Abstract
The rapid spread of the SARS-CoV-2 in the COVID-19 pandemic had raised questions on the route of transmission of this disease. Initial understanding was that transmission originated from respiratory droplets from an infected host to a susceptible host. However, indirect contact transmission of viable virus by fomites and through aerosols has also been suggested. Herein, we report the involvement of fine indoor air particulates with a diameter of ≤ 2.5 µm (PM2.5) as the virus’s transport agent. PM2.5 was collected over four weeks during 48-h measurement intervals in four separate hospital wards containing different infected clusters in a teaching hospital in Kuala Lumpur, Malaysia. Our results indicated the highest SARS-CoV-2 RNA on PM2.5 in the ward with number of occupants. We suggest a link between the virus-laden PM2.5 and the ward’s design. Patients’ symptoms and numbers influence the number of airborne SARS-CoV-2 RNA with PM2.5 in an enclosed environment.
Similar content being viewed by others
Introduction
The Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) is primarily transmitted via respiratory droplets of various sizes1,2,3. Large respiratory droplets (> 5 μm) transmission occur when a person is in close contact with someone4 who has respiratory symptoms such as coughing or sneezing5. Whereas, finer virus-laden respiratory droplets and particulate matters (≤ 5 μm) can remain in the air for an extended period and be carried over greater distances6 > 6 m (such as the outbreak of tuberculosis, measles, and chickenpox)7. Despite numerous studies that have demonstrated the transmission route of SARS-CoV-2 via respiratory droplets, evidence on aerosols-borne transmission remains limited1,8,9.
Transmission of SARS-CoV-2 in a range of particulate matter (PM) from submicrometer and/or supermicrometer have been reported1,10. This suggests that the virus can be transported via solid aerosols. PM2.5 is fine solids with a particle diameter of ≤ 2.5 μm that is suspended in ambient air aerosols.
No correlation was found between the virus concentration and PM’s diameter. Nevertheless, positive correlations between PM2.5 and other respiratory viruses such as the influenza virus have been reported11, emphasizing the possibility of particulate matter as a transport carrier for SARS-CoV-2.
PM2.5 is fine solid aerosols with a particle diameter of ≤ 2.5 μm that is suspended in ambient air. PM2.5 in indoor environments is mainly derived from common outdoor sources such as motor-vehicles, biomass burning, and industrial emissions12,13,14. Prolonged exposure to PM2.5 is particularly detrimental to human health as this fine particulate matter can be easily inhaled and penetrate deep into the lungs15,16. PM2.5 is known to have a significantly longer lifetime in the air where it can be suspended at an extended period compared to respiratory liquid droplets. This longer lifetime of particles may pose a significant viral exposure to healthcare personnel, especially in indoor environments. PM2.5 can also be deposited in indoor environments such as hospitals’ flooring17,18 and any surface materials19,20. This fine particulate matter is readily propagated by tiny turbulent eddies in the air that arise from physical activities such as human movements and walking21,22. Considering the fact that the viability of SARS-CoV-2 on many types of surfaces have been reported (e.g., on metals for 48 h, plastic for 72 h, cardboard for 24 h, and copper for 4 h)23,24, it is likely that the virus on the surface can be potentially lodged on the PM2.5 and redistributed/transported back into the air.
Recent findings based on air particle measurements have suggested that SARS-CoV-2 can be carried by PM2.5 in the air when healthcare workers remove their personal protective equipment (PPE)2,5. Furthermore, it is also suggested that suspended tiny dust in the air could couple with microorganisms of diameter < 5 μm during aerosolization7. Since the diameter of the SARS-CoV-2 is two orders of magnitude smaller—approximately 70–90 nm25, the mechanism/mode of the airborne transport is still unclear and, therefore, worth exploring. In this study, we hypothesize the possible role of PM2.5 as a carrier (or transport agent) for SARS-CoV-2 to remain in the air. In order to prove this hypothesis, we investigated the PM2.5 burden and SARS-CoV-2 from several wards with COVID-19 patients in a hospital.
