Abstract
The aim was to find out the impact of stress, caused by agrotechnological tools on photosynthetic behaviour of apple trees. The apple tree (Malus domestica Borkh.) cultivar Rubin was grafted on dwarfing rootstocks P60, planted in single rows spaced 1.25 m apart with 3.5 m between rows. In contrast to plant senescing reflectance index and nitrogen balance index, the photochemical reflectance index was significantly higher in 2018 compared with 2017. Such differences might be caused by drought stress on the summer and fast recovery before harvest time when measurements were made. The movement of nutrients and water disrupted by trunk incision had significantly negative effect on reflectance indices regardless on the year. Mechanical pruning with trunk incision and calcium-prohexadione lead to decreased dry to fresh weight ratio by 10–12% in first year of treatment. Mechanical pruning had significantly negative impact on photosynthetic rate, compared to pruning by super spindle it decreased 47%. Strong positive correlation between PRI and NBI 0,89–0,94, and strong negative correlations between PRI, NBI and PSRI −0.88 – (−0.91) were determined.
Similar content being viewed by others
Introduction
Not only environmental factors but also plant canopy ant light penetration into canopy strongly influence photosynthetic productivity and plants’ growth. Photosynthesis productivity depends on light, water, CO2, nutrients and even plant structure and architecture1 Light is important for all physiological processes in a plant. From 100% sun light only 40% is typical light interception by orchard systems and 15% (out of 40%) is limited by orchard design and leaf area2. During photosynthesis, solar energy binds to dry matter, so the photosynthetic behavior of the plant can be evaluated. Increasing photosynthesis goes to increase in biomass. Optimal plant photosynthesis system work is the main part of the biomass quantity1,3.
Non-destructive measurement methods easily measure variable optical properties without affecting the leaf at various stages of its development. In one measurement several functionally relevant leaf parameters is done without interruption4,5. Nitrogen (N) is one of the most important components of photosynthetic productivity is the also, it is a part of chlorophyll and it is one of the most important elements for biomass production6. The chlorophyll index (Chl) and the nitrogen balance index (NBI) measure the relative amount of chlorophyll and nitrogen at the same point on the leaf in the same moment and it may convey N dynamics in apple trees systems7,8. Plant synthesizes proteins and favors primary metabolism(nitrogen containing molecules) containing chlorophyll and just a few flavonols is shown by higher nitrogen balance index. NBI status of leaves has a relationship to light availability and light limitation as shown in literature2,5,9.
The Photochemical Reflectance Index (PRI), which is based on leaf reflectance at 531 nm, is suitable for tracking variations in photosynthetic activity from leaf10. PRI is commonly used as an appropriate indicator of abiotic factors that limit photosynthesis11. PRI via the xanthophyll cycle is related with photosystem II (PSII) and it is using as an alternative for light use efficiency. Photochemical reflectance index was applied as an active probe of pigment conversion, it can reflect almost all changes in the pigment content of the xanthophyll cycle12. Other authors have investigated that changes in pigment content and ratio due to leaf growth, aging, or chronic stress play an important role along with xanthophyll pigment epoxidation in the PRI signal. The photochemical reflectance index responds to the chlorophyll / carotenoid ratio in leaves due to species, age, and environmental conditions5,12,13. While PRI characterizes the photosynthetic efficiency, the plant senescing reflectance index (PSRI) is sensitive to the changes of carotenoids and chlorophyll ratio and it was used as a quantitative measure of leaf senescence. They are both commonly used to describe the changing physiological state of vegetation13,14,15. Reflectance spectra in the visible and near infra‐red range of the spectrum, acquired for maple (Acer platanoides L.), chestnut (Aesculus hippocastanum L.), potato (Solanum tuberosum L.), coleus (Coleus blumei Benth.), leaves and lemon (Citrus limon L.) and apple (Malus domestica Borkh.) were studied and it was found that leaf senescence effect is depending on pigment composition14.
