Abstract
The selected A. tricolor accessions contained abundant color attributes, betacyanin, carotenoids, betalains, betaxanthins, and antioxidants potentiality that varied in terms of genotypes. For the first time, we identified 4 betacyanins, and 5 carotenoid compounds in A. tricolor genotypes. The genotype VA14 and VA16 had abundant color attributes, betacyanin such as amaranthine, iso-amaranthine, betanin, iso-betanin, and antioxidants potentiality. These two genotypes having an excellent source of color attributes, betacyanins, betalains, betaxanthins, and antioxidants potentiality could be used as potent antioxidant varieties. The genotype VA11 and VA16 had abundant carotenoid components, such as zeaxanthin, lutein, violaxanthin, neoxanthin, total xanthophylls, and beta-carotene. The genotype VA11 and VA16 had abundant carotenoid components that could be used as carotenoid enrich varieties. It revealed from the correlation study that pigment profiles of A. tricolor genotypes exhibited high quenching capacity of radicals. These accessions have high antioxidant potentials and great opportunity to make drinks, preservatives, and colorant of food products to feed the community deficient in antioxidants. The identified components of betacyanins and carotenoids in A. tricolor require comprehensive pharmacological study. The baseline data on color attributes, betacyanins profile, carotenoids profile, betaxanthins, betalains and antioxidant potentiality obtained in the present study could contribute to pharmacologists for evaluating these components scientifically in A. tricolor.
Similar content being viewed by others
Introduction
Edible stems and leaves of Amaranthus are an inexpensive and abundant source of digestive fiber, protein containing methionine and lysine, vitamin C, carotenoids, minerals1,2,3,4,5,6. It has also abundant antioxidant pigments, such as betacyanin, anthocyanin, betaxanthin, betalain, carotenoids, and chlorophylls7,8,9,10; bioactive phytochemicals including ascorbic acid, flavonoids, and phenolic acids11,12,13,14. These bioactive components of natural origin have a remarkable contribution to the industry of food as these quench reactive oxygen species (ROS) and cure many diseases including cardiovascular diseases, arthritis, cancer, cataracts, retinopathy, atherosclerosis, emphysema, and neurodegenerative diseases15,16,17,18.
The acceptability of foods mostly depends on color, flavor, and taste. For these reasons, recently coloring products of foods become very popular due to meet up the general interest of the people around the globe. These products meet up the general interest of consumers in the safety, nutritional and aesthetic aspects of foods. It meet up the requirement for betacyanin, anthocyanin, carotenoids, and chlorophylls. Amaranthus leafy vegetable is a sole source of betacyanins including amaranthine, iso-amaranthine, betanin, iso-betanin that has important scavenging activity of free radicals19. Betacyanin could be used as a food colorant in low-acid foods. It has higher pH stability than anthocyanins20. Amaranthine, a major pigment of betacyanin in amaranth had very strong quenching capacity of radicals. It is an alternate source of betanins (red beets) for colorant of foods and potential antioxidants19. It is widely adapted to abiotic stresses including drought21,22,23,24 and salinity25,26,27. Red color amaranth has more pigments, such as betacyanin and carotenoids than green color amaranth. Amaranth leaves inhibited the proliferation of colon (Caco-2) cancer cell lines, breast (MCF-7) cancer cell lines, liver (HepG2) and exhibited anticancer potential28.
In many developing countries including Bangladesh and India, deficiency of vitamin A and macular degeneration associated with aging are serious threats among adults and children. In developing countries, age-related macular degeneration is increasing at an alarming rate. Consumption of inadequate macular pigments and pro-vitamin A in regular diet is the main cause of deficiency of vitamin A and macular degeneration associated with aging. So, regular consumption of vitamin A enriches vegetables may be the solution of deficiency of vitamin A and macular degeneration associated with aging. A. tricolor is the inexpensive natural source of pro-vitamin A due to high content of beta-carotene and xanthophylls profile including lutein, violaxanthin, neoxanthin, and zeaxanthin. The carotenoids profile of crops varies markedly in different climatic conditions. Carotenoids profile of the same species may vary due to the difference in eco-geographical regions. Recently, we have been exploring the possibility of pigments and phenolic profiles of A. tricolor including antioxidant potentials as it has abundant natural betacyanins and carotenoids profile containing macular pigments along with phenolic profiles, antioxidant constituents, and antioxidant activities of interest in the industry of foods17,29. In our earlier studies, we selected a few high yields and antioxidant potential A. tricolor genotypes from 43 germplasms. It is the first attempt to study the color attributes, betacyanin, carotenoids, and phenolic profiles, antioxidant constituents, and antioxidant potentials in selected A. tricolor elaborately through high performance liquid chromatograph. Therefore, we ultimately evaluate the possibility of selection of appropriate genotypes for extracting colorful juice as drinks, preservatives, and colorant of food products containing abundant betacyanin, carotenoids, and phenolic profiles, antioxidant constituents, and antioxidant potentials.
