Abstract
Reward motivation is known to enhance cognitive control. However, detrimental effects have also been observed, which have been attributed to overdosing of already high baseline dopamine levels by further dopamine increases elicited by reward cues. Aarts et al. (2014) indeed demonstrated, in 14 individuals, that reward effects depended on striatal dopamine synthesis capacity, measured with [18F]FMT-PET: promised reward improved Stroop control in low-dopamine individuals, while impairing it in high-dopamine individuals. Here, we aimed to assess this same effect in 44 new participants, who had previously undergone an [18F]DOPA-PET scan to quantify dopamine synthesis capacity. This sample performed the exact same rewarded Stroop paradigm as in the prior study. However, we did not find any correlation between reward effects on cognitive control and striatal dopamine synthesis capacity. Critical differences between the radiotracers [18F]DOPA and [18F]FMT are discussed, as the discrepancy between the current and our previous findings might reflect the use of the potentially less sensitive [18F]DOPA radiotracer in the current study.
Similar content being viewed by others
Introduction
Incentive motivation, or motivation activated by external reward cues, is generally thought to enhance cognitive control1,2 and performance-contingent rewards are common across various domains of our society, including sports, education and the workplace. However, negative effects of rewards on cognitive control have also been observed3,4,5,6,7,8,9. For example, it has been demonstrated that when participants received performance-contingent payment for completing various tasks—including tasks that drew primarily on motor skills, memory and creativity—high reward levels had detrimental effects on performance, compared with low and medium reward levels8. The authors argued that high reward levels can shift arousal or motivation levels beyond the optimal level for executing a task, leading to performance decrements, an effect known as choking. However, not everyone chokes under high reward conditions and to gain more insight into this individual variation we must unravel the neural mechanisms underlying these choking effects. Motivational effects have long been associated with striatal dopamine signaling10,11,12 and prior work indeed suggested that individual variation in the effects of motivation on cognitive control depends on dopamine-related functioning, such as dopamine cell loss in Parkinson’s disease, midbrain and striatal BOLD activity, loss aversion and dopamine transporter genotype3,5,6,9,13,14. Detrimental effects of rewards resonate with the notion of a potential overdosing of the dopamine system: Rewards, eliciting dopamine release15, could have beneficial effects in individuals with low dopamine levels by inducing a shift from sub-optimal to optimal dopamine levels, but detrimental effects in individuals with already high dopamine levels by inducing a shift from optimal to supra-optimal dopamine levels16. Building on this work, our previous study17 directly addressed this issue by assessing the effect of reward on cognitive control as a function of dopamine synthesis capacity, measured with 6-[18F]-fluoro-L-m-tyrosine ([18F]FMT) positron emission tomography (PET). Specifically, participants performed a Stroop task after being promised either a high or a low monetary reward upon successful completion of the task. These monetary incentives were demonstrated to enhance Stroop interference control in participants with low baseline dopamine synthesis capacity in the left caudate nucleus, but impair Stroop interference control in participants with high baseline dopamine synthesis capacity in the left caudate nucleus. This study thus advanced our understanding of differential effects of incentives on cognitive control, by demonstrating that incentive motivation can shift dopamine levels to supra-optimal in participants with already high dopamine levels. It is of note that this effect was present only when participants were uninformed (i.e., un-cued) about the congruency of the upcoming Stroop target and not when Stroop targets were preceded by cues informing subjects about their congruency.
The finding that a negative correlation between reward effects and dopamine synthesis capacity was present specifically in the left caudate nucleus strengthened evidence from two other prior studies, implicating specifically the left caudate nucleus in the effects of the dopamine transporter gene DAT1 during rewarded cognitive control3,18. Moreover, this finding generally concurred with evidence from an fMRI study demonstrating enhanced connectivity between the ventral striatum and left caudate nucleus when cognitive demand for reward was high19. The focus of the effect on the caudate nucleus also converged with functional MRI work from a third research group demonstrating a modulation by reward incentives of specifically the caudate nucleus during (oculomotor) control20. Finally, confidence in a negative correlation between individual differences in baseline dopamine levels and reward effects on cognitive control was further increased following a subsequent study in Parkinson’s disease patients, revealing greater beneficial effects of reward on cognitive control in patients with greater dopamine cell loss, measured with CIT-SPECT13.
