Abstract
This study aimed to analyse the effect of playing surface temperature on muscular and thermal response to a repeated-sprint ability (RSA) test in football players. Thirty-two male football players (23 ± 5 years; 1.77 ± 0.06 m; 71.2 ± 6.7 kg) from two squads of a third-division football club participated in the study. An RSA test was carried out at a high surface temperature (45.34 ± 2.53 °C) and low surface temperature (27.21 ± 2.17 °C). Before and after this test, the muscular response of the players was assessed through tensiomyography and thermograms. The results revealed that performance in the RSA test particularly increased at a higher surface temperature, especially in the first 5 m of the 30 m sprint test. While a reduction in maximal radial displacement (Dm) in the biceps femoris post-RSA was observed at lower surface temperatures, a higher temperature on the thigh, hamstring and calf was found in the higher surface temperature group. In conclusion, higher surface temperatures had an influence on players’ thermal and tensiomyographic profile and improved performance in their repeated-sprint ability. These results suggest a need for coaches and players to be aware of these parameters to ensure adequate functionality and safety of the playing surface.
Similar content being viewed by others
Introduction
Football is a sport that demands maximal or near maximal actions of short duration with brief recovery periods1. A top-class player performs intermittent work and 150–250 brief intense actions during a game, and in terms of energy production and energy systems, high-intensity exercise periods are important2. Repeated-sprint ability (RSA)-based exercises are characterized by several sprints interspersed with brief recovery periods between bouts3. The RSA test is recognized as a valid method to reproduce performance decrement and fatigue in football players3. From a practical standpoint, the faster an athlete can accelerate, the greater the chance of success − for instance, evading an opponent or getting possession of the ball4. It has been noted that linear sprints are the most usual actions in goal situations and highlight the importance of maximal efforts and speed during decisive moments of the game5. Moreover, Cuadrado-Peñafiel et al.6 have shown that the performance of an athlete in an RSA test offers relevant information on the explosive ability of footballers, and Padulo et al.7 have highlighted the use of an RSA test as a tool to evaluate an athlete’s ability to recover from sprint efforts. There is a significant correlation between performance in this RSA test (3 sets of 6 × 40 m maximal shuttle sprints) and sprinting or high-intensity running distances covered during official matches in professional football players8. The use of the RSA protocol in previous studies has been based on the intermittent nature of football and the determinant importance of high-intensity actions during the game2,8,9,10. These significant correlations confirm that the RSA test involves the physical capacities taxed during the high-intensity phases of a match9. Furthermore, the authors noted that, as this test includes shuttle sprints, the muscular contractions required for decelerating and to reaccelerate body mass may potentially be beneficial in improving physiological responses and the ability to change direction, so they could be considered an appropriate training exercise for football players. The load of this kind of test can be analysed through different physiological and mechanical variables, such as body temperature11,12 and muscular response13,14.
According to the thermal profile analysis, muscle activity is one of the principal heat sources of the human body15. Therefore, exercise is considered to be one of the strongest influences on skin temperature (Tsk). To understand the concept, Jiang et al.16 briefly noted that Tsk is the result of the heat balance that is generated by the metabolism and heat loss through thermal conduction, forced and natural convection, perspiration and exhalation. It is important to note that body temperature is one of the most commonly used indicators of health status in humans and it is influenced by a tremendously large number of factors17. This research showed that the human thermal portrait is related to the individual fitness and expertise level, aerobic working capacity, blood lactate levels, sport specialization and sweating capacity. Moreover, a strong correlation has been found between the skin and ambient temperature. Tsk was observed to increase proportionally with the ambient temperature18. Additionally, Fisher et al.19 performed a study demonstrating that extreme environmental conditions can significantly affect Tsk. On the other hand, it has been shown that hyperthermia in the muscle temperature of players caused by hot ambient conditions markedly decreases high-intensity running at the end of a game; however, it increases peak sprinting speed20, and, prior to competition, maximal sprint performance may be improved when the muscle temperature is elevated21. Studies of the thermal response following exercise have reported both increases and decreases in Tsk immediately following exercise, depending on the type and duration of the exercise. Normally, increasing Tsk is related to constant and prolonged aerobic tasks21,22 , whereas studies that report decreasing Tsk primarily utilized brief intense or maximal exercises23,24. Malkinson25 affirmed that a greater intensity is related to a major increase in Tsk; however, other works have shown the opposite: there is an indirect relationship between exercise intensity and Tsk12,26. Nevertheless, no studies have been published regarding the effect of the RSA protocol on the thermal profile.
