Abstract
Although shedding of zoonotic brucellae in milk has been demonstrated in natural hosts, these data are still missing for the standard murine infection model. We therefore analysed shedding kinetics and the niche of B. melitensis in murine milk. Pregnant Balb/cByJ mice were intraperitoneally infected with 105 CFU of the 16 M reference strain, a 16 M mCherry mutant or a human isolate. Milk was collected over the course of lactation, and subjected to culture and immunofluorescence assays. Bacteria were also quantified in spleen and mammary glands of maternal mice and in spleen of the litter. The shedding of the three strains did not differ significantly (p = 0.301), ranging from log10 1.5 to 4.04 CFU/ml. A total of 73% of the mice excreted B. melitensis into the milk with peak values at mid-lactation; up to 30 bacteria/cell were found in macrophages and neutrophils. While the bacterial counts in the spleen of lactating females confirmed a well-established infection, only 50% of the pups harboured brucellae in their spleen, including the spleen of an uninfected pup fed by an infected foster mother. In conclusion, the murine model of infection may contribute to a better understanding of the zoonotic transmission of brucellosis.
Similar content being viewed by others
Introduction
Brucellosis is a severe, though neglected, zoonosis with widespread distribution and global impact1. The consequences on public health, animal health and economy are considerable, especially in endemic developing countries, but also in developed countries where the infection is re-emerging in wildlife2. The causative agents are facultative intracellular Gram-negative bacteria of the genus Brucella, including twelve species, of which B. melitensis is responsible for the main burden of disease in humans. Brucella melitensis is mainly isolated and transmitted from sheep and goats3.
In the natural host, brucellae have a predilection for both female and male reproductive organs in sexually mature animals based on immune, hormonal and metabolic factors, including the availability of erythritol. This polyhydric alcohol can be found in high concentrations in placenta, mammary gland and epididymis of most host species, but less in mice and men4. Brucellae enter, replicate and survive in both phagocytic and non-phagocytic cells; macrophages, dendritic cells and trophoblasts are their major target cells. Brucella spp. actually find excellent shelter in macrophages for their spread (shuttle), while evading the immune response of the host (stealthy organism). Replication in trophoblastic cells is strongly influenced by the stage of pregnancy; it increases in late gestation, when the cells actively secrete steroid hormones5. The replication in trophoblasts compromises the integrity of the placenta and shapes in consequence the clinical picture, represented by abortion and reduced fertility in the natural host. Infected animals shed the organisms in high concentrations in uterine discharges after abortion or parturition, and also intermittently or continuously in the colostrum and milk3. Due to the shedding of brucellae in milk, Brucella-contaminated raw milk and unpasteurized dairy products are the most important vehicles of human infection and a considerable public health risk even in non-endemic countries6. In goats infected during pregnancy, Higgins et al. reported shedding of B. melitensis 16 M in milk postpartum (pp); about half of the animals were high level shedders revealing bacterial counts of 104–107 CFU/ml in milk and the others shed at lower concentrations with a declining trend shortly postpartum7. Brucella melitensis was isolated periodically from the milk of ewes up to three years after infection8. Cows shed Brucella abortus up to nine years after infection9. In infected water buffalos, B. abortus was recovered from milk in concentrations up to 104 CFU/ml throughout the lactation period10. The presence of Brucella suis was demonstrated in the milk of pigs as well11. Last but not least, even human milk may contain brucellae and was proven to be contagious to infants12,13.
Mice are the classical infection models widely used for studying the various aspects of pathogenesis after the infection with Brucella, including vertical and pseudo-vertical transmission scenarios14. Mice develop a discoid placenta with a flat surface facing the foetus and an irregular opposite surface adjacent to the uterine wall. The foetal labyrinthine vessels constitute the interhaemal barrier with three trophoblast layers, of which the outer one is cellular and the inner two are syncytial, forming a haemochorial placenta where the foetal chorion is in direct contact with the maternal blood15. Due to the structural similarity to the haemochorial placenta of women, mice are the most frequently used animal model for placenta and pregnancy research in human medicine16. In contrast, ruminants develop a synepitheliochorial placenta type with 100 to 140 cotyledons, which attach to maternal preformed uterine caruncles to form placentomes. Foetal villous trees consist of an epithelial surface layer, the chorionic epithelium or trophoblast, and a mesenchymal core carrying branches of allantochorionic blood vessels, and interdigitate with maternal crypts. Owing to the synepitheliochorial structure, foetal and maternal vascular systems remain completely separated from each other17.
