Abstract
Invasions often accelerate through time, as dispersal-enhancing traits accumulate at the expanding range edge. How does the dispersal behaviour of individual organisms shift to increase rates of population spread? We collate data from 44 radio-tracking studies (in total, of 650 animals) of cane toads (Rhinella marina) to quantify distances moved per day, and the frequency of displacement in their native range (French Guiana) and two invaded areas (Hawai’i and Australia). We show that toads in their native-range, Hawai’i and eastern Australia are relatively sedentary, while toads dispersing across tropical Australia increased their daily distances travelled from 20 to 200 m per day. That increase reflects an increasing propensity to change diurnal retreat sites every day, as well as to move further during each nocturnal displacement. Daily changes in retreat site evolved earlier than did changes in distances moved per night, indicating a breakdown in philopatry before other movement behaviours were optimised to maximise dispersal.
Similar content being viewed by others
Introduction
The rate at which invasive species spread into new areas determines the timeframe of ecological impact, and is critical for management attempts to buffer those impacts. Thus, understanding factors that influence rates of population expansion is an important challenge. That challenge is complicated by extensive interspecific and intraspecific variation in rates of spread, driven by processes both extrinsic and intrinsic to populations1,2. Although the role of environmental and demographic variation is well appreciated, there is also mounting evidence that evolutionary processes contribute strongly to variation in invasion speeds3,4. For example, deterministic evolutionary processes on invasion fronts select for high rates of dispersal and population growth, causing invasions to accelerate5,6,7.
Mathematical models predict that individuals at the leading edge of an expanding population will accumulate phenotypic modifications that increase their dispersal ability8,9, through a spatial analogue of natural selection called spatial sorting10. Laboratory experiments have confirmed that prediction11,12,13, but field studies generally have relied on less direct evidence. Studies of dispersal rate evolution in free-ranging organisms mostly measure either rates of overall population spread14, or geographic variation in morphological, physiological and behavioural traits that plausibly influence dispersal ability. For example, individuals at an expanding range-edge might have larger wings or longer legs than do conspecifics in range-core populations15,16. In such studies, the links between phenotype, dispersal, and rate of population spread are either assumed or dependent upon correlational data.
We can fill in that knowledge gap, at least partly, by gathering empirical data on rates of dispersal of individuals at range-edges compared to range-core regions; but such studies are rare because it is more difficult to measure movement patterns of free-ranging animals than it is to capture those individuals and measure aspects of their phenotype. Even when dispersal rates of free-ranging animals are measured directly, the data typically are restricted to a small number of populations intended to be representative of different phases of the invasion process17,18 or studies at a single site through time19. As a result, no published studies have enough population-level replication to robustly identify general patterns in rates of dispersal of free-ranging individuals through the course of an invasion. However, we have now studied the spatial ecology of one iconic invader—the cane toad, Rhinella marina—in sufficient detail, in enough places, to fulfil this criterion. Native to South America, toads were translocated to Hawai’i in 1932, and from there to northeastern Australia in 1935—in both cases, to control insect pests of commercial agriculture. In Australia, the toads have since greatly expanded their range across the tropics to the west (moving into increasing hot and arid environments) and along the eastern coast to the south (into cooler climates). The westwards expansion across the tropics has been accompanied by a rapid acceleration in rates of expansion of the geographic range of toads14. In the present paper, we collate and analyse raw data from 44 radio-tracking studies on cane toads in three countries (French Guiana, USA and Australia) to explore general patterns of dispersal-relevant behaviour.
Results
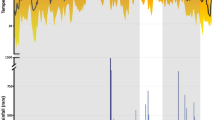
The mean distances moved by radio-tracked toads varied considerably among sites, from < 10 m per day in some locations in the native range and Hawai’i, through to more than 200 m per day at the invasion front in tropical Australia (Fig. 1a). Overall, dispersal distances were highest in tropical northwestern Australia (Northern Territory and Western Australia: see Fig. 1a, Table 1). Statistical analysis showed a significant link between colonisation history and daily displacements: toads closer to the invasion front (i.e., with lower time since colonisation of the site) moved further each day (Table 1). The distances traversed during displacements (i.e., omitting days when the toad returned to the same shelter-site on successive days) showed a similar pattern (Fig. 1b; Table 1), but with some exceptions to the general trend. For example, mean distances per move were as high in one of the native-range sites (Montjoly Beach) as in locations close to the invasion front in tropical Australia (Fig. 1b). For the third variable that we examined, the proportion of nights that toads changed shelter-sites from one day to the next, considerable variation was evident within the regions where toads were relatively sedentary overall (i.e., French Guiana, Hawai’i, Queensland [QLD], New South Wales [NSW]). By contrast, toads in tropical northwestern Australia showed little variation in this behaviour; most individuals shifted to a new location almost every night (Northern Territory [NT] and Western Australia [WA]: Fig. 1c). Despite that variation within locations, ANCOVA detected an overall link between time since colonisation and proportion of nights when toads moved from one shelter site to another: toads changed shelter-sites more often in invasion-front populations than in areas where the animals had been present for longer periods (Table 1).