Methods
Sampling location and indoor air sampling
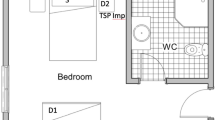
The layout and dimension of the wards are shown in Fig. 1. Each ward had different clusters of infected groups, as illustrated in Table 2 (in the main text) and Fig. 1. Each ward was occupied by one to eighteen COVID-19 patients. As a caveat, during the measurement in this study, hospital’s management staff has deployed three units of air purifiers at ward B, C, and D. During the air sampling measurement, air purifier (FANFIL AP510M, Aire-plus Technology, Singapore) was deployed at ~ 1 m distance in wards C and D, ~ 8 m in ward B, and no air purifier in single occupant room.
PM2.5 was sampled in a single-bed ward (31st March to 4th April 2020) and multiple bed wards (4th–29th April 2020) in a teaching hospital at Kuala Lumpur, respectively. Air sampling was conducted for 48 h during a 29 days sampling period using two types of instruments; an air quality sensor known as AiRBOXSense (AIRBOXSENSE V3.0, UKM Tech. Sdn Bhd, Malaysia)12,26 and a low volume sampler (LVS) (MINIVOL, AirMetrics, USA). Details of AiRBOXSense are described in26. Both instruments were operated side by side in wards occupied by SARS-CoV-2 positive patients. Instruments were treated using ultraviolet light for 20 min (UV) (UV-C 253.7 nm), further disinfected with 70% alcohol and calibrated before being translocated to the next wards. The same instruments were used to avoid variability during sampling.
AiRBOXSense was used to continuously measure PM2.5, while the LVS was used to determine the virus loading in PM2.5 trapped on filter paper (WHATMAN glass microfiber filters, Grade GF/F) with a tight specification of 0.6–0.8 μm particle retention and pure borosilicate glass structure, GF/F. A 5 L min−1 of air was drawn into the AiRBOXSense. While, the Minivol’s pump draws air at 5 L min−1 through a filter paper. The continuous concentration of PM2.5 was recorded and stored in secure digital card (SD card) in the AiRBOXSense. The data synchronously retrieved via THINGSPEAK (The MathWorks Inc, USA) cloud storage and analysed using MATLAB software (The MathWorks Inc, USA).
Each filter paper was collected after 48 h of sampling and stored in a sealed container and kept in − 80 °C laboratory freezer. The filter papers were extracted for viral load analysis using reverse transcription quantitative real time polymerase chain reaction (RT-qPCR) approach.
Calibration of AiRBOXsense
AiRBOXSense was calibrated according to26 1 day before each sampling. Calibration consists of setting a mathematical model describing the relationship between sensor data and reference instruments. The AiRBOXSense unit was calibrated in tandem with the GRIMM (as reference instrument) dust monitor model 1.108 (GRIMM Aerosol, Technik GmbH & Co. KG, Germany). The sensors measuring mass concentration were calibrated using GRIMM Aerosol, which was deployed at a clean area (laboratory) for lower concentration measurement and near to a car exhaust for high concentration measurement. The calibration equations are set by fitting a model during a calibration time interval when AiRBOXSense and GRIMM are co-located.
Viral nucleic acid extraction
Prior to viral nucleic acid extraction, the membrane filter was processed according to35 with slight modifications. The membrane was first divided into four parts and immersed in 1 mL sterile RNase-free water in separate tubes. Each part of the membrane was vortexed for 2 min in 30 s-intervals to release viral particles attached to the membrane. The tubes were then centrifuged at 500 rpm for 1 min to remove debris, and the supernatants were transferred into new microcentrifuge tubes for viral nucleic acid extraction. This process was repeated twice to ensure all virus particles were resuspended into the water. Subsequently, viral nucleic acid extraction was performed using a Viral Nucleic Acid Extraction Kit II (Geneaid Biotech Ltd., Taiwan) according to the manufacturer’s protocol. The purified nucleic acid containing the samples was then kept at − 80 °C for further analysis.
