Abstract
The Capripoxvirus genus includes three agents: Sheeppox virus, Goatpox virus and Lumpy skin disease virus. Related diseases are of economic importance and present a major constraint to animals and animal products trade in addition to mortality and morbidity. Attenuated vaccines against these diseases are available, but afforded cross-protection is controversial in each specie. In this study, groups of sheep, goats and cattle were vaccinated with Romania SPPV vaccine and challenged with corresponding virulent strains. Sheep and cattle were also vaccinated with Neethling LSDV vaccine and challenged with both virulent SPPV and LSDV strains. Animals were monitored by clinical observation, rectal temperature as well as serological response. The study showed that sheep and goats vaccinated with Romania SPPV vaccine were fully protected against challenge with virulent SPPV and GTPV strains, respectively. However, small ruminants vaccinated with LSDV Neethling vaccine showed only partial protection against challenge with virulent SPPV strain. Cattle showed also only partial protection when vaccinated with Romania SPPV and were fully protected with Neethling LSDV vaccine. This study showed that SPPV and GTPV vaccines are closely related with cross-protection, while LSDV protects only cattle against the corresponding disease, which suggests that vaccination against LSDV should be carried out with homologous strain.
Similar content being viewed by others
Introduction
Capripoxvirus genus includes 3 viruses, namely Sheeppox virus (SPPV), Goatpox virus (GTPV) and Lumpy Skin Disease Virus of cattle (LSDV). Diseases related to these viruses are of economic importance as they cause significant damages on meat, milk production, wool and leather quality in addition to carcass depreciation and mortality1,2,3.
SPPV and GTPV are widespread throughout Northern and Central Africa, Middle East, Indian subcontinent, Central Asia, China, Vietnam and Russia4,5. Recent outbreaks have been reported in Azerbaijan, Greece and Bulgaria5. LSDV was first confined in Austral Africa then spread to Middle East areas including Israel, Lebanon, Jordan, Kuwait and Saudi Arabia. Since 2013, the disease has been notified in Turkey, Iran, Cyprus, Greece, Azerbaijan, Georgia, Russia, Bulgaria, Serbia, Albania, Kazakhstan, China, Bangladesh and India4,6,7.
All capripoxviruses are antigenically related to each other and can only be distinguished by molecular characterization8,9. Though SPPV and GTPV are serologically indistinguishable, some authors have reported a host preference with most strains of SPPV and GTPV10.
Vaccination is the only effective method to control the disease in endemic areas. Despite vaccination efforts, only a few countries have successfully eradicated these diseases11. To date, available vaccines are live attenuated specific for small ruminants and cattle. In some countries, people use SPPV to vaccinate against GTPV and LSDV12. Cross-protection between SPPV and GTPV has been already demonstrated for various strains and vaccination with LSDV Kenyan Sheep and Goat pox (KSGP) strain has also been widely used for many years to protect small ruminants against SPPV and GTPV13,14.
Experiments have been performed in some countries to evaluate the cross-protection amongst the 3 members of Capripoxvirus genus15. Immunological studies and field trials have been conducted, but few studies assessed the efficacy of vaccine strains by challenge16,17.
The aim of this study was to evaluate the cross-protection between SPPV, GTPV and LSDV on sheep, goats and cattle. We selected a well-known, highly immunogenic SPPV strain (Romania) and the most used LSDV strain (Neethling) to vaccinate sheep, cattle and goats against their respective diseases. Protection was evaluated by serological monitoring, using neutralizing antibody assays and challenge using corresponding virulent strains.
Results
Vaccine preparation
For the live Romania SPPV vaccine, the virus showed a cytopathic effect starting at day 3 (D3) and was harvested at D4 post-infection (pi) with a titer of 106.0 TCID50/ml. After freeze-drying, the infectious titer of the final vaccine was 105.0 TCID50/vial of 100 doses.
The Neethling vaccine strain showed similar effect starting from D3 and was harvested at D5 pi. The infectious titer was 106.8 TCID50/ml at the harvest and 105.7 TCID50/ vial of 50 doses after vaccine freeze-drying.
Vaccines complied with analytical QC testing. They were sterile, free from adventitious agents and no extraneous agents were detected by qPCR.
Animal vaccination
In the group of sheep vaccinated with Romania SPPV (G1), a slight rise in temperature was noticed 5 to 7 days post vaccination (pv) in 2 sheep with limited inflammation at the injection site. The other sheep remained within the normal range. In the group of goats vaccinated with Romania SPPV (G2), 2 weeks following vaccination, the body temperature remained in the normal range and no clinical signs of GTPV nor inflammation at inoculation site were observed. Cattle vaccinated with Romania SPPV (G3) did not show hyperthermia nor inflammation at the injection site.
Sheep vaccinated with LSDV vaccine (G4) showed a slight increase in temperature between D2 and D4 pv and a limited local reaction was observed in 5 out of 8 animals from D3 pv until D12 pv. Cattle vaccinated with LSDV Neethling (G5) did not show hyperthermia except a slight rise at D7 pv for one cow.
Serology of vaccinated animals, evaluated by Virus Neutralization Test (VNT) revealed 100% of positive animals starting from D14 pv in the group of sheep vaccinated with SPPV Romania (G1), with a maximum titer of 1.6 log10 at D21 pv. In the group of goats (G2), 5 out of 8 goats were positive at D21 pv with a maximum titer of 1.7 log10. Serology of cattle vaccinated with SPPV Romania SPPV (G3) showed no seroconversion as all the animals remained negative.
In the group of sheep vaccinated with LSDV vaccine (G4), only 2 among 8 animals were positive using VNT, starting from D7 pv with a low neutralizing antibody titer (0.3 log10). In the group of cattle vaccinated with LSDV vaccine (G5), 50% of animals were seropositive at D28 pv (Fig. 1).