Muscular response, fatigue of the lower-limb muscles appears to be an important factor in elevating the risk of injury27. Fatigue-inducing actions include muscle stiffness, contraction speed or displacement of the muscle belly14 and can be determined by tensiomyography (TMG), via the application of an electrical stimulus28. TMG has been identified as a reliable method for analysing the risk of injury in athletes14 as it enables the identification of asymmetries in the lower-limb muscles and differences in muscle responses after completing fatiguing efforts such as an RSA test29. More specifically, this technique has been reported to have high reproducibility and reliability in measuring values like contraction time (Tc), half-relaxation time (Tr), delay time (Td), sustained contraction time (Ts) and maximal radial displacement of the muscle belly (Dm) for the medial vastus, lateral vastus, femoris rectus and biceps femoris muscles14,27,28,29,30.
The evolution that artificial turfs have undergone is still unable to prevent the turf from reaching higher temperatures than natural grass. This situation results in football players’ dissatisfaction, decreased performance and the possibility of causing heat-related injuries31. Felipe et al.32 and Sánchez-Sánchez et al.33 showed that high temperatures have a potential effect on the physiological stress of users of artificial turf. As a result, a greater compaction of the surface has been calculated when the temperature increases34. This aspect had an influence on mechanical properties, especially with regard to shock absorption, energy restitution and rotational resistance, making the surface harder35. This causes a reduction in the contact time with the surface, improving the efficiency of running technique and reducing fatigue, with longer step lengths, a more extended knee posture, and higher ankle and hip angular velocities33,35. These findings indicate that a change in an artificial turf’s mechanical properties can affect an athlete’s landing and acceleration mechanics34, thereby improving physical performance. However, negative perceptions of football players have been shown when the surface temperature is increased excessively32.
A large number of studies have been published about RSA in football, however few of them have analysed the effect of RSA on thermal and muscular response in football players. Moreover, the artificial turf temperature reached is considered a risk factor for players’ safety, but no studies have demonstrated the influence of temperature on players’ performance and safety. Therefore, the aim of this study was to analyse the effect of playing surface temperature on muscular and thermal response to a repeated-sprint ability test in football players.
Results
RSA test
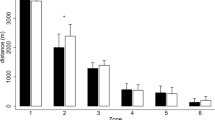
Analysis of the RSA test revealed significant differences in time and accumulated fatigue in relation to the surface temperature (p < 0.05; Fig. 1). Players revealed worse times in the RSA test when the surface temperature was lower, especially in the last sprint, in 5 m (+ 0.14 s; CI 95%: 0.05–0.23; ES: 1.15; p = 0.005) and 30 m (+ 0.24 s; CI 95% 0.09–0.39; ES: 1.25; p = 0.003). The difference in performance deterioration between the last and the first sprint was greater when the surface temperature was lower in RSADEC (+ 4.42%; CI 95% 3.53–5.30; ES: 2.07; p < 0.001) and RSACHANGE (+ 8.14%; CI 95% 5.87–10.40; ES: 2.00; p < 0.001).
30 m (A) and 5 m (B) time and performance deterioration profile; RSADEC (C) and RSACHANGE (D) for the RSA test (7 × 30 m). RSACHANGE: ((worst time – best time)/best time) * 100). RSADEC: ((total sprint time − best time * 7)/best time * 7) * 100). HSTT: high surface temperature test. LSTT: low surface temperature test. *p < 0.05; significant differences between high (white) and low (black) surface temperature (n = 32). Data are presented as mean and SD.