However, the sample size (n = 14) of the key PET study providing the direct evidence for baseline-dependency of reward effects on cognitive control in healthy volunteers was very small for a between-subject correlational design. Such a small sample size is associated not only with low positive predictive value21, but also with high likelihood that effect sizes are biased and overestimated22. Therefore, we here aimed to test the effect found by Aarts et al. using a new, larger participant sample, who had already, as part of a previous study, undergone a PET scan with the radiotracer [18F]fluoro-3,4-dihydroxyphenyl-L-alanine ([18F]FDOPA). Specifically, we hypothesized that the effect of anticipated reward on Stroop interference control depends on individual differences in baseline dopamine synthesis capacity in the left caudate nucleus. We supplemented the analyses with voxel-wise correlations of the behavioral measures with dopamine synthesis capacity. Critically, striatal dopamine synthesis capacity in this new sample had already been indexed in the context of a previous study not reported here (www.trialregister.nl/trial/5959; data of which were previously included in Westbrook et al.23 and Hofmans et al.24), using [18F]DOPA PET. This is unlike the original study, in which [18F]FMT PET was used to index striatal dopamine synthesis capacity. While [18F]DOPA PET is considered less sensitive than [18F]FMT PET, due to increased background noise and cell clearance of radiolabeled metabolites, we anticipated that a strong association between striatal dopamine synthesis capacity and motivational effects on cognitive control should surface also with [18F]DOPA PET.
The present attempt at conceptual replication was driven by our goal to increase our confidence in the role of dopamine synthesis capacity in motivated cognitive control and is of particular interest because a robust mechanistic account of the link between incentive motivation and cognitive control will advance our understanding of who chokes under high reward conditions and why25, a topic of great societal relevance today. A preregistration of this study, data and code are available via https://osf.io/ky9s2/.
Materials and methods
Participants
Forty-five (out of a total of 94) right-handed and native Dutch-speaking volunteers who had participated in a previous [18F]DOPA PET study (protocol NL57538.091.16; trial register NTR6140, http://www.trialregister.nl/trial/5959) accepted the invitation to participate in the current study. All participants gave written informed consent according to the declaration of Helsinki and the experiment was conducted in compliance with and was approved by the local ethics committee (CMO Arnhem-Nijmegen, The Netherlands; Imaging Human Cognition, CMO 2014/288, version 2.2). One dataset was excluded due to an error rate above 33% [36%; mean (SD) = 18 (7) %). With the resulting 44 participants (aged: 19–45 years, mean (SD) = 24 (5.8); 22 women] we adhered to Simonsohn26 recommendation to obtain a sample size at least 2.5 times larger than the original sample size (N = 14). The new sample had 90% power27 to detect a correlation of r = 0.55, which is considerably lower than the correlation of r = 0.75 reported in the original study (two-sided α = 0.0042, see “Data analysis”). The time between the PET scan and this behavioral study ranged between 0.3 and 1.8 years [mean (SD) = 1.0 (0.4)], which is substantially shorter than in the original study [range: 1.0–4.2 years, mean (SD) = 2.3 (1.1)]. Background neuropsychological tests (listening span and behavioral inhibition/activation) had been assessed in the prior [18F]DOPA PET study.
Behavioral paradigm
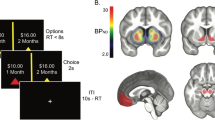
Participants completed the exact same paradigm as in Aarts et al.: a rewarded word-arrow Stroop paradigm, where they responded with a left or right button press to the words “left” or “right” in a left or right pointing arrow, using their right index finger or right middle finger, respectively (Fig. 1a). The direction indicated by the word could either be congruent (same direction as the arrow) or incongruent (opposite direction). Each trial was preceded by a reward cue for a duration of 1–2 s, which indicated either a high (15 cents) or low (1 cent) reward that would be earned on that trial if the participant responded correctly and within the response window. After the reward cue, an information cue was shown on the screen for 1–2 s which was either informative, in which case it announced to the participant whether the trial would be congruent (green circle) or incongruent (red cross), or uninformative, in which case it showed a question mark. The information cues were added in the original study to assess potential anticipatory reward effects on proactive control, i.e. the ability to prepare for the upcoming congruent and incongruent Stroop targets (without being able to prepare a left or right motor response). Reward cues, information cues and congruency were equally divided across 240 trials, which lasted about 30 min.
Schematic of data acquisition. (a) Schematic of an example word-arrow Stroop trial. Participants could either earn a high (15 cents) or low (1 cent) reward for a correct answer within the response window, which was cued at the start of the trial (1–2 s). After that, an information cue was presented for 1–2 s, indicating a congruent (green circle) or incongruent (red cross) trial, or giving no information about the upcoming congruency (grey question mark). Upon seeing the word-arrow Stroop target, participants had to respond to the word with a left or right button-press within the dynamically set response window. (b) Coronal view of our regions of interest including the bilateral caudate nucleus (red), putamen (green) and ventral striatum (blue).