Serological response of animals after vaccination. Graph A presents average antibody titers of positive goats vaccinated with SPV vaccine. Graph B presents average antibody titers of positive sheep vaccinated with LSD and SPV. Graph C presents average antibody titers of cattle vaccinated with SPV and LSD vaccines.
Control animals remained negative during the observation period.
Challenge virus preparation
Sheep used to prepare the SPPV challenge strain showed a huge skin inflammation of 10 cm in diameter at the injection site. Animals were euthanized at D13 pi to collect aseptically lymph and inflammatory tissues. Viral suspension showed a CT value of 13,3 at qPCR, which corresponds approximately to a titer of 107.0 TCID50/ml.
Under the same conditions, goats inoculated with virulent GTPV showed an important erythema followed by a skin inflammation that turned into oozing pustule (Fig. 2). Animals were euthanized at D12 pi and the collected tissue was prepared following the same protocol as SPPV. The CT of the supernatant, obtained by qPCR, was 18,9 which corresponds to 105.5 TCID50/ml
The titer of LSDV challenge virus used in Sciensano was 106.55 TCID50/100 μl.
Experimental infection
After the challenge of sheep with virulent SPPV strain, unvaccinated sheep showed an increase in body temperature between D4 and D10 pi reaching 41.7 °C at D5 pv, in addition to a local reaction at the injection site that appeared at D3 pi. Typical SPPV skin secondary nodules were observed at D10 pi.
Sheep vaccinated with Romania SPPV (G1) exhibited a transient increase in temperature between 39.6 °C and 39.7 °C at D6 and D7 pi. Hypersensitivity reaction at the injection site in the whole group was noticed at D2 pi in the first dilution and disappeared the following day. None of the immunized animals showed clinical signs of SPPV during the observation period. Serological response confirmed the identity of the challenge virus as demonstrated by the increase of neutralizing titer at D14 pi (2.2 log10). Using the protection index protocol, the infectious titer obtained in unvaccinated sheep was 105.7 ID50/ml, significantly higher than that obtained with vaccinated animals (100.75 ID50/ml) (P ≤ 0.05). The protection index (PI) was estimated at 4.7 in sheep vaccinated with Romania strain (G1).
Sheep vaccinated with LSDV vaccine (G4) showed an increase in body temperature between D7 and D8 pi. Hypersensitivity reaction at the injection site was observed at D2 and D3 pi (n = 7), followed at D4 pi, by papules that reached 3 cm of diameter in 3 among 8 vaccinated animals. Two among 8 vaccinated sheep showed a PI of 3 and 4.5, while 3 sheep showed moderate protection (PI: 1, 1 and 2). The other 3 animals did not show any protection.
In the goat group, the 2 unvaccinated control goats challenged with the virulent GTPV virus showed a rise in mean body temperature, between D4 and D9 pi, that reached 41.1 °C at D6 pv. Typical skin papules appeared at injection site at D3 pi and reached 2 cm of diameter at D5 pi. Lesions showed confluence tendency and evolved to eschar and abscess in low dilution range. Secondary lesions were also observed in hairless part of the body at D10 pi.
Goats vaccinated with Romania strain (G2) did not show any rise in body temperature, however, a hypersensitivity reaction at the injection site was observed during 2 days pi (Fig. 3). The reaction disappeared completely before D4 pi. None of the immunized animals showed clinical signs of GTPV during the observation period. Serology confirmed the identity of the challenge virus since all the animals showed an increase of the neutralization titer (2.9 log10) at D7 pi. The obtained titer in the 2 unvaccinated animals was 106.5ID50/0.2 ml, significantly higher than that obtained with vaccinated animals (101.3ID 50/ml) (P ≤ 0.05). The PI was 5,2.
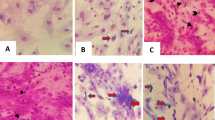
(A) Figure of challenged vaccinated goat showing hypersensitivity reaction and no local inflammations on site of inoculation with10–1 to 10–6 dilutions (left to right) of virulent GPV. (B) figure of challenged unvaccinated goat showing local inflammation on site of inoculation. The arrows show inflammation or hypersensitivity reaction on inoculation site.
All cattle vaccinated with SPPV Romania vaccine (G3) showed, after challenge with virulent LSDV virus, fever with spikes between D7 and D9 pi. Three out of 7 cattle showed important swelling starting from D9 pi and developed localized nodules in one animal and generalized in 2 animals, which developed large pre-scapular lymph node enlargement. The 3 animals were viremic after challenge (Ct = 32). Animals of the control group displayed fever until the end of the trial with a spike at D6 to D8 pi. Three out of 5 animals were viremic starting from D3 pi and developed generalized nodules starting from D13 pi. Regarding the clinical score, there was no significant difference between groups of unvaccinated and vaccinated cattle with Romania SPPV (P > 005).
In the group of cattle vaccinated with Neethling strain (G5), body temperature remained normal and no animal showed clinical signs nor viremia after challenge. Unvaccinated control cattle showed pathognomonic clinical signs of LSDV in 3 out of 5 animals with viremia (between 3 and 17 dpi) and positive buccal swabs. Clinical scores were significantly higher in the group of unvaccinated animals compared to cattle vaccinated with Neethling strain (P ≤ 0.05) (Table 1).
Discussion
Prevention of Capripoxvirus infections in countries where the diseases exist is mainly based on vaccination of susceptible animals18. As the 3 capripoxviruses share major neutralization sites, cross-immunity has been reported, but not experimentally documented4,19.
The use of SPPV vaccine to protect sheep, goats and cattle against infections is the most common4,20. SPPV vaccine strains, such as the Romania SPPV and Yugoslavian RM65, are widely used in endemic countries to protect sheep against their disease,13,21,22,23. Romania SP strain has also been used to vaccinate goats with controversial results. Rao et al. (2000) and Abdelfatah et al. (2018) reported the vaccine to be not protective of goats against GTPV24,25. In Saudi Arabia, Abuelzein et al. (2003) noticed the apparition of Goatpox disease in animals vaccinated with locally produced Romania SPPV vaccine26, whereas the same authors recommended vaccination of goats with the same vaccine at 3 months of age with annual booster27. According to Molla et al. (2017), immunization failure may be linked to vaccination conditions, vaccine reconstitution and coverage rate28. However, although no challenge was carried out to confirm the cross protection, Abdelfatah et al. (2019) observed that vaccination of goats with Romania strain induced cell-mediated immunity with a satisfactory PBMC and lymphocyte proliferation level29.
