Whatman GF/F filter (25 mm) using an in-line vacuum (≤ 100 mmHg) to obtain the small cell size fraction (≤ 5 µm). The filters were extracted in 6 ml of 90% acetone and incubated in the dark at − 20 ºC for 24 h. Raw fluorescence values of the chl a extracts were measured on a Turner Designs 10-AU fluorometer according to the methods of44. Depth-integrated total chl a was determined through trapezoidal integration of the combined size-fractions at four depth measurements made throughout the euphotic zone corresponding to sampling depths of 50%, 30%, 10% and 1% incident irradiance.
Spearman’s rank correlation coefficient (rs) was used to analyze the relationship between the natural logarithm of the total number of pups and the average value (from August to October) of oceanographic variables per year. All statistical analyses were conducted using Statistica version 8.0 (StatSoft. Inc., Tulsa, OK, USA). Statistical significance was defined as p < 0.05.
Results
Galapagos sea lions’ abundance and population trend
From 2014 to 2018, the GSL was recorded at 32 breeding rookeries on 13 islands (Table S1). The estimated population sizes on all islands are shown in Table 1. The 2018 global census resulted in 4,891 individuals counted, representing an estimated 19,929 (95% confidence interval (CI) = 17,617–24,694) animals in total. The greater abundance was estimated in 2014 (21,493; 95% CI = 18,864–26,895 individuals), based on a count of 4,980 animals. Overall, there was a decrease of 23.9% in the number of animals counted from 2014 to 2015 across the archipelago (Fig. 2); whereas from 2015 to 2018 the total of animals counted increased 29%. The average annual growth rate in the archipelago was 1% (slope = 0.01, standard error (SE) = 0.04, p = 0.818, R2 = 0.02) between 2014 and 2018 (Fig. 2).
Galapagos sea lions’ population and pup abundance trend in the Galapagos Archipelago from 2014 to 2018. It includes the annual counts at 32 breeding rookeries on 13 islands distributed throughout the archipelago. Blue and gray circles show the total population and pup population, respectively. Blue and gray dashed lines show the population trend and the pup trend, respectively.
During the study period, larger rookeries were located in the southeastern region (62.0 ± 2.4% of the entire population), following by the central region (23.5 ± 2.3%), the western region (9.6 ± 1.4%), and the northern region (5.0 ± 2.0%). The populations in the central and northern rookeries increased at an average annual growth rate of 4.5% (slope = 0.04, SE = 0.11, p = 0.531, R2 = 0.14) and 15.3% (slope = 0.14, SE = 0.13, p = 0.341, R2 = 0.30) respectively (Fig. S1). The populations in the western and southeastern regions decreased at an average rate of -6.6% (slope = -− 0.07, SE = 0.05, p = 0.291, R2 = 0.35) and − 0.3% (slope = -0.01, SE = 0.03, p = 0.919, R2 = 0.01) respectively, between 2014 and 2018 (Fig. S1).
During the five years of sampling, the annual average (± standard deviation, SD) number of pups was 567 ± 153 individuals. However, pup abundance varied across years in the archipelago, where the highest and the lowest interannual variability were observed in the northern (average coefficient of variation (CV) = 82.5%) and central (CV = 33.4%) rookeries, respectively (Table 2). The pup abundance in the archipelago decreased 40.5% between 2014 and 2016, whereas from 2015 to 2018 it increased 91.5%. The average annual growth rate of pup abundance was 5% between 2014 and 2018 (slope = 0.05, SE = 0.11, p = 0.677, R2 = 0.07) (Fig. 2). This increasing pup trend between 2014 and 2018 was observed in northern and central rookeries at an average annual rate of 100.3% (slope = 0.69, SE = 0.22, p = 0.050, R2 = 0.77) and 12.9% (slope = 0.12, SE = 0.07, p = 0.183, R2 = 0.50), respectively. In the west, pups decreased at an average rate of − 6.7% (slope = − 0.07, SE = 0.15, p = 0.677, R2 = 0.07) between 2014 and 2018; whereas in the southeastern, the average annual growth rate is 3.1% (slope = 0.03, SE = 0.12, p = 0.898, R2 = 0.02).
The number of pups in 2018 was 793 individuals, which was the highest value during the five years. Fifty percent of births occurred in the southeastern rookeries, 37% in the central, 10% in western, and 8% in northern rookeries. There were significant differences between years (Kruskal–Wallis test: H(4) = 13.66, p = 0.008) and regions (Kruskal–Wallis test: H(3) = 16.55, p = 0.001) in the numbers of pups counted from 2014 to 2018. A multiple comparisons test revealed that the numbers of pups counted in 2015 differed significantly from 2017 and 2018 (p < 0.05), and the numbers of pups at northern rookeries differed significantly from the central and southeastern rookeries in all years (p < 0.05).
Galapagos fur seals’ abundance and population trend
From 2014 to 2018, the GFS was recorded at 11 breeding rookeries on seven islands (Table S2). The 2018 global census was the largest, resulting in 3,093 individuals counted. Overall, there was a 38.1% reduction between 2014 and 2015 in the number of animals counted, followed by a 78.7% increase between 2015 and 2018 (Fig. 3). The average annual growth rate in the archipelago was 3% (slope = 0.03, SE = 0.09, p = 0.755, R2 = 0.04) between 2014 to 2018 (Fig. 3).
Galapagos fur seals’ population and pup abundance trend in the Galapagos Archipelago from 2014 to 2018. It includes the annual counts at 11 breeding rookeries on seven islands distributed in the western, northern and central regions of the archipelago which this species inhabits. Blue and gray circles show the total population and pup population, respectively. The blue dashed line shows the population trend.
