Abstract
Recently, many new rice–fish co-culture models have been developed to increase economic and ecological benefits. In this study, we added eels (Monopterus albus) to a rice–crayfish system and conducted a 3-year field investigation to compare the yields and availability of fertilizer N among groups with a low density of eels, high density of eels and no eels. We performed a mesocosm experiment and used an isotope tracer technique to detect the fate of fertilizer N. The results showed that the rice yields significantly improved after the introduction of the eels. However, the introduction of a high density of eels significantly limited the crayfish yield, increased water N and N2O emissions and decreased soil N content. The mesocosm experiment suggested that the use efficiency of fertilizer N was significantly increased after the introduction of the eels. The fertilizer N used by rice was significantly higher in rice–crayfish–eel system than in rice–crayfish system. This study indicated that the introduction of eels may be a good practice for improving yields and availability of fertilizer N in a rice–crayfish system.
Similar content being viewed by others
Introduction
In recent years, a substantial increase in integrated rice–fish culture (IRFC) has been observed in China. By the end of 2010, IRFC covers an area of 1.33 × 106 ha, which accounts for 4.48% of the total rice planting area in China1. The primary concept of IRFC is to culture aquatic animals, e.g. fish, shrimp, crabs and soft shell turtles, in limited paddy space2,3. When compared with monoculture of rice and fish, IRFC has advantages, such as optimizing land resources, saving labour inputs and facilitating field management4. IRFC is considered an effective agriculture mode that can simultaneously provide food security and conserve the environment because almost no pesticides or herbicides are used during rice production5. Moreover, IRFC can increase fertilizer availability and decrease fertilizer application6,7,8.
Nitrogen (N) fertilizer provides an essential nutrient for rice cultivation. In IRFC, the availability of soil or fertilizer N can be enhanced through the complementary use of N by rice and fish9. The activities of aquatic animals can increase N release from the soil and N uptake by rice plants10,11,12,13. Fish excrement and effluents also have a fertilizing effect, which increases the amount of nutrients available to the rice crops14. Excess N temporarily retained in rice fields can be transmitted through the food chain by aquatic animals15. Therefore, when compared with rice monoculture, IRFC can greatly reduce N loss to the environment.
Integrated rice–crayfish (Procambarus clarkii) culture (IRCC) is one of the most popular IRFCs in China. Currently, the total area under IRCC in China is 6 × 105 ha, and 1.2 × 106 t crayfish are produced from paddy fields16. The co-culture of rice and crayfish can improve the soil carbon pool and microbial community structure17. Crayfish activities can contribute to a high rice yield from paddy fields18. However, the explosive increase in crayfish production has greatly limited the economic benefits of IRCC in the past few years. Moreover, crayfish farming relies too much on artificial diets, which may cause water pollution and environmental damage. To increase income and ecological health, many farmers have tried to introduce new species into the rice–crayfish (RC) system to develop new IRFCs with more complex species combinations, such as rice–crayfish–eel and rice–crayfish–turtle co-cultures19.
In China, the Asian swamp eel (Monopterus albus) is an indigenous species with a high economic value. The eels can adapt to the complex environment of rice fields, and they are considered as an ideal species for rice paddy farming20. The introduction of eels to the RC system can improve spatial efficiency, prolong the food chain and increase biodiversity21. Many scholars have detected N availability in the RC system. Previous studies have suggested that IRCC does not significantly increase the N uptake in rice grains, roots and straw when compared with rice monoculture22. Moreover, the co-culture of rice and crayfish may cause more N loss in the form of N2O from the paddy ecosystem23. However, there is limited information on the effects of eel or eel–crayfish disturbance on the N cycle in paddy fields.
In this study, we investigated a rice–crayfish–eel (RCE) system continuously for 3 years. Meanwhile, we performed a mesocosm experiment and used a stable isotope (15N) tracer technique. The aims of this study were to (1) analyse the effects of eel introduction on the yields of rice and crayfish and (2) detect the availability of fertilizer N in the RCE system.
