Abstract
Dietary reconstruction is used to make inferences about the subsistence strategies of ancient human populations, but it may also serve as a proxy to characterise their diverse cultural and technological manifestations. Dental microwear and stable isotope analyses have been shown to be successful techniques for paleodietary reconstruction of ancient populations but, despite yielding complementary dietary information, these techniques have rarely been combined within the same study. Here we present for the first time a comprehensive approach to interpreting ancient lifeways through the results of buccal and occlusal microwear, and δ13C and δ15N isotope analyses applied to the same individuals of prehistoric populations of Hungary from the Middle Neolithic to the Late Bronze Age periods. This study aimed to (a) assess if the combination of techniques yields a more precise assessment of past dietary and subsistence practices, and (b) contribute to our understanding of the dietary patterns of the prehistoric Hungarian populations. Overall, no correlations between microwear and δ13C and δ15N isotope variables were observed, except for a relationship between nitrogen and the vertical and horizontal index. However, we found that diachronic differences are influenced by the variation within the period. Particularly, we found differences in microwear and isotope variables between Middle Neolithic sites, indicating that there were different dietary practices among those populations. Additionally, microwear results suggest no changes in the abrasiveness of the diet, neither food processing methods, despite higher C4 plant resource consumption shown by carbon isotopic signal. Thus, we demonstrate that the integration of dental microwear and carbon and nitrogen stable isotope methodologies can provide complementary information for making inferences about paleodietary habits.
Similar content being viewed by others
Introduction
The transitions between the Neolithic, Copper, and Bronze Ages constitute key periods of important social changes in the configuration of European societies that included the creation of hierarchies and the establishment of agriculture1,2,3. The adoption of an agriculture and/or a pastoral lifestyle is one of the most important events in human history, resulting in significant biological, environmental, cultural and health changes3,4,5,6. By reconstructing the diet of these transitional populations, we can better understand the changes these societies experienced at different levels, including subsistence strategies, landscape use, health, and/or social stratification7,8,9,10,11,12.
In the case of the Great Hungarian Plain (GHP), a series of changes in settlement patterns and subsistence strategies occurred from the Neolithic through the Copper Age and into the Bronze Age13,14 (Table 1). During the transition from the Neolithic to the Copper Age, people dispersed from large settlements to smaller nuclear villages13,15. There was an increased focus on animal husbandry and less reliance on agricultural products, which included an increase in the use of secondary products such as milk and wool16. This shift was also accompanied by a diversification of material culture17,18. Afterwards, over the course of the Bronze Age, the intensification of agriculture and building of extensive trade networks took place due to the arrival of bronze metallurgy from the northern Pontic and Balkan Peninsulas19,20. In addition, there was an increase in millet cultivation in the Middle and Late Bronze Age21,22. During the last phase of the Bronze Age, great economic changes occurred, associated with cultural connections to Central Europe23,24,25.
Dietary patterns and subsistence practices of prehistoric populations have long been inferred from cultural and archaeological remains33,34,35, but these studies are now most often being complemented by a variety of bioarchaeological and molecular approaches7,8,36,37,38,39,40,41. Dental microwear and stable isotope analyses have been largely used to infer dietary practices in Holocene populations36,37,42,43,44,45,46,47. Dental microwear studies on buccal and occlusal surfaces of teeth provide insight into the physical properties of the food ingested by humans and other animals by yielding information about the abrasiveness of the food coupled with how these foods were processed prior to consumption48,49,50,51,52,53. During chewing, pits of different sizes and striations of different lengths and orientations are formed across enamel surfaces caused by particles harder than enamel, such as plant phytoliths, grit, or quartz dust 43,50,54,55,56,57. As such, distinct dietary habits and food processing methods result in different microwear patterns, allowing for the distinction, for example, between individuals who ingested foodstuffs obtained through different subsistence practices, such as foraging and agriculture, or between farming and pastoralism36,37,58,59.
Carbon and nitrogen stable isotope analyses, in turn, are based on the principle that the biochemical composition of the food consumed by animals and humans is preserved in their body tissues60,61,62. The isotopic signature in archaeological human tissues differs from resources consumed and consumers in a predictable isotopic fractionation, with a generally accepted stepwise increase of 0–2‰ for carbon63 and 3–5‰ for nitrogen isotopic signatures64,65,66. Carbon (δ13C) and nitrogen (δ15N) isotope ratios measured from bone collagen of archaeological samples are indicative of the protein consumed by the individual about 10–15 years prior to death67. In brief, δ13C is primarily used to identify the consumption of plants that use different metabolic pathways for fixing carbon during photosynthesis and the animals raised on them. These δ13C differences can then be used to identify the consumption of C3 versus C4 terrestrial resources in past diets68,69,70. Values of carbon ratios may also inform about the consumption of marine foods71,72,73,74 and freshwater resources75,76,77.
In addition to carbon isotope signals, values of δ15N in bone collagen are primarily used to analyse the relative presence of plant versus animal protein in the diet63,78. It is based on the observed increase of δ15N in tissues with the increase of trophic level63,79. Nitrogen isotope signals are also used to distinguish between the consumption of terrestrial foods and marine and freshwater resources64,75. Other environmental factors can also influence δ15N in tissues, such as the consumption of fertilized crops80,81, the health of the individual82, nursing practices83, and/or other environmental factors84,85,86.
Both approaches offer different insights into dietary reconstruction, and although they are complementary, their combined use for paleodietary reconstruction is rare46,87,88,89,90. Still, some research has been done in this line46,89,90, showing interesting results of the combination of buccal dental microwear and stable isotopes and enhancing the importance of integrating both methods. Within dental microwear, analyses combining buccal and occlusal surfaces are also becoming more popular91,92 due to differences in indentation enamel processes, which can provide dietary information over both short and long time scales48,54,55,93,94,95,96. The central aim of this study is to assess whether the combined use of dental microwear and isotope data provide a better understanding of the dietary patterns and the types of food consumed by past Hungarian populations. Remarkably, this study takes a step further, not only by including information from the occlusal surface too, but also and unlike previous studies88,89, by employing the same individuals for all the data obtained. The integration of the data obtained from these three proxies contributes for a dietary approximation with a more fine-grained approach.
Here, we contribute to a better understanding of the dietary practices of people in the GHP from the Middle Neolithic to the Late Bronze Age by studying their diets at different levels: (a) at the individual level, by comparing both techniques in the same individual; (b) at the diachronic level, by observing potential differences between chronologies; and (c) at the regional level, by comparing contemporaneous sites.
Materials
Archaeological human skeletal samples of both sexes from a wide range of ages at death, spanning from the Middle Neolithic to the Late Bronze Age, along with faunal material, were sampled from 17 sites across the GHP (Fig. 1; see details in “Supplementary Information Archaeological sites descriptions” and Supplementary Table S9). Postcranial elements, petrous bones, and dental remains were collected for each individual, when possible, from the osteological collection of the Herman Ottó Museum (Miskolc, Hungary). Sex and age-at-death were estimated based on established bioanthropological methods (described in “Supplementary Information Methods”). Individuals were also assigned sex using ancient DNA (aDNA) methods. In the case of divergence, aDNA sex results were favoured. For those individuals whose sex could not be identified using aDNA, osteological information was used (see more details in “Supplementary Information Methods”).
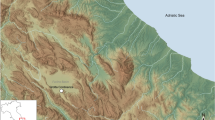
Hungarian map showing the location of sites analysed in the study. QGis free software (version 3.10), (https://www.qgis.org/es/site/) was used to create this map, with the coordinate system ETR89/UTM Zone 30 N. (A) Scale 1:1.200,000; archaeological sites represented: Middle Neolithic (1. Bükkábrány-Bánya; 2. Rásonysápberencs-Szőlő alja; 3. Csincse-Gomba Barna földje (M3- site 14–16); 4. Arnót-Nagy-bugyik; 5. Arnót-Arnóti-oldal Dél); Middle Copper Age (6. Mezőkövesd-Klementina (Szentistván-Reptér); 7. Mezőkövesd-Patakra járó dűlő); Middle Bronze Age (8. Mezőzombor-Községi temető; 9. Mezőkeresztes-Csincsetanya; 10. Nagyrozvágy-Papdomb; 11. Vatta-Dobogó); Late Bronze Age (12. Felsődobsza-site 2; 13. Oszlár-Nyárfaszög (M3-site 32); 14. Mezőkeresztes-Cet halom (M3-site 10); 15. Köröm-Kápolna-domb; 16. Pácin-Alsókenderszer). (B) Scale 1:10,000; Bükkábrány-Bánya lignite mine area with archaeological sites (BB) represented.