During the study period, larger rookeries were located in the western region (77.9 ± 4.8% of the entire population), following by the northern region (18.0 ± 4.7%), and the central region (4.1 ± 0.4%). The average annual growth rate in the western rookeries was 3.3% (slope = 0.03, SE = 0.10, p = 0.772, R2 = 0.03) between 2014 to 2018, while for the northern and central rookeries it was 3.9% (slope = 0.04, SE = 0.07, p = 0.637, R2 = 0.08) and −0.2% (slope = −0.01, SE = 0.07, p = 0.975, R2 = 0.01) respectively (Fig. S2).
During the five years of sampling, the annual average (± SD) number of pups was 398 ± 76 individuals, and the highest and the lowest interannual variability were observed in the central (CV = 50.3%) and western (CV = 18.3%) rookeries, respectively (Table 3). The pup abundance throughout the archipelago decreased by 36.8% between 2014 and 2015, whereas from 2015 to 2016, it increased by 86.8% due to a tripling in total pups in the northern region (Fig. 3). The number of pups increased between 2014 and 2018 in the archipelago at an average annual rate of 6.3% (slope = 0.06, SE = 0.08, p = 0.508, R2 = 0.16) (Fig. 3). This trend was observed in western, northern and central rookeries at an average annual rate of 5.7% (slope = 0.06, SE = 0.08, p = 0.524, R2 = 0.15), 7.6% (slope = 0.07, SE = 0.11, p = 0.641, R2 = 0.08) and 20.9% (slope = 0.19, SE = 0.11, p = 0.174, R2 = 0.51), respectively.
The number of pups counted in 2018 was 447 individuals, which was the second highest value during the five years. Eighty-eight percent of pup’s production occurred in the western rookeries, 8% in northern and 4% in central rookeries. There were no significant differences between years in the numbers of pups counted from 2014 to 2018 (Kruskal–Wallis test: H(4) = 1.39, p = 0.845), but differences were observed between regions (Kruskal–Wallis test: H(2) = 12.52, p = 0.001). A multiple comparisons test revealed that the number of pups at western rookeries differed significantly from the number in the central rookeries (p < 0.05).
Environmental variables and regional pup abundance
The average (± standard deviation) values of ST and chl a (from August to October) recorded from 2014 to 2018 per island in each region are shown in Table 4. There were no significant relationships between the oceanographic variables and the GSL and GFS pup abundances in any region of the archipelago (Tables 5, 6). Pup abundance of GSL showed a slight negative trend with the El Niño 1 + 2 index and ST values in most regions, and a positive trend with the depth-integrated chl a data in the western and southeastern regions (Table 5). While the pup abundance of GFS showed a negative trend with the El Niño 1 + 2 index and ST values, and a positive trend with the depth-integrated chl a data in all regions (Table 6).
In the GSL, the tendency with the El Niño 1 + 2 index is stronger in the north and central regions, less evident in the southeastern region and was not observed in the west (Fig. 4A). The relationships between GSL pup abundance and ST and chl a were as expected (negative with ST and positive with chl a) in the west and southeastern regions, but contrary in the north and central regions. In the GFS, the negative relationships between pup abundance and the El Niño 1 + 2 index (Fig. 4B) and ST values were evident in the three regions which this species inhabits. However, the positive relationship between GFS pup abundance and chl a was evident in the north and west but not in the central region.

taken from the NOAA website. The Niño El 1 + 2 index indicates the intensity of El Niño (positive values) and La Niña (negative values) events, which can be considered weak (SST anomaly of 0.5 to 0.9), moderate (1.0 to 1.4) or, strong (> 1.5).
Annual pup abundance per region of the Galapagos Archipelago concerning the El Niño 1 + 2 index. (A) Galapagos sea lion. (B) Galapagos fur seal. The El Niño 1 + 2 index is the three-month running average of the sea surface temperature (SST) anomalies in the El Niño 1 + 2 region (0–10°S, 90–80°W). The values of the El Niño 1 + 2 index corresponding to October (during the peak in pup births of both species) were plotted for each year and
Discussion
Determining the abundance and population trends of Galapagos pinnipeds is a complex task that depends on the assumptions of techniques through which the census is performed. However, in these polygynous pinnipeds, breeding seasons occur annually within a limited, predictable timeframe35. Therefore, pup production is believed to be the best indicator of rookery status and from which the population trends can be estimated6,11. We report estimates of GSL and GFS population abundances during their reproductive peaks, derived from a direct count method commonly employed in Galapagos otariids8,10, that provides estimated values with narrow confidence intervals.
Galapagos sea lion’s population trend
During the last decades, the Galapagos pinnipeds have experienced significant population declines10,26. Therefore, both species are protected by Ecuadorian laws, and its natural environment is managed under conservation plans29. The current size of the GSL population was estimated at ~ 20,000 individuals and increased at an average annual rate of 1% between 2014 and 2018. For the population estimate, we use correction factors calculated from GSL observations in a central rookery. However, it is important to consider that there may be differences in the phenology of births between regions that might affect the proportion of animals on land during the counts8. The population peaked in 2014, followed by a population decline of 23.8% in 2015, which was associated with the strong El Niño event of that year. Since the abundance and distribution of pinnipeds are influenced by their ability to feed while at sea45,46, oceanographic factors seem to be the main causes of population fluctuations.