Thermal analysis
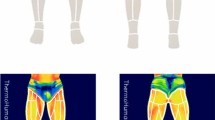
Differences in different muscle temperatures after the RSA test between groups (p < 0.05) were revealed (Table 1). Players showed a higher temperature on the thigh (+ 0.8 °C; CI 95% 0.2–1.3; ES: 1.06; p = 0.006), hamstring (+ 1.1 °C; CI 95% 0.6–1.6; ES: 1.63; p < 0.001) and calf (+ 1.0 °C; CI 95% 0.6–1.4; ES: 1.70; p < 0.001;) at a higher surface temperature; however, no significant differences were found between groups before the RSA test (p > 0.05). The contractile muscle properties of the players were independent of the surface temperature (p > 0.05). However, the players showed a significant reduction in Td (− 1.18 ms; CI 95% − 2.27 to − 0.10; ES: 0.50; p = 0.034) and Dm (− 2.62 mm; CI 95% − 4.02 to − 1.21; ES: 0.85; p = 0.001) in the biceps femoris at a higher surface temperature. Similarly, a reduction of the Dm in the BF after the RSA test was identified when the surface temperature was lower (− 1.37 mm; CI 95% − 2.70 to − 0.05; ES: 0.50; p = 0.042).
Discussion
This study analysed the effect of surface temperature on thermal and muscular response before and after an RSA test in football players. The results revealed that performance in the RSA test increased at a higher surface temperature (45.34 ± 2.53 °C), especially in the 5 m and 30 m of the sprint. On the other hand, significant differences post-RSA were found in players’ thermal profiles, depending on the field temperature. In regard to contractile properties, the RSA test decreased Dm in the BF in both groups (HSTT and LSTT) and Td decreased only in the HSTT post-RSA. However, no significant differences were found regarding field temperature.
In this study, players improved their performance in the RSA protocol at higher field temperatures, probably due to the shock absorption (SA), which is lower when the temperature is high; consequently, the surface becomes harder and the subject is able to run faster33. It is interesting to note that shock absorption is the ability of a surface to reduce the impact force of a load. Contrarily, if the temperature is lower, the SA is higher, thus running efficiency and speed decrease, and consequently a higher level of fatigue will be found35. Similar results were found in Sánchez-Sánchez et al.33, who noted that users’ performance can be improved if the surface becomes too hard because of the lower contact time during running and the less frequent reuse of the stored elastic energy of the surface with a higher damping capacity. Moreover, it has been found that too hard fields36 and too soft fields35 can adversely affect sport performance and increase the risk of injury. Thus, environmental conditions have an influence on the mechanical behaviour of artificial turf, and it has been proven that temperature alters the mechanical properties of the surface. However, other studies found that temperature, in contrast to intensity of use, did not significantly alter the mechanical behaviour of a surface, although Sánchez-Sánchez et al.33 reported a lower SA at a higher surface temperature.
Another element of the present study was muscular response analysed through tensiomyography (TMG), which has been identified as a reliable method for detecting differences in muscle responses after completing fatiguing efforts such as an RSA test29. Delay time (Td), radial displacement (Dm) and contraction time (Tc) in the biceps femoris (BF) and rectus femoris (RF) have been measured at high and low surface temperatures before and after the RSA test. Our study found a decrement in Dm in the BF after the RSA protocol, mostly at high surface temperatures. These results are in line with other investigations that reported a decrease in Dm after high-intensity exercises29. All these studies used variations of high-intensity resistance or endurance training, or interval training, over short periods of time. This reduction is interpreted as an increase in muscle stiffness that occurs after high-intensity efforts37. Furthermore, measurements of Dm were lower following high-load resistance exercise than after workload-matched high-volume training38, and Wiewelhove et al.39 reported that Dm tended to be reduced following six days of high-intensity interval training. In our study, the RSA test caused a similar reduction in Dm. Contrary to our findings, two other studies revealed an increase in Dm after ultra-endurance exercise using TMG, namely an Ironman triathlon40 and an uphill marathon39, whilst others showed no change41. Thus, the type of exercise can determine the contractile properties of the muscle, reducing Dm after high-intensity and brief efforts and increasing it after longer and less intense exercises13. According to the rectus femoris analysed in our study, no significant changes were found in any contractile property of the muscle in the RSA test at high and low temperatures, probably due to the small activation of this muscle in high-intensity efforts like the RSA, in contrast to the BF.