As in other dental microwear studies92,97,98, there was a great amount of teeth in which dental microwear could not be observed for different reasons (e.g. enamel preservation). This fact made that small sample size be a typical unavoidable reality. From a total number of 65 individuals, buccal and occlusal well preserved microwear were obtained in 49 human posterior teeth from different individuals of both sexes and various ages. Teeth present different post-depositional modification, from no detectable to severe modification. Although, experimental studies have shown that post-mortem damage could be discerned from ante-mortem microwear99,100, teeth with severe evidence of post-mortem damage were excluded from the analysis. In the case of occlusal surface, enamel wear related with abrasive diets or the effect of age, affect directly to this surface and obscure dental microwear as wear progresses93. Lower molars were preferred as sample size was higher. Nevertheless, when dental microwear was not possible to be observed in the lower molars, upper molars from the same individual were chosen instead. Carbon and nitrogen stable isotope analysis was carried out on collagen from bone samples of 89 human individuals from both sexes and various ages, along with 29 faunal bone samples (Table 2).
Results
We obtained buccal data from 38 molars (from a total number of 65 molars) from different adult individuals, while 34 molars from different adult individuals were obtained from occlusal microwear (Table 2 and Supplementary Tables S1 and S2). Bone collagen was successfully obtained for all human samples, and for 27 of 29 faunal samples analysed. Three human samples did not meet the acceptable atomic C:N quality range of 2.9–3.6101 and their isotopic data were excluded from further analyses (Table 2 and Supplementary Tables S9 and S10).
Comparison of dental microwear and stable isotope analyses
Within individuals, results from buccal and occlusal microwear analysis were compared to stable isotope data. Spearman correlation results indicated that the majority of microwear variables and stable isotope ratios were not statistically correlated (Table 3, Supplementary Figs. S1 and S2). An exception was δ15N, which was correlated with the vertical (NV/BTN) and horizontal index (NH/BTN) in buccal microwear.
Dental microwear analysis
Descriptive statistics for the buccal surface variables are summarized in Table 4 and detailed in Supplementary Table S1. Total sample consist on 65 individuals from various ages. From this sample, dental microwear was observed on 45 individuals (38 adults and 7 infants). When comparing samples between periods (N = 38), there were differences between the number of striations (BTN; Kruskal–Wallis: X2 = 13.81, p = 0.007) and their length (XT; Kruskal–Wallis: X2 = 14.82, p = 0.005) (Fig. 2, Supplementary Table S3).
Boxplot showing (A) the total number and (B) length of buccal striations, (C) the vertical and (D) horizontal indexes by period. Samples only include individuals ≥ 8 years old (N = 38; see text for explanation). Red triangles show the means, middle horizontal lines represent the medians. Middle Neolithic (MN, n = 16); Middle Copper Age (MCA, n = 11); Late Copper Age (LCA, n = 3); Middle Bronze Age (MBA, n = 6); Late Bronze Age (LBA, n = 2).
When comparing buccal microwear across sites within each period, only Middle Neolithic samples (N = 14) could be analysed, as only those sites with n ≥ 3 (Bükkábrány-Bánya VII, Bükkábrany-Bánya XI/A and Bükkábrany-Bánya XII/B in Fig. 1B) were included for statistical analyses (see “Methods”). Samples from the Bükkábrány-Bánya VII site had significantly more striations than those from Bükkábrány-Bánya XI/A, (BTN; Kruskal–Wallis: X2 = 6.44, p = 0.039). We also found differences in the horizontal (NH/BTN; Kruskal–Wallis: X2 = 6.65, p = 0.036) and vertical indexes (NV/BTN; Kruskal–Wallis: X2 = 6.75, p = 0.034) between the Bükkábrány-Bánya XI/A and Bükkábrány-Bánya XII/B sites (Fig. 3, Supplementary Table S4).
Boxplot representing (A) the total number and (B) length of striations, (C) the vertical and (D) horizontal indexes for buccal microwear of Middle Neolithic sites. Samples only include individuals > 8 years old (N = 14; see text for explanation). Red triangles show the means, the horizontal lines represent the medians. Sites: Bükkábrány-Bánya VII (BB-VII, n = 4); Bükkábrány-Bánya XI/A (BB-XI/A, n = 7); Bükkábrány-Bánya XII/B (BB-XII/B, n = 3).
Differences in buccal microwear between females and males were not significant in any of the comparisons (Supplementary Table S5, Supplementary Figs. S3 and S4). Nevertheless, although non-significant, we found trends in the length of striations (XT) and the horizontal index (NH/BTN) during the Middle Copper Age (Supplementary Fig. S4).
The descriptive data for occlusal surfaces is available in Supplementary Table S2. As in buccal surface, 65 teeth were also analyzed. However, just in 34 adult individuals dental microwear could be observed. In contrast to buccal microwear, we found no significant differences in occlusal dental microwear by period, site, or sex (Supplementary Tables S6–S8, Supplementary Figs. S5–S8).
Stable isotopes analysis
The carbon and nitrogen stable isotope results of faunal and human samples are summarised in Tables 5 and 6 and are detailed in Supplementary Tables S9 and S10.
The local terrestrial fauna analysed (N = 26; Fig. S9) had a range of δ13C of − 22.6 to − 17.9‰ [mean = − 20.5‰ ± 1.0‰ (1σ)], and a range of δ15N of 5.5–11.0‰ (mean = 7.9‰ ± 1.4‰ (1σ)), similar to those previously reported in the area21,102,103. Significant δ15N values are found among domesticates (Kruskal–Wallis: X2 = 6.21, p = 0.044), and particularly pigs show higher enriched δ15N values than sheep/goats (Mann–Whitney: W = 32.50, p = 0.024). High δ15N values are also seen in the GHP site of Polgár-Ferenci-hát104, suggesting they might eat a fair amount of meat and most likely being fed human food scraps.
The range of δ13C values for the human sample, including adults and infants (N = 89; Fig. 4), was − 21.2 to − 14.8‰ [mean = − 19.8‰ ± 1.1‰ (1σ)], and the range of δ15N was 6.9–13.2‰ [mean = 10.8‰ ± 0.9‰ (1σ)]. The human sample HUNG896 had an unusually low δ15N value (Supplementary Table S10), which indicates low consumption of animal protein and/or exclusively plant protein source105, or reflects a positive nitrogen physiological balance (more protein is used for tissue formation and less nitrogen is excreted), as seen in different physiological conditions (e.g. pregnancy, liver disease or growth spurts) in soft tissues (i.e. hair)82. Although it still remains uncertain the detection of physiological stress in archaeological skeletal remains82, presumably this individual might have suffer some kind of physiological stress occurred during a long term (as it is reflected in bone collagen), or at least episodes of nutritionally deficient diets during early years as suggested by the presence of linear enamel hypoplasia in the anterior teeth106,107. That is why we decided to remove this individual from statistical analyses. Some infant individuals showed high δ15N values (HUNG892 and HUNG967), most probably due to breastfeeding effect83,108. Therefore, Infants I (1–6 years109) were also excluded from statistical analyses (see “Methods”).
Average values of stable carbon and nitrogen isotope ratios of human and faunal bone collagen from sites analysed in this study. Human samples include both adults and young individuals (N = 89). Human isotopic values from Middle and Late Bronze Age sites were previously published in22. Domestic fauna includes ovicaprids, cattle and pigs. Errors bars correspond to the standard deviation (1σ).
Ideally, humans’ isotope ratios should be compared relative with other fauna from the same site and specific period of time. However, the faunal sample size available was relatively small to be compared separately for each time period. We decided to group all faunal samples since apparently they do not differ between periods (Supplementary Fig. S9) and the isotopic range found for each of the species are similar to those found in other GHP sites from different periods (Table 5)103,104. With the data employed here, most of the human’s samples potentially fall within the assumed isotopic offset range estimated from the domesticated species used here (ovicaprids, cattle, and pigs). When comparing human samples between different periods (N = 74), significant differences appeared between the isotope ratios (δ13C Kruskal–Wallis: X2 = 23.54, p < 0.000; δ15N Kruskal–Wallis: X2 = 13.37, p = 0.009). Individuals from the Middle Neolithic to the Late Copper Age had similar δ13C values consistent with a diet based on terrestrial C3 resources (Fig. 5). In contrast, individuals from the Late Bronze Age had significantly higher δ13C values (p < 0.05) (Supplementary Table S11) than those in preceding periods, in accordance with previous studies21,22. Nitrogen isotope ratios presented a wide range of values, suggesting a varying amount of animal protein intake (Fig. 5). Late Copper Age δ15N values were significantly higher compared to the rest of the time periods, and the Middle Copper Age samples presented significantly lower δ15N values than their Middle Neolithic predecessors (Supplementary Table S12).
Boxplot showing δ13C and δ15N values for human samples by period (N = 74) (A, B) and by Middle Neolithic sites (N = 26) (C, D). Samples only include individuals ≥ 6 years old (see text for explanation). Red triangles show the means, middle horizontal lines represent the medians. Human isotopic values from Middle and Late Bronze Age sites were previously published in15. Middle Neolithic (MN, n = 33); Middle Copper Age (MCA, n = 17); Late Copper Age (LCA, n = 3); Middle Bronze Age (MBA, n = 15); Late Bronze Age (LBA, n = 6). Sites: Bükkábrány-Bánya VII (BB-VII, n = 7); Bükkábrány-Bánya XI/A (BB-XI/A, n = 10); Bükkábrány-Bánya XII/B (BB-XII/B, n = 9).