Photosynthesis activity is associated not only with the yield of individual leaves, but also with the optimal use of light, considering the penetration of light through the canopy16. Alarcon and Sassenrath17 showed cotton leaf photosynthesis efficient dependence from shapes and canopy architecture. This shows that the effectiveness of photosynthesis can be controlled through the shape of the canopy and the best results were based on the high number of small-to-medium sized leaves. Like all plants, the ability of apple trees to sustain higher photosynthetic rates (PN) depends on the whole tree canopy and its regulation16,17,18. Also, Zhang et al.19 showed that photosynthesis consider from temperature, CO2 and especially light intensity and chlorophyll content. Moreover, photosynthetic rates are sensitive to various stresses like salt-alkaline, salt, water deficiency, shading and other20. One of the reasons for the PN reduction is the stress of water drought and Ohashi et al. measured the decrease by up to 34%21. The inhibition of photosynthetic rate under water stress may be attributed not only to stomatal closure but also on several biochemical and photochemical processes22,23.
‘Rubin’ is large fruit variety. De Salvador et al.24 showed that heavy crop load decreased fruit size and weight. Denser canopy also has negative impact on fruit mass and yield25. Although calcium-prohexadione is using for smaller terminal growth it also has the impact for fruit weight. Usually fruit weight decreases by use of calcium-prohexadion. As Privé et al.26 showen Calcium-prohexadion can increase light penetration into the inner canopy of fully-grown apple trees and thus improve flower bud formation and expected yield.
Water and nutrients movement are disturbed by trunk girdling as well as by trunk incision. Trunk girdling and ringing significantly increase yield but most of the studies shown that photosynthetic behavior decreases in the same time as yields increase27,28,29. Trunk girdling not only increase fruit yield, but also can increase the influence of other agrotechnical tools using foliar spray or fertilization29,30,31,32.
Manual pruning of orchards requires a lot of resources and manpower to reduce the cost of gardening through mechanical pruning, trunk cutting, chemicals and more. Despite the amount of research on the relationship between the optical properties of apple leaves and the rate of photosynthesis in different apple agrotechnology tools, there is little. Thus, the aim was to elucidate the influence of stress caused by light penetration on canopy and agro-technological measures on photosynthetic behavior of apple trees.
Materials and methods
Plant material and growing conditions
A field experiment was carried out in an intensive orchard. The apple tree (Malus domestica Borkh.) cultivar Rubin was grafted on dwarfing rootstocks P60. Apple trees were planted in 2010 year in single rows spaced 1.25 m apart with 3.5 m between rows. Pest and disease management was carried out according to integrated plant protection practices and orchard was not irrigated. Soil conditions of the experimental orchard were following: clay loam, pH 7.3, humus 2.8%, P2O5 255 mg kg−1, K2O 230 mg kg−1. Three single trees were fully randomized. The non-destructive and biometric analysis were performed in harvest time in the end of September. The samples were taken from five leaves in two levels: at 1.0–1.2 m and 1.8–2.0 m above the ground. Five agrotechnological tools were used: (1) each year hand pruning forming slender spindle (control); (2) mechanical pruning (each year) with hand pruning every second year; (3) mechanical pruning (each year); (4) trunk incision using chain saw +mechanical pruning (each year); (5) mechanical pruning (each year) +spraying with calcium-prohexadione. Mechanical pruning goes with hand pruning ones in three years removing the oldest branches from the stem base. Reducing the movement of water and nutrients by cutting the trunk also suppresses tree growth, reduces the number of shoots and at the same time increases the lighting of the canopy at the top of the it. Mechanical pruning, on the other hand, only shortens the branches, without the use of manual pruning, increasing the number of large branches, which increases the darkening of the crown. Also, trees grow actively in the upper part of the canopy, densify and multiply branches, which cast heavy shadows on the entire tree. Using only mechanical pruning, the trees resemble a hedge. While in the case of manual pruning, the large branches are removed from the trunk, there are no large branches at the top of the canopy, and the canopy is easily lightened and does not darken the rest of the crown.