Results and discussion
Color attributes
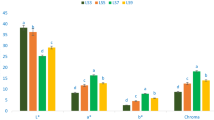
Figure 1 shows the leaf color attributes of four selected A. tricolor. The pronounced variability was noted with regard to b*, lightness (L*), a*, and chroma of the four accessions studied. The range of chroma, a*, b*, and lightness (L*) were 8.15 to 19.73, 7.35 to 17.68, 3.51 to 8.75, and 23.78.34 to 40.35, respectively. The maximum values of lightness was noticed in VA6 (40.35), while the minimum values of lightness was reported in VA14 (23.78) followed by VA16 (26.84). Similarly, the maximum a* (17.68), chroma (19.73), and b* (8.75) value were noted in VA14 followed by VA16, while the lowest a* (7.35), b* (3.51), and chroma (8.15) were found in VA6. We found corroborative findings with the findings of Colonna et al.30 and Sarker and Oba31. Color attributes are essential parameters that have contributed significantly to the choice of consumers, their acceptability and preferability. These are main attributes for testing the potentiality of antioxidants in vegetables30. Deep red accessions VA14 and VA16 had high values of yellowness and redness representing the occurrence of plentiful pigments including betaxanthin, anthocyanins, betacyanin, and betalain. Conversely, the accession VA6 exerted low values of yellowness and redness representing the occurrence of low pigments including betaxanthin, anthocyanins, betacyanin, and betalain. The four bright red color accessions have ample betacyanin pigments showing better stability at pH 5–7 and lower temperatures (<14 °C)32. These accessions have great opportunity to make drinks, preservatives and colorant of food products.
Betacyanin components
The betacyanin is responsible for the red-violet and maroon color in the appearance of A. tricolor genotypes. Table 1 shows main fragment ions (MS2), molecular ion, retention time, λmax, and detected betacyanin components. The liquid chromatograph separated values of betacyanin components from four genotypes (VA16, VA14, VA11, and VA6) were compared with standard betanin (Betanin from red beet were used as standards) using the respective peaks of the compounds. Figure 2 represents the individual betacyanin components and their relative proportions in the extracts. The HPLC determined betacyanin exhibited 2 principal peaks (peak1 and peak2) at 538 nm (Table 1). Peak1 and peak2 were eluted before the peaks of betanin and iso-betanin standards (peak3 and peak4), representing a greater intensity of glycosylation33. The betacyanin components amaranthine and iso-amaranthine were abundant pigments in Amaranthus34. Cai et al.35 also reported that amaranthine as well as iso-amaranthine (C15 epimer of amaranthine) were widely distributed and available (91.5%) in forty accessions of thirty seven species of eight genera of Amaranthus. Betacyanin components amaranthine and betanin have betanidin (unit of aglycone). Betanin has glucoside in lieu of glucuronosylglucoside of amaranthine. Both peak3 and peak4 represented the molecule at m/z 551 [M + H]+, matching the mass of glucoside of betanidin, and their aglycone ion (main fragment ion) were at m/z 389 [M-glucose + H]+. In A. tricolor, both peak1 and peak2 represented the molecule at m/z 727 [M + H]+, corresponding to glucuronosylglucoside of betanidin. Both peak1 and peak2 represented the major fragment ions at m/z 551 [M-glucuronic acid + H]+, and with the same aglycone ion at m/z 389 [M-glucuronosylglucose + H]+ (Table 1). Based on the UV–Vis and MS data presented in Table 1 and those obtained by others, peak1 and peak2 were tentatively identified as amaranthine and iso-amaranthine (C15 epimer), respectively32,33.
Betacyanin profiles in four selected A. tricolor leafy vegetables, Amaranthine (mg 100 g−1), Iso-amaranthine (mg 100 g−1), Betanin (mg 100 g−1), Iso-betanin (mg 100 g−1), Total betacyanin (mg 100 g−1), different letters in the bar are differed significantly by Duncan Multiple Range Test ((P < 0.01), (n = 3).
Considering betacyanin components, amaranthine was the most abundant betacyanin followed by iso-amaranthine in A. tricolor genotypes (Fig. 2). Our obtained amaranthine, iso-amaranthine, betanin and iso-betanin content in A. tricolor genotype VA14 and VA16 were greater than the results of Stinzing et al.33 in A. spinosus stem. Amaranthine, iso-amaranthine, betanin, iso-betanin, and total betacyanin ranged from 7.82 to 22.84, 5.32 to 12.83, 3.37 to 6.84, 2.21 to 5.32, and 19.60 to 46.52 mg 100 g−1, respectively (Fig. 2). The highest amaranthine (22.84 mg 100 g−1), betanin (6.84 mg 100 g−1), iso-betanin (5.32 mg 100 g−1) and total betacyanin (46.52 mg 100 g−1) were obtained from the genotype VA14 followed by the genotype VA16. Cai et al.19 also reported that amaranthine was the major compounds in betacyanin components in the plants of Amaranthaceae which corroborated with the present findings. Whereas, the genotype VA16 showed the highest iso-amaranthine (12.83 mg 100 g−1) followed by VA14. In contrast, the genotype VA11 showed the lowest amaranthine (7.82 mg 100 g−1), iso-amaranthine (5.32 mg 100 g−1), iso-betanin (2.21 mg 100 g−1) and total betacyanin (19.60 mg 100 g−1) and the genotype VA6 exhibited the lowest betanin (3.37 mg 100 g−1). Within leafy vegetables, amaranth is a unique and excellent source of betacyanin components including amaranthine, iso-amaranthine, betanin, iso-betanin. These betacyanin components have high scavenging activity of free radicals and act as an excellent antioxidants19. Betacyanin components could be used as a food colorant in low-acid foods as these components have higher pH stability compared to anthocyanins20. Amaranthine, a major pigment of betacyanin components in amaranth had very strong quenching capacity of radicals and also act as an excellent antioxidants. It is an alternate source of betanins (red beets) for colorant of foods and potential antioxidants19. Hence, the genotypes VA14 and VA16 containing high betacyanin profiles including amaranthine, iso-amaranthine, betanin, iso-betanin could be used for extracting colorful juice as drinks, preservatives, and colorant of food products.
