Abstract
Few studies have investigated the measurement of oxygen uptake (\({\dot{\text{V}}}\)O2) in tracheostomized patients undergoing unassisted breathing trials (UBTs) for liberation from mechanical ventilation (MV). Using an open-circuit, breath-to-breath method, we continuously measured \({\dot{\text{V}}}\)O2 and relevant parameters during 120-min UBTs via a T-tube in 49 tracheostomized patients with prolonged MV, and calculated mean values in the first and last 5-min periods. Forty-one (84%) patients successfully completed the UBTs. The median \({\dot{\text{V}}}\)O2 increased significantly (from 235.8 to 298.2 ml/min; P = 0.025) in the failure group, but there was no significant change in the success group (from 223.1 to 221.6 ml/min; P = 0.505). In multivariate logistic regression analysis, an increase in \({\dot{\text{V}}}\)O2 > 17% from the beginning period (odds ratio [OR] 0.084; 95% confidence interval [CI] 0.012–0.600; P = 0.014) and a peak inspiratory pressure greater than − 30 cmH2O (OR 11.083; 95% CI 1.117–109.944; P = 0.04) were significantly associated with the success of 120-min UBT. A refined prediction model combining heart rate, energy expenditure, end-tidal CO2 and oxygen equivalent showed a modest increase in the area under the receiver operating characteristic curve of 0.788 (P = 0.578) and lower Akaike information criterion score of 41.83 compared to the traditional prediction model including heart rate and respiratory rate for achieving 48 h of unassisted breathing. Our findings show the potential of monitoring \({\dot{\text{V}}}\)O2 in the final phase of weaning in tracheostomized patients with prolonged MV.
Similar content being viewed by others
Introduction
Liberating patients from prolonged mechanical ventilation (MV) remains a challenge. Prolonged MV is generally defined as the requirement of at least 6 h of MV for > 21 consecutive days1,2, although reported periods of ventilator dependence range from 2 days to 4 weeks3. These patients account for about 10% of those with acute respiratory failure as a consequence of persistent respiratory failure or complications, and they have a very poor prognosis, with 1-year survival of only 40–50%3. This status of chronic critical illness also imposes a significant care burden on intensive care units (ICUs)4, with an associated mean incremental cost of $1522 per day for patients in the United States5. Successful liberation from MV is, therefore, an important goal in the care of surviving subacute patients. However, only 30–53% of patients with prolonged MV can be successfully liberated from MV3.
Frequent screening for weaning and early initiation of unassisted breathing trials (UBTs) have been reported to lead to a higher weaning rate6. Although patients may be able to tolerate the initial reduction in MV settings and proceed with a final UBT, failure at this stage may be encountered due to respiratory, cardiac, neuromuscular, psychological, metabolic, anemia, and nutritional problems7. This condition may be associated with insufficient strength and endurance of respiratory muscles due to the increased respiratory mechanical load8. In addition, the change from MV support to unassisted breathing can increase the work of breathing (WOB)9. However, measuring WOB may not be practical in a post-acute setting due to the need for invasive placement of pressure sensors.
Oxygen uptake (\({\dot{\text{V}}}\)O2) assesses the global ability of oxygen intake by the respiratory system, delivery by the cardiovascular system, and use by working tissues. In physiologic studies, maximum \({\dot{\text{V}}}\)O2 is traditionally used to evaluate cardiorespiratory capacity10. For example, in cardiopulmonary exercise tests it is used to differentiate the cause of dyspnea11, in which an incremental external work rate is usually applied to determine whether the \({\dot{\text{V}}}\)O2 is above the anaerobic threshold12,13,14,15. The kinetics of \({\dot{\text{V}}}\)O2 during exercise have also been correlated with the prognosis in patients with chronic heart failure13,16,17,18,19,20.
Previous studies have shown that the change in \({\dot{\text{V}}}\)O2 when reducing ventilator support can be used as an alternative measurement to represent the change in WOB in patients receiving ventilator support21. Although previous studies have evaluated associations between changes in \({\dot{\text{V}}}\)O2 at different levels of ventilator support and the ability to spontaneously breathe and the outcomes of weaning19,22,23, few studies have investigated measurements of the kinetics of \({\dot{\text{V}}}\)O2 in tracheostomized patients undergoing UBTs. Understanding the kinetics of \({\dot{\text{V}}}\)O2 and associated metabolic and spirometry parameters may help clinicians to differentiate the cause of weaning failure in this last stage. Therefore, the aim of this pilot study was to investigate the kinetics of \({\dot{\text{V}}}\)O2 during UBTs in patients with prolonged MV.
Methods
Design and setting
This prospective study was conducted from January to December 2018 at the Respiratory Care Center (RCC), a dedicated weaning unit, of National Taiwan University Hospital, Taiwan. All of the experimental protocols were conducted in accordance with the Declaration of Helsinki. The Research Ethics Committee B of National Taiwan University Hospital approved the study (#20170003RINB). The RCC in this study has 15 beds, and treats patients requiring prolonged MV from ICUs using an integrated delivery system according to the regulations of the National Health Insurance program in Taiwan, which was introduced in 1995. Stable prolonged MV patients from ICUs are transferred to the RCC for liberation from MV, and patients who remain under MV after 6 weeks are transferred to the respiratory care ward for long-term ventilator care. In 2014, the unit implemented a standardized weaning protocol6, and the decision to wean is determined by the consensus of attending physicians, residents and respiratory therapists. Patients who can tolerate the liberation process and a final 5 days of continuous unassisted breathing are transferred to a general ward, while those who are not successfully liberated after at least two weaning trials are given long-term respiratory care.