To vaccinate cattle against LSDV, partial cross-protection with SPPV vaccines has been documented from the field in the Middle East and Africa with SPPV RM65, Bakirkoy and Romania strains4. Vaccination failure after the use RM65 strain in cattle has been cited by Brenner et al. (2009) and Abutarbush et al. (2014). Indeed, during the LSDV outbreak of 2006 in Israel, an incomplete protection of herds was detected, as 11% of vaccinated cattle showed LSDV symptoms12. The same results were reported in Jordan (Abutarbush et al., 2014) where a partial protection was provided by RM65 vaccine, although morbidity and mortality rates were lower compared to unvaccinated herds30. In another study, it appeared that 10 time doses of RM65 vaccine induced high antibody response, but no challenge has been performed to evaluate cell-mediated protection17. Sevik and Dorgan (2016) also reported failure of vaccination with SPPV Bakirkoy strain to protect cattle in the field against LSDV31. Similarly, Abdallah et al. (2018) and Zeedan et al. (2019) described cases of infected cattle emerging from a herd vaccinated with Romania vaccine in Egypt32,33. Studies on humoral and cellular response upon vaccinated cattle against LSDV with Romania strain showed an induction of humoral response, proliferation of lymphocytes and stimulation of Interferon gamma and IL4, but no challenge was carried out to confirm the protection15,34,35. Also, Mikhael et al. (2016) did not perform any challenge, however they observed that Romania SPPV vaccine did not protect sufficiently cattle against LSDV and concluded on the need to use homologous strain for cattle vaccination36. In another study, Mikhael et al. (2017) reported that combination of SPPV and GTPV may induce better protection of cattle against LSDV but the homologous strain remains the best37. Similar results were reported by Aboul Soud et al. (2018), who showed that no serological response was induced in cattle vaccinated with Romania strain while a trivalent Capripox vaccine (made up of SP Romania, GTPV Held and KSGP 0180) induced antibodies in 66% of vaccinated animals38.
Vaccination of sheep with GTPV vaccine has been rarely reported. Agrawal et al. (1997) described a weak protection at the challenge on a group of 6 sheep vaccinated with a not named strain of GTPV39. Nevertheless, protection of cattle vaccinated with Kedong and Isiolo GTPV has been reported against LSDV challenge40. The GTPV Gorgan strain is also commonly used in Iran to protect cattle against LSDV along with SPPV RM6535 and in a recent experiment, Gorgan strain has been reported to be more immunogenic than KSGP 0180 and Neethling strains of LSDV41. This experimental study was based on challenge and cellular immune response (by Delayed-Type Hypersensitivity). Such observations should however be confirmed, as protection may depend on the replicative capacity of the virus in the animal and on the vaccine production process.
The use of LSDV to prevent infections of sheep and goats has not been reported to our knowledge. However, Kenya LSDV strain (long time considered as a Sheep Goatpox (SGP) has been extensively used to protect small ruminants against their respective diseases with controversial results. The KSGP 0180 and KSGP 0240 have been reported to induce incomplete protection of cattle against LSDV in Egypt and Ethiopia42,43,44. In sheep, similar vaccination failure has also been reported against SGPV using the KSGP O-180 strain4.
The discrepancy reported on cross-protection between Capripoxvirus genus members prompted us to conduct this experiment on a vaccination/challenge study using SPPV vaccine (Romania strain) to protect sheep, goats and cattle against SPPV, GTPV and LSDV respectively. In parallel, we also conducted a LSDV (Neethling strain) vaccination trial to protect sheep and cattle against SPPV and LSDV. Strains used in this experiment, have been tested before to be immunogenic in target species at the recommended dose of vaccination.
We chose live vaccines as they are the most common in the field and known to confer a solid immunity when used properly for the target species17,45.
Romania SPPV strain was selected because it has been used worldwide to prevent infections in small ruminants and is known to confer a high level of protection in sheep46. When used for mass vaccination, results are conclusive in the field and if vaccination pressure is regularly maintained, it may lead to disease eradication in the country47,48. Romania SPPV vaccine strain grows very well on different primary cells and also on Vero cell line which are suitable for vaccine preparation to avoid unavailability and potential adventitious contaminants. However, uncontrolled serial passages of virus on continuous heterologous cell line like Vero cells may limit the capacity of the strain to replicate in animals, affecting its immunogenicity49. This phenomenon has been observed with KSGP and Neethling strains, passed several times on Vero cells, that showed to be ineffective to protect cattle against the infection50. Regarding LSDV, Neethling strain is widely used and has been involved in the eradication of the disease in many countries, despite post-vaccination reported effects (Neethling disease)51.
To vaccinate animals against Capripoxvirus diseases, the minimal recommended vaccine dose is 102.5 TCID50 for small ruminants and 103.5 TCID50 for cattle23,52,53. In our study, we used a dose of 103.0 TCID50 for sheep and goats and 104.0 TCID50 for cattle, which are the most common used doses, to secure replication of the vaccine strain in animals. Used vials for animal vaccination were titrated on cells to ensure that the animals received the right dose.
Vaccination monitoring was conducted using VNT that detects protective IgG specifically. Sheep vaccinated with Romania SPPV strain were all seropositive at D14 pv with a maximum neutralizing titer of 1.6 log10 and 5 out of 8 goats vaccinated with Romania strain were positive at D21 with a maximum neutralizing titer of 1.7 log10. The reported kinetics of antibody response showed an increase in the titer despite the fact that Capripoxviruses induce mainly a cell-mediated immunity15,35. Those results are in agreement with Bhanuprakash et al. and Boumart et al., who cited an increase in neutralizing antibodies between D14 and D21 pv54,55.
