Abstract
Zika virus (ZIKV) is an emerging flavivirus transmitted to humans by Aedes mosquitos. ZIKV can be transmitted from mother to fetus during pregnancy and can cause microcephaly and other birth defects. Effective vaccines for Zika are yet to be approved. Detection of the ZIKV is based on serological testing that often shows cross-reactivity with the Dengue virus (DENV) and other flaviviruses. We aimed to assemble a highly specific anti-Zika antibody panel to be utilized in the development of a highly specific and cost-effective ZIKV rapid quantification assay for viral load monitoring at point-of-care settings. To this end, we tested the affinity and specificity of twenty one commercially available monoclonal and polyclonal antibodies against ZIKV and DENV envelope proteins utilizing nine ZIKV and twelve DENV strains. We finalized and tested a panel of five antibodies for the specific detection and differentiation of ZIKV and DENV infected samples.
Similar content being viewed by others
Introduction
Isolated in 1947 from a rhesus monkey1 the Zika virus (ZIKV) is a member of the virus family Flaviviridae (genus Flavivirus). ZIKV is transmitted by various species of Aedes mosquitoes2,3 and was not considered critical to global health until the last decade, after a series of outbreaks on several Pacific islands4,5,6,7. In the United States, the first outbreak of Zika was reported in 2016 with a total of 5,168 symptomatic cases in the continental US and 35,395 cases in Puerto Rico8,9. In 2015 and 2016, total 1,673,272 cases were reported in Brazil10. To date, Zika virus has circulated to all continents with the exception of Europe and Antarctica11. Without the availability of an effective vaccine the prevention of disease transmission is dependent on the early diagnosis of the virus to determine the site and size of an outbreak and the effectiveness of vector control measures12.
ZIKV is an enveloped virus with a single-stranded, positive sense RNA genome coding for three structural proteins (C, prM/M, and E) and seven non-structural proteins (NS1, NS2A, NS2B, NS3, NS4A, NS4B, and NS5)13. Dengue viruses (DENV1-4), of the genus Flavivirus, are structurally and genetically related to ZIKV14,15 and are also transmitted by Aedes spp. mosquitoes14. The envelope (E) protein of both ZIKV and DENV, is highly immunogenic and is expressed on the surface of the virus to mediate the binding and the membrane fusion of the target cell16,17.
The viral envelope protein consists of three main domains (ED I, II, III). Neutralizing antibodies (primarily IgG class) against the ED I and ED II domains are more prone to show cross-reactivity between DENV and ZIKV due to a higher homology (ED I (35%) and ED II (51%)) compared to the ED III domain (29%)18. As a result of the considerable structural and genetic similarities between ZIKV and DENV, neutralizing antibodies often show cross-reactivity in serological assays. Immunological cross-reactivity between ZIKV and DENV have already been reported during the Yap State (Micronesia) outbreak19. Furthermore, in the Americas and Africa, DENV and yellow fever virus, also a member of the Flaviviridae family, can be found in the same geographical areas, and DENV present symptoms similar to ZIKV, hence a precise differential diagnosis among these viruses is critical to implement the proper monitoring and prevention strategies20,21,22,23.
Currently, identification of ZIKV infection is accomplished by i) testing the serum to detect viral nucleic acid using RT-PCR, ii) testing the serum for the presence of the non-structural 1 (NS1) protein antigen or iii) serological assays to determine the presence of virus-specific immunoglobulin IgG and IgM antibodies using enzyme-linked immunosorbent assay (ELISA)3,24. Unfortunately, ZIKV IgM-ELISA displays high specificity, but poor sensitivity, while the ZIKV IgG-ELISA are characterized by low specificity and cross-reactivity in patients previously exposed to dengue infections25. Other assays, such as the plaque reduction neutralization test (PRNT), can be performed to measure virus-specific neutralizing antibodies but show high accuracy only after day 7 of the disease onset2,26,27,28, are labor-intensive, expensive and time-consuming. Similarly, RT-PCR assays, although highly specific29,30,31, are expensive and require multiple labor-intensive sample preparation steps. Considering all the factors and limitations of the Zika detection methods currently utilized, there is an unmet need to develop a rapid, inexpensive, minimally labor-intensive, and highly specific detection assay for ZIKV that can be utilized in point of care settings without the access to specialized equipment and facilities.