As for Tc, no significant differences were found in our investigation before or after the RSA test in the low and high surface temperature groups. However, conflicting results were reported for Tc, which showed a post-fatigue increase40 or decrease39, or no change29.
According to the physiological load, no significant differences were found in the HR variability parameter between high and low temperature groups during the RSA test. Similarly to studies by Hughes et al.42 and Sánchez-Sánchez et al.33, there was no influence of the surface on a player’s internal load, although physical performance was different according to the surface temperature. In conclusion, surface temperature had an influence on the players’ thermal and tensiomyographic profile and on their repeated-sprint ability test performance. This study increases knowledge about the effect of an RSA test on the thermal and tensiomiography responses of football players because no studies have been published about this topic so its application in this area could be useful. This study contributes towards an adequate use of artificial turf with the objective of converting it into a safe surface that ensures a good performance for football players.
Methods
Subjects
A group of 32 male football players (23 ± 5 years; 1.77 ± 0.06 m; 71.2 ± 6.7 kg) participated in the study. The subjects were part of two squads of a third-division football club, and they all provided written consent before participating in the research, in accordance with the Declaration of Helsinki. Test procedures and possible risks were explained in detail in the written consent. Two test sessions were performed across two different days. Each session included two tests: high temperature surface test (HSTT, at 16:00) and low surface temperature test (LSTT, at 20:00). Subjects were randomly distributed into two groups. The first group performed the first test in HSTT (day 1) and afterward in LSTT (day 2). The second group performed the first test in LSTT (day 1) and afterward in HSTT (day 2). All participants were healthy and not injured, and they were required not to perform any exhausting activity at least 24 h before testing.
Procedures
The data collection process took place immediately after the end of the regular season. The experimental tests were carried out during four different sessions distributed over two consecutive days, two sessions each day. Sessions 1 and 3 took place at 16:00 h, and sessions 2 and 4 at 20:00 h. The main part of the test protocol consisted of a repeated-sprint ability (RSA) test carried out at ambient temperature. The mean surface temperature during the tests carried out at 16:00 and 20:00 was 45.34 ± 2.53 °C and 27.21 ± 2.17 °C, being categorized as high surface temperature tests (HSTTs) and low surface temperature tests (LSTTs), respectively. The ambient temperature oscillated between 30 and 35 °C. All temperature measures were performed using a PCE-T 318 (PCE Holding GmbH, Meschede, Germany). Before and after the RSA test, the muscular response of the players was assessed by means of tensiomyography (TMG-100 System electrostimulator, TMG-BMC d.o.o., Ljubljana, Slovenia), and digital thermal images (thermograms) were recorded for the front and rear surfaces of the lower limb by means of a FLIR T420bx infrared thermographic camera (FLIR Systems Inc., Wilsonville, OR, USA). Players’ heart rate (HR) was continuously monitored during the RSA test by means of an HR monitoring band (Firstbeat Technologies Ltd., Finland). The ambient temperature and surface temperature at the time of performing the RSA test were recorded.