Participants
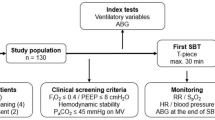
Patients admitted to the RCC were screened and considered eligible if they fulfilled all of the following criteria: age 20 years or older, tracheostomized after intubation for acute respiratory failure, requiring at least 10 days of continuous MV, and no fever or clinical evidence of active infection. In addition, the ventilator settings had to be successfully reduced to and maintained at low-level support for at least 24 h, including the following: pulse oximetry (SpO2) > 90%, PaCO2 < 52 mmHg, respiratory rate (RR) < 35/min under a pressure support mode with inspired oxygen fraction < 0.4, and positive end-expiratory pressure < 8 cmH2O. Patients were excluded if they met any of the following criteria: unstable hemodynamics, active bleeding, documented dynamic airway collapse by bronchoscopic examination, frequent seizures, myoclonus, tremors or involuntary movements.
Measurement of \({\dot{\text{V}}}\)O2, metabolic and spirometry parameters during UBT
After written informed consent had been obtained, the ventilator settings were reduced to a pressure support level ≤ 10 cmH2O and positive end-expiratory pressure of 5 cmH2O under an inspired oxygen fraction of 0.4, and the patient was observed for at least 8 h to ensure stable respiratory and hemodynamic status. The patient then underwent a UBT for 120 min through an open breathing circuit comprised of a T-tube connected to a central oxygen source with an oxygen fraction of 0.4 and air flow of 10 L/min. To measure the \({\dot{\text{V}}}\)O2 data panel, a gas exchange and spirometry measuring kit (D-lite + spirometry kit, GE Healthcare, IL, USA) was bridged between the tracheostomy tube and T-tube, with the pressure sensor and gas sampling pole maintained at a non-dependent position to the kit to avoid occlusion by respiratory secretions and condensed water. During the UBT session, \({\dot{\text{V}}}\)O2 was continuously measured using a commercial system (CARESCAPE Monitor B650 and Engström Carestation, GE Healthcare, IL, USA) based on the following calculations: O2 inspired and expired were calculated by multiplying the % of O2 within inspired air (FIO2) by the volume of air inspired (VI), or % of O2 within expired air (FEO2) by the volume of air expired (VE). \({\dot{\text{V}}}\)O2 was calculated as follows: \({\dot{\text{V}}}\)O2 = (VI × FIO2)—(VE × FEO2). During the session, 11 other parameters were also recorded or calculated, including production of carbon dioxide (\({\dot{\text{V}}}\) CO2), heart rate (HR), RR, tidal volume (VT), minute ventilation (VE), end-tidal CO2 (EtCO2), O2 pulse (calculated as \({\dot{\text{V}}}\)O2/HR), EqO2, equivalent for carbon dioxide (EqCO2), respiratory quotient (RQ), and energy expenditure (EE).
During the UBT session, the patients were allowed to receive necessary bedside care, including suctioning of airway secretions, administration of nutrition, intravenous fluids, oral and intravenous medications, and physical restraints if needed, however percussion, rehabilitation activities, nebulized medications, and bedside procedures were avoided. Once the oxygen uptake tracing had been stabilized after transition to the T-tube, defined as \({\dot{\text{V}}}\)O2 variability < 15% within 15 min, data of \({\dot{\text{V}}}\)O2 and the other 11 parameters were formally recorded. For analysis of the recorded data, observations outlying the range of mean ± 1 standard deviation during each 5-min block were replaced by the mean value of the previous 5-min period. Each parameter in the first 5 min was analyzed as the beginning phase. The last 5-min period of a trial regardless of failure or success was classified as the end phase. The first and last 5-min periods of the 12 parameters during each UBT session were then compared.
Clinical data collection, analysis and outcome classification
For each patient, we collected the following data from the institutional healthcare information system: age, gender, co-morbidities, etiology of respiratory failure and reason to initiate MV, and date of initiating ventilator support. The following clinical and physiological data were also collected before starting the UBT: body height, weight, Glasgow coma scale, lung compliance, airway resistance, venous blood gas analysis, and weaning parameters including maximal inspiratory pressure (PImax), maximal expiratory pressure (PEmax), rapid shallow breathing index (RSBI), and minute ventilation. During the UBT, blood oxygen saturation was continuously measured using pulse oximetry, while blood pressure was measured every 15 min using an electronic sphygmomanometer.
The definition of a failed UBT was adopted from the literature7 as follows: systolic blood pressure > 180 mmHg; HR > 120% of baseline, or the development of arrhythmia; RR > 150% of baseline; SpO2 < 90%; blood CO2 level or EtCO2 level increase > 8 mmHg, or serum pH < 7.2; and clinical judgment of a primary physician who was not involved in this study, including intolerable subjective dyspnea, accessory muscle use, diaphoresis, cyanosis, or loss of consciousness. Reasons for reconnecting to MV and the time to reconnect were recorded.