Recent population estimates differ from the first census in 1978, probably reflecting differences in the methods used. In 1978, a direct and at-distance count of 8,000 animals suggested an estimated population of 40,000 individuals30, while in 2001, the direct and at-distance count was 4,937 sea lions, resulting in an estimated 16,000 individuals26. As the rationales behind the correction factors were not provided, these estimates may not be compared. Since 2012, counts have followed a standardized method based on the management plan for Galapagos sea lion’s conservation29. As animal counts for 2018 remain close to that recorded in 2001 and that the average age of reproducing individuals is ~ 10 years with a typical lifespan ≥ of 18 years47, the population may have been relatively stable over the past 20 years. If so, this suggests that a population reduction of 50% is maintained in the last four generations (1978–2018), which would ratify its classification as an endangered species27. Thus, the GSL maintains residual effects of the strong 1982/83 and 1997/98 El Niño events22,26,48 from which they have not been able to recover due to the recurrence of El Niño events every 4–5 years49,50.
The GSL breeds on almost all the islands of the archipelago and approximately 62% of the population inhabit the southeastern region. The El Malecón rookery, on San Cristobal Island, is the largest in the archipelago (Table S1) and maintains an annual increase close to 2% reported by10, during favorable oceanographic conditions. After an El Niño event, the GSL populations increase mainly in the central and southeastern rookeries, while northern and western rookeries maintain a reduced population for a longer period. This is explained by the marine habitat characteristics and the population dynamics, which differ in each region of the Galapagos Archipelago51,52, influencing the abundance of the GSL and determining the population trends of its rookeries. The characteristics of shelf habitat could lead to a restriction of food resources25. The northern region has few areas where benthic habitat is attainable to GSL, which causes them to increase their feeding effort53, making this region a more demanding environment for GSL, reflected in its population size. Oceanographic conditions show upwellings in the western region32. These high levels of marine productivity could favor the trophic requirements of the GSL and, consequently, their population growth. However, in this region, the GSL limits its foraging effort and probably its population size to reduce competition with GFS24,25, which has significantly larger populations. Therefore, the slow population recovery in the northern and western regions could be associated with ecological conditions, their rookeries’ size and with the low pup production that is maintained. These conditions confirm that the population trend of a single region or island should not be considered representative of the entire archipelago8,10.
Pup abundance varied across years throughout the archipelago, decreasing about 40% during the El Niño event, and increased at an average annual rate of 5% between 2014 and 2018. These trends were maintained in the southeastern rookeries, while in other regions, the recovery rate was lower. This suggests that northern and western regions are less important as a breeding rookery, and rather these sites could be used as haul-out areas for GSL. The breeding success and growth of rookeries around the archipelago are linked to feeding resources available for mothers54,55,56. Therefore, a low trophic efficiency related to a different diet in females from the northern and western regions could be reflected in the annual pup abundance. There are spatial differences in the GSL diet that suggests the presence of specific foraging areas with distinct prey components for each region53. The western region has the greatest diversity of prey species, however, low-calorie fish as myctophids are their main prey. While the main prey in the central and southeastern regions are larger fish of better caloric quality, such as sand bass and mottled scorpionfish53,55.
Galapagos fur seal’s population trend
GFS populations have undergone a drastic decline related to inter-annual warming events and anthropogenic stressors, such as the 19th-century commercial sealing operations in the Galapagos Archipelago28,57. Like GSL, its population peaked in 2014 with a 38% decline in 2015 during the strong El Niño event conditions. The total population in the archipelago has always been low in comparison to GSL, however, these conditions are not maintained in sympatric rookeries as Cabo Hammond and Cabo Douglas, where the number of GFS is approximately five times that of GSL. The potential trophic overlap between GFS and GSL seems to be linked to environmental variability since these otariids tend to overlap in their foraging zones during inter-annual warm events such as El Niño24. These conditions could favor the GFS populations, since being the most abundant in the western region, they would easily displace the GSL populations. This behavior has already been observed on the coasts of Uruguay, where the South American fur seal (Arctocephalus australis) is the most abundant species, a product of a gradual displacement of the South American sea lion (Otaria flavences), that has significantly decreased its population58.
Most individuals are concentrated on a few rookeries in the western and northern islands of the archipelago, with 76.7% of the population found in Fernandina and Isabela islands. Western islands are situated in a region of strong upwelling and high productivity33,34, suggesting a geostrophic association to this hotspot within a tropical environment in the equatorial Pacific Ocean24,25. The GFS is the smallest of all otariids and exhibits an unusually restricted geographic range for a pinniped23,59, accompanied by high fidelity to its breeding rookeries that might even have reduced their genetic variability60. The link between high levels of philopatry and resource availability in otariids could exert strong geographical effects on its population dynamics45,46. It is then possible that the regional population trends of GFS are related to high-quality habitat and greater availability of resources24,61.
Pup abundances varied across years, decreasing about 36.7% during the El Niño event. The average annual growth rate was calculated in 6.3% between 2014 and 2018. This trend was maintained in western rookeries, while in other regions the recovery rates were lower, suggesting that the central and northern regions are less important as breeding rookeries and that these sites could be recolonization areas. There are disproportionately large populations in the western region concerning central and northern regions. The Santiago Island population in the central region is the smallest rookery, and apparently, it is a rookery which has been stable for the past two decades. Likewise, the Pinta Island population in the north region constituted 14% of the entire Galapagos population and showed an important population increase between 2001 and 2018. These results indicate the most significant increase was from 2001 (40 individuals counted26) to 2014–18 (an average of 313 individuals counted), making it the third-largest rookery to date. A dispersal of individuals from the western region to Pinta Island to escape crowding and local competition could explain this population increase. Population dispersions motivated by density-dependent elements have been reported in other pinnipeds as a strategy to facilitate their population growth62,63.
