Abstract
In early infancy, melody provides the most salient prosodic element for language acquisition and there is huge evidence for infants’ precocious aptitudes for musical and speech melody perception. Yet, a lack of knowledge remains with respect to melody patterns of infants’ vocalisations. In a search for developmental regularities of cry and non-cry vocalisations and for building blocks of prosody (intonation) over the first 6 months of life, more than 67,500 melodies (fundamental frequency contours) of 277 healthy infants from monolingual German families were quantitatively analysed. Based on objective criteria, vocalisations with well-identifiable melodies were grouped into those exhibiting a simple (single-arc) or complex (multiple-arc) melody pattern. Longitudinal analysis using fractional polynomial multi-level mixed effects logistic regression models were applied to these patterns. A significant age (but not sex) dependent developmental pattern towards more complexity was demonstrated in both vocalisation types over the observation period. The theoretical concept of melody development (MD-Model) contends that melody complexification is an important building block on the path towards language. Recognition of this developmental process will considerably improve not only our understanding of early preparatory processes for language acquisition, but most importantly also allow for the creation of clinically robust risk markers for developmental language disorders.
Similar content being viewed by others
Introduction
The ability to perceive and produce the time varying vocal fundamental frequency (fo) (i.e., melody) is an extremely important component of auditory information and an essential suprasegmental aspect of spoken language. Each language is characterized by specific musical elements in the form of prosody, that is, its intonation system and constituent rhythm1. Human adults2 and even newborns are able to distinguish different languages using prosodic cues3,4,5,6, particularly melody (intonation). Most, if not all, of the linguistic and paralinguistic functions of intonation systems seem to be shared by languages of even widely different origins, a fact which strongly points to their universal role for language acquisition1.
Infants recognize auditory information of the surrounding language on the basis of prosodic cues, mainly melody, the prototypical musical element of language7,8,9,10,11,12,13,14. They do so long before they are capable of perceiving the segmental characteristics of speech such as consonants, vowels, or syllables. Infants apply their knowledge of melody in order to segment the continuous speech stream into meaningful parts (prosodic phrasing)15,16 and experience how protowords begin to emerge from the melody17.
Melody contour is probably the most important entity to be memorized and imitated by infants13,18,19,20,21,22. A newborn’s cortical structures are active in the processing of prosodic information, particularly melody contour, as typified in the exaggerated melodic expression of infant-directed speech (IDS)23,24. IDS exemplifies how messages are conveyed to the preverbal human infant by melody variation18,20,25,26,27. According to Falk28,29, the special, melodic form of vocal communication that exists between a mother and her infant (IDS) could well have co-evolved in prehistoric time. The essential characteristic of IDS is the emotional content conveyed via melody, rather than words. The well-developed perception of melody and rhythm in the foetus30, newborn31,32,33 and young infant21,34,35,36,37 leaves no doubt that the human infant has a specific sensitivity to musical sound features36,38,39,40.
During the first two months, vocal messages are almost entirely and very effectively coded in the laryngeally produced cry melody, while supralaryngeal mechanisms are still immature41,42. In addition to crying, from about the end of the second month of life onwards, new sound properties emerge within the non-cry vocal repertoire of infants. These properties (e.g., consonant- and vowel-like elements)42,43, are the result of maturing supralaryngeal mechanisms that often change the sound to be recognized as seemingly more “speech-like”. In most of these non-cry vocalisations, melody still serves as a kind of scaffolding and forms together with supralaryngeally produced constituents the characteristic overall shape (gestalt) of the sound17,44,45.
Given the importance of melody for language acquisition, there remains a dramatic lack of knowledge with respect to the starting point of melodic variation and age-related development of melody patterns in infants’ cry and non-cry vocalisations. Commonly used models of vocal development describe the emerging speech capacity by focussing on the occurrence of segmental (syllable-like) precursors, sometimes called “protophones” after Oller46,47,48. However, the specific protophone typology of pre-speech sounds is lacking the crucial importance of prosodic development and hence, does not fully conform with or capture a young infant’s entire vocal behavior49.
Considerable importance is placed on the fo patterns of non-cry vocalisations within the field of pre-speech development and language acquisition10,42,49,50,51,52,53,54. In contrast to melody in non-cry vocalisations, cry melody is often ignored in models of vocal development towards language47,55. The first universal step towards prosody (intonation and rhythm), and hence language development for the young infant requires coordination of respiratory and laryngeal activity for the production of melody variations (phonation)56,57. There is compelling evidence for a huge melody repertoire in infants’ spontaneous natural crying, recorded in a relatively ‘relaxed context’ in the presence of the mother (i.e., when the infants were hungry or thirsty)45,58,59,60. This is completely different to the fo pattern of cries elicited by application of a painful stimulus. The medical importance of vocal fo and related parameters was often exclusively documented in the area of infant cry diagnostics by eliciting pain-induced cries61,62,63,64,65,66,67. For many years, this approach hindered the recognition of the melody repertoire typical of spontaneous natural crying, which sets human infants apart from other primates68.
In earlier work and, remarkably even more recently, infant crying has been viewed as essentially stereotypic, similar to primate calls69,70,71. This view has since been refuted35,44,45,49,72,73,74,75,76,77,78. The mitigated, melodic cries of human infants are at some level, similar to simple musical melodies (“glissandi smoothly slurred or swept over a certain frequency interval”17; p.643) and could provide raw material for prosodic constituents of later language17,37,58. The same is true for prosodic properties of non-cry vocalisations.
The Melody Development Model (MD-Model), initially proposed by Wermke and Mende45 postulated that early vocal development is reflected by a sequence from simple to complex melody contours that span across infant crying and non-cry vocalisations providing important building blocks for prosody and language development. Furthermore, this complexity pattern is assumed to be regular, uni-directional for each vocalisation type, and universal in nature (ibid.). Melodic complexity is exhibited as an increase in the number of arc-like substructures of fo contour (melody arcs) for the combinatorial assembly of multiple-arc (complex) melodies of cry and non-cry vocalisations during the first months of life17,45,72,77.
While newborn infant crying and early non-cry vocalisations (e.g., vocants as defined by Martin 198143) show a melody contour that is still rather simple i.e. single-arc-like (see Fig. 1a,b), melody becomes more complex, i.e. multiple-arc-like with increasing age (see Fig. 1c,d). By the second to third month of life and depending on age-specific factors like individual fitness79 or sex hormone concentration during mini-puberty73,80,81 or the surrounding language75,82,83, melody structure in both vocalisation types becomes more complex, i.e. multiple-arc-like with increasing age.