In the USA, ZIKV diagnostic assays for either detection of antibodies or nucleic acid from ZIKV were not available before 2016. Since 2016 the FDA has issued an Emergency Use Authorization (EUA) for fourteen molecular-based assays for the detection of genetic material in samples of bodily fluids, such as serum and urine, along with five serological-based assays for the detection of antibodies against ZIKV in the blood32. Among the assays one (CDC Zika MAC-ELISA) utilizes noninfectious ZIKV-like particles, another one (InBios) uses recombinant ZIKV E glycoprotein whereas the rest utilize recombinant ZIKV NS1 antigen33. Although individual serologic assays have a prolonged window of detection, they also have disadvantages. The CDC and the InBios assays show lower specificity34 due to the similarities in the antigenic structure of the E protein between DENV and ZIKV while the majority of the other assays shows lower sensitivity34,35,36. The current ZIKV serological assays only comprise IgM class antibodies and to date, there is no FDA EUA approved IgG based ELISA assay due to the higher potentiality of cross-reactivity. CDC guideline suggests additional PRNT testing if a sample results are positive with any of the above-mentioned assays37. Therefore, the development of tools that allow ZIKV E protein detection in various experimental conditions is of utmost importance. The selection of highly specific and non-cross-reactive antibodies is the first step for the development of an effective detection platform for the ZIKV.
In this study, we have evaluated the sera cross-reactivity of twenty one monoclonal and polyclonal antibodies against the ZIKV and DENV E proteins to assemble a highly specific panel of antibodies for the specific detection and differentiation of ZIKV from DENV. The antibodies were tested against nine ZIKV and twelve DENV strains. Next, the panel of selected antibodies were tested with deidentified ZIKV and DENV viral culture lysates and their lower limit of detection was determined by western blot.
Materials and methods
Antibodies and viruses
We utilized twenty one commercially available antibodies purified from either mouse hybridoma cell line or rabbit against the ZIKV or DENV E protein (Table S1). HRP-conjugated anti-rabbit IgG and anti-mouse IgG antibodies were used as secondary antibodies (GE Healthcare Life Science). Twelve different strains of DENV and nine different strains of ZIKV were obtained from the ATCC and BEI resources repositories (Table S2). Four virus samples (one DENV and three ZIKV) were quantified by foci-forming assay38 and plaque assay39 and utilized for the antibody panel specificity and affinity testing.
Cell culture
Vero cells were grown in Dulbecco's Modified Eagle Medium with 10% Fetal Bovine Serum and 1% Gentamicin at 37° C in 5% CO2.
SDS-PAGE and western blot
ZIKV/DENV samples were mixed with 2 × Laemmli Sample Buffer containing 10% β-mercaptoethanol and heated at 95 °C for 5 min. Samples containing either ZIKV or DENV were separated utilizing a 10% SDS–polyacrylamide, electroblotted onto a nitrocellulose membrane (Thermofisher). The membrane was blocked at room temperature for 30 min in 5% milk–TBST (50 mM Tris, 150 mM NaCl and 0.2% Tween-20) and then, probed with the primary antibodies (60 min) listed in Table 1, the HRP conjugated secondary antibody (1:5,000 dilution in 2.5% milk–TBST, for 60 min) and stained using the Supersignal West Femto Maximum Sensitivity substrate (Thermofisher). Luminescence was quantified utilizing an Odyssey classic imaging system (LICOR Biosciences, Bad Homburg, Germany). All assays were run as independent duplicates.
RNA extraction, cDNA synthesis, and real-time qPCR
RNA was extracted using Trizol Reagent (Invitrogen) according to the manufacturer’s protocol. 11 µl of RNA (out of total 50 µl of RNA) was used for cDNA synthesis with SuperScript Reverse Transcriptase system (Invitrogen, California, USA). 2 µl of (1:3 diluted) synthesized cDNA was used for RT-qPCR analysis using Green-2-Go qPCR mastermix (Bio Basic, New York, USA.) with a pair of primers specific for NR–50355 and NR-50245 genomic sequences (5′-GCAAACTGTCGTGGTTCTAG-3′, 5′-CTTTGCACCATCCATCTCAG-3′). Synthesized DNA from a conserved 429 nt region of the ZIKV genome was used as standard. PCR amplifications were performed on an AriaMx Real-time PCR System (Agilent, California, USA) for thermal cycling and SYBR detection with three technical replicates for each sample. The quantification of the ZIKV samples was determined by comparing the cycle threshold (Cq) value based on the standard curve generated by the known DNA samples amount.