Statistical analyses
Data of clinical and baseline characteristics are expressed as median (interquartile range [IQR]) for continuous variables and were compared using the Wilcoxon rank-sum test; categorical variables were compared using the chi-square or Fisher's exact test. The moving average of every 5-min time block was performed to smooth the trend of \({\dot{\text{V}}}\)O2 and the 11 other parameters. Observations outlying the range of mean ± 1 standard deviation during each 5-min block were replaced by the mean value of the previous 5-min period.
Parameters at the beginning phase and end phase were reported as median (IQR) and were compared using the Wilcoxon rank–sum test. Physiological changes and \({\dot{\text{V}}}\)O2 kinetics during the UBT were compared using a non-parametric method according to the UBT results.
Univariate logistic regression was used to analyze the results of 2-h UBT and 48-h weaning results. Odds ratios (ORs), 95% confidence intervals (CIs), and P values are reported. The significant predictors in univariate analysis were included in multivariate logistic regression analysis. Variables assessed in the multivariate logistic regression analysis for 48-h outcomes included physiological parameters during UBT, such as HR and RR, and the parameters related to \({\dot{\text{V}}}\)O2. Receiver operating characteristic (ROC) curves were plotted, and the Youden Index was used to identify the best cut-off point. Akaike information criterion (AIC) score was calculated for each model. Data were analyzed using STATA software version 14.0 (StataCorp, College Station, TX, USA). Figures depicting \({\dot{\text{V}}}\)O2 and relevant data were compiled using R software (R version 3.5.3).
Ethics approval and consent to participate
All the experiment protocol for involving humans was in accordance to Taiwan national guidelines in the manuscript. This prospective observational study was approved by the Research Ethics Committee of National Taiwan University Hospital (approval number 20170003RINB).
Results
Participants
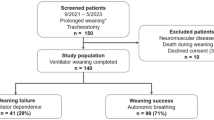
Seventy-two consecutive patients were screened, of whom nine did not provide informed consent, two failed to proceed to the initiation of unassisted breathing due to medical conditions, and three did not undergo tracheostomy throughout the stay. Of the 58 patients who underwent a UBT session with measurements of \({\dot{\text{V}}}\)O2 and relevant parameters, nine were further excluded because their collected data were insufficient to meet the analysis criteria as a result of interrupted data recording involving the beginning 20 min and the last 20 min toward the test end-point, mostly due to profuse airway secretion obstructing the sampling channel for \({\dot{\text{V}}}\)O2. Of the 49 patients who underwent a UBT with valid data for analysis, 41 who completed the 120-min test without meeting the criteria of failure in the UBT session as the primary end-point were enrolled as the success group. In the eight patients in the failure group, the UBT was terminated prematurely at a median duration of 80 min (IQR, 34–96) due to clinical manifestations including significant desaturation in five patients, tachycardia with > 20% HR increase in four, intractable tachypnea in four, and hypercapnia in one. Table 1 summarizes the clinical and demographic characteristics of all enrolled patients (n = 49). Their median age was 67 years (IQR, 61–77), and 35 (71.4%) were male. The most common comorbidities were hypertension (49.0%), diabetes mellitus (32.7%), and solid organ malignancy (28.6%). Pneumonia was the most common etiology (53.1%) of the initial acute respiratory failure. The median duration of MV before the measurement of \({\dot{\text{V}}}\)O2 kinetics was 31 days (IQR, 25–51). There were no significant differences in the clinical and demographic characteristics between the success and failure groups (Table 1).
For the secondary end-point, 35 (85%) of the 41 patients in the success group reached 48 h of unassisted breathing without meeting the criteria of weaning failure. Overall, 34 (83%) patients in the success group at the primary end-point were eventually liberated from MV upon discharge from the weaning unit, with a median duration of 31 days (IQR, 20–64) of MV at the unit. The seven patients who failed could not undergo further weaning due to sepsis and septic shock (five patients) and signs of limited endurance as judged by the clinicians (two patients).
Among the eight patients who failed the 120-min UBT, one was a 68-year-old man with cerebellar epithelioid glioblastoma who failed the 120-min UBT according to \({\dot{\text{V}}}\)O2 measurements, but was finally weaned successfully and discharged. His \({\dot{\text{V}}}\)O2 level increased by 46.5%, and the failed UBT was associated with an acute onset of atrial fibrillation with rapid ventricular response, which was improved by amiodarone. We followed his \({\dot{\text{V}}}\)O2 in the following UBT, which showed a 0.8% decrease in \({\dot{\text{V}}}\)O2, and he was thus successfully weaned. The other seven patients had not been weaned upon RCC discharge.
Physiological measurements at baseline
Table 2 summarizes the physiological data obtained before the UBT and comparisons between the two groups. The data of traditional weaning parameters, lung mechanics and blood gas analysis suggested adequate preparedness for weaning. The success group had a lower body mass index (22.3 [IQR, 19.8–24.8] vs. 26.7 [23.7–30.5] kg/m2, P = 0.0127) and higher PImax (− 40.0 [− 50.0 to − 32.0] vs. − 27.5 [− 30.0 to − 22.5] cmH2O, P = 0.002) than the failure group. Other characteristics were similar between the two groups, including level of consciousness, weaning parameters, lung mechanics measured by inspiratory hold, and baseline arterial CO2 partial pressure.