Dispersal traits of cane toads. (A) Mean daily displacements by radio-tracked cane toads (Rhinella marina) from sites within the species’ native range in French Guiana, and in invaded areas of Hawai'i and Australia (QLD = Queensland; NSW = New South Wales; NT = Northern Territory; WA = Western Australia). (B) Mean displacements by radio-tracked cane toads (Rhinella marina) on nights when the toads changed diurnal shelter-sites. Data are shown from sites within the species’ native range in French Guiana, and in invaded areas of Hawai'i and Australia (QLD = Queensland; NSW = New South Wales; NT = Northern Territory; WA = Western Australia). (C) The proportion of days when radio-tracked cane toads (Rhinella marina) changed shelter-sites rather than returning to the same shelter-site on consecutive days. Data are shown from sites within the species’ native range in French Guiana, and in invaded areas of Hawai'i and Australia (QLD = Queensland; NSW = New South Wales; NT = Northern Territory; WA = Western Australia). The Figure plots raw data (means and standard errors) but statistical analyses were based on ln-transformed data. Photographs by J. DeVore.
An ANCOVA with region as the factor revealed that the effect of invasion history on movement parameters differed among regions for distances moved but not for the proportion of days with changes in shelter-sites (interaction years since colonised*region effects: Table 1). We thus repeated the ANCOVAs on subsets of data from regions that contained sites differing in times since colonisation by cane toads (i.e., northwestern or eastern Australia). All three movement variables were linked to time since colonisation in tropical northwestern Australia. The two distance variables increased closer to the invasion front whereas the proportion of nights when toads changed shelters decreased closer to the front (Table 1). In eastern Australia (QLD plus NSW), neither of the distance variables were significantly linked to time since colonisation, but toads closer to the invasion front shifted shelter-sites more frequently than did conspecifics from long-established populations (Table 1).
Discussion
Our review of data on free-ranging cane toads reveals a pattern that is largely consistent with theory. In their native range, cane toads are relatively sedentary; a situation that was maintained following introduction to the islands of Hawai’i. The continuous invasion of northern Australia over 80 years is, however, associated with the emergence of highly dispersive phenotypes on the invasion front. Especially in terms of daily distances moved, the geographic pattern is strong (Fig. 1a). Studies on captive-raised offspring of Australian toads have documented significant heritability of dispersal behaviour, suggesting that these are evolved changes rather than phenotypically plastic responses to local conditions15,20.
The cline in dispersal rates across northern Australia was first observed some years ago17,18, but until a recent telemetry study21, the spatial ecology of cane toads within their native range was poorly known. This was an important missing piece of the puzzle, because (despite strong theoretical expectations) the Australian pattern may have been driven not by increased dispersal on the invasion front, but by reduced dispersal in the long-established parts of the range. Comparison with dispersal rates in the native range (based on studies in both beachside and forested habitats21) now allows confidence as to the direction of evolution in Australia. Previously published information on dispersal in the native range came from Central America, and were based on studies of a toad species now recognised as R. horribilis rather than R. marina22,23. Based on those reports, the two species may exhibit similarly low vagility in the native range. Although his methods were not directly comparable to ours (reliant on spooling and mark-recapture not telemetry), Bayliss24 recorded modest overall dispersal for cane toads in Amazonian Brazil; net displacements were around 30 m per day, within the range we recorded in French Guiana. Overall, available data support the idea that cane toads within their native range are similar to those in long-established Australian populations. Individuals move nomadically, but their dispersal is constrained to relatively slow, short movements. In Puerto Rico (the stepping-stone population between French Guiana and Hawai’i25), Carpenter and Gillingham26 also reported strong fidelity to water holes in Puerto Rican cane toads.