Results
Validation of the specificity of anti-ZIKV and anti-DENV E protein antibodies
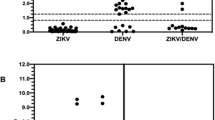
We obtained 19 antibodies raised against either the ZIKV or DENV Envelope (E) protein and two pan-Flavivirus (anti-E protein) antibody with specificity for DENV-1, 2, 3, 4, Japanese encephalitis virus, West Nile virus, yellow fever virus and ZIKV (Table S1). We obtained tissue culture supernatants for twelve different strains of DENV (subtypes 1–4) and nine different strains of ZIKV from the ATCC and BEI Resources repositories (Table S2). E protein amino acid sequences were aligned for both ZIKV/DENV strains using Clustal Omega 2.1 to determine their structural differences/identities with each other (Supplementary Figs. S1 and S2). Moreover, phylogenetic analysis was performed to show evolutionary relationships for all ZIKV/DENV E proteins (Supplementary Fig. S3). The protein quantity of each viral lysates was quantified using standard BCA protein assay reagent (Pierce, Rockford, IL, USA) in 96 well plates following the manufacturer guidelines (Supplementary Fig. S4). Five ZIKV viral isolates exhibited the same E sequence which are from central/south America region, nevertheless their viral preparations and the titer are different. SDS PAGE/Western blot assays were carried out for all the possible antibody/viral strain combinations to determine the specificity and cross-reactivity of all the antibodies tested (Supplementary Figs. S5 and S6). A western blot approach was chosen given the higher sensitivity and specificity compared to standard ELISA assays and other quantitative/qualitative immunohistochemical techniques. Although this technique might display lower sensitivity if the antibody recognizes with high affinity a higher-order structural conformation of the epitope. Tables 1 and 2 summarize the results of the antibodies tested with the ZIKV and DENV strains respectively.
13 out of the 21 the antibodies tested showed reactivity with the E protein from either ZIKV, DENV or both (Figs. S5, S6 and Tables 1, 2). Ten out of the eleven anti-Zika antibodies demonstrate good to excellent reactivity with the ZIKV E protein (Fig. S5, Table 2); however, four of these antibodies displayed marked cross-reactivity with multiple DENV strains. One anti-ZIKV antibodies did not recognize either DENV or ZIKV E proteins. Three of the anti-Zika antibodies that specifically recognized the ZIKV E protein also recognized a higher molecular weight (MW) protein, possibly of cellular origin, but did not cross-react with the DENV E protein. Only three of the eight anti DENV E protein antibodies tested recognized one or more of the DENV strains, but none exhibited cross-reactivity with the ZIKV E protein or with cellular proteins. Moreover, the pan-Flavivirus antibodies, with the exception of a weak signal from one of the DENV type 1 samples for Ab NR-50327, did not recognize any of the ZIKV or DENV samples. This can be partially explained by the fact that some of these antibodies (NR-4757, NR255, NR-50327) might recognize structured epitopes and their activity in denaturing PAGE/western blot assays had not been previously tested.
Overall, our data indicate that only a subset of the commercially available antibodies can be reliably utilized in the analysis of ZIKV and DENV samples given the marked cross-reactivity observed in four of the ZIKV specific antibodies and the fact that one third of the tested antibodies were unable to recognize any of the tested ZIKV or DENV strains.
Validation of a ZIKV/DENV antibody panel
A panel of five antibodies was selected based on their specificity and affinity as determined by the western blot data presented in Figs. S5, S6 and Tables 1, 2. The antibodies selected can be divided into three categories: (i) three anti-ZIKV E protein specific antibodies (BF1176-56, GTX634157, GTX133314), (ii) one anti-DENV E serotype 2 protein specific antibodies (GTX127277) and iii) one antibody that can recognize both the ZIKV and DENV E proteins (GTX133326). Antibodies that showed a high affinity for the ZIKV E protein but also recognized other unknown antigens, possibly of cellular origin, were excluded from the panel.
The antibodies selected were validated utilizing three ZIKV and one DENV viral samples of known titer. (Figs. 1, S7 and data Summary in Table 3). Each viral preparation was analyzed at different concentrations (from 5 × 106 to 8 × 103 FFU/mL or PFU/mL) to determine both the specificity and sensitivity of the antibody panel. The testing of the viral samples carried out with the ZIKV/DENV antibody panel identified the DENV (sample 1) and three ZIKV (samples 2,3,4) viral preparations. The limit of detection for each antibody, determined by western blot, is shown in Table 4, with a lower limit varying for each antibody and virus tested and ranging from 4.0 × 104 PFU/mL (for ZIKV specific and ZIKV + DENV specific antibodies) to 5.0 × 106 FFU/mL for DENV specific antibodies. Overall the panel of antibody we selected was able to accurately specify and differentiate between ZIKV and DENV type 2 infected samples.