In contrast, the histological structure of the mammary gland is similar in most mammalian species18. The functional unit of the mammary gland in all of these species is the alveolus, a hollow sphere surrounded by an embracing blood system. Epithelial cells synthesize and secret milk apically into the alveolar lumen19. The alveoli open into secondary mammary tubules, which form the mammary duct20. The blood-milk barrier inhibits the leakage of milk components from the luminal side into the blood due to epithelial tight junctions. During infection, the integrity of the alveolar epithelium is compromised and therefore enables the flow of aqueous molecules, ions, water, and microbes21. Pathogens will find ideal conditions to multiply, once they are inside the gland. Here, they trigger immunologic responses because of the damage of mammary alveoli and ductal epithelium. Whereas contagious or environmental mastitis results from an ascending infection through the teat canal, the subclinical mastitis due to brucellosis is caused by haematogenous dissemination of the pathogen22,23.
Neither the shedding of Brucella, nor its histopathological impact on the mammary gland, nor, and most importantly, its cellular niche in milk has yet been studied in the murine model of infection. We therefore characterized the quantitative burden and the kinetics of shedding in murine milk of experimentally infected Balb/cByJ mice. Our main objectives were, (i) to quantify the shedding of B. melitensis in murine milk, (ii) to evaluate the pathology and the nature of colonization resulting from infection in mammary glands, and (iii) to identify the cellular niche (if any) of B. melitensis in murine milk and mammary glands.
Results
Neither the infection with (i) the B. melitensis mCherry mutant strain (mCh), nor with (ii) the B. melitensis 16 M reference strain (16 M), nor with (iii) the human B. melitensis isolate (FS) resulted in changes of behaviour or food intake. In addition, body temperature was not elevated and abortions could not be recorded. Finally, five mother mice in the mCh group and seven mother mice each in the 16 M and FS group and their litters were included in the study. The experiment was terminated in accordance with the ethical protocol when the predefined end point was reached by the pups (less than 50% weight compared to the control litter). The loss of weight coincided with insufficient milk production of the dam (<10 µl per sample).
B. melitensis is shed in murine milk
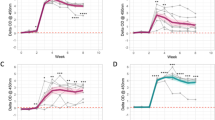
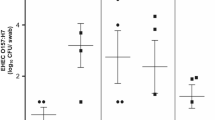
To assess the presence of B. melitensis in murine milk, pregnant Balb/cByJ mice were inoculated i.p. with 2 × 105 CFU B. melitensis and milked at several points in time postpartum. In the mCh group, 80% of the mice (4/5) shed B. melitensis in milk, whereas in the 16 M group and in the FS group, 70% of the mice (5/7) in each group shed B. melitensis in milk. The quantitative results did not differ significantly among groups (p = 0.301) (Fig. 1). Therefore, the three group results were merged (mCh: mice no. 1–5, 16 M: mice no. 6–12, FS: mice no. 13–19, resulting in 19 mother mice altogether) and further statistical analyses were run together, regardless of the strain used for infection.
The majority of mice shed B. melitensis intermittently
A total of 73% of the mice (14 out of 19) shed B. melitensis in their milk. The majority of mice were milked 3 to 4 times (15/19, 79%), three mice 5 to 7 times (16%) and one mouse only once (5%).
About half of the mice (9/19) revealed bacterial shedding in milk between log10 1.5 and 2.9 CFU/ml. A total of 26% of the mice (5/19; mice no. 3–5, 8 and 14) showed at least once high shedding rates (≥log10 3 CFU/ml). It is noteworthy that 26% of the mice (5/19) did not excrete Brucella at any time during lactation. The individual kinetics usually showed intermittent shedding (79%, 11/14 mice) and merely three persistent shedders (16%) (Table 1). Taking the 76 milk samples collected from all mice together, 51.3% were negative (39/76), 39.5% (30/76) revealed a moderate contamination with Brucella between log10 1.5 and 2.99 CFU/ml and seven milk samples (9%) exceeded log10 3 CFU/ml. Positive milk samples were most frequently recorded from day 6–7 pp onwards with a maximum at day 10 pp (71.4%, 5/7 mice). The shedding rate showed the highest mean (1.65 CFU/ml) at day 12 pp (Fig. 2). However, the time effect of this qualitative and quantitative rise of the shedding comparing the seven defined sampling time points was non-significant (p = 0.2182).
B. melitensis was localized in murine milk macrophages and PMN
Somatic cell counts in the milk of shedding mice were slightly increased compared to control mice and dominated by macrophages and PMN, whereas lymphocytes and epithelial cells were only sporadically found. Macrophages and PMN often contained lipid globules and amorphous globular protein (Fig. 3A). Brucella melitensis was mainly found in macrophages with considerable numbers up to 30 bacteria/cell and in PMN with up to 15 bacteria/cell (Fig. 3B). In contrast, brucellae were rarely found extracellularly and free-floating without association to cells in the milk. The cellular localization of B. melitensis did not differ between the various sampling time points.