Reverse-transcription quantitative real-time polymerase chain reaction (RT-qPCR) analysis
The primers and probes used in the detection of SARS-CoV-2 were 2019-nCoV_N1, and 2019-nCoV_N2 combined primer/probe mixes purchased from Integrated DNA Technology (IDT). The information on primers and probes were included in Table 1. Human RNase P primer was not included as a control in this analysis because this study was not conducted using specimen from human. RT-qPCR was carried out using a THUNDERBIRD One-step RT-qPCR kit (Toyobo Co., Ltd., Japan) according to the manufacturer’s protocol. The annealing temperature of the primers was set at 55 °C, as suggested by Centres for Disease Control and Prevention or CDC (2020)28. Detection of SARS-CoV-2 using the RT-qPCR approach with a BIORAD iQ5 Real-Time PCR machine (BIORAD, USA) as described by CDC (2020) with slight modifications. A standard curve was also generated using 2019-nCoV Positive Control (nCoVPC) with a series of tenfold dilutions from 2 × 105 to 2 copies/µL of the control template. The amplification efficiency and R2 value were recorded, and the standard curve was used to estimate the viral RNA of SARS-CoV-2 on the membrane.
Results and discussion
Indoor PM2.5
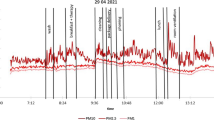
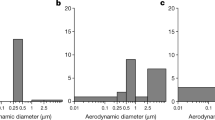
All 48 h average of PM2.5 concentration measurements and samplings were taken in COVID-19 wards as illustrated in Fig. 2 and Table 2. The highest concentration of indoor PM2.5 was measured in general ward B (23.27 µg m−3) on the 4th April, while the lowest 48 h average concentration was measured in general ward D (6.23 µg m−3) on the 22nd April as shown in Fig. 2. The General ward B was occupied by a cluster of patients from the same institution and was observed to have the most activity among the patients. Higher PM2.5 concentrations can be contributed by physical activities such as movements of health workers and patients21,27,29. The PM2.5 concentrations measured in this study are slightly lower than reported in a European urban hospital30.
Virus RNA analysis
SARS-CoV-2 RNA was isolated from filter membranes collected from the LVS. Only the N1 nucleocapsid gene was successfully detected in RT-qPCR in certain wards. According to the Emergency Use Authorization (EUA), detection of either the N1 or N2 gene is considered positive for the presence of SARS-CoV-230. We detected positive results for SARS-CoV-2 genes in the single room Ward A (74 ± 117.1 copies μL−1) and General Ward B (10 ± 7.44 copies μL−1). The viral genomes extracted from the filter paper were of heterogenous mixture. This contributes to the high standard deviation in the virus copy number as heterogeneous nucleic acid template was used in RT-qPCR and the presence of SARS-CoV-2 genome was relatively low. Nonetheless, the cycle threshold (CT) value was < 4030, confirming the positive detection of SARS-CoV-2 in our samples (Table 2). Due to operational restriction imposed by the hospital, the sample size was limited and replication was not possible.
The uniqueness in the result is that viral RNA was still able to be detected in the single occupancy ward (Ward A). Ward A is a small enclosed room (22 m2) with a lavatory attached. The frequent use of the lavatory by the symptomatic patient is likely to result in the increase of viral shedding activity in the room. We suspect that virus-laden PM2.5 generated from the shedding activity circulated within the enclosed room despite low PM2.5 concentration (11.25 µg m−3), thus explaining the spike in the data. The degree of viral shedding (from the patients) due to symptoms such as coughing, sneezing, diarrhoea, etc. has been reported to influence the number of virus particles in the environment1,5. It is suggested that the increased virus particles (due to shedding) in a poorly ventilated environment might increase the virus-PM2.5 assemblage9,19,31. A study done by5 reported that they were not able to detect SARS-CoV-2 in all of their tested air samples. However, they highlighted that their short sampling time of 15 min–4 h might not represent total air volume in the ward and the presence of SARS-CoV-2 might have possibly been diluted during air exchanges in the ward. In contrast, viral RNA was able to be detected in this study when air sampling duration was extended.
