Abstract
Toxoplasmic encephalitis is an AIDS-defining condition. The decline of IFN-γ-producing CD4+ T cells in AIDS is a major contributing factor in reactivation of quiescent Toxoplasma gondii to an actively replicating stage of infection. Hence, it is important to characterize CD4-independent mechanisms that constrain acute T. gondii infection. We investigated the in vivo regulation of IFN-γ production by CD8+ T cells, DN T cells and NK cells in response to acute T. gondii infection. Our data show that processing of IFN-γ by these non-CD4 cells is dependent on both IL-12 and IL-18 and the secretion of bioactive IL-18 in response to T. gondii requires the sensing of viable parasites by multiple redundant inflammasome sensors in multiple hematopoietic cell types. Importantly, our results show that expansion of CD8+ T cells, DN T cells and NK cell by S4B6 IL-2 complex pre-treatment increases survival rates of mice infected with T. gondii and this is dependent on IL-12, IL-18 and IFN-γ. Increased survival is accompanied by reduced pathology but is independent of expansion of TReg cells or parasite burden. This provides evidence for a protective role of IL2C-mediated expansion of non-CD4 cells and may represent a promising lead to adjunct therapy for acute toxoplasmosis.
Similar content being viewed by others
Introduction
Toxoplasma gondii (T. gondii) is an obligate intracellular parasite of the phylum Apicomplexa1. It is estimated that one-third of the world’s population is infected with T. gondii. In most individuals, infection is asymptomatic and leads to chronic, life-long persistence of T. gondii-containing cysts, primarily in brain and muscle tissue2. Active disease, also known as toxoplasmosis, usually occurs after reactivation of encysted parasites, and is often associated with immunosuppression. If untreated, toxoplasmosis may be fatal. Additionally, serious eye disease has been reported as a result of infection with T. gondii3 and, if a primary infection occurs during pregnancy, abortion, stillbirth and fetal abnormalities can occur2,4. Whereas an acute infection is generally mediated by the fast-replicating tachyzoite stage of the parasite, the persistent tissue cysts, characteristic of a chronic infection, contain slow-replicating bradyzoites. Currently, treatment of toxoplasmosis is limited to the acute disease and requires prolonged exposure to anti-toxoplasmosis drugs for the duration of the immunosuppression5,6.
Containment of chronic T. gondii infection requires functional T-cell responses, in particular interferon gamma (IFN-γ)-producing CD4+ T cells2,7. In the absence of CD4+ T cells, IFN-γ, its receptor or downstream effector molecules, such as inducible nitric oxide synthase (iNOS), susceptibility and disease are severely exacerbated8,9,10,11. Accordingly, co-infection with human immunodeficiency virus (HIV), which impairs CD4+ T cells during its reproduction, is one of the major reactivation factors. In fact, toxoplasmic encephalitis accompanied by low numbers of CD4+ T cells is considered to be an AIDS-defining condition in HIV+ individuals12.
In addition to antigen-specific CD4+ T cells11, innate immune cells, such as NK cells and neutrophils also contribute significantly to the production of host-protective IFN-γ13,14. In particular, the recognition of T. gondii-derived profilin via Toll-like receptor (TLR)-11, which drives myeloid differentiation primary-response protein 88 (MyD88)-dependent IL-12 secretion by dendritic cells, is considered a crucial upstream pathway of protective IFN-γ secretion15,16. MyD88 or IL-12 knock-out mice are also susceptible to T. gondii infection17,18. Furthermore, elegant studies by Hunter and colleagues showed that T cell-intrinsic ablation of MyD88 also impacts severely on the control of the parasite19. These findings indicate that, in addition to IL-12, cytokine-driven IFN-γ secretion in response to T. gondii also relies on IL-18, an IL-1 family cytokine originally known as IFN-γ-inducing factor, which requires cell-intrinsic MyD88 signaling20,21. IL-18 is particularly important for the rapid secretion of IFN-γ by cells of the immune system, in particular NK cells, CD8+ memory T cells and double negative (DN) γδ T cells22.
Proteolytic cleavage of IL-18 from biologically inactive pro-IL-18 requires caspase-123 and the activation of cytosolic inflammasome sensors23. Deficiencies in caspase-1, IL-1824,25 and the inflammasome sensors NLRP1 and NLRP324,26 are associated with compromised immunity to T. gondi and several intracellular bacterial pathogens27. Hence, the positive impact of targeting IL-18-mediated IFN-γ production on protective immunity has been demonstrated in models of Listeria monocytogenes, Mycobacterium tuberculosis and Salmonella enterica infection28,29,30.
Given that control of acute toxoplasmosis depends on a delicate balance between limiting immunopathology and maintaining parasite killing, in the present study, we interrogated the regulation of IL-18-driven IFN-γ production in vivo. We discovered that bioactive IL-18 is dependent on the sensing of viable parasites by multiple redundant inflammasome sensors in multiple hematopoietic cell types, leading to the hypothesis that enhancement of this innate response could be harnessed to prevent disease resulting from infection with T. gondii. We therefore investigated if treatment with S4B6-containing IL2C, an IL2 complex that can boost NK and CD8+ T cell numbers31, could prevent acute lethal toxoplasmosis.
Results
Toxoplasma-driven IFN-γ secretion by non-CD4 immune cells following oral infection with brain cysts or intravenous (i.v.) infection with tachyzoites
Given that control of acute toxoplasmosis critically depends on IFN-γ7 and non-CD4 immune cell types, such as CD8+ T cells, DN T cells and NK cells, are prime IFN-γ producers, we wanted to delineate the mechanistic requirements of IFN-γ production by these cell types in response to T. gondii. We furthermore wanted to explore whether responses were similar after oral infection (a common natural route of infection), i.v. infection with tachyzoites (modelling blood transfusion, a rare but significant—for the individual—route of infection32 and the often used purely experimental i.p. route of infection with tachyzoites.
We first inoculated naïve B6 mice with 10, 40 or 100 T. gondii ME49 cysts and assessed IFN-γ production by viable splenic CD3+CD4+, CD3+CD8+, CD3+CD4–CD8– (DN) T cells and CD3–NKp46+ cells 1 day and 5 days after inoculation. Whereas no IFN-γ production was observed 1 day after inoculation, a significant increase in IFN-γ-secreting cells was detected at 5 days after inoculation in spleen (Fig. 1a,b), mesenteric lymph nodes (MLN) (Fig. 1e,f) and Peyer’s Patches PP (Fig. S1a,b). Up to 10% of CD8+ T cells and DN T Cells and up to 50% of all NK cells stained IFN-γ+, particularly following inoculation with 40 and 100 cysts.
Toxoplasma-driven IFN-γ secretion by non-CD4 immune cells following oral infection with brain cysts or intravenous (i.v.) infection with tachyzoites. (a,b) Percent of IFN-γ+ cells amongst total viable CD3+CD4+, CD3+CD8+, CD3+CD4–CD8– (DN) T cells and CD3–NKp46+ cells in the spleen 1 day (a) or 5 days (b) after B6 mice were inoculated orally with 10, 40 or 100 T. gondii ME49 brain cysts. (c) Percent of IFN-γ+ cells amongst total viable CD3+CD4+, CD3+CD8+, CD3+CD4–CD8– (DN) T cells and CD3–NKp46+ cells in the spleen 2, 24, 48 or 72 h after B6 mice were injected i.v. with 107 T. gondii ME49 tachyzoites. (d) Percent of IFN-γ+ cells amongst total viable CD3+CD4+, CD3+CD8+, CD3+CD4–CD8– (DN) T cells and CD3–NKp46+ cells in the spleen 24 h after B6 mice were injected i.p. with 107 T. gondii ME49 tachyzoites. (e,f) Percent of IFN-γ+ cells amongst total viable CD3+CD4+, CD3+CD8+, CD3+CD4–CD8– (DN) T cells and CD3–NKp46+ cells in mesenteric lymph nodes (MLN) 1 day (e) or 5 days (f) after B6 mice were inoculated orally with 10, 40 or 100 T. gondii ME49 brain cysts. (g) Percent of IFN-γ+ cells amongst total viable CD3+CD4+, CD3+CD8+, CD3+CD4–CD8– (DN) T cells and CD3–NKp46+ cells in MLN 2, 24, 48 or 72 h after B6 mice were injected i.v. with 107 T. gondii ME49 tachyzoites. (h) Percent of IFN-γ+ cells amongst total viable CD3+CD4+, CD3+CD8+, CD3+CD4–CD8– (DN) T cells and CD3–NKp46+ cells in MLN 24 h after B6 mice were injected i.p. with 107 T. gondii ME49 tachyzoites. (i–k) Serum concentrations of IL-6 (i), TNFα (j) and IL-10 (k) at 2–72 h after B6 mice were injected i.v. with 107 T. gondii ME49 tachyzoites. Results are presented as pooled data means ± SEM from at least two pooled independent experiments (n = 5–10 mice per group). See also Figure S1.
We also investigated if rapid IFN-γ production could be induced by inoculation with tachyzoites via the i.v. and i.p. routes using a short-term in vivo exposure model in which naïve B6 mice were exposed to T. gondii tachyzoites for a maximum of 72 h. When mice were injected i.v. or i.p. with 105 tachyzoites, no significant IFN-γ production could be seen in either spleen, MLN or PP within 72 h (Fig. S1e). However, i.v. or i.p. inoculation with 107 tachyzoites led to secretion of IFN-γ by CD3+CD8+, CD3+CD4–CD8– (DN) T cells and CD3–NKp46+ cells in spleen, MLN and PP as early as 2–24 h after inoculation (Fig. 1c,d, g, h; Fig. S1c,d), mirroring the results seen 5 days after a cyst inoculation (Fig. 1b,f). Importantly, at 24 h after tachyzoite inoculation, levels of other acute inflammatory mediators, such as IL-6, TNFα and IL-10, were almost indistinguishable from naïve mice (Fig. 1i–k), indicating that these cytokines were not impacting on protective IFN-γ responses 24 h after i.v. infection.
These results show that i.v., i.p. tachyzoite infections and oral brain cyst infections induce almost identical acute immune responses. Given that it is difficult to quantify the number of bradyzoites within brain cysts used for oral infection and, moreover, dissemination patterns following oral infection are erratic in individual mice33, we subsequently focused on IFN-γ secretion 24 h after i.v. injection of tachyzoites as our primary readout for further dissection of the underlying mechanistic requirements.
