Abstract
Pneumonia is a leading cause of mortality among older adults worldwide. Recently, several studies reported that frailty was associated with mortality among older adults hospitalized due to respiratory infectious diseases, including pneumonia. However, it is unknown whether frailty is associated with susceptibility to and severity of pneumonia in functionally-independent community-dwelling older adults. In this study, we examined whether frailty increased the susceptibility to pneumonia and hospitalization in older adults. We used cross-sectional data from the Japan Gerontological Evaluation Study; the data was collected by using mail-based, self-reported questionnaires from 177,991 functionally-independent community-dwelling older adults aged ≥ 65 years. Our results showed that frailty was significantly associated with both occurrence of and hospitalization due to pneumonia after adjustments with covariates; (Preference ratio {PR} 1.92, 95% confidence interval {95% CI} [1.66–2.22] and PR 1.80, 95% CI [1.42–2.28], respectively, p < 0.001 for the both). Pre-frailty was associated only with the occurrence of pneumonia. Besides, the instrumental activity of daily living, physical strength, nutrition status, oral function, homeboundness, and depression status in frail older adults were associated with either or both occurrence of and hospitalization due to pneumonia. Our results suggest that frailty influenced the susceptibility to and severity of pneumonia in older adults.
Similar content being viewed by others
Introduction
Pneumonia is a major cause of mortality and morbidity among older adults worldwide1. Community-acquired pneumonia (CAP) is a leading cause of morbidity among community-dwelling older adults in many countries and is different from hospital-acquired pneumonia (HAP) and ventilator-associated pneumonia (VAP). The overall incidence rates of CAP have been estimates as 1790–4000 in Japan, 2015; 630–1463 in the US, 2015, and 1400 in Europe, 2013 per 100,000 older adults aged ≥ 65 years2,3,4. A recent report showed that 6.8 million episodes of clinical pneumonia, including CAP, resulted in hospital admissions of older adults globally in 20155.
Frailty is a state of increased vulnerability to stressors, including infectious diseases, which develops as a consequence of age‐related decline in multiple physiological and psychological systems, including the central nervous, endocrine, skeletal muscle, and immune systems6,7. A recent report showed that the pooled prevalence rates of frail older adults in 62 countries were 22–26%, based on the deficit accumulation model8. Several reports have recently shown that frailty was associated with increased severity and mortality of hospitalized older adults due to respiratory infectious diseases. Hewitt and colleagues showed that frailty in older adults hospitalized due to COVID-19 was associated with in-hospital mortality9. Lees and colleagues reported that the frailty of hospitalized older adults with influenza and acute respiratory illness was associated with lower odds of recovery10. Luo and colleagues recently reported that frailty was associated with the severity of CAP and mortality among hospitalized older adults11. However, it was unknown whether frailty was related to the susceptibility to and severity of pneumonia among community-dwelling older adults.
Therefore, we examined whether frailty was associated with susceptibility to pneumonia and hospitalization among functionally-independent community-dwelling older adults aged ≥ 65 years. We assessed associations between each physical or psychological condition in frailty and the prevalence and severity of pneumonia.
Methods
Study sample
The present study had a cross-sectional design and uses data from the Japan Gerontological Evaluation Study (JAGES). This project investigated the social determinants of health among non-institutionalized and functionally independent people aged 65 years or older, who did not receive benefits from public long-term care insurance (LTCI) between September 2016 and January 2017:
“The LTCI system was introduced in Japan in 2000 to address the demands of older persons with disabilities based on the concept of a user-oriented social insurance system with support for independence. Older people with a certification for LTCI service needs can utilize facility services, in-home services, and community-based services depending on their physical and cognitive impairments”12.
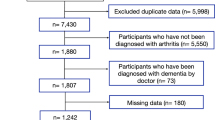
We used the data “JAGES 2016,” which was obtained from self-reported questionnaires mailed to and filled-in by community-dwelling individuals in 39 municipalities in 2016. In the JAGES 2016 wave, self-administered questionnaires were mailed to functionally independent adults aged 65 years or older who did not receive benefits from the LTCI insurance in Japan. The survey was conducted in the municipalities between September 2016 and January 2017. The data included 180,021 individuals who answered the questionnaires with the basic items (response rate was 70.2%). Participants who did not answer questions regarding age or sex were excluded (n = 2030). The data consisted of individuals who did not receive benefits from the LTCI on 1st April 2016. The data consisted of a three-stage hierarchal structure. The individual data were nested into 720 communities based on elementary or junior high school districts, and these communities were further nested into 39 municipalities.