After intraperitoneal inoculation of B. melitensis in pregnant mice, the bacteria were found in murine milk macrophages and neutrophils. Extracellular brucellae sticked to the surface of epithelial cells. (A) Somatic cell counts in murine milk revealed 60% macrophages, 25% lymphocytes, 15% neutrophils, and a few epithelial cells (light microscopy, 100x oil immersion). (B) Milk samples were analysed using confocal microscopy for cell specific marker antigens, namely F4/80 (macrophages), CD3 (lymphocytes), Ly6G (PMN) and pan-keratin (C11, epithelial cells). The microscopic images enabled the resolution of nucleic acids stained by DAPI (blue), cell antigens (green) and Brucella (red).
The mammary glands and the litter inconsistently harboured B. melitensis
After intraperitoneal inoculation of B. melitensis, bacteria were isolated from the spleen of all mice (mean: log10 4.6 CFU/g). In 5 out of 19 (26%) lactating females, B. melitensis was detected in the mammary gland. In contrast, 9 out of 10 (90%) females that lost their entire litter due to infanticide and were sacrificed shortly postpartum (according to our study protocol) harboured Brucella in the mammary gland (mean: log10 2.14 CFU/g (data not shown)). The average litter included 4 pups and about half of the pups were infected which was diagnosed by the bacterial load in the spleen. However, the prevalence of positive pups in a litter ranged from 0 to 100%. One of the infected pups originated from an uninfected foster mother of the control group but was adopted and fed for five days by an infected mother (mouse no. 17, strain FS) (Table 2).
B. melitensis was localized in resident macrophages in the mammary gland
Histologically, neutrophilic and lymphocytic cell infiltrations were observed in the interstitial tissue of the mammary glands that extended into the muscular layers resulting in a mild, subacute, interstitial mastitis. At the end of the lactation period (time of sacrifice), the mammary tissue was moderately atrophic and showed a lobular inflammation with infiltrating macrophages, lymphocytes and PMN. In case of residual milk in the alveolar lumen, coccobacilli were found in the milk macrophages. The milk duct epithelium and periductal stroma was flattened and marked by a squamous meta- and hyperplasia, and was also infiltrated with lymphocytes and macrophages. The lumen of the ducts contained PMN, lymphocytes, macrophages and cell debris. Figure 4A shows the histopathology of a mammary duct stained with HES. In the ductal lumen, intracellular coccoid bacteria were found (Fig. 4B). Furthermore, specific and localized foci of non-cell associated brucellae in the lumen of mammary ducts were visible (Fig. 4C). Immunofluorescence also showed Brucella in macrophages in the mammary tissue (Fig. 4D).
Histopathology and immunohistochemistry of murine mammary tissue infected with B. melitensis. (A) HES staining of a teat showing focal interstitial infiltration of lymphocytes, macrophages and neutrophils (10×). (B) HES staining of a murine mammary gland showing a lactiferous duct filled with milk, containing lymphocytes, macrophages and neutrophils as well as intracellular coccobacilli (10×) marked by blue arrows. (C) Immunohistochemistry of the mammary gland showing immunolabeled extracellular B. melitensis (10×). (D) Fluorescence immunolabeling of mammary tissue, analysed by confocal laser scanning microscopy, localized Brucella (red) in macrophages (green, F4/80).
Discussion
We characterized the kinetics of B. melitensis shedding in murine milk after experimental infection during gestation. Furthermore, we identified the cellular niche of the bacteria in the milk and evaluated histological changes in the mammary gland.
Shedding kinetics and cellular niche of B. melitensis in murine milk
Though several studies on Brucella transmission already quantified the bacterial load in milk and the mammary gland of the target species, we present here the first report on the isolation and quantification of Brucella from murine milk in the standard model of infection. Among 19 B. melitensis infected mice, 14 (73%) shed the pathogen in their milk. About half of the mice (9/19) revealed moderate contamination loads of the milk during the lactation period, whereas 26% (5/19) of the mice were high level shedders with >log10 3 CFU/ml. It was previously reported, that water buffalos naturally infected with B. abortus heterogeneously shed brucellae in milk. Most of the animals shed at low levels, but 16% of the infected buffalos were classified as “super-spreaders” since they consistently shed high Brucella concentrations in the milk with ≥ log10 4 CFU/ml10.