The challenge of small ruminants vaccinated with SPPV Romania and LSDV Neethling was conducted according to the protocol by Fassi-Fehri et al.56. This method allows quantitative assessment of the conferred immunity and is based on the titers obtained from the challenge virus in vaccinated and unvaccinated animals. We selected this method because it is the one used routinely for years to conduct potency testing for SPPV vaccines. The method works perfectly for sheep and goats.
During the observation period, unvaccinated sheep and goats showed typical symptoms of SPPV and GTPV respectively. Unvaccinated animals were euthanized at D12 pi because of symptom severity, and virus recovery was conducted successfully from skin lesions in both sheep and goats. Serology also showed increase in the VNT titer after challenge, confirming the use of the homologous virulent strain in each species. The challenge dose allowed virus titration in sheep and goats and comparison between high and low immunogenic vaccines. Protection index in vaccinated sheep and goats was between 4.7 and 5.2, giving evidence of complete and long-lasting protection in those species against SPPV and GTPV infections56.
In the group of sheep vaccinated with LSDV Neethling, only 2 animals among 8 were serologically positive with a low antibody titer. The challenge showed partial protection, as only 2 animals were fully protected (positive also in serology), 3 partially protected and 3 others not protected. To our knowledge, this is the first time an experiment was carried out to test efficacy of Neethling strain in sheep. Challenge of goats vaccinated with LSDV was not performed in this experiment.
In cattle, vaccination monitoring using VNT showed 50% of positive cattle vaccinated with LSDV while no antibody response was detected in cattle blood three weeks after vaccination with SPPV vaccine. The discrepancy between cattle and small ruminants has been reported by many authors, who highlighted the fact that cattle do not all (100%) react to vaccination41,57,58, suggesting that a challenge is required to confirm protection of cattle against the disease. To perform challenge on cattle against LSDV virulent strain, Sciensano laboratory adopted a semi-quantitative method. Challenge of LSDV Neethling vaccinated cattle revealed full protection as no animal showed symptoms of LSDV, while 3 out of 5 control animals displayed numerous nodules accompanied by fever. Partial protection was observed in vaccinated animals with Romania strain as 4 out of 7 challenged calves showed protection while 3 animals displayed fever, viremia and typical lesions of LSDV. Our study demonstrated partial protection against LSDV when we used SPPV vaccine, which complies with Michael et al. observations59. The control group showed infection of 3 among 5 challenged cattle, which is in agreement with the experiment by Gari et al., who described clinical disease in only 3 out of 5 animal controls41. Besides, previous authors reported that only half of the infected animals may show clinical signs of LSDV60,61.
Molecular analysis of the 3 Capripoxviruses was investigated by several authors. Tulman et al. (2002) sequenced and analyzed complete genomes of several strains of SPPV, GTPV and LSDV and reported presence of a parent LSDV-like virus8. These results were confirmed by Biswas et al. (2019) who analyzed 36 different strains and showed a loss of 5 ORFs in the SPPV/GTPV lineage9. In another study, analysis of partial fragment of B22R gene showed a specific deletion in SPPV Romania comparatively to GTPV and LSDV62. Rouby et al. (2018) also detected 21-nucleotide deletion within RPO30 gene of SPPV in general63. Many studies have confirmed that GTPV is closely related to LSDV than SPPV is to LSDV64,65, which can explain the protection conferred to cattle vaccinated by GTPV Gorgan strain against LSDV. Additional studies are required to support our conclusions.
Capripox infections are now considered as emerging diseases which threaten new geographic areas (austral Africa and Europe). In most of the endemic countries, there are mixed flocks with goats and sheep living in promiscuity and, until now, there is no universal solution to avoid capripoxvirus diseases spreading except vaccination. The current study showed that goats and sheep vaccinated with Romania SP vaccine are well protected against a challenge with virulent GTPV and SPPV strains. However, Neethling strain did not protect sheep against the disease after challenge. Cattle vaccinated with Neethling strain showed full protection against LSDV virulent strain, while vaccination with SPPV strain did not give full protection against infection with the virulent LSDV virus. Preferably, vaccination should be conducted with the homologous Neethling strain.
Methods
Vaccines preparation
The SPPV vaccine used for the experiment was prepared in MCI laboratory. It is a commercial vaccine based on Romania strain of ovine origin, attenuated by Precausta et al.46 and manufactured by MCI, Morocco. The vaccine strain has been shown to provide strong protection of sheep beyond 24 months66. The strain was sequenced in Sciensano and the Full Genome Sequence (FGS) confirmed the identity of the virus (Unpublished data). For the antigen preparation, the master seed virus was passed three times on primary testis cells maintained in Dulbecco’s Modified Eagle’s Medium (DMEM) with 10% irradiated fetal calf serum. The inoculation was carried out using a Multiplicity of Infection (M.O.I) of 0.01. Viral suspension was harvested when 80% of cytopathic effect (CPE) was observed, generally at day 4 pi. The viral suspension was then stored at −80 °C before use. Sterility, identity, purity testing and titration were carried out as part of the quality control of the intermediate product. The live vaccine was prepared from the virus suspension by addition of the stabilizer (4% peptone, 8% sucrose and 2% glutamate) and freeze-dried in LSI lyodryer. The vaccine was tested for sterility, identity, purity and infectious titer. To vaccinate, we used an animal dose of 103.0TCID50 for small ruminants and 104.0 TCID50 for cattle.