Tensiomyography (TMG)
The muscular response and the lateral symmetry of both the rectus femoris (RF) and biceps femoris (BF) were assessed. The contraction time (Tc), delay time (Td) and maximal radial displacement of the muscle belly (Dm) were recorded under basal conditions and after the RSA test. Figure 2 represents the meaning of those variables during a typical muscle contraction. The stimulus was of 1 ms, giving four stimuli to each muscle, varying the amplitude (25, 50, 75 and 100 mAp). The RF was measured with the subject in a supine position with a knee flexion of 120° with the help of a triangular foam cushion. The BF was measured with the subject in a prone position and the knee flexed 5º with the help of a foam cushion43.
Dm (mm) is a parameter that reflects the maximal radial displacement of the muscle belly as a consequence of muscle contraction and depends on the flexibility and tone of the muscular tissue. Therefore, Dm values increase when the explosive force is developed, involving high movement amplitude, and decrease under the conditions of a high muscular tone. Td (ms) is the time lapse between the transmission of the electric stimulus and the onset of muscle contraction (10% Dm). Tc (ms) is the time between the moment when the muscular contraction is 10% of the Dm and the moment when the contraction reaches 90% of the Dm.
The response to the electric stimulus was measured by placing a digital Dc-Dc transducer Trans-Tek (GK 40, Panoptik d.o.o., Ljubljana, Slovenia) perpendicular to the muscle belly, along with two self-adhesive electrodes (TMG electrodes, TMG-BMC d.o.o. Ljubljana, Slovenia) placed equidistantly at a distance of 50–60 mm from the digital transducer. Sensor and electrode positions were marked with a permanent marker to ensure that all measurements were performed at the same point44,45. All measurements were carried out by the same expert technician.
Thermograms
Data collection followed the standards proposed by the European Association of Thermology44. Thermograms were assessed before and after the RSA tests in an air-conditioned room, registering skin temperatures to a sensitivity of < 0.1 °C. All the thermograms were taken in similar conditions: the room temperature was maintained at 23 ± 0.3 °C, air humidity ranged between 35 and 45%, and the distance between the subject and the camera was always 3 m. Subjects began and finished the RSA test one after the other, so the time duration between exercise and testing time was the same for each participant. Tests were preceded by a 10-min acclimatization period for all subjects, surpassing the minimum time of 8 min of stabilization proposed by Roy et al.46. For each participant and session, two series of thermograms were performed on the front and rear surfaces of the lower limbs (lower legs and thighs) in a standing position. The first was taken 15 min before the RSA test, and the second 15 min after. Prior to and during the procedure, the subjects were asked to avoid any sudden and intense movement, or rubbing, scratching or crossing their legs. During the tests, subjects were dressed in shorts and sport shoes, so the selected skin surface areas were continuously exposed during the exercises and the measurements. Body regions of interest (ROIs) analysed included the thighs, hamstring and calf. Computerized image analysis allowed selection of the measurement area on the thermograms. These areas were selected by a rectangle bounded by the software Flir Tools (FLIR Systems Inc., Wilsonville, OR, USA), which provided the average temperature (Tmean) from each analysed ROI. The index of human skin emissivity was assumed to be 0.9847.
Repeated-sprint ability test (RSA)
The RSA test included seven repeated sprints of 30 m, with 20 s of active recovery between each sprint. Four pairs of photocells (Witty, Microgate, Bolzano, Italy) placed at 0, 5 and 30 m were used to assess performance in this test. This test was performed according to the methodology proposed in previous studies48. The best sprint time (RSABEST), the mean time (RSAMEAN), the total time (RSATT), the per cent sprint decrement RSADEC ((7-sprints total time − best time * 7)/best time * 7) * 100) and the per cent difference between best and worst sprints during the RSA test (RSACHANGE ((worst time – best time)/best time) * 100) were also calculated48. Before the RSA, participants carried out a standardized warm-up consisting of 5 min of running, 5 min of joint mobility and three bouts of 30 m sprints of increasing intensity. The warm-up concluded with two 30 m sprints at maximum intensity with 4 min of active recovery. These two previous sprints were used as a control measure to ensure that players would perform the RSA test at maximal intensity. During this test, if the time of the first sprint was higher (> 5%) than the best individual sprint performed prior to the beginning of the test, the RSA test was not considered valid and the player had to repeat the test after 5 min of recovery34.