Photochemical reflectance index (PRI) and Plant senescing reflectance index (PSRI)
Spectral reflectance was measured using a leaf spectrometer (CID Bio-Science, USA) from 9 to 12 am. Reflection spectra obtained from the leaves were used to calculate photochemical reflectance index (PRI) and plant senescing reflectance index (PSRI). PRI shows changes in the xanthophyll cycle, using the following formula:
where R570 and R531 represent the leaf reflectance integrated over a 10 nm wavelength band centred on 570 and 531 nm, respectively
PSRI was proposed as being sensitive to the senescence phase of plant development and it’s calculated by formula:
where R678, R500 and R570 represent the leaf reflectance integrated over a 10 nm wavelength band centred on 678, 500 and 570 nm, respectively
Nitrogen balance index (NBI)
NBI was evaluated using no-destructive measurement of leaf chlorophyll and flavonoid content (related to Nitrogen/Carbon allocation) in the epidermis (Dualex ®4, USA).
Photosynthetic rate was determined at 9:00–12:00 am by using a LI-6400XT portable open flow gas exchange system (Li-COR Biosciences, Lincoln, USA). Reference air [CO2] (400 μmol mol−1), light intensity (1000 μmol m−2 s−1) and the flow rate of gas pump (500 mmol s −1) were set.
Biometric measurements
To determine the leaf area (cm2), twenty leaves were randomly sampled from all tree canopy and measured with a leaf area meter (AT Delta – T Device, UK). The dry mass of twenty leaves was determined by drying apple tree leaves in 70 °C for 48 h. (Venti cell 222, Medcenter Einrichtungen, Gräfeling, Germany) to constant weight33.
Meteorogical conditions
The meteorogical data were collected from ‘iMetos’ meteorogical in an intensive orchard. The air temperature and precipitation in last two years were very variable (Fig. 1). Air temperature was close to multi-annual, while precipitation during the vegetation period (May to September) was much higher than multi-annual (average of 100 years), especially on harvest time in 2017. While during bloom period the precipitation was very low in both years. Drought year of research was characterized by a higher than usual temperature and low and very variable precipitation. Natural drought was announced in Lithuania that year (Lithuania, lat. 55°N, 2017–2018). Although rainfall in 2018 is significantly lower than in 2017 and average of multiannual precipitation, given that apple trees are perennial woody plants, we do not generally consider drought stress. However, on harvest time precipitation level was very low in 2018. On July 12, 2018, precipitation was 52 mm during one day, when on the other days it was only 0–8 mm per day.
Statistical analysis
The data was processed using XLStat software (Addinsoft, 2019, USA, 2019), The data were processed using two-way ant three-way analysis of variance (ANOVA) Analysis of variance was performed with Duncan’s least significant difference test (P < 0.05).
Results
Mechanical pruning results denser crown and light penetration into canopy. The lower light penetration into the crown resulted the decrease of PRI (25–100%) (Fig. 2A). However calcium-prohexadione increased light penetration into canopy at the same time increased PRI 45% compared to control. The movement of nutrients and water disrupted by trunk incision has significantly negative effect PRI. PRI was more than 50% lower by using trunk incision compared to control. Mechanical pruning increased PSRI to 30% in the lower part of the canopy, but decreased 50–65% in the top of the canopy (Fig. 2B).
Photochemical reflectance (A) and plant senescence reflectance (B) indices in apple trees on harvest time in average of two years. 1. Each year hand pruning forming slender spindle (control); 2. Mechanical pruning (each year) with hand pruning every second year; 3. Mechanical pruning (each year); 4. Trunk incision using chain saw + mechanical pruning (each year); 5. Mechanical pruning (each year) + spraying with calcium-prohexadione. Averages followed by different letter within the same figure indicate significant differences according to the Duncan’s least significant difference test (P < 0.05). Error bars shows standard deviation.
Nitrogen balance index showed significant differences only in the lower part of canopy. Mechanical pruning with manual pruning increased NBI by 38% compared to control. Meanwhile, using trunk cut, the NBI variation is insignificant compared to the control (Fig. 3). NBI increased reached the highest values by the use of mechanical pruning with hand pruning in the lower part of the canopy in 2017 (Fig. S1)
Nitrogen balance index in apple trees on harvest time. 1. Each year hand pruning forming slender spindle (control); 2. Mechanical pruning (each year) with hand pruning every second year; 3. Mechanical pruning (each year); 4. Trunk incision using chain saw + mechanical pruning (each year); 5. Mechanical pruning (each year) + spraying with calcium-prohexadione. Averages followed by different letter indicate significant differences according to the Duncan’s least significant difference test (P < 0.05). Error bars shows standard deviation.