Results
Yields in field investigation
Table 1 shows the average yields and total N content of rice, crayfish and eels. The rice yields decreased significantly in the control paddies during the investigation (P < 0.05, Table S1). However, no significant changes were observed in the yields of rice, crayfish and eels in the LD and HD groups (P > 0.05, Table S1). From 2018 to 2019, the rice yields were significantly higher in the LD and HD groups than in the control group (P < 0.05, Table S1). However, the crayfish yields in the HD group were significantly lower than that in the LD and control paddies (P < 0.05, Table S1). The eel yields were significant higher in the HD group than in the LD group (P < 0.05, Table S1). The total N content of rice, crayfish and eels had similar spatial and temporal trends with the rice yields.
Total N content in the water and soils
Figure 1 shows the changes in water and soil N during the 3-year investigation. No significant changes in water and soil N were observed during the 3 years (P > 0.05, Table S2). The average content of total N in the water was significantly higher in the HD group than in the control paddy (P < 0.05, Table S2). From August to October, the total N content in the water was significantly higher in the HD group than in the control group (P < 0.05, Table S2). However, no significant differences in water N were observed among the three groups from June to July (P > 0.05, Table S2). In contrast, the soil N was significantly lower in the HD group than in the control and LD paddies (P < 0.05, Table S2).
Average values of total N in the water (A) and soils (B) and monthly changes in total N in the water (C) of the different co-culture systems: rice–fish culture (C), rice–crayfish–eel with a low density of eels (LD) and rice–crayfish–eel with a high density of eels (HD). The lowercase letters represent a significant difference among the three co-culture systems (P < 0.05).
N2O emission and NH3 volatilization
Figure 2 shows the variations in N2O emission and NH3 volatilization from the three rice–fish groups. The average N2O flux was significantly higher in the HD group than in the other two groups (P < 0.05, Table S3). In August and October, the N2O flux was significantly higher in HD group than in LD and control groups (P < 0.05, Table S3). However, no significant differences in N2O flux were observed in the other 3 months among three groups (P > 0.05, Table S3).
Average values of N2O emission (A) and NH3 volatilization (B) and monthly changes in N2O (C) and NH3 (D) in the different co-culture systems: rice–fish culture (C), rice–crayfish–eel with a low density of eels (LD) and rice–crayfish–eel with a high density of eels (HD). The lowercase letters represent a significant difference among the three co-culture systems (P < 0.05).
No significant differences in average NH3 volatilization were observed among the three groups (P > 0.05, Table S3). The NH3 flux was significantly higher in HD group than in control group (P < 0.05, Table S3) from September to October. However, there were no significant differences in NH3 flux among three groups from June to August (P > 0.05, Table S3).
Fate of fertilizer N
Figure 3 shows the fate of fertilizer N in the RCE and RC systems. No significant differences in the total N content of rice (P = 0.228) and crayfish (P = 0.334) were observed between the RCE and RC systems after the mesocosm experiment. The use efficiency of fertilizer N was significantly higher in RCE (54.39%) than in RC (36.78%; P = 0.009). The proportion of fertilizer N used by crayfish was significantly higher in RC (7.78%) than in RCE (0.82%; P = 0.003). In contrast, the proportion of fertilizer N used by rice was significantly lower in RC (29.00%) than in RCE (37.30%; P = 0.048). In the RCE system, about 16.27% of N was transferred to the eels from the fertilizer.
The total N content of the organisms (A), fate of fertilizer N (B,C) and N use efficiency (D) in the mesocosm experiment. RCE and RC represent rice–crayfish–eel and rice–crayfish systems, respectively. *indicates that no samples were collected from the mesocosm. The lowercase letters represent a significant difference among the different species (P < 0.05). The capital letters represent a significant difference between REC and RC (P < 0.05).