The translocation of cane toads to Hawai’i in 1932, with subsequent release of thousands of their progeny in sugar-growing areas on all the major islands25, has resulted in disjunct populations of toads on the dry (leeward) sides of islands and more continuous distribution on the wetter (windward) sides27. In Hawai’i, the toad population did not have thousands of square kilometres of uncolonised suitable habitat through which to invade. Instead, toad populations on Hawai’i experienced strong environmental gradients associated with elevation and between moist and dry sides of each island. Even in areas with high rainfall, porous volcanic soils restrict the availability of moisture and hence make long-distance dispersal risky27. The strong spatial heterogeneity and limited vacant habitat on Hawai’i likely explains why cane toads have not evolved increased rates of dispersal there (Fig. 1): spatial heterogeneity directly selects against long-distance movements28, and fine-grained spatial heterogeneity can increase the strength of genetic drift such that it overwhelms selection29.
The cane toads that were translocated from Hawai’i to Queensland in 1935 encountered a very different opportunity. Here, there was a vast area of suitable habitat to the west of their introduction point. Arid habitats in southwestern Queensland restricted the toads’ geographic spread to the southwest, leaving possible routes of spread in two main directions—northwest through the tropics to the Northern Territory and then Western Australia; and south along the east coast down into New South Wales14,30. The toads exploited both of those opportunities, but with different invasion dynamics. The northwestern front accelerated to > 50 km per annum (from an initial spread rate of < 15 km per annum) whereas the southern front moved very slowly (< 10 km per annum14,31). In keeping with those rates of population spread, invasion-front toads in northwestern Australia exhibit rapid dispersal whereas southern (NSW) toads do not.
The acceleration in rate of dispersal in northwestern Australia reflects the joint effects of spatial and temporal evolutionary processes6. First, the leading tip of the invasion is a region of low conspecific density and populations experience exponential growth. Here, any trait that increases the reproductive rate is favoured. This is standard r-selection, and can be seen as a race between genetic variants through time. Second, only the most dispersive individuals make it to the leading edge of the invasion in every generation and so only genes that confer high dispersal rates will be on the invasion front. This is spatial sorting and can be seen as a race between genetic variants through space10. On continuous invasion fronts, evolutionary increases in reproductive rate steepen the invasion front and further increase the strength of spatial sorting9,32.
That acceleration has not occurred in eastern Australia, either in rates of population expansions31,33 or in the movements of individual toads (Fig. 1a). This again likely reflects environmental heterogeneity, albeit in this case manifesting as the latitudinal gradient in temperature and rainfall seasonality that will ultimately place a limit on the toads’ distribution in the south34. Low ambient temperatures at night in northern New South Wales appear to limit the rate of spread of these tropical amphibians35, and, in keeping with the hypothesis of temperature limitation, the toad front in New South Wales has expanded more rapidly during decades with unusually high ambient temperatures31.
The overall rate of daily dispersal of a cane toad (Fig. 1a) is driven by two variables: how far a toad travels when it leaves its previous shelter-site before selecting a new one (Fig. 1b) and the proportion of nights on which a toad moves to a new shelter rather than returning to the previous one (Fig. 1c). Both of these variables shift relative to recency of colonisation of a site (above), but in different ways. Distances travelled were linked more consistently to invasion chronology (Fig. 1a,b) than was frequency of movement from one shelter-site to another. Indeed, changes in shelter-sites were slightly less frequent, not more frequent, close to the invasion front in tropical Australia (Fig. 1c). That non-intuitive pattern likely reflects the toads’ spread into arid regions where moist retreat sites are difficult to find, and hence (as in Hawai’i) favouring re-use of known sites, particularly in the dry season. Toads shifted to new shelter-sites on most nights in some (but not all) populations from throughout the species’ range, including in regions where overall rates of daily dispersal were low (Fig. 1a). Thus, one of the movement parameters that influences dispersal rate (propensity to change shelter-sites frequently) was more variable at a local scale than was the other parameter (distance moved per night). Interestingly, the frequency of changes in shelter-sites decreased closer to the invasion front in tropical northwestern Australia (where the front has accelerated: Fig. 1c) but increased closer to the invasion front in eastern Australia (where the front has not accelerated: Fig. 1c).