The LSDV Neethling attenuated strain of South Africa origin was used to prepare the live attenuated LSDV vaccine in our laboratory. This strain has been attenuated through 61 passages on chorio-allantoic membrane and has been used as a vaccine strain for decades in Africa, Middle East and Europe recently52,67. The identity of the Neethling vaccine strain we used was confirmed by full genome sequencing (unpublished data). For the antigen preparation, the master seed virus was passed three times on primary testis maintained in Dulbecco’s Modified Eagle’s Medium (DMEM) with 10% irradiated fetal calf serum. The viral suspension was harvested after 5 days of incubation at 35 °C. Sterility, identity, purity and titration were performed as part of the quality control of the intermediate product. The vaccine was freeze-dried according the same protocol applied for SPPV. To vaccinate, we used an animal dose of 103.0 TCID50 for small ruminants and 104.0 TCID50 for cattle.
For SPPV and LSDV vaccine reconstitution, a sterile diluent based on phosphate saline solution was used. Vaccines were injected directly after reconstitution.
Vaccination with SPPV and LSDV vaccines
The experiment was conducted on animals housed in the high containment ABSL3. Animals were from Morocco local breeds, aged 6 to 8 months and tested negative by virus neutralization test (VNT) as described below.
Three groups of 4 sheep (G1), 8 goats (G2) and 13 cattle (G3) were subcutaneously (SC) vaccinated using Romania SPPV vaccine. Two groups of 8 sheep (G4) and 22 cattle (G5) were SC vaccinated using Neethling LSDV vaccine. All animals were monitored during 28 days for rectal temperature, clinical signs and inflammation at the injection site. Serum samples were collected weekly until D28 pv.
Challenge virus preparation
Infection was carried out according to international guidelines described for the care and handling of experimental animals, chapter 7.8 of the Terrestrial Animal Health Code and Directive 2010/63/UE of the European commission68,69. The protocol was submitted and approved by the Internal Laboratory Committee.
To prepare the challenge SPPV virus, we used the virulent SPPV Held strain, a Turkish strain, passaged on lamb testis cells56 and provided by IAV Institute in 2014. The virus was prepared by SC inoculation of 10 ml of viral suspension into 2 sheep flanks to obtain a giant pustule. After 13 days, sheep were euthanized to collect the subcutaneous inflammatory tissue at the injection site. The collected tissue was minced using sterile scissors and grounded after adding 2 volumes of PBS with 5% of antibiotics (penicillin and streptomycin). The suspension was centrifuged at 3500 RPM for 30 minutes. The supernatant was then aliquoted and stored at −80 °C for further use.
To prepare the challenge GTPV virus, we used the virulent GTPV Vietnam strain, originated from an infected goat lung10 and obtained from IAH Pirbright UK in 2012. Preparation of the challenge virus was the same as described above.
The virulent LSDV Israeli field isolate (106.5 TCID50/100 μl) was used to inoculate vaccinated and unvaccinated control cattle in the confined facilities of Sciensano laboratory in Belgium.
Challenge test
Vaccine potency testing was carried out by a challenge trial, under ABSL3 conditions using the virulent SPPV and GTPV strains for small ruminants. Vaccine potency testing on cattle was subcontracted to the European Reference Laboratory, Sciensano, in Belgium. Animal experiments were carried out in accordance with the international guidelines for care and handling of experimental animals.
Four sheep of G1 and 8 sheep of G4 were challenged at D28 pv with a virulent SPPV strain. Four goats from G2 were challenged at D28 pv with a virulent GTPV strain. Seven cattle from G3 and 7 cattle from G5 were challenged with a virulent LSDV strain. Two sheep, 2 goats and 5 cattle were kept unvaccinated and challenged by their respective virulent strains for challenge validation (Table 2).
Small ruminants were challenged using the protection index protocol56 which consists on a virus titration by intradermal injection of serial dilutions (10−1 to 10−6) on shaved flank, 5 inoculation points per dilution and 0.2 ml per injection in each animal. The obtained titer on vaccinated animals with average of the group is compared with the average titer obtained in unvaccinated animals. The protective index (PI) represents the difference between the 2 titers (in log). During the challenge trial, animals were daily observed for specific symptoms, temperature and local inflammation at the injection site. Serum samples were collected weekly for serology until D14 pi. Animals were all euthanized 2 weeks after infection (D14 pi); the control animals were euthanized when severe generalized symptoms appeared.
Cattle were challenged with virulent LSDV strain 21 days pv by intradermal (ID) (4 points of 250 µl each) and IV (2 ml) routes. Following inoculation, cattle were monitored for appearance of clinical signs during 21 days then euthanized. Clinical response in challenged animals was measured using clinical reaction score (CRS).
Laboratory testing
Vaccinated animals were monitored weekly for antibody response by VNT as described in the OIE Terrestrial Manual23,53. Blood was collected before and weekly after vaccination into dry tubes, which were centrifuged. Serum was then harvested and stored until use. Obtained sera were heat inactivated and serial 1:3 dilutions were mixed with a constant dose of SPPV or LSDV virus then incubated for one hour. Cell suspension was then added (OA3: ATCC® CRL-6546 or MBDK: ATCC® CCL22) and CPE was observed after 7 days incubation. Positive control without serum and negative without virus as well as cell control are introduced in the assay for validation. The neutralizing antibody titer was calculated according to Reed and Muench method70.
Organs of euthanized animals were analyzed using qPCR. DNA was extracted from the inflammatory tissue, using ISOLATE II Genomic DNA Kit (Bioline) following the manufacturer’s instructions. The extracted DNA was amplified using TaqMan Universal PCR Master Mix. The reactions were run on the Applied Biosystems 7500. The test was performed in 96-well Optical Reaction Plates using primers described by Bowden et al.71. Amplification conditions were: 95 °C during 10 min; and 45 cycles of 95° 15 s and 60 °C 1 min. Data were analyzed using the 7500 System software. Results were generated by determination of the threshold cycle (CT).
Statistical analysis
For statistical analysis, we started with the test of equality of variances (F-test) to check if the two treatments have the same variance, in order to choose the appropriate test for the average comparison. Where variances are equal, the test of equality of expectations would be used i.e. for two observations of equal variances. If not, the equality of expectations test for two observations of different variances would be used.