Carotenoid profiles
Table 2 represents the data on the molecular ion, main fragment ions in MS2, λmax, retention time, and identified carotenoid compounds. The LC separated carotenoid values from four genotypes (VA16, VA14, VA11, and VA6) were compared with standard carotenoid compounds masses through the corresponding peaks of the compounds. A total of five carotenoid compounds were identified in A. tricolor. Among them, four compounds were xanthophylls (Lutein, violaxanthin, neoxanthin, and zeaxanthin) and one compound was pro-vitamin A (beta-carotene). Figure 3 represents the identified carotenoid profiles along with total xanthophylls and total carotenoids of four selected A. tricolor leaves, respectively.
Within xanthophylls, lutein was identified as the most abundant carotenoids followed by violaxanthin and neoxanthin while the content of zeaxanthin was very low in A. tricolor genotypes (Fig. 3). Except for violaxanthin, we obtained much greater lutein, neoxanthin, zeaxanthin, total xanthophylls, beta-carotene, and total carotenoid contents in A. tricolor genotype compared to the contents of A. gangeticus genotype of Raju et al.36. There were no prominent variations in zeaxanthin, lutein, violaxanthin, neoxanthin, total xanthophylls, beta-carotene, and total carotenoids content of the studied genotypes, albeit those showed the significant differences among studied genotypes. Zeaxanthin, lutein, violaxanthin, neoxanthin, total xanthophylls, beta-carotene, and total carotenoids ranged from 3.28 to 4.84, 18.34 to 25.12, 11.05 to 15.51, 7.67 to 10.77, 40.34 to 56.24, 21.43 to 31.01, and 65.85 to 87.25 mg 100 g−1, respectively (Fig. 3). The highest zeaxanthin (4.84 mg 100 g−1), lutein (18.34 mg 100 g−1), violaxanthin (15.51 mg 100 g−1), neoxanthin (10.77 mg 100 g−1), total xanthophylls (56.24 mg 100 g−1), and total carotenoids (87.25 mg 100 g−1) were obtained from the genotype VA11 followed by the genotype VA16. In contrast, the genotype VA14 showed the lowest zeaxanthin (3.28 mg 100 g−1), lutein (18.34 mg 100 g−1), violaxanthin (11.05 mg 100 g−1), neoxanthin (7.67 mg 100 g−1), total xanthophylls (40.34 mg 100 g−1), and total carotenoids (65.85 mg 100 g−1). The genotype VA11 exhibited the highest beta-carotene (31.01 mg 100 g−1), followed by the genotype VA16. In contrast, the genotype VA6 showed the lowest beta-carotene (21.43 mg 100 g−1) (Fig. 3).
Percentage of zeaxanthin, lutein, violaxanthin, neoxanthin, total xanthophylls, beta-carotene to total carotenoids ranged from 4.98 to 6.15, 27.85 to 29.27, 16.78 to 19. 79, 11.65 to 13.53, 61.26 to 68.75, and 31.25 to 38.74 mg 100 g−1 FW, respectively (Fig. 4). The genotype VA6 demonstrated the highest percentage of zeaxanthin, lutein, violaxanthin, neoxanthin, total xanthophylls to total carotenoids followed by VA16 and VA11 albeit this genotype exhibited the lowest beta-carotene. In contrast, VA14 showed the lowest percentage of zeaxanthin, lutein, violaxanthin, neoxanthin, total xanthophylls albeit this genotype exhibited the highest beta-carotene. It indicated from the results that the percentage of total xanthophylls to carotenoids along with its components had the opposite relationship with the percentage of beta-carotene content to carotenoids of A. tricolor genotypes. (Fig. 4).
In this study, we found remarkable pigments profile such as betacyanins, betalains, betaxanthins, and carotenoid profiles such as lutein, violaxanthin, neoxanthin, zeaxanthin, total xanthophylls, beta-carotene in A. tricolor genotypes. The results of total carotenoids of our study corroborated with the results of Khanam and Oba37 where they observed higher carotenoids in the red amaranth genotype compared to green amaranth. The genotype VA11 and VA16 contained higher lutein, violaxanthin, neoxanthin, zeaxanthin, total xanthophylls, beta-carotene, and total carotenoids compared to the genotype VA14 and VA6. Hence, the carotenoid profiles of A. tricolor genotype could be an important parameter for consumers, playing a crucial role in detoxification of ROS in the human body and preventing antiaging and many degenerative human diseases17,29. Our result showed that the A. tricolor genotype is an excellent source of lutein, violaxanthin, neoxanthin, zeaxanthin, total xanthophylls, beta-carotene, and total carotenoids among leafy vegetables that has important free radical-scavenging activity19. A. tricolor genotypes VA11 and VA16 had high carotenoid profiles, such as zeaxanthin, lutein, violaxanthin, neoxanthin, total xanthophylls, beta-carotene, and total carotenoids content. The genotypes VA11 and VA16 might be utilized as high-yielding varieties. These genotypes containing abundant carotenoid profiles could be used for extracting colorful juice as drinks, preservatives, and colorant of food products. It revealed from the current investigation that VA11 and VA16 have abundant carotenoids that explore new insight for comprehensive pharmacological study.