Outcome variables
The occurrence of pneumonia and hospitalization due to it in the past year from September 2016 to January 2017 were the outcome variables. The occurrence of pneumonia was assessed by asking “Did you fall sick in the past year?” and instructing the participants to select an appropriate answer from the following items: “Influenza,” “Pneumonia”, and “none of them.” Hospitalization due to pneumonia was assessed for participants who answered “Influenza” or “pneumonia” in the previous question by asking “If the sickness was due to influenza or pneumonia, were you hospitalized with relation to it?” and instructing the participants to select an answer from the following options: “Not hospitalized,” “Hospitalized due to influenza,” “Hospitalized due to pneumonia,” “Contracted influenza while I was hospitalized for other diseases,” and “Developed pneumonia while I was hospitalized for other diseases.” Only participants who answered “Hospitalized due to pneumonia” were considered to be hospitalized. The participants who answered “Influenza” in the first question and “Hospitalized due to pneumonia” in the second were included because influenza can cause primary viral or secondary bacterial pneumonia13.
Frailty
Frailty was assessed using the Kihon Check List (KCL)14. KCL was developed by the Japanese Ministry of Health, Labor and Welfare to identify older adults requiring LTCI. KCL was included in the self-administered questionnaires of the JAGES 2016 wave. KCL consists of 25 questions classified into the following seven categories: instrumental activity of daily living (IADL), physical strength, nutritional status, oral function, homeboundness, cognitive function, and depressive mood (Supplemental Table 1). The scores from the KCL was well correlated with the validated assessments of physical strength, nutritional state, cognitive function, depressive mood, and the number of frailty phenotypes defined by the Cardiovascular Health Study criteria (CHS)14. Frailty was categorized into three groups based on KCL scores: robust, 0–3; pre-frail, 4–7; and frail, ≥ 8; scores were calculated from the KCL questions, which were validated with the pre-frail and frail categories established by the CHS criteria14. KCL variables were generated for each of the seven categories. Each KCL variable was categorized into three groups, based on answers to questions: 0, not applicable; 1, applicable; and 2 or ≥ 2 applicable.
Covariates
Age was classified into two groups (65–74 years and ≥ 75 years). Educational attainment was categorized into five groups: < 6 years, 6–9 years, 10–12 years, ≥ 13 years, and others. Equivalized income was calculated by dividing the normalized household gross income in 2015 by the square root of the number of household members, and was categorized into five groups: < 0.5, 0.50–0.99, 1.00–1.99, 2.00–3.99, and ≥ 4.00 million yen. The household structure was assessed by asking the respondents the question, “Who do you live with?”. They were asked to choose from the following options: “no one,” “spouse,” “son,” “daughter,” “spouse of child,” “grandchild,” “brother or sister, “father,” “mother,” “father-in-law,” “mother-in-law,” and “other.” The responses were classified into six groups as follows: living alone, living with a spouse, living with children, living with a spouse and children, living in three-generation households (living with/without a spouse, but with one of the sons/daughters/son’s or daughter’s spouse and grandchildren), and living in a household structure other than the above five categories. Marital status was assessed by asking, “What is your marital status?” and participants were instructed to select one from the following five options: “Married,” “Widowed,” “Divorced,” “Never married” and “Other.” Smoking status was assessed by asking, “Do you smoke cigarettes?” and the participants were instructed to select appropriate answers from the following items: “Never smoked,” “Quit smoking ≥ 5 years ago”, “Quit smoking < 5 years ago,” “Smoke sometimes” and “Smoke almost every day.” The population density of the municipality was categorized as follows: metropolitan (density over 4000 people per km2), urban (density between 1,500 and 4000 people per km2), semi-urban (density between 1000 and 1499 people per km2), and rural (density below 1,000 people per km2)15. A municipality dummy variable was generated to adjust for differences in municipalities’ policies in preventing frailty16. The diabetes, respiratory, heart, kidney/prostate gland, hematological, or immune disease status was assessed by asking participants whether they were receiving any treatment or experiencing after-effects of any of the above diseases. Pneumococcal vaccination status was assessed by asking the participants, “Did you get a pneumococcal vaccination in the last five years? They chose from the following options: “No,” “Yes, I got vaccinated using my municipality's subsidy”, and “Yes, but I did not get vaccinated using my municipality's subsidy”. The participants who chose the two latter options were categorized as vaccinated.
Statistical analysis
Multilevel Poisson regression analyses with random intercepts were performed to assess associations between frailty and pneumonia/hospitalization after adjusting for all the covariates. The data were structured in three levels: the individuals were nested within their elementary or junior high school districts and the districts were further nested within the municipalities. The covariates consist of all the covariates at individual-level and the municipality dummy variable used to adjust for differences in municipalities’ policies in older adult’s health including preventing frailty and pneumonia at municipality-level16,17. The individual-level covariates were: age group, sex, educational attainment, equivalized income, household structure, marital status, smoking status, municipality population density, diabetes, respiratory disease, heart disease, kidney/prostate gland disease, hematological/immune disease, and pneumococcal vaccination. The prevalence ratios (PRs) and 95% confidence intervals (95% CIs) were calculated after adjusting for all covariates. We used Stata version 14.2 (StataCorp., College Station, TX, USA) for all analyses, with a 2-tailed significance level set at 5%.