In our infection model, we observed that more mice were shedding towards mid-lactation and the total burden of brucellae in milk peaked quantitatively at day 10 pp. In contrast, goats excrete B. melitensis most frequently at the beginning of the lactation period7. Though the kinetic trend was obvious in our study, the smaller sample sizes at the beginning and at the end of the sampling period, leading to lower volumes of murine milk, may have biased the results. Moreover, we worked exclusively with nulliparous females. In mammals, nulliparous females tend to provide less milk during their first lactation than multiparous females. Without continuous monitoring of Brucella concentrations in milk during subsequent lactation periods, it is difficult to say whether multiparous females still shed Brucella in their milk, and if so, in which quantity. In sheep, experimentally infected with a single shot of B. melitensis, shedding in milk persisted for at least three reproductive cycles post-infection and the percentage of sheep shedding Brucella in milk was 79%, 64%, and 38% over the first, second, and third reproductive cycle post-infection, respectively8.
Brucella was localized in murine milk macrophages and PMN, with up to 30 bacteria per cell. Macrophages derive from blood monocytes, once entering tissues they promote both innate and acquired immune responses. The innate functions of macrophages include acidification and destruction of the phagocytosed bacteria by reactive oxygen and nitrogen species24. Brucella, however, can reside in macrophages which actually should eliminate the invading bacteria. According to our understanding, Brucella-containing phagocytes migrate from the blood stream to the mammary alveolus due to the compromised blood-milk-barrier and subsequently into the milk. This contamination pathway was already suggested for goats22. PMN are the second line of immune defence in mammary gland infections; their numbers are up to 50 times higher during the first hours of mammary gland infection in bovines25. Similar to macrophages, PMN phagocyte and kill bacteria but PMN also phagocyte milk fat globules22,26. We confirmed the presence of fat globules in PMN and also in murine milk. Extracellular brucellae were seen rarely in the milk and were most likely leftovers of lysed phagocytic cells. Brucellae were not detected in lymphocytes or epithelial cells, although Brucella is known to persist in uterine epithelial cells27 and is able to replicate in murine B lymphocytes28.
Histopathology and tissue burden of spleen and mammary gland
No clinical symptoms of brucellosis were observed in pregnant mice although bacteriological results revealed 100% infection of the females with high splenic tissue burden. Demars and colleagues demonstrated, that intraperitoneal inoculation results in a widely disseminated infection in mice affecting almost every organ29. In our study, Brucella could be isolated from the spleen of all mice, but the bacteriological results of the mammary glands varied. Brucella melitensis was merely isolated from the mammary gland of 5 out of 19 lactating mice, of which one mouse (no. 17) did not even shed the pathogen. The low isolation rate from mammary tissue of breastfeeding mice compared to the high prevalence of Brucella-positive mammary glands in non-breastfeeding mice (90%) suggests that the recovery of brucellae from the mammary tissue substantially depends on the residual milk volume in the gland. Meador et al. studied the effect of nursing on the quantity of B. abortus in the mammary tissue of goats30. Non-nursed mammary glands revealed high titres of brucellae in milk, moderate interstitial mastitis, and brucellar antigen in macrophages primarily located in alveolar and ductal lumina. In contrast, nursed mammary glands showed fewer brucellae in milk, minimal inflammatory changes, and no detectable brucellar antigen in histological sections30. We therefore conclude that bacterial shedding in murine milk is usually more frequent and constant than the occurrence of Brucella in the mammary gland or mammary lesions. It was shown that B. abortus was more often isolated from milk of infected cows (12/21 cows; 57%) than from the mammary tissue (8/21 cows; 38%)23. Histologically, the lesions in the mammary gland of mice mainly corresponded to the lesions described in target species. Brucella abortus caused only mild lesions in the mammary glands of goats such as multifocal to diffuse, mild interstitial mastitis with infiltration of macrophages27,30. This was recently confirmed in cattle23, showing that cows infected with B. abortus developed a mild interstitial inflammatory reaction in the mammary gland. However, we further observed atrophied, squamous ductal epithelium which might have been caused by the mechanical manipulations of the miniscule teats of mice during milking.
Despite of the high prevalence of B. melitensis in milk and mammary glands of mice, only 50% of their pups were found to be infected. Interestingly, the antibody titres we measured in the lactoserum (data not shown) revealed no differences regardless of the presence of Brucella in milk or the infectious status of the pups. The incomplete transmission of virulent B. abortus 544 from pregnant mice to the foetus in utero was already demonstrated31. Furthermore, vaccination reduced the amount of infected pups in this study by 50% which was linked to a placental barrier protection mediated by T- and B- lymphocytes31.