(a–d) Melody (fundamental frequency—fo) contour diagrams (left) exemplifying simple (a: cry, b: non-cry) and complex (c: cry, d: non-cry) patterns together with the corresponding frequency spectra (time representation above) of the sounds (up to 4 kHz). The frequency scale of the melody diagrams is logarithmic, and the diagram grid marks semitone (fo) vs. time distances. Note that spectra a. and c. also display the inspiratory noise following the vocalisation. For the original audio files of these vocalisations see Supplementary Information.
To date, there is a small number of studies that have investigated melody development in infant vocalisation and few that have focused on melody development during the first six months of life13,42,45,59. The seminal paper by Kent and Murray (1982)42 examined the non-cry vocalisations in 3, 6 and 9-month-old infants during vocal interaction and play situations. Based on visual inspection of frequency spectrograms, they analysed “simple” and “complex” fo shapes to characterize intonation patterns produced by these age groups. Only 11% of multiple-arc (“complex”) patterns were observed at the age of 3 months (ibid., Table II p. 358). By 6 months, the number of complex melody contours had increased to 22% but reduced to 10% by 9 months. This may point to a developmental course of first increasing-then-decreasing production of complex melody contours in infant non-cry vocalisation between the age of 3 and 9 months. A longitudinal study of “speech quality” (segmental sound quality: vocalic versus syllabic vocalisations) and melodic complexity (suprasegmental prosodic features) in the non-cry vocalisations of infants between 2 and 6 months was reported by Hsu et al. (2000)59. However, melody contour was coded only qualitatively based on perceptual impressions. The researchers found a curvilinear trend that seemed to parallel the pattern reported by Kent and Murray (1982)42 of increasing then decreasing production of complex melodies in non-cry vocalisations. However, the decline in melody complexity was found at an earlier age (i.e., beginning at five months of age) compared to Kent and Murray (ibid.). The most comprehensive report on melody complexity development in infant crying was provided by Wermke and Mende45. The spontaneous cries of 270 infants were analysed for melodic complexity across the first five months of life. The authors described three developmental phases of cry melody, (1) birth to 8 weeks: initialization phase (increase of complex pattern from 30 to 52%), (2) 8 to 12 weeks: stabilization phase (no further increase) and (3) 12 to 18 weeks: modification phase (further increase up to a rate of about 65% cries exhibiting a complex melody; p. 34) during the third month of life was interpreted to be due to the emerging interaction of melody and resonance frequencies (resulting from vocal tract maturation). To the best of our knowledge, there are no further developmental studies of the melody features of infant vocalisations over the first six months of life, especially those capturing both cry and non-cry vocalisations.
To establish a more comprehensive model of early vocal development and gain a better understanding of early language (prosody) acquisition, we sought to investigate whether there is a developmental pattern of the production of complex melodies across the range of vocalisations produced during the first six months of life (i.e., encompassing cry and non-cry vocalisation types). The aim of the study was to perform an objective developmental analysis of prosodic precursors in the form of melody in healthy infants between 1 and 180 days of life in their cry and non-cry vocalisations. Based on the MD-Model by Wermke and Mende45, we hypothesised that both cry and non-cry vocalisations produced by infants would show a characteristic developmental increase in complex (multiple-arc) melodies.
Methods
Study design
Longitudinal analysis of complex melody pattern development among healthy young infants, stratified by cry and non-cry vocalisations from the baby sound archive at the Center of Pre-Speech Development and Developmental Disorders (University of Wuerzburg).
Participants and datasets
Healthy, term-born (≥ 37 gestational weeks) monolingual German infants from birth to the age of 180 days of life. Exclusion criteria were any kind of hearing disorder or developmental disorders over the observation period. The available database totalled 67,629 vocalisations from 277 infants; comprising of 56,537 spontaneous cry utterances from 227 infants (115 boys; 50.7%) recorded between 1 and 180 days of life, and 11,092 non-cry vocalisations (cooing/babbling sounds) from 50 infants (24 boys; 47.1%) recorded between 60 and 180 days. Only one infant appeared in both cry and non-cry data sets (for more details see Table S1 in Supplementary information).
Procedure
The archive contains anonymized audio files (wav format) of the original recording sessions (sequences of cry and non-cry vocalisations) as well as all the individual sounds, which were previously manually segmented using a commercially available system (CSL 4500; KayPENTAX, USA). Here, we used all cry and non-cry sounds available in the archive from our participants. Original recordings were approved by the respective ethical boards (ethics committee of the Charité, Humboldt University Berlin and ethics committee of the medical faculty of the University Wuerzburg) and were carried out in accordance with relevant guidelines and regulations; informed consent signed by parents was given. Finally, all recordings were archived as anonymized data sets. Each parent had a minimum of a high school education and the monthly family income was reflective of a middle class standard of living.
Cry vocalisations (spontaneous, naturally occurring crying) were recorded under comparable conditions in a hospital (first week) and at home, respectively (e.g., before breastfeeding, relaxed, pain-free manner). Non-cry-vocalisations were recorded in infants’ homes during joyful mother–infant interactions. All vocalisations were spontaneously uttered by the infants and any elicitation of vocalising was avoided. The length of an individual recording session ranged from about 1 to 3 min (crying) and 1 to 30 min (non-cry vocalisations).