Betaxanthins, betalains and radical scavenging capacity
Betaxanthins, betalains, and antioxidant capacity (AC) varied significantly among the studied A. tricolor leafy vegetable genotypes (Fig. 5). Betaxanthins exhibited much prominent variations regarding genotypes. Betaxanthins varied from 26.87 mg 100 g−1 in VA11 to 55.21 mg 100 g−1 in VA14. Similarly, betalains varied from 46.47 mg 100 g−1 in VA11 to 101.73 mg 100 g−1 in VA14. Antioxidant capacity (DPPH) varied from 12.27 µg g−1 (VA6) to 29.38 µg g−1 (VA14). The maximum antioxidant capacity (DPPH) was noted in VA14 followed by VA16 and VA11. Conversely, VA6 demonstrated the minimum antioxidant capacity (DPPH). Antioxidant capacity (ABTS+) varied from 26.69 µg g−1 to 63.79 µg g−1. A. tricolor accession VA14 demonstrated the maximum antioxidant capacity (ABTS+) followed by VA16. On the contrary, antioxidant capacity (ABTS+) was the minimum in VA6. Our results were corroborated with the findings of Khanam and Oba37. They reported greater betaxanthins, betalains, and antioxidant capacity in the red color amaranth compared to green color amaranth. The A. tricolor genotype VA14 and VA16 contained higher betaxanthins, betalains, and antioxidant capacity compared to the genotype VA6 and VA11. Hence, these antioxidant constituents of A. tricolor genotype could be an important parameter for consumers, playing a crucial role in quenching of ROS, curing many degenerative diseases and preventing anti-aging and17,29. Our result revealed that A. tricolor contained ample betaxanthins, betalains, and antioxidant potentials among leafy vegetables that have important free radical-scavenging activity19.
Betaxanthins, betalins, and antioxidant capacity in four selected A. tricolor leafy vegetables, betaxanthins (mg 100 g−1 FW), betalains (mg 100 g−1 FW), AC (DPPH) = Antioxidant capacity (DPPH) (TEAC µg g−1 DW), AC (ABTS+) = Antioxidant capacity (ABTS+) (TEAC µg g−1 DW), different letters in the blue and green bars are differed significantly by Duncan Multiple Range Test ((P < 0.01), (n = 3).
In this study, we found remarkable color attributes, betacyanin profiles such as amaranthine, iso-amaranthine, betanin, iso-betanin, carotenoid profiles, such as zeaxanthin, lutein, violaxanthin, neoxanthin, total xanthophylls, beta-carotene, total carotenoids, betaxanthins, betalains, and antioxidant capacity in A. tricolor genotypes. Our results were corroborated with the findings of Khanam and Oba37. They reported greater antioxidant capacity, betacyanins, betaxanthins, betalains, total carotenoids in the red color amaranth compared to green color amaranth. Betacyanin, total carotenoids, antioxidant capacity (ABTS+) and antioxidant capacity (DPPH) of current investigation were corroborated with the findings of A. tricolor by Khanam et al.38. We found 2 to 3 time’s higher β-carotene in red color amaranth compared β-carotene of Raju et al.36 in A. gangeticus. The β-carotene of the leaves of red color amaranths were 2 to 3 times and green color amaranths were twice than the β-carotene of A. caudatus leaves29. Li et al.39 in A. hypochondriacus leaves noticed the highest quenching capacity of radicals (ORAC and FRAP methods) in comparison to the leaves of A. caudatus. They also noted the maximum quenching capacity of radicals (FRAP) in leaves in comparison to seed, stalks, sprouts, and flowers. The genotypes VA14 and VA16 had high color attributes, betacyanin profiles, betaxanthins, betalains, and antioxidant capacity. The genotypes VA14 and VA16 containing high antioxidant profiles and high quenching capacity of radicals might be utilized as high-yielding varieties. It revealed from the current investigation that quenching capacity of radicals have high possibility to feed the community deficient in antioxidants. These genotypes also could be used for extracting colorful juice as drinks, preservatives, and colorant of food products.
The correlation coefficient study
Correlation of betacyanins, betaxanthins, betalains, and antioxidant capacity of A. tricolor leafy vegetables are shown in Table 3. Total betacyanins and betaxanthins, betalains had highly significant positive associations among themselves, with total carotenoids, antioxidant capacity (DPPH and ABTS+). It revealed that total betacyanins betaxanthins, and betalains, exhibited strong antioxidant activity. Total xanthophylls, total carotenoids, and β-carotene had positive and significant interrelationships with β-carotene, antioxidant capacity (ABTS+ and DPPH), and total carotenoids which revealed high quenching capacity of radicals of major carotenoids. The findings of current investigation were corroborated with the results of our earlier study of drought and salt-stressed A. tricolor24.
In conclusions, we identified betacyanin profiles, such as amaranthine, iso-amaranthine, betanin, iso-betanin, carotenoid profiles, such as zeaxanthin, lutein, violaxanthin, neoxanthin, total xanthophylls, beta-carotene, and total carotenoids, betaxanthins, betalains, and antioxidant potentiality (DPPH and ABTS+) in A. tricolor genotypes. A. tricolor genotype VA14 and VA16 had abundant betacyanin components, betaxanthins, betalains and antioxidant potentiality might be utilized as high-yielding varieties. VA11 and VA16 had abundant carotenoids components might be utilized as high-yielding varieties. It revealed from the correlation study that all phytopigments of A. tricolor exhibited good quenching capacity of radicals. It also revealed from the current investigation that these accessions had ample antioxidants to quench ROS and had high possibility to study pharmacologically in details. These genotypes also could be used for extracting colorful juice as drinks, preservatives, and colorant of food products. The data on color attributes, betacyanins, carotenoids, betaxanthins, betalains and antioxidant potentiality obtained in the present study could contribute to the scientists for scientific evaluation of these active compounds in A. tricolor.
Methods
Experimental materials
We selected four high yields and antioxidant potential genotypes from 43 genotypes. It is the first report on color attributes, betacyanin and carotenoid profiles, and antioxidant potentials in A. tricolor.
Design and layout
We executed the experiment in three replicates following a completely randomized block design (RCBD) at Bangabandhu Sheikh Mujibur Rahman Agricultural University. Each genotype was grown in 1 m2 experimental plot following 20 cm and 5 cm distance between rows and plants, respectively.