As in the original study, before the actual task, participants completed 3 practice blocks. The first one to familiarize them with the information cues (12 trials), the second one to familiarize them with the reward cues (32 trials), and a third one—similar to the actual experiment—to set the initial response windows for the different trial types (48 trials). The initial response windows were set as the average response time per trial type (high or low reward; informed or uninformed; congruent or incongruent). During the actual experiment, the response windows were adapted after correct responses within the time window (-25 ms) or too late responses (+ 25 ms). After every block of 30 trials participants received feedback on their performance, showing their obtained reward on that block, the maximum reward that could have been obtained, their misses (too late), their errors (in time but wrong) and their reward for the total experiment so far, which remained on screen for 15 s. Due to the dynamic response windows, obtained reward was similar across participants and trial types (mean (SD) = €9.31 (0.95) for the entire experiment). The task was performed on a computer running on Windows 7 and a screen resolution of 1920 × 1080p, and the stimuli were shown using Presentation (version 20.2, 2018).
PET acquisition
PET scans were carried out at the Department of Nuclear Medicine of the Radboud university medical center, using a Siemens PET/CT scanner and the radiotracer [18F]DOPA. Participants received 150 mg of carbidopa and 400 mg of entacapone 50 min before scanning, to minimize peripheral metabolism of [18F]DOPA and thereby increase central [18F]DOPA availability. The procedure started with a low dose CT scan (approximately 0.75 mCi) followed by a bolus injection of [18F]DOPA into an antecubital vein and an 89 min dynamic PET scan (approximately 5 mCi). The data were divided into 24 frames (4 × 1, 3 × 2, 3 × 3, 14 × 5 min) and reconstructed with weighted attenuation correction and time-of-flight recovery, scatter corrected, and smoothed with a 3 mm full-width-at-half-maximum (FWHM) kernel.
Structural MRI
A high-resolution anatomical scan, T1-weighted MP-RAGE sequence (repetition time = 2,300 ms, echo time = 3.03 ms, 192 sagittal slices, field of view = 256 mm, voxel size 1 mm isometric) was acquired using a Siemens 3 T MR scanner with a 64-channel coil. These were used for coregistration and spatial normalization of the PET scans.
PET analysis
PET data were preprocessed and analyzed using SPM12 (https://www.fil.ion.ucl.ac.uk/spm/). All frames were realigned for motion correction and coregistered to the anatomical MRI-scan, using the mean PET image of the first 11 frames. Dopamine synthesis capacity was computed as the [18F]DOPA influx constant per minute (Ki) per voxel relative to the grey matter of the cerebellum, using Gjedde-Patlak graphical analysis28. The individual cerebellum grey matter masks were obtained by segmenting the individuals’ anatomical MRI scan, using Freesurfer (https://surfer.nmr.mgh.harvard.edu/). The Ki values were calculated based on the PET frames from the 24th to 89th minute. We then extracted average Ki values from six regions of interest (ROIs)—left and right caudate nucleus, putamen and ventral striatum—defined using masks based on an independent functional connectivity-analysis of the striatum29 (Fig. 1b). These ROIs are different from the ROIs used by Aarts et al.17, which were specified according to guidelines described by Mawlawi et al.30. An overlay of the two sets of ROIs are displayed in Supplementary Fig. S1. Supplementary analyses reveal a high Pearson correlation coefficient between the mean Ki values extracted from the two sets of ROIs (all r > 0.96). Analyses assessing the relationship between dopamine synthesis capacity in the left caudate nucleus as specified according to Mawlawi et al. and the effect of reward on Stroop interference can be found in the Supplementary information, including Supplementary Fig. S2). For voxel-wise group analyses, the Ki maps were normalized to MNI space and smoothed using an 8 mm FWHM kernel.
Data analysis
We expected a linear relationship between dopamine synthesis capacity and the effect of reward on Stroop interference. This prediction derives from the hypothesis that there is a negative quadratic relationship between dopamine signaling and cognitive performance16, such that both too little and too much dopamine is detrimental for performance: in low-dopamine participants, a putative increase in dopamine release in response to the promise of reward will positively affect performance by shifting dopamine levels from suboptimal to optimal. Conversely, in high-dopamine participants, the same reward promise will negatively affect performance by shifting dopamine levels from optimal to supra-optimal. See Supplementary Figs. S3 and S4 for an exploration of nonlinear relationships between dopamine signaling and cognitive performance.