When comparing isotope ratios by site for each period, only those sites with n ≥ 3 were included for statistical analyses. Late Neolithic, Late Copper Age and Late Bronze Age periods were not used in this analysis as they were represented by either a single individual per site or one site per period (Table 6). We found significant statistical differences in δ15N values between Middle Neolithic sites (Bükkábrány-Bánya VII, Bükkábrany-Bánya XI/A and Bükkábrany-Bánya XII/B in Fig. 1B); Kruskal–Wallis: X2 = 8.40, p = 0.014), but no differences in δ13C values (Kruskal–Wallis: X2 = 4.06, p = 0.125). Differences were significant between Bükkábrány-Bánya XII/B and the other sites (Fig. 5, Supplementary Table S13). However, we found no significant statistical differences among Middle Copper Age sites (Bükkábrány-Bánya XI/B and 7-Mezőkövesd-Patakra járó dűlő in Fig. 1A; δ13C Mann–Whitney: W = 17.50, p = 0.599; δ15N Mann–Whitney: W = 26.50, p = 0.596) (Supplementary Fig. S10). Middle Bronze Age sites (8-Mezőzombor-Községi temető and 11-Vatta-Dobogó in Fig. 1A) only presented significant differences for δ15N values (δ13C Mann–Whitney: W = 8.00, p < 0.090; δ15N Mann–Whitney: W = 37.00, p = 0.015). Notably, markedly higher δ13C values from the Vatta-Dobogó site indicate a signature indicative of C4 plant consumption (Supplementary Fig. S11), suggesting a differential intensification of crops during that time period.
Differences between females and males (N = 49) were not statistically significant for either δ13C or δ15N values (δ13C Mann–Whitney: W = 284.5, p = 0.808; δ15N Mann–Whitney: W = 218.5, p = 0.116). These were not statistically different when comparing females and males by period (Supplementary Table S14, Supplementary Fig. S12).
Discussion
In Middle Neolithic to Late Bronze Age populations that lived in the GHP, dental microwear and stable isotope analyses have been shown to provide complementary information rather than correlated variables. Microwear data from both occlusal and buccal surfaces show the abrasiveness of food, which might be related to cultural and technological changes such as food processing or dietary preferences42,44,46,53,96,110. By contrast, isotope data indicate the types of resources consumed by individuals63,78. Each approach describes dietary patterns at different levels. Therefore, they can be combined in paleodietary research to explore dietary preferences in past populations at short (occlusal microwear) and long (buccal microwear and stable isotopes combined) timescales, within individuals and across groups46,89,90.
There were significant positive and negative correlations between δ15N and the buccal variables of vertical (NV/BTN) and horizontal (NH/BTN) index, respectively. This means that individuals with high δ15N values (presumably with high meat consumption) are characterized as having a relatively more vertical and less horizontals striations. These results are in accordance with studies suggesting meat-eaters usually present high frequencies of vertical striations on the enamel surface due to the movements of the mandible while chewing meat49,111. In contrast, chewing hard foods, such as cereals, requires more horizontal movements49,111, which increases the horizontal index.
Generally, we did not find significant differences between periods, sites or sex in occlusal dental microwear. The lack of differences in the occlusal surfaces might be due to the so-called “last supper effect”, which consists of a fast renewal of occlusal enamel, thus recording the diet of the most recent days or weeks48,93,112. This contrast with a slower turnover of the buccal surface, which stabilizes at maturity50,54, and provides information of the diet over time. Nevertheless, we found interesting trends among the samples on the occlusal surface, which we discuss below.
We observed significant diachronic differences between the Middle Neolithic and the rest of the periods in the buccal microwear analysis, with the exception of Late Copper Age individuals. The low number of striations (BTN) observed from the Middle Neolithic might indicate that foods were highly processed, and thus less abrasive, and/or that a high proportion of consumed foods were soft, such as meat, which left little wear on the enamel surface44,49,59,96,110,111. The high nitrogen isotopic values [mean = 11.0 ± 0.8‰ (1σ)] were similar to those found at other Middle Neolithic sites21,104. Freshwater resources consumption (e.g. fish) can potentially explain high δ15N values, due to longer and complex foodwebs77,113, and to its proximity to freshwater resources access for some of the sites. However, although not completely discarded, the consumption of freshwater fish would not necessary lead to significant nitrogen isotope enrichment. Even if the freshwater fish represented 20% of human’s diet, the isotopic effect is quite small (+ 0.7‰)66. Additionally, freshwater fish consumption would have resulted in a more 13C-depleted values, as documented in other Mesolithic and Early Neolithic individuals from the same geographical area21,114. The carbon isotopic signal from bone collagen of Middle Neolithic sites are indicative of a C3 terrestrial protein source.
Assuming a range for trophic level enrichment generally accepted in the literature and observed in the archaeological record of the GHP102,103 (ca 0–1‰ for δ13C and ca 2–3‰ for δ15N), high δ15N could be explained by high reliance on meat consumption, particularly pig meat with significant high enriched 15N ratios among domesticates. However that would have required that 100% of the protein in diet had contributed essentially from terrestrial mammalian fauna66, ignoring that other dietary macronutrients, such as carbohydrates and lipids, are required to meet body’s energy requirements115,116. Another possibility is the ingestion of young animals meat, which shows higher δ15N due to, most probably, nursing signal117. Different patterns on bone assemblages were found on Middle Neolithic sites of the northern part of the GHP. Findings in Nagykálló-Harangod yielded several young animals (mostly sheep)118, while in Kompolt-Kíser high amounts of elderly individuals were found, probably due to their complex exploitation (skin meet, wool, milk and/or meat)119. Unfortunately, the zooarchaeological small sample size makes difficult assess the extent to which livestock exploitation prevailed in the sites presented in this study. High δ15N values may be the result of a combined effect of fertilized crops and meat consumption, as suggested elsewhere21,80,103. Long-term experimental studies have shown an increase in δ15N values in modern manured crops80,120. This has led some researchers to suggest that there has been an overestimation of animal protein intake in Neolithic paleodietary reconstructions121. Thus, these researchers propose that the combined effects of fertilized crops and animal protein consumption in Early Neolithic European farmers should result in bone collagen δ15N values of approximately 9–11‰80,121. For Middle Neolithic samples represented here, manured crops consumption, together with animal protein intake, in our opinion, is the hypothesis more feasible to us to explain high δ15N values.
However, we found that there are differences in buccal microwear and isotope ratios among Middle Neolithic sites. Particularly, samples from the Bükkábrány-Bánya VII site tended to have more total buccal striations (BTN) when compared to other Middle Neolithic sites, which were significantly different from those found at Bükkábrány-Bánya XI/A. This might indicate that the diet at Bükkábrány-Bánya VII was more abrasive due to the consumption of hard foods and/or to the inclusion of hard extrinsic particles in food50,53,55. Bükkábrány-Bánya XII/B samples instead presented the lowest BTN, indicating that the foods in their diets were less abrasive. Although not statistically significant, we observed the same trend for the total number of occlusal striations in both Middle Neolithic sites. This is also in accordance with δ15N values, where in generally high isotopic values were observed for all Middle Neolithic sites; in particular, we further found significantly more consumption of meat, a minimally abrasive resource, by the individuals at Bükkábrány-Bánya XII/B. Although we found significant differences between Bükkábrány-Bánya XI/A and Bükkábrány-Bánya XII/B sites in the horizontal and vertical indexes, the values of both indexes within Bükkábrány-Bánya XII/B were similar (around 0.2%), indicating they ate a mixed diet. Differences between the Middle Neolithic sites might have been due to cultural differences, particularly the type of food intake and/or how food was processed (e.g., with millstones) or cooked (e.g., in pottery vessels), as has been observed in other prehistoric populations37,42,44,122. It is important to keep in mind that Bükkábrány-Bánya XI/A and Bükkábrány-Bánya XII/B are closer to each other than either of them is to Bükkábrány-Bánya VII123 (Fig. 1B), which may have affected cultural and economic patterns, as well as connections across the sites. With the information recovered from the distribution of grave goods, dietary pattern differences between Bükkábrány-Bánya sites cannot be attributed to differences in terms of social status. Further studies are needed to assess whether there were other differences in these settlements.