Vocalisation analysis
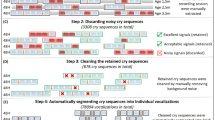
A cry or non-cry vocalisation was defined as an utterance produced on the expiratory phase of a single respiratory cycle and identified acoustically as the onset and offset of acoustic energy in the waveform. Frequency spectrograms were automatically calculated for each vocalisation using the CSL 4500 (KayPENTAX, USA). Based on visual inspections of the spectrograms, phonatory noise phenomena and phenomena like sudden fo shifts or subharmonics were identified. These well-known features of young infants’ vocalisations84,85,86 are often caused by strong nonlinearities in the restoring forces resulting from an extremely large amplitude-to-length ratio of the very young infants’ vocal folds84,85. Based on audio-visual inspections of the spectrograms (cf. previous detailed description86), vocalisations containing broad regions of phonatory noise (e.g., creaky sounds) and/or subharmonics or a highly unstable pattern caused by sudden frequency shifts (register changes) or marked vibrato-like phenomena were excluded from melody pattern analysis. The fo and its course over time (melody) cannot be reliably determined in those signals. The same was true for most vocalisations shorter than 300 ms to avoid effects from vegetative noises and sounds with background noise (e.g., parent’s voice). All vocalisations without a well-defined melody structure were identified by audio-visual analyses and categorized as exhibiting “no pattern” and subsequently excluded from the analysis. This means that “no pattern” represented a fuzzy class of vocalisations without a clearly definable melody. The remaining vocalisations were assigned as either containing a “simple” or “complex” melody pattern (see Melody Complexity Analysis). Statistical analysis revealed that both cry and non-cry vocalisation types had a decreasing occurrence of “no pattern” over age, but occurrence was lower in the non-cry vocalisations and decreased quicker than that for the cries:
For cries, the median age of cries without a well-defined melody (‘no pattern’) was 51 days, Q1 = 25 days, Q3 = 79 days, range: 1–173 days. Looking at the change over age, using a multi-level linear regression, there was a significant decrease in this pattern with increasing age (p < 0.001), given by the equation:
For non-cry vocalisations, the median age of sounds without a well-defined melody (‘no pattern’) was 113 days, Q1 = 96 days, Q3 = 140 days, range: 60–180 days. Looking at the change over age, there was a significant decrease in this pattern with increasing age (p < 0.001), given by the equation:
For determining the melody pattern, an automatic fo measurement (melody contour analysis) was required using PRAAT v. 6.0.387. PRAAT uses an autocorrelation method for fo analysis88. A post-processing verification included removal of high-frequency modulation noise and artefacts. In cases of obvious fo-tracking problems of the automatic routine, fo determination was manually repeated using PRAAT. Using specially developed lab intern software, melody diagrams were drawn and a low-pass filter (Gaussian filtering) was applied with a cut-off frequency of about 40 Hz to eliminate high-frequency modulation noise and artifacts44. This time-consuming, but robust analysis method, applied to each individual vocalisation, guaranteed that the subsequent melody pattern analysis was based on correct fo contours.
Melody complexity analysis
Melody complexity analysis was performed using specific in-lab software (CDAP, pw-project), which was implemented as a routine procedure at the Center for Pre-Speech Development and Developmental Disorders. Using the fo data calculated with PRAAT, the CDAP software allows for flexible drawings of melody diagrams and quantitative melody contour analysis. To classify melodies in simple versus complex pattern, for each sound melody the number of single arcs was identified (cf. detailed description in Supplementary information). A melody arc was defined as being longer than 150 ms and as exhibiting a frequency amplitude (FM-amplitude) of at least three (cry) or two (non-cry) semitones17. In agreement with preceding studies17,44,45,58, a complex melody structure was defined as exhibiting ≥ two arcs and/or intra-melodic breaks between arcs by glottal oscillatory pauses or marked laryngeal constrictions that generate rhythmical variations of the acoustic Gestalt. Examples for rhythmic variations of complex (multiple-arc melodies) are available in several previous publications37,45,58,89.
Based on these objective criteria, all cry melodies were analysed and subdivided into those with only a simple (single-arc) melody (Fig. 1a,b), those with a complex (multiple-arc) melody (Fig. 1c,d), while those having “no pattern” had already been excluded during pre-processing (the excluded samples, see paragraph Vocalization Analysis).
Statistical analysis
The reporting of analyses was informed by the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines; an international, collaborative initiative of epidemiologists, methodologists, statisticians, researchers and journal editors involved in the conduct and dissemination of observational studies (see: https://www.strobe-statement.org/)90. Analyses were stratified by melodies of cry and non-cry vocalisations. Summary statistics of infant’s sex, age and vocalisation signals were recorded, and the overall signal classification distributions were reported. Bubble plots (Figs. 2, 3) were then drawn, charting the proportion of vocalisations with complex melodies recorded by age (measured in days). Here, the bubble area reflected the relative numbers of vocalisations recorded at that age but ignored the nested nature of the data. The binary data (simple vs. complex melody pattern) were then analysed using fractional polynomial multi-level mixed effects logistic regression models, with unstructured covariance terms. Such models demonstrate flexibility and efficiency in modelling longitudinal developmental data, account for the hierarchical dependences associated with serial vocalisation data measurements nested within children over time, and minimise undesirable artifacts including edge effects and waves91,92. That is, results from polynomial regression models have a propensity to produce artefacts in higher order fitted curves—such as abrupt changes near the variable extremes, leading to unrealistic predictive data patterns93. Despite their flexibility and utility, fractional polynomial multi-level mixed effects logistic regression models have been rarely applied to non-Gaussian dependent variables94. Random intercept multi-level mixed effects logistic regression models were first investigated to determine the number of age related terms and their power function. Consistent with the recommendations of Royston and Sauerbrei (2008)95 degree-2 fractional polynomial powers of infant age were considered from the set (− 2; − 1; − 0.5; 0; 0.5; 1; 2; 3). The best models were then selected by minimising the deviance statistic, and the Χ2 test used to investigate deviance differences between models. These best fractional polynomial models defined the functional relationship of age (measured in days) to the binary complexity data for pursuant investigations. Once specified, both random intercept and random coefficient models were next investigated, and the Bayesian Information Criterion (BIC) used to select between these competing models96. The BIC rewards for goodness-of-fit to the data but penalises for model complexity, with the preferred model balancing these opposing demands and yielding the lowest BIC statistic. A sex difference was then tested, treating sex as a fixed effect. All statistical analyses and associated graphs were performed using Stata SE version 16.0 (StataCorp, College Station, TX, USA), and α = 0.05 defined significance.
Bubble plot of the proportion of complex melodies in cries over age together with the mean prediction line derived from the final multi-level mixed effects logistic regression model. Note: the bubble size gives the relative number. Here, the median bubble size represented n = 197, with Q1 = 88, Q3 = 331 and range: 2–1804.
Bubble plot of the proportion of complex melodies in non-cry vocalisations over age together with the mean prediction line derived from the final multi-level mixed effects logistic regression model. Note: the bubble size gives the relative number. Here, the median bubble size represented n = 102, with Q1 = 45, Q3 = 143 and range: 2–347.
Results
Regression analysis
Cry vocalisations
the mean recorded infant age of vocalisations was 56 days (SD = 38 days, range: 1, 173 days), with 21,789 (38.5%) signals defined to complex, 20,917 (37.0%) as simple, and 13,831 (24.5%) had ‘no pattern’ (and were subsequently set to missing). Overall, 226 infants had at least one recorded spontaneous cry utterances classified as simple or complex, with an average of 189 such recordings (range: 1, 910). Figure 2 presents a bubble plot of the crude proportion of complex cries (using the number of simple and complex cry signals as the denominator) by infant age (in days), without accounting for the serial nature of cries nested within children. A non-linear mean pattern is apparent within Fig. 2.