The main effect of interest was the correlation between the effect of reward on Stroop interference (in terms of response times) on uninformed trials and dopamine synthesis capacity in the left caudate nucleus, as was observed in the original study. For completeness, we also explored the other five ROIs. We analyzed response times (RTs) of all correct trials, including trials on which participants were “too late”, and error rates. Participants with error rates above 33% were excluded. We ran separate repeated measures analyses of variance (rmANOVA) for each region of interest and two dependent variables: Stroop interference on RT and on error rate (mean RT or error rate on incongruent trials minus mean RT or error rate on congruent trials). The within-subjects factors were REWARD (low, high) and INFORMATION (uninformed, informed), and [18F]DOPA Ki in the left or right caudate nucleus, putamen, or ventral striatum was a covariate of interest. The analyses were performed using the ezANOVA function from the ez package31 in R (version 3.4.2). We corrected for multiple comparisons (6 ROIs, 2 dependent variables), resulting in a Bonferroni-corrected alpha value of 0.0042. Pearson’s correlations were calculated between the Ki values of the six ROIs and the effect of reward on Stroop interference in terms of RT on uninformed trials for comparison with the original study17. We supplemented the analyses with voxel-wise correlations between the reward effect on Stroop interference and dopamine synthesis capacity within the voxels comprising the entire striatum (the sum of the 6 regions of interest, specified above). Statistical significance was defined as family-wise error corrected p < 0.05 at peak coordinate, after small volume correction for all voxels within the striatal region of interest.
Although striatal [18F]DOPA uptake shows high test–retest reliability within a time frame of 2 years32, we performed additional regression analyses, separately for each of the six ROIs, to assess whether any effects of the interaction between REWARD and dopamine synthesis capacity on Stroop interference depended on time between the PET scan and the behavioral testing day, while also including age and gender in the model, using the lm function from the stats package in R.
We could not directly compare baseline dopamine synthesis capacity between the original and the current study, because the PET tracer differed between the two studies. However, to appreciate possible differences between the main findings of the current study and that of the original study, it is important to analyze comparability of the sample (Table 1). We therefore compared the two samples in terms of age, neuropsychological assessment (listening span and behavioral inhibition / activation) and overall performance in terms of error rates and RT, using Welch’s t-tests. We then compared reward effects on Stroop interference in terms of RT between the two studies, with the hypothesis that reward would decrease interference in individuals with lower baseline dopamine synthesis capacity and increase interference in individuals with higher baseline dopamine synthesis capacity16,17. We assessed differences in mean using a Welch’s t-test and differences in variances using a Levene’s test. Moreover, given the well-established link between dopamine and response vigor11,12,33, we assessed the effect of baseline dopamine synthesis capacity on response times, both the main effect and in interaction with reward, in the current and the original study using an rmANOVA (Supplementary Tables S1, S2).
To allow for quantification of evidence for or against our hypotheses, we additionally report Bayesian individual effects analyses performed in JASP (version 0.10.2.0), with default JASP Cauchy priors. The BFinclusion reflects how strongly the data support inclusion of a factor. We performed a sequential Bayesian correlation to illustrate evidence accumulation against the previously found correlation between the effect of reward on Stroop interference and dopamine synthesis capacity in the left caudate nucleus after observing the new data. Data from both studies were included; dopamine synthesis capacity values were separately standardized (z-scored) for both [18F]DOPA and [18F]FMT Ki values.
All continuous independent variables ([18F]DOPA Ki values, time between the PET and behavioral session, overall response times and age) were mean centered.
Results
Participants performed more poorly on incongruent than congruent trials (RT: F(1,43) = 185.3, p = 3.438e−17, BFINC = 3.217e+14; error rate: F(1,43) = 137.7, p = 5.394e−15, BFINC = 6.434e+13), on uninformed than informed trials (RT: F(1,43) = 35.0, p = 4.829e−7, BFINC = 3.217e+14; error rate: F(1,43) = 34.1, p = 6.224e−7, BFINC = 4.949e+13) and low reward than high reward trials (RT: F(1,43) = 14.2, p = 4.974e−4, BFINC = 3.217e+14; error rate: F(1,43) = 0.2, p = 0.662, BFINC = 4.949e+13). These results validate the task manipulation, and they are similar to findings in the original study.
Crucially, and in contrast with the original study, there was no interaction effect between REWARD, INFORMATION and dopamine synthesis capacity in any of the six ROIs on Stroop interference in terms of response times or error rates (Table 2). For completeness, we also report the effect of reward on Stroop interference independent of the factor INFORMATION (Table 2). Pearson’s correlations between baseline dopamine synthesis capacity and the effect of reward on Stroop interference on uninformed trials only revealed no significant associations (all r <|0.22|, p > 0.158, BF < 0.575; Fig. 2). Importantly, the 95% confidence interval for the correlation between dopamine synthesis capacity in the left caudate nucleus and the effect of reward on Stroop interference in the present study (r = − 0.06, p = 0.700, 95% CI [− 0.35, 0.24]) did not overlap with that of the originally reported effect of r = 0.75. Upon visual inspection of Fig. 2, we additionally explored a quadratic relationship between dopamine synthesis capacity in the left and right caudate nucleus and the effect of motivation on Stroop interference in terms of RT on uninformed trials, but did this not yield significant results (page 9 of the Supplementary information). Moreover, a supplementary rmANOVA and Pearson’s correlation analysis revealed no relationship between dopamine synthesis capacity in the left caudate nucleus as specified according to Mawlawi et al.30 and used in Aarts et al.17 and the effect of reward on Stroop interference (Supplementary Fig. S2).