Differences between antibody titers obtained in vaccinated animals were tested for significance. Infectious and protective titers obtained post challenge in SPPV and GTPV vaccinated small ruminants and SPPV and LSDV vaccinated sheep were also tested for significance. Comparison of vaccinated and unvaccinated cattle was performed following clinical scoring obtained after challenge. Values of P ≤ 0.05 were considered significant.
Ethics and consent to participate
Animal experiments were carried out in accordance with the international guidelines for care and handling of experimental animals described in chapter 7.8 of the Terrestrial Animal Health Code and Directive 2010/63/UE of the European commission. The protocol was submitted and approved by the Internal Ethic Committee for animal experiment in MCI santé animale.
References
Yeruham, I., Yadin Van. Ham, M., Bumbarov, V., Soham, A. & Peri, S. Economic and epidemiological aspects of an outbreak of sheeppox in a dariy sheep flock. Vet. Rec. 160, 236–237 (2007).
Babiuk, S., Bowden, T. R., Boyle, D. B., Wallace, D. B. & Kitching, R. P. Capripoxviruses: an emerging worldwide threat to sheep, goats and cattle. Transboundary and Emerging Diseases 55, 263–272 (2008).
Bhanuprakash, V. et al. Animal poxvirus vaccines: a comprehensive review. Expert review of vaccines. 11, 1355–1374 (2012).
Tuppurainen, E. S. M. et al. Review: Capripoxvirus Diseases: Current Status and Opportunities for Control. Transboundary and Emerging Diseases 64, 729–745 (2017).
Hurisa, T. T., Jing, Z., Jia, H., Chen, G. & He, X. B. A Review on Sheeppox and Goatpox: Insight of Epidemiology, Diagnosis, Treatment and Control Measures in Ethiopia. J Infect Dis Epidemiol 4, 057, https://doi.org/10.23937/2474-3658/1510057 (2018).
World Animal Health Information Database (WAHID), 2019: https://www.oie.int/wahis_2/public/wahid.php/Diseaseinformation/Diseasetimelines (accessed 18 September 2019).
EFSA (European Food Safety Authority), Calistri, P. et al. Scientific report on lumpy skin disease: III. Data collection and analysis. EFSA Journal 17(3), 5638, https://doi.org/10.2903/j.efsa.2019.5638 (2019). 26 pp.
Tulman, E. R. et al. The genomes of sheeppox and goatpox viruses. J. Virol. 76, 6054–6061 (2002).
Biswas, S. et al. Extended sequencing of vaccine and wild-type capripoxvirus isolates provides insights into genes modulating virulence and host range. Transbound Emerg Dis. 00, 1–18, https://doi.org/10.1111/tbed.13322 (2019).
Babiuk, S. et al. Yemen and Vietnam capripoxviruses demonstrate a distinct host preference for goats comparend with sheep. J.Gen.Virol. 90, 105–114 (2009).
https://www.cabi.org/isc/datasheet/81537, accessed 26 September 2019
Brenner, J. et al. Appearance of skin lesions in cattle populations vaccinated against lumpy skin disease: statutory challenge. Vaccine 27, 1500–1503 (2009).
Tuppurainen, E. S. M. et al. Characterization of sheep pox virus vaccine for cattle against lumpy skin disease virus. Antiviral Research 109, 1–6 (2014).
Abutarbush, S.M. Lumpy Skin Disease (Knopvelsiekte, Pseudo-Urticaria, Neethling Virus Disease, Exanthema Nodularis Bovis). In: Bayry J (eds.) Emerging and Re-emerging Infectious Diseases of Livestock. Springer International Publishing, Gewerbestrasse 11, 6330 Cham, Switzerland, pp: 309-326. (2017)
Norian, R., Ahangaran, N. A., Varshovi, H. R. & Azadmehr, A. Evaluation of humoral and cell-mediated immunity of two capripoxvirus vaccine strains against lumpy skin disease virus. Iran J Virol. 10, 1–11, https://doi.org/10.21859/isv.10.4.1 (2016).
Norian, R., Afzal Ahagran, N., Varshovi, H. R. & Azadmehr, A. Comparative efficacy of two heterologous capripox vaccines to control Lumpy Skin Disease in cattle. Bulgarian Journal of Veterinary Medicine 22, 171–179 (2017).
Abutarbush, S. M. & Tuppurainen, E. S. M. Serological and clinical evaluation of the Yugoslavian RM65 sheep pox strain vaccine use in cattle against lumpy skin disease. Transbound Emerg Dis. 00, 1–7, https://doi.org/10.1111/tbed.12923 (2018).
Achour, H.A, Bouguedour, R., Bouhbal, A., Guechtouli, A., Aouissat, M. Comparative study of the immunogenicity of some attenuated strains of sheep pox virus and of a virus/immune serum vaccine. Rev. sci. tech. Off. int. Epiz (2000)
Quinn, P. J. et al. Veterinary microbiology and microbial disease, Wiley-Blackwell, Second edition (2011)
Teffera, M. & Babiuk, S. Potential of Using Capripoxvirus Vectored Vaccines Against Arboviruses in Sheep, Goats, and Cattle. Front Vet Sci. 6, 450 (2019).
Kitching, R. P. Vaccines for lumpy skin disease, sheep pox and goat pox. Dev Biol (Basel) 114, 161–7 (2003).
EFSA AHAW Panel (EFSA Panel on Animal Health and Welfare). Scientific Opinion on sheep and goat pox. EFSA Journal 12(11), 3885, https://doi.org/10.2903/j.efsa.2014.3885 (2014). 122 pp.
OIE: Sheep pox and Goat Pox. Manual of Diagnostic Tests and Vaccines for Terrestrial Animals. Chapter 3.7.12 (2018)
Rao, T. V. S. & Bandyopadhyay, S. K. A comprehensive review of goat pox and sheep pox and their diagnosis. Animal Health Research Reviews 1(2), 127–136 (2000).