The last population monitoring carried out in 2002 indicated a reduction of more than 60% in a period of 24 years relative to counts from 197826,30. However, these results are not the best estimates of GFS abundance28. Our results demonstrate GFS populations seem to have remained stable over the past few decades. The average count for 2014 to 2018 is close to the count in 2001, suggesting minimal changes in abundance over the last 20 years. Age structure data are not available for GFS. However, based on information from other Arctocephalus, the average age of reproduction may be estimated anywhere between 9 and 12 years of age, with maximum longevity of 20 years64,65. This shows that the GFS population maintains a 50% reduction during the last four generations (1978–2018), which suggests the conservation status of this species should remain Endangered. This important population decline may also be associated with the impacts of strong El Niño events in 1982/83 and 1997/98, which caused acute shortages of prey and high rates of mortality48,66.
From the management perspective, there is a great interest to know the GFS population size. Unfortunately, there is little information on the population dynamics of this species that allows estimating specific correction factors for the counts on land. As an alternative, the GSL correction factors could be applied in the GFS counts taking into account that both pinnipeds display certain similarities in their life histories. For example, both otariids exhibit high fidelity to their breeding rookeries10,26,30 and feeding grounds55,61, show similarities in the maternal care and reproductive strategies35,67,68 and other adaptation strategies to the Galapagos environment69. In this way, the current population size of GFS was estimated at ~ 13,000 individuals (Table S3). However, we do not rule out the possibility of over-or underestimates of the GFS population as the correction factors to counts are not species-specific. Thus, further studies are needed to improve our knowledge and the accuracy of these estimates.
Oceanographic conditions and pup abundance
Galapagos pinnipeds reflect short-term changes in response to inter-annual warm events such as the El Niño event22,48, which generate a strong depletion of main prey availability and changes in foraging patterns24,66. The oceanographic conditions recorded in the Galapagos Archipelago show that the effects of the El Niño event on the population dynamics of GSL and GFS tend to remain for at least the next two years, and after this time there is a notable recovery. Both species are top predators in the region24,51, therefore, long-term warming would produce a progressive habitat change affecting multiple generations70. California sea lions (Zalophus californianus) and Guadalupe fur seals (Arctocephalus townsendi) also showed a decline in the pups’ production during the El Niño event 2014–2015, which, in both otariids, was associated with nutritional stress in females and the potential impact on reproductive success15. While northern elephant seals (Mirounga angustirostris) populations of Baja California show a decline in their abundance because the animals are not migrating as far south to avoid thermal stress (metabolic rate increases) that is associated with warming sea and its effect on air temperature71. Thus, it is essential to establish accurate population trends in pinnipeds during the presence of anomalous oceanographic events to predict potential changes under climate change scenarios.
The interannual variability in GSL pup abundance showed a positive relationship with the chlorophyll-a values and a negative relationship with the subthermocline temperature values in western and southeastern regions. Both environmental variables averaged over the period from August to October in each year. However, unexpectedly the northern and central regions showed an inverse relationship with both variables, which could be due to the oceanographic and demographic characteristics of these regions. The northern region is characterized by having a slight influence of the South Equatorial Current and Panama Bight Cyclonic Gyre that causes a low marine production32, while the central region is considered a mixing area33. Both conditions limit the resilience of GSL females since after anomalous oceanographic events (such as El Niño) they usually need between one and two years to regain their reproductive synchrony47,72. While the upwellings of the western region33, and the greater abundance of females in the southeast region10 could compensate or facilitate the generation of new cohorts.
The GFS pup abundance showed a positive relationship with the chlorophyll-a values and a negative relationship with the subthermocline temperature values in all regions, except for the central region that showed a negative relationship with the chlorophyll-a values. This discrepancy could be explained by the limited GFS population in the only rookery located in the central region, a condition that would decrease the birth rate of the species. Climate variability strongly influences marine productivity, with repercussions in the trophic food web all the way up to top predators73,74. Thus, chlorophyll-a is highly correlated with other variables, such as temperature in the ocean33,34, and is a good indicator of the climate variability that affects the reproductive success of Galapagos pinnipeds and the abundance of their prey10,75. GSL and GFS exhibited seasonal fluctuations according to the availability of their main prey66,75. This flexibility in the trophic behavior has enabled them to adapt to the changing conditions of the Galapagos Archipelago and thereby improve their survival55,76.
The Galapagos Archipelago is a region where levels of marine productivity are unpredictable compared to other upwelling systems at higher latitudes77,78. This creates an ecological challenge for Galapagos pinnipeds, so their population trend changes could be associated with a diet change79,80. In years of unusually good feeding conditions, females apparently regulate feeding efforts by staying ashore longer than by shortening their foraging trips67,68. In contrast, during anomalous low-prey conditions (El Niño years), females extend their trips to the sea, abandoning their pups and increasing mortality in this age category35,48. The presence of El Niño conditions also impacts California sea lion and South American sea lion (Otaria flavences) populations modifying main prey availability, increasing foraging effort, and thus decreasing the number of time females spend on land, which causes a negative effect on pups’ body condition and survival14,15. The GSL and GFS are highly philopatric species and restrict their foraging trips to areas located within 70 km from the coast25,61. While GSL shows a trophic specialization accompanied by different foraging strategies and different prey55,81, during ENSO events, some overlap may occur in GSL foraging zones, accompanied by a change of foraging strategies and main prey choices75.
The energetic costs of living in a warm climate with lower resource availability may have selected for a reduction in metabolic rate in other otariids59. This highlights the fundamental role of the physical environment in shaping the physiology of these species56,59. Reduced prey availability in El Niño years causes females to increase their foraging effort (more time at sea), so the pups receive less energy and their survivability decreases14,48. There is a negative relationship between sea temperature levels and the early development of GSL pups, thus, during warm conditions, there are smaller pups with reduced weight36,82. Both species are among the otariids with the longest lactation periods (2–3 years35). The extension of maternal investment in these otariids is a response mechanism to increase the chances of pup survival when populations are exposed to food stress due to drastic changes in oceanographic conditions35,83.