The effect of reward on Stroop interference (RT: incongruent–congruent) on uninformed trials plotted as a function of dopamine synthesis capacity in the six ROIs. Shaded area around the regression line represents 95% confidence interval. N = 44.; RT (ms) = response time in milliseconds; Ki = [18F]DOPA uptake, reflecting dopamine synthesis capacity. Effect in grey was the correlation observed in Aarts et al. to be significant.
Voxel-wise analyses of the effect of reward on Stroop interference on uninformed trials confirmed the lack of significant correlations with any of the voxels within the striatum (Fig. 3). Separate multiple regression analyses for each ROI further confirmed the lack of a significant interaction between REWARD, INFORMATION and dopamine synthesis capacity or between REWARD and dopamine synthesis capacity on Stroop interference in terms of RT or error rate (Table 3). Additionally, time between PET and behavioral testing, age and gender did not affect the interaction between REWARD, INFORMATION and dopamine synthesis capacity or the interaction between REWARD and dopamine synthesis capacity on Stroop interference in terms of RT or error rate (Table 3). To further illustrate evidence against a correlation between the effect of REWARD on Stroop interference and dopamine synthesis capacity in the left caudate nucleus on uninformed trials, we ran a sequential Bayesian correlation including the data from both the original study and the current study. This revealed a strong increase in evidence in favor of a correlation when including participants from the original study, followed by a strong decline in evidence when including participants from the current study, culminating in moderate evidence against a correlation (Fig. 4).
Association of baseline dopamine synthesis capacity with the effect of reward on Stroop interference on uninformed trials. Voxels showing a positive (red) or negative (blue) regression coefficient on the effect of a promised reward on Stroop interference in terms of response times on uninformed trials. Dual-coded and simultaneously displaying the contrast estimate (x axis) and t values (y axis). The hue indicates the size of the contrast estimate, and the opacity indicates the height of the t value. The z coordinates correspond to the standard MNI brain. No voxels survive p < 0.05 peak-level corrected (FWE) or p < 0.001 uncorrected. Plotted using a procedure introduced by Allen et al.44 and implemented by Zandbelt45.
Sequential analysis showing progression of the Bayes Factor (BF) as new participants (n) enter the analysis. Values above 1 represent evidence for a correlation between dopamine synthesis capacity in the left caudate nucleus and a motivation effect on Stroop interference on uninformed trials, whereas values below 1 represent evidence against a correlation. Each dot represents the BF after inclusion of the next participant. Dark grey dots represent the 14 participants from Aarts et al. entered first; light grey dots represent the 44 participants from the current study. Order of including participants within each group was at random.
Average age differed significantly between the original and the current study (original study: mean = 28.1 years old; current study: mean = 24.3 years old; t(47) = -3.4, p = 0.001; Table 1). To assess whether this could have caused the lack of effect of interest in the current study, we repeatedly discarded the youngest participant from our current dataset until age no longer differed between the studies, before rerunning the rmANOVAs. This resulted in a dataset including 26 participants (mean age = 26.9 years old; t(37) = -0.9, p = 0.379). However, we did not observe a significant REWARD (by INFORMATION) by dopamine synthesis capacity interaction effect on Stroop interference (Supplementary Table S3).
Similarly, individual average RTs across trials differed significantly between the original and the current study (original study: mean = 397.5 ms; current study: mean = 346.9 ms; t(40) = -4.1, p = 2.0e−4; Table 1). We therefore repeatedly discarded the fastest participant from our current dataset until the average RTs no longer differed, resulting in a dataset including 29 participants (mean RT = 371.8; t(39) = -1.9, p = 0.064). However, we did not observe a significant REWARD (by INFORMATION) by dopamine synthesis capacity effect on Stroop interference (Supplementary Table S4). We additionally ran a multiple linear regression for each ROI including the terms REWARD, INFORMATION, dopamine synthesis capacity and individual average RT across all trials, including all interactions, which confirmed the lack of a significant effect of average RTs (Supplementary Table S5).