The kinetics of \({\dot{\text{V}}}\)O2, metabolic and spirometry parameters during the unassisted breathing trial
Table 1S of Additional File 1 shows comparisons of \({\dot{\text{V}}}\)O2 and other parameters between the two groups and comparisons between the beginning and the last 5 min of UBT. There were no significant differences in the 12 parameters at the beginning phase of measurements between the two groups. However, toward the end of the UBT, the failure group had more significant changes in \({\dot{\text{V}}}\)O2 (+ 18.0% vs. − 2.7%, P = 0.009), \({\dot{\text{V}}}\)CO2 (+ 15.1% vs. + 1.4%, P = 0.025), HR (+ 14.9% vs. + 0%, P = 0.001), and energy expenditure (EE) (+ 16.6% vs. − 1.6%, P = 0.004) than the success group (Table S1 of Additional File 1).
In the failure group, the median \({\dot{\text{V}}}\)O2 increased significantly (from 235.8 [154.2–292.6] to 298.2 [188.6–314.0] ml/min; P = 0.025). This change was not significant in the success group (from 223.1 [192.8–250.7] to 221.6 [198.1–252.4] ml/min; P = 0.505) (Fig. 1A). Patients in the success group typically showed a stable \({\dot{\text{V}}}\)O2 throughout the 120 min (Fig. 1B), while patients in the failure group typically showed either a steadily increasing \({\dot{\text{V}}}\)O2 (Fig. 1C) or an abrupt increase in \({\dot{\text{V}}}\)O2 (Fig. 1D).
Comparisons of \({\dot{\text{V}}}\)O2 changes in the success and failure groups, and representative \({\dot{\text{V}}}\)O2 kinetic changes in one success and two failure patients. (A) Changes in \({\dot{\text{V}}}\)O2 from the beginning to the end of the UBT were significant in the failure group but not in the success group. Three distinct kinetics of \({\dot{\text{V}}}\)O2 are demonstrated in this figure. (B) This is a patient who successfully completed the UBT. The \({\dot{\text{V}}}\)O2 remained stable during the whole UBT. (C) This is a patient who failed the UBT. The \({\dot{\text{V}}}\)O2 kept increasing along with the heart rate until the end of the UBT due to increasing heart rate > 20% and dropped after decompensation because the UBT was not stopped immediately. A slow increase in \({\dot{\text{V}}}\)O2 was the major failure pattern in the failure group. (D) Patient C had a steeper increase in \({\dot{\text{V}}}\)O2 during the UBT. The patient had impaired lung mechanics due to pleural effusion and poor lung compliance. The work of breathing may have kept increasing to cause the steeper increase in \({\dot{\text{V}}}\)O2.
Figure 2 shows comparisons of \({\dot{\text{V}}}\)O2 and other relevant parameters between the beginning and last 5-min periods of UBT in the two groups. In addition to \({\dot{\text{V}}}\)O2 (Figs. 1A and 2A), the failure group also had a significantly increased HR (beginning 5 vs. last 5 min, 98.0 vs. 109.4 beats/min; P = 0.018) and EE (Fig. 2L) (1551.9 vs. 1917.8 kcal/day; P = 0.012). In contrast, the success group had increased tidal volume (Fig. 2G) (290.1 vs. 306.3 ml; P = 0.006), increased ventilatory EqCO2 (Fig. 2J) (39.9 to 41.4; P = 0.027) and decreased EtCO2 (Fig. 2H) (36.7 vs. 36.4 mmHg; P = 0.002).
Comparisons of the measured and derived parameters between the beginning and end of the 120-min unassisted breathing trials. The Wilcoxon-Rank sum paired test was used to calculate changes at the first 5 min and last 5 min of the UBT. The \({\dot{\text{V}}}\)O2 (a), heart rate (c) and energy expenditure (l) increased significantly in the failure group but not in the success group. In contrast, the tidal volume (g) increased and the end-tidal CO2 (h) decreased significantly in the success group but not in the failure group. Meanwhile, the respiratory rate (2e) and minute ventilation (f) did not change in either group. CO2 output (\({\dot{\text{V}}}\)CO2) showed an increasing trend in the failure group but not significantly (b). Trends of decreases in the respiratory quotient (RQ) and ventilatory equivalent of oxygen (EqO2) were observed in most of the failure patients (i, k). The EqCO2 and oxygen pulse revealed no specific trend in either group (j, d).
Univariate analysis for the primary outcome revealed that PImax, decreased \({\dot{\text{V}}}\)O2, HR, EE, EtCO2, and increased EqO2 were significantly associated with 120-min UBT outcomes (Table 2S). We only entered PImax and % change of \({\dot{\text{V}}}\)O2 into the multivariate analysis model because there was high collinearity between the % change of \({\dot{\text{V}}}\)O2 and EE. The % change of HR, ventilatory EqCO2, and EtCO2 were also not included due to very low event numbers. The cutoff values of \({\dot{\text{V}}}\)O2 (> 17%) and PImax (greater than − 30 cmH2O) were determined by maximizing the Youden Index. Table 3 summarizes the results of multivariate logistic regression analysis for the primary outcome using \({\dot{\text{V}}}\)O2 and PImax as explanatory variables. We found that an increase of > 17% in \({\dot{\text{V}}}\)O2 in the beginning period (OR 0.084; 95% CI 0.012–0.600; P = 0.014) and a peak inspiratory pressure greater than − 30 cmH2O (OR 11.083; 95% CI 1.117–109.944; P = 0.04) were significantly associated with the success of 120-min UBT (Table 3).