Conclusions and recommendations
The monitoring of Galapagos pinnipeds between 2014 and 2018, incorporating annual population-wide surveys, has recorded short-term changes in response to inter-annual warming events such as the El Niño event. It is understood that there are multiple reasons for monitoring these endangered species. This work highlights key parameters that need to be measured to understand population trends, while also providing up-to-date information of their abundance to assist with the prioritizing of rookeries. Our results show that population information of both species may have localized relevance. Therefore, adequate protection probably requires that each rookery be managed according to its specific conditions. For example, rookeries identified as important breeding sites should receive additional protection because they may become source populations for other rookeries. The Galapagos pinniped monitoring program must continue providing data to help local resource managers make effective decisions. Spatial abundance data and knowledge of population trends allow establishing effective protected areas and facilitate wildlife management. Complementary research programs investigating the feeding patterns, health status, and pup development need to be coordinated, along with demographic information to understand the population dynamics of Galapagos pinnipeds. Finally, we recommend that the potential funds designed for monitoring be maintained over time and that the global census be continued in the long-term to accurately predict the GSL and GFS population trends in the archipelago.
References
Baylis, A. M. et al. Disentangling the cause of a catastrophic population decline in a large marine mammal. Ecology 96, 2834–2847 (2015).
Hernández-Camacho, C. & Trites, A. Population viability analysis of Guadalupe fur seals Arctocephalus townsendi. Endanger. Species Res. 37, 255–267 (2018).
Lee, O. A., Burkanov, V. & Neill, W. H. Population trends of northern fur seals (Callorhinus ursinus) from a metapopulation perspective. J. Exp. Mar. Biol. Ecol. 451, 25–34 (2014).
Williams, P. J., Hooten, M. B., Womble, J. N., Esslinger, G. G. & Bower, M. R. Monitoring dynamic spatio-temporal ecological processes optimally. Ecology 99(3), 524–535 (2018).
Wege, M. et al. Trend changes in sympatric Subantarctic and Antarctic fur seal pup populations at Marion Island. Southern Ocean. Mar. Mam. Sci. 32(3), 960–982 (2016).
McIntosh, R. R. et al. Understanding meta-population trends of the Australian fur seal, with insights for adaptive monitoring. PLoS ONE 13(9), e0200253. https://doi.org/10.1371/journal.pone.0200253 (2018).
Harwood, J. & Prime, J. H. Some factors affecting the size of British grey seal populations. J. Appl. Ecol. 15, 401–411 (1978).
Trillmich, F. et al. On the challenge of interpreting census data: Insights from a study of an endangered pinniped. PLoS ONE 12(5), e0154588. https://doi.org/10.1371/journal.pone.0154588 (2016).
Taylor, R. H., Barton, K. J., Wilson, P. R., Thomas, B. W. & Karl, B. Population status and breeding of New Zealand fur seals (Arctocephalus forsteri) in the Nelson–northern Marlborough region, 1991–94. New. Zeal. J. Mar. Fresh. 29, 223–234 (1995).
Riofrío-Lazo, M., Arreguín-Sánchez, F. & Páez-Rosas, D. Population abundance of the endangered galapagos Sea Lion Zalophus wollebaeki in the Southeastern galapagos archipelago. PLoS ONE 12(1), e0168829. https://doi.org/10.1371/journal.pone.0168829 (2017).
García-Aguilar, M. C., Elorriaga-Verplancken, F. R., Rosales-Nanduca, H. & Schramm, Y. Population status of the Guadalupe fur seal (Arctocephalus townsendi). J. Mammal. 99(6), 1522–1528 (2018).
Forcada, J., Trathan, P. N., Reid, K. & Murphy, E. J. The effects of global climate variability in pup production of Antarctic fur seals. Ecology 86(9), 2408–2417 (2005).
Taylor, B. L., Martinez, M., Gerrodette, T. & Barlow, J. Lessons from monitoring trends in abundance of marine mammals. Mar. Mam. Sci. 23, 157–175 (2007).
Soto, K. H., Trites, A. W. & Arias-Schreiber, M. The effects of prey availability on pup mortality and the timing of birth of South American sea lions (Otaria flavescens) in Peru. J. Zool. 264, 419–428 (2004).
Elorriaga-Verplancken, F., Sierra-Rodríguez, G., Rosales-Nanduca, H., Acevedo-Whitehouse, K. & Sandoval-Sierra, J. Impact of the 2015 El Niño-Southern Oscillation on the Abundance and Foraging Habits of Guadalupe Fur Seals and California Sea Lions from the San Benito Archipelago. Mexico. PLoS ONE. 11(5), e0155034. https://doi.org/10.1371/journal.pone.0155034 (2016).
Villegas-Amtmann, S., Simmons, S. E., Kuhn, C. E., Huckstadt, L. A. & Costa, D. P. Latitudinal range influences the seasonal variation in the foraging behavior of marine top predators. PLoS ONE 6(8), e23166. https://doi.org/10.1371/journal.pone.0023166 (2011).
Hoskins, A. J., Costa, D. P., Wheatley, K. E., Gibbens, J. R. & Arnould, J. P. Influence of intrinsic variation on foraging behaviour of adult female Australian fur seals. Mar. Ecol. Prog. Ser. 526, 227–239 (2015).