To establish that the discrepancy between the studies does not reflect differences in the dynamic range of the key variable of interest, we also compared the means and variances of the reward effects on Stroop interference on uninformed trials in terms of RT between the two studies. The two participant samples did not differ significantly from each other in terms of their means and variances (Fig. 5), as revealed by a Welch’s t-test (original study: mean = 0.07 ms; current study: mean = − 3.2 ms; t(20) = − 0.3, p = 0.767; Table 1) and Levene’s test (F(1,56) = 0.2, p = 0.660), respectively.
The effect of promised reward on Stroop interference (RT incongruent trials minus RT congruent trials) on uninformed trials in the original study (Aarts et al.) and the current replication attempt. Individual data points and probability distribution. Error bars represent 95% confidence interval around the mean. Plotted using R_rainclouds.R46.
Discussion
The current study reveals no evidence for an interaction between monetary incentives and dopamine synthesis capacity, indexed with [18F]DOPA PET, on Stroop interference. Bayesian analyses in fact provide evidence in favor of a lack of a relationship between dopamine synthesis capacity and reward effect on Stroop interference. Our conclusion is therefore not consistent with the earlier findings by Aarts et al.17.
It is possible that the discrepancy between the findings of the two studies reflects the use of [18F]DOPA in the present study, as opposed to [18F]FMT used in the original study. [18F]DOPA is a substrate for catechol-O-methyltransferase (COMT) in the periphery. Metabolites can cross the blood–brain-barrier and will distribute evenly throughout the brain, enhancing background noise relative to the use of [18F]FMT, which is not a substrate for COMT34. However, this is mainly a concern when one is interested in brain areas with low dopamine levels, as opposed to the dopamine-rich striatum. Moreover, entacapone was administered before PET scanning to inhibit peripheral COMT metabolism, further reducing the risk of a too low signal-to-noise ratio. [18F]DOPA and [18F]FMT also differ in their metabolic actions after decarboxylation by aromatic amino acid decarboxylase (AAAD), including higher affinity of [18F]DOPA metabolites compared with [18F]FMT metabolites for the vesicular monoamine transporter, leading to increased cell clearance of radiolabeled [18F]DOPA metabolites35. Indeed, differences in aging effects on dopamine synthesis capacity measured with [18F]DOPA and [18F]FMT have been observed, possibly owing to age-related changes in post-AADC metabolism36. However, this would mostly be a concern for extended scanning times, as [18F]DOPA behaves as an irreversibly bound tracer in the first 90 min after tracer injection, during which their uptake rates are tightly correlated34,35.
Another possibility is that the discrepancy between the original and the current study was introduced by group differences in sample characteristics. However, differences in overall response times and age did not explain the lack of significant effects in the current study. According to the dopamine overdose hypothesis16, monetary incentives might enhance Stroop interference control in participants with very low average levels of baseline dopamine, whereas those incentives would impair control in participants with very high average levels. Sampling only participants with intermediate dopamine levels should lead to very small reward effects. However, a comparison of reward effects between the two studies demonstrated similar means and variances within the two samples. We therefore argue that the current result decreases our belief in the previously observed correlation between motivational effects on cognitive control and baseline dopamine synthesis capacity.
Notably, this conclusion would not imply that dopamine transmission is not important for the motivation of cognitive control, because brain dopamine levels are a function not only of dopamine synthesis capacity, but also of other factors, including transporter density, dopamine receptor availability, dopamine release and genetic make-up. Thus, the current study cannot refute hypothesized correlations between motivational effects on cognitive control and other measures of dopamine function. For example, the current design does not disconfirm previously demonstrated and replicated links between motivation, cognitive control and polymorphisms in the dopamine transporter gene3,14,18, dopamine release37 or dopamine-related disease status13,38,39. Similarly, the current failure to replicate does not undermine other studies demonstrating a link between dopamine synthesis capacity and cognitive motivation indexed with other tasks, such as delay discounting40, cognitive effort discounting23,24 or reward-based reversal learning41. Nevertheless, the presently observed lack of effect reduces our confidence in the link between dopamine synthesis capacity and the effect of a promised reward on Stroop interference and stresses the need for further studies.
Data availability
The data and analysis scripts used in this article will be made publicly available after manuscript acceptance at the following web address: https://doi.org/10.34973/s0fm-3e10. Prior to accessing and downloading the shared data, users must create an account. It is possible to use an institutional account or a social ID from Google, Facebook, Twitter, LinkedIn or Microsoft. After authentication, users must accept the Data Use Agreement (DUA), after which they are automatically authorized to download the shared data. The DUA specifies whether there are any restrictions on how the data may be used. The Radboud University and the Donders Institute for Brain, Cognition and Behaviour will keep these shared data available for at least 10 years.
References
Krawczyk, D. C., Gazzaley, A. & D’Esposito, M. Reward modulation of prefrontal and visual association cortex during an incentive working memory task. Brain Res. 1141, 168–177 (2007).
