Abstract
The longitudinal relationship between smoking status and risk of developing visual impairment (VI) remains unclear. We examined the relationship of smoking status and urinary cotinine level, an objective measure of smoking, with incidence of VI. This cohort study included 279,069 individuals free of VI who were followed for up to 8.8 years (median 4.8 years). VI was defined as when bilateral visual acuity was worse than 0.5 (cutoffs of 0.3 Logarithm of the Minimum Angle of Resolution). During 1,324,429.8 person-years of follow-up, 7852 participants developed new-onset bilateral VI. Self-reported current smoking status was associated with increased risk of developing VI in both men and women, with a stronger association in women (P for interaction = 0.01). Multivariable adjusted hazard ratios (95% confidence intervals) for incident VI comparing current smokers to never-smokers were 1.14 (1.04–1.25) in men and 1.52 (1.28–1.80) in women. Urinary cotinine levels of ≥ 100 ng/ml were significantly associated with increased risk of incident VI, and these associations remained when introducing changes in urinary cotinine and other confounders during follow-up as time-varying covariates. Cigarette smoking assessed based on self-report and urinary cotinine level was associated with increased incidence of VI. Our findings identify smoking as an independent risk factor for VI.
Similar content being viewed by others
Introduction
Visual impairment (VI) or blindness is an important public health issue that was estimated to affect 440 million people in 2015 worldwide1,2. VI reduces vision-related quality of life, which results in increases of risk of dependency, accidents, falls, and depression3,4,5,6. VI itself is reported to be related to high risk of mortality7,8,9. Age-related macular degeneration (AMD) and cataracts are the leading ocular diseases related to severe bilateral VI, and the prevalence of these diseases increases with age10. However, the cause and mechanism of VI is not fully elucidated, other than being related to specific eye diseases such as AMD, cataract, glaucoma, and diabetic retinopathy. Indeed, Visual acuity (VA) reflects the integrity of the visual system and is considered as a “vital sign” of ocular function11. In this context, VI appears to be an important sign of integral ocular health. Therefore, it is important to identify modifiable risk factors for VI to establish preventive strategies to decrease VI-associated health burden and social cost, and preserve quality of life.
Cigarette smoking is the leading preventable cause of premature mortality worldwide and is closely associated with a wide range of diseases12. Smoking is associated with worsening of some ocular morbidities, especially AMD and cataracts13. Most studies have focused on the detrimental effects of cigarette smoking on individual specific eye diseases such as AMD14,15,16 and cataracts17,18. Only a few studies have addressed the relationship between smoking and VI19,20,21,22, and in existing studies, cigarette smoking was only one of many behavioral factors rather than a main exposure variable, and the results did not provide detailed information about smoking. Furthermore, many previous studies were limited by cross-sectional design, lack of consideration of sex differences, ambiguous temporal relationship of smoking with VI, small sample size, and subjective methods to measure smoking, mainly relying on self-reports. Cotinine, the main metabolite of nicotine, is a reliable objective measure of cigarette smoking status that is useful to minimize the misclassification of smoking status based on self-report23.
Despite the health risks related to cigarette smoking and the tobacco control policies in Korea, such as inclusion of health warning labels and images on cigarette packages, increasing smoke-free public places, anti-smoking campaigns and education, and increasing tobacco prices prices24,25, the high prevalence of smoking is still observed in Korea, approximately 49.8% of men and 4.2% of women in 2015 according to the 2017 report of the World Health Organization26,27.
Therefore, we examined the longitudinal relationships between smoking status, pack-years, and urinary cotinine level as an objective measure of smoking with the risk of developing VI in a large cohort of participants who underwent health screening examinations, while accounting for time-dependent measures of change in smoking status and other covariates during follow-up.
Results
At baseline, the mean (standard deviation) ages of men and women participants were 38.5 (7.9) years, and 36.6 (7.8) years, respectively (Tables 1, 2). The prevalence of never, former, and current smokers based on self-reports were 27.7%, 35.9%, and 36.4%, respectively, among men and 90.3%, 7.4%, and 2.3%, respectively, among women. Among men, compared with never-smokers, current smokers were more likely to be older, drink alcohol, and have worse lipid profiles and to have high levels of body mass index (BMI), liver enzymes, Homeostatic Model Assessment for Insulin Resistance (HOMA-IR), and total energy intake, while they were less likely to have a high educational level (Table 1). Among women, current smokers were more likely to drink alcohol and have high levels of BMI, triglycerides, but have lower HOMA-IR scores, and they were less likely to have a high educational level than never-smokers (Table 2).