Statistical analyses
SPSS 21.0 (IBM Corp, New York, USA) was used for the data analysis. A descriptive analysis (mean ± SD) of the TMG test results, the temperatures of the analysed body surfaces and the performance parameters of the RSA test was performed. Kolmogorov–Smirnov analyses indicated all data were normally distributed; therefore, parametric tests were used. Two groups were separated according to the surface temperature, using a K-means analysis. The high and low temperature groups had a surface temperature of 45.34 ± 2.53 °C and 27.21 ± 2.17 °C, respectively. Two-way analysis of variance (ANOVA) was used to analyse the differences in the TMG and the thermographic variables regarding the time (before and after the exercise) and temperature (HSTT and LSTT). Differences in performance and between the first and last sprint in the RSA test at both temperatures were analysed using a two-way ANOVA. A Bonferroni post hoc test was employed to study pairwise differences. The confidence interval and the effect size (ES; Cohen’s d) of the pre- to post-differences for all variables (CI of 95%) were calculated. The ES was evaluated with the following criteria: 0 to 0.2 = trivial; 0.2 to 0.5 = small; 0.5 to 0.8 = moderate; and > 0.8 = large49. The level of significance was established at p < 0.05.
Ethical statement
The study protocol was approved by the Local Ethics Committee (Toledo Hospital; CEIC61). All research was performed in accordance with the Code of Ethics of the World Medical Association (Declaration of Helsinki) and all of the participants signed an informed consent form in which the test procedures and possible risks were explained.
References
Spencer, M., Bishop, D., Dawson, B. & Goodman, C. Physiological and metabolic responses of repeated-sprint activities. Sports Med. 35, 1025–1044 (2005).
Bangsbo, J., Mohr, M. & Krustrup, P. Physical and metabolic demands of training and match-play in the elite football player. J. Sports Sci. 24, 665–674 (2006).
Pau, M., Ibba, G. & Attene, G. Fatigue-induced balance impairment in young soccer players. J. Athl. Train. 49, 454–461 (2014).
Rampinini, E. et al. Effect of match-related fatigue on short-passing ability in young soccer players. Med. Sci. Sports Exerc. 40, 934–942 (2008).
Faude, O., Koch, T. & Meyer, T. Straight sprinting is the most frequent action in goal situations in professional football. J. Sports Sci. 30, 625–631 (2012).
Cuadrado-Peñafiel, V., Párraga-Montilla, J., Ortega-Becerra, M. & Jiménez-Reyes, P. Repeated sprint ability in professional soccer vs. professional futsal players. E-balonmano.com: Revista de Ciencias del Deporte 10, 89–98 (2014).
Padulo, J. et al. The effect of slope on repeated sprint ability in young soccer players. Res. Sports Med. 24, 320–330 (2016).
Rampinini, E., Coutts, A. J., Castagna, C., Sassi, R. & Impellizzeri, F. Variation in top level soccer match performance. Int. J. Sports Med. 28, 1018–1024 (2007).
Bravo, D. F. et al. Sprint vs. interval training in football. Int. J. Sports Med. 29, 668–674 (2008).
Calderón-Pellegrino, G., Paredes-Hernández, V., Sánchez-Sánchez, J., García-Unanue, J. & Gallardo, L. Effect of the fatigue on the physical performance in different small-sided games in elite football players. J. Strength Cond. Res. https://doi.org/10.1519/jsc.0000000000002858 (2018).
Maior, A. S., Leporace, G., Tannure, M. & Marocolo, M. Profile of infrared thermography in elite soccer players. Motriz J. Phys. Ed. 23, e101654 (2017).
Nakayama, T., Ohnuki, Y. & Kanosue, K. Fall in skin temperature during exercise observed by thermography. Jpn. J. Phys. 31, 757–762 (1981).