Both buccal microwear and δ15N values suggest that there was a change in dietary preferences among Middle Copper Age individuals. Significantly higher BTN values than those found in Middle Neolithic individuals indicate they were consuming a more abrasive diet such as cereals, or to the presence of exogenous particles, such as grit, as a result of processing or cooking methods44,50,57,58. In keeping with this finding, lower δ15N values also indicate lower animal protein intake, in accordance with a lower reliance on softer resources, and/or on manured crops. Isotopic faunal offsets suggest that potentially they could have relied on cattle, pig and sheep/goats as an animal protein source, although only ovicaprid remains were recovered from this period in this study. In contrast, Late Copper Age individuals presented significant δ15N-rich nitrates compared to other periods, suggesting a higher meat intake (potentially from all domesticated fauna represented in this study), and/or consumption of fertilized crops or animals that consumed them80,103. Late Copper Age individuals had lower BTN values, which is related to less abrasive foods, such as meat110. Additionally, despite not being significant, there was a tendency for the values of the vertical indexes on the buccal surfaces to be high, which also might indicate that there was more consumption of meat49. The nitrogen isotope signal in these Late Copper Age individuals was noticeably higher (mean = 12.0 ± 0.3‰ (1σ)) than those from other Late Copper Age sites21. These results are in accordance with findings from other Late Copper Age sites that suggest an increase in animal product use during this period16, but not from other Middle Copper Age sites103. As Giblin103 argued for Early Copper Age site differences, this could be due to regional site differences in meat consumption and nitrogen enrichment, rather than differences in subsistence practices. More Copper Age sites, particularly from the Middle and Late Copper Age, need to be analysed to better understand if the increase in animal products was a general trend or instead a local or regional process.
The few Middle and Late Bronze Age individuals analysed for buccal microwear presented high mean values of BTN and XT variables, which indicates the consumption of an abrasive diet50,55 and/or less consumption of processed food43. Millet consumption starts to be noticeable in bone collagen as δ13C values suggest22, although it probably would have been identified in earlier and more individuals if bone or enamel apatite were also tested (see60). Higher consumption of this C4 plant resource was not probably the cause of the abrasiveness increase, since no significant positive correlations were found for BTN and XT with δ13C values. The nitrogen isotope signal, although high (MBA mean: 10.8 ± 0.9‰ (1σ); LBA mean: 10.9 ± 1.1‰ (1σ)), suggests a decreased reliance on animal products (potentially from the livestock represented here), and/or manured crops as compared to Late Copper Age individuals22.
There were no significant sex differences among any of the periods analyzed. Despite a limited sample size, there was an overarching trend in XT and NH/TN in the Middle Copper Age individuals for females and males: females presented shorter, more horizontally oriented buccal striations than males, which is consistent with higher cereal intake. Males’ higher δ15N values suggest they may have consumed more animal protein than females. This trend is supported by other studies in Holocene populations45,124. This issue needs more careful research, and additional studies using dental microwear and stable isotopes should be undertaken to explore potential sex-related differences and their dietary manifestations.
Conclusion
As demonstrated here, the integration of dental microwear and stable isotope approaches employed within individuals can provide a more complete overview of the dietary preferences of past human populations. Our findings suggest these two proxies are complementary, confirming their significance in paleodietary research, and we encourage future studies its combined use.
In general, the level of abrasiveness in diet, revealed by buccal dental microwear variables, is correlated with the isotopic signal at the individual/local level when interpreting δ15N results for meat consumption. In addition, the results obtained here show that changes in C4 plant consumption in the prehistoric Hungarian populations did not necessarily entail changes in diet abrasiveness, either in the C4 resource itself (grain) or the way it is processed (extrinsic particles). It is the case of Middle Bronze Age sites where higher reliance on millet (C4) consumption did not imply differences in the abrasiveness of their diet.
This research demonstrates the importance of studying the variation between different sites from the same time period. We have shown the characteristics of each Middle Neolithic site, and, at the individual level, confirmed that both dietary proxies, dental microwear and stable isotopes, point toward the same interpretation. We also found that previous interpretations regarding an increase in the consumption of animal products by the Middle Copper Age could not be confirmed in the Middle Copper Age sites analysed here.
We found no significant differences in dietary composition between females and males for each period. However, results for Middle Copper Age females and males suggest a differential trend in proportions of cereal and meat consumed. Further research involving bone apatite and/or tooth enamel, as well as the inclusion of more samples would contribute significantly to better understand these results.
Methods
Dental microwear analysis
Prior to microscopic analysis, molars were washed with cotton swabs soaked in 96% alcohol, then left to air dry completely125. Polyvinylsiloxane dental impression material (Coltene President Ligh Body©) was used to mold the tooth crowns. High resolution casts were made using EPO-TEK 301. The replicas were sputter coated with gold, and aluminum tape was attached from the stub to the molar to improve the conductivity of electrons126. We analysed the buccal and occlusal surfaces of the molars under an FEI Quanta 600 Environmental Scanning Electron Microscope (ESEM) held in the Scientific and Technical Resources Service of the University Rovira i Virgili (Tarragona, Spain), using previously-published parameters43.
Analysis of buccal microwear
Micrographs (1024 × 832 pixels) were taken with ESEM at 100× of the middle third of the surface. The images were enhanced to increase the contrast and facilitate the observation of striations125 with the free software GIMP (version 2.10.22), (https://gimp.es/). Then, digital images were cropped to cover an area of 0.56 mm2 following125. Afterwards, the total number of striations in the buccal surface (BTN), the average length (XT, in µm), and their orientation (0°–180°, divided into 45° intervals according to their orientation: horizontal (H) (0°–22.5°; 157.5°–180°), vertical (V) (67.5°–112.5°), mesiodistal (MD) (Lower Left Molar/Upper Right Molar [LL/UR]: 112.5°–157.5°; Upper Left Molar/Lower Right Molar [UL/LR]: 22.5°–67.5°); distomesial (DM) (Lower Left Molar/Upper Right Molar [LL/UR]: 22.5°–67.5°; Upper Left Molar/Lower Right Molar [UL/LR]: 112.5°–157.5°)50 were computed using open-source image processing software, Image J127 (version 1.52p), (https://imagej.nih.gov/ij/index.html). Two orientation indexes that track broad differences in dietary patterns across groups49,50 were calculated: total horizontal/total number (NH/BTN) and total vertical/total number (NV/BTN).
Analysis of occlusal microwear
Micrographs (1024 × 832 pixels) were taken with ESEM at 500× of facet 9, which is located on the distobuccal cusp on the first and second molars128,129. Facet 9 is a crushing and grinding surface, which experiences both compression and shearing during chewing130,131. The images were also enhanced, and the contrast modified using GIMP. Each image was cropped to 700 × 500 pixels, which represents approximately 0.03 mm2 of the molar’s surface112,122. On this surface, four variables were considered: the number of striations (OTN), the number of pits, their area, and the percentage of pits (Pits/(TN + Pits) × 100)42. Striations were defined and measured as in buccal surface, whereas pits are defined as features with a length to width ratio ≤ 4:193. Both features (striations and pits) were computed and recorded with ImageJ.
Observer error
There is a potentially high level of inter-observer error of the dental microwear132. For that reason, one author (RH) was responsible for the measurement of buccal and occlusal dental microwear.
Collagen extraction and isotope analyses
Carbon and nitrogen stable isotope analyses were carried out on human bones from different individuals of both sexes and various ages, along with several faunal bones (Table 2 and Supplementary Tables S9 and S10). Previously published isotopic data from Bronze Age samples22 (24 individuals) were also included in the analyses. Human postcranial samples were preferentially selected for collagen extraction except in those cases where only skull fragments were available (Supplementary Table S10).
Sample preparation and collagen extractions were performed at the University College Dublin (UCD) School of Archaeology lab facilities and UCD Conway Institute (Dublin, Ireland). Bone fragments were prepared by cutting a sample of approximately 0.2–1.0 g from the original sample material using a handheld rotary tool with a diamond-coated cutting wheel. The outer surfaces were then abraded using a diamond coated burr. Collagen extraction followed a modified Longin method133. Bone samples were first weighed and then demineralized in 0.5 M HCl at 4 °C until soft and pliable. Demineralization time varied between bone samples, from just over a week to 3 weeks. Samples were then rinsed in deionized water three times and gelatinized in pH 3 HCl solution at 70 °C for roughly 48 h. The resulting gelatine solutions were filtered using Ezee filters and then freeze-dried. Subsequently, aliquots of approximately 0.35–0.60 mg were prepared in duplicate in tin capsules for mass spectrometry analyses.
Stable isotope analyses were carried out following the routine procedures at Light Stable Isotope Mass Spectrometry Laboratory of the Department of Geological Science at University of Florida (Gainesville, USA). A Thermo Electron DeltaV Advantage isotope ratio mass spectrometer was used coupled with a ConFlo II interface linked to a Carlo Erba NA 1500 CNS Elemental Analyzer. The accuracy and precision of the measurements, based on repeated measurements of two international laboratory standards USGS40 and USGS41, was ± 0.05‰ (1σ) for δ13C and ± 0.06‰ (1σ) for δ15N. All δ13C results are expressed in standard delta notation relative to Vienna PeeDee Belemnite (V-PDB). All δ15N results are expressed in standard delta notation relative to air N2 (AIR).
Ancient DNA analyses
Ancient DNA (aDNA) analyses were performed using human petrous bones following standard procedures to determine the sex of some of the individuals (Table 2). The cochlea of one petrous bone from each individual was isolated with a sandlaster134,135. Powder was obtained from the isolated cochlea using a mixer mill (Retsch MM400), and aliquots of 50–75 mg were used for DNA extraction. The risk of modern DNA contamination was reduced by carrying out all steps in a lab dedicated to the preparation of ancient bone samples at the UCD Conway Institute of Biomolecular and Biomedical Research (University College Dublin, Ireland), as recommended by136,137,138. While handling ancient bone samples, researchers wore overall suits, double gloves, hair nets, and face masks; all surfaces were cleaned and decontaminated with DNA-ExitusPlus between sample preparation and UV irradiation was also used to ensure effective decontamination took place.