To understand the predictive value of \({\dot{\text{V}}}\)O2-associated parameters combined with traditional physiological parameters, we compared the areas under the receiver operating characteristic (ROC) curves (AUCs) to predict the secondary outcome, i.e., reaching 48 h of unassisted breathing without meeting the criteria of weaning failure. Univariate analysis for the primary outcome revealed that decreased HR, EE, EtCO2, and increased EqO2 were associated with the success of achieving 48 h of unassisted breathing (Table 2S). Using the traditional definition of a failed spontaneous breathing trial as an increase in HR > 20% and increase in RR > 50%, resulted in a sensitivity of 0 to predict failure at 48 h. In our fragile prolonged MV patients, the traditional criteria for the failure of a spontaneous breathing trial were not sensitive enough to predict the long-term outcomes. Therefore, we included the parameters related to \({\dot{\text{V}}}\)O2 into the prediction model. The multivariate logistic regression analysis for 48-h outcomes including the percentage change of HR and the percentage RR showed an AUC of 0.7440, with an AIC score of 42.75. Combining the percentage change of HR, EE, EtCO2 and EqO2 resulted in a modest increase in the AUC to 0.7880 (P = 0.578) with a decrease in the AIC score to 41.83 to predict the 48-h outcome. The percentage change of \({\dot{\text{V}}}\)O2 was not associated with the 48-h outcome, and was therefore not included as a predictor (Fig. 3).
Receiver operating characteristic (ROC) curves of traditional and refined criteria for predicting success in achieving 48-h unassisted breathing. A refined criterion combining heart rate (HR), energy expenditure, end-tidal CO2 and oxygen equivalent showed increased area under the receiver operating characteristic curve (AUC) of 0.788 and decreased Akaike information criterion (AIC) score of 41.83, compared with traditional criteria including HR and respiratory rate (AUC = 0.744, P = 0.578, AIC score = 42.75) for achieving 48 h of unassisted breathing.
Discussion
In this study, we found differences in several parameters between the success and failure groups, and a significant association between an increase in \({\dot{\text{V}}}\)O2 during the session and failure to complete the trial. Derivative parameters of \({\dot{\text{V}}}\)O2 also showed good predictive value for 48-h UBT success. To the best of our knowledge, the prognostic significance of the kinetics of \({\dot{\text{V}}}\)O2 during open-circuit UBT in tracheostomized patients receiving prolonged MV has not previously been reported. We also showed the practical feasibility of open-circuit, continuous measurements and calculation of \({\dot{\text{V}}}\)O2 and relevant parameters during a 120-min UBT for tracheostomized patients receiving prolonged MV. Our findings suggest a possible model to understand the mechanism of weaning failure in patients with prolonged MV.
Reports in the literature have suggested the theoretical and practical potential of \({\dot{\text{V}}}\)O2 measurements to understand the level and kinetics of WOB in mechanically ventilated patients. In 1988, Jubran et al. continuously measured mixed venous oxygen saturation (SvO2) and calculated cardiac output using a pulmonary artery catheter in 19 ventilated patients who underwent a spontaneous breathing trial. They demonstrated that the patients who failed the spontaneous breathing trial had a decrease in SvO2, increase in O2 extraction ratio and decrease in O2 transport during the spontaneous breathing trial24. However, the measurements of O2 extraction, O2 transport, and mixed venous oxygen require the use of an invasive pulmonary artery catheter24. Developed decades ago, non-invasive, spirometry-derived, breath-to-breath \({\dot{\text{V}}}\)O2 measurements have been widely used to measure pulmonary gas exchange during exercise. The clinical estimation of WOB requires measuring the pressure–volume loop using invasive esophageal pressure monitoring25,26. To avoid such invasive monitoring, the concept of \({\dot{\text{V}}}\)O2 of respiratory muscles (\({\dot{\text{V}}}\)O2, resp) was proposed to represent the WOB, which is calculated as the difference between the \({\dot{\text{V}}}\)O2 measured during controlled MV and during spontaneous breathing27,28,29,30. The number of patients requiring prolonged MV continues to increase, and the relatively non-invasive technique used in this study to measure \({\dot{\text{V}}}\)O2 during a UBT may be of potential application in real-world care.