Rapid IFN-γ secretion in response to T. gondii requires IL-12 and IL-18
Whereas the role of IL-12 in IFN-γ secretion is well established for T. gondii2, rapid production of IFN-γ in response to other intracellular pathogens, such as S. enterica, L. monocytogenes and M. tuberculosis has also been linked to the upstream effects of IL-1828,29. To interrogate whether or not, and how early, IFN-γ secretion in response to T. gondii also requires IL-18, we exposed naïve B6 mice to T. gondii ME49 tachyzoites and treated the animals with neutralizing monoclonal antibodies (mAb) to IL-12, IL-18 or IL-12 and IL-18 immediately after inoculation. We focused on NK cells for these and subsequent experiments, since this was the cell type for which the highest proportion of cells stained positive for IFN-γ following inoculation with T. gondii (see Fig. 1). At 24 h after exposure, we assessed IFN-γ secretion by NK cells in the spleen ex vivo. Neutralization of IL-12 and IL-18 significantly reduced IFN-γ production, with IL-12 contributing approximately 50% and IL-18 approximately 30–40% of the response (Fig. 2a). Consistent with this, where IL-12 levels in the serum of infected mice peaked at approximately 2 h after inoculation, the levels of IL-18 mirrored those of IFN-γ for up to 72 h (Fig. 2b). The significant reduction of rapid IFN-γ production in Il18–/– mice, and the almost complete absence of rapid IFN-γ production in anti-IL-12-treated Il18–/– mice, further confirmed a direct correlation between IL-12, IL-18 and IFN-γ secretion (Fig. 2c,d). Furthermore, treatment with anti-IL-12 and/or anti-IL-18 also reduced concentrations of IFN-γ, IL-12 and IL-18 in the serum of infected mice in an additive manner (Fig. 2d–f). These results suggest a hierarchical relationship in which a primary IL-12-driven IFN-γ response is followed by an IL-18-dominant IFN-γ response. We concluded that innate IFN-γ secretion in response to T. gondii is driven by the secretion of IL-12 and IL-18.
Rapid IFN-γ production in response to T. gondii requires IL-12 and IL-18. (a) Percent of IFN-γ+ cells amongst total viable CD3–NKp46+ cells in the spleen 24 h after B6 mice were injected i.v. with 107 T. gondii ME49 tachyzoites. Some mice received an i.p. injection of 200 µg mAb against IL-18 and/or IL-12 immediately after injection of T. gondii. (b) Serum concentrations of IL-18, IL-12p70 and IFN-γ at various time points after B6 mice were injected i.v. with 107 T. gondii ME49 tachyzoites. (c) Percent of IFN-γ+ cells amongst total viable CD3–NKp46+ cells in the spleen 24 h after B6 or Il18−/− mice were injected i.v. with 107 T. gondii ME49 tachyzoites. Some mice received an i.p. injection of 200 µg mAb against IL-12 immediately after injection of T. gondii. (d) Serum concentrations of IL-18 24 h after B6 or Il18−/− mice were injected i.v. with 107 T. gondii ME49 tachyzoites. Some mice received an i.p. injection of 200 µg mAb against IL-12 immediately after injection of T. gondii. (e) Serum concentrations of IFN-γ 24 h after B6 or Il18−/− mice were injected i.v. with 107 T. gondii ME49 tachyzoites. Some mice received an i.p. injection of 200 µg mAb against IL-18 and/or IL-12 immediately after injection of T. gondii. (f) Serum concentrations of IL-12 24 h after B6 or Il18−/− mice were injected i.v. with 107 T. gondii ME49 tachyzoites. Some mice received an i.p. injection of 200 µg mAb against IL-18 and/or IL-12 immediately after injection of T. gondii. Results are presented as individual data points (a,c,d,e,f) or as means ± SEM (b) of 4–15 mice per group from at least two pooled independent experiments. Statistical analyses: One-way ANOVA followed by Dunnett’s multiple comparison test. Significant differences are indicated by asterisks: *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001.
IL-18-driven IFN-γ secretion to T. gondii depends on multiple redundant inflammasomes
Given that the molecular mechanisms that lead to T. gondii-mediated IL-12 secretion are well characterized, we focused our attention on the host signaling pathways required for IL-18-driven IFN-γ production, using a panel of genetically modified mouse strains. Secretion of bioactive IL-18 depends on the enzymatic cleavage of pro-IL-18 by caspase-123. Activation of caspase-1 involves the sensing of danger molecules or stress signals via upstream cytosolic PRRs, so called inflammasomes, a process that can be enhanced and controlled via TRIF-dependent caspase-11 activation. A significantly increased percentage of IFN-γ+ NK cells was seen in Caspase1/11–/– double KO mice, Nlrp1−/−mice, Nlrp3−/− mice and heterozygous Nlrp1+/−Nlrp3+/−, Nlrp1−/−Nlrp3+/− and Nlrp1+/−Nlrp3−/− mice infected with T. gondii versus uninfected mice, and this response could be almost completely prevented by additional anti-IL-12 treatment (Fig. 3a, Table S1). Caspase1/11–/– double KO mice, Nlrp1−/−mice and Nlrp3−/− mice produced statistically significantly less IFN-γ following injection with T. gondii ME49 tachyzoites compared with B6 mice but, counter-intuitively, heterozygous Nlrp1+/−Nlrp3+/−, Nlrp1−/−Nlrp3+/− and Nlrp1+/−Nlrp3−/− mice did not; this may indicate a statistical rather than biological significance to these particular data, the key observation being that all mice are capable of generating significant numbers of IFN-γ+ cells.
IL-18-driven IFN-γ secretion to T. gondii depends on multiple redundant inflammasomes. (a) Percent of IFN-γ+ cells amongst total CD3–NKp46+ cells in the spleen 24 h after i.v. injection of 107 T. gondii ME49 tachyzoites into B6 mice and different mouse strains lacking either Caspase1/11, Nlrp1, Nlrp3 or Nlrp1 and Nlrp3. (b) Serum IL-18 concentrations 24 h after i.v. injection of 107 T. gondii ME49 tachyzoites into B6 mice and different mouse strains lacking either Caspase1/11, Nlrp1, Nlrp3 or Nlrp1 and Nlrp3. Results are presented as individual data points of 3–25 mice per group from at least two pooled independent experiments. Statistical analyses: Two-way ANOVA followed by Tukey’s post hoc test. Significant differences within each mouse genotype are indicated by asterisks: *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001; n.s. not significant. Further significance values are shown in Tables S1 and S2.
As expected, Caspase1/11–/– mice did not secrete significant levels of IL-18 following T. gondii inoculation (Fig. 3b), indicating that the remaining IFN-γ response in Caspase1/11–/– mice is driven by IL-12. Surprisingly, when we tested mice deficient in the upstream NLR family pyrin domain-containing proteins 1 and 3 (NLRP1 and NLRP3), NLR molecules that had been implicated previously in recognition of T. gondii24, both knockout strains secreted indistinguishable amounts of IL-18 compared with B6 mice (Fig. 3b; Table S2). This data suggested a redundant role for NLRP1 and NLRP3. However, even double knockout and heterozygous Nlrp1+/−Nlrp3+/−, Nlrp1−/−Nlrp3+/− and Nlrp1+/−Nlrp3−/− mice secreted high levels of IL-18 after exposure to T. gondii ME49 tachyzoites (Fig. 3b; Table S2), suggesting that additional PRR molecules must be involved in sensing of T. gondii invasion in vivo. Taken together these results indicate that rapid IFN-γ secretion in vivo in response to T. gondii depends on the inflammasome → caspase-1→ IL-18 axis, and that T. gondii likely activates at least three different inflammasomes in vivo.
Toxoplasma gondii activates inflammasomes in multiple cell types
To further investigate the role of cytosolic PRRs in sensing T. gondii invasion, and to potentially target inflammasome activation for preventive or therapeutic intervention strategies, we next tried to identify the T. gondii-sensing cell type in vivo. To do this, we made use of a red fluorescent protein (RFP) tagged T. gondii ME49 (T. gondii ME49-RFP) strain to track parasite uptake by different immune cell subsets in the spleen. Twenty-four hours after tachyzoite injection, T. gondii ME49-RFP also induced rapid IFN-γ secretion by splenic CD3+CD4+, CD3+CD8+, CD3+CD4–CD8– (DN) T cells and CD3–NKp46+ cells (Fig. 4a) and high levels of serum IL-18 (Fig. 4b), similar to wild-type T. gondii ME49 (see Figs. 1 and 2). Approximately 0.5% of all splenocytes contained T. gondii ME49-RFP in vivo 24 h after inoculation (Fig. 4c). Sorted RFP+ cells secreted significantly more IL-18 ex vivo compared to RFP− cells (Fig. 4d), and further surface phenotyping revealed that T. gondii ME49-RFP was primarily contained in monocytes, neutrophils and CD8α+ dendritic cells (Fig. 4e,f). Splenic MHC-II+CD11c+ DCs, CD11b+Ly6G+ neutrophils and CD11b+Ly6C+ monocytes each comprised approximately 20–30% of all RFP-containing cells after i.v. tachyzoite injection. Only very few T cells, B cells and macrophages appeared to harbor parasites (Fig. 4e,f). To investigate if cell types that contained T. gondii ME49-RFP parasites also activated inflammasomes, we performed intracellular staining for the inflammasome adaptor molecule apoptosis-associated speck-like protein containing a carboxy-terminal CARD (ASC), and measured the activation of caspase-1 with a fluorescent inhibitor that only binds to activated caspase-1 (FLICA FAM-YVAD-FMK)29. Consistent with the uptake of T. gondii ME49-RFP by different cell types, T. gondii ME49-RFP parasite-harboring neutrophils, monocytes and DCs also expressed higher levels of ASC and FAM-YVAD compared with RFP− cells and FMO controls (Fig. 4g). Collectively, these results indicate that T. gondii infection activates multiple redundant inflammasomes in multiple different hematopoietic cell-types in vivo.
T. gondii activates inflammasomes in multiple cell types. (a) Percent of IFN-γ+ cells amongst total viable splenic CD3+CD4+, CD3+CD8+, CD3+CD4–CD8– (DN) T cells and CD3–NKp46+ cells in naïve mice 24 h after i.v. injection of 107 T. gondii ME49-RFP tachyzoites. (b) Serum IL-18 levels in naïve mice 24 h after i.v. injection of 107 T. gondii ME49-RFP tachyzoites. (c) Representative FACS plots showing total viable splenic RFP+ cells 24 h after i.v. injection of 107 T. gondii ME49-RFP tachyzoites. (d) IL-18 levels in supernatant of sorted RFP+ and RFP− cells after incubation at 37 °C for 24 h. (e) Representative FACS plots showing gated RFP+ neutrophils. Monocytes, macrophages, dendritic cells (DCs) and T/B cells 24 h after i.v. injection of 107 T. gondii ME49-RFP tachyzoites. (f) Enumeration of RFP+ cell types shown in (e). (g) Representative histograms of cell type-specific gated RFP+ and RFP− cells showing expression levels of ASC (left panels) or FAM-YVAD (right panels) 24 h after i.v. injection with 107 T. gondii ME49-RFP tachyzoites. FMO control for ASC panels are cells from infected animals that did not get stained with anti-ASC-Alexa488 but all other antibodies. FMO control for FAM-YVAD are cells from mice that were injected with T. gondii ME49-RFP but did not receive an injection with FLICA FAM-YVAD. MFI values ± SD for FAM-YVAD expression in RFP+ and RFP− neutrophils, monocytes and DCs from three mice shown in table. Results are presented as individual data points (d,f), pooled data means ± SEM (a,b) and representative FACS plots (c,e) and histograms (g) of 6–9 mice from two or three pooled independent experiments. Statistical analyses: One-way ANOVA followed by Dunnett’s multiple comparison test (a) or Student’s t-test (b,d,g); significant differences are indicated by asterisks: *p < 0.05; **p < 0.01; ***p < 0.001.