Intercultural practices
Recommended fertilizer doses, compost, and adequate cultural management were practiced40. During final land preparation, total compost (10 ton/ha), triple superphosphate, murate potash and gypsum fertilizers were applied. Whereas, urea was applied in three equal installments i. e., during final land preparation, 10 days after sowing of seed (DAS) and 20 DAS. The urea, triple superphosphate, murate potash and gypsum fertilizers were applied at the rate of 200, 100, 150, and 30 kg/ha, respectively. As the compost was made from rotten cow dung and rice straw, it released the nutrient evenly throughout the experiment period. It also helped to hold the nutrients of chemical fertilizers and released these nutrients evenly. For maintaining the exact spacing of plants in a row, proper thinning was executed. Weeds of experimental plots were regularly removed through proper weeding and hoeing. We provide regular irrigation in the experimental plots for maintaining the proper growth of vegetable amaranth. We collected the leaf samples at 30 days old plant.
Solvent and reagents
Solvent: acetone, hexane, and methanol. Reagents: acetonitrile (HPLC grade), dichloromethane, and acetic acid, betanin, beta-carotene (98%), lutein (99%), neoxanthin (95%), violaxanthin (98%), zeaxanthin (98%), phenolic acids, and flavonoids, 2, 2-dipyridyl, ABTS+, DPPH (2, 2-diphenyl1-picrylhydrazyl), ammonium acetate, petroleum ether, sodium chloride, sodium sulphate, Trolox (6-hydroxy-2, 5, 7, 8-tetramethyl-chroman-2-carboxylic acid), potassium persulfate, sodium carbonate, aluminum chloride Folin-Ciocalteu reagent, hexahydrate, potassium acetate, quercetin, and salicylic acid.
Determination of color attributes
We measured the color attributes C*, L*, b*, and a* using a TES-135A Plus color meter (Taiwan) in fifteen replicates. Positive b value (+b*) represents yellowness, while negative b value (−b*) indicates blueness. Positive a value (+a*) indicates the magnitude of redness, while negative a value (−a*) indicates magnitude of greenness. L* represents lightness and C* value represents magnitude of leaf color designated as chroma. The chroma value was estimated following the formula, Chroma C* = (a2 + b2)1/2.
Extraction of samples for LC-MS and HPLC analysis
Ten ml methanol (80%) containing acetic acid (1%) was added in leaf sample (1 g). The mixture was thoroughly homogenized and transferred to a 50 ml tightly capped test tube. The test tubes were placed in a Scientific Industries Inc. shaker (USA) for fifteen h at 400 rpm. A MILLEX®-HV syringe filter, USA (0.45 µm) was utilized to filter the mixture. It was centrifuged for fifteen min at 10,000 × g. Betacyanin components were detected from this filtrate. Betacyanin analysis in the samples could interfere through precipitation of methanol with the proteins and other insoluble substances in the samples. A Polymeric Reversed-Phase cartridges (33 µ) (StrataTM-X, USA) was utilized to purify betacyanin. The samples were extracted in triplicates.
Betacyanin analysis through HPLC
The method previously described by Stintzing et al.33 was followed to determine betacyanin components in A. tricolor leaf sample using HPLC. The high performance liquid chromatograph Shimadzu SCL10Avp, Kyoto, Japan was furnished with a degasser (DGU-14A), an LC-10Avp binary pumps, and a Shimadzu SPD-10Avp detector (UV–vis). A STR ODS-II, 150 × 4.6 mm I.D., CTO-10AC column (Shinwa Chemical Industries, Ltd., Kyoto, Japan) was utilized to detected the betacyanin components. The binary mobile phase was pumped with solvent A (acetic acid 6% v/v) in water and solvent B (acetonitrile) at the flow rate of 1 ml/min for seventy min. High performance liquid chromatograph was run using a gradient program containing acetonitrile 0–15% for forty five min, 15–30% for fifteen min, 30–50% for five min and 50–100% for five min. Exactly 35 °C temperature was continued in the column with 10 μl injection volume. The betacyanin was simultaneous monitored by setting the detector at 538 nm. The betacyanin components were detected through comparing UV–vis spectra and retention time with their respective standard compounds. We confirmed the betacyanin components through the method of mass spectrometry. The betacyanin components were detected in triplicates. The betacyanin components were estimated as mg 100 g−1 of fresh weight. The LC-MS analysis was done using a mass spectrometer (Tokyo, Japan) equipped with HPLC (Agilent 1100 Series) and a detector coupled with on-line ElectroSpray Ionization (ESI) source. The LC-MS analysis was done in negative ion mode with the needle voltage at −2000 V and the range of the column elutes was m/z 0–1000. Extract constituents were identified by LC-MS-ESI analysis.
Quantification of betacyanin components
Calibration curves of the respective standards were used to quantify individual phenolic compounds. The betanin was dissolved in methanol (80%) to prepare the stock solutions of 10, 20, 40, 60, 80, and 100 mg/ml. A standard curves were made from these solutions to quantify the individual betacyanin components with external standards. The retention times, co-chromatography, and UV spectral characteristics of samples spiked with available standards (commercially) to identify and match the betacyanin components. Betanin standard was used to prepare standard curves based on the equimolecular conversion for estimating amaranthine and iso-amaranthine in the different samples.
Sample preparation for extraction of carotenoids
The fresh leaves were thoroughly washed and dried using blotting papers. All precautions were taken to protect photo-isomerization and photo-oxidation and prevent any losses of carotenoids. Sampling was performed at 20 °C with subdued lighting. We ground the dried leaves using a mechanical blender. The powdered leaves were kept in a bag (self-sealing) containing aluminum foil inside the bag at below −20 °C.