SARS-CoV-2 RNA was also detected in General Ward B. General Ward B is a larger room (~ 100 m2) consisting of 18 occupied beds with two air purifying units installed at a distance of farther away from the LVS. The amount of SARS-CoV-2 collected in the particulate matter is significantly lower than from Ward A despite the higher number of patients and concentration of PM2.5 (17.58 µg m−3). Such a low viral load in the PM2.5 could be attributed to the minimal viral shredding despite the high particulate matter. These particulate matters suspended in the air could be derived from floor and surfaces32,33 as a result of the high occupants’ activities in ward B.
Virus-laden PM2.5 was not detected in Wards C and D despite having similar ward size. The number of patients in Ward C is similar to Ward B, whereas the number of patients in Ward D is half of that of Wards C. The patients in Ward C and Ward D were also diagnosed with mild symptoms. The non-detection of the virus in these wards may be due to very low virus shedding from the patients. Another possible factor to explain the absence of SARS-CoV-2 RNA in PM2.5 is that the LVS in Ward C (and also Ward D) was positioned adjacent to an air purifier. Although air-purifier’s effectiveness in removing PM2.5 remains unclear, air-filtration has been reported to reduce viral loading in air9,32,33.
Our results clearly indicated that SARS-CoV-2 RNA is present within sampling of the Ambient’s particles. Hence, it is crucial to determine whether these RNAs came from intact virus particles or are merely RNA from non-infectious virus particles. The detection of SARS-CoV-2 viral RNA on surfaces was previously reported on a cruise ship, the Diamond Prince, even after 17 days after the evacuation of passengers34. In addition, the CDC pointed out that the infectivity of the detected particles was still uncertain. A study carried out in a CDC facility showed that SARS-CoV-2 could remain infectious up to 72 h on various types of surfaces24. Thus, it is suggested that infectious virus be determined by culturing of virus residing on the PM2.5 onto appropriate cell culture. However, our study could not show a direct link between the concentration of PM2.5 and SARS-CoV-2. We did find that PM2.5 generated from human activities in healthcare facilities can influence the presence of SARS-CoV-2 RNA in indoor environments. Furthermore, the degree of viral shedding from symptomatic patients may also influence the presence of SARS-CoV-2 RNA on PM2.5. Therefore, we recommend that all possible precautions against airborne transmission in indoor environments should be taken seriously.
References
Liu, Y. et al. Aerodynamic analysis of SARS-CoV-2 in two Wuhan hospitals. Nature. https://doi.org/10.1038/s41586-020-2271-3 (2020).
Chan, J. F. W. et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lancet 395, 514–523 (2020).
Huang, C. et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395, 497–506 (2020).
World Health Organization. Infection prevention and control of epidemic- and pandemic-prone acute respiratory infections in health care. (World Health Organization, Geneva, 2014).
Ong, S. W. et al. Air, surface environmental, and personal protective equipment contamination by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) from a symptomatic patient. JAMA 16, 1610–1612 (2020).
Dbouka, T. & Drikakisb, D. On coughing and airborne droplet transmission to humans. Phys. Fluids 32, 053310. https://doi.org/10.1063/5.0011960 (2020).
Ather, B., Edemekong, P. F. Airborne Precautions. (StatPearls Publishing, Treasure Island, FL, 2020).
Peng, X. et al. Transmission routes of 2019-nCoV and controls in dental practice. Int. J. Oral Sci. 12, 9 (2020).
Morawska, L., & Cao, J. Airborne transmission of SARS-CoV-2: The world should face the reality. Environ. Int. 139, 105730 (2020).
Guo, Z. et al. Aerosol and surface distribution of severe acute respiratory syndrome coronavirus 2 in hospital wards, Wuhan, China. Emerg. Infect. Disease J. 26, 1583–1591 (2020).
Su, W. et al. The short-term effects of air pollutants on influenza-like illness in Jinan, China. BMC Public Health. 19, 1319 (2019).