Abd-Elfatah, E. B., El-Mekkawi, M. F. & Aboul-Soud, E. A. Capripoxviruses of small ruminants: control and evaluating the future update efficacy of a current vaccine in Egypt. Advances in Environmental Biology. 12(12), 11–16, https://doi.org/10.22587/aeb.2018.12.12.3 (2018).
Abu elzein, E. et al. Observations on natural and experimental infection of sheep and goats with a virulent field Capripoxvirus with high affinity to goats. Veterinary arshiv 73(3), 119–131 (2003).
Abu elzein, E., Housawi, F., Al-Afaleq, A. & Ibrahim, A. Protection of goats, with a Sheeppox Vaccine, against a virulent field Capripoxvirus with high affinity to goats. Scientific Journal of King Faisal University (Basic and Applied Sciences) 5(2), 1425 (2004).
Molla, W., Frankena, K., Gari, G., de Jong, M.C.M. Field study on the use of vaccination to control the occurrence of lumpy skin disease in Ethiopian cattle. Preventive Veterinary Medicine https://doi.org/10.1016/j.prevetmed.2017.08.019 (2017)
Abd-Elfatah, E. B., El-Mekkawi, M. F., Aboul-Soud, E. A., Fawzi, E. M. & El-Soally, S. A. Immunological response of a new trivalent capripoxvirus vaccine in pregnant ewes and does. Slov Vet Res 56(Suppl 22), 445–55 (2019).
Abutarbush, S. M. Efficacy of vaccination against lumpy skin disease in Jordanian cattle. Vet Rec. 175(12), 302 (2014).
Sevik, M. & Dogan, M. Epidemiological and Molecular Studies on Lumpy Skin Disease Outbreaks in Turkey during 2014-2015. Transboundary and Emerging Diseases 64, 1268–1279 (2016).
Abdallah, F. M., El Damaty, H. M. & Kotb, G. F. Sporadic cases of lumpy skin disease among cattle in Sharkia province, Egypt: Genetic characterization of lumpy skin disease virus isolates and pathological findings. Veterinary World 11(8), 1150–1158 (2018).
Zeedan, G. S. G. et al. Detection of lumpy skin disease virus in cattle using real-time polymerase chain reaction and serological diagnostic assays in different governorates in Egypt in 2017. Veterinary World, EISSN: 2231-0916 (2019).
Khafagy, H. A., Abdelwahab, M. G., Mustafa, A. M. & Saad, M. A. Preparation and field evaluation of live attenuated sheep pox vaccine for protection of calves against lumpy skin disease. Benha Veterinary Medical Journal 31(2), 1–7 (2016).
Varshovi, H.R., Norian, R., Azadmehr, A., Afzal Ahangaran, N. Immune response characteristics of Capri pox virus vaccines following emergency vaccination of cattle against lumpy skin disease virus. IJVST, 9; (2017)
Mikhael C.A., Ibrahim, M.M, Saad, M.A. Efficacy of Alternative Vaccination with Attenuated Sheep Pox and Inactivated Lumpy Skin Disease Vaccines against Lumpy Skin Disease. SCVMJ, XXI (2) (2016)
Mikhael, C. A., Nakhla, O. E. & Mohamed, N. A. Study on the capability of a dual capripox vaccine in protection of cattle against LSD infection. Journal of veterinary medical research 24(1), 224–233 (2017).
Aboul-Soud, E.A., Kafafy, M.H., El-Suall, S.A., Zaghloul, M.A., Mikhael, C.A. Preparation of trivalent vaccine against lumpy skin disease using different capripox viral strain. International J. of Vet. Science and Medicine (inpress) (2018).
Agrawal, S. K. & Soman, J. P. Assessment of immune efficacy of attenuated live goat pox vaccine against sheep pox and contagious ecthyma. Indian Vet.J 74, 1016–18 (1997).
Coackley, W. & Capstick, P. B. Protection of cattle against lumpy skin disease. Res. Vet. Sci. 1961 12, 123–127 (1961).
Gari, G. et al. Evaluation of the safety, immunogenicity and efficacy of threecapripoxvirus vaccine strains against lumpy skin disease virus. Vaccine 33, 3256–3261 (2015).
Salib, A. F. & Osman, A. H. Incidence of lumpy skin disease among Egyptian cattle in Giza Governorate, Egypt. Veterinary World 4(4), 162–167 (2011).
Ayelet, G. et al. Lumpy skin disease: preliminary vaccine efficacy assessment and overview on outbreak impact in dairy cattle at Debre Zeit, central Ethiopia. Antiviral Res 98, 261–265 (2013).
Tassew, A., Assefa, A., Gelaye, E., Bayisa, B. & Ftiwi, M. Identification and Molecular Characterization of Lumpy Skin Disease Virus in East Hararghe and East Shoa Zone. Oromia Regional State” ARC Journal of Animal and Veterinary Sciences 4(3), 1–16 (2018).
Ben-Gera, J., Klement, E., Khinich, E., Stram, Y. & Shpigel, N. Y. Comparison of the efficacy of Neethling lumpy skin disease virus andx10RM65 sheep-pox live attenuated vaccines for the prevention of lumpy skin disease – The results of a randomized controlled field study. Vaccine 33(38), 4837–4842, https://doi.org/10.1016/j.vaccine.2015.07.071 (2015).
Precausta, P., Kato, F. & Vellut, G. A new freeze-dried living virus vaccine against sheep-pox. Comp Immunol Microbiol Infect Dis 1, 305–319 (1979).
Fakri, F. et al. Development and field application of a new combined vaccine against Peste des Petits Ruminants and Sheep Pox. Trials Vaccinol 4, 33–37 (2015).
Mirzaie, K., Barani, S. M. & Bokaie, S. A review of sheep pox and goat pox: perspective of their control and eradication in Iran. J. Adv. Vet. Anim. Res. 2(4), 373–381 (2015).