Ethical consideration
The process of obtaining informed consent in the present study was as follows: the questionnaire was sent by mail with the explanation of the study; the participants read the written explanation about the purpose of study and replied. Hence, we considered that informed consent was provided by those who replied and sent back the questionnaire. The JAGES protocol in 2016 was approved by the ethics committee of National Center for Geriatrics and Gerontology (No. 992) and the ethics committee of Chiba University (No. 2493). We followed the STROBE Statement to report our observational study. This study was performed in accordance with the principles of the Declaration of Helsinki. Informed consent was obtained from all participants.
Results
The relationship between prevalence rates of pneumonia and hospitalization with frailty and other characteristics of older adults
Table 1 compares the prevalence rates of pneumonia in the past year with the characteristics of community-dwelling older adults. Frail and pre-frail older adults were 3- and 1.5-times, respectively, more likely to contract pneumonia than non-frail adults. Older adults, with ≥ 1 applicable question in KCL related to IADL, physical strength, nutrition status, oral function, homeboundness, cognitive function, and depression status, were likely to contract pneumonia than those with a score = 0 (none of the questions were applicable). Older adults aged ≥ 75 years, who were male, had lower education/income, who lived in three-generation households, had “other” marital status, who had quit smoking < 5 years ago, and who lived in rural or semi-urban areas were more likely to contract pneumonia than those in other categories. Older adults with diabetes, respiratory, heart, kidney/prostate gland, and hematological/immune diseases, and those who received pneumococcal vaccination were more likely to contract pneumonia more than those without any disease.
Table 2 compares the prevalence rates of hospitalization due to pneumonia with the characteristics of older adults. Frail and pre-frail older adults were 3.3- and 1.6-times, respectively, more likely to be hospitalized than non-frail adults. Older adults, with ≥ 1 applicable question in KCL from all the seven categories were more likely to be hospitalized than those with a score = 0 (none of the questions were applicable). Older adults aged ≥ 75, who were male, had lower education/income, who lived in three-generation households, had “other” marital status, who had quit smoking < 5 years ago, and who lived in rural areas were more likely to be hospitalized than those in other categories. Older adults with diabetes, respiratory, heart, kidney/prostate gland, and hematological/immune diseases and who received pneumococcal vaccination were more likely to be hospitalized than those without any disease.
Frailty was associated with susceptibility to and severity of pneumonia among community-dwelling older adults
Figure 1 shows PR and 95% CI of the association between frailty and the occurrence of pneumonia in the past year among the community-dwelling older adults (upper side of the figure). After adjusting with all the covariates, both frailty and pre-frailty in older adults were significantly associated with the occurrence of pneumonia compared to non-frailty (PR 1.92 [95% CI 1.66–2.22] and PR 1.30 [95% CI 1.14–1.48], respectively, p < 0.001 for the both).
Prevalence ratios (PR) and 95% confidence intervals (95% CI) of associations between the occurrence of pneumonia and frailty or each of the Kihon checklist (KCL) categories in community-dwelling older adults. Numbers in KCL scores represent how many questions were applicable to older adults. *p < 0.05, **p < 0.01, ***p < 0.001. IADL, Instrumental Activity of Daily Living.
Figure 2 shows PR and 95% CI of the association between frailty and the hospitalization due to pneumonia among the community-dwelling older adults (upper side of the figure). After adjusting with all the covariates, frailty in older adults was significantly associated with the hospitalization due to pneumonia compared to non-frailty (PR 1.80 [95% CI 1.42–2.28], p < 0.001). Pre-frailty was not significantly associated with hospitalization compared to non-frailty (PR 1.23 [95% CI 0.98–1.53]).
Prevalence ratios (PR) and 95% confidence intervals (95% CIs) of associations between the hospitalization due to pneumonia and frailty or each of the Kihon checklist (KCL) categories in community-dwelling older adults. Numbers in KCL scores represent how many questions were applicable to older adults. *p < 0.05, **p < 0.01, ***p < 0.001. IADL, Instrumental Activity of Daily Living.
Associations between each of the KCL categories and pneumonia/hospitalization among community-dwelling older adults
Figure 1 shows PRs and 95% CIs of associations between each of the KCL categories and the occurrence of pneumonia in the past year (from middle to bottom of the figure). After adjusting with all the other KCL categories and covariates, the lower or lowest IADL (1 or ≥ 2 KCL questions were applicable) was negatively and significantly associated with the occurrence of pneumonia (PR 0.90, 95% CI [0.84–0.97], p < 0.01 or PR 0.79, 95% CI [0.72–0.86], p < 0.001, respectively). The lowest physical strength was significantly associated with the occurrence of pneumonia in older adults (PR 1.12, 95% CI [1.03–1.22], p < 0.01). The lower nutrition status was significantly associated with the occurrence of pneumonia (PR 1.09, 95% CI [1.004–1.14], p < 0.05). The lower or lowest oral function was significantly associated with the occurrence of pneumonia in older adults (PR 1.18, 95% CI [1.10–1.27] or PR 1.27, 95% CI [1.17–1.39], respectively, p < 0.001 for the both). The worse and worst cases of depression mode were significantly associated with the occurrence of pneumonia in older adults (PR 1.10, 95% CI [1.006–1.19], p < 0.05 and PR 1.13, 95% CI [1.04–1.23], p < 0.01, respectively).