Trunk incision and calcium-prohexadione significantly decreased DW/FW ratio to 10–12% (Fig. 4). Compared to other agrotechnological tools, mechanical pruning resulted in significant decrease of photosynthetic rate (PN). PN decreased by 47% using only mechanical pruning compared to pruning by super spindle. Mechanical pruning with hand pruning, trunk incision and calcium-prohexadione also resulted in significant decrease of PN 10–20% compared to control.
Dry and fresh weight ratio (A) and photosynthetic rate (B) in apple trees on harvest time. 1. Each year hand pruning forming slender spindle (control); 2. Mechanical pruning (each year) with hand pruning every second year; 3. Mechanical pruning (each year); 4. Trunk incision using chain saw + mechanical pruning (each year); 5. Mechanical pruning (each year) + spraying with calcium-prohexadione. Averages followed by different letter within the same figure indicate significant differences according to the Duncan’s least significant difference test (P < 0.05). Error bars shows standard deviation.
Mechanical pruning increased apple yield by 45% but decreased fruit size by 4% in the average of two years (Fig. 5). Meanwhile trunk incision has no significant impact on yield but fruit mass decreased by 5.2% compared to control. Calcium-prohexadione has no significant effect for yield and fruit mass. There was no significant differences between mechanical pruning all treatment, however all treatments was significant higher compared to control in 2017. Meanwhile mechanical pruning reached 70 t ha−1 yield in 2018 (Fig. S2).
Total yield t/ha (columns) and mean apple weight g differences. 1. Each year hand pruning forming slender spindle (control); 2. Mechanical pruning (each year) with hand pruning every second year; 3. Mechanical pruning (each year); 4. Trunk incision using chain saw + mechanical pruning (each year); 5. Mechanical pruning (each year) + spraying with calcium-prohexadione Averages followed by different letter on yield and on mean fruit mass indicate significant differences according to the Duncan’s least significant difference test (P < 0.05). Error bars shows standard deviation.
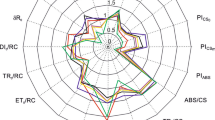
The treatment values were subjected to a principal component analysis (PCA). The first two components accounted for the majority of variation in the data set (88.59%). The variation of 82.60% could be explained by the first principal component (PC1) and 5.99% by the droughty principal component (PC2). Strong positive correlation between PRI and NBI 0,89–0.94, and strong negative correlations between PRI, NBI and PSRI −0.88 – (−0.91) were found (Fig. 6).
Discussion
Wong et al.34 identified three causes of PRI variation, including changes in chlorophyll-carotenoid ratio (seasonality), light intensity, but also the effect of temperature on PRI (PRI and PSII efficiency). In their research is shown that at lower temperatures and dark green leaves, PRI values increase. Similarly, Weng et al.35 found that the relationship between the efficiency of PRS II and PRI varies with temperature and leaf color, they argue that lower PRI values are associated with increased illumination of mango tree leaves. While getting into the details of other authors’ data, and considering that this research comes from autumn measurements, when daytime temperatures drop, we find that during mechanical pruning, PRI responds more to temperature than to light intensity (Fig. 1). Mechanical pruning resulted in a denser apple tree shingle in the lower part of the tree, which caused the trap to release heat from the ground and prevent the wind from spreading. In addition, the leaves were light green due to poor lighting. Perhaps this is why PRI rates remained low at harvest (Fig. 2A). Optimal PSRI values ranged from −0.1 to 0.214, and most trees were in good condition except for the trees treated by mechanical pruning with and without manual pruning. PSRI indices showed that the leaves of apple trees affected by mechanical pruning were did not reached the stage of aging at harvest time (Fig. 2B).
Denser planting of apple trees resulted less light penetration into canopy, thus lower NBI values were obtained36. Other studies we have done, and other authors have found that intense sunlight can reduce photosynthesis and promote metabolism36,37. Cronin and Lodge37 found that low light increased nitrogen content in leaf by 53%. In this study NBI decreased in top of the canopy by 25% (Fig. 3). Yang et al.9 demonstrated significant increases in N concentration in all Abies fabri (Mast.) organs by response to drought. We see that NBI increased significantly only in the sunny and droughty years (Fig. S1A,B)
Summer pruning and trunk incision is useful tool for better light penetration to overly dense canopies2. Meanwhile trunk incision disturbs water and nutrients movement and inhibits the vital functions and photosynthesis of the apple tree as decrease of photosynthetic rate was observed (Fig. 3).