IL-18-driven IFN-γ secretion to T. gondii depends on viable parasites but is independent of secreted GRA proteins
Next, we assessed if rapid IFN-γ secretion in response to T. gondii required viable parasites or could be induced by soluble factors. To this end, naïve B6 mice were injected with either live, heat-killed or sonicated T. gondii ME49 tachyzoites. Inoculation with live parasites induced significantly increased IFN-γ secretion by NK cells and increased serum IL-18 levels compared to heat-killed or sonicated parasites (Fig. 5a,b). To exclude the possibility that heat inactivation and sonication destroyed soluble factors that could potentially drive this response, we also injected naïve B6 mice with HFF cell debris, which had been re-suspended in the T. gondii ME49 culture supernatant. This treatment also failed to induce IFN-γ and IL-18 secretion (Fig. 5a,b). These results indicated that viable parasites are required to initiate an IFN-γ response, suggesting that T. gondii virulence factors may play a critical role. Evidence from studies that have investigated the mechanistic framework of how intracellular bacterial pathogens activate inflammasomes in vivo, suggests that secreted effector molecules and/or distinct structural proteins are critically required34. Apicomplexan parasites also secrete effector molecules with distinct host-modulatory properties35. In particular, dense granule (GRA) proteins have been shown to play important roles in the maintenance of the parasitophorous vacuole (PCV), for the intracellular lifestyle and to exert host-modulatory functions36. We further probed the parasite-derived factors that might drive early, IL-18-dependent IFN-γ secretion by exposing naïve B6 mice to a panel of T. gondii strains to test if GRA proteins are required for IL-18-driven IFN-γ secretion. Hence, we infected mice with a mutant strain of T. gondii ME49 that lacks ASP5, a critical requirement for secretion of GRA proteins37, as well as strains lacking GRA20 or GRA23, two proteins that contain the PEXEL motif required for PCV exit. No significant difference in the levels of serum IL-18 and NK cell-produced IFN-γ was observed 24 and 48 h after inoculation with T. gondii ME49 ASP5-deficient tachyzoites compared with inoculation of a wildtype T. gondii ME49 (Fig. 5c,d), suggesting that ASP5-driven GRA export is dispensable for inflammasome activation. Similarly, inoculation with GRA20-deficient or GRA23-deficient parasites did not significantly reduce IFN-γ secretion in the absence of IL-12 (Fig. S2). We also tested another Type II T. gondii isolate, DEG (T. gondii DEG), which had been implicated in reduced IL-1β secretion following in vitro infection of macrophages24 but, similar to inoculation with T. gondii ME49 ASP5-deficient parasites, inoculation with T. gondii DEG did not lead to reduced levels of serum IL-18 and NK cell-produced IFN-γ in this model (Fig. 5c,d). At 48 h after DEG tachyzoite inoculation, the levels of serum IL-18 were, if anything, slightly higher compared with inoculation of T. gondii ME49, though this was not statistically significant (Fig. 5d). These data indicate that ASP5-dependent secretion of GRA proteins does not affect IL-18-driven IFN-γ secretion in vivo and highlights the diverging mechanisms that underlie in vitro IL-1β and in vivo IL-18 secretion in response to T. gondii.
IL-18 driven IFN-γ secretion to T. gondii depends on parasite viability but is independent of secreted GRA proteins. (a) Percent of IFN-γ+ cells amongst total viable splenic CD3–NKp46+ cells in naïve mice 24 h after i.v. injection of live 107 T. gondii ME49, heat-killed or sonicated ME49 tachyzoites, or HFF debris with culture supernatant. (b) Serum IL-18 levels in naïve mice 24 h after i.v. injection of live 107 T. gondii ME49 heat-killed or sonicated ME49 tachyzoites, or HFF debris with culture supernatant. (c) Percent of IFN-γ+ cells amongst total viable splenic CD3–NKp46+ cells in naïve mice 24 and 48 h after i.v. injection of live 107 T. gondii ME49, DEG or ME49ΔASP5 tachyzoites.(d) Serum IL-18 levels in naïve mice 24 and 48 h after i.v. injection of live 107 T. gondii ME49, DEG or ME49ΔASP5 tachyzoites. Results are presented as individual data points of 4–15 mice per group from at least two pooled independent experiments. Statistical analyses: One-way ANOVA per time-point followed by Dunnett’s multiple comparison test; significant differences are indicated by asterisks: ***p < 0.001; ****p < 0.0001; n.s. not significant. See also Figure S2.
IL2C treatment expands IL-18-responsive IFN-γ-secreting cell subsets
Collectively, the results presented so far raise the prospect that, if the ability of non-CD4 cells to invoke inflammasome-dependent, IL18-driven production of IFN-γ can be enhanced, it may be possible to control acute toxoplasmosis in AIDS. Hence, we investigated if targeted expansion of non-CD4 cells with IL2C treatment can achieve this. First, naïve mice were treated i.p. with IL2C complex on four consecutive days (Fig. 6a) and, 24 h after the last IL2C injection, immune cell expansion was assessed by flow cytometry relative to untreated animals. As reported previously38, IL2C treatment led to a significant expansion of memory CD8+ T cells, NK cells and DN T cells in spleen and MLN (Fig. 6b,c) though increases observed in the PP were not statistically significant (Fig. 6d). Next we assessed if IL2C-expanded and non-expanded CD8+ T cells, DN T cells and NK cells responded similarly to T. gondii infection. IL2C-treated and untreated mice were infected with 107 ME49 tachyzoites for 24 h (Fig. 6a). The percentage of CD8+ T cells, DN T cells and NK cells producing IFN-γ was almost indistinguishable between IL2C-treated and untreated mice (Fig. 6e). The number of IFN-γ+ NK cells, IFN-γ+ CD8+ T cells and IFN-γ+ DN T cells increased 3–30 fold following IL2C treatment (Fig. 6f). Similarly, IL2C pretreatment significantly increased systemic IFN-γ levels in the serum after i.v. infection (Fig. 6g) but, as expected, did not lead to a significant change in the levels of serum IL-18 (Fig. 6h). We also assessed the expression of IL18R and IL12R on the surface of IFN-γ+ and IFN-γ− cells. IFN-γ+ NK cells (data for CD8+ T cells and DN T cells not shown) expressed significantly higher levels of IL18R and IL12R compared to IFN-γ− NK cells (Fig. 6i,j). Taken together, these results show that IL2C-expanded cells respond similarly to non-expanded cells and that the effect of IL2C treatment is to numerically expand IFN-γ producing cells that maintain higher IL-18R and IL-12R levels of expression compared to IFN-γ− cells.
IL2C treatment expands IL-18-responsive IFN-γ-secreting cell subsets. (a) Experimental plan showing that naïve B6 mice were treated i.p. with IL2C on four consecutive days. One day after the last administration, some mice were euthanized to isolate cells from spleens, mesenteric lymph nodes (MLN) and Peyer’s Patches (PP). Other mice were injected i.v. with 107 T. gondii ME49 tachyzoites 2 days after the last IL2C treatment mice and cells were isolated 24 h later. (b–d) Numbers of CD3+CD8+, CD3+CD4–CD8– (DN) and CD3−NKp46+ cells (enumerated by FACS) in spleens (b), MLN (c) and PP (d) of naïve B6 mice that were treated i.p. with IL2C on four consecutive days and euthanased 1 day after the last administration. (e,f) Proportions (e) and total numbers (f) of CD3–NKp46+ CD3+CD4+, CD3+CD8+ and CD3+CD4–CD8– (DN) IFN-γ+ cells (assessed by FACS) in spleens of B6 mice that were treated i.p. with IL2C on four consecutive days and, 2 days after the last IL2C treatment, were injected i.v. with 107 T. gondii ME49 tachyzoites and cells isolated 1 day later. (g,h) IFN-γ (g) and IL-18 (h) serum concentrations of B6 mice that were treated i.p. with IL2C on four consecutive days and, 2 days after the last IL2C treatment, were injected i.v. with 107 T. gondii ME49 tachyzoites, and serum collected 1 day later. (i,j) Expression of IL18R (i) and IL12R (j) on IFN-γ− (blue histogram) and IFN-γ+ CD3–NKp46+ cells after i.v. infection with 107 T. gondii ME49 tachyzoites with (orange histogram) or without (red histogram) IL2C treatment. Results are presented as pooled data means ± SEM from at least two pooled independent experiments with 5–6 mice per group (b,c,d,e,f), with individual data points shown for (g,h), and as representative histograms and individual data points of mean fluorescent intensity for (i,j). Statistical analyses: One-way ANOVA followed by Dunnett’s multiple comparison test (a,b,c,d,e,f); significant differences are indicated by asterisks: * p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001; n.s. not significant.
IL2C pre-treatment protects mice from acute lethal toxoplasmosis independently of TReg expansion and parasite burden
To definitively assess if IL2C-mediated expansion of IL-18-responsive IFN-γ-secreting non-CD4 cell subsets can prevent lethal toxoplasmosis in mice, we used the well-established oral inoculation model with T. gondii ME49 bradyzoite-containing brain cysts. As above, naïve B6 mice were treated i.p. with IL2C for four consecutive days (Fig. 7a). IL2C treatment was accompanied by a weight loss from which mice recovered within a few days (data not shown). Forty-eight hours after the last IL2C treatment, mice were inoculated orally with 10 or 40 T. gondii ME49 cysts and were assessed for weight loss and consequent survival, whereby mice were euthanized when weight loss exceeded 20% of body weight, in accordance with Animal Ethics Committee of James Cook University Approvals A2138 and A2324. All mice that had been inoculated with 40 cysts and 87% of mice that had been inoculated with 10 cysts, but had not received IL2C injections, succumbed within 14 days after inoculation (Fig. 7b,c). In contrast, IL2C pre-treatment extended survival in mice that had been inoculated with 40 cysts up to 36 days, and approximately 40% of mice that had been inoculated with 10 cysts survived until day 60 (Fig. 7b,c).