Figure 2 shows PRs and 95% CIs of associations between each of the KCL categories and the hospitalization due to pneumonia in the past year (from middle to bottom of the figure).
After adjusting with all the other KCL categories and covariates, the lowest IADL was significantly associated with the hospitalization due to pneumonia (PR 1.24, 95% CI [1.01–1.53], p < 0.05). The lowest physical strength was significantly associated with the hospitalization due to pneumonia in older adults (PR 1.32, 95% CI [1.07–1.64], p < 0.01). The lower or lowest nutrition status was significantly associated with the hospitalization due to pneumonia (PR 1.54, 95% CI [1.30–1.82], p < 0.001 or PR 1.71, 95% CI [1.25–2.34], p < 0.01, respectively). The worst homeboundness was significantly associated with the hospitalization due to pneumonia in older adults (PR 1.60, 95% CI [1.11 − 2.12], p < 0.05]).
Discussion
We examined whether frailty was associated with the occurrence of and hospitalization due to pneumonia compared to non-frailty in community-dwelling older adults. Our analysis showed that frailty was significantly associated with both occurrence of and hospitalization due to pneumonia after adjusting for all covariates (Figs. 1 and 2) and that pre-frailty was significantly associated with the occurrence (Fig. 1). Besides, IADL, physical strength and nutrition status were significantly associated with the both occurrence of and hospitalization due to pneumonia, oral function and depression status were significantly associated with the occurrence, and homeboundness was significantly associated with the hospitalization (Figs. 1 and 2). Our results also suggested that frailty was associated with both susceptibility to and severity of pneumonia, and the six physical or psychological conditions in frailty were associated with either or both of them in community-dwelling older adults.
Several reports have recently shown that frailty was related to mortality in older adults hospitalized due to respiratory infectious diseases9,10,11. However, it was unknown whether frailty is associated with susceptibility to and severity of pneumonia in community-dwelling older adults. Our results showed that frailty was significantly associated with both occurrence of and hospitalization due to pneumonia in community-dwelling older adults, suggesting that frailty was associated with susceptibility to and severity of pneumonia in these adults (Figs. 1 and 2). Therefore, our results indicate the possibility that frailty may be one of the risk factors related to CAP and hospitalization in community-dwelling older adults. The pneumonia cases were likely CAP because HAP and VAP were excluded by asking corresponding questions to the participants (see “Outcome variables” in Methods). Our results also suggest that frailty may be an important indicator in the prevention of pneumonia among community-dwelling older adults, not only be an indicator of the mortality in older adults hospitalized for pneumonia as recently reported11.
Our results showed that low IADL was negatively and significantly associated with the occurrence of pneumonia after adjusting for all other KCL categories and covariates (Fig. 1). However, a negative association between low IADL and pneumonia has not been reported so far. Several reports have shown that community-dwelling older adults with low IADL were less social than those with normal IADL18,19,20. Social participation was reported to be associated with influenza among community-dwelling older adults21. Taken together, low IADL may present fewer opportunities for infection with respiratory infectious pathogens. However, our results showed that the lowest IADL was significantly associated with hospitalization among older adults (Fig. 2). Reichard and colleagues reported that adults with low IADL delayed or forewent receiving healthcare services twice more often than those with normal IADL, even if they were insured22. Older adults with the lowest IADL may have done the same when compared to those with normal IADL; they might have delayed visiting hospitals even if they had perceived subjective symptoms of pneumonia and hence, the pneumonia may have worsened and become severe leading to hospitalization.
The decline in immune function, so-called “immune senescence”, occurs in frail older adults; this is associated with the malfunction of the cellular and humoral immune systems6,23. Several research groups have reported that antibodies against influenza were more prevalent in physically active older adults than in sedentary individuals after vaccination; exercise enhanced the production of immunoglobulin A (IgA) secreted by the salivary gland in adults aged ≥ 6524,25,26. IgA is important for mucosal immunity as frontline protection against infections. Our results showed that the lowest physical strength was significantly associated with both occurrence of and hospitalization due to pneumonia (Figs. 1 and 2). Older adults with the lowest physical strength may be more susceptible to infections by pneumonia-causing pathogens due to decreased immunity, including the poor secretion of IgA; this may enable pathogens to invade the upper and lower respiratory tracts causing pneumonia and subsequent hospitalization.