Guinet, C., Jouventin, P. & Georges, J. Y. Long term population changes of fur seals Arctocephalus gazella and Arctocephalus tropicalis on subantarctic (Crozet) and subtropical (St. Paul and Amsterdam) islands and their possible relationship to El Niño southern oscillation. Antarct. Sci. 6(4), 473–478 (1994).
Chilvers, B. L., Wilkinson, I. S. & Childerhouse, S. New Zealand sea lion, Phocarctos hookeri, pup production 1995 to 2006. New. Zeal. J. Mar. Fresh. 41, 205–213 (2007).
Weise, M. J. & Harvey, J. T. Temporal variability in ocean climate and California sea lion diet and biomass consumption: implications for fisheries management. Mar. Ecol. Prog. Ser. 373, 157–172 (2008).
Robinson, H., Thayer, J., Sydeman, W. J. & Weise, M. J. Changes in California sea lion diet during a period of substantial climate variability. Mar. Biol. 165, 169. https://doi.org/10.1007/s00227-018-3424-x (2018).
Trillmich, F. & Dellinger, T. The Effects of El Niño on Galapagos Pinnipeds. In: Pinnipeds and El Niño. Ecological Studies (Analysis and Synthesis) (eds. Trillmich, F., & Ono, K. A.) 66–74 (Springer, Heidelberg, 1991).
Churchill, M., Boessenecker, R. & Clementz, M. Colonization of the Southern Hemisphere by fur seals and sea lions (Carnivora: otariidae), revealed by combined evidence phylogenetic and Bayesian biogeographic analysis. Zool. J. Linn. Soc. 172, 200–225 (2014).
Páez-Rosas, D., Aurioles-Gamboa, D., Alava, J. J. & Palacios, D. Stable isotopes indicate differing foraging strategies in two sympatric otariids of the Galapagos Islands. J. Exp. Mar. Bio. Ecol. 425, 44–52 (2012).
Villegas-Amtmann, S., Jeglinski, J. W. E., Costa, D. P., Robinson, P. W. & Trillmich, F. Individual foraging strategies reveal niche overlap between endangered Galapagos Pinnipeds. PLoS ONE 8(8), e70748. https://doi.org/10.1371/journal.pone.0070748 (2013).
Alava, J. J. & Salazar, S. Status and conservation of otariids in Ecuador and the Galapagos Islands. In: Sea lions of the world (eds. Trites, A. W., et al.) 495–519 (Fairbanks, 2006).
Trillmich, F. Zalophus wollebaeki: The IUCN Red List of Threatened Species. e.T41668A45230540; https://dx.doi.org/https://doi.org/10.2305/IUCN.UK.2015-2.RLTS.T41668A45230540.en (2015).
Trillmich, F. Arctocephalus galapagoensis. The IUCN Red List of Threatened Species. e.T2057A45223722; https://dx.doi.org/https://doi.org/10.2305/IUCN.UK.2015-2.RLTS.T2057A45223722.en (2015).
Páez-Rosas, D. & Guevara, N. Management strategies and conservation status of Galapagos sea lion populations at San Cristóbal Island, Galapagos, Ecuador. In: Tropical Pinnipeds: Bio- Ecology, Threats and Conservation (ed. Alava, J. J.) 159–175 (Taylor & Francis Group, Abingdon, 2017).
Trillmich, F. Galapagos sea lions and fur seals. Noticias de Galápagos. 29, 8–14 (1979).
Wolf, J. B. et al. Tracing early stages of species differentiation: Ecological, morphological and genetic divergence of Galápagos sea lion populations. BMC Evol. Biol. 8, 150. https://doi.org/10.1186/1471-2148-8-150 (2008).
Palacios, D. Seasonal patterns of sea surface temperature and ocean color around the Galapagos: regional and local influences. Deep Sea Res. Part II Top. Stud. Oceanogr. 51, 43–57 (2004).
Palacios, D., Bograd, S., Foley, D. & Schwing, F. Oceanographic characteristics of biological hot spots in the North Pacific: A remote sensing perspective. Deep Sea Res. Part II Top. Stud. Oceanogr. 53(3), 250–269 (2006).
Schaeffer, B. et al. Phytoplankton biomass distribution and identification of productive habitats within the Galapagos Marine Reserve by MODIS, a surface acquisition system. Remote Sens. Environ. 112(6), 3044–3054 (2008).
Trillmich, F. & Wolf, J. B. Parent-offspring and sibling conflict in Galapagos fur seals and sea lions. Behav. Ecol. Sociobiol. 62, 363–375 (2008).
Mueller, B., Pörschmann, U., Wolf, J. B. & Trillmich, F. Growth under uncertainty: the influence of marine variability on early development of Galapagos sea lions. Mar. Mam. Sci. 27, 350–365 (2011).
Bonnell, M. L. & Ford, R. G. California sea lion distribution: a statistical analysis of aerial transect data. J. Wildl. Manage. 51, 13–20 (1987).
Edgar, G. J. et al. Conservation of threatened species in the Galapagos Marine Reserve through identification and protection of marine key biodiversity areas. Aqua. Conserv. Mar. Freshw. Ecosyst. 18, 955–968 (2008).
Edgar, G. J., Banks, S., Fariña, J. M., Calvopiña, M. & Martínez, C. Regional biogeography of shallow reef fish and macro-invertebrate communities in the Galapagos archipelago. J. Biogeogr. 31, 1107–1124 (2004).
Ruttenberg, B. I., Haupt, A. J., Chiriboga, A. I. & Warner, R. R. Patterns, causes and consequences of regional variation in the ecology and life history of a reef fish. Oecologia 145, 394–403 (2005).