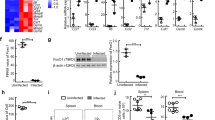
IL2C pre-treatment protects mice from acute, lethal toxoplasmosis independently of TReg expansion and parasite burden. (a) Experimental plan showing that naïve B6 mice were treated i.p. with IL2C on four consecutive days or left untreated. Two days after the last IL2C treatment, mice were inoculated orally with 10 or 40 T. gondii ME49 brain cysts and survival was assessed over time. (b,c) Percent survival of B6 mice, untreated or treated with IL2C before being inoculated orally with 40 (b) or 10 (c) T. gondii ME49 brain cysts. (d) Experimental plan showing that naïve B6 mice were treated as described in (a) with IL2C-treated animals receiving weekly i.p. injections with mAb and survival being assessed over time. (e,f) Percent survival of B6 mice, inoculated orally with 10 T. gondii ME49 brain cysts, untreated or treated with IL2C and mAb against IFN-γ, CD8, NK1.1, Thy1.2 or control rIgG (e), or with mAb against IL-12, IL-18, IFN-γ or control rIgG (f). (g,h) Gross pathology of the intestines (g) or livers (h) and parasite burden in the spleen (i) and IL-10 levels in the serum (j) of B6 mice, untreated or treated with IL2C, 2, 4 and/or 9 days after being inoculated orally with 10 T. gondii ME49 brain cysts. (k) Representative FACS plots of CD3+CD4+CD25+Foxp3+ regulatory T cells from MLN and lamina propria (LP) of B6 mice, untreated or treated with IL2C, 2 days after being inoculated orally with 10 T. gondii ME49 brain cysts. (l,m) Numbers of regulatory T cells from MLN (l) and LP (m) of B6 mice, untreated or treated with IL2C, 2, 4 or 9 days after being inoculated orally with 10 T. gondii ME49 brain cysts. Results are presented as individual data points (g,h,i), pooled data means (b,c,e,f,j,l,m) or representative FACS plots (k) from two to three pooled independent experiments with 5–15 mice per group. Statistical analyses: One-way ANOVA followed by Dunnett’s multiple comparison test (g,h,l,m), Student’s t-test (i) or Log-rank (Mantel–Cox) test (b,c,e,f); significant differences are indicated by asterisks: *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001. BDL, below detection limit.
Importantly, depletion of NK cells, CD8+ T cells, Thy1.2+ cells (expressed on all T cells and immature NK cells39 or IFN-γ from mice that had been treated with IL2C for four days and had been inoculated with 10 T. gondii ME49 cysts with neutralizing antibodies reversed IL2C-mediated increase in survival (Fig. 7d,e), indicating that IL2C-mediated cell expansion directly correlated with increased survival. Similarly, neutralization of IL-18, IL-12 or IFN-γ, reversed the protective phenotype (Fig. 7d,f). All mice that were not treated with IL2C succumbed to the infection by day 16, with a median survival of 11 days (Fig. 7f). Whilst 67% of IL2C-treated mice that received control rat IgG survived until day 60, the median survival for mice treated with anti-IFN-γ was 10.5 days, 13 days for mice treated with anti-IL-12 and 14 days for mice treated with anti-IL-18 (Fig. 7f). All mice that survived until day 60 were assessed for T. gondii brain cysts and all contained cysts in their brain, indicating that all mice were infected and that survival was not due to a failure of the infection to establish.
To assess if IL2C pre-treatment also impacts on measurable disease parameters other than survival, we also assessed pathology, parasite burden, serum cytokine levels and TReg numbers in MLN and lamina propria (LP) at 2, 4 and 9 days following oral cyst infection. At 9 days after infection, IL2C pre-treated mice displayed significantly reduced gross pathology of gut and liver (Fig. 7g,h) in the absence of any effect on parasite burden (Fig. 7i); thus, parasite burden was below detection limits in IL2C-treated and untreated animals at 2 days after infection and progressively increased at the same rate in both groups through days 4 and 9 post-infection (Fig. 7i). Due to the low infectious dose of 10 cysts, inflammatory cytokines such as IFN- γ, IL-6, IL-12 and TNF were not detectable in any mice at 2 and 4 days after infection and, although detectable at day 9 post-infection, there was no significant difference in the levels of these cytokines in IL2C-treated versus untreated mice (data not shown). Similarly, systemic IL-10 levels (Fig. 7j) and TReg numbers in MLN and lamina propria were not increased by IL2C injections (Fig. 7k–m) suggesting a role for IL2C pre-treatment independent of the previously reported TReg expansion with JES6-1A12-containing IL2C40,41. Collectively, these results demonstrate a protective role of IL2C pre-treatment in acute lethal murine toxoplasmosis that is dependent on IL-12, IL-18 and IFN-γ but is independent from effects on parasite burden.
Discussion
Non-CD4 cells, such as CD8+ T cells, DN T cells and NK cells, have been implicated in early control of severe infections with intracellular pathogens, including T. gondii, M. tuberculosis and Salmonella2,29. Our study provides a mechanistic framework for how T. gondii activates IFN-γ secretion by protective CD8+ T cells, DN T cells and NK cells. In particular, we demonstrate that in vivo IL-18-driven IFN-γ secretion in response to T. gondii likely requires the activation of at least three different inflammasomes. The involvement of both NLRP1 and NLRP3 has been shown in other, distinct models of toxoplasmosis24 but, in the model presented here, only Caspase1/11−/− mice but not Nlrp1−/−, Nlrp3−/− and Nlrp1±/−Nlrp3±/− mice were devoid of circulating IL-18 after T. gondii infection. These results suggest that a third sensor for in vivo T. gondii detection must exist in addition to NLRP1 and NLRP324,42. This conclusion is underscored by the fact that Caspase1/11−/− mice but not Nlrp1−/−, Nlrp3−/− and Nlrp1±/−Nlrp3±/− mice all maintained significant levels of IFN-γ+ NK cells after 24 h infection with T. gondii. Furthermore, we show that inflammasome activation occurred in CD8α+ DCs, inflammatory monocytes and neutrophils, cell types that have also been implicated in IL-12 secretion in response to T. gondii2. These results imply a high level of redundancy in the cell type that senses T. gondii infection as well as in the host inflammasome signaling pathway. This is in contrast to the often very specific recognition of viral and bacterial infections by one particular inflammasome in a distinct cell type28,29,43,44,45,46. It is likely that this divergence highlights the evolutionary complexity of parasites and suggests that more highly evolved organisms have developed a more complex inflammasome-dependent interplay with their hosts. In line with this hypothesis, it was shown recently in vitro that T. gondii also activates the NLRC4 and AIM2 inflammasomes in human fetal small epithelial cells47, as well as the expression of NLRP6, NLRP8 and NLRP13 in THP-1 macrophages48. Given that myeloid cell subsets often express distinct arsenals of PRRs on the cell surface and intracellularly, their ability to recognize and interact with T. gondii differs. Subsequently, the identification and characterization of distinct myeloid cell types producing IL-18 in response to T. gondii may foster innovative strategies for targeted interventions.
Toxoplasma gondii appears to activate both NLRP1 and NLRP324, yet the specificity of this activation remains elusive. While NLRP3 activation in response to T. gondii is influenced by P2X7 receptor-dependent potassium efflux and the induction of reactive oxygen species47,49,50,51, the exact mechanisms of how T. gondii activates multiple inflammasomes remain enigmatic. It is also interesting to note that in vitro infection of mouse macrophages and human monocytes with T. gondii only leads to the secretion of IL-1β, but not IL-1824,52. In contrast, in vivo infection in mice leads to significant secretion of IL-18 but not IL-1β24. It has even been suggested that in vitro infection of human neutrophils leads to evasion of NLRP3 activation and IL-1β secretion53. Furthermore, in vitro activation of inflammasomes differs between T. gondii strains, and is predominantly induced by Type II parasites24. These findings suggest that T. gondii has evolved sophisticated diverging effector mechanisms to manipulate inflammasome biology in different host cell subsets, and suggest that secreted effector molecules and/or distinct structural proteins may underlie inflammasome activation. It is, therefore, interesting that Nlrp1±/−Nlrp3±/− mice did not show reduced IL-18 secretion after infection with T. gondii. It is important to note that in mice the Nlrp1 locus is on the same chromosome as the Nlrp3 gene, meaning that the generation of rare double knockout offspring relies on recombination rather than inheritance. It will therefore be important to further investigate the role of Nlrp1 and 3 with alternative methods, such as CRISPR/Cas9 and/or chemical inhibition.
Our study has ruled out ASP5-dependent GRA proteins37, the most abundant family of T. gondii-derived effector molecules35, as the primary activator of inflammasomes. GRA molecules influence several host cell pathways54 and are required for the transport of small molecules across the parasitophorous vacuole55. These results do not exclude GRA proteins that don’t depend on ASP5 for export, and further studies will have to investigate the role of ASP5-independent GRA proteins as well as rhoptry proteins and other surface structures in driving this process. In particular, the recently described MYR1 protein export system56,57,58 may be valuable in answering if secreted effector molecules are at all required to initiate inflammasome activation.
It is tempting to speculate that the overall purpose of activating multiple inflammasomes in multiple cell types is to drive an inflammatory host response that mediates the progression of T. gondii into the chronic cyst phase, while at the same time preventing activation of parasite- killing mechanisms. Toxoplasma can invade and replicate in virtually all nucleated cell types of warm-blooded animals. From an evolutionary perspective, it is not surprising that the arms race between the host and the parasite has led to the evolution of numerous strategies to activate the immune system (from the parasite’s perspective) and to sense the invasion (from the host’s perspective). The fundamental differences between the habitats and the composition of the immune system of susceptible warm-blooded host species may require T. gondii to activate as many different inflammasome sensors as possible. It is well established that T. gondii requires a pro-inflammatory, IFN-γ-dominated immune response to form cysts7. Because transmission is critical for the parasite’s survival and completion of the life cycle, it is maladaptive for T. gondii to kill its host. This may explain why IFN-γ neutralization is fatal, because IFN-γ deficiency favors tachyzoite replication and prevents cyst formation. Furthermore, these findings may also explain why T. gondii cysts reactivate after HIV co-infection in humans; HIV destroys CD4+ T cells, a prime IFN-γ producer. Hence, we reasoned that a viable adjunct therapy in T. gondii/ HIV co-infection might be achieved by boosting IFN-γ-producing NK cells, CD8+ T cells and DN T cells to prevent acute toxoplasmosis.