Fitting fractional polynomial multi-level mixed effects logistic regression models, the best model containing two powers of age, namely age−1 and age1 was significantly better than the best model containing one power of age (p < 0.001) or the model with a linear function of age (p < 0.001). This preferred random intercept model resulted in a BIC = 55,597. Extending the multi-level mixed effects logistic regression model to include both random intercept and random slopes for children yielded BIC = 54,815, a value superior to the random intercept only model. In this model, the fixed effects components were given by:
where Π is the predicted binary response, and value 1 indicates complex cries while 0 indicates simple cries. This fixed effects function is also drawn on Fig. 2. Each of the fixed effects age terms were statistically significant (both p < 0.001); as were the random effects terms, with variability given by: constant SD = 1.13 (95% CI: 0.98, 1.30); age−1 SD = 5.94 (95% CI: 5.07, 6.96); age1 SD = 0.013 (95% CI: 0.011, 0.015); corr(age−1, age) = 0.56 (95% CI: 0.41, 0.72); corr(constant, age−1) = − 0.55 (95% CI: − 0.69, − 0.40); and, corr(constant, age) = − 0.82 (95% CI: − 0.89, − 0.75). No sex difference was identified (p = 0.13).
Non-cry vocalisations
the mean recorded infant age of vocalisations was 125 days (SD = 32 days, range: 60, 180 days), with 4527 (40.8%) signals defined to complex, 5428 (48.9%) as simple, and 1137 (10.3%) as having ‘no pattern’ (and were subsequently set to missing). Simple or complex cooing/babbling sounds were recorded from 50 infants; with an average of 199 such sounds (range: 1, 1332 sounds). Figure 3 presents a bubble plot of the crude proportion of complex signals (using the number of simple and complex melodies as the denominator) by infant age (in days), without accounting for the serial melody vocalisations nested within children. Again, a non-linear mean pattern is suggested within this figure.
This non-linear pattern was confirmed by the application of fractional polynomial multi-level mixed effects logistic regression models. The best model contained two powers of age, namely age2 and ln(age) × age2, and was significantly better than the best models containing one power (p < 0.001) or a linear function of age (p < 0.001). This preferred random intercept model yielded a BIC = 13,235. Extending the multi-level mixed effects logistic regression model to also include random slopes for children, the algorithm failed to converge when both age2 and ln(age) × age2 terms were simultaneously considered. However, the model including a random intercept and a random slope component for age2 yielded BIC = 13,040, a value superior to the random intercept only model. In this superior model, the fixed effects components were given by:
where Π is the predicted binary response, and value 1 indicates complex melodies whereas the value 0 indicates simple melodies. These fixed effects function is superimposed on Fig. 3 as the solid line. The fixed effects for the age terms were statistically significant (both p < 0.001); as were the children’s random effects terms, with variability given by: constant SD = 1.066 (95% CI: 0.725, 1.568); age2 SD = 5.177E−05 (95% CI: 3.435E−05, 7.804E−05); and, corr(constant, age2) = − 0.813 (95% CI: − 0.999, − 0.614). Again, no differences between boys and girls was identified (p = 0.64).
Discussion
Based on longitudinal analysis of melody structure (simple vs. complex pattern), we found that human infants acquire a repertoire of complex vocal melodies over the first six months of life, with rapid gains early on. Particularly impressive was the rapid increase of the prediction curve for melody complexity development derived from the final multi-level mixed effects logistic regression model in crying (Fig. 2). This behaviour demonstrates that human infants natural spontaneous crying (in contrast to pain cries) is much more than a simple alarm signal. That is, it seems unlikely and highly ineffective for a simple alarm signal to be characterized by rapid changes in the pattern and occurrence of complex melody.
The curve predicts a cry repertoire with about 53% of the vocalisations exhibiting a complex melody at the end of the first month and a further increase thereafter. This rapid gain requires mature functioning of neuro-physiological mechanisms underlying melody production, which corresponds to the fast brain growth at this early age97,98. For example, Holland et al.97 found that brain development is most rapid during the neonatal period with 64% of whole brain growth occurring within the first 90 days” (ibid. p. 6).
The early occurrence of complex cry melodies would further suggest a ‘preparatory’ intrauterine development. The muscles of the larynx are an integral part of the respiratory system. Like other respiratory muscles, they undergo considerable use prior to birth99. From birth, newborns are capable of a highly developed laryngeal-respiratory control that serves breathing and phonation41,49,56,57. This aptitude facilitates survival, but at the same time enables the newborn to play with his laryngeal options for vocalising and hence, to quickly acquire complex melody patterns.
Intrauterine preparation also involves perceptive components. Indeed, there is evidence that late-term fetuses demonstrate the ability to discriminate their native language, to which they were exposed in utero, from an unknown language100,101. These findings suggest that fetuses encode suprasegmental characteristics of speech (melody, rhythm) rather than segmental features, which is due to segmental information being filtered out by tissue and fluid before it reaches the fetus. Prenatal sensitivity to prosodic features was demonstrated in newborns, who exhibited a preference for a low-pass filtered maternal voice and language (focused on melody) in contrast to natural voice during their first days of life5.
The non-cry vocalisations produced by the infants were found to parallel a similar developmental path observed earlier for cry vocalisations. Specifically, the model curve for the non-cry vocalisations initially also shows a low percentage of complex melodies (about 30%) and a fast increase in their share of complex melodies over the next 2.5 months up to about 50% (Fig. 3). This vocal development is consistent with the course of brain development and reported perceptive performances of infants at this age102. From both, a phylogenetic and ontogenetic perspective on spoken language evolution, it seems essential that the rapid gain in producing crying characterized by complex melodies occurs long before supralaryngeal maturation (vocal tract maturation) allows for pre-articulatory activities in sound production.