Table 3 shows the relationship between self-reported smoking status, smoking pack-years, and the incidence of binocular VI in men and women separately. During 1,324,429.8 person-years of follow-up (median follow-up of 4.8 years; interquartile range, 2.6–6.9 years; maximum 8.8 years), 7852 participants (men, 3549; women, 4303) developed new-onset binocular VI. Based on self-reports, current smoking was significantly associated with an increased incidence of bilateral VI, and this association was stronger in women (P for interaction = 0.01). After adjustments for potential confounders, multivariable adjusted hazard ratio (HR) and 95% confidence interval (CI) for incident VI comparing former and current smokers to never-smokers were 1.00 (0.92–1.10) and 1.14 (1.04–1.25), respectively, in men and 1.11 (1.00–1.23) and 1.52 (1.28–1.80), respectively, in women. In time-dependent models in which updated smoking status and confounders were treated as time-varying covariates, these associations were also observed. Increasing baseline smoking pack-years were positively associated with the incidence of bilateral VI in a dose–response manner in both men and women (P for trend < 0.05), and this association did not differ by sex (P for interaction = 0.098). For men, multivariable adjusted HR (95% CI) for incident VI comparing < 10, 10–19.9, and ≥ 20 pack-years to 0 pack-year were 0.98 (0.90–1.07), 1.11 (1.01–1.22) and 1.13 (1.01–1.23), respectively (P for trend = 0.008). For women, multivariable adjusted HR (95% CI) for incident VI comparing < 10, and ≥ 10 pack-years to 0 pack-years were 1.20 (1.05–1.37), and 1.28 (0.88–1.87), respectively (P for trend = 0.004). When introducing changes in smoking pack-years and confounders during follow-up as time-varying covariates, the association between pack-years and incident VI was similar in women but attenuated in men.
Urinary cotinine level, an objective marker of smoking, was associated with increased risk of incident VI in men and women (all P for trend < 0.05), with no significant interaction by sex (P for interaction = 0.742) (Table 4). Multivariable-adjusted HR (95% CI) for incident VI comparing urinary cotinine ≥ 100 ng/ml to < 50 ng/ml was 1.17 (1.08–1.27) in men and 1.22 (1.002–1.48) in women. In the time-dependent model, the association between urinary cotinine levels ≥ 100 ng/ml and incident VI was stronger than in the previous model.
In subgroup analyses (Supplementary Table S1), the association between smoking status and VI was consistently observed without significant interaction by age (< 50 vs. ≥ on years), alcohol intake (< 20 vs. ≥ 20 g/day), health-enhancing physical activity (no vs. yes), BMI (< 25 vs. 25 kg/m2), diabetes (no vs. yes), or hypertension (no vs. yes).
In sensitivity analyses using unilateral VI (either right or left VI) as an endpoint instead of bilateral VI, the association between smoking and VI was consistently observed (Supplementary Table S2).
Discussion
This large cohort study of young and middle-aged Korean men and women demonstrated that current smokers were at higher risk of incident VI than never smokers, and this association remained after adjustments for various confounding factors. These independent and positive associations were stronger in women. Smoking pack-years also showed a dose–response relationship with new-onset VI. In women, the risk increased even with relatively low pack-years (< 10 pack-years) compared to men. The lack of a significant positive association between highest pack-years (≥ 10 pack-years) with incidence of VI in women was probably due to insufficient sample size. Urinary cotinine level, an objective marker of smoking, was associated with increased risk of VI in both men and women, and these associations became evident when changes in urinary cotinine level and other confounders during follow-up were treated as time-varying covariates. Our findings suggest that cigarette smoking is an independent risk factor for VI and that women are more susceptible to the deleterious effects of smoking on vision than men.
A few studies evaluated the effects of smoking on VI. According to the Beaver Dam Eye Study, a prospective cohort study of 9548 individuals aged 43 years or older free of VI at baseline with a maximum follow-up of 20 years, current and former smoking were related to decreased vision, and incident VI was higher in current smokers compared to never-smokers with fully-adjusted odds ratios of 2.01 (95% CI 0.87–4.61)19. In a cross-sectional study of 12,233 Chinese participants, presenting VI was associated with smoking (odds ratio: 1.55; 95% CI 1.31–1.83), but this study did not include detailed information on smoking such as current vs. former smoking or smoking pack-years21. A cross-sectional study by Merle et al.22 using nationally representative French sample demonstrated that heavy smokers with ≥ 20 pack-years had a 1.48 (95% CI 1.03–2.14)-fold increase in the odds of presenting VI compared to never smokers, whereas no association with VI was observed among smokers with < 20 pack-years with a corresponding odds ratio of 0.98 (95% CI 0.72–1.32). Another cross-sectional study by Aljied et al.20 based on the Canadian Longitudinal Study on Aging included 30,097 adults aged between 45 and 85 years and showed that the odds (95% CI) of presenting VI in former and current smokers were 1.04 (0.93–1.17) and 1.52 (95% CI 1.25–1.85) compared to never smokers. However, previous studies were limited by cross-sectional design20,21,22, and lack of detailed information regarding smoking such as current vs. former smoking21 and smoking pack-years20,21. Additionally, all of these studies relied on self-reported smoking and did not incorporate changes in smoking status during follow-up. The present cohort study of relatively young men and women demonstrated positive and independent associations of both subjective and objective measures of cigarette smoking with the development of VI, while incorporating smoking status and confounders at both baseline and follow-up visits.