Interleukin-2 and Interleukin-15 are critical cytokines for the maturation and survival of IL-18 responsive DN T cells, NK cells and CD8+ T cells59,60,61,62,63,64. The role of IL-15 in immunity to T. gondii remains controversial65,66, but IL-2 knockout mice are highly susceptible to T. gondii infection67, and injection of recombinant IL-2 enhances survival of Toxoplasma-infected mice68,69. Because IL-2 and IL-15 signaling depends on trans-presentation70,71, complexing IL-2 with anti-IL-2 (IL2C) or IL-15 with IL-15RαFc (IL15C) significantly enhances their biological activity in vivo31,72. Importantly, the binding site of the anti-IL2 clone used in the IL2C determines whether a preferential expansion of regulatory T cells (TReg; anti-IL-2 clone JES6-1A12) or CD8+ T cell, NK cells and DN T cells occurs (anti-IL-2 clone S4B6)31,73.
Using JES6-1A12-containing IL2C, Akbar et al.41 showed that selective expansion of TReg cells in Type I T. gondii RH-infected animals improved control of the parasite. It was also demonstrated that TReg expansion with JES6-1A12-containing IL2C can overcome the competition for bioavailable IL-2 by regulatory and effector T cells, leading to reduced immunopathology and morbidity during acute Type II T. gondii ME49 infection40. These studies are in line with other reports showing a collapse of TReg cells during acute T. gondii infection due to IL-2 starvation and an overall protective role of TRegs in acute T. gondii-mediated immunopathogenesis74,75,76,77. In contrast to JES6-1A12-containing IL2C, S4B6-containing IL2C has been shown to boost NK cell and memory CD8+ T cell numbers in mice and to enhance their cytolytic capacity against viral infections, malaria78 and cancer cells70,79,80,81. Short-term exposure of naïve mice to IL2C containing S4B6 has also been shown to enhance resistance and immunity against Listeria monocytogenes infection82. Our study is the first to show a protective effect of S4B6-containing IL2C pre-treatment in toxoplasmosis and our results suggest that IL2C pre-treatment can protect mice from lethal toxoplasmosis via distinct mechanisms, depending on the IL-2 mAb clone used to prepare the cytokine complex. Thus, JES6-1A12-containing IL2C seems to compensate for the limited bioavailability of IL-2 for Treg survival during acute T. gondii infection, leading to reduced immunopathology, whereas S4B6-containing IL2C, whilst also reducing pathology without affecting parasite burden, does so in a Treg-independent manner. Our findings do not definitively rule out a role for Treg function or local (i.e., gut) IL-10 in SB46 IL2C treated mice (e.g., via use of Treg ot IL-10 depleted or knockout mice) but our results do show that S4B6 IL2C treatment has no effect on systemic IL-10 levels, further pointing towards a Treg-independent function. Based on our results, we conclude that S4B6-containing IL2C seems to favor survival and expansion of IL-18-driven IFN-γ secretion, possibly driving parasites towards stage conversion and cyst formation. It is, hence, tempting to speculate that both types of IL2C could have a synergistic effect if applied together.
Cytokine complex-mediated immunotherapy has not only attracted attention in models of infectious diseases but also in the cancer field83. IL2C treatment reduces viral load in a mouse model of gamma-herpesvirus infection84 and impacts positively on mouse melanoma85 and BCL1 leukemia86. More recently, IL2C treatment has also been tested successfully in cancer models in combination with immune checkpoint blockade87. IL-15/IL-15Rα-Fc complexes (IL15C) have also been shown to expand CD8+ T cell, DN T cell and NK cell populations, and to protect mice against cerebral malaria via the induction of IL-10-producing NK cells78. Whether IL15C would also be protective in our model of lethal toxoplasmosis remains to be investigated. Taken together, these results suggest that cytokine complex treatment may be a more broadly applicable adjunct therapy in infectious diseases, but also highlight that the protective mechanisms may differ between different pathogens and cytokine complex types used. To our knowledge, no data are available yet on any clinical use of IL2C and IL15C in humans. It will be important to consider the hyper-inflammatory response that can be attributed to IL2C and IL15C treatment and, hence, careful consideration should be taken before using cytokine complexes clinically in the context of toxoplasmosis.
In summary, here we delineate a mechanistic framework for how IFN-γ is produced by non-CD4 cell types in vivo in response to T. gondii, including a crucial role for parasite viability and inflammasome-dependent IL-18 secretion. Our results demonstrate that in vivo inflammasome activation in response to T. gondii occurs in multiple myeloid cell types and indicate the existence of an unidentified T. gondii-sensing component. Additionally, our study excludes T. gondii-derived, ASP5-dependent, dense granule proteins as the main activators of inflammasomes in vivo. The observation that both IL-12 and IL-18 neutralization reverses the host protective role of CD8+ T cells, DN T cells and NK cell-produced IFN-γ during T. gondii infection highlights the redundancy and functional interchangeability of both cytokines during T. gondii infection. This combination of observations led us to the hypothesis that enhancement of inflammasome-dependent, IL18-driven production of IFN-γ by non-CD4 cells may be a route to control acute toxoplasmosis in AIDS. In accord with this, we provide compelling evidence for a protective role of IL2C pre-treatment in lethal toxoplasmosis. We demonstrate that IL2C-mediated expansion of CD8+ T cells, NK cells and DN T cells protects mice against acute disease and death in an IFN-γ-dependent manner. Hence, we conclude that inducing immune responses that lead to the expansion of IFN-γ-secreting CD8+ T cells, DN T cells and NK cells could be a crucial feature of improved toxoplasmosis intervention strategies, perhaps most particularly in the context of HIV co-infection and AIDS.
Methods
Mice
C57BL/6 J and Arc(S) mice were purchased from the Animal Resource Center (Perth, Australia). Knockout mice (Caspase1/11–/–, Nlrp1−/−, Nlrp3–/– and Il18−/−) were bred and maintained at the Australian Institute of Tropical Health and Medicine, James Cook University, Cairns and Townsville, Australia. Double knockout mice (Nlrp1−/−Nlrp3−/−) mice were bred by sequentially crossing Nlrp1−/−88 and Nlrp3−/− mice. Genotyping was performed using the following primer pairs: Nlrp3-F 5′-GCTCAGGACATACGTCTGGA-3′,Nlrp3-R 5′-TGAGGTCCACATCTTCAAGG-3′,Nlrp3-R2 5′-TTGTAGTTGCCGTCGTCGTCCTT-3′,Nlrp1 WT: Nalp1aF 5′-TGGAAGGAAGGCAAGCTTTA-3′; Nalp1aR 5′-ACCCAGGGAACTTCACACAG-3′; Nlrp1 mutant: Nalp1aF 5′-TTTAGAGCTTGACGGGGAAA-3′; Nalp1aR 5′-GGAAGGACTTCCCACCCTAA-3′. The following mice were used for experiments: Nlrp1−/−Nlrp3−/−, Nlrp1+/−Nlrp3−/− and Nlrp1−/−Nlrp3+/−. For infection experiments, all mice were sex- and age-matched, and kept in our BSL 2 animal facility under specific pathogen-free (SPF) conditions.
Parasites
Type II T. gondii strains ME49, ME49-RFP, ME49 GRA20-deficient, ME49 GRA23-deficient, ME49 ASP5-deficient and DEG (ATCC, ATC50855) were maintained by continuous passage in human foreskin fibroblasts (HFF; ATCC, ATCSCRC1041) in DMEM supplemented with 10% FCS, penicillin, streptomycin and L-glutamine at 37 °C and 5% CO2. Parasites were harvested from recently lysed cell monolayers, passed through a 26G needle and a 3 µm TSTP Isopore™ membrane filter and concentrated by centrifugation at 500 g for 10 min. The pellet of tachyzoites was re-suspended in sterile PBS. Parasites were counted using a Neubauer hemocytometer and diluted to the required infectious dose in sterile PBS.
Generation of T. gondii ME49 Gra20 and Gra23 knockouts
We employed a CRISPR/Cas9 approach to insert frameshifts within the first 20 nt of the start of the coding sequence of gra20 and gra23 in T. gondii Me49 with consequential disruption of the final translated proteins. Inverse PCR was used to exchange the sgRNA of UPRT with the sgRNA for GRA20 with Ph-sgRNA_TgGRA20mutF (5′-ATGCATAGCCGGAACTGCGTGTTTTAGAGCTAGAAATAGC-3′) and Ph-genCas9mutR (5′-AACTTGACATCCCCATTTAC-3′) to yield plasmid pCAS9sgGRA20. Similarly, inverse PCR was used to exchange the sgRNA of UPRT with the sgRNA for GRA23 with Ph-sgRNA_TgGRA23mutF (5′- GCAGCGCGTGCGGGAAGCAGGTTTTAGAGCTAGAAATAGC-3′) and Ph-genCas9mutR (5′-AACTTGACATCCCCATTTAC-3′) to yield plasmid pCAS9sgGRA23. Transfection of T. gondii Me49 was carried out as described previously89. Twenty-four hours post-transfection, transiently transfected GFP+ parasites were purified by flow cytometry as previously described90 and individual GRA20 and GRA23 KO clones were further purified using two rounds of limiting dilution cloning. Sanger sequencing of PCR products was used to confirm disruption of the gra20 and gra23 ORFs.
Infections and monitoring
To isolate T. gondii ME49 bradyzoite containing cysts, the brains of chronically infected Arc(S) mice (injected i.p. with 500 tachyzoites of T. gondii ME49 > 8 weeks prior) were removed, homogenized in sterile PBS, and subjected to centrifugation in a discontinuous Percoll gradient. Cysts were counted using a Neubauer hemocytometer and diluted in sterile PBS. For experiments, B6 mice were inoculated with 10, 40 or 100 cysts by oral inoculation. For mechanistic studies, B6 mice were injected i.v. in the lateral tail vein with 107 tachyzoites of T. gondii ME49, mutant strains on the T. gondii ME49 background or the Type II strain T. gondii DEG in a volume of 200 µl. For heat inactivation, T. gondii ME49 tachyzoites were grown as described above, enumerated, and washed twice with PBS before incubation at 62º C in a water bath for 1 h. Effective killing was verified by addition of heat-killed parasites to a HFF cell monolayer.
All mice were monitored as stipulated by Animal Ethics Committee of James Cook University Approvals A2138, A2324. Chronically infected Arc(S) mice were monitored weekly for signs of morbidity and were euthanized using carbon dioxide asphyxiation for brain cyst harvesting. Mice infected orally with T. gondii-containing brain cysts were monitored daily for signs of disease and were euthanized using carbon dioxide asphyxiation at distinct time-points after infection for immunological readouts or when ethical endpoints were reached for survival experiments. Death was never used as an endpoint. Mice infected i.v. or i.p. with T. gondii tachyzoites were monitored daily for signs of disease and were euthanized using carbon dioxide asphyxiation 2 to 72 h after infection.
Isolation of leukocytes
Spleens, mesenteric lymph nodes and Peyer’s Patches were extracted and mechanically disrupted by pushing cells through a 70 µm cell strainer. Subsequently, red-cell depleted, single-cell suspensions were prepared as described elsewhere39. Lamina propria cells were isolated from the ileum as published previously with minor modifications91.