Discussion
This study demonstrated that the introduction of eels at a low density significantly increased rice yields in the RC system. Moreover, no significant changes in rice and crayfish yields of the LD group occurred during the investigation. The results suggest that eels and crayfish can be bred simultaneously in paddies, despite their predation relationship. Moreover, the rice yields may be enhanced by multispecies complex rearing in the rice field. This viewpoint was also demonstrated by other scholars24,25. Lin and Wu found that the rice yields were significantly higher in the rice–frog–fish system than in rice–fish co-culture and rice monoculture25. In the rice fields, the eels prefer high temperatures and are usually active in the rice-growing platform. In contrast, the crayfish are aquatic animals that prefer shady areas and usually live at the bottom of ditches. The living spaces of the eels and crayfish do not completely coincide. The addition of eels can restrict the activities of crayfish in the rice-planting platform, thus reducing the destruction of rice roots by the crayfish. Therefore, an appropriate number of eels may promote the sustainable development of the RCE system.
This study also suggests that the crayfish yields were significantly suppressed by a high density of eels, although the rice yields were significantly increased. Previous studies have shown that bioturbation by eels is beneficial for maintaining the ecological security of paddies and rice yields because they prey on insect pests26. However, the eels also feed on benthic animals and fishes. The crayfish, especially the juvenile ones, may become the major food source of eels in the RCE system27. In rice fields, cultivation of crayfish would mainly require self-propagation and self-breeding; thus, the crayfish juveniles would probably be heavily preyed on by the eels, leading to an inevitable population degradation. In addition, we found that the concentration of water N in high-density group was significantly enhanced. However, the variation of ammonia can alter the duration and intensity of agonistic interactions in the crayfish28. Therefore, the decline in crayfish populations could also be caused by cannibalism.
In this study, the introduction of eels considerably increased water N and decreased soil N. Moreover, previous studies have suggested that the process of N release is affected by many abiotic and biotic factors, e.g. temperature, mobility and rearing density29. We found that the water N content was significantly higher in the HD group than in the control group from August to October. In addition, bioturbation by the eels in the rice platform may have loosened the soil structure, thus increasing pore size and sediment permeability and fertilizer N uptake by rice. Therefore, the N content of rice also significantly increased in the groups with eels.
The emission of greenhouse gases, e.g. N2O and NH3, is one of the main methods of N loss from rice fields. In this study, the emission of N2O was significantly increased after the introduction of a high density of eels. Some behaviours of aquatic animals, e.g. digging burrows and foraging, can promote gas exchange among soil, water and atmosphere as well as enhance soil Eh, which contributes to the production of N2O through nitrification30. Moreover, the N substrates used for nitrification and denitrification can be obtained from the excretions of crayfish and eels. We found that NH3 volatilization was higher in the HD group than in the LD and control groups. This was possibly attributable to an increase in ionized ammonium (NH4+). Hargreaves considered that NH3 volatilization is determined by the equilibrium between unionized ammonia (NH3) and ionized ammonium (NH4+)31. All three groups showed a trend of increase in NH3 flux from June to September. This may be because the ingestion and excretion of crayfish and eels may be accelerated with an increase in temperature, thus increasing the concentrations of NH3 and NH4+.
In the mesocosm experiment, we found that the use efficiency of fertilizer N significantly improved with the introduction of eels in the RC system. However, the proportion of fertilizer N in the crayfish was significantly lower in the RCE system than in the RC system because the feeding habitat of crayfish may have been affected by the introduction of eels. A previous study indicated that about 58.6–65.3% of crayfish diets originated from aquatic plants, zooplankton and organic debris in the RC co-culture system15. However, the eel activities restricted the crayfish to the benthic zone, thus greatly reducing the probability of the crayfish of feeding on plants. Therefore, the N of crayfish in the RCE culture was mainly derived from the artificial diet, although no significant differences in total N were found between the RC and RCE systems. Wan et al. reported that muscle quality can be significantly improved in integrated RC culture (when compared with crayfish monoculture in ponds) because the crayfish can ingest more plant fibre32. Therefore, the quality of crayfish may be indirectly degraded in the RCE system, although their yields did not decrease significantly. To sum up, the introduction of appropriate amount of eels into rice–crayfish system may improve the availability of nitrogen fertilizer without increasing nitrogen loss to the environment.