Mitsutake and colleagues recently reported that socially-isolated and homebound older adults were significantly less likely to use outpatient and home medical care than those with neither characteristic, which may eventually lead to serious health problems that require more intensive treatment27. Our result showed that homeboundness was significantly associated with hospitalization due to pneumonia (Fig. 2). Older adults with homeboundness may not go to hospitals even if they recognize ills due to pneumonia. In addition, it may be difficult for physicians to have opportunities to diagnose pneumonia in those because they rarely visit hospitals. As a result, pneumonia in those with homeboundness may be severer and result in more hospitalizations. On the other hand, several reports have shown that homebound older adults were physically and psychologically unhealthy compared to those without homebound28,29,30. However, the physical and psychological conditions consisting of the IADL, physical strength, nutrition status, oral function, cognitive function and depression status were adjusted for the regression models to analyze the association between the hospitalization and homeboundness in our statistical analyses (Fig. 2). It suggests that there were the other reasons why the homeboundness was associated with the hospitalization among older adults than the physical or psychological frailty.
Several reports have already shown that poor nutrition is associated with CAP and hospitalization31,32,33. The decline in oral function, including poor swallowing and cough reflex, causes dysphagia, which induces aspiration pneumonia in frail older adults4,34,35,36,37. Geriatric depression is associated with the malfunction of the immune system and increased susceptibility to infection38,39,40. Our results showed that the nutrition status, oral function, and depression status were significantly associated with either or both occurrence of and hospitalization due to pneumonia (Figs. 1 and 2). The concordance between our results and the reported studies on the associations between the poor conditions and infectious diseases, including pneumonia, suggests that frailty is associated with susceptibility to pneumonia or hospitalization.
Our study has some limitations. First, this study has a cross-sectional design. Therefore, the possibility of pneumonia causing frailty cannot be completely excluded. However, the prevalence rate of pneumonia was 3.0% among all frail older adults (Table 1). Second, the occurrence of and hospitalization due to pneumonia were self-reported by the participants and not collected from medical records from the physicians in-charge. However, it is unlikely that they arbitrarily diagnosed themselves with pneumonia or non-medical professionals did so and they trusted it because most Japanese citizens have good medical access due to the national health coverage41. Third, we did not categorize the medical records based on the type of pneumonia, for example, CAP, HAP, or VAP. However, we expect most cases to be CAP because we excluded cases of nosocomial pneumonia with a question (see Methods). Besides, all the participants were functionally-independent community-dwelling older adults who did not receive benefits form the LTCI. Forth, it is possible that older adults with severe pneumonia could have been automatically excluded from the survey if the pneumonia was too severe to answer the questionnaire. Fifth, an information bias may have occurred in the survey for smoking if the participants did not honestly answer for their smoking status as self-reported-based assessments tend to underestimate smoking rates than the biological tests42. Therefore, our assessment might have counted the occurrence of pneumonia/hospitalization in the participants who did not honestly answer their smoking status despite actual smoking as pneumonia in those who never smoked or quit smoking (Tables 1 and 2). Sixth, a recall bias may have occurred in the survey for the pneumococcal vaccination. The question asked participants the vaccination status in the last five years, which they might not have been able to remember the vaccination if they had got it several years before the survey. Therefore, there may be participants who got the vaccination but did not remember the vaccination status during the survey (Tables 1 and 2). Besides, those who got the vaccination but contracted pneumonia could have been able to remember the vaccination status better than those who got the vaccination and did not contract pneumonia. As a result, a ratio of those who got the vaccination and contracted pneumonia may be overestimated (Tables 1 and 2).
Conclusion
It was unknown whether frailty was associated with the susceptibility to and severity of pneumonia among community-dwelling older adults, although recent reports have shown that it is related to mortality due to respiratory infectious diseases in hospitalized older adults. We examined whether frailty was associated with the occurrence of and hospitalization due to pneumonia in functionally-independent community-dwelling older adults. Our results showed that frailty was significantly associated with both occurrence of and hospitalization due to pneumonia and that pre-frailty was significantly associated with the occurrence after adjusting for all covariates. Our results showed that six of all the seven physical and psychological frailty were significantly associated with either occurrence of, hospitalization due to pneumonia, or the both as follows: the oral function and depression mode were associated with the occurrence, the homeboundness was associated with the hospitalization, and IADL, physical strength and nutrition status were associated with the both. Our results suggested that frailty was associated with susceptibility to and severity of pneumonia, which is a leading cause of mortality and morbidity among community-dwelling older adults worldwide. This study showed frailty as a risk factor for increased susceptibility to and severity of pneumonia among community-dwelling older adults. It is necessary to assess whether frailty is also significantly associated with CAP diagnosed by medical doctors and the causal relationship between frailty and pneumonia should be confirmed in longitudinal studies in the future.