Scoring of pathology
Gross pathology of ileum and liver was scored visually using a scoring system adapted from Melgar et al.92. For the ileum, the consistency of the intestinal contents, the degree of swelling and amount of angiogenesis were assessed. This system is based on an ascending scale of severity, for each parameter, as follows: 0 (no abnormality),1 (minimal),2 (moderate),or 3 (severe). For the liver, the colour and appearance of the organ were assessed on an ascending scale of severity from 0 (normal colour and appearance); 1 (blotchy appearance with some areas exhibiting change in colour); 2 (entire organ pale in colour); or 3 (entire organ pale in colour with visible signs of necrosis). Scores for each parameter were added together to give a total score for each animal.
Parasite burden
Parasite burden was measured in the whole spleen of individual mice using a microtitre dilution method adapted from Buffet et al.93 It was necessary to determine parasite burden in the spleen rather than the intestine because it was impossible to harvest immune cells for analysis from the intestine and determine parasite burden in the same animal, however, we have demonstrated previously that the parasite burden in the spleen accurately mirrors that in the intestine50. Briefly, prior to the experiment, 96 well plates were seeded with HFF cells and allowed to become confluent. One row was allocated per mouse and each mouse was done in duplicate. Spleens were removed and single-cell suspensions were made by passing through a 70-µm cell strainer. Cells were pelleted at 1500g, and then resuspended in RPMI 1,640 containing 5% FCS at a concentration of 1 × 107 cells/ml. Two hundred microliters of spleen cell suspension was added to the first well of a 96-well plate and then serially diluted 1/2 across the plate. Plates were incubated at 37 °C in 5% CO2 for 7 days before wells were examined for the presence of parasites. A score of parasite burden was allocated based on the last column in which parasites were visible.
Flow cytometry
To assess expression of surface antigens and IFN-γ secretion, viable, red blood cell-depleted single-cell suspensions were stained with monoclonal antibodies (all from BD Pharmingen) against CD4 (clone GK1.5), CD8α (clone 53-6.7), CD3 (clone 145-2C11), NKp46 (clone 29A1.4), CD44 (clone 1M7), CD90.1 (clone 30-H12), CD11b (clone M1/70), CD11c (clone HL3), MHC-II (clone M5/114), Ly6G (clone 1A8), Ly6C (clone AL-21), CD19 (clone 1D3), F4/80 (clone BM8), or IFN-γ detection antibody (Miltenyi Biotec, Germany), IL-18Ralpha (R&D Systems) or IL-12Rbeta1 (CD212, BD). CD3+CD4+CD25+Foxp3+ regulatory T cells were identified using the Foxp3/Transcription Factor Staining Buffer Set (eBioscience). After washing the cells, samples were analyzed using a FACSCantoII or FortessaX20 analyzers (BD Biosciences, CA). Propidium iodide (2 μg/ml) or Fixable Viability Dye e780 (BD) was added to exclude dead cells. Flow cytometry data were analyzed using FlowJo software (Treestar, CA). For all flow cytometry-based analyses, cells were first gated on singlets, followed by dead cell exclusion, scatter characteristics and surface marker expression. All samples contained Blank Calibration Particles (BD) to allow cell enumeration.
Assessment of ex vivo IFN-γ secretion
Ex vivo IFN-γ secretion by distinct lymphocyte subsets was assessed as described previously29. Briefly, mice were injected i.v., i.p., or p.o. with different doses of T. gondii ME49 cysts or tachyzoites (as described in figure legends). At different time points after injection of parasites (as described in figure legends), organs were removed aseptically, single cell suspensions were prepared and red blood cells were lysed. Cells (106 were stained with the ‘Mouse IFN-γ secretion assay detection kit’ (Miltenyi Biotec, Germany) according to the manufacturer’s instructions and IFN-γ secretion was analyzed by flow cytometry. Cells were first gated on live, single lymphocytes, followed by separation into CD3+NKp46− and CD3−NKp46+ cells. CD3+NKp46− cells were further gated into CD4+, CD8+ and CD4−CD8− subsets. CD3−NKp46+, CD3+NKp46−CD4+, CD3+NKp46−CD8+ and CD3+NKp46−CD4−CD8− cells were assessed for IFN-γ secretion.
Detection of in vivo inflammasome activation by flow cytometry
Detection of apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC) assembly was performed as described previously94. Briefly, mice were injected with 107 T. gondii ME49-RFP tachyzoites and euthanased 24 h later. Cells were stained for surface molecules, fixed, permeabilized and stained with rabbit anti-ASC antibody (Santa Cruz Biotechnology) for 45 min at room temperature. Subsequently, a secondary anti-rabbit Alexa488 antibody (Life Technologies) was added for 45 min at room temperature. A FMO control without anti-goat Alexa488 was included.
Detection of active caspase-1 by flow cytometry was performed using the carboxyfluorescein FLICA kit (FAM-YVAD-FMK, Immunochemistry Techniques, Bloomington, MN). B6 mice were injected with 107 T. gondii ME49-RFP tachyzoites and 23 h later FAM-YVAD-FMK (diluted in DMSO and PBS) was injected intravenously. Splenic cells were analyzed by FACS 1 h later as described above (24 h after T. gondii ME49-RFP injection). Mice that received T. gondii ME49-RFP but no FAM-YVAD-FMK were used as FMO control.
Single cells were gated for RFP expression and RFP+ cells were analyzed for expression of neutrophil, macrophage, monocyte, dendritic cell, T cell and B cell specific surface markers and positivity in green fluorescence as shown in Fig. 4.
Multiplex and ELISA
Blood for serum analysis was taken post mortem from the aorta abdominalis and collected in serum separator tubes (BD), left for 30 min at room temperature, followed by centrifugation at 12,000 g for 3 min. Sera were stored at –20 °C until analysis. Measurements were performed using CBA (BD Biosciences, CA) or ELISA (elisakit.com, Australia) according to manufacturers’ instructions. Samples were acquired on a FACSCantoII (BC Biosciences, CA) or a FLUOstar Omega ELISA Reader (BMG Labtech).
IL-2/anti-IL-2 complex-mediated cell expansion
IL-2/anti-IL-2 complexes (IL2C) were prepared as described previously38. Briefly, 1.5 µg of recombinant mouse IL-2 (Peprotech) and 10 µg of anti-IL-2 mAb (clone S4B6, Walter and Eliza Hall Institute [WEHI] antibody facility, Melbourne, Australia) were mixed, incubated at 37 °C for 30 min, and administered i.p. in a volume of 200 µl for four consecutive days. Mice were monitored and weighed daily during the IL2C treatment period.
Antibody-mediated cell depletion and cytokine neutralization
For cytokine neutralization and cell depletion, monoclonal antibodies against IL-12, IL-18, IFN-γ, CD8, NK1.1, Thy1.2 and rat IgG were purchased from the WEHI antibody facility or from BioXCell (NH, USA). A total of 200 µg of anti-IL-18 (clone YIGIF74-1G7; Cat. No.: BE0237), anti-IFN-γ (clone HB170-15), anti-IL12 (clone C17.8), anti-NK1.1 (clone PK136), anti-CD8 (clone 2.43), anti-Thy1.2 (clone 30H12) or control rat IgG were injected i.p. weekly in a volume of 200 µl.
Statistics
Statistical analysis was performed using GraphPad Prism, GraphPad software, San Diego, CA as indicated in individual figure legends. These included: one-way analysis of variance (ANOVA), followed by Dunnett’s multiple comparison test for most data sets; two-way ANOVA followed by Tukey’s post hoc test for data presented in Fig. 3; two-tailed Student’s t tests for data presented in Fig. 7i; a Log-rank (Mantel–Cox) test to compare significance for survival experiments in Figs. 7b,c,e,f. A P value of less than 0.05 was considered significant.
Ethics statement
All experiments were approved and conducted according to Australian animal protection law and in accordance with requirements of the Animal Ethics Committee of James Cook University (Approvals A2138, A2324). Death was never used as an endpoint.
Data availability
All data are available within the manuscript and Supplementary Information.
References
Seeber, F. & Steinfelder, S. Recent advances in understanding apicomplexan parasites. F1000Research https://doi.org/10.12688/f1000research.7924.1 (2016).
Yarovinsky, F. Innate immunity to Toxoplasma gondii infection. Nat. Rev. Immunol. 14, 109–121. https://doi.org/10.1038/nri3598 (2014).
Zhang, Y., Lin, X. & Lu, F. Current treatment of ocular toxoplasmosis in immunocompetent patients: A network meta-analysis. Acta Trop. 185, 52–62. https://doi.org/10.1016/j.actatropica.2018.04.026 (2018).
Fallahi, S., Rostami, A., Nourollahpour Shiadeh, M., Behniafar, H. & Paktinat, S. An updated literature review on maternal-fetal and reproductive disorders of Toxoplasma gondii infection. J. Gynecol. Obstet. Hum. Reprod. 47, 133–140. https://doi.org/10.1016/j.jogoh.2017.12.003 (2018).
Dard, C. et al. Management of toxoplasmosis in transplant recipients: An update. Expert Rev. Anti Infect. Ther. https://doi.org/10.1080/14787210.2018.1483721 (2018).
Rajapakse, S., Weeratunga, P., Rodrigo, C., de Silva, N. L. & Fernando, S. D. Prophylaxis of human toxoplasmosis: A systematic review. Pathog. Glob. Health 111, 333–342. https://doi.org/10.1080/20477724.2017.1370528 (2017).
Sturge, C. R. & Yarovinsky, F. Complex immune cell interplay in the gamma interferon response during Toxoplasma gondii infection. Infect. Immun. 82, 3090–3097. https://doi.org/10.1128/iai.01722-14 (2014).
Scharton-Kersten, T. M., Yap, G., Magram, J. & Sher, A. Inducible nitric oxide is essential for host control of persistent but not acute infection with the intracellular pathogen Toxoplasma gondii. J. Exp. Med. 185, 1261–1273 (1997).
Scharton-Kersten, T. M. et al. In the absence of endogenous IFN-gamma, mice develop unimpaired IL-12 responses to Toxoplasma gondii while failing to control acute infection. J Immunol 157, 4045–4054 (1996).
Suzuki, Y., Orellana, M. A., Schreiber, R. D. & Remington, J. S. Interferon-gamma: the major mediator of resistance against Toxoplasma gondii. Science 240, 516–518 (1988).
Gazzinelli, R., Xu, Y., Hieny, S., Cheever, A. & Sher, A. Simultaneous depletion of CD4+ and CD8+ T lymphocytes is required to reactivate chronic infection with Toxoplasma gondii. J. Immunol. 149, 175–180 (1992).