Extraction of carotenoids
We extracted carotenoids following the procedure of Lakshminarayana et al.41. We extracted carotenoids with acetone (ice-cold) until the powdered leaves became colorless. To prevent oxidation of carotenoid rapid extraction in acetone (ice-cold) was performed. Petroleum ether (100 ml) and 25% aqueous sodium chloride (w/v) (100 ml) were added to leaf extract (50 ml) in a funnel. After mixing, we separated the upper layer. We repeated it for three times (final volume: 250 ml). We dried the extract over 20 g anhydrous sodium sulphate and filtered through a filter paper (Whatman No.1). A rotary evaporator was used to evaporate the filtrate at 35 °C to dryness. We re-dissolved the dried filtrate in hexane (known volume) and an aliquot (100 µl) for analyzing it using high performance liquid chromatograph. We carried out homogenization, extraction, and sample handling under dim yellow light at 4 °C to reduce photo-oxidation and photo-isomerization of carotenoids.
HPLC analysis
We estimated the carotenoid profiles in A. tricolor leaf sample using high performance liquid chromatograph (Shimadzu SCL10Avp, Kyoto, Japan)41. A variable detector, binary pumps, and a degasser were equipped with the HPLC system. Briefly, the carotenoids were separated on a column (STR ODS-II, 150 × 4.6 mm I.D., Kyoto, Japan). The carotenoids were separated using methanol:acetonitrile:dichloromethane with a ratio of 20:60:20 (v/v/v) containing ammonium acetate (0.1%) as a mobile phase. For HPLC analysis, leaves (20 µl) were injected under the isocratic condition at a flow rate of 1 ml/min. The detector was set at 450 nm. The SPD-10AVP detector were equipped with Shimadzu model LC-10Avp series to confirm the peak of carotenoids in comparison with their retention time of standard chromatograms. While the characteristic spectrum record with a PDA detector was taken to confirm the λmax values of these compounds. We quantified the carotenoid profiles estimating the area of peak of corresponding standards.
Betaxanthins content measurement
The leaves of A. tricolor leafy vegetables were extracted in methyl alcohol (80%) containing ascorbate (50 mM) to measure betaxanthins following the method of Sarker and Oba42. A Hitachi, U-1800 spectrophotometer (Tokyo, Japan) was utilized to read the absorbance at 475 nm for betaxanthins. We calculated the betaxanthins as miligrams per gram of fresh weight equivalent to indicaxanthin.
Antioxidant capacity assay
Thirty days old A. tricolor leaves were harvested. Antioxidant capacity assay, the leaves were dried in the air in a shade. Aqueous methanol (40 ml, 90%) was utilized to extract grounded dried leaves (1 g) from each cultivar in a capped bottle (100 ml). A Thomastant T-N22S (Japan) shaking water bath was utilized to extract leaf samples for 1 h. A MILLEX®-HV syringe filter (0.45 µm) (USA) was used to filter the homogenized mixture. After centrifugation for fifteen min at 10,000 × g, the antioxidant capacity was estimated from the extract.
Diphenyl-picrylhydrazyl (DPPH) radical degradation method43,44 was used to estimate the antioxidant activity. We added DPPH solution (250 µM, 1 ml) to extract (10 µl) in a test tube (in triplicate). Finally we added 4 ml distilled water the extract and kept in the dark (30 min). A spectrophotometer (Hitachi, Tokyo, Japan) was used to take the absorbance at 517 nm. The antioxidant activity was also estimated following ABTS+ assay45,46. To prepare two stock solutions separately ABTS+ solution of 7.4 mM and potassium persulfate of 2.6 mM were used. We mixed both solutions in equal proportion to prepare the working solution at room temperature. The working solution was allowed to react in the dark for 12 h. One hundred fifty μl extract was added to 2.85 ml of ABTS+ solution and allowed to react in the dark for 2 h. For the preparation of the solution, one ml of ABTS+ solution was mixed with sixty ml of methanol. A spectrophotometer (Hitachi, Tokyo, Japan) was utilized to take the absorbance at 734 nm. The inhibition of ABTS+ (%) and DPPH (%) with control were utilized to calculate quenching capacity of radicals using the equation:
Where, A. sample is the absorbance of the test compound and A. blank is the absorbance of the control reaction [10 µl methanol (DPPH), 150 μl methanol (ABTS+) instead of extract]. The quenching capacity of radicals were calculated as μg g−1 of dry weight equivalent to Trolox.
Statistical analysis
Statistix 8 software was used to analyze the data for analysis of variance (ANOVA)47. Duncan’s Multiple Range Test (DMRT) at a 1% level of probability was used to compare the means. The results were reported as the mean ± SD of three separate replicates.
Ethical statement
The lab and field experiments in this study were carried out as per guidelines and recommendations of “Biosafety Guidelines of Bangladesh” published by the Ministry of Environment and Forest, Government of the People’s Republic of Bangladesh (2005).
Data availability
Data used in this manuscript will be available to the public.
References
Sarker, U., Islam, M. T., Rabbani, M. G. & Oba, S. Genotypic variability for nutrient, antioxidant, yield and yield contributing traits in vegetable amaranth. J. Food Agri. Environ. 12, 168–174 (2014).
Sarker, U., Islam, M. T., Rabbani, M. G. & Oba, S. Variability, heritability and genetic association in vegetable amaranth (A. tricolor). Span. J. Agric. Res. 13, 1–8, https://doi.org/10.5424/sjar/2015132-6843 (2015a).
Sarker, U., Islam, M. T., Rabbani, M. G. & Oba, S. Genotype variability in composition of antioxidant vitamins and minerals in vegetable amaranth. Genetika. 47, 85–96 (2015b).
Sarker, U., Islam, M. T., Rabbani, M. G. & Oba, S. Genetic variation and interrelationship among antioxidant, quality and agronomic traits in vegetable amaranth. Turk. J. Agric. For. 40, 526–535 (2016).