Pessoa, L. & Engelmann, J. B. Embedding reward signals into perception and cognition. Front. Neurosci. 4, 17 (2010).
Aarts, E. et al. Striatal dopamine mediates the interface between motivational and cognitive control in humans: evidence from genetic imaging. Neuropsychopharmacology 35, 1943–1951 (2010).
Aarts, E., van Holstein, M. & Cools, R. Striatal dopamine and the interface between motivation and cognition. Front. Psychol. 2, 163 (2011).
Chib, V. S., De Martino, B., Shimojo, S. & O’Doherty, J. P. Neural mechanisms underlying paradoxical performance for monetary incentives are driven by loss aversion. Neuron 74, 582–594 (2012).
Mobbs, D. et al. Choking on the money. Psychol. Sci. 20, 955–962 (2009).
Zedelius, C. M., Veling, H. & Aarts, H. Boosting or choking: how conscious and unconscious reward processing modulate the active maintenance of goal-relevant information. Conscious. Cogn. 20, 355–362 (2011).
Ariely, D., Gneezy, U., Loewenstein, G. & Mazar, N. Large stakes and big mistakes. Rev. Econ. Stud. 76, 451–469 (2009).
Chib, V. S., Shimojo, S. & O’Doherty, J. P. The effects of incentive framing on performance decrements for large monetary outcomes: behavioral and neural mechanisms. J. Neurosci. 34, 14833–14844 (2014).
Mohebi, A. et al. Dissociable dopamine dynamics for learning and motivation. Nature 570, 65–70 (2019).
Robbins, T. W. & Everitt, B. J. A role for mesencephalic dopamine in activation: commentary on Berridge (2006). Psychopharmacology 191, 433–437 (2007).
Salamone, J. D., Yohn, S. E., López-Cruz, L., San Miguel, N. & Correa, M. Activational and effort-related aspects of motivation: neural mechanisms and implications for psychopathology. Brain 139, 1325–1347 (2016).
Aarts, E. et al. Aberrant reward processing in Parkinson’s disease is associated with dopamine cell loss. Neuroimage 59, 3339–3346 (2012).
van Holstein, M. et al. Human cognitive flexibility depends on dopamine D2 receptor signaling. Psychopharmacology. 218, 567–578 (2011).
Schultz, W. Dopamine neurons and their role in reward mechanisms. Curr. Opin. Neurobiol. 7, 191–197 (1997).
Cools, R. & D’Esposito, M. Inverted-U-shaped dopamine actions on human working memory and cognitive control. Biol. Psychiatry 69, e113–e125 (2011).
Aarts, E. et al. Dopamine and the cognitive downside of a promised bonus. Psychol. Sci. 25, 1003–1009 (2014).
Aarts, E. et al. Reward modulation of cognitive function in adult attention-deficit/hyperactivity disorder: a pilot study on the role of striatal dopamine. Behav. Pharmacol. 26, 227–240 (2015).
Schmidt, L., Lebreton, M., Cléry-Melin, M. L., Daunizeau, J. & Pessiglione, M. Neural mechanisms underlying motivation of mental versus physical effort. PLoS Biol. 10, (2012).
Harsay, H. A. et al. Functional connectivity of the striatum links motivation to action control in humans. J. Neurosci. 31, 10701–10711 (2011).
Heston, T. F. & King, J. M. Predictive power of statistical significance. World J. Methodol. 7, 112–116 (2017).
Button, K. S. et al. Power failure: Why small sample size undermines the reliability of neuroscience. Nat. Rev. Neurosci. 14, 365–376 (2013).
Westbrook, A. et al. Dopamine promotes cognitive effort by biasing the benefits versus costs of cognitive work. Science 367, 1362–1366 (2020).
Hofmans, L. et al. Methylphenidate boosts choices of mental labor over leisure depending on baseline striatal dopamine. Preprint at https://www.biorxiv.org/content/10.110 (2020).
Silston, B. & Mobbs, D. Dopey dopamine: high tonic results in ironic performance. Trends Cogn. Sci. 18, 340–341 (2014).
Simonsohn, U. Small telescopes: detectability and the evaluation of replication results. Psychol. Sci. 26, 559–569 (2015).
Faul, F., Erdfelder, E., Buchner, A. & Lang, A. G. Statistical power analyses using G*Power 3.1: tests for correlation and regression analyses. Behav. Res. Methods 41, 1149–1160 (2009).
Patlak, C. S., Blasberg, R. G. & Fenstermacher, J. D. Graphical evaluation of blood-to-brain transfer constants from multiple-time uptake data generalizations. J. Cereb. Blood Flow Metab. 5, 584–590 (1983).