Oksenhendler, E. et al. Toxoplasma gondii pneumonia in patients with the acquired immunodeficiency syndrome. Am. J. Med. 88, 18n–21n (1990).
Hunter, C. A., Subauste, C. S., Van Cleave, V. H. & Remington, J. S. Production of gamma interferon by natural killer cells from Toxoplasma gondii-infected SCID mice: regulation by interleukin-10, interleukin-12, and tumor necrosis factor alpha. Infect. Immun. 62, 2818–2824 (1994).
Denkers, E. Y., Gazzinelli, R. T., Martin, D. & Sher, A. Emergence of NK1.1+ cells as effectors of IFN-gamma dependent immunity to Toxoplasma gondii in MHC class I-deficient mice. J. Exp. Med. 178, 1465–1472 (1993).
Yarovinsky, F. et al. TLR11 activation of dendritic cells by a protozoan profilin-like protein. Science 308, 1626–1629. https://doi.org/10.1126/science.1109893 (2005).
Plattner, F. et al. Toxoplasma profilin is essential for host cell invasion and TLR11-dependent induction of an interleukin-12 response. Cell Host Microbe 3, 77–87. https://doi.org/10.1016/j.chom.2008.01.001 (2008).
Scanga, C. A. et al. Cutting edge: MyD88 is required for resistance to Toxoplasma gondii infection and regulates parasite-induced IL-12 production by dendritic cells. J. Immunol. 168, 5997–6001 (2002).
Hou, B., Benson, A., Kuzmich, L., DeFranco, A. L. & Yarovinsky, F. Critical coordination of innate immune defense against Toxoplasma gondii by dendritic cells responding via their Toll-like receptors. Proc. Natl. Acad. Sci. USA 108, 278–283. https://doi.org/10.1073/pnas.1011549108 (2011).
LaRosa, D. F. et al. T cell expression of MyD88 is required for resistance to Toxoplasma gondii. Proc. Natl. Acad. Sci. USA 105, 3855–3860. https://doi.org/10.1073/pnas.0706663105 (2008).
Sun, J. & Lanier, L. NK cell development, homeostasis and function: parallels with CD8+ T cells. Nat. Rev. Immunol. 11, 645–657 (2011).
O’Donnell, H. et al. Toll-like receptor and inflammasome signals converge to amplify the innate bactericidal capacity of T helper 1 cells. Immunity 40, 213–224. https://doi.org/10.1016/j.immuni.2013.12.013 (2014).
Arend, W., Palmer, G. & Gabay, C. IL-1, IL-18, and IL-33 families of cytokines. Immunol. Rev. 223, 20–38 (2008).
Schroder, K. & Tschopp, J. The inflammasomes. Cell 140, 821–832 (2010).
Gorfu, G. et al. Dual role for inflammasome sensors NLRP1 and NLRP3 in murine resistance to Toxoplasma gondii. mBio https://doi.org/10.1128/mBio.01117-13 (2014).
Cai, G., Kastelein, R. & Hunter, C. A. Interleukin-18 (IL-18) enhances innate IL-12-mediated resistance to Toxoplasma gondii. Infect. Immun. 68, 6932–6938 (2000).
Zamboni, D. S. & Lima-Junior, D. S. Inflammasomes in host response to protozoan parasites. Immunol. Rev. 265, 156–171. https://doi.org/10.1111/imr.12291 (2015).
Helbig, E. T., Opitz, B. & Sander, L. E. Adjuvant immunotherapies as a novel approach to bacterial infections. Immunotherapy 5, 365–381. https://doi.org/10.2217/imt.13.17 (2013).
Kupz, A. et al. ESAT-6-dependent cytosolic pattern recognition drives noncognate tuberculosis control in vivo. J. Clin. Investig. https://doi.org/10.1172/JCI84978 (2016).
Kupz, A. et al. NLRC4 inflammasomes in dendritic cells regulate noncognate effector function by memory CD8(+) T cells. Nat. Immunol. 13, 162–169 (2012).
Soudja, S. M. et al. Memory-T-cell-derived interferon-gamma instructs potent innate cell activation for protective immunity. Immunity 40, 974–988. https://doi.org/10.1016/j.immuni.2014.05.005 (2014).
Boyman, O., Kovar, M., Rubinstein, M., Surh, C. & Sprent, J. Selective stimulation of T cell subsets with antibody-cytokine immune complexes. Science 311, 1924–1927 (2006).
Tenter, A. M., Heckeroth, A. R. & Weiss, L. M. Toxoplasma gondii: from animals to humans. Int. J. Parasitol. 30, 1217–1258. https://doi.org/10.1016/s0020-7519(00)00124-7 (2000).
Boyle, J. P., Saeij, J. P. & Boothroyd, J. C. Toxoplasma gondii: inconsistent dissemination patterns following oral infection in mice. Exp. Parasitol. 116, 302–305. https://doi.org/10.1016/j.exppara.2007.01.010 (2007).
Malik, A. & Kanneganti, T. D. Inflammasome activation and assembly at a glance. J. Cell Sci. 130, 3955–3963. https://doi.org/10.1242/jcs.207365 (2017).
Melo, M. B., Jensen, K. D. C. & Saeij, J. P. J. Toxoplasma gondii effectors are master regulators of the inflammatory response. Trends in parasitology 27, 487–495. https://doi.org/10.1016/j.pt.2011.08.001 (2011).
Hakimi, M. A. & Bougdour, A. Toxoplasma’s ways of manipulating the host transcriptome via secreted effectors. Curr. Opin. Microbiol. 26, 24–31. https://doi.org/10.1016/j.mib.2015.04.003 (2015).
Hammoudi, P. M. et al. Fundamental roles of the golgi-associated toxoplasma aspartyl protease, ASP5, at the host–parasite interface. PLoS Pathog. 11, e1005211. https://doi.org/10.1371/journal.ppat.1005211 (2015).
Kupz, A., Zedler, U., Staber, M. & Kaufmann, S. H. A mouse model of latent tuberculosis infection to study intervention strategies to prevent reactivation. PLoS ONE 11, e0158849. https://doi.org/10.1371/journal.pone.0158849 (2016).
Kupz, A. et al. Contribution of Thy1+ NK cells to protective IFN-γ production during Salmonella Typhimurium infections. Proc. Natl. Acad. Sci. 110, 2252–2257. https://doi.org/10.1073/pnas.1222047110 (2013).
Oldenhove, G. et al. Decrease of Foxp3+ Treg cell number and acquisition of effector cell phenotype during lethal infection. Immunity 31, 772–786. https://doi.org/10.1016/j.immuni.2009.10.001 (2009).
Akbar, H., Dimier-Poisson, I. & Moire, N. Role of CD4+ Foxp3+ regulatory T cells in protection induced by a live attenuated, replicating type I vaccine strain of Toxoplasma gondii. Infect Immun 83, 3601–3611. https://doi.org/10.1128/iai.00217-15 (2015).
Witola, W. et al. NALP1 influences susceptibility to human congenital toxoplasmosis, proinflammatory cytokine response, and fate of Toxoplasma gondii-infected monocytic cells. Infect. Immun. 79, 756–766 (2011).
Vladimer, G. I. et al. The NLRP12 inflammasome recognizes yersinia pestis. Immunity 37, 96–107. https://doi.org/10.1016/j.immuni.2012.07.006 (2012).
Fernandes-Alnemri, T. et al. The AIM2 inflammasome is critical for innate immunity to Francisella tularensis. Nat Immunol 11, 385–393 (2010).
Soudja, S. M. H., Ruiz, A., Marie, J. & Lauvau, G. Inflammatory monocytes activate memory CD8(+) T and innate NK lymphocytes independent of cognate antigen during microbial pathogen invasion. Immunity 37, 549–562 (2012).
Kastenmüller, W., Torabi-Parizi, P., Subramanian, N., Lämmermann, T. & Germain, R. N. A spatially-organized multicellular innate immune response in lymph nodes limits systemic pathogen spread. Cell 150, 1235–1248 (2012).
Quan, J. H. et al. P2X7 receptor mediates NLRP3-dependent IL-1beta secretion and parasite proliferation in Toxoplasma gondii-infected human small intestinal epithelial cells. Parasit. Vectors 11, 1. https://doi.org/10.1186/s13071-017-2573-y (2018).
Chu, J. Q. et al. Production of IL-1beta and Inflammasome with Up-regulated expressions of NOD-like receptor related genes in Toxoplasma gondii-Infected THP-1 macrophages. Korean J. Parasitol. 54, 711–717. https://doi.org/10.3347/kjp.2016.54.6.711 (2016).
Correa, G. et al. Inflammatory early events associated to the role of P2X7 receptor in acute murine toxoplasmosis. Immunobiology 222, 676–683. https://doi.org/10.1016/j.imbio.2016.12.007 (2017).
Miller, C. M. et al. Dysregulation of the inflammatory response to the parasite, Toxoplasma gondii, in P2X7 receptor-deficient mice. Int. J. Parasitol. 41, 301–308. https://doi.org/10.1016/j.ijpara.2010.10.001 (2011).
Moreira-Souza, A. C. A. et al. The P2X7 receptor mediates Toxoplasma gondii control in macrophages through canonical NLRP3 inflammasome activation and reactive oxygen species production. Front. Immunol. 8, 1257. https://doi.org/10.3389/fimmu.2017.01257 (2017).
Gov, L., Schneider, C. A., Lima, T. S., Pandori, W. & Lodoen, M. B. NLRP3 and potassium efflux drive rapid IL-1beta release from primary human monocytes during Toxoplasma gondii infection. J Immunol 199, 2855–2864. https://doi.org/10.4049/jimmunol.1700245 (2017).
Lima, T. S., Gov, L. & Lodoen, M. B. Evasion of human neutrophil-mediated host defense during Toxoplasma gondii infection. mBio https://doi.org/10.1128/mBio.02027-17 (2018).
Nadipuram, S. M. et al. In vivo biotinylation of the toxoplasma parasitophorous vacuole reveals novel dense granule proteins important for parasite growth and pathogenesis. mBio 7, e00808-00816. https://doi.org/10.1128/mBio.00808-16 (2016).
Gold, D. A. et al. The toxoplasma dense granule proteins GRA17 and GRA23 mediate the movement of small molecules between the host and the parasitophorous vacuole. Cell Host Microbe 17, 642–652. https://doi.org/10.1016/j.chom.2015.04.003 (2015).
Franco, M. et al. A novel secreted protein, MYR1, is central to toxoplasma’s manipulation of host cells. mBio 7, e02231-e12215. https://doi.org/10.1128/mBio.02231-15 (2016).
Marino, N. D. et al. Identification of a novel protein complex essential for effector translocation across the parasitophorous vacuole membrane of Toxoplasma gondii. PLoS Pathog 14, e1006828. https://doi.org/10.1371/journal.ppat.1006828 (2018).