Our observation that melody development in both cry and non-cry vocalisations is characterized by an increase in complexity points to a similar strategy in the acquisition and rehearsal of prosodic building blocks as postulated by the MD-Model, introduced by Wermke and Mende45. It emphasizes spontaneous crying being as important as non-cry vocalizations (cooing, babbling) during the developmental process. In contrast to past simplistic suggestions that human infant crying is a fixed and monotonous reflex response comparable to animal vocalizations71, we see a developmental progression of vocal growth. Our study further elucidates the prominence of melody variation in cry development. The early occurrence of complex melodies also confirms a previous report suggesting that by approximately two months of age, a majority of spontaneous cry vocalisations should contain complex melodies. Otherwise, the infant may be at risk for an early language impairment79,89. This hypothesis is supported by a recent study of Francois et al. (2017)103. The authors demonstrated that neonatal brain responses for sung streams predicted expressive vocabulary at 18 months. These findings further corroborate the importance of melody production and perception for language development long before “speech-like” vocalisations emerge.
There is major agreement between scientists of several disciplines, that the first universal steps taken by an infant on his way to spoken language include melodic-rhythmic aptitudes, with respect to both perceptive and productive performances. A systematic increase in melody complexity in cry and non-cry vocalisations, as demonstrated here, provides the raw material for later language prosody. This takes place to an extent that has been often underestimated in spontaneous crying and is continued in non-cry vocalisations. Consequently, infants’ melodic sound characteristics, i.e. early building blocks of prosody, are crucial in order to characterize their path to spoken language in the first months of life.
While the model curve of complex melodies among the cry repertoire did not decline at the end of the observation period, occurrence of complex melodies in non-cry vocalisations slightly decreased from about 4.5 to 5 months. This is most likely due to the new constituents of non-cry vocalisations that emerged to interact with the overall melodic contour, namely vowel-like (vocants) and consonant-like elements (closants). During the process of spoken language acquisition, the human infant must modify his laryngeally produced melodies repeatedly and tune them to the resonance frequencies of a vocal tract that continues to grow and change42,44. The tuning in non-cry vocalisations is additionally challenged by an increasing articulatory activity required to produce syllabic combinations in babbling from about five to six months on. This new developmental period requires a temporary “regression” in melody development to establish vocal development on a higher hierarchical level17. Thereafter, the infant begins to intentionally imitate intonation patterns of the surrounding language(s) in consonant–vowel syllable sequences in babbling17,44,45. The identification of primitive precursors of later articulatory speech elements (closants, constrictions), observed in early cry and non-cry vocalisations49, show the close interaction of suprasegmental and segmental phonatory activity by the infant at an early age. A logical next step in our line of research is to examine the possible interaction between vocant and closant articulations and melodic complexity.
The study provides the first statistical model to demonstrate a systematic melody development in cry and non-cry vocalisations of infants. Our data revealed a strong developmental continuity in spontaneous crying with respect to melody complexity across the first 180 days. Additionally, there was a continuous increase in complex melodies in non-cry development with a slight reduction occurring at approximately 140 days. Recognition of this developmental process will considerably improve not only our understanding of early preparatory processes for spoken language acquisition, but most importantly also allow for the creation of clinically robust risk markers for developmental language disorders. This is the crucial prerequisite to enable us to develop innovative therapies for infants at-risk for developing language disorders. This developmental model could help to better understand why the human infant acquires so quickly and seemingly effortlessly such a complex faculty as language.
Data availability
Because the participants did not give explicit written consent that their data can be made publicly available, data will not be shared.
References
Hirst, D. & Di Cristo, A. Intonation Systems. A Survey of Twenty Languages (Cambridge University Press, Cambridge, 1998).
Vicenik, C. & Sundara, M. The role of intonation in language and dialect discrimination by adults. J. Phon. 41, 297–306. https://doi.org/10.1016/j.wocn.2013.03.003 (2013).
Mehler, J. et al. A precursor of language acquisition in young infants. Cognition 29, 143–178. https://doi.org/10.1016/0010-0277(88)90035-2 (1988).
Nazzi, T., Floccia, C. & Bertoncini, J. Discrimination of pitch contours by neonates. Infant Behav. Dev. 21, 779–784. https://doi.org/10.1016/S0163-6383(98)90044-3 (1998).
Byers-Heinlein, K., Burns, T. C. & Werker, J. F. The roots of bilingualism in newborns. Psychol. Sci. 21, 343–348. https://doi.org/10.1177/0956797609360758 (2010).
Chong, A. J., Vicenik, C. & Sundara, M. Intonation plays a role in language discrimination by infants. Infancy 23, 795–819. https://doi.org/10.1111/infa.12257 (2018).
Perszyk, D. R. & Waxman, S. R. Infants’ advances in speech perception shape their earliest links between language and cognition. Sci. Rep. 9, 3293. https://doi.org/10.1038/s41598-019-39511-9 (2019).
Bertoncini, J., Bijeljac-Babic, R., Jusczyk, P. W., Kennedy, L. J. & Mehler, J. An investigation of young infants’ perceptual representations of speech sounds. J. Exp. Psychol. Gen. 117, 21–33. https://doi.org/10.1037//0096-3445.117.1.21 (1988).
Háden, G. P., Németh, R., Török, M. & Winkler, I. Predictive processing of pitch trends in newborn infants. Brain Res. 1626, 14–20. https://doi.org/10.1016/j.brainres.2015.02.048 (2015).
Hallé, P. A., de Boysson-Bardies, B. & Vihman, M. M. Beginnings of prosodic organization: intonation and duration patterns of disyllables produced by Japanese and French infants. Lang. Speech 34, 299–318. https://doi.org/10.1177/002383099103400401 (1991).
Vihman, M. M. Phonological Development. The Origins of Language in the Child (Blackwell, Cambridge, 1996).
Cirelli, L. K., Trehub, S. E. & Trainor, L. J. Rhythm and melody as social signals for infants. Ann. N. Y. Acad. Sci. https://doi.org/10.1111/nyas.13580 (2018).
Gratier, M. & Devouche, E. Imitation and repetition of prosodic contour in vocal interaction at 3 months. Dev. Psychol. 47, 67–76. https://doi.org/10.1037/a0020722 (2011).
Mendoza, J. K. & Fausey, C. M. Everyday music in infancy. PsyArXiv https://doi.org/10.31234/osf.io/sqatb (2019).
Jusczyk, P. W. Narrowing the distance to language: One step at a time. J. Commun. Disord. 32, 207–222. https://doi.org/10.1016/s0021-9924(99)00014-3 (1999).
Frazier, L., Carlson, K. & Clifton, C. Prosodic phrasing is central to language comprehension. Trends Cogn. Sci. 10, 244–249. https://doi.org/10.1016/j.tics.2006.04.002 (2006).
Wermke, K. & Mende, W. In The Oxford Handbook of Social Neuroscience (eds Decety, J. & Cacioppo, J. T.) 624–648 (Oxford University Press, New York, 2011).