Our finding of an association between increased \({\dot{\text{V}}}\)O2 during a 120-min UBT and test failure may suggest that an increase in WOB is an underlying physiologic mechanism for failing a weaning session. Events that can develop during a weaning session include progressive atelectasis of air spaces with loss of end-expiratory lung volume, increasing retention of airway secretions with worsening airway resistance, and loss of coordination of the respiratory muscles resulting in unfavorable breathing movements. However, as these conditions are difficult to measure non-invasively during an open-circuit weaning period, the development of better tools is needed for a deeper understanding of these physiologic alterations. Our findings are also compatible with a previous study by Teixeira et al. who demonstrated increased WOB calculated using airway pressure measurements during a 120-min spontaneous breathing trial in patients before a failed extubation26, although they focused on endotracheally intubated patients rather than tracheostomized patients receiving prolonged MV. Previous reports have suggested that measuring WOB or \({\dot{\text{V}}}\)O2 may be a promising approach to assess the prognosis of MV weaning, and that increased WOB during a spontaneous breathing trial in endotracheally intubated patients may suggest a worse weaning outcome25,26. The \({\dot{\text{V}}}\)O2, resp-to-\({\dot{\text{V}}}\)O2 ratio has also been associated with weaning outcomes23, despite uncertain results, and several studies have reported an association between a ratio > 15% and extubation failure19,27,29,30,31,32,33,34,35,36. A previous study also suggested that the detection of an obvious increase in \({\dot{\text{V}}}\)O2 during a UBT without reaching a steady state may suggest increasing WOB33. We obtained similar results using a clinically feasible, non-invasive method. Moreover, this monitoring modality may also provide a practical guide to adjust the weaning process so that earlier termination of unassisted breathing may be considered before overt signs of weaning failure or exhaustion can be detected. Furthermore, it may be easier to make the decision to continue ongoing unassisted breathing in situations where the patient is showing subjective distress without clinical failure. Bridging measures to step down to unassisted breathing using a slower weaning process37, such as applying external continuous positive airway pressure to recruit the atelectatic area, and to remove airway secretions or accumulating pleural effusion, may also be provided earlier when the monitoring suggests a trend of increasing \({\dot{\text{V}}}\)O2.
We also demonstrated that a combination of physiologic parameters related to \({\dot{\text{V}}}\)O2 with traditional variables may provide better predictive value in weaning success. Traditional measurements of changes in heart and respiratory rates, despite being sensitive to detect UBT failure, are not specific to the underlying causes for respiratory failure. Several studies have suggested the potential application of these derived parameters in assessing weaning potential, such as failure to increase O2 pulse during exercise implies the inability to increase cardiac stroke volume, and that this is related to a poor prognosis in heart failure patients38,39. Other studies have shown a higher EqO2 in patients succeeding in spontaneous breath trials, and that a decreased EqO2 implies poor ventilatory compensation in prolonged mechanically ventilated patients40,41. Our observations, therefore, may implicate that incorporating the analysis of gas exchange during UBT may help to better predict the long-term outcomes of these patients. In this study, the traditional criteria to define a failed UBT, including HR increase > 20%, RR increase > 50% and EtCO2 increase > 8 mmHg, appeared to be less sensitive in the patients with prolonged MV. The combination including EE and EqO2 resulted in a higher AUC to predict the 48-h weaning outcomes, and therefore it may represent a promising prediction model. However, further studies are needed to validate our findings.
There are several limitations to this study. First, this was a single-center study which included only a small number of patients admitted to the RCC, with diverse causes of respiratory failure and clinical situations contributing to prolonged MV. The small sample size limits the generalizability of the study findings, and a further validation study is definitely needed. Nevertheless, the findings of this study may elucidate the possible reasons for clinical failure during a 120-min UBT session. Second, interpretation of the measured data may have been complicated by the presence of respiratory secretions, necessary clinical care activities, and background noise, making it difficult to measure the data over a long duration in this open-circuit real-time model. A possible alternative method would be multiple intermittent measurements, taking necessary interruptions into account. Third, the small number in the failure group (eight patients) meant that we could not perform multivariate analysis including more potential variables such as clinical characteristics and weaning parameters commonly measured before UBTs. Prospective studies including more patients in relatively specific patient groups may provide more insight into the kinetics of \({\dot{\text{V}}}\)O2 and the contribution to weaning failure in tracheostomized patients. Fourth, the main reason we chose successful 48-h unassisted breathing was that this study lacked serial daily measurements of \({\dot{\text{V}}}\)O2. As the primary outcome of this study was the results of a 120-min UBT, the design was limited by the fact that we did not take serial measurements from the same patient during repeated UBTs, which would have ideally been conducted every day. Therefore, we were unable to investigate the daily evolutional change in \({\dot{\text{V}}}\)O2 during serial daily UBTs, and we could only investigate the kinetics of \({\dot{\text{V}}}\)O2 during one UBT session. It is probable that some of the patients who initially had less promising \({\dot{\text{V}}}\)O2 data improved during the following days of training, and thus had improved \({\dot{\text{V}}}\)O2 data and at the same time a higher probability of successful weaning. Serial measurements might be needed to understand the association with successful weaning as defined by clinical criteria, such as unassisted breathing for at least 5 days. Further studies are needed to validate the prediction model we propose in this study. Finally, this study was based on a non-invasive design and thus did not calculate WOB traditionally using esophageal pressure measurements; therefore, the exact role of WOB in weaning failure, as suggested by \({\dot{\text{V}}}\)O2 in our study, requires further validation. Researchers may also consider measuring the airway pressure as an alternative method to calculate the WOB, as suggested by other researchers26, as complementary data to validate the predictability by \({\dot{\text{V}}}\)O2.
In conclusion, an increase in \({\dot{\text{V}}}\)O2 in tracheostomized patients who fail a UBT may provide a deeper insight into the understanding of weaning failure in patients with prolonged MV. Further large-scale studies are warranted to understand and validate the relationships among WOB, kinetics of \({\dot{\text{V}}}\)O2, and weaning outcomes.
Data availability
The datasets used and analyzed in the current study are available from the corresponding author on reasonable request.