Naor, A. et al. MYR1-dependent effectors are the major drivers of a host cell’s early response to toxoplasma, including counteracting MYR1-independent effects. mBio 9, e02401-02417. https://doi.org/10.1128/mBio.02401-17 (2018).
Carson, W. E. et al. Interleukin (IL) 15 is a novel cytokine that activates human natural killer cells via components of the IL-2 receptor. J. Exp. Med. 180, 1395–1403 (1994).
Zwirner, N. & Domaica, C. Cytokine regulation of natural killer cell effector functions. BioFactors 36, 274–288 (2010).
Ku, C. C., Murakami, M., Sakamato, A., Kappler, J. & Marrack, P. Control of homeostasis of CD8(+) memory T cells by opposing cytokines. Science 288, 675–678 (2000).
Sprent, J. & Surh, C. D. Generation and maintenance of memory T cells. Curr. Opin. Immunol. 13, 248–254 (2001).
Li, X. C. et al. IL-15 and IL-2: a matter of life and death for T cells in vivo. Nat. Med. 7, 114–118 (2001).
Nishimura, H. et al. IL-15 is a novel growth factor for murine gamma delta T cells induced by Salmonella infection. J. Immunol. 156, 663–669 (1996).
Lieberman, L. A., Villegas, E. N. & Hunter, C. A. Interleukin-15-deficient mice develop protective immunity to Toxoplasma gondii. Infect. Immun. 72, 6729–6732. https://doi.org/10.1128/iai.72.11.6729-6732.2004 (2004).
Khan, I. A. et al. Treatment with soluble interleukin-15Ralpha exacerbates intracellular parasitic infection by blocking the development of memory CD8+ T cell response. J. Exp. Med. 195, 1463–1470 (2002).
Villegas, E. N., Lieberman, L. A., Carding, S. R. & Hunter, C. A. Susceptibility of interleukin-2-deficient mice to Toxoplasma gondii is associated with a defect in the production of gamma interferon. Infect. Immun. 70, 4757–4761 (2002).
Sharma, S. D., Hofflin, J. M. & Remington, J. S. In vivo recombinant interleukin 2 administration enhances survival against a lethal challenge with Toxoplasma gondii. J. Immunol. 135, 4160–4163 (1985).
Shirahata, T., Muroya, N., Ohta, C., Goto, H. & Nakane, A. Enhancement by recombinant human interleukin 2 of host resistance to Toxoplasma gondii infection in pregnant mice. Microbiol. Immunol. 37, 583–590 (1993).
Stoklasek, T. A., Schluns, K. S. & Lefrançois, L. Combined IL-15/IL-15Rα immunotherapy maximizes IL-15 activity in vivo. J. Immunol. 177, 6072–6080 (2006).
Stonier, S. & Schluns, K. Trans-presentation: a novel mechanism regulating IL-15 delivery and responses. Immunol. Lett. 127, 85–92 (2010).
Votavova, P., Tomala, J. & Kovar, M. Increasing the biological activity of IL-2 and IL-15 through complexing with anti-IL-2 mAbs and IL-15Rα-Fc chimera. Immunol. Lett. 159, 1–10. https://doi.org/10.1016/j.imlet.2014.01.017 (2014).
Shevach, E. M. Application of IL-2 therapy to target T regulatory cell function. Trends Immunol. 33, 626–632. https://doi.org/10.1016/j.it.2012.07.007 (2012).
Liu, Y. et al. Adoptive transfer of Treg cells counters adverse effects of Toxoplasma gondii infection on pregnancy. J. Infect. Dis. 210, 1435–1443. https://doi.org/10.1093/infdis/jiu265 (2014).
Tenorio, E. P., Fernandez, J., Castellanos, C., Olguin, J. E. & Saavedra, R. CD4+ Foxp3+ regulatory T cells mediate Toxoplasma gondii-induced T-cell suppression through an IL-2-related mechanism but independently of IL-10. Eur. J. Immunol. 41, 3529–3541. https://doi.org/10.1002/eji.201141507 (2011).
Olguin, J. E. et al. Adoptive transfer of CD4(+)Foxp3(+) regulatory T cells to C57BL/6J mice during acute infection with Toxoplasma gondii down modulates the exacerbated Th1 immune response. Microbes Infect. 17, 586–595. https://doi.org/10.1016/j.micinf.2015.04.002 (2015).
Salinas, N., Olguin, J. E., Castellanos, C. & Saavedra, R. T cell suppression in vitro during Toxoplasma gondii infection is the result of IL-2 competition between Tregs and T cells leading to death of proliferating T cells. Scand. J. Immunol. 79, 1–11. https://doi.org/10.1111/sji.12120 (2014).
Burrack, K. S. et al. Interleukin-15 complex treatment protects mice from cerebral malaria by inducing interleukin-10-producing natural killer cells. Immunity 48, 760-772.e764. https://doi.org/10.1016/j.immuni.2018.03.012 (2018).
Epardaud, M. et al. Interleukin-15/interleukin-15Rα complexes promote destruction of established tumors by reviving tumor-resident CD8+ T cells. Can. Res. 68, 2972–2983. https://doi.org/10.1158/0008-5472.can-08-0045 (2008).
Dubois, S., Patel, H. J., Zhang, M., Waldmann, T. A. & Müller, J. R. Preassociation of IL-15 with IL-15Rα-IgG1-Fc enhances its activity on proliferation of NK and CD8+/CD44 high T cells and its antitumor action. J. Immunol. 180, 2099–2106 (2008).
Huntington, N. D. et al. IL-15 trans-presentation promotes human NK cell development and differentiation in vivo. J. Exp. Med. 206, 25–34. https://doi.org/10.1084/jem.20082013 (2009).
Hamilton, S. E., Schenkel, J. M., Akue, A. D. & Jameson, S. C. IL-2 Complex treatment can protect naive mice from bacterial and viral infection. J. Immunol. 185, 6584–6590. https://doi.org/10.4049/jimmunol.1001215 (2010).
Boyman, O., Kolios, A. G. & Raeber, M. E. Modulation of T cell responses by IL-2 and IL-2 complexes. Clin. Exp. Rheumatol. 33, S54-57 (2015).
Molloy, M. J., Zhang, W. & Usherwood, E. J. Cutting edge: IL-2 Immune complexes as a therapy for persistent virus infection. J. Immunol. 182, 4512–4515. https://doi.org/10.4049/jimmunol.0804175 (2009).
Kamimura, D. et al. IL-2 in vivo activities and antitumor efficacy enhanced by an anti-IL-2 mAb. J. Immunol. 177, 306–314. https://doi.org/10.4049/jimmunol.177.1.306 (2006).
Tomala, J., Chmelova, H., Mrkvan, T., Rihova, B. & Kovar, M. In Vivo Expansion of activated naive CD8+ T cells and NK cells driven by complexes of IL-2 and Anti-IL-2 monoclonal antibody as novel approach of cancer immunotherapy. J. Immunol. 183, 4904–4912. https://doi.org/10.4049/jimmunol.0900284 (2009).
Caudana, P. et al. IL2/anti-IL2 complex combined with CTLA-4, but not PD-1, blockade rescues antitumor NK cell function by regulatory T-cell modulation. Cancer Immunol. Res. https://doi.org/10.1158/2326-6066.cir-18-0697 (2019).
Masters, S. L. et al. NLRP1 inflammasome activation induces pyroptosis of hematopoietic progenitor cells. Immunity 37, 1009–1023. https://doi.org/10.1016/j.immuni.2012.08.027 (2012).
Shen, B., Brown, K. M., Lee, T. D. & Sibley, L. D. Efficient gene disruption in diverse strains of Toxoplasma gondii using CRISPR/CAS9. mBio 5, e01114-01114. https://doi.org/10.1128/mBio.01114-14 (2014).
Striepen, B., He, C. Y., Matrajt, M., Soldati, D. & Roos, D. S. Expression, selection, and organellar targeting of the green fluorescent protein in Toxoplasma gondii. Mol. Biochem. Parasitol. 92, 325–338 (1998).
Weigmann, B. et al. Isolation and subsequent analysis of murine lamina propria mononuclear cells from colonic tissue. Nat. Protoc. 2, 2307–2311. https://doi.org/10.1038/nprot.2007.315 (2007).
Melgar, S., Karlsson, A. & Michaelsson, E. Acute colitis induced by dextran sulfate sodium progresses to chronicity in C57BL/6 but not in BALB/c mice: correlation between symptoms and inflammation. Am. J. Physiol. Gastrointest. Liver Physiol. 288, G1328-1338. https://doi.org/10.1152/ajpgi.00467.2004 (2005).
Buffet, P. A., Sulahian, A., Garin, Y. J., Nassar, N. & Derouin, F. Culture microtitration: a sensitive method for quantifying Leishmania infantum in tissues of infected mice. Antimicrob. Agents Chemother. 39, 2167–2168 (1995).
Sester, D. P. et al. Assessment of inflammasome formation by flow cytometry. Curr. Protoc. Immunol. 114, 144011–144029. https://doi.org/10.1002/cpim.13 (2016).
Acknowledgments
This work was supported by the National Health and Medical Research Council of Australia (NHMRC) through a CJ Martin Biomedical Early Career Fellowship (APP1052764) and a Career Development Level 1 Fellowship (APP1140709) to A.K. A.K and C.M.M. also acknowledge support through a Capacity Building Grant (15031) provided by the Australian Institute of Tropical Health and Medicine (AITHM) via the Queensland Government. We thank Michael E. Grigg (NIH, USA) for generously providing the T. gondii ME49-RFP, and Seth Masters (WEHI, Melbourne, Australia) for providing Nlrp1−/− and Nlrp3−/− mice. We are grateful to Dominique Soldati-Favre (University of Geneva) for her insights into GRA proteins.
Author information
Authors and Affiliations
Contributions
A.K., C.M.M. and N.C.S. conceived the study; A.K., C.M.M, R.A.W., J.A.W., P.R.G. and S.P. performed experiments; P.M.H. and P.R.G. provided reagents and intellectual input. A.K. performed data analysis and wrote the manuscript. N.C.S and C.M.M. commented extensively on, and revised the manuscript; all coauthors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kupz, A., Pai, S., Giacomin, P.R. et al. Treatment of mice with S4B6 IL-2 complex prevents lethal toxoplasmosis via IL-12- and IL-18-dependent interferon-gamma production by non-CD4 immune cells. Sci Rep 10, 13115 (2020). https://doi.org/10.1038/s41598-020-70102-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-70102-1
This article is cited by
-
Upregulated dual oxidase 1-induced oxidative stress and caspase-1-dependent pyroptosis reflect the etiologies of heart failure
BMC Molecular and Cell Biology (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.