Discussion and conclusion
In this study, we evaluated the reactivity of commercially available anti-DENV and anti-ZIKV E protein antibodies utilizing several DENV and ZIKV isolates. We observed that, although 13 out of 21 of the antibodies tested were reactive to one or more viral samples, eight failed to recognize viral antigens in any of the 21 viral samples tested. Viruses from different isolates were recognized with different affinity by the same antibody, this is likely to be due to both differences in the sequences and structure of the viral E protein and differences in the viral titer of the samples tested. Quantification by qPCR of the isolate NR-50355 and NR-50245 (1 × 108 and 3 × 107 genome copies/mL respectively, Fig. S8) confirmed a strict correlation between the western blot data (Tables 1 and 2) and viral titer. Furthermore, the signal detected with all ZIKV strains by four of the anti-Zika E protein antibodies (BF-1176-56, MBS5304716, GTX133325 and GTX133326) confirmed that the viral titer in all the ZIKV samples were sufficiently high to be easily detected by western blot. Nevertheless, it is plausible that other types of assays or different experimental conditions might result in the detection of the viral proteins with some of the antibodies and failed to work in our assays. Moreover, the E protein of three DENV isolates (NR-86, NR-49757 and NR-84) was not recognized by any of the DENV specific antibody tested, although, a band corresponding to a higher MW cellular protein was observed with three of the ZIKV antibodies (Fig S6). However, additional dengue antibodies can be tested in the future. Overall, we validated 3 anti-DENV E protein antibodies, 6 anti-ZIKV E protein antibodies and 4 antibodies that recognized both DENV and ZIKV E proteins. We selected a panel of 5 antibodies (3 ZIKV specific, 1 DENV specific and one cross-reactive for both ZIKV and DENV) for their specificity and sensitivity. The antibody panel was validated utilizing three ZIKV and one DENV samples. Analysis of the viral samples correctly identified the DENV and ZIKV viruses with an upper limit of detection of roughly 5.0 × 106 PFU/mL.
Currently, the FDA emergency-use-authorized and CDC-developed IgM antibody capture enzyme-linked immunosorbent assay (MAC-ELISA) is considered as the gold standard for serological fluids testing for ZIKV, although a PRNT test is required to confirm the positive results32,40. The panel of antibodies that were reported here can also be used for developing rapid ZIKV specific, and possibly DENV specific if other DENV-specific antibodies are added to it, detection and quantification assays, to be used at port of entry, urgent care centers, and other resource-limited settings if integrated with technologies such as microfluidic channeled41, optical photonic crystal42, colorimetric analysis43,44,45,46,47, and plasmon resonance48.
References
Dick, G., Kitchen, S. & Haddow, A. Zika virus (I). Isolations and serological specificity. Trans. R. Soc. Trop. Med. Hyg. 46, 509–520 (1952).
Wong, S.S.-Y., Poon, R.W.-S. & Wong, S.C.-Y. Zika virus infection—the next wave after dengue?. J. Formos. Med. Assoc. 115, 226–242 (2016).
Herrada, C., Kabir, M. A., Altamirano, R. & Asghar, W. Advances in Diagnostic methods for Zika Virus infection. J. Med. Devices 12, 4080211 (2018).
Duffy, M. R. et al. Zika virus outbreak on Yap Island, federated states of Micronesia. N. Engl. J. Med. 360, 2536–2543 (2009).
Cao-Lormeau, V.-M. et al. Zika virus, French polynesia, South pacific, 2013. Emerg. Infect. Dis. 20, 1085 (2014).
Dupont-Rouzeyrol, M. et al. Co-infection with Zika and dengue viruses in 2 patients, New Caledonia, 2014. Emerg. Infect. Dis. 21, 381 (2015).
Tognarelli, J. et al. A report on the outbreak of Zika virus on Easter Island, South Pacific, 2014. Adv. Virol. 161, 665–668 (2016).
Marini, G., Guzzetta, G., Rosà, R. & Merler, S. First outbreak of Zika virus in the continental United States: a modelling analysis. Eurosurveillance 22, 30612 (2017).