Conclusions
This study demonstrated the possibility of co-culture of crayfish and eels in rice fields. The addition of eels at a low density can promote the rice yield, while maintaining crayfish yield and N content in the environment. However, an overabundance of eels can cause a decline in crayfish yield. Moreover, total N content of the water and N2O emission increased significantly after the introduction of eels at a high density. More fertilizer N was used by rice and less N entered the crayfish from the fertilizer in the RCE system than in the RC system. The recycling of N in the field shows that the availability of fertilizer in the RC system can be effectively improved after the introduction of an appropriate number of eels.
Materials and methods
Field investigation
This study was performed between from May 2017 and October 2019 at Xinsheng Aquaculture Professional Cooperative (121° 0′ 56″ N, 30° 58′ 17″ E) in Qingpu District, Shanghai, Eastern China. This region has a subtropical monsoon climate with a mean monthly air temperature of 17.6 ± 2.3 °C and mean monthly precipitation of 126.9 ± 24.6 mm.
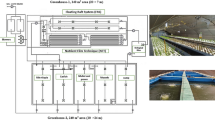
Each RC paddy (667 m2) had a rice-growing area (80% of the total area), aquaculture area (10%) and ridge area (10%; Fig. 4A). In the aquaculture area, a 1.2 m deep ditch was dug to provide a more comfortable habitat for the crayfish and eels. The ridge had a height of 40 cm, and it was covered with a high-density polyethylene film to prevent the aquatic animals from escaping. Every May, rice (Oryza sativa L., Qing-Xiang-Ruan-Geng) seedlings were transplanted from a nursery into the paddies at a planting density of 20 × 20 cm (one seedling on each hill). Moreover, the juvenile crayfish weighing 1.5 ± 0.3 g were released into the paddies according to the standard of 45,000 juveniles per hectare, and the crayfish were allowed to self-propagate inside the rice paddies. A total of nine RC paddies were divided into three groups according to the rearing density of the eels: control group (C), low-density group (LD) and high-density group (HD) with rearing densities of 0, 6000 and 12,000 ind. ha−1, respectively. The LD and HD groups were supplemented with juvenile eels at a density of 2000 and 4000 ind. ha−1 in June 2018 and 2019. The average weights of juvenile eels in 2017, 2018 and 2019 were 21.4 ± 1.8, 24.1 ± 0.9 and 26.8 ± 1.1 g, respectively. All juvenile crayfish and eels were purchased from Shanghai Xiangsheng Aquaculture Cooperative. In the aquaculture area, floating plants, such as duckweed (Lemna minor L.) and foxtail (Myriophyllum spicatum L.), covered one-third of the water surface. The soil contained 20.6–23.7 g kg−1 of organic matter, 0.7–1.2 g kg−1 of total N and 0.31–0.37 g kg−1 of total P.
Only basal fertilizer was used for rice cultivation, and it contained 587 kg ha−1 of urea (46.4% N), 625 kg ha−1 of superphosphate and 150 kg ha−1 of potassium chloride. Every day, 500 g of commercial fish diet (5.83% N) was applied, and no pesticides or herbicides were used in the paddies.
In late August, the mature crayfish and eels were collected using ground cages to measure the aquatic product yields. The immature crayfish and eels were returned to the paddy fields during the collection. After the rice was harvested, the rice grains were air-dried and weighed to estimate the rice yield. The N content of the rice grains and aquatic animals was determined using the semi-micro Kjeldahl method33. Before testing, rice grains, crayfish and eels were weighted, dried at 65 °C and ground. Then, all the samples were digested with concentrated sulphuric acid (H2SO4) and hydrogen peroxide.
Water samples were collected every month during the co-culture period. Three duplicate 500 mL water samples were collected from 0 to 10 cm below the surface in the aquaculture area; the three subsamples were combined to obtain one sample per paddy. In the laboratory, the total N content of the water was analysed using UV spectrophotometry after digestion by alkaline potassium persulfate oxidation.