MacGregor, L. J., Ditroilo, M., Smith, I. J., Fairweather, M. M. & Hunter, A. M. Reduced radial displacement of the gastrocnemius medialis muscle after electrically elicited fatigue. J. Sport Rehabil. 25, 241–247 (2016).
Rey, E., Lago-Peñas, C., Lago-Ballesteros, J. & Casáis, L. The effect of recovery strategies on contractile properties using tensiomyography and perceived muscle soreness in professional soccer players. J. Strength Cond. Res. 26, 3081–3088 (2012).
Ferreira, J. J. et al. Exercise-associated thermographic changes in young and elderly subjects. Ann. Biomed. Eng. 36, 1420–1427 (2008).
Jiang, L. et al. A perspective on medical infrared imaging. J. Med. Eng. Technol. 29, 257–267 (2005).
Fernández-Cuevas, I. et al. Classification of factors influencing the use of infrared thermography in humans: a review. Infrared Phys. Technol. 71, 28–55 (2015).
Deng, Z.-S. & Liu, J. Mathematical modeling of temperature mapping over skin surface and its implementation in thermal disease diagnostics. Comput. Biol. Med. 34, 495–521 (2004).
Fisher, G., Foster, E. B. & Pascoe, D. D. Equilibration period following exposure to hot or cold conditions when using infrared thermography. Thermol. Int. 18, 95–100 (2008).
Mohr, M., Nybo, L., Grantham, J. & Racinais, S. Physiological responses and physical performance during football in the heat. PlosOne 7, e39292 (2012).
Clark, R., Mullan, B. & Pugh, L. Skin temperature during running: a study using infra-red colour thermography. J. Physiol. 267, 53–62 (1977).
de Andrade Fernandes, A. et al. Measuring skin temperature before, during and after exercise: a comparison of thermocouples and infrared thermography. Physiol. Meas. 35, 189 (2014).
Bonnett, P., Hare, D., Jones, C., Ring, E. & Hare, C. Some preliminary observations of the effects of sports massage on heat distribution of lower limb muscles during a graded exercise test. Thermol. Int. 16, 143–149 (2006).
Merla, A., Mattei, P. A., Di Donato, L. & Romani, G. L. Thermal imaging of cutaneous temperature modifications in runners during graded exercise. Ann. Biomed. Eng. 38, 158–163 (2010).
Malkinson, T. J. In Proceedings of the Second Joint 24th Annual Conference and the Annual Fall Meeting of the Biomedical Engineering Society, Engineering in Medicine and Biology. 1297–1298 (IEEE).
Arfaoui, A., Bertucci, W. M., Letellier, T. & Polidori, G. Thermoregulation during incremental exercise in masters cycling. J. Sci. Cycling 3, 33–40 (2014).
Small, K., McNaughton, L., Greig, M. & Lovell, R. The effects of multidirectional soccer-specific fatigue on markers of hamstring injury risk. J. Sci. Med. Sport 13, 120–125 (2010).
Rusu, L. D. et al. Tensiomyography method used for neuromuscular assessment of muscle training. J. Neuroeng. Rehabil. 10, 67 (2013).
Wiewelhove, T. et al. Tensiomyographic markers are not sensitive for monitoring muscle fatigue in elite youth athletes: a pilot study. Front. Physiol. 8, 406 (2017).
Carrasco, L., Sanudo, B., de Hoyo, M., Pradas, F. & Da Silva, M. E. Effectiveness of low-frequency vibration recovery method on blood lactate removal, muscle contractile properties and on time to exhaustion during cycling at VO 2max power output. Eur. J. Appl. 111, 2271–2279 (2011).
Villacañas, V., Sánchez-Sánchez, J., García-Unanue, J., López, J. & Gallardo, L. The influence of various types of artificial turfs on football fields and their effects on the thermal profile of surfaces. P. I. Mech. Eng. P-J. Spo. 231, 11–32 (2016).