Wellington, G. M., Strong, A. E. & Merlen, G. Sea surface temperature variation in the Galápagos Archipelago: a comparison between AVHRR nighttime satellite data and in-situ instrumentation (1982–1988). Bull. Mar. Res. 69, 27–42 (2001).
Le Boeuf, B. J. et al. Size and distribution of the California sea lion population in Mexico. Proc. Calif. Acad. Sci. 43, 77–85 (1983).
Fielder, P. C. & Talley, L. D. Hydrography of the tropical Eastern Pacific: a review. Prog. In Ocean. 69, 143–180 (2006).
Brand, L. E., Guillard, R. R. & Murphy, L. S. A method for therapid and precise determination of acclimated phytoplankton reproduction rates. J. Plankton. Res. 3, 193–201 (1981).
Riedman, M. The Pinnipeds: seals, sea lions and walruses (University of California Press, 1990).
Costa, D. P., Weise, M. J. & Arnould, J. P. Potential influences of whaling on the status and trends of pinniped populations (University of California Press, 2006).
Kalberer, S., Meise, K., Trillmich, F. & Krüger, O. Reproductive performance of a tropical apex predator in an unpredictable habitat. Behav. Ecol. Sociobiol. 72, 108. https://doi.org/10.1007/s00265-018-2521-7 (2018).
Trillmich, F. & Limberger, D. Drastic effects of El Niño on Galapagos pinnipeds. Oecologia 67(1), 19–22 (1985).
Cai, W. et al. Increasing frequency of extreme El Niño events due to greenhouse warming. Nat. Clim. Change. 4, 111–116 (2014).
Wang, G., et al. Continued increase of extreme El Niño frequency long after 1.5 C warming stabilization. Nat. Clim. Change. 7, 568–572 (2017).
Páez-Rosas, D., Rodríguez-Pérez, M. & Riofrío-Lazo, M. Competition influence in the segregation of the trophic niche of otariids: a case study using isotopic bayesian mixing models in Galapagos pinnipeds. Rapid Commun. Mass Spectrom. 28, 2550–2558 (2014).
Jeglinski, J. W. E., Wolf, J. B., Werner, C., Costa, D. & Trillmich, F. Differences in foraging ecology align with genetically divergent ecotypes of a highly mobile marine top predator. Oecologia 179, 1041–1052 (2015).
Páez-Rosas, D. & Aurioles-Gamboa, D. Spatial variation in the foraging behaviour of the Galapagos sea lions (Zalophus wollebaeki) assessed using scat collections and stable isotope analysis. J. Mar. Biol. Assoc. U.K. 94, 1099–1107 (2014).
Drago, M. et al. Stable Isotopes Reveal Long-Term Fidelity to Foraging Grounds in the Galapagos Sea Lion (Zalophus wollebaeki). PLoS ONE 11(1), e0147857. https://doi.org/10.1371/journal.pone.0147857 (2016).
Páez-Rosas, D., Villegas-Amtmann, S. & Costa, D. P. Intraspecific variation in feeding strategies of Galapagos sea lions: A case of trophic specialization. PLoS ONE 12(10), e0185165. https://doi.org/10.1371/journal.pone.0185165 (2017).
Villegas-Amtmann, S., McDonald, B., Páez-Rosas, D., Aurioles-Gamboa, D. & Costa, D. P. Adapted to change: Low energy requirements in a low and unpredictable productivity environment, the case of the Galapagos sea lion. Deep Sea Res. Part II Top. Stud. Oceanogr. 140, 94–104 (2017).
Heller, E. Mammals of the Galapagos Archipelago: Exclusive of the Cetacea (University of Harvard Press, 1904).
Franco-Trecu, V., Aurioles-Gamboa, D., Arim, M. & Lima, M. Pre-partum and postpartum trophic segregation between sympatrically breeding female Arctocephalus australis and Otaria flavenscens. J. Mammal. 93(2), 514–521 (2012).
Trillmich, F. & Kooyman, G. L. Field metabolic rate of lactating female Galapagos fur seals (Arctocephalus galapagoensis): the influence of offspring age and environment. Comp. Biochem. Phys. A. 129(4), 741–749 (2001).
Lopes, F. et al. Fine-scale matrilineal population structure in the Galapagos fur seal and its implications for conservation management. Conserv. Genet. 16, 1099–1113 (2015).
Jeglinski, J. W. E., Goetz, K. T., Werner, C., Costa, D. P. & Trillmich, F. Same size–same niche? Foraging niche separation between sympatric juvenile Galapagos sea lions and adult Galapagos fur seals. J. Anim. Ecol. 82, 694–706 (2013).
Lowry, L. F., Frost, K. J., Davis, R., DeMaster, D. P. & Suydam, R. S. Movements and behavior of satellite-tagged spotted seals (Phoca largha) in the Bering and Chukchi Seas. Polar Biol. 19, 221–230 (1998).
O’Corry-Crowe, G., Gelatt, T., Rea, L., Bonin, C. & Rehberg, M. Crossing to safety: dispersal, colonization and mate choice in evolutionarily distinct populations of Steller sea lions. Eumetopias jubatus. Mol. Ecol. 23, 5415–5434 (2014).
Forcada, J., Trathan, P. N. & Murphy, E. J. Life history buffering in Antarctic mammals and birds against changing patterns of climate and environmental variation. Glob. Chang. Biol. 14, 2473–2488 (2008).
Pacifici, M. et al. Generation length for mammals. Nat. Conserv. 5, 87–94 (2013).
Dellinger, T. & Trillmich, F. Fish prey of the sympatric Galapagos fur seals and sea lions: seasonal variation and niche separation. Can. J. Zool. 77, 1204–1216 (1999).