Fernald, A. Intonation and communicative intent in mothers’ speech to infants. Is the melody the message?. Child Dev. 60, 1497–1510 (1989).
Fernald, A. In The Adapted Mind (eds Barkow, J. H. et al.) 391–428 (Oxford University Press, New York, 1992).
Papoušek, M., Bornstein, M. H., Nuzzo, C., Papoušek, H. & Symmes, D. Infant responses to prototypical melodic contours in parental speech. Infant Behav. Dev. 13, 539–545. https://doi.org/10.1016/0163-6383(90)90022-Z (1990).
Trehub, S. E. Musical predispositions in infancy. Ann. N. Y. Acad. Sci. 930, 1–16. https://doi.org/10.1111/j.1749-6632.2001.tb05721.x (2001).
Dowling, W. J. & Fujitani, D. S. Contour, interval, and pitch recognition in memory for melodies. J. Acoust. Soc. Am. 49(Suppl 2), 524+. https://doi.org/10.1121/1.1912382 (1971).
Cheng, Y., Lee, S.-Y., Chen, H.-Y., Wang, P.-Y. & Decety, J. Voice and emotion processing in the human neonatal brain. J. Cogn. Neurosci. 24, 1411–1419. https://doi.org/10.1162/jocn_a_00214 (2012).
Saito, Y. et al. The function of the frontal lobe in neonates for response to a prosodic voice. Early Hum. Dev. 83, 225–230. https://doi.org/10.1016/j.earlhumdev.2006.05.017 (2007).
Tomlinson, G. A Million Years of Music. The Emergence of Human Modernity (Zone Books, New York, 2015).
Fernald, A. & Mazzie, C. Prosody and focus in speech to infants and adults. Dev. Psychol. 27, 209–221. https://doi.org/10.1037//0012-1649.27.2.209 (1991).
Fernald, A. Approval and disapproval: Infant responsiveness to vocal affect in familiar and unfamiliar languages. Child Dev. 64, 657–674 (1993).
Falk, D. Prelinguistic evolution in early hominins: Whence motherese?. Behav. Brain Sci. 27, 491–503. https://doi.org/10.1017/s0140525x04000111 (2004).
Falk, D. The, “putting the baby down” hypothesis: Bipedalism, babbling, and baby slings. Behav. Brain Sci. 27, 526–534. https://doi.org/10.1017/S0140525X0448011X (2004).
Lecanuet, J. P., Graniere-Deferre, C., Jacquet, A. Y. & DeCasper, A. J. Fetal discrimination of low-pitched musical notes. Dev. Psychobiol. 36, 29–39 (2000).
DeCasper, A. J. & Fifer, W. P. Of human bonding: Newborns prefer their mothers’ voices. Science 208, 1174–1176. https://doi.org/10.1126/science.7375928 (1980).
Winkler, I. et al. Newborn infants can organize the auditory world. Proc. Natl. Acad. Sci. U. S. A. 100, 11812–11815. https://doi.org/10.1073/pnas.2031891100 (2003).
Winkler, I., Háden, G. P., Ladinig, O., Sziller, I. & Honing, H. Newborn infants detect the beat in music. Proc. Natl. Acad. Sci. U. S. A. 106, 2468–2471. https://doi.org/10.1073/pnas.0809035106 (2009).
Chang, H. W. & Trehub, S. E. Infants’ perception of temporal grouping in auditory patterns. Child Dev. 48, 1666–1670 (1977).
Trehub, S. E. & Thorpe, L. A. Infants’ perception of rhythm: categorization of auditory sequences by temporal structure. Can. J. Psychol. 43, 217–229. https://doi.org/10.1037/h0084223 (1989).
Trehub, S. E. The developmental origins of musicality. Nat. Neurosci. 6, 669–673. https://doi.org/10.1038/nn1084 (2003).
Armbrüster, L. et al. Musical intervals in infants’ spontaneous crying over the first 4 months of life. Folia Phoniatr. Logop. 19, 1–12. https://doi.org/10.1159/000510622 (2020).
Cross, I. Communicative development: Neonate crying reflects patterns of native-language speech. Curr. Biol. 19, R1078–R1079. https://doi.org/10.1016/j.cub.2009.10.035 (2009).
Brown, S. A joint prosodic origin of language and music. Front. Psychol. 8, 1894. https://doi.org/10.3389/fpsyg.2017.01894 (2017).
Cross, I. Music, mind and evolution. Psychol. Music. 29, 95–102. https://doi.org/10.1177/0305735601291007 (2016).
Bosma, J. F., Truby, H. M. & Lind, J. Cry motions of the newborn infant. Acta Paediatr. 54, 60–92. https://doi.org/10.1111/j.1651-2227.1965.tb09309.x (1965).
Kent, R. D. & Murray, A. D. Acoustic features of infant vocalic utterances at 3, 6, and 9 months. J. Acoust. Soc. Am. 72, 353–365. https://doi.org/10.1121/1.388089 (1982).
Martin, J. A. M. In Voice, Speech, and Language in the Child. Development and Disorder (Bd. 4) (eds Arnold, G. E. et al.) (Springer, Vienna, 1981).
Wermke, K., Mende, W., Manfredi, C. & Bruscaglioni, P. Developmental aspects of infant’s cry melody and formants. Med. Eng. Phys. 24, 501–514. https://doi.org/10.1016/S1350-4533(02)00061-9 (2002).
Wermke, K. & Mende, W. In Melodies, Rhythm and Cognition in Foreign Language Learning (eds Fonseca-Mora, M. C. & Gant, M.) 24–47 (Cambridge Scholars Publishing, Newcastle upon Tyne, 2016).
Oller, D. K. In Precursors of early speech New York (eds Lindblom, B. & Zetterstrom, R.) 21–35 (Stockton Press, New York, 1986).
Oller, D. K. The Emergence of the Speech Capacity (Lawrence Erlbaum Associates, Mahwah, N.J., 2000).
Oller, D. K. et al. Infant boys are more vocal than infant girls. Curr. Biol. 30, R426–R427. https://doi.org/10.1016/j.cub.2020.03.049 (2020).
Robb, M. P. et al. Laryngeal constriction phenomena in infant vocalizations. J. Speech Lang. Hear. Res. 63, 49–58. https://doi.org/10.1044/2019_JSLHR-S-19-0205 (2020).