The DNA extraction, library preparation, and sequencing steps for Neolithic and Copper Age samples were performed in a physically separated aDNA lab at UCD and at the University of Vienna, following standard stringent anti-contamination protocols. A detailed description of DNA extraction, library preparation, and sequencing steps is provided in the “Supplementary Information Methods”. For Bronze Age samples, these steps were performed at Harvard University as part of a separate study.
Statistical analyses
Given the small sample size of some chronological groups, the use of non-parametric statistic was required139. Although non-parametric tests were done, with this kind of sample we also relied on overarching trend and data visualization when the groups were small140,141.
To investigate the relationship between microwear and stable isotope approaches, Spearman correlation tests were performed between microwear variables, and δ13C and δ15N, for each individual. Biplot graphs were generated for graphical visualization, including Ordinary Least Squares (OLS) regressions with a 95% confidence interval (Supplementary Figs. S1 and S2).
For general dietary trends, human samples were compared from diachronic (between time periods), synchronic (between sites), and between-sex perspectives by employing non-parametric statistical analyses. Isotopic values from faunal samples were also statistically compared. Kruskal–Wallis tests were chosen when comparing more than two groups (e.g. between periods). Mann–Whitney tests were applied for pairwise comparisons (i.e. compare two groups).
For dental microwear, only individuals 8 years and older were considered for statistical analyses, as children over 8 years have been shown to have similar dietary patterns to adults within the same time periods92. For isotope analyses, only data from adults and subadults 6 years and above of known age were considered, as they have been shown to have similar isotopic signals in the same geographical areas and time periods103. Boxplots were created to illustrate the general tendencies of the samples. Human isotope ratios were also compared with fauna isotope ratios. Faunal samples from all sites were grouped and compared with human samples for each time period separately. We assumed an isotopic fractionation range from consumed to consumers of a + 2–3‰ for δ15N and + 0–1‰ for δ13C, as it is the range that generally previous studies have found103,104. All statistical data was performed with R142, and graphs were generated using the package ggplot2143. All data generated are included in this article (and in “Supplementary Information” files).
Ethics
All necessary permits were obtained for the described study, which complied with all relevant regulations and ethical approval (Herman Ottó Múzeum).
Data availability
All data generated or analysed during this study are included in this published article (and its “Supplementary Information files”).
References
Price, T. D. & Bar-Yosef, O. Traces of inequality at the origins of agriculture in the ancient Near East. In Pathways to Power (eds Price, T. D. & Feinman, G. M.) 147–168 (Springer, 2010).
Larsen, C. S. The agricultural revolution as environmental catastrophe: Implications for health and lifestyle in the Holocene. Quat. Int. 150, 12–20 (2006).
von Cramon-Taubadel, N., Stock, J. T. & Pinhasi, R. Skull and limb morphology differentially track population history and environmental factors in the transition to agriculture in Europe. Proc. R. Soc. B Biol. Sci. 280, 20131337 (2013).
Larsen, C. S. Biological changes in human populations with agriculture. Annu. Rev. Anthropol. 24, 185–213 (1995).
Bellwood, P. S. First Farmers: The Origins of Agricultural Societies (Blackwell Publishing, 2005).
Pinhasi, R. & Stock, J. T. Human Bioarchaeology of the Transition to Agriculture (Wiley, 2011).
Buzon, M. R. & Bombak, A. Dental disease in the Nile Valley during the New Kingdom. Int. J. Osteoarchaeol. 20, 371–387 (2010).
Moghaddam, N., Müller, F., Hafner, A. & Lösch, S. Social stratigraphy in Late Iron Age Switzerland: stable carbon, nitrogen and sulphur isotope analysis of human remains from Münsingen. Archaeol. Anthropol. Sci. 8, 149–160 (2016).
Waterman, A. J., Silva, A. M. & Tykot, R. Stable isotopic indicators of diet from two late prehistoric burial sites in Portugal: an investigation of dietary evidence of social differentiation. Open J. Archaeom. 2(5258), 22–27 (2014).
Iacumin, P., Di Matteo, A., Usai, D., Salvatori, S. & Venturelli, G. Stable isotope study on ancient populations of central Sudan: Insights on their diet and environment. Am. J. Phys. Anthropol. 160, 498–518 (2016).
Lambert, P. M. Health versus fitness: Competing themes in the origins and spread of agriculture?. Curr. Anthropol. 50, 603–608 (2009).
Larsen, C. S. The bioarchaeology of health crisis: Infectious disease in the past. Annu. Rev. Anthropol. 47, 295–313 (2018).
Horváth, L. A. & Virág, Z. M. History of the Copper Age. In Hungarian Archaeology at the Turn of the Millennium (ed. Visy, Z.) 125–127 (Ministry of National Cultural Heritage, Teleki László Foundation, New York, 2003).
Parkinson, W. A. Integration, interaction, and tribal ‘cycling’: The transition to the Copper Age on the Great Hungarian Plain. In The Archaeology of Tribal Societies (ed. Parkinson, W. A.) 391–438 (International Monographs in Prehistory. Archaeology Series 15, 2002).
Chapman, J. The Neolithic in the Morava-Danube confluence area: A regional assessment of settlement pattern. In Selevac: A Neolithic Village in Yugoslavia (ed. Tringham, R. K.) 13–43 (Institute of Archaeology, University of California, 1990).
Hoekman-Sites, H. A. & Giblin, J. I. Prehistoric animal use on the Great Hungarian Plain: A synthesis of isotope and residue analyses from the Neolithic and Copper Age. J. Anthropol. Archaeol. 31, 515–527 (2012).
Whittle, A. W. R. Europe in the Neolithic: The creation of new worlds (Cambridge University Press, 1996).
Visy, Z. Hungarian Archaeology at the Turn of the Millenium (Ministry of National Cultural Heritage & Teleki László Foundation, 2003).
Sherratt, A. The emergence of élites: Earlier Bronze Age Europe, 2500–1300 BC. In Prehistoric Europe: An Illustrate History (ed. Cunliffe, B.) 244–276 (Oxford University Press, 1998).
Kulcsár, G. The Early Bronze Age. In Hungarian Archaeology in the Turn of the Millennium (ed. Visy, Z.) 141–142 (Ministry of National Cultural Heritage & Teleki László Foundation, 2003).
Gamarra, B. et al. 5000 years of dietary variations of prehistoric farmers in the Great Hungarian Plain. PLoS ONE 13, e0197214 (2018).
McCall, A. The Relationship of Stable Isotopes to Great Hungarian Plain Diet and Mobility Through the Neolithic, Copper Age, Bronze Age, and Iron Age. (PhD Dissertation, University College of Dublin, 2020).
Fischl, K. P., Kiss, V., Kulcsár, G. & Szeverényi, V. Transformations in the Carpathian Basin around 1600 BC. In 1600 BC—Cultural Change in the Shadow of the Thera-Eruption? (ed. Meller, H.) 355–372 (Landesamt für Denkmalpflege und Archäologie Sachsen-Anhalt-Landesmuseum für Vorgeschichte, 2013).
Harding, A. Reformation in Barbarian Europe. In Prehistoric Europe: An Illustrate History (ed. Cunliffe, B.) 304–335 (Oxford University Press, 1998).
Csányi, M. The Tumulus culture: Invaders from the west. In Hungarian Archaeology at the Turn of the Millennium (ed. Visy, Z.) 161–163 (Ministry of National Cultural Heritage & Teleki László Foundation, 2003).
Dani, J. A kurgánok és az építőik: Az Alföld a bronzkor hajnalán. Magyar Régészet 9, 1–19 (2020).
Horváth, T. 4000–2000 BC in Hungary: The age of transformation. Annales Universitatis Apulensis Series Historica 20, 51–112 (2016).
Kulcsár, G. Glimpses of the Third Millennium BC in the Carpathian Basin. In Moments in Time: Papers Presented to Pál Raczky on his 60th Birthday (eds Anders, A. & Kulcsár, G.) 643–659 (L’Harmattan, 2013).
Kulcsár, G. & Szeverényi, V. Transition to the Bronze Age: Issues of continuity and discontinuity in the first half of the Third Millennium BC in the Carpathian Basin. In Transitions to the Bronze Age. Interregional Interaction and Socio-Cultural Change in the Third Millennium BC Carpathian Basin and Neighbouring Regions (eds Heyd, V. et al.) 67–92 (Archaeolingua, 2013).
Heyd, V. M. Das Zeitalter der Ideologien: Migration, interaktion & expansion im prähistorischen Europa des 4. und 3. Jahrtausend v Chr. In Transitional Landscapes? The 3rd Millennium BC in Europe (eds Furholt, M. et al.) 53–85 (Habelt, 2016).