We recorded the infection of a primarily non-infected pup which has been suckled by an infected foster mother for five days. Bosseray et al. showed that the infection of infant mice, born by non-infected dams but drinking the milk from infected foster mothers, is a rare event32. Although the mammary glands of nursing mice were colonized with Brucella, less than one percent (1/114) of the pups was infected32. It remains to be elucidated, why the transmission to murine offspring is incomplete and which conditions finally trigger the infection of the foetus in utero and of the pups. Pseudo-vertical transmission through colostrum or milk12 and vertical transmission in utero33 may also occur in human brucellosis cases, but these kinds of transmission are rare events although placentation in mice and men is similar.
Conclusions
Our results closed some of the knowledge gaps on brucellosis in the murine model of infection. We characterized the shedding pattern of B. melitensis in milk peaking in the mid-lactation period and clearly identified the cellular niche of B. melitensis in milk macrophages and neutrophils. The colonization of the mammary gland by Brucella using blood macrophages as a vehicle resulted in a mild interstitial mastitis. Even though, the research on murine brucellosis has its own value, the mouse model may also contribute to further exploration of zoonotic brucellosis mainly transmitted from animals to men through the consumption of unpasteurized dairy products.
Material and Methods
Ethical approval
The experimental setting and the procedures in this study were in accordance with current European legislation (Council Directive 86/609/EEC) and the corresponding Belgian law “Arrêté royal relatif à la protection des animaux d’expérience du 6 avril 2010 publié le 14 mai 2010”. The Animal Welfare Committee of the Université de Namur (Belgium) reviewed and approved the complete protocol (number of application approval: GIN 18-308). Animals were monitored daily and all experiments were performed under biosafety level 3 containment according to Council Directive 98/81/EC of 26 October 1998.
With limited food resources, the number of pups per litter declines (Weber and Olsson, 2008). We therefore improved postpartum conditions, and optimized the diet of the mothers with high protein (12.5–18%) and lipid contents (5%), supplemented with calcium and phosphorus (0.4%), vitamin A (500 IU/kg) and vitamin D (150 IU/kg). To reduce maternal stress and increase pup survival, the number of pups was limited to a maximum of 5 per litter. In case a litter included fewer pups, adoptions were carried out as described by Weber and Olsen34.
Bacterial strains and mice
Wild-type SPF Balb/cByJ mice aged 8–12 weeks were purchased from Charles River Laboratories (Ecully, France). Females were synchronized three days on the bedding of males. Two females and one male were housed together at a time. After five days of mating, the male was removed. Gestation was determined manually starting from day 11. At day 14 of the gestation period, the pregnant mice were inoculated intraperitoneally (i.p.) with 2 × 105 CFU B. melitensis in 500 μl phosphate buffered saline (Gibco PBS, ThermoFisher Scientific, Merelbeke, Belgium). In the first group, a mutant strain of Brucella melitensis 16 M was used, which steadily expresses the fast maturing variant of the red fluorescent protein DsRed (mCherry, mCh)35,36. In the second group, the unmodified B. melitensis 16 M reference strain (16 M) was used. In the third group, the field strain B. melitensis L311/15 (FS) isolated from a Turkish patient in Belgium was used37. Bacteria were grown in biosafety level 3 laboratory facilities overnight in 2YT media (Luria-Bertani broth with double quantity of yeast extract) shaking at 37 °C, and were washed twice in PBS (3,500 rpm, 10 min) before inoculating the mice. The control animals were inoculated with the same volume of PBS. The infectious doses were validated by plating serial dilutions of the inoculum. Those females that lost their litter due to infanticide as well as the females that resorbed the foetuses in utero were sacrificed for bacteriology of the spleen and mammary gland.
Bacteriological quantification of B. melitensis in murine milk and tissue
Mice were milked, in group mCh, between day 2 and day 17 pp, in group 16 M between day 6 and day 13 pp, and in group FS between day 7 and day 14 pp according to the protocol of Gómez-Gallego et al.38 with slight modifications based on Muranishi et al.39. Briefly, females were separated from the litter for 6 h, and after exogenous oxytocin stimulation (2.5 IU i.p.), the females were anesthetized for 35–45 min with a cocktail of xylazine (10 mg/kg) and ketamine (100 mg/kg) in sterile saline solution administrated i.p. (0.1 ml/10 g body weight). Milk was collected manually and milk volume was recorded. The milking frequency was adapted from daily to every other day after the mCh group, because of the high mortality rate among pups. Moreover, the milk volume taken was limited to a maximum of 150 µl/milking. In accordance with ISO 4833-2:2013, milk samples were initially diluted tenfold in 2YT. For quantification, 100 µl of serial decimal dilutions of the milk were plated in duplicate on tryptone agar containing antibiotic supplement (Brucella Selective Supplement SR83; ThermoFisher Scientific) and 10% equine serum, known as Farrel’s medium. Plates were incubated for up to 12 days at 37 °C under aerobic conditions and controlled for bacterial growth every 48 h. Typically, bacterial colonies were visible within the first five days. To avoid false-negative results in the quantification process, an enrichment step was additionally performed. Remaining milk samples were incubated for 24 h at 37 °C and another 100 µl were plated on Farrel’s medium and incubated as described above. Brucellae were identified phenotypically40.