Wermke, K. et al. Acoustic properties of comfort sounds of 3-month-old Cameroonian (Nso) and German infants. Speech Lang. Hear. 16, 149–162. https://doi.org/10.1179/2050572813Y.0000000010 (2013).
DePaolis, R. A., Vihman, M. M. & Nakai, S. The influence of babbling patterns on the processing of speech. Infant Behav. Dev. 36, 642–649. https://doi.org/10.1016/j.infbeh.2013.06.007 (2013).
De Boysson-Bardie, B. How Language Comes to Children: From Birth to Two Years (MIT Press, Cambridge, 2001).
Stark, R. E., Rose, S. N. & Benson, P. J. Classification of infant vocalization. Br. J. Disord. Commun. 13, 41–47. https://doi.org/10.3109/13682827809011324 (1978).
Robb, M. P. & Saxman, J. H. Developmental trends in vocal fundamental frequency of young children. J. Speech Hear. Res. 28, 421–427. https://doi.org/10.1044/jshr.2803.427 (1985).
Buder, E., Warlaumont, A. & Oller, D. K. In Comprehensive Perspectives on Speech Sound Development and Disorders (eds Peter, B. & MacLeod, A. A. N.) 103–134 (Nova Publishers, New York, 2013).
Boliek, C. A., Hixon, T. J., Watson, P. J. & Morgan, W. J. Vocalization and breathing during the first year of life. J. Voice 10, 1–22. https://doi.org/10.1016/s0892-1997(96)80015-4 (1996).
Wermke, K. et al. The vocalist in the crib: The flexibility of respiratory behaviour during crying in healthy neonates. J. Voice https://doi.org/10.1016/j.jvoice.2019.07.004 (2019).
Wermke, K. & Mende, W. Musical elements in human infants’ cries: In the beginning is the melody. Musicae Scientiae 13, 151–173. https://doi.org/10.1177/1029864909013002081 (2009).
Hsu, H.-C., Fogel, A. & Cooper, R. B. Infant vocal development during the first 6 months: Speech quality and melodic complexity. Inf. Child Develop. 9, 1–16. https://doi.org/10.1002/(SICI)1522-7219(200003)9:1%3c1::AID-ICD210%3e3.0.CO;2-V (2000).
Shinya, Y., Kawai, M., Niwa, F., Imafuku, M. & Myowa, M. Fundamental frequency variation of neonatal spontaneous crying predicts language acquisition in preterm and term infants. Front. Psychol. 8, 2195. https://doi.org/10.3389/fpsyg.2017.02195 (2017).
Michelsson, K. Cry analyses of symptomless low birth weight neonates and of asphyxiated newborn infants. Acta Paediatr. Scand. Suppl. 216, 1–45 (1971).
Michelsson, K., Eklund, K., Leppänen, P. & Lyytinen, H. Cry characteristics of 172 healthy 1-to 7-day-old infants. Folia Phoniatr. Logop. 54, 190–200. https://doi.org/10.1159/000063190 (2002).
Raes, J., Michelsson, K., Dehaen, F. & Despontin, M. Cry analysis in infants with infectious and congenital disorders of the larynx. Int. J. Pediatr. Otorhinolaryngol. 4, 157–169. https://doi.org/10.1016/0165-5876(82)90091-x (1982).
LaGasse, L. L., Neal, A. R. & Lester, B. M. Assessment of infant cry: Acoustic cry analysis and parental perception. Ment. Retard. Dev. Disabil. Res. Rev. 11, 83–93. https://doi.org/10.1002/mrdd.20050 (2005).
Lester, B. M. Developmental outcome prediction from acoustic cry analysis in term and preterm infants. Pediatrics 80, 529–534 (1987).
Vohr, B. R. et al. Abnormal brain-stem function (brain-stem auditory evoked response) correlates with acoustic cry features in term infants with hyperbilirubinemia. J. Pediatr. 115, 303–308. https://doi.org/10.1016/s0022-3476(89)80090-3 (1989).
Goberman, A. M. & Robb, M. P. Acoustic examination of preterm and full-term infant cries: the long-time average spectrum. J. Speech Lang. Hear. Res. 42, 850–861. https://doi.org/10.1044/jslhr.4204.850 (1999).
Kappeler, P. M. & Silk, J. (eds) Mind the Gap (Springer, Berlin, 2010).
Lieberman, P., Harris, K. S., Wolff, P. & Russell, L. H. Newborn infant cry and nonhuman primate vocalization. J. Speech Hear. Res. 14, 718–727. https://doi.org/10.1044/jshr.1404.718 (1971).
Soltis, J. The signal functions of early infant crying. Behav. Brain Sci. 27, 443–458. https://doi.org/10.1017/S0140525X0400010X (2004).
Oller, D. K. et al. Language origins viewed in spontaneous and interactive vocal rates of human and bonobo infants. Front. Psychol. 10, 729. https://doi.org/10.3389/fpsyg.2019.00729 (2019).
Wermke, K. Habilitation. Humboldt-Universität zu Berlin https://doi.org/10.18452/13810 (2002).
Wermke, K., Hain, J., Oehler, K., Wermke, P. & Hesse, V. Sex hormone influence on human infants’ sound characteristics: melody in spontaneous crying. Biol. Lett. 10, 20140095. https://doi.org/10.1098/rsbl.2014.0095 (2014).
Mampe, B., Friederici, A. D., Christophe, A. & Wermke, K. Newborns’ cry melody is shaped by their native language. Curr. Biol. 19, 1994–1997. https://doi.org/10.1016/j.cub.2009.09.064 (2009).
Prochnow, A., Erlandsson, S., Hesse, V. & Wermke, K. Does a ‘musical’ mother tongue influence cry melodies? A comparative study of Swedish and German newborns. Musicae Scientiae https://doi.org/10.1177/1029864917733035 (2017).
Tonkova-Yampolskaya, R. V. In Studies of Child Language Development (eds Ferguson, C. A. & Slobin, D. I.) 128–138 (Holt Rinehart and Winston, New York, 1973).
Wermke, K., Mende, W., Borschberg, H. & Ruppert, R. Changes of voice parameters and melody patterns during the first year of life in human twins. J. Acoust. Soc. Am. 105, 1303–1304. https://doi.org/10.1121/1.424751 (1999).
Wermke, K. & Robb, M. P. Fundamental frequency of neonatal crying: Does body size matter?. J. Voice 24, 388–394. https://doi.org/10.1016/j.jvoice.2008.11.002 (2010).