Reményi, L. A bronzkori településtörténeti változások értelmezése az új kronológiai adatok alapján (Die Interpretation von bronzezeitlichen siedlungsgeschichtlichen Änderungen mit Hilfe der neuen chronologischen Angaben). In Relationes rerum. Régészeti tanulmányok Nagy Margit tiszteletére (Relationes rerum. Archäologische Studien zu Ehren von Margit Nagy) (ed. Korom, A.) 47–56 (Archaeolingua, 2018).
Szabó, G. Problems with the periodization of the Early Bronze Age in the Carpathian Basin in light of the older and recent AMS radiocarbon data (A Kárpát-medencei kora bronzkor periodizációjának nehézségei a régi és az újabb AMS radiokarbon adatok tükrében). Archeometriai Műhely 14, 99–116 (2017).
Bogaard, A., Bending, J. & Jones, G. Archaeobotanical evidence for plant husbandry and use. In The Early Neolithic on the Great Hungarian Plain. Investigations of the Körös culture site Ecsegfalva 23, County Békés (ed. Whittle, A.) 421–446 (Archaeological Institute of the Hungarian Academy of Science, 2007).
Martín, P., Saladié, P., Nadal, J. & Vergès, J. M. Butchered and consumed: Small carnivores from the Holocene levels of El Mirador Cave (Sierra de Atapuerca, Burgos, Spain). Quat. Int. 353, 153–169 (2014).
Dunne, J. et al. Milk of ruminants in ceramic baby bottles from prehistoric child graves. Nature 574, 246–248 (2019).
Schmidt, C. W., Beach, J. J., McKinley, J. I. & Eng, J. T. Distinguishing dietary indicators of pastoralists and agriculturists via dental microwear texture analysis. Surf. Topogr. Metrol. Prop. 4, 14008 (2015).
Schmidt, C. W. et al. Dental microwear texture analysis of Homo sapiens sapiens: Foragers, farmers, and pastoralists. Am. J. Phys. Anthropol. 169, 207–226 (2019).
Eshed, V., Gopher, A. & Hershkovitz, I. Tooth wear and dental pathology at the advent of agriculture: New evidence from the levant. Am. J. Phys. Anthropol. 130, 145–159 (2006).
El Zaatari, S. & Hublin, J. Diet of Upper Paleolithic modern humans: Evidence from microwear texture analysis. Am. J. Phys. Anthropol. 153, 570–581 (2014).
Masotti, S., Varalli, A., Goude, G., Moggi-Cecchi, J. & Gualdi-Russo, E. A combined analysis of dietary habits in the Bronze Age site of Ballabio (northern Italy). Archaeol. Anthropol. Sci. 11, 1029–1047 (2019).
Beck, J., Bonilla, M.D.-Z., Bocherens, H. & Rio, P. D. Feeding a third millennium BC mega-site: Bioarchaeological analyses of palaeodiet and dental disease at Marroquíes (Jaén, Spain). J. Anthropol. Archaeol. 52, 23–43 (2018).
Molleson, T., Jones, K. & Jones, S. Dietary change and the effects of food preparation on microwear patterns in the Late Neolithic of Abu Hureyra, northern Syria. J. Hum. Evol. 24, 455–468 (1993).
Romero, A., Martínez-Ruiz, N. & De Juan, J. Non-occlusal dental microwear in a Bronze-Age human sample from east Spain. Anthropologie 42, 65–70 (2004).
Polo-Cerdá, M., Romero, A., Casabó, J. & De Juan, J. The Bronze Age burials from Cova Dels Blaus (Vall d′Uixó, Castelló, Spain): An approach to palaeodietary reconstruction through dental pathology, occlusal wear and buccal microwear patterns. HOMO J. Comp. Hum. Biol. 58, 297–307 (2007).
Fontanals-Coll, M., Subirà, M. E., Bonilla, M.D.-Z., Duboscq, S. & Gibaja, J. F. Investigating palaeodietary and social differences between two differentiated sectors of a Neolithic community, La Bòbila Madurell-Can Gambús (north-east Iberian Peninsula). J. Archaeol. Sci. Rep. 3, 160–170 (2015).
Salazar-García, D. C., Romero, A., García-Borja, P., Subirà, M. E. & Richards, M. P. A combined dietary approach using isotope and dental buccal-microwear analysis of human remains from the Neolithic, Roman and Medieval periods from the archaeological site of Tossal de les Basses (Alicante, Spain). J. Archaeol. Sci. Rep. 6, 610–619 (2016).
Irvine, B., Erdal, Y. S. & Richards, M. P. Dietary habits in the Early Bronze Age (3rd millennium BC) of Anatolia: A multi-isotopic approach. J. Archaeol. Sci. Rep. 24, 253–263 (2019).
Grine, F. E. Dental evidence for dietary differences in Australopithecus and Paranthropus: A quantitative analysis of permanent molar microwear. J. Hum. Evol. 15, 783–822 (1986).
Lalueza, C., Péréz-Perez, A. & Turbón, D. Dietary inferences through buccal microwear analysis of Middle and Upper Pleistocene human fossils. Am. J. Phys. Anthropol. 100, 367–387 (1996).
Pérez-Pérez, A., Lalueza, C. & Turbón, D. Intraindividual and intragroup variability of buccal tooth striation pattern. Am. J. Phys. Anthropol. 94, 175–187 (1994).
El-Zaatari, S. Occlusal microwear texture analysis and the diets of historical/prehistoric hunter-gatherers. Int. J. Osteoarchaeol. 20, 67–87 (2010).
Mahoney, P. Human dental microwear from Ohalo II (22,500–23,500 cal BP), southern Levant. Am. J. Phys. Anthropol. 132, 489–500 (2007).
Romero, A. & De Juan, J. Intra-and interpopulation human buccal tooth surface microwear analysis: Inferences about diet and formation processes. Anthropologie 45, 61–70 (2007).
Romero, A. & De Juan, J. SEM, teeth, and palaeoanthropology: The secret of ancient human diets. In Scanning Electron Microscopy for the Life Sciences (ed. Schatten, H.) 236–256 (Cambridge University Press, 2012).
Teaford, M. F. & Lytle, J. D. Brief communication: Diet-induced changes in rates of human tooth microwear: A case study involving stone-ground maize. Am. J. Phys. Anthropol. 100, 143–147 (1996).
Fox, C. L. & Pérez-Pérez, A. The diet of the Neanderthal Child Gibraltar 2 (Devil’s Tower) through the study of the vestibular striation pattern. J. Hum. Evol. 24, 29–41 (1993).
Gügel, I. L., Grupe, G. & Kunzelmann, K. Simulation of dental microwear: Characteristic traces by opal phytoliths give clues to ancient human dietary behavior. Am. J. Phys. Anthropol. 114, 124–138 (2001).
Romero, A., Ramírez-Rozzi, F. V., De Juan, J. & Pérez-Pérez, A. Diet-related buccal dental microwear patterns in Central African pygmy foragers and Bantu-speaking farmer and pastoralist populations. PLoS ONE 8, e84804 (2013).
Organ, J. M., Teaford, M. F. & Larsen, C. S. Dietary inferences from dental occlusal microwear at mission San Luis de Apalachee. Am. J. Phys. Anthropol. 128, 801–811 (2005).
Lee-Thorp, J. A. On isotopes and old bones. Archaeometry 50, 925–950 (2008).
Katzenberg, M. A. Stable isotope analysis: A tool for studying past diet, demography, and life history. In Biological Anthropology Human Skeleton (eds Katzenberg, M. A. & Grauer, A. L.) 413–441 (Willey-Blackwell, 2008).
Schwarcz, H. P., White, C. D. & Longstaffe, F. J. Stable and radiogenic isotopes in biological archaeology: Some applications. In Isoscapes Understanding Movement, Pattern, and Process on Earth Through Isotope Mapping (eds West, J. B. et al.) 335–356 (Springer, 2010).
Bocherens, H. & Drucker, D. Trophic level isotopic enrichment of carbon and nitrogen in bone collagen: Case studies from recent and ancient terrestrial ecosystems. Int. J. Osteoarchaeol. 13, 46–53 (2003).
Minagawa, M. & Wada, E. Stepwise enrichment of 15N along food chains: Further evidence and the relation between δ15N and animal age. Geochim. Cosmochim. Acta 48, 1135–1140 (1984).
Schoeninger, M. J. & DeNiro, M. J. Nitrogen and carbon isotopic composition of bone collagen from marine and terrestrial animals. Geochim. Cosmochim. Acta 48, 625–639 (1984).
Hedges, R. E. M. & Reynard, L. M. Nitrogen isotopes and the trophic level of humans in archaeology. J. Archaeol. Sci. 34, 1240–1251 (2007).
Hedges, R. E. M., Clement, J. G., Thomas, C. D. L. & O’Connell, T. C. Collagen turnover in the adult femoral mid-shaft: Modeled from anthropogenic radiocarbon tracer measurements. Am. J. Phys. Anthropol. 133, 808–816 (2007).