Control, C. f. D. 2016 Case Counts in the US, https://www.cdc.gov/zika/reporting/2016-case-counts.html (2016).
de Oliveira, W. K. et al. Infection-related microcephaly after the 2015 and 2016 Zika virus outbreaks in Brazil: a surveillance-based analysis. The Lancet 390, 861–870 (2017).
Boyer, S., Calvez, E., Chouin-Carneiro, T., Diallo, D. & Failloux, A.-B. An overview of mosquito vectors of Zika virus. Microbes Infect. 20, 646–660 (2018).
Rather, I. A. et al. Prevention and control strategies to counter dengue virus infection. Frontiers in cellular and infection microbiology 7, 336 (2017).
Dai, L. et al. Structures of the Zika virus envelope protein and its complex with a flavivirus broadly protective antibody. Cell Host Microbe 19, 696–704 (2016).
Uno, N. & Ross, T. M. Dengue virus and the host innate immune response. Emerg Microbes Infect. 7, 167 (2018).
Suwanmanee, S. & Luplertlop, N. Dengue and Zika viruses: lessons learned from the similarities between these Aedes mosquito-vectored arboviruses. J. Microbiol. 55, 81–89 (2017).
Faye, O. et al. Molecular evolution of Zika virus during its emergence in the 20th century. PLoS Negl. Trop. Dis. 8, e2636 (2014).
Keasey, S. L. et al. Antibody responses to Zika Virus infections in environments of flavivirus endemicity. Clin. Vaccine Immunol https://doi.org/10.1128/cvi.00036-17 (2017).
Tran, A. & Theel, E. S. The role of serologic testing for Zika virus infection. Rev. Med. Microbiol. 29, 1–7 (2018).
Priyamvada, L., Hudson, W., Ahmed, R. & Wrammert, J. Humoral cross-reactivity between Zika and dengue viruses: implications for protection and pathology. Emerg. Microbes Infect. 6, e33 (2017).
Stettler, K. et al. Specificity, cross-reactivity and function of antibodies elicited by Zika virus infection. Science https://doi.org/10.1126/science.aaf8505 (2016).
Escadafal, C. et al. Rapid molecular assays for the detection of yellow fever virus in low-resource settings. PLoS Negl Trop Dis 8, e2730 (2014).
Faria, N. R. et al. Genomic and epidemiological monitoring of yellow fever virus transmission potential. Science 361, 894–899 (2018).
Lee, C. T. et al. Zika virus surveillance and preparedness—New York City, 2015–2016. Morb. Mortal. Wkly Rep. 65, 629–635 (2016).
Moulin, E., Selby, K., Cherpillod, P., Kaiser, L. & Boillat-Blanco, N. Simultaneous outbreaks of dengue, chikungunya and Zika virus infections: diagnosis challenge in a returning traveller with nonspecific febrile illness. New Microbes New Infections 11, 6–7 (2016).
Kikuti, M. et al. Diagnostic performance of commercial IgM and IgG enzyme-linked immunoassays (ELISAs) for diagnosis of Zika virus infection. Virol. J. 15, 108. https://doi.org/10.1186/s12985-018-1015-6 (2018).
Balmaseda, A. et al. Antibody-based assay discriminates Zika virus infection from other flaviviruses. Proc. Natl. Acad. Sci. USA 114, 8384–8389 (2017).
Rockstroh, A. et al. Specific detection of dengue and Zika virus antibodies using envelope proteins with mutations in the conserved fusion loop. Emerg Microbes Infect. 6, e99 (2017).
Lindsey, N. P. et al. Ability to serologically confirm recent Zika virus infection in areas with varying past incidence of dengue virus infection—United States and territories, 2016. J. Clin. Microbiol. https://doi.org/10.1128/JCM.01115-17 (2017).
Santiago, G. A. et al. Performance of the Trioplex real-time RT-PCR assay for detection of Zika, dengue, and chikungunya viruses. Nat. Commun. 9, 1391 (2018).
L’Huillier, A. G. et al. Evaluation of Altona Diagnostics RealStar Zika Virus RT-PCR Test Kit for Zika virus PCR testing. J. Clin. Microbiol. 55, 1576–1584 (2017).
Balmaseda, A. et al. Comparison of four serological methods and two RT-PCR assays for diagnosis and surveillance of Zika. J. Clin. Microbiol. https://doi.org/10.1128/JCM.01785-17 (2018).