To date, the pathophysiology of smoking and the incidence of VI remain unclear, however, several explanations to ocular disease or effects on eyes after smoking are suggestive. Cigarette smoke is composed of thousands of hazardous compounds including toxic particles, gases, chemicals, and heavy metals28,29. Increased oxidative stress by reactive oxygen species directly and indirectly damage the lens, and heavy metals can accumulate on the lens, which causes cataracts even in young adults18,29. Oxidative stress due to free radicals also damages the retina and can cause age-related macular degeneration13,30. Smoking damages the optic nerve due to ischemic changes, and furthermore, smoking promotes inflammation that can aggravate the course of uveitis and thyroid-associated orbitopathy13. Besides the pre-existing ocular diseases, smoking may cause pathological changes in eyes by decreasing the choroidal vessel circulation31,32 and accelerating endothelial apoptosis, which diminishes the endothelial function of the cornea due to reactive oxygen species29, and DNA damage33. There are some reports of reduced contrast sensitivity associated with low serum levels of manganese and zinc34, and activated nicotinic acetylcholine receptors, the functional units of visual processing35, in heavy smokers.
In the present study, the associations between current smoking, smoking pack-years, and risk for developing VI were stronger in women, although only a small proportion (2.3%) of women were current smokers. In Korea, the overall prevalence of smoking in 2016 was 39.7% in men and 3.3% in women36, similar to our sample. Smoking prevalence remained unchanged for 10 years from 2003 in women, whereas that of men decreased consistently36. The mechanism underlying stronger associations of smoking with development of VI among women is not fully understood. Although controversial, there are a few studies suggesting that women are more susceptible to the development of smoking-related diseases than men37,38,39. Additionally, a previous study showed that women are less likely to quit smoking and more likely to transit from being a former smoker to a current smoker40. These differences among women might also be attributed to menstrual cycles and hormone variation41, greater nicotine dependence40,42, or lower level of education21. It is also well known that individuals with higher education levels tend to engage in more preventive care and are more likely to have healthy behaviors21. In this study, the prevalence of high education attainment was lower among women, which was the lowest in current women smokers. However, after adjustment for education level and other health behaviors, the association between smoking and development of VI remained significant. Further studies are required to confirm these findings and to elucidate the mechanism underlying potential sex differences in the associations between smoking and the risk of VI.
This study’s main strengths lie in its design as a large-scale cohort study of young and middle-aged men and women, and the inclusion of a wide range of confounders as well as detailed information on smoking status based on both subjective and objective measures of smoking status. These factors enabled us to demonstrate independent and dose–response associations of subjective and objective smoking with new-onset of VI and to identify differences between men and women while incorporating changes in smoking status and other covariates during follow-up.
There are several limitations in this study. First, data for best-corrected VA was not available. We defined VI based on presenting VA of participants whose refractive errors may have been partially corrected with their own glasses/lenses. However, presenting VA reflects the real vision of individuals in everyday life9 and is widely used in population-based and other clinical studies8,9,20. Second, the present study used both self-reported and an objective estimate of smoking status, however, the half-life of nicotine metabolism is about 16–20 h. Therefore, measurement errors may result in errors in estimates of actual smoking status of irregular smokers, or people who prevised the cotinine test and avoided smoking temporarily before health screening examination. Third, the history of ocular diseases was determined based on self-reporting of physician-diagnosed ocular diseases. Specific causes of VI were not addressed in this study. Thus, we cannot exclude some degree of residual confounding related to measurement errors and other unmeasured confounding factors, which might have affected the observed associations between smoking and incident VI. Fourth, environmental tobacco smoking was not considered. In Korea, the prevalence of environmental tobacco smoking in workplace was 57.2% among men and 38.7% among women in 201343. As second-hand smoking also increases incidence of diseases, such as lung cancer and deaths44, VI in non-smokers may have affected by environmental tobacco smoking. Fifth, the data on urinary creatinine concentrations were not available, limiting our ability to control for dilution effects45,46. Sixth, the exposure to occupational hazards such as organic solvents may affect as potential confounding factors to VI. Organic solvents affect the retina and optic nerve with neurotoxicity resulting color vision loss and lower visual function47,48. In our study, participants are mostly white-collar workers; thus the results are less likely to be affected by occupational hazards related to organic solvents. Additionally, nicotine metabolism can vary across ethnicity/race and our findings might not be generalizable to other race/ethnic groups. Finally, our study findings were derived from relatively healthy young and middle-aged educated Koreans with high accessibility to healthcare, and might not be generalizable to other populations with different characteristics.