Sarker, U., Islam, M. T., Rabbani, M. G. & Oba, S. Genotypic diversity in vegetable amaranth for antioxidant, nutrient and agronomic traits. Indian J. Genet. Plant Breed. 77, 173–176 (2017).
Chakrabarty, T., Sarker, U., Hasan, M. & Rahman, M. M. Variability in mineral compositions, yield and yield contributing traits of stem amaranth (Amaranthus lividus). Genetika. 50, 995–1010 (2018).
Sarker, U., Islam, M. T., Rabbani, M. G. & Oba, S. Variability in total antioxidant capacity, antioxidant leaf pigments and foliage yield of vegetable amaranth. J. Integrative Agric. 17, 1145–1153 (2018a).
Sarker, U., Islam, M. T., Rabbani, M. G. & Oba, S. Antioxidant leaf pigments and variability in vegetable amaranth. Genetika. 50, 209–220 (2018b).
Sarker, U., Islam, M. T., Rabbani, M. G. & Oba, S. Phenotypic divergence in vegetable amaranth for total antioxidant capacity, antioxidant profile, dietary fiber, nutritional and agronomic traits. Acta Agric. Scandinavica Section B- Soil and Plant Sci. 68, 67–76 (2018c).
Sarker, U. & Oba, S. Nutraceuticals, antioxidant pigments, and phytochemicals in the leaves of Amaranthus spinosus and Amaranthus viridis weedy species. Sci. Rep. 9, 20413, https://doi.org/10.1038/s41598-019-50977-5 (2019a).
Sarker, U., Oba, S. & Daramy, M. A. Nutrients, minerals, antioxidant pigments and phytochemicals, and antioxidant capacity of the leaves of stem amaranth. Sci. Rep. 10, 3892, https://doi.org/10.1038/s41598-020-60252-7 (2020).
Sarker, U. & Oba, S. Nutrients, minerals, pigments, phytochemical, and radical scavenging activity in Amaranthus blitum leafy vegetable. Sci. Rep. 10, 3868, https://doi.org/10.1038/s41598-020-59848-w (2020a).
Sarker, U., Hossain, M. M. & Oba, S. Nutritional and antioxidant components and antioxidant capacity in green morph Amaranthus leafy vegetable. Sci. Rep. 10, 1336, https://doi.org/10.1038/s41598-020-57687-3 (2020).
Sarker, U. & Oba, S. Protein, dietary fiber, minerals, antioxidant pigments and phytochemicals, and antioxidant activity in selected red morph Amaranthus leafy vegetable. PLoS One 14, 0222517, https://doi.org/10.1371/journal.pone.0222517 (2019b).
Dusgupta, N. & De, B. Antioxidant activity of some leafy vegetables of India: A comparative study. Food Chem. 101, 471–474 (2007).
Isabelle, M. et al. Antioxidant activity and profiles of common fruits in Singapore. Food Chem. 123, 77–84 (2010).
Repo-Carrasco-Valencia, R., Hellstrom, J. K., Philava, J. M. & Mattila, P. H. Flavonoids and other phenolic compounds in Andean indigenous grains: Quinoa (Chenopodium quinoa), kaniwa (Chenopodium pallidicaule) and kiwicha (Amaranthus caudatus). Food Chem. 120, 128–133 (2010).
Steffensen, S. K. et al. Variations in the polyphenol content of seeds of field grown Amaranthus genotypes. Food Chem. 129, 131–138 (2011).
Cai, Y., Sun, M. & Corke, H. Antioxidant activity of betalains from plants of the Amaranthaceae. J. Agric. Food Chem. 51, 2288–2294 (2003).
Stintzing, F. C. & Carle, R. Betalains-emerging prospects for food scientists. Trends Food Sci. Technol. 18, 514–525 (2007).
Sarker, U. & Oba, S. Catalase, superoxide dismutase and ascorbate-glutathione cycle enzymes confer drought tolerance of A. tricolor. Sci. Rep. 8, 16496, https://doi.org/10.1038/s41598-018-34944-0 (2018a).
Sarker, U. & Oba, S. Drought Stress Effects on Growth, ROS Markers, Compatible Solutes, Phenolics, Flavonoids and Antioxidant Activity in A. tricolor. Appl. Biochem. Biotechnol. 186, 999–1016, https://doi.org/10.1007/s12010-018-2784-5 (2018b).
Sarker, U. & Oba, S. Drought stress enhances nutritional and bioactive compounds, phenolic acids and antioxidant capacity of Amaranthus leafy vegetable. BMC Plant Biol. 18, 258, https://doi.org/10.1186/s12870-018-1484-1 (2018c).
Sarker, U. & Oba, S. Response of nutrients, minerals, antioxidant leaf pigments, vitamins, polyphenol, flavonoid and antioxidant activity in selected vegetable amaranth under four soil water content. Food Chem. 252, 72–83 (2018d).
Sarker, U., Islam, M. T. & Oba, S. Salinity stress accelerates nutrients, dietary fiber, minerals, phytochemicals and antioxidant activity in Amaranthus tricolor leaves. PLOS One. 13, e0206388, https://doi.org/10.1371/journal.pone.0206388 (2018).
Sarker, U. & Oba, S. Augmentation of leaf color parameters, pigments, vitamins, phenolic acids, flavonoids and antioxidant activity in selected A. tricolor under salinity stress. Sci. Rep. 8, 12349, https://doi.org/10.1038/s41598-018-30897-6 (2018e).
Sarker, U. & Oba, S. Salinity stress enhances color parameters, bioactive leaf pigments, vitamins, polyphenols, flavonoids and antioxidant activity in selected Amaranthus leafy vegetables. J. Sci. Food Agric. 99, 2275–2284, https://doi.org/10.1002/jsfa.9423 (2019c).