Sinitsyna, O. A. et al. Further-attenuated measles vaccine: Virus passages affect viral surface protein expression, immunogenicity and histopathology pattern in vivo. Research in VirologyVolume 141(Issue 5), 517–531 (1990).
Babiuk. et al. Vaccines Against LSD and Vaccination Strategies in Lumpy Skin Disease (2018) https://doi.org/10.1007/978-3-319-92411-3_12
Tuppurainen, E. S. M. et al. Field observations and experiences gained from the implementation of control measures against lumpy skin disease in South-East Europe between 2015 and 2017, Preventive Veterinary Medicine (2018), https://doi.org/10.1016/j.prevetmed.2018.12.006
Davies, F. G. Lumpy skin disease of cattle: a growing problem in Africa and the Near East. World Anim. Rev. 68, 37–42 (1991).
OIE: Lumpy skin disease. Manual of Diagnostic Tests and Vaccines for Terrestrial Animals. Chapter 3.4.12 (2018)
Bhanuprakash, V., Indrani, B. K., Hegde, R., Kumar, M. M. & Moorthy, A. R. S. A classical live attenuated vaccine for sheep pox. Trop Anim Health Prod. 36, 307–20 (2004).
Boumart, Z. et al. Comparative innocuity and efficacy of live and inactivated sheeppox vaccines. BMC Vet. Res. 12(1), 133, https://doi.org/10.1186/s12917-016-0754-0 (2016).
Fassi-Fehri, M., El-Harrak, M., Johnson, D., Abbadi, M. & El-Idrissi, A. H. Etude expérimentale de l’immunité anticlaveleuse post-vaccinale. Annales de Recherches Vétérinaires, INRA Editions 15(1), 59–64 (1984).
Tilahun, Z., Berecha, B., Simenew, K. & Reta, D. Towards effective vaccine production: a controlled field trial on the immunological response of three lumpy skin disease vaccine strains in dairy farms. Academic Journal of Animal Diseases 3(3), 14–26 (2014).
Milovanović, M. et al. Humoral immune response to repeated lumpy skin disease virus vaccination and performance of serological tests. BMC Veterinary Research 15, 80, https://doi.org/10.1186/s12917-019-1831 (2019).
Michael, A. et al. Control of lumpy skin disease outbreak in Egypt with Romanian sheep pox vaccine. Assiut Veterinary Medical Journal 36, 173–180 (1997).
Osuagwuh, U. I., Bagla, V., Venter, E. H., Annandale, C. H. & Irons, P. C. Absence of lumpy skin disease virus in semen of vaccinated bulls following vaccination and subsequent experimental infection. Vaccine 25, 2238–2243 (2007).
Annandale, C. H., Irons, P. C., Bagla, V. P., Osuagwuh, U. I. & Venter, E. H. Sites of persistence of lumpy skin disease virus in the genital tract of experimentally infected bulls. Reprod. Domest. Anim. 45, 250–255 (2010).
Chibssa, T. R. et al. An HRM Assay to Differentiate Sheeppox Virus Vaccine Strains from Sheeppox Virus Field Isolates and other Capripoxvirus Species. Scientific Reports 9, 6646, https://doi.org/10.1038/s41598-019-43158-x (2019).
Rouby, S.R. RPO30 Gene based PCR for Detection and Differentiation of Lumpy Skin Disease Virus and Sheep Poxvirus Field and Vaccinal Strains. Veterinary Sciences: Research and Reviews, 4(1): 1-8.
Lamien, C. E. et al. Real time PCR method for simultaneous detection, quantitation and differentiation of Capripoxviruses. Journal of Virological Methods 171, 134–140 (2011).
Le Goff, C. et al. Capripoxvirus G-protein-coupled chemokine receptor: a host-range gene suitable for virus animal origin discrimination. Jounal of general virology, volume 9, issue 8 (2009)
Kitching, R. P. Passive protection of sheep against capripoxvirus. Res Vet Sci. 2, 247–5 (1986).
Klement, E. et al. Neethling vaccine proved highly effective in controlling lumpy skin disease epidemics in the Balkans. Preventive Veterinary Medicine, https://doi.org/10.1016/j.prevetmed.2018.12.001 (2018)
Directives EU Commission. Protection des animaux utilisés à des fins scientifiques. J Off l’Union Eur. 276, 1–162 (2010).
OIE. Terrestrial Animal Health Code. Use of Animals In Research And Education. In: Chapter 7.8, OIE Terrestrial Animal Health Code; 1 –10 (2016).
Reed, L. & Muench, H. A simple method of estimating fifty percent endpoints. Amer Jour Hyg 27, 493 (1938).
Bowden, T. R., Babiuk, S., Parkyn, G. R., Copps, J. S. & Boyle, D. B. Capripoxvirus tissue tropism and shedding: a quantitative study in experimentally infected sheep and goats. Virologyn 371, 380–393 (2008).
Acknowledgements
All authors gratefully acknowledge Dr Kris DeClerq, Dr Andy Haegman and Sciensano team for performing challenge trials and laboratory analysis. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
J.H. and Z.Ba. carried out the experiment, M.J. performed the molecular biology analysis, J.H. performed serological analysis, participated in data analysis and interpretation and wrote the manuscript, Z.Bo. reviewed the manuscript, K.T. and O.F. participated in the design and the follow up of the study, M.E. participated in the design of the study, manuscript drafting, data analysis and interpretation. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hamdi, J., Bamouh, Z., Jazouli, M. et al. Experimental evaluation of the cross-protection between Sheeppox and bovine Lumpy skin vaccines. Sci Rep 10, 8888 (2020). https://doi.org/10.1038/s41598-020-65856-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-65856-7
This article is cited by
-
An insight into emergence of lumpy skin disease virus: a threat to Indian cattle
Archives of Microbiology (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.