In the present cohort study, both subjective and objective measures of smoking were positively and independently associated with an increased risk of development of VI in both men and women, with a stronger association in women. Current smoking was consistently associated with increased risk of VI, whereas former smoking was not. Our results suggest that smoking is an independent risk factor of decreased vision and that incidence of VI may be preventable by cessation of smoking or avoidance of smoking initiation. Tobacco control policies regarding health promotion and education would have important role to prevent VI in addition to other smoking-induced diseases. Further studies are needed to evaluate the exact pathogenesis of VI according to smoking and sex differences in biological and physiological effects of smoking on VI.
Methods
Study population
The Kangbuk Samsung Health Study is a cohort study of Korean adults who underwent comprehensive annual or biennial health examinations at the clinics of Kangbuk Samsung Hospital Total Healthcare Screening Center in Seoul and Suwon, South Korea49. In Korea, all employees are mandatory for health care by law under National Health Insurance. Eye examination with visual acuity is one of the essential items in basic health care in Korea. This study was designed to use data routinely collected as part of health screening examinations where questionnaires, blood tests, imaging examination, and procedures (e.g., ultrasound, endoscopy) are components of health screening exams49,50. More than 80% of the participants were employees of companies and local governmental organizations or their spouses. In South Korea, the Industrial Safety and Health Law requires annual or biennial health-screening exams of all employees to be offered free of charge. An office worker receives biennial health-screening examinations, and a blue-collar worker receives annual health-screening examinations. The remaining participants voluntarily paid for screening examinations at the health screening center.
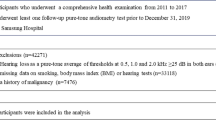
The present analysis included all study participants who received comprehensive health examinations including vision tests between 2011 and 2017, and who underwent at least one follow-up vision test prior to December 31, 2019 (N = 336,262; Fig. 1). A total of 57,193 participants (age range 18–89 years old) met one of the following exclusion criteria at baseline: missing data for smoking status, BMI, or VA (n = 33,529); history of cancer (n = 7476); VI defined as presenting VA < 0.5 (n = 12,449); or history of physician-diagnosed ocular diseases including cataracts, macular degeneration, glaucoma, and retinopathy (n = 8797). Some individuals met more than one exclusion criterion, and thus a total of 279,069 participants were included in the final analysis. The baseline characteristics according to missing visual acuity information is presented as Supplementary Table S3.
We followed all related tenets of the Declaration of Helsinki and this study was approved by the Institutional Review Board of Kangbuk Samsung Hospital (KBSMC 2020-04-025), which waived the requirement for written informed consent due to the use of de-identified data obtained as part of routine health screening examinations.
Data collection
Physical measurements, vision tests, and laboratory measurements of urine and blood were assessed every 1–2 years as part of the routine screening program at baseline and follow-up visits. Demographic characteristics, smoking habits and other lifestyle factors, and medical history were collected at each visit using standardized, self-administered questionnaires as previously described49. Physician-diagnosed eye diseases including cataracts, glaucoma, dry eye, macular degeneration, and any type of retinopathy were also assessed. Questions regarding smoking included lifetime and current smoking status, smoking duration, and number of cigarettes per day. Participants who had smoked < 100 cigarettes during their lifetime were classified as never-smokers. Participants who had smoked > 100 cigarettes in their lifetime were further categorized into two groups as follows: current smokers and former smokers who no longer smoked at all at the time of their screening examination. In Korea, 20 cigarettes are sold per pack. Pack-years were calculated as the product of smoking duration and cigarettes per day divided by 20. Alcohol consumption was categorized as none, < 20 g of ethanol/day, and ≥ 20 g of ethanol/day. The Korean version of the International Physical Activity Questionnaire Short Form was used to assess physical activity level51,52 which was classified into three categories: inactive, minimally active, and health-enhancing physically active as described previously53. Usual dietary intake over the past year was assessed using a Korean version of a 106-item self-administered food frequency questionnaire54. CVD and cancer were defined as physician-diagnosed heart disease or stroke and physician-diagnosed malignancy of any type, respectively.
Height, weight, and blood pressure (BP) were measured by trained nurses. Obesity was defined as BMI ≥ 25 kg/m2 according to Asian-specific criteria55. Hypertension was defined as systolic BP ≥ 140 mmHg, diastolic BP ≥ 90 mmHg, self-reported history of hypertension, or current use of antihypertensive medications.
Fasting blood measurements included glucose, glycated hemoglobin (HbA1c), lipid profiles, insulin, and high sensitivity C-reactive protein. Insulin resistance was assessed with the following HOMA-IR equation: fasting blood insulin (uU/ml) × fasting blood glucose (mmol/l)/22.5. Diabetes mellitus (DM) was defined as fasting serum glucose ≥ 126 mg/dl, HbA1c ≥ 6.5%, or current use of antidiabetic medications.