At the end of the experiment, females were sacrificed by double cervical dislocation. The litter was sacrificed by a lethal overdose of isoflurane (IsoFlo w/w 100% liquid, Zoetis, Zaventem, Belgium). The spleen and the second thoracic mammary complex were taken aseptically, homogenized in PBS/0.1% X100 Triton (Sigma-Aldrich, Machelen, Belgium) and plated in decimal serial dilutions in duplicates on Farrell’s medium.
Milk cytology
The remaining milk was centrifuged at 14,000 rpm for 15 min. The supernatant was removed and the lactoserum in the intermediate phase separated for further analysis. The milk cell pellet was prepared for a Kieler sediment smear. Cells were fixed overnight at 4 °C in 4% formaldehyde at pH 7.4 (VWR, Leuven, Belgium), washed twice in PBS and re-suspended in 0.1 ml PBS. A total of 10 µl of the cell suspension was spread in a monolayer over a 3 cm2 field on a microscope slide41 and stained with Giemsa (Sigma-Aldrich, Overijse, Belgium) for light microscopy. Up to 100 cells, including macrophages, epithelial, lymphocytic and neutrophilic cells, were identified and counted at a 1000-fold magnification.
Histology
The mammary glands were fixed in 4% formaldehyde at 4 °C for 24 h, and washed twice in PBS for 30 min. Organs were dehydrated and embedded in paraffin. Tissue sections (6 µm) were mounted on SuperFrost microscope slides (ThermoFisher Scientific). Serial sections were stained with haematoxylin eosin saffron (HES) according to standard protocols.
For immunohistochemistry and immunofluorescence staining, tissue sections were dewaxed. Immunohistochemistry was performed as described by Nicaise et al.42. In brief, after the sections had been rehydrated with PBS, endogenous peroxidase activity was inhibited with 3% hydrogen peroxide for 10 min and blocked for 1 hour using 5% normal serum. Slides were then incubated with a rabbit anti-Brucella serum (1/500) for 1 h at room temperature, followed by a biotinylated secondary anti-rabbit antibody (1/100, VECTASTAIN), visualized with avidin-horseradish peroxidase (1/200, VECTASTAIN) and diaminobenzidine with haematoxylin counterstaining, and finally mounted with DPX mounting medium for histology (Merck, Darmstadt, Germany). Immunofluorescence was performed on monolayer milk smears and tissue sections as described by Demars et al.29. In brief, slides were blocked for 20 min and incubated for 1 h with one of the following mAbs or reagents: FITC-coupled 1.45-2C11 (anti-CD3, eBiosciences, 1/200), FITC-coupled 1A8 (anti-Ly6G, Biolegend, 1/200), FITC-coupled Pan-Keratin (anti-C11, CellSignalling Technology, 1/200) or PE-coupled BM8 (anti-F4/80, eBiosciences, 1/200) for cells. Bacteria were stained simultaneously with a rabbit anti-Brucella serum (1/500). The secondary anti-rabbit antibody was coupled to Alexa-647 (Life Technologies, 1/400). Slides were mounted with ProLong Antifade containing DAPI (ThermoFisher Scientific). Labelled tissue sections were visualized with an Axiovert M200 inverted microscope (Zeiss, Jena, Germany) equipped with a high-resolution monochrome camera (AxioCam HR, Zeiss). Images (1,384 × 1,036 pixels, 0.16 μm/pixel) were taken sequentially for each fluorochrome with A-Plan 10x/0.25 N.A. and LD-Plan-NeoFluar 63x/0.75 N.A. dry objectives and recorded as eight-bit grey-level.zvi files. Three slides per organ from three different animals were analysed resulting in representative images for the experiment.
Statistics
Quantitative data were recorded, processed and analysed in Microsoft Excel and SPSS. The non-parametric Kruskal-Wallis test was applied to detect statistical differences between the strains and their shedding in milk and tissue. The frequencies of contamination and bacterial loads per milking time point, compiled for milking day 1–3 pp (time point 1), day 4–5 pp (time point 2), day 6–7 pp (time point 3), day 8–9 pp (time point 4), day 10–11 pp (time point 5), day 12–13 pp (time point 6) and day 14–17 pp (time point 7), were analysed by the FREQ procedure and subjected to a Jonckheere-Terpstra test for associations between pathogen load and time point of milking using a significance level of 0.05. Data were plotted in Excel and Prism GraphPad.