Wermke, K., Leising, D. & Stellzig-Eisenhauer, A. Relation of melody complexity in infants’ cries to language outcome in the second year of life: A longitudinal study. Clin. Linguist. Phon. 21, 961–973. https://doi.org/10.1080/02699200701659243 (2007).
Quast, A., Hesse, V., Hain, J., Wermke, P. & Wermke, K. Baby babbling at five months linked to sex hormone levels in early infancy. Infant Behav. Dev. 44, 1–10. https://doi.org/10.1016/j.infbeh.2016.04.002 (2016).
Wermke, K., Quast, A. & Hesse, V. From melody to words: The role of sex hormones in early language development. Horm. Behav. 104, 206–215. https://doi.org/10.1016/j.yhbeh.2018.03.008 (2018).
Wermke, K. et al. Fundamental frequency variation in crying of Mandarin and German neonates. J. Voice 31(255), e25-255.e30. https://doi.org/10.1016/j.jvoice.2016.06.009 (2017).
Wermke, K. et al. Fundamental frequency variation within neonatal crying: Does ambient language matter?. Speech Lang. Hear. 19, 211–217. https://doi.org/10.1080/2050571X.2016.1187903 (2016).
Mende, W., Herzel, H. & Wermke, K. Bifurcations and chaos in newborn infant cries. Phys. Lett. A 145, 418–424. https://doi.org/10.1016/0375-9601(90)90305-8 (1990).
Titze, I. R. Principles of Voice Production (Prentice Hall, Englewood Cliffs, 1994).
Fuamenya, N. A., Robb, M. P. & Wermke, K. Noisy but effective. Crying across the first 3 months of life. J. Voice 29, 281–286. https://doi.org/10.1016/j.jvoice.2014.07.014 (2015).
Boersma, P. & Weenink, D. Praat: Doing phonetics by computer [Computer program]. Version 6.0.37; http://www.praat.org/.
Boersma, P. Accurate short-term analysis of the fundamental frequency and the harmonic-to-noise ratio of a sampled sound. Proc. Inst. Phonetic Sci. Univ. Amster. 17, 97–110 (1993).
Wermke, K. et al. Cry melody in 2-month-old infants with and without clefts. Cleft Palate Craniofac. J. 48, 321–330. https://doi.org/10.1597/09-055 (2011).
von Elm, E. et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. Ann. Intern. Med. 147, 573–577. https://doi.org/10.7326/0003-4819-147-8-200710160-00010 (2007).
Johnson, W., Balakrishna, N. & Griffiths, P. L. Modeling physical growth using mixed effects models. Am. J. Phys. Anthropol. 150, 58–67. https://doi.org/10.1002/ajpa.22128 (2013).
Tilling, K., Macdonald-Wallis, C., Lawlor, D. A., Hughes, R. A. & Howe, L. D. Modelling childhood growth using fractional polynomials and linear splines. Ann. Nutr. Metab. 65, 129–138. https://doi.org/10.1159/000362695 (2014).
Royston, P. & Altman, D. G. Approximating statistical functions by using fractional polynomial regression. J. R. Stat. Soc. Ser. D. 46, 411–422. https://doi.org/10.1111/1467-9884.00093 (1997).
Ryoo, J. H., Long, J. D., Welch, G. W., Reynolds, A. & Swearer, S. M. Fitting the fractional polynomial model to non-Gaussian longitudinal data. Front. Psychol. 8, 1431. https://doi.org/10.3389/fpsyg.2017.01431 (2017).
Royston, P. & Sauerbrei, W. Multivariable Model-Building. A Pragmatic Approach to Regression Analysis Based on Fractional Polynomials for Modelling Continuous Variables (John Wiley & Sons, Hoboken, 2008).
Schwarz, G. Estimating the dimension of a model. Ann. Stat. 6, 461–464 (1978).
Holland, D. et al. Structural growth trajectories and rates of change in the first 3 months of infant brain development. JAMA Neurol. 71, 1266–1274. https://doi.org/10.1001/jamaneurol.2014.1638 (2014).
Dubois, J. et al. The early development of brain white matter: A review of imaging studies in fetuses, newborns and infants. Neuroscience 276, 48–71. https://doi.org/10.1016/j.neuroscience.2013.12.044 (2014).
Harding, R. Function of the larynx in the fetus and newborn. Annu. Rev. Physiol. 46, 645–659. https://doi.org/10.1146/annurev.ph.46.030184.003241 (1984).
Lee, G. Y. & Kisilevsky, B. S. Fetuses respond to father’s voice but prefer mother’s voice after birth. Dev. Psychobiol. 56, 1–11. https://doi.org/10.1002/dev.21084 (2014).
Kisilevsky, B. S. et al. Fetal sensitivity to properties of maternal speech and language. Infant Behav. Dev. 32, 59–71. https://doi.org/10.1016/j.infbeh.2008.10.002 (2009).
Polka, L., Masapollo, M. & Ménard, L. Who’s talking now? Infants’ perception of vowels with infant vocal properties. Psychol. Sci. 25, 1448–1456. https://doi.org/10.1177/0956797614533571 (2014).
François, C. et al. Enhanced neonatal brain responses to sung streams predict vocabulary outcomes by age 18 months. Sci. Rep. 7, 12451. https://doi.org/10.1038/s41598-017-12798-2 (2017).
Acknowledgements
We are grateful to all parents and infants who supported our work over all the years. We also are very thankful to PhD students and research assistants who were engaged in recording and pre-processing data over many years. Particularly, we thank Peter Wermke for his hugely helpful work in handling and managing the enormous amount of involved data and information, for automatically producing all frequency spectrograms and melody contours as well as providing the labintern software for melody complexity measurement. The Open Access Publication Fund of the University of Wuerzburg supported this publication.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to this project. K.W. gathered the vocalisations in several of her previous projects. All authors jointly analysed the data, with the statistical analysis performed by P.J.S., and wrote and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wermke, K., Robb, M.P. & Schluter, P.J. Melody complexity of infants’ cry and non-cry vocalisations increases across the first six months. Sci Rep 11, 4137 (2021). https://doi.org/10.1038/s41598-021-83564-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-83564-8
This article is cited by
-
Frühe Sprachleistungen im Fragebogen ELFRA
HNO (2024)
-
Exploring individual differences in musical rhythm and grammar skills in school-aged children with typically developing language
Scientific Reports (2023)
-
Infant cries convey both stable and dynamic information about age and identity
Communications Psychology (2023)
-
A self-training automatic infant-cry detector
Neural Computing and Applications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.