Urinary cotinine level was measured using the DRI Cotinine Assay (Microgenics Corp., Fremont, CA, USA) with a modular P800 chemistry analyzer (Roche Diagnostics, Tokyo, Japan). A urine cotinine cut-off point of 100 ng/ml is used as a reference point in the screening center of our hospital, since nonsmoker urine levels are reported not to exceed 100 ng/ml56. Urinary cotinine 50 ng/ml has also been widely used to distinguish tobacco use vs. no tobacco use according to the SRNT Subcommittee on Biochemical Verification57. Therefore, in the present study, urinary cotinine levels were categorized into three groups: (1) < 50 ng/ml; (2) 50–99 ng/ml; (3) ≥ 100 ng/ml.
Presenting VA with habitual correction if worn was assessed for each eye using an international standard chart that is based on Early Treatment Diabetic Retinopathy Study (ETDRS) scales at 3 m with distance. Presenting VA (either VA of naked eyes or corrected VA) was used because it represents the VA of individuals in the real world9. VA was recorded as a decimal, which is the usual unit in clinical practice in Korea, and then converted to Logarithm of the Minimum Angle of Resolution (logMAR) scale. For the main analysis, VI was defined as when VA was worse than 0.5 (cutoffs of 0.5; 20/40 Snellen; 0.3 logMAR) in both eyes19,20,22.
Statistical analysis
The baseline characteristics of the study participants are presented according to smoking status. The relationship between smoking status and incident VI differed significantly by sex; therefore, analyses were performed separately for men and women. Pack-years were categorized as never (0), > 0–10, > 10–20, and ≥ 20 pack-years. Few women participants (n = 64) were identified as having ≥ 20 pack-years and were thus combined with the category of > 10–20 years, resulting in a category of ≥ at pack-years in women.
The primary endpoint was the development of binocular VI, defined as VA < 0.5 in both eyes. The participants were followed from their baseline examination until either the development of binocular VI or the last health exam conducted prior to December 31, 2019, whichever came first. The incidence rate was calculated as incident cases divided by the number of person-years of follow-up. Since new-onset VI, if it did occur, would have occurred at an unknown time point between the visit at which VI was diagnosed by vision tests and the prior visit, a parametric proportional hazards model was used to account for this type of interval censoring (stpm command in Stata)58. In these models, the baseline hazard function was parameterized with restricted cubic splines in log time with four degrees of freedom.
The HR and 95% CI were calculated for incident VI according to smoking status, pack-years smoked, and urinary cotinine levels. Models were initially adjusted for age and then further adjusted for BMI, alcohol intake (0 g/day, < 20 g/day, ≥ 20 g/day, or unknown), physical activity level (inactive, minimally active, health-enhancing physically active, or unknown), education level (high school graduate or less, community college or university graduate, graduate school or more, or unknown), total calorie intake (in quintiles or missing), history of diabetes (no vs. yes), history of hypertension (no vs. yes) and history of cardiovascular disease (no vs. yes) (multivariable adjusted model). To evaluate the effects of changes in smoking status and other covariates over time during follow-up, we conducted additional analyses introducing smoking status and other covariates as time-varying covariates. The proportional hazards assumption was assessed by examining graphs of estimated log (− log) survival; ultimately, no violations of the assumption were found. To test for linear trends, we included the median value of each category (pack-years and urinary cotinine) as continuous variables in the models.
Subgroup analyses were performed by age (< 50 vs. ≥ 50 years), alcohol intake (< 20 vs. ≥ 20 g/day), health-enhancing physical activity (no vs. yes), BMI (< 25 vs. 25 kg/m2), diabetes (no vs. yes), and hypertension (no vs. yes). Interactions between smoking category and subgroup characteristics were tested using likelihood ratio tests that compared models with vs. without multiplicative interaction terms.
All analyses were carried out using STATA version 16.0 (Stata Corp LP, College Station, TX, USA). All P values less than 0.05 were considered to be statistically significant.
Data availability
All data generated or analyzed during this study are included in this published article.
References
Flaxman, S. R. et al. Global causes of blindness and distance vision impairment 1990–2020: A systematic review and meta-analysis. Lancet. Glob. Health 5, e1221–e1234. https://doi.org/10.1016/s2214-109x(17)30393-5 (2017).
Bourne, R. R. A. et al. Magnitude, temporal trends, and projections of the global prevalence of blindness and distance and near vision impairment: A systematic review and meta-analysis. Lancet. Glob. Health 5, e888–e897. https://doi.org/10.1016/S2214-109X(17)30293-0 (2017).