O’Leary, M. H. Carbon isotope fractionation inplants. Phytochemistry 20, 553–567 (1990).
van der Merwe, N. J. & Medina, E. The canopy effect, carbon isotope ratios and foodwebs in Amazonia. J. Archaeol. Sci. 18, 249–259 (1991).
van der Merwe, N. J. & Vogel, J. C. 13C content of human collagen as a measure of prehistoric diet in Woodland North America. Nature 276, 815–816 (1978).
Tauber, H. 13C evidence for dietary habits of prehistoric man in Denmark. Nature 292, 332–333 (1981).
Chisholm, B. S., Nelson, D. E. & Schwarcz, H. P. Stable-carbon isotope ratios as a measure of marine versus terrestrial protein in ancient diets. Science (80-). 216, 1131–1132 (1982).
Richards, M. P. & Hedges, R. E. M. Stable isotope evidence for similarities in the types of marine foods used by Late Mesolithic humans at sites along the Atlantic coast of Europe. J. Archaeol. Sci. 26, 717–722 (1999).
Fischer, A. et al. Coast–inland mobility and diet in the Danish Mesolithic and Neolithic: Evidence from stable isotope values of humans and dogs. J. Archaeol. Sci. 34, 2125–2150 (2007).
Katzenberg, M. A. & Weber, A. Stable isotope ecology and palaeodiet in the Lake Baikal region of Siberia. J. Archaeol. Sci. 26, 651–659 (1999).
Dufour, E., Bocherens, H. & Mariotti, A. Palaeodietary implications of isotopic variability in Eurasian lacustrine fish. J. Archaeol. Sci. 26, 617–627 (1999).
Lillie, M., Richards, M. P. & Jacobs, K. Stable isotope analysis of 21 individuals from the Epipalaeolithic cemetery of Vasilyevka III, Dnieper Rapids region, Ukraine. J. Archaeol. Sci. 30, 743–752 (2003).
Shoeninger, M. J. & DeNiro, M. J. Nitrogen and carbon isotopic composition of bone collagen from marine and terrestrial animals. Geochim. Cosmochim. Acta 48, 625–639 (1984).
O’Connell, T. C., Kneale, C. J., Tasevska, N. & Kuhnle, G. G. C. The diet-body offset in human nitrogen isotopic values: A controlled dietary study. Am. J. Phys. Anthropol. 149, 426–434 (2012).
Bogaard, A., Heaton, T. H. E., Poulton, P. & Merbach, I. The impact of manuring on nitrogen isotope ratios in cereals: Archaeological implications for reconstruction of diet and crop management practices. J. Archaeol. Sci. 34, 335–343 (2007).
Fraser, R. et al. Manuring and stable nitrogen isotope ratios in cereals and pulses: Towards a new archaeobotanical approach to the inference of land use and dietary practices. J. Archaeol. Sci. 38, 2790–2804 (2011).
Reitsema, L. J. Beyond diet reconstruction: Stable isotope applications to human physiology, health, and nutrition. Am. J. Hum. Biol. 25, 445–456 (2013).
Fuller, B. T., Fuller, J. L., Harris, D. A. & Hedges, R. E. M. Detection of breastfeeding and weaning in modern human infants with carbon and nitrogen stable isotope ratios. Am. J. Phys. Anthropol. 129, 279–293 (2006).
Heaton, T. H. E. The 15N/14N Ratios of Plants in South Africa and Namibia: Relationship to climate and coastal/saline environments. Oecologia 74, 236–246 (1987).
Ambrose, S. H. Effects of diet, climate and physiology on nitrogen isotope abundances in terrestrial foodwebs. J. Archaeol. Sci. 18, 293–317 (1991).
Craine, J. M. et al. Global patterns of foliar nitrogen isotopes and their relationships with climate, mycorrhizal fungi, foliar nutrient concentrations, and nitrogen availability. New Phytol. 183, 980–992 (2009).
Grine, F. E., Sponheimer, M., Ungar, P. S., Lee-Thorp, J. & Teaford, M. F. Dental microwear and stable isotopes inform the paleoecology of extinct hominins. Am. J. Phys. Anthropol. 148, 285–317 (2012).
Pérez-Pérez, A., Martínez, L. M., Gómez, M., Estebaranz-Sánchez, F. & Romero, A. Correlations among dietary proxies in African fossil hominins: Dental buccal microwear, occlusal textures and 13C stable isotope. J. Archaeol. Sci. Rep. 22, 384–391 (2018).
García-González, R. et al. Dietary inferences through dental microwear and isotope analyses of the Lower Magdalenian individual from El Mirón Cave (Cantabria, Spain). J. Archaeol. Sci. 60, 28–38 (2015).
Hogue, S. H. & Melsheimer, R. Integrating dental microwear and isotopic analyses to understand dietary change in east-central Mississippi. J. Archaeol. Sci. 35, 228–238 (2008).
García-González, R., Sánchez-Puente, Z., Arsuaga, J. L. & Carretero, J. M. Dietary inferences from dental microwear patterns in Chalcolithic populations from the Iberian Peninsula: The case of El Portalón de Cueva Mayor (Sierra de Atapuerca, Burgos, Spain) and El Alto de la Huesera (Álava, Spain). Archaeol. Anthropol. Sci. 11, 3811–3823 (2018).
Hernando, R. et al. Inferring childhood dietary maturation using buccal and occlusal deciduous molar microwear: A case study from the recent prehistory of the Iberian Peninsula. Archaeol. Anthropol. Sci. 12, 30 (2020).
Teaford, M. F. Dental microwear and dental function. Evol. Anthropol. Issues News Rev. 3, 17–30 (1994).
Teaford, M. F., Ungar, P. S., Taylor, A. B., Ross, C. F. & Vinyard, C. J. In vivo rates of dental microwear formation in laboratory primates fed different food items. Biosurface Biotribol. 3, 166 (2017).
Ungar, P. S. Dental evidence for the diets of Plio-Pleistocene hominins. Am. J. Phys. Anthropol. 146, 47–62 (2011).
Romero, A., Galbany, J., De Juan, J. & Pérez-Pérez, A. Brief communication: Short- and long-term in vivo human buccal-dental microwear turnover. Am. J. Phys. Anthropol. 148, 467–472 (2012).
Galbany, J. & Pérez-Pérez, A. Buccal enamel microwear variability in Cercopithecoidea primates as a reflection of dietary habits in forested and open savanna environments. Anthropologie 42, 13–20 (2004).
Correia, M. A., Foley, R. & Lahr, M. M. Applying dental microwear texture analysis to the living: Challenges and prospects. Am. J. Phys. Anthropol. https://doi.org/10.1002/ajpa.24133 (2020).
King, T., Andrews, P. & Boz, B. Effect of taphonomic processes on dental microwear. Am. J. Phys. Anthropol. 108, 359–373 (1999).
Romero, A., Martínez-Ruiz, N., Amorós, A. & De Juan, J. Microdesgaste dental in vivo: Modelo preliminar para interpretar su formación y variabilidad en grupos humanos antiguos. Rev. Española Antropol. Física 24, 5–18 (2004).
DeNiro, M. J. Postmortem preservation and alteration of in vivo bone collagen isotope ratios in relation to palaeodietary reconstruction. Nature 317, 806–809 (1985).
Bickle, P. & Whittle, A. The First Farmers of Central Europe: Diversity in LBK Lifeways (Oxbow Books, 2013).
Giblin, J. Isotope analysis on the Great Hungarian Plain: An exploration of mobility and subsistence strategies from the Neolithic to the Copper Age (PhD Dissertation). (The Ohio State University, 2011).
Whittle, A. Hungary. In The First Farmers of Central Europe. Diversity in LBK Lifeways (eds Bickle, P. & Whittle, A.) 49–97 (Oxbow Books, 2013).
Petzke, K. J., Boeing, H. & Metges, C. C. Choice of dietary protein of vegetarians and omnivores is reflected in their hair protein 13C and 15N abundance. Rapid Commun. Mass Spectrom. An Int. J. Devoted to Rapid Dissem. Up‐to‐the‐Minute Res. Mass Spectrom. 19, 1392–1400 (2005).
Temple, D. H. & Goodman, A. H. Bioarcheology has a “health” problem: Conceptualizing “stress” and “health” in bioarcheological research. Am. J. Phys. Anthropol. 155, 186–191 (2014).
Reid, D. J. & Dean, M. C. Brief communication: The timing of linear hypoplasias on human anterior teeth. Am. J. Phys. Anthropol. 113, 135–139 (2000).
Fernández-Crespo, T., Czermak, A., Lee-Thorp, J. A. & Schulting, R. J. Infant and childhood diet at the passage tomb of Alto de la Huesera (north-central Iberia) from bone collagen and sequential dentine isotope composition. Int. J. Osteoarchaeol. 28, 542–551 (2018).
Martin, R. & Saller, K. Lehrbuch der Anthropologie, in Systematischer Darstellung (Gustav Fischer Verlag, 1957).