Felipe, J. L. et al. A qualitative vision of artificial turf football fields: elite players and coaches. SAJRSPER 35, 105–120 (2013).
Sánchez-Sánchez, J. et al. Influence of the mechanical properties of third-generation artificial turf systems on soccer players’ physiological and physical performance and their perceptions. PlosOne 9, e0111368 (2014).
Charalambous, L., und Wilkau, H. C. V. L., Potthast, W. & Irwin, G. The effects of artificial surface temperature on mechanical properties and player kinematics during landing and acceleration. J. Sport Health Sci. 5, 355–360 (2016).
Sánchez-Sánchez, J. et al. Effect of structural components, mechanical wear and environmental conditions on the player–surface interaction on artificial turf football pitches. Mater. Des. 140, 172–178 (2018).
Gallardo, L. et al. Effect of extrinsic factors and structural components on sport functionality of artificial turf surfaces. P. I. Mech. Eng. P-J. Spo. 233, 135–144 (2019).
Torii, M., Yamasaki, M., Sasaki, T. & Nakayama, H. Fall in skin temperature of exercising man. Br. J. Sports Med. 26, 29–32 (1992).
Raeder, C. et al. Assessment of fatigue and recovery in male and female athletes after 6 days of intensified strength training. J. Strength Cond. Res. 30, 3412–3427 (2016).
Wiewelhove, T. et al. Markers for routine assessment of fatigue and recovery in male and female team sport athletes during high-intensity interval training. PlosOne 10, e0139801 (2015).
García-Manso, J. M. et al. Effect of high-load and high-volume resistance exercise on the tensiomyographic twitch response of biceps brachii. J. Electromyogr. Kinesiol. 22, 612–619 (2012).
Pišot, R. et al. Whole muscle contractile parameters and thickness loss during 35-day bed rest. Eur. J. Appl. 104, 409–414 (2008).
Hughes, M. et al. Effects of playing surface on physiological responses and performance variables in a controlled football simulation. J. Sports Sci. 31, 878–886 (2013).
Šimunič, B. Between-day reliability of a method for non-invasive estimation of muscle composition. J. Electromyogr. Kinesiol. 22, 527–530 (2012).
Ammer, K. & Ring, E. F. J. Standard procedures for infrared imaging in medicine. In Biomedical Engineering Handbook (CRC Press, Boca Raton, 2006).
Sánchez-Sánchez, J. et al. Repeated sprint ability and muscular responses according to the age category in elite youth soccer players. Front. Physiol. 10, e175 (2019).
Roy, R., Boucher, J. P. & Comtois, A. S. Validity of infrared thermal measurements of segmental paraspinal skin surface temperature. J. Manipulat. Physiol. Ther. 29, 150–155 (2006).
Rodrigues, H. F. et al. Precise determination of the heat delivery during in vivo magnetic nanoparticle hyperthermia with infrared thermography. Phys. Med. Biol. 62, 4062 (2017).
Camm, A. J. et al. Heart rate variability: standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation 93, 1043–1065 (1996).
Cohen, J. Quantitative methods in psychology: a power primer. Psychol. Bull. 112, 1155–1159 (1992).
Author information
Authors and Affiliations
Contributions
All authors participated importantly and collaboratively in this study. G.C.P. participated in the writing of the paper, J.G.U. and E.C. contributed to data interpretation and analysis, J.S.S and L.G. revised the paper, J.G.U. prepared the figures and tables, V.P.H. participated in the design of the work, and finally, J.L.F. contributed to the acquisition of the data. All these authors have reviewed and approved this manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Calderón-Pellegrino, G., Gallardo, L., Paredes-Hernández, V. et al. Influence of artificial turf temperature on physical performance and muscle contractile properties in football players after a repeated-sprint ability test. Sci Rep 10, 12747 (2020). https://doi.org/10.1038/s41598-020-69720-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-69720-6
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.