Water deficit is one of the greatest limiting factor for photosynthesis and biomass accumulation38,39,40. The negative impact of the water deficit on woody plants has been extensively studied in trees (such as Alnus glutinosa, Betula pendula, Cercis canadensis, Fraxinus excelsior Pinus sylvestris, Picea abies, Populus tremula, Quercus robur, Robinia pseudoacacia)41,42,43. However, Kozlowski and Pallardy44 observations of woody plants adaptation to drought stress showed that trees have developed mechanisms to deal with dehydration conditions that are different from herbaceous plants. The amount of unstructured carbohydrates may also reflect tree drought, which may lead to a decrease in the concentration of unstructured carbohydrates, but as Gaul et al. In45, this usually only occurs in the later stages of drought.
Water intake increases with increased root fine growth, deep root formation, and solvent accumulation to reduce the water potential in the root tissue. Only then does the drought level become too severe, and the plants react by activating the mechanisms that protect tissues from cell damage, mainly by the action of protective solutes and proteins46,47. Trees that have been touched after a short drought have no major changes, even some tree species are able to recover quickly after a drought48,49. During treatment years was one drought episode form May till July (one day hard rainfall in July, and no rain other days), then another drought episode from the end of August till harvest time appeared. Apple trees were affected by short-term drought in 2018 but form the PRI and PSRI values it can be stated that the trees quickly recovered and functioned even better than in normal years.
Denser canopy formed by mechanical pruning decreases another limiting factor – light penetration into canopy. PN is up to 50% lower in shade leaves compared to sun leaves50. Negative shade impact for trees photosynthesis also was described in several research like for Acer rubrum and Betula papyrifera51, Fagus syltica52, Ginkgo biloba53, Malus domestica54. However, shading can have positive impact for photosynthesis when it goes with drought but in our research there was no significant impact of shading55.
In agreement to Rze56 that calcium-prohexadione had no significant impact for fruit weight for apple cultivar ‘Rubin’ (Fig. 4). During the arid years calcium-prohexadione even increased ‘Rubin’ fruit weight. In our research the yields of the apple trees after prohexadione-Ca exposure were significantly bigger compared to untreated trees, meanwhile, other authors demonstrates similar results between treated and untreated trees57,58.
Conclusion
Strong positive correlation between PRI and NBI 0,89–0,94, and strong negative correlations between PRI, NBI and PSRI −0.88 – (−0.91) were found. Trunk incision disturbed nutrients and water movement from roots to canopy that resulted the significant decrease of photochemical reflectance index, photosynthetic rate, fruit mass and accelerated leaf aging. The lower light penetration in denser canopy formed using mechanical pruning also resulted in significant decrease of photosynthetic indices (PRI, PSRI, Pn). Mechanical pruning increased apple yield upto 45% but decreased fruit size by 4% in two years average. In general, the recommendation for industrial orchards is hand pruning every second year, mechanical pruning combined with reduced manual pruning lowered orchard maintenance but retained apple yield and photosynthetic activity.
References
Long, S. P., Zhu, X. G., Naidu, S. L. & Ort, D. R. Can improvement in photosynthesis increase crop yields? Plant Cell Environ. 29, 315–330 (2006).
Wünsche, J. N., & Lakso A. N. Apple Tree Physiology-Implications for Orchard and Tree Management. 43rd Annual IDFTAConference, 82–88 (2000)
Hüner, N. P. A., Dahal, K., Bode, R., Kurepin, L. V. & Ivanov, A. G. Photosynthetic acclimation, vernalization, crop productivity and ‘the grand design of photosynthesis’. J. Plant Physiol. 203, 29–43 (2016).
Sala, F., Arsene, G. G., Iordănescu, O. & Boldea, M. Leaf area constant model in optimizing foliar area measurement in plants: A case study in apple tree. Sci. Hortic. (Amsterdam) 193, 218–224 (2015).