Data availability
All data used are from the JAGES study and are not third-party data. All enquiries are to be addressed at the JAGES data management committee via e-mail: dataadmin.ml@jages.net. All JAGES datasets have ethical or legal restrictions for public deposition due to inclusion of sensitive information from the human participants. Following the regulation of local governments which cooperated on our survey, the JAGES data management committee has imposed the restrictions upon the data.
References
Troeger, C. et al. Estimates of the global, regional, and national morbidity, mortality, and aetiologies of lower respiratory tract infections in 195 countries: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Infect. Dis. 17, 1133–1161. https://doi.org/10.1016/s1473-3099(17)30396-1 (2017).
Morimoto, K. et al. The burden and etiology of community-onset pneumonia in the aging Japanese population: a multicenter prospective study. PLoS ONE 10, e0122247. https://doi.org/10.1371/journal.pone.0122247 (2015).
Jain, S. et al. Community-acquired pneumonia requiring hospitalization among US adults. N. Engl. J. Med. 373, 415–427. https://doi.org/10.1056/NEJMoa1500245 (2015).
Torres, A., Peetermans, W. E., Viegi, G. & Blasi, F. Risk factors for community-acquired pneumonia in adults in Europe: a literature review. Thorax 68, 1057–1065. https://doi.org/10.1136/thoraxjnl-2013-204282 (2013).
Shi, T. et al. Global and regional burden of hospital admissions for pneumonia in older adults: a systematic review and meta-analysis. J. Infect. Dis. 222, S570–S576. https://doi.org/10.1093/infdis/jiz053 (2020).
Clegg, A., Young, J., Iliffe, S., Rikkert, M. O. & Rockwood, K. Frailty in elderly people. Lancet 381, 752–762. https://doi.org/10.1016/s0140-6736(12)62167-9 (2013).
Fried, L. P. et al. Frailty in older adults: evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 56, M146-156. https://doi.org/10.1093/gerona/56.3.m146 (2001).
O’Caoimh, R. et al. Prevalence of frailty in 62 countries across the world: a systematic review and meta-analysis of population-level studies. Age Ageing https://doi.org/10.1093/ageing/afaa219 (2020).
Hewitt, J. et al. The effect of frailty on survival in patients with COVID-19 (COPE): a multicentre, European, observational cohort study. Lancet Public Health 5, e444–e451. https://doi.org/10.1016/s2468-2667(20)30146-8 (2020).
Lees, C. et al. Frailty hinders recovery from influenza and acute respiratory illness in older adults. J. Infect. Dis. 222, 428–437. https://doi.org/10.1093/infdis/jiaa092 (2020).
Luo, J., Tang, W., Sun, Y. & Jiang, C. Impact of frailty on 30-day and 1-year mortality in hospitalised elderly patients with community-acquired pneumonia: a prospective observational study. BMJ Open 10, e038370. https://doi.org/10.1136/bmjopen-2020-038370 (2020).
Yamada, M. & Arai, H. Long-term care system in Japan. Ann. Geriatr. Med. Res. 24, 174–180. https://doi.org/10.4235/agmr.20.0037 (2020).
Rothberg, M. B., Haessler, S. D. & Brown, R. B. Complications of viral influenza. Am. J. Med. 121, 258–264. https://doi.org/10.1016/j.amjmed.2007.10.040 (2008).
Satake, S. et al. Validity of the Kihon checklist for assessing frailty status. Geriatr. Gerontol. Int. 16, 709–715. https://doi.org/10.1111/ggi.12543 (2016).
Mochida, Y., Yamamoto, T., Fuchida, S., Aida, J. & Kondo, K. Does poor oral health status increase the risk of falls?: The JAGES project longitudinal study. PLoS ONE 13, e0192251. https://doi.org/10.1371/journal.pone.0192251 (2018).
Sato, K. et al. Intensity of community-based programs by long-term care insurers and the likelihood of frailty: multilevel analysis of older Japanese adults. Soc. Sci. Med. 245, 112701. https://doi.org/10.1016/j.socscimed.2019.112701 (2020).
Murakami, Y. et al. Factors associated with PPSV23 coverage among older adults in Japan: a nationwide community-based survey. BMJ Open 9, e030197. https://doi.org/10.1136/bmjopen-2019-030197 (2019).
Ke, Y., Jiang, J. & Chen, Y. Social capital and the health of left-behind older adults in rural China: a cross-sectional study. BMJ Open 9, e030804. https://doi.org/10.1136/bmjopen-2019-030804 (2019).
Fujihara, S. et al. Does community-level social capital predict decline in instrumental activities of daily living? A JAGES prospective cohort study. Int. J. Environ. Res. Public Health https://doi.org/10.3390/ijerph16050828 (2019).
Tomioka, K., Kurumatani, N. & Hosoi, H. Social participation and the prevention of decline in effectance among community-dwelling elderly: a population-based cohort study. PLoS ONE 10, e0139065. https://doi.org/10.1371/journal.pone.0139065 (2015).