Soil samples were collected after the rice-planting period. In each paddy, three samples were collected from a rice-planting area of 0.25 m × 0.25 m × 0.10 m. All the soil samples were air-dried, ground, passed through a 0.15 mm sieve and digested with K2SO4–CuSO4–Se solution. Then, the semi-micro Kjeldahl method was used to test the total N content of the soil.
The N2O flux rate was measured using the static chamber method34. The size of the chamber was 1.0 m × 1.0 m × 1.0 m. The N2O samples were collected every half month between 8:30 and 10:30 AM from June to October. In each paddy, four gas samples were collected using 40 mL vacuum tubes at 10 min intervals (0, 10, 20 and 30 min after chamber closure). All samples were analysed with gas chromatography (GC 2010; Shimadzu, Kyoto, Japan). The N2O flux rate was calculated using the following equation:
where F is the N2O flux rate (μg N m−2 h−1); ρ, density of N2O at the standard state (μg m−3); h, height of the chamber (m); T, average temperature in the chamber during gas collection and dC/dt, concentration variation rate of N2O.
The ammonia volatilization flux was measured with a continuous airflow enclosure method35. The NH3 flux was measured every half month from 09:00 to 11:00 AM during the rice-planting period. NH3 was absorbed using boric acid, and 0.01 M H2SO4 was used to titrate the solution to determine the rate of NH3 volatilization. The ammonia volatilization flux was calculated using the following equation:
where F denotes the ammonia volatilization flux (mg N m−2 h−1); V, volume of H2SO4 titrated (L); C, concentration of H2SO4 (mol L−1); A, area of the chamber base (m2) and t, continuous measurement time.
In this study, all the data were shown as mean ± standard error of the mean (SEM) values. One-way ANOVA and Tukey’s test (SPSS V.16.0) were used to compare the differences of the yields and total N content among the three groups and three investigated years.
Mesocosm experiment
Between May and October 2019, the mesocosm experiment was conducted at Shanghai Academy of Agricultural Sciences. Each mesocosm consisted of an experimental plot (1.2 m × 1.2 m × 0.6 m) covered with a high-density polyethylene film (Fig. 4B). In each experiment plot, 30 kg of soil from Xinsheng Aquaculture Professional Cooperative was used to construct a rice-planting platform and an aquaculture ditch (40 cm in depth). The platform area was about three-fourth of the cross-sectional area of the plot.
A total of six mesocosms were constructed: three experimental plots (RCE) and three control plots (RC). In each plot, the rice seedlings were planted in hills (one seedling per hill) within rows in May, with 20 cm between rows and 20 cm between hills in the same row for the experimental and control plots. The fertilizers used in each plot contained 84.5 g of urea (N content, 46.8%; 15N abundance, 10.15%), 90 g of superphosphate and 15 g of potassium chloride. The duckweed was planted in the aquaculture area, and it covered 30% of the aquaculture zone. Mudsnails (Cipangopaludina cathayensis, 500 g) were added to each plot. After a month, 12 crayfish were cultured in each simulated paddy, and two eels were reared in each experiment plot. The proportion of crayfish and eels was set according to that in the LD group of field investigation. The crayfish feed was supplied once every day, and the daily allowance was about 3% of the estimated crayfish weight in each mesocosm. The rice and aquatic products were harvested in October.
Rice, crayfish and eel samples were collected to measure the total N content and 15N abundance. The total N content of the soil and organism samples were measured using the semi-micro Kjeldahl method after digestion with concentrated H2SO4 and hydrogen peroxide. The 15N abundance was measured in all samples by using the MAT-271 isotope mass spectrometer (Finnigan MAT, California). The accumulation of N in rice, crayfish and eels from N fertilizer was calculated using the following equations:
where A% E is the difference between the 15N abundance of the samples or 15N-labelled fertilizers and natural abundance of 15N.
The independent-samples t-test was used to determine the differences in total N, N use efficiency and percentage of N derived from fertilizer between RCE and RC at 95% confidence level by using SPSS 16.0 (P value < 0.05 was considered statistically significant).
References
Hu, L. L. et al. Development of rice–fish system: today and tomorrow. Chin. J. Eco Agric. 23, 268–275 (2015).