Kulmala, J. et al. Visual acuity and mortality in older people and factors on the pathway. Ophthalm. Epidemiol. 15, 128–134. https://doi.org/10.1080/09286580701840388 (2008).
Dhital, A., Pey, T. & Stanford, M. R. Visual loss and falls: A review. Eye (Lond., England) 24, 1437–1446. https://doi.org/10.1038/eye.2010.60 (2010).
Brown, G. C., Brown, M. M., Stein, J. D. & Smiddy, W. E. Vision-related quality of life associated with unilateral and bilateral ocular conditions. Ophthalmology 125, 965–971. https://doi.org/10.1016/j.ophtha.2017.12.033 (2018).
Park, H. Y., Ryu, H., Kang, H. Y., Lee, H. & Kwon, J. W. Clinical and economic burden of visual impairment in an aging Society of South Korea. Asia Pac. J. Public Health 27, 631–642. https://doi.org/10.1177/1010539515588944 (2015).
Zhang, T., Jiang, W., Song, X. & Zhang, D. The association between visual impairment and the risk of mortality: A meta-analysis of prospective studies. J. Epidemiol. Community Health 70, 836–842. https://doi.org/10.1136/jech-2016-207331 (2016).
Foong, A. W. et al. Visual acuity and mortality in a Chinese population. The Tanjong Pagar Study. Ophthalmology 115, 802–807. https://doi.org/10.1016/j.ophtha.2007.04.066 (2008).
Freeman, E. E., Egleston, B. L., West, S. K., Bandeen-Roche, K. & Rubin, G. Visual acuity change and mortality in older adults. Invest. Ophthalmol. Vis. Sci. 46, 4040–4045. https://doi.org/10.1167/iovs.05-0687 (2005).
Weih, L. M., VanNewkirk, M. R., McCarty, C. A. & Taylor, H. R. Age-specific causes of bilateral visual impairment. Arch. Ophthalmol. (Chicago, Ill: 1960) 118, 264–269. https://doi.org/10.1001/archopht.118.2.264 (2000).
Kenneth Walker, H., Dallas Hall, W. & Willis Hurst, J. Clinical Methods Ch*** 115 563–564 (Butterwirths, New York, 1990).
Lim, S. S. et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet (Lond., England) 380, 2224–2260. https://doi.org/10.1016/s0140-6736(12)61766-8 (2012).
Nita, M. & Grzybowski, A. Smoking and eye pathologies. A systemic review. Part II. Retina diseases, uveitis, optic neuropathies, thyroid-associated orbitopathy. Curr. Pharm. Des. 23, 639–654. https://doi.org/10.2174/1381612823666170111095723 (2017).
Evans, J. R., Fletcher, A. E. & Wormald, R. P. 28,000 Cases of age related macular degeneration causing visual loss in people aged 75 years and above in the United Kingdom may be attributable to smoking. Br. J. Ophthalmol. 89, 550–553. https://doi.org/10.1136/bjo.2004.049726 (2005).
Ying, G. S. et al. Baseline predictors for five-year visual acuity outcomes in the comparison of AMD treatment trials. Ophthalmol. Retina 2, 525–530. https://doi.org/10.1016/j.oret.2017.10.003 (2018).
Zhang, X. et al. Smoking and visual impairment among older adults with age-related eye diseases. Prev. Chron. Dis. 8, A84 (2011).
Lindblad, B. E., Hakansson, N. & Wolk, A. Smoking cessation and the risk of cataract: A prospective cohort study of cataract extraction among men. JAMA Ophthalmol. 132, 253–257. https://doi.org/10.1001/jamaophthalmol.2013.6669 (2014).
Kar, T., Ayata, A., Aksoy, Y., Kaya, A. & Unal, M. The effect of chronic smoking on lens density in young adults. Eur. J. Ophthalmol. 24, 682–687. https://doi.org/10.5301/ejo.5000454 (2014).
Klein, R., Lee, K. E., Gangnon, R. E. & Klein, B. E. Relation of smoking, drinking, and physical activity to changes in vision over a 20-year period: The Beaver Dam Eye Study. Ophthalmology 121, 1220–1228. https://doi.org/10.1016/j.ophtha.2014.01.003 (2014).
Aljied, R., Aubin, M. J., Buhrmann, R., Sabeti, S. & Freeman, E. E. Prevalence and determinants of visual impairment in Canada: Cross-sectional data from the Canadian Longitudinal Study on Aging. Can. J. Ophthalmol. 53, 291–297. https://doi.org/10.1016/j.jcjo.2018.01.027 (2018).
Yan, X., Chen, L. & Yan, H. Socio-economic status, visual impairment and the mediating role of lifestyles in developed rural areas of China. PLoS One 14, e0215329. https://doi.org/10.1371/journal.pone.0215329 (2019).