The consistently high frequency of shelter-site changes in tropical Australia compared to the ancestral condition (as in coastal French Guiana and Hawai’i: Fig. 1c) suggests that in the course of this invasion, increased rates of dispersal were first achieved by reducing fidelity to specific shelter-sites. An increase in distance moved per night was the second step. Willingness to abandon a previously-used shelter-site requires only a minor behavioural shift. Essentially it is simply choosing not to return home; this may incur little or no cost in the tropical wet-season36. Such a shift immediately increases the dispersal rate of the population, but it also exposes other dispersal-relevant traits—such as movement rate, stamina, and path straightness—to spatial sorting. Thus, the progression in displacement behaviour we see in toads may apply generally: the stepwise evolutionary increases in overall dispersal rate during an invasion may be achieved first by a breakdown in site fidelity and only later by optimisation of other dispersal-relevant traits.
Importantly, cane toads also adjust their movement patterns to local conditions, with moist conditions increasing the probability of a toad emerging at night from its diurnal retreat37, and moving long distances38. The spatial distribution of local resources—such as areas for sheltering, feeding, rehydrating and breeding—also influences movements21,39. Thus, the behaviours seen in invasion-front cane toads (i.e., frequent changes in shelter-sites, and long-distance displacements) are manifested in all populations of cane toads, even in the native range—but only by some individuals, some of the time21,36. In the course of their international diaspora, some populations of cane toads—those in northwestern Australia—have shifted to constitutive expression of these previously-facultative traits. All cane toads are capable of rapid dispersal, but exhibit those rates only under specific conditions40,41. That scenario may be common; for example, Komodo dragons have profound capacities for navigation and movement, despite most individuals never leaving the valley in which they were born42. Given sufficient evolutionary pressure, these latent abilities can become the predominant phenotype: at the tropical Australian invasion front, all toads disperse rapidly, and do so as long as conditions allow it.
Future work could usefully examine geographic variation in other traits that influence overall rates of dispersal. For example, one of the most efficient ways to disperse long distances is to keep moving in the same direction night after night. Cane toads at the invasion front in tropical northwestern Australia tend to move in this way, and this path straightness is heritable20. It is easy to imagine other behavioural traits that would enhance rates of dispersal, such as a preference for moving along open linear corridors rather through dense vegetation40. More detailed studies of cane toads at different stages of the invasion process could explore routes as well as rates of dispersal (e.g., avoidance of dispersal barriers), and the timing and abiotic correlates of dispersal behaviour (what conditions of darkness and moisture elicit emergence at dusk?).
To evaluate generalities in the evolutionary processes driving rapid evolution during biological invasions, it would also be of interest to examine other examples. Is it generally true, for example, that evolutionary innovation is first driven by changes in behaviour, before morphological and physiological traits evolve43,44,45? In many cases, some level of behavioural shift is required to impose selection on non-behavioural traits. For example, soapberry bugs (Leptocoris tagalicus) likely exhibited at least a partial switch in hostplant preference prior to evolving longer mouthparts that enhanced the bugs’ ability to feed on the invasive plant46, and lizards (Anolis carolinensis) presumably shifted to higher perches following invasion by competitors before evolving morphological adaptations (larger toepads) that facilitate arboreal activity47. Analogously, cane toads appear to have evolved more frequent shifts among refuge-sites prior to evolving enhanced rates of daily dispersal.
In summary, our data on cane toads provide strong evidence for shifts in dispersal behaviour in the course of a biological invasion. Importantly, we have measured actual rates of dispersal rather than relying upon putative links between an individual’s phenotype and its likely rate of dispersal. Our data reinforce the emerging conclusion that we need to incorporate evolutionary thinking into our approach to wildlife management; the speed and magnitude of shifts in important traits (such as those driving dispersal propensity) may be high enough that we ignore them at our peril.
Methods
Study species
The cane toad (Rhinella marina; Bufo marinus in earlier literature) is among the largest anuran species (exceptionally, to > 1 kg in mass), and is native to northern South America east of the Andes48 (Fig. 2). Members of the same clade from west of the Andes, previously allocated to R. marina, are now recognised as a separate species, R. horribilis48. Bufonid anurans are capable of sustained locomotion49, but most field studies of toads report sedentary behaviour interspersed (in some species) with long seasonal migrations50,51,52. Studies in Australia suggest that newly-metamorphosed toads are highly philopatric to the natal waterbody until they grow large enough to resist desiccation and thus, can move into the more arid surrounding habitat53. Adult cane toads are primarily inactive by day (but see Pettit et al.54 for exceptions), within cool moist retreat-sites37. The animals emerge at night to forage, rehydrate and breed27,40. Because dispersal occurs by night, the linear distance between locations of diurnal retreat-sites used on successive days offers a simple metric of dispersal.