Fernando, C. H. Rice field ecology and fish culture—an overview. Hydrobiologia 259, 91–113 (1993).
Halwart, M. Biodiversity and nutrition in rice-based aquatic ecosystems. J. Food Compos. Anal. 19, 747–751 (2006).
McClain, W. R. & Romaire, R. P. Crawfish culture: a Louisiana aquaculture success story. World Aquacult. 35, 60–61 (2004).
Xie, J. et al. Ecological mechanisms underlying the sustainability of the agricultural heritage rice–fish coculture system. Proc. Natl. Acad. Sci. USA 108, E1381–E1387 (2011).
Oehme, M., Frei, M., Razzak, M. A., Dewan, S. & Becker, K. Studies on nitrogen cycling under different nitrogen inputs in integrated rice–fish culture in Bangladesh. Nutr. Cycl. Agroecosyst. 79, 181–191 (2007).
Li, C. F. et al. Studies on nitrogen cycling in integrated rice–duck, rice–fish ecosystems. J. Agro Environ. Sci. 27, 1326–1334 (2008).
Vromant, N., Chau, N. T. H. & Ollevier, F. The effect of rice-seeding rate and fish stocking on the floodwater ecology of the trench of a concurrent, direct-seeded rice–fish system. Hydrobiologia 457, 105–117 (2001).
Hu, L. L. et al. Productivity and the complementary use of nitrogen in the coupled rice–crab system. Agric. Syst. 178, 102742 (2020).
Clark, K. L., Branch, L. C., Hierro, J. L. & Villarreal, D. Burrowing herbivores alter soil carbon and nitrogen dynamics in a semi-arid ecosystem, Argentina. Soil Biol. Biochem. 103, 253–261 (2016).
Ritvo, G., Kochba, M. & Avnimelech, Y. The effects of common carp bioturbation on fishpond bottom soil. Aquaculture 242, 345–356 (2004).
John, K., Jauker, F., Marxsen, J., Zaitsev, A. S. & Wolters, V. Earthworm bioturbation stabilizes carbon in non-flooded paddy soil at the risk of increasing methane emissions under wet soil conditions. Soil Biol. Biochem. 91, 127–132 (2015).
Wang, Q., Wang, W., Ma, X. Z., Chen, Z. Z. & Yu, Y. Q. The effects of integrated ricecrab production on soil physical and chemical properties. Hubei Agric. Sci. 50, 3948–3952 (2011).
Hu, L. L. et al. The productivity of traditional rice–fish co-culture can be increased without increasing nitrogen loss to the environment. Agric. Ecosyst. Environ. 177, 28–34 (2013).
Li, C., Cheng, Y. X., Guan, Q. Z., Xi, Y. W. & Li, J. Y. Using stable isotopes to estimate the effects of different carbon/nitrogen feeds on feeding habit of Procambarus clarkii in the rice–crayfish co-culture system. J. Fish. China 42, 1778–1786 (2018).
Guo, Y. F., Zhu, Z. W., Ma, D. W. & Tang, J. Q. The development report of China’s crayfish industry in 2017. China Fish. 13, 8–17 (2017).
Si, G. H. et al. Changes in soil microbial community composition and organic carbon fractions in an integrated rice–crayfish farming system in subtropical China. Sci. Rep. 7, 2856 (2017).
Peng, C. L. et al. Effects of long-term integrated rice–crayfish model on yield and nitrogen use efficiency of direct-seeding rice under different nitrogen application rates. J. Henan Agric. Sci. 49, 15–21 (2020).
Shao, N. L., Guo, Y., Shen, H., Chen, J. M. & Wei, H. Water quality comparison and economic benefit analysis of rice–crayfish–eel and rice–crayfish–eel–turtle co-cultures. Fish. Sci. Tech. Inf. 43(1), 33–36 (2016).
Zeng, B. P. & Wang, W. B. Seasonal population dynamics of Pallisentis (Neosentis) celatus (Acanthocephala: Quadrigyridae) in the intestine of the rice-field eel Monopterus albus in China. J. Helminthol. 81, 415–420 (2007).