Merle, B. M. J. et al. Unhealthy behaviours and risk of visual impairment: The CONSTANCES population-based cohort. Sci. Rep. 8, 6569. https://doi.org/10.1038/s41598-018-24822-0 (2018).
Connor Gorber, S., Schofield-Hurwitz, S., Hardt, J., Levasseur, G. & Tremblay, M. The accuracy of self-reported smoking: A systematic review of the relationship between self-reported and cotinine-assessed smoking status. Nicotine Tobacco Res. 11, 12–24. https://doi.org/10.1093/ntr/ntn010 (2009).
Khang, Y. H., Yun, S. C., Cho, H. J. & Jung-Choi, K. The impact of governmental antismoking policy on socioeconomic disparities in cigarette smoking in South Korea. Nicotine Tobacco Res. 11, 262–269. https://doi.org/10.1093/ntr/ntn036 (2009).
Han, M. A. The price of tobacco and its effects on smoking behaviors in Korea: The 2015 Korea Community Health Survey. Prev. Med. 120, 71–77. https://doi.org/10.1016/j.ypmed.2019.01.010 (2019).
World Health Organization. Geneva: World Health Organization; 2017. WHO report on the Global Tobacco Epidemic. http://www.who.int/tobacco/surveillance/policy/country_profile/kor.pdf?ua=1.
Gunter, R., Szeto, E., Jeong, S. H., Suh, S. & Waters, A. J. Cigarette Smoking in South Korea: A narrative review. Korean J. Fam. Med. 41, 3–13. https://doi.org/10.4082/kjfm.18.0015 (2020).
Solberg, Y., Rosner, M. & Belkin, M. The association between cigarette smoking and ocular diseases. Surv. Ophthalmol. 42, 535–547. https://doi.org/10.1016/s0039-6257(98)00002-2 (1998).
Nita, M. & Grzybowski, A. Smoking and eye pathologies. A systemic review. Part I. Anterior eye segment pathologies. Curr. Pharm. Des. 23, 629–638. https://doi.org/10.2174/1381612822666161129152041 (2017).
Makrynioti, D., Zagoriti, Z., Koutsojannis, C., Morgan, P. B. & Lagoumintzis, G. Ocular conditions and dry eye due to traditional and new forms of smoking: A review. Contact Lens Anterior Eye J. Brit. Contact Lens Assoc. https://doi.org/10.1016/j.clae.2020.02.009 (2020).
Bettman, J. W., Fellows, V. & Chao, P. The effect of cigarette smoking on the intraocular circulation. AMA Arch. Ophthalmol. 59, 481–488. https://doi.org/10.1001/archopht.1958.00940050037002 (1958).
Ayhan, Z., Kaya, M., Ozturk, T., Karti, O. & HakanOner, F. Evaluation of macular perfusion in healthy smokers by using optical coherence tomography angiography. Ophthalm. Surg. Lasers Imaging Retina 48, 617–622. https://doi.org/10.3928/23258160-20170802-03 (2017).
Ballinger, S. W. et al. Mitochondrial genome damage associated with cigarette smoking. Cancer Res. 56, 5692–5697 (1996).
Uz, E. et al. The relationship between serum trace element changes and visual function in heavy smokers. Acta Ophthalmol. Scand. 81, 161–164. https://doi.org/10.1034/j.1600-0420.2003.00032.x (2003).
Fernandes, T. M. P., Almeida, N. L. & Santos, N. A. D. Effects of smoking and smoking abstinence on spatial vision in chronic heavy smokers. Sci. Rep. 7, 1690. https://doi.org/10.1038/s41598-017-01877-z (2017).
Chang, Y., Kang, H. Y., Lim, D., Cho, H. J. & Khang, Y. H. Long-term trends in smoking prevalence and its socioeconomic inequalities in Korea, 1992–2016. Int. J. Equity Health 18, 148. https://doi.org/10.1186/s12939-019-1051-x (2019).
Huxley, R. R. & Woodward, M. Cigarette smoking as a risk factor for coronary heart disease in women compared with men: A systematic review and meta-analysis of prospective cohort studies. Lancet (Lond., England) 378, 1297–1305. https://doi.org/10.1016/s0140-6736(11)60781-2 (2011).
Silverman, E. K. et al. Gender-related differences in severe, early-onset chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 162, 2152–2158. https://doi.org/10.1164/ajrccm.162.6.2003112 (2000).
Peters, S. A., Huxley, R. R. & Woodward, M. Do smoking habits differ between women and men in contemporary Western populations? Evidence from half a million people in the UK Biobank study. BMJ Open 4, e005663. https://doi.org/10.1136/bmjopen-2014-005663 (2014).
Smith, P. H., Bessette, A. J., Weinberger, A. H., Sheffer, C. E. & McKee, S. A. Sex/gender differences in smoking cessation: A review. Prev. Med. 92, 135–140. https://doi.org/10.1016/j.ypmed.2016.07.013 (2016).