Cane toad (Rhinella marina) with radio-transmitter attached to waist belt. Photograph by Matthew Greenlees. Cane toads were exported from their native range in the Guianas to the Caribbean, before being taken from Puerto Rico to Oahu, Hawai’i. Toads were further spread across the Hawai’ian islands of Kauai, Molokai, Lanai, Maui, and Hawai’i (i.e., “the Big Island”, where tracking data were collected) from 1933 to 1935. Toads from Oahu were also transported to Queensland (QLD), Australia. Here we have depicted the regions in which toad movements have been radio-tracked in red. Within Australia, the red arrows indicate the two routes of spread from the original introduction site (northwestern and eastern), and the black dotted line indicates the extent of their current distribution.
Cane toads are a prolific invasive species, translocated to many places around the world in attempts to control insect pests in commercial sugarcane plantations. These toads were taken from French Guiana and Guyana to sugar-cane plantations in Puerto Rico (via Martinique, Barbados, and/or Jamaica) in the early nineteenth century, and 150 individuals from Puerto Rico were translocated to Hawai’ian plantations in 193225. Three years later, 101 toads from Hawai’i were brought to northeastern Queensland, Australia, and their progeny released in sugar-growing areas over much of the eastern Queensland coast. The toads failed to control insect pests55 but dispersed rapidly. Rates of invasion accelerated from around 10 km per annum to > 50 km per annum as the toads spread westward through Queensland and the Northern Territory into Western Australia14,38,56, but spread rates remained low as the toads moved south into cooler regions of southern Queensland and New South Wales30,31,33.
Methods
We compiled original data from 44 radio-tracking studies (total of 29 sites, 650 toads; n = 3–75 toads tracked per site) that we conducted using consistent methods. Adult cane toads were captured in the wild, fitted with small radio-transmitters on waist belts (Holohil model PD-2, 2.5 gm, < 3% of toad body mass), released at the site of capture, and then relocated on a daily basis (where possible) for at least 5 days thereafter. We did not include data from spool-tracking studies24,57, or from studies with longer intervals between relocations39. Most of the studies were conducted by members of the same research group, over a 16-year period (2005 to 2020). In some cases, studies were conducted at the same sites several years apart, and hence with different times since initial colonisation by toads. All studies were conducted during weather conditions that facilitate toad activity (warm, with at least intermittent rain37,38), and we excluded data from toads that had been subjected to experimental treatments (such as translocation, captive rearing, or removal of parasites58,59) that might change their dispersal behaviour.
From the available data, we extracted information on two variables: (1) mean distance moved per day (from one diurnal shelter site to the next, including occasions when the same shelter was used) and (2) the proportion of nights that the toad changed diurnal retreat-sites (i.e., was found in different shelters > 5 m apart on successive days). From these two variables we also calculated (3) daily distance moved in cases where the toad changed shelter-sites (i.e., omitting zeros for times when toads did not change shelter-sites from one day to the next).
Ethical note
This paper is based on a collation of data from previous research; no additional animals were handled in the course of the current work. In the original studies, all experimental protocols were approved by institutional committees (University of Sydney Animal Care and Ethics Committee; James Cook University Animal Care and Ethics Committee) and were conducted in accordance with all relevant guidelines and regulations, including the ARRIVE guidelines.
Analysis
We classified each study site in terms of time since invasion by toads (based on historical records14,60). Table 2 provides data on study site locations, the seasonal timing of radio-tracking, and the number of toads tracked. We ln-transformed the dependent variables to improve normality; for proportion of shelter-site changes our statistical analysis used ln (1-proportion of changes). To examine the predicted increase in dispersal-rate attributes close to the invasion front, we used ANCOVA with the dispersal parameter (e.g., ln daily distance travelled) as the dependent variable, years since invasion of the site as a covariate, and population identity as a random factor (to allow for measures on multiple toads within a single population). For the purposes of this analysis, we allocated the four native-range sites an arbitrary “age since colonisation” of 100 years (i.e., marginally longer than any of the invaded sites). To evaluate whether or not regions differed in the effects of invasion history (time since colonisation) on movement parameters, we used an ANCOVA with region as the factor and invasion history as the covariate, plus the interaction term between region and invasion history. We then conducted ANCOVAs separately on data for the two regions within which sites with a range of invasion histories were sampled (eastern Australia and tropical northwestern Australia).