Trillmich, F. Attendance behavior of Galapagos sea lions. In: Fur Seals: Maternal Strategies on Land and at Sea. (eds. Gentry, R. L. & Kooyman, G. L.) 196–208 (Princeton University Press, 1986).
Trillmich, F. Maternal investment and sex-allocation in the Galapagos fur seal. Arctocephalus galapagoensis. Behav. Ecol. Sociobiol. 19(3), 157–164 (1986).
Riofrío-Lazo, M. & Páez-Rosas, D. Galapagos sea lions and fur seals: adaptations to environmental variability. In: Ethology and Behavioral Ecology of Otariids and the Odobenid (eds. Campagna, C. & Harcourt, R.) (Springer Science & Business Media, Germany, 2021).
Adame, F., Elorriaga-Verplancken, F., Beier, E., Acevedo-Whitehouse, K. & Pardo, M. The demographic decline of a sea lion population followed multi-decadal sea surface warming. Sci. Rep. 10, 10499. https://doi.org/10.1038/s41598-020-67534-0 (2020).
García-Aguilar, M. C., Turrent, C., Elorriaga-Verplancken, F. R., Arias-Del-Razo, A. & Schramm, Y. Climate change and the northern elephant seal (Mirounga angustirostris) population in Baja California. Mexico. PLoS ONE. 13(2), e0193211. https://doi.org/10.1371/journal.pone.0193211 (2018).
Mueller, B., Porschmann, U., Wolf, J. B. W. & Trillmich, F. Growth under uncertainty: the influence of marine variability on early development of Galapagos sea lions. Mar. Mammal. Sci. 27, 350–365 (2011).
Doney, S. C. et al. Climate change impacts on marine ecosystems. Ann. Rev. Mar. Sci. 4, 11–37 (2011).
Wernberg, T. et al. An extreme climatic event alters marine ecosystem structure in a global biodiversity hotspot. Nat. Clim. Change. 3, 78–82 (2013).
Páez-Rosas, D., Moreno-Sánchez, X., Tripp-Valdez, A., Elorriaga-Verplancken, F. & Carranco-Narváez, S. Changes in the Galapagos sea lion diet as a response to El Niño-Southern Oscillation. Reg. Stud. Mar. Sci. 40, 101485. https://doi.org/10.1016/j.rsma.2020.101485 (2020).
Urquía, D. & Páez-Rosas, D. δ13C and δ15N values in pup whiskers as a proxy for the trophic behavior of Galapagos sea lion females. Mamm. Biol. 96, 28–36 (2019).
Martin, J. H. et al. Testing the iron hypothesis in ecosystems of the equatorial Pacific-Ocean. Nature 371(6493), 123–129 (1994).
Sakamoto, C.M., et al. Surface seawater distributions of inorganic carbon and nutrients around the Galapagos Islands: results from the PlumEx experiment using automated chemical mapping. Deep Sea Res. Part 2 Top Stud. Oceanogr. 45(6), 1055–1071 (1998).
Fritz, L. W. & Hinckley, S. A. Critical review of the regime shift “junk food” nutritional stress hypothesis for the decline of the western stock of Steller sea lion. Mar. Mammal. Sci. 21, 476–518 (2005).
McClatchie, S. et al. Food limitation of sea lion pups and the decline of forage off central and southern California. R. Soc. Open Sci. 3, 150628. https://doi.org/10.1098/rsos.150628 (2016).
Villegas-Amtmann, S., Costa, D., Tremblay, Y., Aurioles-Gamboa, D. & Salazar, S. Multiple foraging strategies in a marine apex predator, the Galapagos Sea Lion. Mar. Ecol. Prog. Ser. 363, 299–309 (2008).
Kraus, C. et al. Mama’s boy: sex differences in juvenile survival in a highly dimorphic large mammal, the Galapagos sea lion. Oecologia 171, 893–903 (2013).
Gentry, R. L. & Kooyman, G. L. Fur seals: Maternal Strategies on Land and at Sea (Princeton University Press, 1987).
Acknowledgements
We thank the Galapagos National Park Directorate (GNPD) for providing logistical support and for permitting us to collect the data used in this study. We also thank the Universidad San Francisco de Quito (USFQ) for financial and logistical support during the preparation of this manuscript. We are grateful to GNPD research technicians, rangers, and the crew of the "M/V Sierra Negra" ship for their help in collecting and processing the biological material used in this study. We offer special gratitude to all members of the USFQ/UNC/GNPD research expeditions for providing their invaluable work during the collection of data. We are grateful for anonymous reviewers that greatly helped to clarify our manuscript. Finally, we thank Carlos Mena, Steve Walsh, Diego Quiroga and all staff of the Galapagos Science Center (GSC) for providing the facilities for information processing and analysis.
Funding
This work was supported by Galapagos Science Center (Galapagos research expeditions 2014-2018).
Author information
Authors and Affiliations
Contributions
Conceptualization: D.P-R. and M.R-L. Collected census data: D.P-R., J.T., E.E. Collected oceanographic data: A.M and H.S. Designed field methodology: D.P-R. and J.T. Funding acquisition: D.P-R., A.M. and H.S. Formal analysis: D.P-R. and M.R-L. Writing-original draft: D.P-R. and M.R-L. Writing-review and editing: D.P-R., J.T., E.E., A.M., H.S., and M.R-L. All authors gave final approval for publication.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Páez-Rosas, D., Torres, J., Espinoza, E. et al. Declines and recovery in endangered Galapagos pinnipeds during the El Niño event. Sci Rep 11, 8785 (2021). https://doi.org/10.1038/s41598-021-88350-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-88350-0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.