References
Adone, R. & Pasquali, P. Epidemiosurveillance of brucellosis. Rev. sci. tech. (Off. int. Epiz.) 32(1), 199–205 (2013).
Lambert, S. et al. High shedding potential and significant individual heterogeneity in naturally-infected Alpine ibex (Capra ibex) with Brucella melitensis. Front Microbiol 9, 1065, https://doi.org/10.3389/fmicb.2018.01065 (2018).
OIE (Off. int. Epiz., Paris, France) in Brucellosis Vol. Chapter 2.4.1 (2016).
Al-Tawfiq, J. A. & Memish, Z. A. Pregnancy associated brucellosis. Recent Pat Antiinfect Drug Discov 8, 47–50 (2013).
Carvalho Neta, A. et al. Pathogenesis of bovine brucellosis. Vet J 184, 146–155, https://doi.org/10.1016/j.tvjl.2009.04.010 (2010).
Jansen, W., Linard, C., Noll, M., Nöckler, K. & Al Dahouk, S. Brucella-positive raw milk cheese sold on the inner European market: A public health threat due to illegal import? Food Control 100, 130–137, https://doi.org/10.1016/j.foodcont.2019.01.022 (2019).
Higgins, J. L., Gonzalez-Juarrero, M. & Bowen, R. A. Evaluation of shedding, tissue burdens, and humoral immune response in goats after experimental challenge with the virulent Brucella melitensis strain 16M and the reduced virulence vaccine strain Rev. 1. PLoS One 12, e0185823, https://doi.org/10.1371/journal.pone.0185823 (2017).
Tittarelli, M. et al. The persistence of Brucella melitensis in experimentally infected ewes through three reproductive cycles. J Vet Med B Infect Dis Vet Public Health 52, 403–409, https://doi.org/10.1111/j.1439-0450.2005.00885.x (2005).
Lapraik, R. D. & Moffat, R. Latent bovine brucellosis. Vet Rec 111, 578–579 (1982).
Capparelli, R. et al. Heterogeneous shedding of Brucella abortus in milk and its effect on the control of animal brucellosis. J Appl Microbiol 106, 2041–2047, https://doi.org/10.1111/j.1365-2672.2009.04177.x (2009).
Diaz Aparicio, E. Epidemiology of brucellosis in domestic animals caused by Brucella melitensis, Brucella suis and Brucella abortus. Rev Sci Tech 32, 43–51 (2013).
Tikare, N. V., Mantur, B. G. & Bidari, L. H. Brucellar meningitis in an infant–evidence for human breast milk transmission. J Trop Pediatr 54, 272–274 (2008).
Tuon, F. F., Gondolfo, R. B. & Cerchiari, N. Human-to-human transmission of Brucella - a systematic review. Trop Med Int Health 22, 539–546 (2017).
Grillo, M. J., Blasco, J. M., Gorvel, J. P., Moriyon, I. & Moreno, E. What have we learned from brucellosis in the mouse model? Vet Res 43, 29, https://doi.org/10.1186/1297-9716-43-29 (2012).
Georgiades, P., Ferguson-Smith, A. C. & Burton, G. J. Comparative developmental anatomy of the murine and human definitive placentae. Placenta 23, 3–19 (2002).
Carter, A. M. Animal models of human placentation–a review. Placenta 28, 27 (2007).
Wooding, F. B. Analysis of the structure of the ruminant placenta: methods of fixation, embedding, and antibody localization at light and electron microscope levels. Methods Mol Med 121, 315–322 (2006).
Heid, H. W. & Keenan, T. W. Intracellular origin and secretion of milk fat globules. Eur J Cell Biol 84, 245–258, https://doi.org/10.1016/j.ejcb.2004.12.002 (2005).
Masedunskas, A., Chen, Y., Stussman, R., Weigert, R. & Mather, I. H. Kinetics of milk lipid droplet transport, growth, and secretion revealed by intravital imaging: lipid droplet release is intermittently stimulated by oxytocin. Mol Biol Cell 28, 935–946, https://doi.org/10.1091/mbc.E16-11-0776 (2017).
Reece, W. O., Erickson, H. H., Goff, J. P. & Uemura, E. E. (eds.) Dukes’ Physiology of Domestic Animals, 13th Edition. Vol. 13 pp. 760, Wiley Backwell (2015).
Kobayashi, K., Oyama, S., Numata, A., Rahman, M. M. & Kumura, H. Lipopolysaccharide disrupts the milk-blood barrier by modulating claudins in mammary alveolar tight junctions. PLoS One 8, e62187–e62187, https://doi.org/10.1371/journal.pone.0062187 (2013).