Weinberger, A. H. et al. Systematic and meta-analytic review of research examining the impact of menstrual cycle phase and ovarian hormones on smoking and cessation. Nicotine Tobacco Res. 17, 407–421. https://doi.org/10.1093/ntr/ntu249 (2015).
Shiffman, S., Sweeney, C. T. & Dresler, C. M. Nicotine patch and lozenge are effective for women. Nicotine Tobacco Res. 7, 119–127. https://doi.org/10.1080/14622200412331328439 (2005).
Choi, S., Kim, Y., Park, S., Lee, J. & Oh, K. Trends in cigarette smoking among adolescents and adults in South Korea. Epidemiol. Health 36, e2014023. https://doi.org/10.4178/epih/e2014023 (2014).
Park, S. et al. Attributable fraction of tobacco smoking on cancer using population-based nationwide cancer incidence and mortality data in Korea. BMC Cancer 14, 406. https://doi.org/10.1186/1471-2407-14-406 (2014).
Biochemical verification of tobacco use and cessation. Nicotine Tobacco Res. 4, 149–159. https://doi.org/10.1080/14622200210123581 (2002).
Jatlow, P., McKee, S. & O’Malley, S. S. Correction of urine cotinine concentrations for creatinine excretion: Is it useful?. Clin. Chem. 49, 1932–1934. https://doi.org/10.1373/clinchem.2003.023374 (2003).
Jiménez Barbosa, I. A., Boon, M. Y. & Khuu, S. K. Exposure to organic solvents used in dry cleaning reduces low and high level visual function. PLoS One 10, e0121422. https://doi.org/10.1371/journal.pone.0121422 (2015).
Gong, Y. et al. Visual dysfunction in workers exposed to a mixture of organic solvents. Neurotoxicology 24, 703–710. https://doi.org/10.1016/s0161-813x(03)00034-2 (2003).
Chang, Y. et al. Metabolically healthy obesity and development of chronic kidney disease: A cohort study. Ann. Intern. Med. 164, 305–312. https://doi.org/10.7326/M15-1323 (2016).
Chang, Y. et al. Metabolically-healthy obesity and coronary artery calcification. J. Am. Coll. Cardiol. 63, 2679–2686. https://doi.org/10.1016/j.jacc.2014.03.042 (2014).
Craig, C. L. et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 35, 1381–1395. https://doi.org/10.1249/01.Mss.0000078924.61453.Fb (2003).
Chun, M. Y. Validity and reliability of Korean version of international physical activity questionnaire short form in the elderly. Korean J. Fam. Med. 33, 144–151. https://doi.org/10.4082/kjfm.2012.33.3.144 (2012).
Ryu, S. et al. Relationship of sitting time and physical activity with non-alcoholic fatty liver disease. J. Hepatol. 63, 1229–1237. https://doi.org/10.1016/j.jhep.2015.07.010 (2015).
Ahn, Y. et al. Validation and reproducibility of food frequency questionnaire for Korean genome epidemiologic study. Eur. J. Clin. Nutr. 61, 1435–1441. https://doi.org/10.1038/sj.ejcn.1602657 (2007).
Wen, C. P. et al. Are Asians at greater mortality risks for being overweight than Caucasians? Redefining obesity for Asians. Public Health Nutr. 12, 497–506. https://doi.org/10.1017/s1368980008002802 (2009).
Haufroid, V. & Lison, D. Urinary cotinine as a tobacco-smoke exposure index: A minireview. Int. Arch. Occup. Environ. Health 71, 162–168. https://doi.org/10.1007/s004200050266 (1998).
Verification, S. S. O. B. Biochemical verification of tobacco use and cessation. Nicotine Tobacco Res. 4, 149–159. https://doi.org/10.1080/14622200210123581 (2002).
Royston, P. & Parmar, M. K. Flexible parametric proportional-hazards and proportional-odds models for censored survival data, with application to prognostic modelling and estimation of treatment effects. Stat. Med. 21, 2175–2197. https://doi.org/10.1002/sim.1203 (2002).
Acknowledgements
We would like to thank our staff members at the Kangbuk Samsung Health Study for their hard work, dedication, and continuing support.
Author information
Authors and Affiliations
Contributions
Concept and design: S.R. Acquisition, analysis, or interpretation of data: S.Y.H., Y.C. Drafting of the manuscript: S.Y.H., S.R. Critical revision of the manuscript for important intellectual content: all authors. Statistical analysis: Y.C., S.R. Administrative, technical, or material support: H.S., C.Y.C. Supervision: C.Y.C., S.R.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Han, S.Y., Chang, Y., Shin, H. et al. Smoking, urinary cotinine levels and incidence of visual impairment. Sci Rep 11, 398 (2021). https://doi.org/10.1038/s41598-020-79865-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-79865-z
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.