References
MacIntyre, N. R. et al. Management of patients requiring prolonged mechanical ventilation: report of a NAMDRC consensus conference. Chest 128, 3937–3954. https://doi.org/10.1378/chest.128.6.3937 (2005).
Béduneau, G. et al. Epidemiology of weaning outcome according to a new definition. The WIND Study. Am. J. Respir. Crit. Care Med. 195, 772–783. https://doi.org/10.1164/rccm.201602-0320OC (2016).
Nelson, J. E., Cox, C. E., Hope, A. A. & Carson, S. S. Chronic critical illness. Am. J. Respir. Crit. Care Med. 182, 446–454 (2010).
Lone, N. I. & Walsh, T. S. Prolonged mechanical ventilation in critically ill patients: epidemiology, outcomes and modelling the potential cost consequences of establishing a regional weaning unit. Crit. Care 15, R102 (2011).
Dasta, J. F., McLaughlin, T. P., Mody, S. H. & Piech, C. T. Daily cost of an intensive care unit day: the contribution of mechanical ventilation. Crit. Care Med. 33, 1266–1271 (2005).
Jubran, A. et al. Effect of pressure support vs unassisted breathing through a tracheostomy collar on weaning duration in patients requiring prolonged mechanical ventilation: a randomized trial. JAMA 309, 671–677 (2013).
Boles, J.-M. et al. Weaning from mechanical ventilation. Eur. Respir. J. 29, 1033–1056 (2007).
Jubran, A. & Tobin, M. J. Pathophysiologic basis of acute respiratory distress in patients who fail a trial of weaning from mechanical ventilation. Am. J. Respir. Crit. Care Med. 155, 906–915 (1997).
Pinsky, M. R. Breathing as exercise: the cardiovascular response to weaning from mechanical ventilation. Intensive Care Med. 26, 1164–1166 (2000).
Hawkins, M. N., Raven, P. B., Snell, P. G., Stray-Gundersen, J. & Levine, B. D. Maximal oxygen uptake as a parametric measure of cardiorespiratory capacity. Med. Sci. Sports Exerc. 39, 103–107. https://doi.org/10.1249/01.mss.0000241641.75101.64 (2007).
Ross, R. M. ATS/ACCP statement on cardiopulmonary exercise testing. Am. J. Respir. Crit. Care Med. 167, 1451. https://doi.org/10.1164/ajrccm.167.10.950 (2003).
Weisman, I. M. & Zeballos, R. J. An integrated approach to the interpretation of cardiopulmonary exercise testing. Clin. Chest Med. 15, 421–445 (1994).
Zhang, Y. Y. et al. O2 uptake kinetics in response to exercise. A measure of tissue anaerobiosis in heart failure. Chest 103, 735–741 (1993).
Lajoie, C., Laurencelle, L. & Trudeau, F. Physiological responses to cycling for 60 minutes at maximal lactate steady state. Can. J. Appl. Physiol. 25, 250–261 (2000).
Wilkerson, D. P., Koppo, K., Barstow, T. J. & Jones, A. M. Effect of work rate on the functional “gain” of Phase II pulmonary O2 uptake response to exercise. Respir. Physiol. Neurobiol. 142, 211–223. https://doi.org/10.1016/j.resp.2004.06.001 (2004).
Sietsema, K. E., Ben-Dov, I., Zhang, Y. Y., Sullivan, C. & Wasserman, K. Dynamics of oxygen uptake for submaximal exercise and recovery in patients with chronic heart failure. Chest 105, 1693–1700 (1994).
Cohen-Solal, A. et al. Prolonged kinetics of recovery of oxygen consumption after maximal graded exercise in patients with chronic heart failure: analysis with gas exchange measurements and NMR spectroscopy. Circulation 91, 2924–2932 (1995).
Schalcher, C. et al. Prolonged oxygen uptake kinetics during low-intensity exercise are related to poor prognosis in patients with mild-to-moderate congestive heart failure. Chest 124, 580–586 (2003).
Shikora, S. A., Benotti, P. N. & Johannigman, J. A. The oxygen cost of breathing may predict weaning from mechanical ventilation better than the respiratory rate to tidal volume ratio. Arch. Surg. 129, 269–274. https://doi.org/10.1001/archsurg.1994.01420270045011 (1994).
Mancini, D. M. et al. Value of peak exercise oxygen consumption for optimal timing of cardiac transplantation in ambulatory patients with heart failure. Circulation 83, 778–786 (1991).
Wasserman, K., Hansen, J. E., Sue, D. Y., Stringer, W. W. & Whipp, B. J. Principles of exercise testing and interpretation: including pathophysiology and clinical applications, vol 206 (Lippincott Williams & Wilkins, Philadelphia, 1999).
Putensen, C. & Wrigge, H. Clinical review: biphasic positive airway pressure and airway pressure release ventilation. Crit. Care 8, 492. https://doi.org/10.1186/cc2919 (2004).
Bellani, G. et al. Increase of oxygen consumption during a progressive decrease of ventilatory support is lower in patients failing the trial in comparison with those who succeed. Anesthesiology 113, 378–385. https://doi.org/10.1097/ALN.0b013e3181e81050 (2010).