Data availability
Data are available in the Dryad Data Repository https://doi.org/10.5061/dryad.tb2rbp021.
References
Melbourne, B. A. & Hastings, A. Highly variable spread rates in replicated biological invasions: Fundamental limits to predictability. Science 325, 1536–1539 (2009).
Lewis, M. A., Petrovskii, S. V. & Potts, J. R. The Mathematics Behind Biological Invasions (Springer, 2016).
Phillips, B. L. Evolutionary processes make invasion speed difficult to predict. Biol. Invasions 17, 1949–1960 (2015).
Peischl, S., Kirkpatrick, M. & Excoffier, L. Expansion load and the evolutionary dynamics of a species range. Am. Nat. 185, E81–E93 (2015).
Burton, O. J., Travis, J. M. J. & Phillips, B. L. Trade-offs and the evolution of life-histories during range expansion. Ecol. Lett. 13, 1210–1220 (2010).
Phillips, B. L. & Perkins, T. A. Spatial sorting as the spatial analogue of natural selection. Theor. Ecol. 12, 155–163 (2019).
Deforet, M., Carmona-Fontaine, C., Korolev, K. S. & Xavier, J. B. Evolution at the edge of expanding populations. Am. Nat. 194, 291–305 (2019).
Travis, J. M. J. & Dytham, C. Dispersal evolution during invasions. Evol. Ecol. Res. 4, 1119–1129 (2002).
Bouin, E. et al. Invasion fronts with variable motility: Phenotype selection, spatial sorting and wave acceleration. C. R. Math. 350, 761–766 (2012).
Shine, R., Brown, G. P. & Phillips, B. L. An evolutionary process that assembles phenotypes through space rather than through time. Proc. Natl. Acad. Sci. USA 108, 5708–5711 (2011).
Williams, J. L., Kendall, B. E. & Levine, J. M. Rapid evolution accelerates plant population spread in fragmented experimental landscapes. Science 353, 482–485 (2016).
Weiss-Lehman, C., Hufbauer, R. A. & Melbourne, B. A. Rapid trait evolution drives increased speed and variance in experimental range expansions. Nat. Commun. 8, 14303 (2017).
Ochocki, B. M. & Miller, T. E. Rapid evolution of dispersal ability makes biological invasions faster and more variable. Nat. Commun. 8, 14315 (2017).
Urban, M. C., Phillips, B. L., Skelly, D. K. & Shine, R. The cane toad’s (Chaunus [Bufo] marinus) increasing ability to invade Australia is revealed by a dynamically updated range model. Proc. R. Soc. B 274, 1413–1419 (2007).
Phillips, B. L., Brown, G. P. & Shine, R. Evolutionarily accelerated invasions: The rate of dispersal evolves upwards during the range advance of cane toads. J. Evol. Biol. 23, 2595–2601 (2010).
Chuang, A. & Peterson, C. R. Expanding population edges: Theories, traits, and trade-offs. Glob. Change Biol. 22, 494–512 (2016).
Phillips, B. L., Brown, G. P., Travis, J. M. & Shine, R. Reid’s paradox revisited: The evolution of dispersal kernels during range expansion. Am. Nat. 172, S34–S48 (2008).
Alford, R. A., Brown, G. P., Schwarzkopf, L., Phillips, B. L. & Shine, R. Comparisons through time and space suggest rapid evolution of dispersal behaviour in an invasive species. Wildl. Res. 36, 23–28 (2009).
Lindström, T., Brown, G. P., Sisson, S. A., Phillips, B. L. & Shine, R. Rapid shifts in dispersal behavior on an expanding range edge. Proc. Natl. Acad. Sci. USA 110, 13452–13456 (2013).
Brown, G. P., Phillips, B. L. & Shine, R. The straight and narrow path: The evolution of straight-line dispersal at a cane toad invasion front. Proc. R. Soc. B 281, 20141385 (2014).
DeVore, J., Ducatez, S. & Shine, R. Spatial ecology of cane toads (Rhinella marina) in their native range: A study from French Guiana. Sci. Rep. 11, 11817 (2021).
Brattstrom, B. H. Homing in the giant toad, Bufo marinus. Herpetologica 18, 176–180 (1962).