Hua, L., Brandt, E. T., Meullenet, J., Zhou, Z. & Ungar, P. S. An in vitro study of dental microwear formation using the BITE Master II chewing machine. Am. J. Phys. Anthropol. 158, 769–775 (2015).
Hinton, R. J. Differences in interproximal and occlusal tooth wear among prehistoric Tennessee Indians: Implications for masticatory function. Am. J. Phys. Anthropol. 57, 103–115 (1982).
Teaford, M. F. & Tylenda, C. A. A new approach to the study of tooth wear. J. Dent. Res. 70, 204–207 (1991).
Lillie, M. C. & Richards, M. Stable isotope analysis and dental evidence of diet at the mesolithic-neolithic transition in Ukraine. J. Archaeol. Sci. 27, 965–972 (2000).
Whittle, A., Bartosiewicz, L., Boric, D., Pettitt, P. & Richards, M. In the beginning: New radiocarbon dates for the Early Neolithic in northern Serbia and south-east Hungary. Antaeus 25, 63–117 (2002).
Speth, J. D. & Spielmann, K. A. Energy source, protein metabolism, and hunter-gatherer subsistence strategies. J. Anthropol. Archaeol. 2, 1–31 (1983).
Cordain, L. et al. Plant-animal subsistence ratios and macronutrient energy estimations in worldwide hunter-gatherer diets. Am. J. Clin. Nutr. 71, 682–692 (2000).
Jay, M. & Richards, M. P. Diet in the Iron Age cemetery population at Wetwang Slack, East Yorkshire, UK: Carbon and nitrogen stable isotope evidence. J. Archaeol. Sci. 33, 653–662 (2006).
Bárány, A. Animal bone find material from the Nagykálló-Harangod Middle Neolithic settlement (County Szabolcs-Szatmár-Bereg, NE-Hungary). Commun. Archaeoligicae Hungariae 2017, 39–50 (2018).
Bartosiewicz, L. A lelőhely állatcsontanyaga. Archaeozoológia—Animal bones from the site. Archaeozoology. In Kompolt-Kistér. Újkőkori, Bronzkori, Szarmata és Avar lelőhely. Leletmentő Ásatás az M3-as Autópálya Nyomvonalán. Neolithic, Bronze Age, Sarmatian and Avar site. Rescue Excavation at the M3 Motorway (eds Petercsák, T. & Szabó, J. J.) 279–338 (Heves Megyei Régészeti Közlemények, 1999).
Szpak, P., Longstaffe, F. J., Millaire, J.-F. & White, C. D. Stable isotope biogeochemistry of seabird guano fertilization: Results from growth chamber studies with Maize (Zea mays). PLoS ONE 7, e33741 (2012).
Bogaard, A. et al. Crop manuring and intensive land management by Europe’s first farmers. Proc. Natl. Acad. Sci. 110, 12589–12594 (2013).
Schmidt, C. W. Dental microwear evidence for a dietary shift between two nonmaize-reliant prehistoric human populations from Indiana. Am. J. Phys. Anthropol. 114, 139–145 (2001).
Kalli, A. & Tutkovics, E. K. Archaeological research on the area of the Bükkábrány lignite mine in the valley of the Csincse Stream. Hung. Archaeol. E-Journal 2017 Spring, 1–11 (2017).
Jarosová, I. Dietary inferences using buccal microwear analysis on the LBK population from Vedrovice, Czech Republic. Anthropol. 46, 175–184 (2008).
Galbany, J., Martínez, L. M. & Pérez-Pérez, A. Tooth replication techniques, SEM imaging and microwear analysis in primates: Methodological obstacles. Anthropologie 42, 5–12 (2004).
Fiorenza, L., Benazzi, S. & Kullmer, O. Do it yourself: Morphology, wear and 3D digital surface models: Materials and techniques to create high-resolution replicas of teeth. J. Anthropol. Sci. 87, 211–218 (2009).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671 (2012).
Maier, W. & Schneck, G. Functional morphology of hominoid dentitions. J. Hum. Evol. 11, 693–696 (1982).
Mahoney, P. Dental microwear from Natufian hunter-gatherers and early Neolithic farmers: Comparisons within and between samples. Am. J. Phys. Anthropol. 130, 308–319 (2006).
Kay, R. F. & Hiiemae, K. M. Jaw movement and tooth use in recent and fossil primates. Am. J. Phys. Anthropol. 40, 227–256 (1974).
Tausch, J., Kullmer, O. & Bromage, T. G. A new method for determining the 3D spatial orientation of molar microwear. Scanning 37, 446–457 (2015).
Galbany, J. et al. Error rates in buccal-dental microwear quantification using scanning electron microscopy. Scanning 27, 23–29 (2005).
Longin, R. New Method of collagen extraction for radiocarbon dating. Nature 230, 241–242 (1971).
Pinhasi, R. et al. Optimal ancient DNA yields from the inner ear part of the human petrous bone. PLoS ONE 10, e0129102 (2015).
Pinhasi, R., Fernandes, D. M., Sirak, K. & Cheronet, O. Isolating the human cochlea to generate bone powder for ancient DNA analysis. Nat. Protoc. 14, 1194–1205 (2019).
Hofreiter, M., Jaenicke, V., Serre, D., von Haeseler, A. & Pääbo, S. DNA sequences from multiple amplifications reveal artifacts induced by cytosine deamination in ancient DNA. Nucleic Acids Res. 29, 4793–4799 (2001).
Poinar, H. N. The top 10 list: Criteria of authenticity for DNA from ancient and forensic samples. Int. Congr. Ser. 1239, 575–579 (2003).
Llamas, B. et al. From the field to the laboratory: Controlling DNA contamination in human ancient DNA research in the high-throughput sequencing era. STAR Sci. Technol. Archaeol. Res. 3, 1–14 (2017).
Hernando, R. et al. Exploring the utility of optical microscopy versus scanning electron microscopy for the quantification of dental microwear. Quat. Int. 569–570, 5–14 (2020).
Smith, R. J. The continuing misuse of null hypothesis significance testing in biological anthropology. Am. J. Phys. Anthropol. 166, 236–245 (2018).
Weissgerber, T. L., Milic, N. M., Winham, S. J. & Garovic, V. D. Beyond bar and line graphs: Time for a new data presentation paradigm. PLoS Biol. 13, e1002128 (2015).
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, Vienna, 2020).
Wickham, H. ggplot2: Elegant Graphics for Data Analysis (Springer-Verlag, 2016).
Acknowledgments
The authors would like to thank Rebeca Revilla for preparing the maps in Fig. 1. We thank Carlos Tornero, for comments on the isotope results, and János Dani, for comments on the time-span on Carpathian’s Basin Prehistory. We also thank to Anna Szecsenyi-Nagy, Nadine Rohland, David Reich and his wetlab team to kindly provide information of Bronze Age individuals. Samples were collected and prepared within the Horizon 2020-Marie Slodowska Curie Actions (IF-2015-703373) project held by B.G. R.H is funded by predoctoral URV Martí-Franquès Research Grant (URV 2019PMF-PIPF-59). BG is funded by the Beatriu de Pinós Post-doctoral fellowship (2017 BP 00210) from AGAUR, Government of Catalonia. DF was supported by an Irish Research Council Post-Graduate grant GOIPG/2013/36. A.M. was supported by Goverment of Ireland Postgraduate Scholarship Programme grant GOIPG/2015/2275. T.H., K.K and T.S.Z developed their work within the Hungarian Research, Development and Innovation Office (project id. FK128013). The Institut Català de Paleoecologia Humana i Evolució Social (IPHES-CERCA) has received financial support from the Spanish Ministry of Science and Innovation through the “María de Maeztu” program for Units of Excellence (CEX2019-000945-M). This project was partially funded by AGAUR (Ref. 2017SGR1040), URV (Ref. 2019PFR-URV-91) and the MICINN/FEDER (Ref. PGC2018-093925-B-C32).
Author information
Authors and Affiliations
Contributions
R.H. and B.G. conceived and designed this research. Both authors contributed equally to this work. T.S., T.H., A.B., A.K., E.K.T., K.K., K.Kr., J.K., P.C., Á.K., A.H., M.H., K.T., R.P. provided materials, physical anthropological, archaeozoological and archaeological data and background for the research. R.H., B.G., O.C., K.S. and R.S. performed the laboratory analyses. D.F. performed the bioinformatics for aDNA data. R.H. and B.G. interpreted the results and wrote the original draft with inputs from M.L. All the authors reviewed and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hernando, R., Gamarra, B., McCall, A. et al. Integrating buccal and occlusal dental microwear with isotope analyses for a complete paleodietary reconstruction of Holocene populations from Hungary. Sci Rep 11, 7034 (2021). https://doi.org/10.1038/s41598-021-86369-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-86369-x
This article is cited by
-
The diet of settled Neolithic farmers of east-central Europe: isotopic and dental microwear evidence
Archaeological and Anthropological Sciences (2023)
-
Stable carbon and nitrogen isotopes identify nuanced dietary changes from the Bronze and Iron Ages on the Great Hungarian Plain
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.