Shobugawa, Y., Fujiwara, T., Tashiro, A., Saito, R. & Kondo, K. Social participation and risk of influenza infection in older adults: a cross-sectional study. BMJ Open 8, e016876. https://doi.org/10.1136/bmjopen-2017-016876 (2018).
Reichard, A., Stransky, M., Phillips, K., McClain, M. & Drum, C. Prevalence and reasons for delaying and foregoing necessary care by the presence and type of disability among working-age adults. Disabil. Health J. 10, 39–47. https://doi.org/10.1016/j.dhjo.2016.08.001 (2017).
Pansarasa, O. et al. Altered immune system in frailty: Genetics and diet may influence inflammation. Ageing Res. Rev. 54, 100935. https://doi.org/10.1016/j.arr.2019.100935 (2019).
Bachi, A. L. et al. Increased production of autoantibodies and specific antibodies in response to influenza virus vaccination in physically active older individuals. Res. Immunol. 3, 10–16. https://doi.org/10.1016/j.rinim.2013.01.001 (2013).
Sakamoto, Y. et al. Effect of exercise, aging and functional capacity on acute secretory immunoglobulin A response in elderly people over 75 years of age. Geriatr. Gerontol. Int. 9, 81–88. https://doi.org/10.1111/j.1447-0594.2008.00502.x (2009).
Kimura, F. et al. The effects of walking exercise training on immune response in elderly subjects. Int. J. Sport Health Sci. 4, 508–514. https://doi.org/10.5432/ijshs.4.508 (2006).
Mitsutake, S. et al. Influence of co-existing social isolation and homebound status on medical care utilization and expenditure among older adults in Japan. Arch. Gerontol. Geriatr. 93, 104286. https://doi.org/10.1016/j.archger.2020.104286 (2021).
Ornstein, K. A. et al. Epidemiology of the homebound population in the United States. JAMA Intern. Med. 175, 1180–1186. https://doi.org/10.1001/jamainternmed.2015.1849 (2015).
Herr, M., Latouche, A. & Ankri, J. Homebound status increases death risk within two years in the elderly: results from a national longitudinal survey. Arch. Gerontol. Geriatr. 56, 258–264. https://doi.org/10.1016/j.archger.2012.10.006 (2013).
Qiu, W. Q. et al. Physical and mental health of homebound older adults: an overlooked population. J. Am. Geriatr. Soc. 58, 2423–2428. https://doi.org/10.1111/j.1532-5415.2010.03161.x (2010).
Almirall, J., Serra-Prat, M., Bolibar, I. & Balasso, V. Risk Factors for community-acquired pneumonia in adults: a systematic review of observational studies. Respiration 94, 299–311. https://doi.org/10.1159/000479089 (2017).
Riquelme, R. et al. Community-acquired pneumonia in the elderly. Clinical and nutritional aspects. Am. J. Respir. Crit. Care Med. 156, 1908–1914. https://doi.org/10.1164/ajrccm.156.6.9702005 (1997).
Ma, H. M., Yu, R. H. & Woo, J. Recurrent hospitalisation with pneumonia is associated with higher 1-year mortality in frail older people. Intern. Med. J. 43, 1210–1215. https://doi.org/10.1111/imj.12258 (2013).
Almirall, J. et al. Oropharyngeal dysphagia is a risk factor for community-acquired pneumonia in the elderly. Eur. Respir. J. 41, 923–928. https://doi.org/10.1183/09031936.00019012 (2013).
Nishida, T., Yamabe, K. & Honda, S. Dysphagia is associated with oral, physical, cognitive and psychological frailty in Japanese community-dwelling elderly persons. Gerodontology 37, 185–190. https://doi.org/10.1111/ger.12455 (2020).
Ebihara, S., Ebihara, T. & Kohzuki, M. Effect of aging on cough and swallowing reflexes: implications for preventing aspiration pneumonia. Lung 190, 29–33. https://doi.org/10.1007/s00408-011-9334-z (2012).
Langmore, S. E., Skarupski, K. A. & Fries, B. E. Predictors of aspiration pneumonia in nursing home residents. Dysphagia 17, 298–307. https://doi.org/10.1007/s00455-002-0072-5 (2002).
Gale, S. D., Berrett, A. N., Erickson, L. D., Brown, B. L. & Hedges, D. W. Association between virus exposure and depression in US adults. Psychiatry Res. 261, 73–79. https://doi.org/10.1016/j.psychres.2017.12.037 (2018).
Morimoto, S. S. & Alexopoulos, G. S. Immunity, aging, and geriatric depression. Psychiatr Clin. N. Am. 34, 437–449. https://doi.org/10.1016/j.psc.2011.02.006 (2011).