Sani, H. A., Rahmat, A., Ismail, M., Rosli, R. & Endrini, S. Potential anticancer effect of red spinach (Amaranthus gangeticus) extract. Asia Pacific J. Clinical Nutri. 13, 396–400 (2004).
Venskutonis, P. R. & Kraujalis, P. Nutritional components of amaranth seeds and vegetables: a review on composition, properties, and uses. Comp. Rev. Food Sci. Food Saf. 12, 381–412 (2013).
Colonna, E., Rouphael, Y., Barbieri, G., De & Pascale, S. Nutritional quality of ten leafy vegetables harvested at two light intensities. Food Chem. 199, 702–710 (2016).
Sarker, U. & Oba, S. Antioxidant constituents of three selected red and green color Amaranthus leafy vegetable. Sci. Rep. 9, 18233, https://doi.org/10.1038/s41598-019-52033-8 (2019d).
Cai, Y., Sun, M. & Corke, H. Characterization and application of betalain pigments from plants of the Amaranthaceae. Trends Food Sci. Technol. 16, 370–376 (2005).
Stintzing, F. C. et al. Betacyanins and phenolic compounds from Amaranthus spinosus L. and Boerhavia erecta L. Plants Food Hum. Nutri. 59, 1–8 (2004).
Cai, Y., Sun, M., Wu, H., Huang, R. & Corke, H. Characterization and quantification of betacyanin pigments from diverse Amaranthus species. J. Agric. Food Chem. 46, 2063–2070 (1998).
Cai, Y., Sun, M. & Corke, H. Identification and distribution of simple and acylated betacyanins in the Amaranthaceae. J. Agric. Food Chem. 49, 1971–1978 (2001).
Raju, M., Varakumar, S., Lakshminarayana, R., Krishnakantha, P. T. & Baskaran, V. Carotenoid composition and vitamin A activity of medicinally important green leafy vegetables. Food Chem. 101, 1598–1605 (2007).
Khanam, U. K. S. & Oba, S. Bioactive substances in leaves of two amaranth species, Amaranthus lividus and A. hypochondriacus. Canadian J. Plant Sci. 93, 47–58 (2013).
Khanam, U. K. S., Oba, S., Yanase, E. & Murakami, Y. Phenolic acids, flavonoids and total antioxidant capacity of selected leafy vegetables. J. Functional Foods. 4, 979–987 (2012).
Li, H. et al. Characterization of phenolics, betacyanins and antioxidant activities of the seed, leaf, sprout, flower and stalk extracts of three Amaranthus species. J. Food Compos. Anal. 37, 75–81 (2015).
Rashad, M. M. I. & Sarker, U. Genetic variations in yield and yield contributing traits of green amaranth. Genetika 52, 391–404 (2020).
Lakshminarayana, R., Raju, M., Krishnakantha, T. P. & Baskaran, V. Determination of major carotenoids in few Indian leafy vegetables by HPLC. J. Agric. Food Chem. 53, 2838–2842 (2005).
Sarker, U. & Oba, S. Nutritional and bioactive constituents and scavenging capacity of radicals in Amaranthus hypochondriacus. Sci. Rep. https://doi.org/10.1038/s41598-020-71714-3 (accepted) (2020b).
Sarker, U. & Oba, S. Phenolic profiles and antioxidant activities in selected drought-tolerant leafy vegetable amaranth. Sci. Rep. https://doi.org/10.1038/s41598-020-71727-y (accepted) (2020c).
Sarker, U. & Oba, S. The response of salinity stress-induced A. tricolor to growth, anatomy, physiology, non-enzymatic and enzymatic antioxidants. Front Plant Sci. https://doi.org/10.3389/fpls.2020.559876 (accepted) (2020d).
Sarker, U. & Oba, S. Polyphenol and flavonoid profiles and radical scavenging activity in selected leafy vegetable Amaranthus gangeticus. BMC Plant Biol. (accepted) (2020e).
Hasan-Ud-Daula, M. & Sarker, U. Variability, heritability, character association, and path coefficient analysis in advanced breeding lines of rice (Oryza sativa L.). Genetika. 52, 711–726 (2020).
Sarker, U., Hossain, M. N., Iqbal, M. A. & Oba, S. Bioactive components and radical scavenging activity in selected advance lines of salt-tolerant vegetable amaranth. Front Nutr. (accepted) (2020f).
Author information
Authors and Affiliations
Contributions
U.S. initiated the research work and conceived the study; U.S. performed the experiments; U.S. performed biochemical analysis and statistical analysis; U.S. drafted, edited, interpreted data and prepared the manuscript; S.O. edited the manuscript, provided valuable suggestions during the experiment.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sarker, U., Oba, S. Leaf pigmentation, its profiles and radical scavenging activity in selected Amaranthus tricolor leafy vegetables. Sci Rep 10, 18617 (2020). https://doi.org/10.1038/s41598-020-66376-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-66376-0
This article is cited by
-
Bioactive polyphenolic compounds and antioxidant potentials of two leafy vegetables in Bangladesh: the Momordica charantia and the Ipomoea aquatica
Food Production, Processing and Nutrition (2024)
-
Manure-biochar compost mitigates the soil salinity stress in tomato plants by modulating the osmoregulatory mechanism, photosynthetic pigments, and ionic homeostasis
Scientific Reports (2024)
-
Amaranthus crop for food security and sustainable food systems
Planta (2024)
-
Color attributes, betacyanin, and carotenoid profiles, bioactive components, and radical quenching capacity in selected Amaranthus gangeticus leafy vegetables
Scientific Reports (2021)
-
Nutraceuticals, phytochemicals, and radical quenching ability of selected drought-tolerant advance lines of vegetable amaranth
BMC Plant Biology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.