Zug, G. R. & Zug, P. B. The marine toad Bufo marinus: A natural history resumé of native populations. Smithson. Contrib. Zool. 284, 1–58 (1979).
Bayliss, P. The ecology of post-metamorphic Bufo marinus in central Amazonian savanna, Brazil. Unpublished Ph.D. thesis (The University of Queensland, 1995).
Turvey, N. Cane Toads: A Tale of Sugar, Politics and Flawed Science (Sydney University Press, 2013).
Carpenter, C. C. & Gillingham, J. C. Water hole fidelity in the marine toad, Bufo marinus. J. Herpetol. 21, 158–161 (1987).
Ward-Fear, G., Greenlees, M. J. & Shine, R. Toads on lava: Spatial ecology and habitat use of invasive cane toads (Rhinella marina) in Hawai’i. PLoS ONE 11, e0151700 (2016).
Hastings, A. Can spatial variation alone lead to selection for dispersal? Theor. Popul. Biol. 24, 244–251 (1983).
Möbius, W., et al. The collective effect of finite-sized inhomogeneities on the spatial spread of populations in two dimensions. Preprint at http://arxiv.org/abs/1910.05332 (2019).
Urban, M. C., Phillips, B. L., Skelly, D. K. & Shine, R. A toad more traveled: The heterogeneous invasion dynamics of cane toads in Australia. Am. Nat. 171, E134–E148 (2008).
Macgregor, L. F., Greenlees, M., de Bruyn, M. & Shine, R. An invasion in slow motion: The spread of invasive cane toads (Rhinella marina) into cooler climates in southern Australia. Biol. Invasions 23(11), 3565–3581 (2021).
Perkins, A. T., Phillips, B. L., Baskett, M. L. & Hastings, A. Evolution of dispersal and life history interact to drive accelerating spread of an invasive species. Ecol. Lett. 16, 1079–1087 (2013).
Seabrook, W. Range expansion of the introduced cane toad Bufo marinus in New South Wales. Aust. Zool. 27, 58–62 (1991).
Kearney, M. R. et al. Modelling species distributions without using species distributions: The cane toad in Australia under current and future climates. Ecography 31, 423–434 (2008).
McCann, S. M., Kosmala, G. K., Greenlees, M. J. & Shine, R. Physiological plasticity in a successful invader: Rapid acclimation to cold occurs only in cool-climate populations of cane toads (Rhinella marina). Conserv. Physiol. 6, cox072 (2018).
Schwarzkopf, L. & Alford, R. A. Nomadic movement in tropical toads. Oikos 96, 492–506 (2002).
Seebacher, F. & Alford, R. A. Movement and microhabitat use of a terrestrial amphibian (Bufo marinus) on a tropical island: Seasonal variation and environmental correlates. J. Herpetol. 33, 208–214 (1999).
Phillips, B. L., Brown, G. P., Greenlees, M., Webb, J. K. & Shine, R. Rapid expansion of the cane toad (Bufo marinus) invasion front in tropical Australia. Austral Ecol. 32, 169–176 (2007).
Tingley, R. & Shine, R. Desiccation risk drives the spatial ecology of an invasive anuran (Rhinella marina) in the Australian semi-desert. PLoS ONE 6, e25979 (2011).
Brown, G. P., Phillips, B. L., Webb, J. K. & Shine, R. Toad on the road: Use of roads as dispersal corridors by cane toads (Bufo marinus) at an invasion front in tropical Australia. Biol. Conserv. 133, 88–94 (2006).
Pettit, L. J., Greenlees, M. J. & Shine, R. Is the enhanced dispersal rate seen at invasion fronts a behaviourally plastic response to encountering novel ecological conditions? Biol. Lett. 12, 20160539 (2016).
Jessop, T. S. et al. Exploring mechanisms and origins of reduced dispersal in island Komodo Dragons. Proc. R. Soc. B 285, 20181829 (2018).
Mayr, E. Animal Species and Evolution (Harvard University Press, 1963).
Duckworth, R. A. The role of behavior in evolution: A search for mechanism. Evol. Ecol. 23, 513–531 (2009).
Muñoz, M. M. & Losos, J. B. Thermoregulatory behavior simultaneously promotes and forestalls evolution in a tropical lizard. Am. Nat. 191, E15–E26 (2017).
Carroll, S. P. et al. And the beak shall inherit–evolution in response to invasion. Ecol. Lett. 8, 944–951 (2005).