Gamon, J. A. & Surfus, J. S. Assessing leaf pigment content and activity with a reflectometer. New Phytol. 143, 105–117 (1999).
Agati, G. et al. Fluorescence-based versus reflectance proximal sensing of nitrogen content in Paspalum vaginatum and Zoysia matrella turfgrasses. Eur. J. Agron. 45, 39–51 (2013).
Cerovic, Z. G., Masdoumier, G., Ghozlen, Ben, N. & Latouche, G. A new optical leaf-clip meter for simultaneous non-destructive assessment of leaf chlorophyll and epidermal flavonoids. Physiol. Plant. 146, 251–260 (2012).
Overbeck, V., Schmitz, M., Tartachnyk, I. & Blanke, M. Identification of light availability in different sweet cherry orchards under cover by using non-destructive measurements with a DualexTM. Eur. J. Agron. 93, 50–56 (2018).
Yang, Y., Guo, J., Wang, G. & Yang, L. Effects of drought and nitrogen addition on photosynthetic characteristics and resource allocation of Abies fabri seedlings in eastern Tibetan Plateau. New forests 43(4), 505–518 (2012).
Filella, I. et al. PRI assessment of long-term changes in carotenoids/chlorophyll ratio and short-term changes in de-epoxidation state of the xanthophyll cycle. International Journal of Remote Sensing 30(17), 4443–4455 (2009).
Merlier, E., Hmimina, G., Bagard, M., Dufrêne, E. & Soudani, K. Potential use of the PRI and active fluorescence for the diagnosis of the physiological state of plants under ozone exposure and high atmospheric vapor pressure deficit. Photochemical & Photobiological Sciences 16(8), 1238–1251 (2017).
Gamon, J. A., Field, C. B., Fredeen, A. L. & Thayer, S. Assessing photosynthetic downregulation in sunflower stands with an optically-based model. Photosynth. Res. 67, 113–25 (2001).
Garbulsky, M. F., Peñuelas, J., Gamon, J., Inoue, Y. & Filella, I. The photochemical reflectance index (PRI) and the remote sensing of leaf, canopy and ecosystem radiation use efficiencies. A review and meta-analysis. Remote Sens. Environ. 115, 281–297 (2011).
Merzlyak, M. N., Gitelson, A. A., Chivkunova, O. B. & Rakitin, V. Y. Non‐destructive optical detection of pigment changes during leaf senescence and fruit ripening. Physiologia Plantarum. 106, 135–141 (1999).
Sims, D. A. & Gamon, J. A. Relationships between leaf pigment content and spectral reflectance across a wide range of species, leaf structures and developmental stages. Remote Sens. Environ. 81, 337–354 (2002).
Song, Q., Zhang, G. & Xin-Guang, Z. Optimal crop canopy architecture to maximise canopy photosynthetic CO 2 uptake under elevated CO2 – a theoretical study using a mechanistic model of canopy photosynthesis. Functional Plant Biology. 40(2), 109–124 (2013).
Alarcon, V. J. & Sassenrath, G. F. Optimizing canopy photosynthetic rate through PAR modeling in cotton (Gossypium spp.) crops. Elsevier B.V. 119, 142–152 (2015).
Li, K. T. & Lakso, A. N. Photosynthetic characteristics of apple spur leaves after summer pruning to improve exposure to light. Hort Science. 39, 969–972 (2004).
Zhang, H., Tao, Y. & Hu, J. Photosynthetic rate prediction model of cucumber seedlings fused chlorophyll content. Transactions of the Chinese Society of Agricultural Machinery 46(8), 259–263 (2015).
Liu, J. & Shi, D. Photosynthesis, chlorophyll fluorescence, inorganic ion and organic acid accumulations of sunflower in responses to salt and salt-alkaline mixed stress. Photosynthetica. 48, 127–134 (2010).
Ohashi, Y., Nakayama, N., Saneoka, H. & Fujita, K. Effects of drought stress on photosynthetic gas exchange, chlorophyll fluorescence and stem diameter of soybean plants. Biologia Plantarum. 50, 138–141 (2006).
Long, S. P., Humphries, S. & Falkowski, P. G. Photoinhibition of photosynthesis in nature. Annu. Rev. Plant Physiol. 85, 990–995 (1994).