Nadzir, M. S. M. et al. The impact of movement control order (MCO) during pandemic COVID-19 on local air quality in an urban area of Klang Valley, Malaysia. Aerosol Air Qual. Res. https://doi.org/10.4209/aaqr.2020.04.0163 (2020).
Marcazzan, G. M., Vaccaro, S., Valli, G. & Vecchi, R. Characterisation of PM10 and PM2.5 particulate matter in the ambient air of Milan (Italy). Atmos. Environ. 35(27), 4639–4650 (2001).
Zhang, Y.-L. & Cao, F. Is it time to tackle PM25 air pollutants in China from biomass-burning emissions?. Environ. Pollut. 202, 217–219 (2015).
Xing, Y. F., Xu, Y. H., Shi, M. H. & Lian, Y. X. The impact of PM25 on the human respiratory system. J. Thorac. 8(1), 69–74. https://doi.org/10.3978/j.issn.2072-1439.2016.01.19 (2016).
Zwoździak, A., Sówka, I., Worobiec, A., Zwoździak, J. & Nych, A. The contribution of outdoor particulate matter (PM1, PM2.5, PM10) to school indoor environment. Indoor Built Environ. 24(8), 1038–1047. https://doi.org/10.1177/1420326X14534093 (2015).
Wang, X., Bi, X., Sheng, G. & Fu, J. Hospital indoor PM10/PM2.5 and associated trace elements in Guangzhou, China. Sci. Total Environ. 366(1), 124–135 (2006).
Gemenetzis, P., Moussas, P., Arditsoglou, A. & Samara, C. Mass concentration and elemental composition of indoor PM2.5 and PM10 in University rooms in Thessaloniki, northern Greece. Atmos. Environ. 40(17), 3195–3206 (2006).
Qian, J., Ferro, A. R. & Fowler, K. R. Estimating the resuspension rate and residence time of indoor particles. J. Air Waste Manag. Assoc. 58(4), 502–516 (2012).
Liu, C. et al. Influence of natural ventilation rate on indoor PM2.5 deposition. Build. Environ. 144, 357–364 (2018).
Chatoutsidou, S. E. et al. Indoor/outdoor particulate matter number and mass concentration in modern offices. Build. Environ. 92, 462–474 (2015).
Hänninen, O. O. et al. Reduction potential of urban PM2.5 mortality risk using modern ventilation systems in buildings. Indoor Air 15, 246–256 (2005).
National Ambient Air Quality Standards (NAAQS). https://doi.org/10.4135/9781483381503.n817.
van Doremalen, N. et al. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N. Engl. J. Med. 382(16), 1564–1567 (2020).
Kim, J. M. et al. Identification of coronavirus isolated from a patient in Korea with COVID-19. Osong Public Health Res. Perspect. 11(1), 3 (2020).
Alhasa, K. M. et al. Calibration model of a low-cost air quality sensor using an adaptive neuro-fuzzy inference system. Sensors. 18, 4380 (2018).
Slezakova, K., Pereira, M. & Alvim-Ferraz, M. Influence of tobacco smoke on the elemental composition of indoor particles of different sizes. Atmos. Environ. 43, 486–493 (2009).
Arfaeinia, H. Evaluation of public health impacts related to urban air pollution in Shiraz and Bushehr, Iran. Int. J. Pharm. Technol. 7(3), 9811–9824 (2015).
Galindo, N., Yubero, E., Nicolás, J. F., Varea, M. & Crespo, J. Characterization of metals in PM1 and PM10 and health risk evaluation at an urban site in the western Mediterranean. Chemosphere 201, 243–250 (2018).
Orig3n, Inc. Accelerated Emergency Use Authorization (EUA) summary Orig3n 2019 novel coronavirus (COVID-19) test (Orig3n, Inc.). Orig3n 2019 Novel Coronavirus (COVID-19) Test EUA Summary. https://www.fda.gov/media/136873/download (2020).
Mainka, A., Zajusz-Zubek, E. & Kaczmarek, K. PM2.5 in urban and rural nursery schools in Upper Silesia, Poland: Trace Elements Analysis. Int. J. Environ. Res. Public Health. 12(7), 7990–8008 (2015).