Stuart, Y. E. et al. Rapid evolution of a native species following invasion by a congener. Science 346, 463–466 (2014).
Acevedo, A. A., Lampo, M. & Cipriani, R. The cane or marine toad, Rhinella marina (Anura, Bufonidae): Two genetically and morphologically distinct species. Zootaxa 4103, 574–586 (2016).
Reilly, S. M. et al. Conquering the world in leaps and bounds: Hopping locomotion in toads is actually bounding. Funct. Ecol. 29, 1308–1316 (2015).
Griffis-Kyle, K. L., Kyle, S. & Jungels, J. Use of breeding sites by arid-land toads in rangelands: Landscape-level factors. Southwest. Nat. 56, 251–255 (2011).
Sinsch, U. Movement ecology of amphibians: From individual migratory behaviour to spatially structured populations in heterogeneous landscapes. Can. J. Zool. 92, 491–502 (2014).
Cayuela, H. et al. Determinants and consequences of dispersal in vertebrates with complex life cycles: A review of pond-breeding amphibians. Q. Rev. Biol. 95, 1–36 (2020).
Child, T., Phillips, B. L., Brown, G. P. & Shine, R. The spatial ecology of cane toads (Bufo marinus) in tropical Australia: Why do metamorph toads stay near the water? Austral Ecol. 33, 630–640 (2008).
Pettit, L., Ducatez, S., DeVore, J. L., Ward-Fear, G. & Shine, R. Diurnal activity in cane toads (Rhinella marina) is geographically widespread. Sci. Rep. 10, 5723 (2020).
Shine, R., Ward-Fear, G. & Brown, G. P. A famous failure: Why were cane toads an ineffective biocontrol in Australia? Conserv. Sci. Pract. 2, e296 (2020).
Shine, R., Everitt, C., Woods, D. & Pearson, D. J. An evaluation of methods used to cull invasive cane toads in tropical Australia. J. Pest Sci. 91, 1081–1091 (2018).
Silvester, R., Greenlees, M., Shine, R. & Oldroyd, B. Behavioural tactics used by invasive cane toads (Rhinella marina) to exploit apiaries in Australia. Austral Ecol. 44, 237–244 (2019).
Finnerty, P. B., Shine, R. & Brown, G. P. The costs of parasite infection: Effects of removing lungworms on performance, growth and survival of free-ranging cane toads. Funct. Ecol. 32, 402–415 (2018).
Pettit, L., Greenlees, M. & Shine, R. The impact of transportation and translocation on dispersal behaviour in the invasive cane toad. Oecologia 184, 411–422 (2017).
Kraus, F. Alien Reptiles and Amphibians: A Scientific Compendium and Analysis (Springer, 2008).
McCann, S., Greenlees, M. J. & Shine, R. On the fringe of the invasion: The ecological impact of cane toads in marginally suitable habitats. Biol. Invasions 19, 2729–2737 (2017).
S. Kaiser et al., unpubl. Data.
Finnerty, P., Shine, R. & Brown, G. P. Survival of the faeces: Does a nematode lungworm adaptively manipulate the behaviour of its cane toad host? Ecol. Evol. 8, 4606–4618 (2018).
Brown, G. P., Kelehear, C., Pizzatto, L. & Shine, R. The impact of lungworm parasites on rates of dispersal of their anuran host, the invasive cane toad. Biol. Invasions 18, 103–114 (2016).
G. Ward-Fear et al., unpubl. Data.
Acknowledgements
We thank Crystal Kelehear for assistance. The work was funded by the Australian Research Council (FL120100074, to RS).
Author information
Authors and Affiliations
Contributions
R.S. had the idea for this paper, and assembled and analysed the data. R.S. and B.L.P. wrote the paper, with input from the other authors. All authors participated in collecting data for this paper, and all read the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shine, R., Alford, R.A., Blennerhasset, R. et al. Increased rates of dispersal of free-ranging cane toads (Rhinella marina) during their global invasion. Sci Rep 11, 23574 (2021). https://doi.org/10.1038/s41598-021-02828-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-02828-5
This article is cited by
-
Developments in the study of poison frog evolutionary ecology I: social interactions, life history and habitat use across space and ontogeny
Evolutionary Ecology (2024)
-
Spatial ecology of the invasive Asian common toad in Madagascar and its implications for invasion dynamics
Scientific Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.