Cornic, G. Drought stress inhibits photosynthesis by decreasing stomatal aperture - not by affecting ATP synthesis. Trends Plant Sci. 5, 187–188 (2000).
Salvador, F. R., Fisichella, M. & Fontanari, M. Correlations between fruit size and fruit quality in apple trees with high and standard crop load levels. J. Fruit Ornam. Plant Res. 14, 113–122 (2006).
Greene, D. W. The effect of repeat annual applications of prohexadione-calcium on fruit set, return bloom, and fruit size of apples. Hort Science. 43, 376–379 (2008).
Privé, J. P. et al. Preliminary results on the efficacy of apple trees treated with the growth retardant proxadione- calcium (Apogee) in Eastern Canada. Acta Horticulture. 636, 37–144 (2004).
Teryl, R. R. & Larry, E. W. Net CO2 Assimilation and Carbohydrate Partitioning of Grapevine Leaves in Response to Trunk Girdling and Gibberellic Acid Application. Plant Physiology. 89(4), 1136–1140 (1989).
Rufato, L. et al. Effect of trunk girdling on growth and crop yield of ‘Pacham’s triumph’ pear. Acta Hortic. 1094, 265–268 (2015).
Fayek, M. A., Yehia, T. A. & Farag, A. M. Effect of Ringing and Amino Acids Application on Improving Fruiting of Le Conte Pear Trees. Journal of Horticultural Science & Ornamental Plants. 33(1), 1–10 (2011).
Wargo, J. M., Merwin, I. A. & Watkins, C. B. Nitrogen fertilization, midsummer trunk girdling, and AVG treatments affect maturity and quality of “Jonagold” apples. Hort Science. 39(3), 493–500 (2004).
Glenn, D. M. & Campostrini, E. Girdling and summer pruning in apple increase soil respiration. Scientia Horticulturae. 129(4), 889–893 (2011).
Zhao, Y. et al. Influence of girdlind and foliar-applied urea on apple (Malus Domestica L.) fruit quality. Pakistan Journal of Botany. 45(5), 1609–1615 (2013).
Leach, K. A. & Hameleers, A. The effects of a foliar spray containing phosphorus and zinc on the development, composition and yield of forage maize. Grass and Forage Science 56, 311–315 (2001).
Wong, C. Y. S. & Gamon, J. A. Three causes of variation in the photochemical reflectance index (PRI) in evergreen conifers. New Phytol 206, 187–195 (2015).
Weng, J. H., Jhaung, L. H., Lin, R. J. & Chen, H. Y. Relationship between photochemical efficiency of photosystem II and the photochemical reflectance index of mango tree: merging data from different illuminations, seasons and leaf colors. Tree Physiol. 30(4), 469–78 (2010).
Laužikė, K. & Samuolienė, G. Leaf optical properties reflect changes of photosynthetic indices in apple trees. Research for Rural Development. 2, 90–94 (2018).
Cronin, G. & Lodge, D. Effects of Light and Nutrient Availability on the Growth, Allocation, Carbon/nitrogen Balance, Phenolic Chemistry, and Resistance to Herbivory of Two Freshwater Macrophytes. Oecologia 137(1), 32–41 (2003).
Medrano, H., Escalona, J. M., Bota, J., Gulías, J. & Flexas, J. Regulation of photosynthesis of C3 plants in response to progressive drought: Stomatal conductance as a reference parameter. Annals of Botany. 89, 895–905 (2002).
Ma, X. et al. Biomass accumulation, allocation, and water-use efficiency in 10 Malus rootstocks under two watering regimes. Agroforestry Systems. 80(2), 283–294 (2010).
Jiménez, S., et al. Physiological, biochemical and molecular responses in four Prunus rootstocks submitted to drought stress. Tree Physiology. 33(10) (2013)
Griffin, J. J., Ranney, T. G. & Pharr, D. M. Heat and drought influence photosynthesis, water relations, and soluble carbohydrates of two ecotypes of redbud (Cercis canadensis). Journal of the American Society for Horticultural Science. 129(4), 497–502 (2004).
Pliūra, A. et al. Response of juvenile progeny of seven forest tree species and their populations to simulated climate change-related stressors, heat, elevated humidity and drought. IForest. 11(3), 374–388 (2018).