Kiecolt-Glaser, J. K. & Glaser, R. Depression and immune function: central pathways to morbidity and mortality. J. Psychosom. Res. 53, 873–876. https://doi.org/10.1016/s0022-3999(02)00309-4 (2002).
Ikegami, N. et al. Japanese universal health coverage: evolution, achievements, and challenges. Lancet 378, 1106–1115. https://doi.org/10.1016/s0140-6736(11)60828-3 (2011).
Connor Gorber, S., Schofield-Hurwitz, S., Hardt, J., Levasseur, G. & Tremblay, M. The accuracy of self-reported smoking: a systematic review of the relationship between self-reported and cotinine-assessed smoking status. Nicotine Tob. Res. 11, 12–24, doi:https://doi.org/10.1093/ntr/ntn010 (2009).
Acknowledgements
This study used data from JAGES, which was conducted by the Nihon Fukushi University Center for Well-being and Society. We gratefully appreciate all participants who generously spent time and collaborated in that study. This work was supported by the Japan Science and Technology Agency-Supported Program on Open Innovation Platform with Enterprises, Research Institute and Academia (Grant no JPMJOP1831; Development issue No. 5). We thank Dr. Atsuo Amano, Department of Preventive Dentistry, Graduate School of Dentistry, Osaka University, Japan for his helpful comments on the preparation of this manuscript. We thank Editage (www.editage.com) for English language editing.
Funding
This study used data from JAGES. The present study was supported by Grant-in-Aid for Scientific Research (15H01972, 15H04781, 15H05059, 15K03417, 15K03982, 15K16181, 15K17232, 15K18174, 15K19241, 15K21266, 15KT0007, 15KT0097, 16H05556, 16K09122, 16K00913, 16K02025, 16K12964, 16K13443, 16K16295, 16K16595, 16K16633, 16K17256, 16K17281, 16K19247, 16K19267, 16K21461, 16K21465, 16KT0014, 17K04305,17K34567, 17K04306, 25253052, 25713027, 26285138, 26460828, 26780328, 18H03018, 18H04071, 18H03047, 18H00953, 18H00955, 18KK0057, 19H03901, 19H03915, 19H03860, 19K04785, 19K10641,19K11657,19K19818, 19K19455, 19K24060, 19K20909, 20H00557) from JSPS (Japan Society for the Promotion of Science); Health Labour Sciences Research Grants (H26-Choju-Ippan-006, H27-Ninchisyou-Ippan-001 H28- Choju-Ippan-002, H28-Ninchisyou-Ippan-002, H29-Chikyukibo-Ippan-001, H30-Jyunkankinado-Ippan-004, 18H04071, 19FA1012, 19FA2001) from the Ministry of Health, Labour and Welfare, Japan; the Research and Development Grants for Longevity Science from Japan Agency for Medical Research and development (AMED) (JP17dk0110027, JP18dk0110027, JP18ls0110002, JP18le0110009, JP20dk0110034, JP20dk0110037), the Research Funding for Longevity Sciences from National Center for Geriatrics and Gerontology (24–17, 24–23, 29–42, 30–30, 30–22, 20–19); Open Innovation Platform with Enterprises, Research Institute and Academia(OPERA, JPMJOP1831)from the Japan Science and Technology (JST); a grant from the Japan Foundation For Aging And Health (J09KF00804), a grant from Innovative Research Program on Suicide Countermeasures (1–4), a grant from Sasakawa Sports Foundation, a grant from Japan Health Promotion & Fitness Foundation, a grant from Chiba Foundation for Health Promotion & Disease Prevention, the 8020 Research Grant for fiscal 2019 from the 8020 Promotion Foundation (adopted number: 19–2-06), a grant from Niimi University (1915010), and grants from Meiji Yasuda Life Foundation of Health and Welfare. The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the respective funding organizations.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by K.S. and Y.S. The first draft of the manuscript was written by K.S. and Y.S. J.A. and K.K. prepared the subsequent versions of the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Iwai-Saito, K., Shobugawa, Y., Aida, J. et al. Frailty is associated with susceptibility and severity of pneumonia in older adults (A JAGES multilevel cross-sectional study). Sci Rep 11, 7966 (2021). https://doi.org/10.1038/s41598-021-86854-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-86854-3
This article is cited by
-
Frailty, periinterventional complications and outcome in patients undergoing percutaneous mitral and tricuspid valve repair
Clinical Research in Cardiology (2024)
-
Acute organ injury and long-term sequelae of severe pneumococcal infections
Pneumonia (2023)
-
Prognostic significance of frailty in hospitalized elderly patients with community-acquired pneumonia: a retrospective cohort study
BMC Geriatrics (2023)
-
Acceleration of sarcopenia in elderly patients who develop asymptomatic pneumonia shadow within one year after surgery for early gastric cancer
BMC Surgery (2023)
-
The comparison of prognoses between total and distal gastrectomy for gastric cancer in elderly patients ≥ 80 years old
Surgery Today (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.