Piray, P., Den Ouden, H. E. M., Van Der Schaaf, M. E., Toni, I. & Cools, R. Dopaminergic modulation of the functional ventrodorsal architecture of the human striatum. Cereb. Cortex 27, 485–495 (2017).
Mawlawi, O. et al. Imaging human mesolimbic dopamine transmission with positron emission tomography: I. Accuracy and precision of D2 receptor parameter measurements in ventral striatum. J. Cereb. Blood Flow Metab. 21, 1034–1057 (2001).
Lawrence, M. A. ez: Easy Analysis and Visualization of Factorial Experiments. R package version 4.4–0. https://CRAN.R-project.org/package=ez (2016).
Egerton, A., Demjaha, A., McGuire, P., Mehta, M. A. & Howes, O. D. The test-retest reliability of 18F-DOPA PET in assessing striatal and extrastriatal presynaptic dopaminergic function. Neuroimage 50, 524–531 (2010).
Niv, Y., Daw, N. D., Joel, D. & Dayan, P. Tonic dopamine: opportunity costs and the control of response vigor. Psychopharmacology. 191, 507–520 (2007).
Becker, G. et al. Comparative assessment of 6-[18F]fluoro-L-m-tyrosine and 6-[18F]fluoro-L-dopa to evaluate dopaminergic presynaptic integrity in a Parkinson’s disease rat model. J. Neurochem. 141, 626–635 (2017).
Doudet, D. J. et al. Evaluation of dopaminergic presynaptic integrity: 6-[18 P]Fluoro-l-dopa versus 6-[18 P]Fluoro-L-m-Tyrosine. J. Cereb. Blood Flow Metab. 19, 278–287 (1999).
DeJesus, O. T., Endres, C. J., Shelton, S. E., Jerome Nickles, R. & Holden, J. E. Noninvasive assessment of aromatic L-amino acid decarboxylase activity in aging rhesus monkey brain in vivo. Synapse 39, 58–63 (2001).
Jonasson, L. S. et al. Dopamine release in nucleus accumbens during rewarded task switching measured by [11C]raclopride. Neuroimage 99, 357–364 (2014).
Manohar, S. G. et al. Reward pays the cost of noise reduction in motor and cognitive control. Curr. Biol. 25, 1707–1716 (2015).
Timmer, M. H. M., Aarts, E., Esselink, R. A. J. & Cools, R. Enhanced motivation of cognitive control in Parkinson’s disease. Eur. J. Neurosci. 48, 2374–2384 (2018).
Smith, C. T. et al. Modulation of impulsivity and reward sensitivity in intertemporal choice by striatal and midbrain dopamine synthesis in healthy adults. J. Neurophysiol. 115, 1146–1156 (2016).
Cools, R. et al. Striatal dopamine predicts outcome-specific reversal learning and its sensitivity to dopaminergic drug administration. J. Neurosci. 29, 1538–1543 (2009).
Daneman, M. & Carpenter, P. A. Individual differences in working memory during reading. J. Verbal Learning Verbal Behav. 19, 450–466 (1980).
Carver, C. & White, T. Behavioral inhibition, behavioral activation, and affective responses to impending reward and punishment: The BIS/BAS Scales. J. Pers. Soc. Psychol. 67, 319–333 (1994).
Allen, E. A., Erhardt, E. B. & Calhoun, V. D. Data visualization in the neurosciences: overcoming the curse of dimensionality. Neuron 74, 603–608 (2012).
Zandbelt, B. Slice display. figshare https://doi.org/10.6084/m9.figshare.4742866 (2017).
Allen, M., Poggiali, D., Whitaker, K., Marshall, T. & Kievit, R. RainCloudPlots tutorials and codebase (Version v1.1). Zenodo https://doi.org/10.5281/zenodo.3368186 (2018).
Acknowledgements
The work was funded by a Vici grant from the Netherlands Organization for Scientific Research (NWO) (Grant No. 453-14-015) awarded to RC. We thank Lieke van Lieshout and Felix Linsen for assistance during data collection.
Author information
Authors and Affiliations
Contributions
Conceptualization, R.C., L.H. and E.A.; methodology E.A., R.C. and L.H.; software, E.A. and L.H.; formal analysis, L.H. and R.B.; investigation, L.H.; writing—original draft, L.H.; writing—review and editing, L.H., R.C., E.A., R.-J.V., R.B., and J.I.M.; visualization, L.H.; supervision, R.C. and R.-J.V.; project administration, L.H. and J.I.M.; funding acquisition, R.C.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hofmans, L., van den Bosch, R., Määttä, J.I. et al. The cognitive effects of a promised bonus do not depend on dopamine synthesis capacity. Sci Rep 10, 16473 (2020). https://doi.org/10.1038/s41598-020-72329-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-72329-4
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.