Liu, X. et al. Effects of soil water and nitrogen availability on photosynthesis and water use efficiency of Robinia pseudoacacia seedlings. Journal of Environmental Sciences (China). 25(3), 585–595 (2013).
Kozlowski, T. T. & Pallardy, S. G. Acclimation and adaptive responses of woody plants to environmental stresses. The botanical review 68(2), 270–334 (2002).
Gaul, D., Hertel, D., Borken, W., Matzner, E. & Leuschner, C. Effects of experimental drought on the fine root system of mature Norway spruce. Forest Ecology and Management. 256, 1151–1159 (2008).
Tuberosa, R. Phenotyping for drought tolerance of crops in the genomics era. Frontiers in physiology 3, 347 (2012).
Claeys, H. & Inzé, D. The agony of choice: how plants balance growth and survival under water-limiting conditions. Plant physiology 162(4), 1768–1779 (2013).
Pretzsch, H., Schütze, G. & Uhl, E. Resistance of European tree species to drought stress in mixed versus pure forests: evidence of stress release by inter‐specific facilitation. Plant Biology 15, 483–495 (2013).
Hartmann, H., Ziegler, W. & Trumbore, S. Lethal drought leads to reduction in nonstructural carbohydrates in Norway spruce tree roots but not in the canopy. Functional Ecology 27, 413–427 (2013).
Lichtenthaler, H. K., Alexander, A., Michal, V., Marek, J. K. & Otmar, U. Differences in Pigment Composition, Photosynthetic Rates and Chlorophyll Fluorescence Images of Sun and Shade Leaves of Four Tree Species. Plant Physiology and Biochemistry. 45(8), 577–88 (2007).
Kubiske, M. E. & Pregitzer, K. S. Ecophysiological responses to simulated canopy gaps of two tree species of contrasting shade tolerance in elevated CO2. Funct. Ecol. 11, 24–32 (1997).
Lichtenthaler, H. K., Babani, F., Langsdorf, G. & Buschmann, C. Measurement of differences in red chlorophyll fluorescence and photosynthetic activity activity between sun and shade leaves by fluorescence imaging. Photo- synthetia. 38, 521–529 (2000).
Pandey, S., Kumar, S. & Nagar, P. K. Photosynthetic performance of Ginkgo biloba, L. grown under high and low irradiance. Photosynthetica. 4, 505–511 (2003).
Willaume, M., Pierre, É. L. & Hervé, S. Light Interception in Apple Trees Influenced by Canopy Architecture Manipulation. Trees - Structure and Function. 18(6), 705–13 (2004).
Lopez, G. et al. Effect of shading and water stress on light interception, physiology and yield of apple trees. Agricultural Water Management 210(January), 140–148 (2018).
Rze, B. Effect of bioregulators and summer pruning on growth and cropping of ‘Rubin’ apple trees. Folia Hortic. 18(2), 37–46 (2006).
Medjdoub, R., Val, J. & Blanco, A. Prohexadione-Ca inhibits vegetative growth of ‘Smoothee Golden Delicious’ apple trees. Scientia Hort. 101, 243–253 (2004).
Šabajevienė, G. et al. The effect of ambient air temperature and substrate moisture on the physiological parameters of spring barley. Zemdirbyste-Agriculture. 95(4), 71–80 (2008).
Acknowledgements
This work was carried out within the framework of long-term research program ‘Horticulture: agrobiological basics and technologies’ implemented by Lithuanian Research Centre for Agriculture and Forestry.
Author information
Authors and Affiliations
Contributions
N.U. and G.S. were involved in experimental design, N.U. was responsible for conducting of the experiments and data of yield, V.Š.S. and K.L. performed the analysis, K.L. statistical data analyses and wrote the manuscript and prepared figures. K.L., G.S., N.U. and V.Š.S. wrote methodical part of manuscript. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Laužikė, K., Sirgedaitė-Šėžienė, V., Uselis, N. et al. The Impact of Stress Caused By Light Penetration and Agrotechnological Tools on Photosynthetic Behavior of Apple Trees. Sci Rep 10, 9177 (2020). https://doi.org/10.1038/s41598-020-66179-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-66179-3
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.