Hagbom, M. et al. Ionizing air affects influenza virus infectivity and prevents airborne-transmission. Nat. Sci. Rep. 5, 11431. https://doi.org/10.1038/srep11431 (2015).
Kujundzic, E., Matalkah, F., Howard, C. J., Hernandez, M. & Miller, S. L. UV air cleaners and upper-room air ultraviolet germicidal irradiation for controlling airborne bacteria and fungal spores. J. Occup. Environ. Hygiene. 3, 536–546 (2006).
Moriarty, L. F., Plucinski, M. M., Marston, B. J. et al. Public health responses to COVID-19 outbreaks on cruise ships—Worldwide, February–March 2020. MMWR Morb. Mortal (2020).
Thedell, T., Boles, C. L., Cwiertny, D. W., Brown, G. D., Qian, J., & Nonnenmann, M. W. Comparisons of a novel air sampling filter material, wash buffers and extraction methods in the detection and quantification of influenza virus. BioRxiv. 441154 (2018).
Acknowledgements
We convey our appreciation to Universiti Kebangsaan Malaysia (UKM) for permitting us to perform the sampling at their teaching hospital (Hospital Canselor Tuanku Muhriz UKM or HCTM), despite the country being held under the Movement Control Order (lockdown). This study was supported by the UKM Special Fund for COVID-19 no. COVID-2020-004. Dr. Mohd Shahrul would like to thank UKM for Grant nos. GUP-2018-135 and GGP-2019-015 for instrumentation support. Additionally, we would like to thank the hospital’s healthcare workers at Ward 7C, 6E, and 6H. We would also like to thank the staff from the HCTM microbiology laboratory for assistance in regards to sample storage and Dr. Bernard L H Saw for the assistance in the ward’s dimension measurement. We are also thankful to Innosens Technology (M) Sdn. Bhd for their assistance in AiRBOXSense’s deployment. We would like to specifically convey our appreciation to Prof Dato' Ir Dr. Abdul Wahab Mohammad for his encouragement to conduct this study especially during the initial stages of lockdown. We also like to thank Dr. Rose Norman from the United Kingdom for her assistance in proof reading this article. Finally, we would like to send our gratitude to all of the brave healthcare workers in Malaysia and worldwide during this time of crisis.
Author information
Authors and Affiliations
Contributions
M.S.M.N. conceptualized the idea. M.S.M.N., N.S.M.N., N.I., K.C. and Z.Z.R. aided in the study design. M.S.M.S. performed indoor air sampling procedures. M.S.M.N. drew the wards layout. N.S.M.N., C.W.Y. and N.I. did the qRT-PCR and rRT-PCR analyses. M.S.M.N., N.S.M.N., K.C., C.W.Y., and C.Y.L. wrote the early and the latest versions of the manuscript. All authors contributed equally to the data analysis and interpretations. All authors co-wrote the final version of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nor, N.S.M., Yip, C.W., Ibrahim, N. et al. Particulate matter (PM2.5) as a potential SARS-CoV-2 carrier. Sci Rep 11, 2508 (2021). https://doi.org/10.1038/s41598-021-81935-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-81935-9
This article is cited by
-
Country-specific determinants for COVID-19 case fatality rate and response strategies from a global perspective: an interpretable machine learning framework
Population Health Metrics (2024)
-
Revealing the Potential Effects and Risk of Microplastics from Face Mask on Human Health and Environment: A interlink Review on COVID-19 and Particulate Matter (PM2.5)
Water, Air, & Soil Pollution (2024)
-
Quantitative evaluation of the impact of indoor relative humidity on deposition of aerosols generated during tooth grinding in a real-world clinical setting
Clinical Oral Investigations (2024)
-
Utilizing a Low-Cost Air Quality Sensor: Assessing Air Pollutant Concentrations and Risks Using Low-Cost Sensors in Selangor, Malaysia
Water, Air, & Soil Pollution (2024)
-
Real-time environmental surveillance of SARS-CoV-2 aerosols
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.