Lv, W. Q. et al. Microplastic pollution in rice–fish co-culture system: a report of three farmland stations in Shanghai, China. Sci. Total Environ. 652, 1209–1218 (2019).
Si, G. H., Yuan, J. F., Peng, C. L., Xia, X. G. & Cheng, J. P. Nitrogen and phosphorus cycling characteristics and balance of the integrated rice–crayfish system. Chin. J. Eco Agric. 27, 1309–1318 (2019).
Sun, Z. C. et al. Effects of straw returning and feeding on greenhouse gas emissions from integrated rice–crayfish farming in Jianghan Plain, China. Environ. Sci. Pollut. Res. 26, 11710–11718 (2019).
Wan, N. F. et al. Multispecies co-culture promotes ecological intensification of vegetable production. J. Clean. Prod. 257, 120851 (2020).
Lin, K. M. & Wu, J. P. Effect of introducing frogs and fish on soil phosphorus availability dynamics and their relationship with rice yield in paddy fields. Sci. Rep. 10, 21 (2020).
Wan, N. F. et al. Ecological intensification of rice production through rice–fish co-culture. J. Clean. Prod. 234, 1002–1012 (2019).
Li, Q. M. et al. Influence of consecutive integrated rice–crayfish culture on phosphorus fertility of paddy soils. Land Degrad. Dev. 29, 3413–3422 (2018).
Edwards, D. D., Klotz, K. L. & Moore, P. A. Exposure to sublethal ammonia concentrations alters the duration and intensity of agonistic interactions in the crayfish, Orconectes rusticus. Bull. Environ. Contam. Toxicol. 100, 189–194 (2018).
Sun, G. & Fang, Y. Bioturbation Effects of Benthic Animals 83–89 (Science Press, Beijing, 2013).
Xu, X. Y. et al. Effect of rice–crayfish co-culture on greenhouse gases emission in straw-puddled paddy fields. Chin. J. Eco Agric. 25, 1591–1603 (2017).
Hargreaves, J. A. Nitrogen biogeochemistry of aquaculture ponds. Aquaculture 166, 181–212 (1998).
Wan, J. J., Chen, Y. M., Shao, J. J., Tang, J. P. & Shen, M. F. Comparative analysis on muscle quality of red swamp crayfish (Procambarus clarkia) cultured under different aquaculture modes in Xuyi region. Chin. J. Anim. Nutr. 32, 965–972 (2020).
Zheng, H. J., Zuo, J. C., Wang, L. Y., Li, Y. J. & Liao, K. T. 15N isotope tracing of nitrogen runoff loss on red soil sloping uplands under simulated rainfall conditions. Plant Soil Environ. 62, 416–421 (2016).
Liu, S. et al. Methane and nitrous oxide emissions reduced following conversion of rice paddies to inland crab–fish aquaculture in Southeast China. Environ. Sci. Technol. 50, 633–642 (2016).
Feng, Y. et al. Biochar applied at an appropriate rate can avoid increasing NH3 volatilization dramatically in rice paddy soil. Chemosphere 168, 1277–1284 (2017).
Acknowledgements
This study was supported by the Shanghai Municipal Agricultural Commission [Grant Number (2018) 1-28] and SAAS Program for Excellent Research Team [nong ke chuang 2017 (A-03)].
Author information
Authors and Affiliations
Contributions
W.L. wrote the manuscript. Q.Y. collected and analyzed the data. W.L. contributed to the plot management. W.Z. designed and performed the experiment.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lv, W., Yuan, Q., Lv, W. et al. Effects of introducing eels on the yields and availability of fertilizer nitrogen in an integrated rice–crayfish system. Sci Rep 10, 14818 (2020). https://doi.org/10.1038/s41598-020-71884-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-71884-0
This article is cited by
-
Distinct bacterial communities in the environmental water, sediment and intestine between two crayfish-plant coculture ecosystems
Applied Microbiology and Biotechnology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.