FDA. Zika Virus Response Updates from FDA (FDA, Silver Spring, 2018).
Theel, E. S. & Hata, D. J. Diagnostic testing for Zika Virus: A Post-Outbreak Update. J. Clin. Microbiol. https://doi.org/10.1128/JCM.01972-17 (2018).
Granger, D. et al. Serologic testing for Zika virus: comparison of three Zika virus IgM-screening enzyme-linked immunosorbent assays and initial laboratory experiences. J. Clin. Microbiol. 55, 2127–2136 (2017).
Sloan, A. et al. Evaluation of the Diasorin Liaison® XL Zika Capture IgM CMIA for Zika virus serological testing. Diagn. Microbiol. Infect. Dis. 90, 264–266 (2018).
Herrada, C. A., Kabir, M. A., Altamirano, R. & Asghar, W. Advances in Diagnostic Methods for Zika Virus Infection. J. Med. Devices 12, 040802 (2018).
Oduyebo, T. et al. Update: interim guidance for health care providers caring for pregnant women with possible Zika Virus Exposure—United States (Including U.S. Territories). MMWR Morb Mortal Wkly Rep 66, 781–793. https://doi.org/10.15585/mmwr.mm6629e1 (2017).
Sessions, O. M. et al. Discovery of insect and human dengue virus host factors. Nature 458, 1047 (2009).
Agbulos, D. S., Barelli, L., Giordano, B. V. & Hunter, F. F. Zika virus: quantification, propagation, detection, and storage. Curr. Protoc. Microbiol. 43, 15 (2016).
Musso, D. & Gubler, D. J. Zika virus. Clin. Microbiol. Rev. 29, 487–524 (2016).
Asghar, W. et al. Engineering long shelf life multi-layer biologically active surfaces on microfluidic devices for point of care applications. Sci. Rep. 6, 21163 (2016).
Shafiee, H. et al. Nanostructured optical photonic crystal biosensor for HIV viral load measurement. Sci. Rep. 4, 4116 (2014).
Coleman, B., Coarsey, C., Kabir, M. A. & Asghar, W. Point-of-care colorimetric analysis through smartphone video. Sens. Actuators B 282(221), 251 (2018).
Chin, C. D. et al. Microfluidics-based diagnostics of infectious diseases in the developing world. Nat. Med. 17, 1015–1019 (2011).
Coleman, B., Coarsey, C. & Asghar, W. Cell phone based colorimetric analysis for point-of-care settings. Analyst 144, 1935–1947 (2019).
Coarsey, C., Coleman, B., Kabir, M. A., Sher, M. & Asghar, W. Development of a flow-free magnetic actuation platform for an automated microfluidic ELISA. RSC Adv. 9, 8159–8168. https://doi.org/10.1039/C8RA07607C (2019).
Kabir, M. A., Zilouchian, H., Sher, M. & Asghar, W. Development of a Flow-Free Automated Colorimetric Detection Assay Integrated with Smartphone for Zika NS1. Diagnostics 10, 42 (2020).
Inci, F. et al. Nanoplasmonic quantitative detection of intact viruses from unprocessed whole blood. ACS Nano 7, 4733–4745 (2013).
Acknowledgements
We acknowledge research support from Florida Department of Health (FDOH) 7ZK10, NIH R15AI127214, Institute for Sensing and Embedded Networking Systems Engineering (I-SENSE) Research Initiative Award, FAU Faculty Mentoring Award, Humanity in Science Award, and a start-up research support from College of Engineering and Computer Science, Florida Atlantic University, Boca Raton, FL.
Author information
Authors and Affiliations
Contributions
Conceptualization: M.A.K, M.C., W.A. Data curation: M.A.K., M.C. Formal analysis: M.A.K, M.C. Funding acquisition: M.C., W.A. Methodology: M.C, W.A. Resources: S.S., M.G.B., R.S.A., S.B., M.C., W.A. Supervision: M.C., W.A. Validation: M.A.K., R.S.A. Visualization: M.A.K, M.C. Writing—original draft: M.A.K., M.C. Writing—review & editing: M.A.K., M.C., W.A., M.G.B., R.S.A., S.B.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kabir, M.A., Soto-Acosta, R., Sharma, S. et al. An antibody panel for highly specific detection and differentiation of Zika virus. Sci Rep 10, 11906 (2020). https://doi.org/10.1038/s41598-020-68635-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-68635-6
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.