Jubran, A., Mathru, M., Dries, D. & Tobin, M. J. Continuous recordings of mixed venous oxygen saturation during weaning from mechanical ventilation and the ramifications thereof. Am. J. Respir. Crit. Care Med. 158, 1763–1769. https://doi.org/10.1164/ajrccm.158.6.9804056 (1998).
Fiastro, J. F., Habib, M. P., Shon, B. Y. & Campbell, S. C. Comparison of standard weaning parameters and the mechanical work of breathing in mechanically ventilated patients. Chest 94, 232–238. https://doi.org/10.1378/chest.94.2.232 (1988).
Teixeira, C., Teixeira, P. J., de Leon, P. P. & Oliveira, E. S. Work of breathing during successful spontaneous breathing trial. J. Crit. Care 24, 508–514. https://doi.org/10.1016/j.jcrc.2008.10.013 (2009).
Hubmayr, R. D., Loosbrock, L. M., Gillespie, D. J. & Rodarte, J. R. Oxygen uptake during weaning from mechanical ventilation. Chest 94, 1148–1155 (1988).
Shikora, S. A., MacDonald, G. F., Bistrian, B. R., Kenney, P. R. & Benotti, P. N. Could the oxygen cost of breathing be used to optimize the application of pressure support ventilation?. J. Trauma 33, 521–526 (1992) (discussion 526–527).
Weyland, W. et al. Oxygen cost of breathing for assisted spontaneous breathing modes: investigation into three states of pulmonary function. Intensive Care Med. 21, 211–217 (1995).
Miwa, K., Mitsuoka, M., Takamori, S., Hayashi, A. & Shirouzu, K. Continuous monitoring of oxygen consumption in patients undergoing weaning from mechanical ventilation. Respir. Int. Rev. Thorac. Dis. 70, 623–630. https://doi.org/10.1159/000075209 (2003).
Shikora, S. A. et al. Work of breathing: reliable predictor of weaning and extubation. Crit. Care Med. 18, 157–162 (1990).
Kemper, M., Weissman, C., Askanazi, J., Hyman, A. I. & Kinney, J. M. Metabolic and respiratory changes during weaning from mechanical ventilation. Chest 92, 979–983 (1987).
Mitsuoka, M., Kinninger, K. H., Johnson, F. W. & Burns, D. M. Utility of measurements of oxygen cost of breathing in predicting success or failure in trials of reduced mechanical ventilatory support. Respir. Care 46, 902–910 (2001).
Annat, G. J. et al. Oxygen cost of breathing and diaphragmatic pressure-time index. Measurement in patients with COPD during weaning with pressure support ventilation. Chest 98, 411–414 (1990).
Field, S., Kelly, S. M. & Macklem, P. T. The oxygen cost of breathing in patients with cardiorespiratory disease. Am. Rev. Respir. Dis. 126, 9–13. https://doi.org/10.1164/arrd.1982.126.1.9 (1982).
Mamary, A. J. et al. Survival in Patients Receiving Prolonged Ventilation: Factors that Influence Outcome. Clin. Med. Insights Circ. Respir. Pulm. Med. 5, 1. https://doi.org/10.4137/CCRPM.S6649 (2011).
Lin, S.-J. et al. Maximal expiratory pressure is associated with reinstitution of mechanical ventilation after successful unassisted breathing trials in tracheostomized patients with prolonged mechanical ventilation. PLoS ONE 15, e0229935. https://doi.org/10.1371/journal.pone.0229935 (2020).
Lavie, C. J., Milani, R. V. & Mehra, M. R. Peak exercise oxygen pulse and prognosis in chronic heart failure. Am. J. Cardiol. 93, 588–593. https://doi.org/10.1016/j.amjcard.2003.11.023 (2004).
Teboul, J. L., Monnet, X. & Richard, C. Weaning failure of cardiac origin: recent advances. Crit. Care 14, 211. https://doi.org/10.1186/cc8852 (2010).
Ellens, T., Kaur, R., Roehl, K., Dubosky, M. & Vines, D. Ventilatory equivalent for oxygen as an extubation outcome predictor: a pilot study. Can. J. Respir. Ther. 55, 65–71. https://doi.org/10.29390/cjrt-2019-007 (2019).
Lee, C.-S. et al. Hypercapnic ventilatory response in the weaning of patients with prolonged mechanical ventilation. Can. Respir. J. 7381424–7381424, 2017. https://doi.org/10.1155/2017/7381424 (2017).
Acknowledgements
This work was supported by National Taiwan University Hospital (Grant #107-N4064) and the Ministry of Science and Technology, Taiwan (Grants #108-2314-B-002-192 and #109WFA0111164).
Author information
Authors and Affiliations
Contributions
I.-H.L., Y.-W.K., J.-S.J. and H.-D.W. participated in the conception of the study and design of study methods. I.-H.L., Y.-W.K., F.-C.L., C.-W.W. and J.-C.C. participated in the collection of data. I.-H.L., Y.-W.K., J.-S.J., P.-H.K., Y.-C.C. and C.-K.H. participated in the analysis of data. I.-H.L., Y.-W.K. and J.-S.J. participated in manuscript preparation. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lee, IH., Kuo, YW., Lin, FC. et al. Kinetics of oxygen uptake during unassisted breathing trials in prolonged mechanical ventilation: a prospective pilot study. Sci Rep 10, 14301 (2020). https://doi.org/10.1038/s41598-020-71278-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-71278-2
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.