Meador, V. P., Deyoe, B. L. & Cheville, N. F. Pathogenesis of Brucella abortus infection of the mammary gland and supramammary lymph node of the goat. Vet Pathol 26, 357–368 (1989).
Xavier, M. N., Paixao, T. A., Poester, F. P., Lage, A. P. & Santos, R. L. Pathological, immunohistochemical and bacteriological study of tissues and milk of cows and fetuses experimentally infected with Brucella abortus. J Comp Pathol 140, 149–157, https://doi.org/10.1016/j.jcpa.2008.10.004 (2009).
Ichikawa, M. et al. Breast milk macrophages spontaneously produce granulocyte–macrophage colony-stimulating factor and differentiate into dendritic cells in the presence of exogenous interleukin-4 alone. Immunology 108, 189–195, https://doi.org/10.1046/j.1365-2567.2003.01572.x (2003).
Sharma, N., Singh, N. & Bhadwal, M. Relationship of Somatic Cell Count and Mastitis: An Overview. Asian-Australas J Anim Sci 24, https://doi.org/10.5713/ajas.2011.10233 (2011).
Opdebeeck, J. P. Mammary gland immunity. J Am Vet Med Assoc 181, 1061–1065 (1982).
Meador, V. P., Hagemoser, W. A. & Deyoe, B. L. Histopathologic findings in Brucella abortus-infected, pregnant goats. Am J Vet Res 49, 274–280 (1988).
Goenka, R., Parent, M. A., Elzer, P. H. & Baldwin, C. L. B cell-deficient mice display markedly enhanced resistance to the intracellular bacterium Brucella abortus. J Infect Dis 203, 1136–1146 (2011).
Demars, A. et al. Route of infection strongly impacts the host-pathogen relationship. Front Immunol 10, 1589–1589, https://doi.org/10.3389/fimmu.2019.01589 (2019).
Meador, V. P., Deyoe, B. L. & Cheville, N. F. Effect of nursing on Brucella abortus infection of mammary glands of goats. Vet Path 26, 369–375, https://doi.org/10.1177/030098588902600502 (1989).
Bosseray, N. & Plommet, M. Serum- and cell-mediated immune protection of mouse placenta and fetus against a Brucella abortus challenge: expression of barrier effect of placenta. Placenta 9, 65–79 (1988).
Bosseray, N. Mother to young transmission of Brucella abortus infection in mouse model. Ann Rech Vet 13, 341–349 (1982).
Cacace, M. L., Claros, E. A., Erazu, K. A., Escobar, G. I. & Lucero, N. E. Congenital brucellosis in an infant. Vector Borne Zoonotic Dis 13, 513–515, https://doi.org/10.1089/vbz.2012.1165 (2013).
Weber, E. M. & Olsson, I. A. S. Maternal behaviour in Mus musculus sp.: An ethological review. Appl Anim Behav Sci 114, 1–22, https://doi.org/10.1016/j.applanim.2008.06.006 (2008).
Shaner, N. C. et al. Improved monomeric red, orange and yellow fluorescent proteins derived from Discosoma sp. red fluorescent protein. Nat Biotechnol 22, 1567–1572, https://doi.org/10.1038/nbt1037 (2004).
Copin, R. et al. In situ microscopy analysis reveals local innate immune response developed around Brucella infected cells in resistant and susceptible mice. PLoS Pathog 8, e1002575, https://doi.org/10.1371/journal.ppat.1002575 (2012).
Hanot Mambres, D. et al. Imported human brucellosis in Belgium: Bio and molecular typing of bacterial isolates, 1996–2015. PLoS One 12, e0174756, https://doi.org/10.1371/journal.pone.0174756 (2017).
Gómez-Gallego, C. et al. A method to collect high volumes of milk from mice (Mus musculus). Anal Vet Murcia 29, 55–61 (2014).
Muranishi, Y. et al. Method for collecting mouse milk without exogenous oxytocin stimulation. BioTechniques 60, https://doi.org/10.2144/000114373 (2016).
Alton, G. G., Jones, L. M., Angus, R. D. & Verger, J. M. Techniques for the brucellosis laboratory. Institut National de la Recherche Agronomique (INRA) (1988).
Quehl, A. G. Kielwein: Leitfaden der Milchkunde und Milchhygiene. Verlag Paul Parey, Berlin und Hamburg 1976. Food/Nahrung 21, 648–648, https://doi.org/10.1002/food.19770210727 (1977).
Nicaise, C. et al. Phagocyte-specific S100A8/A9 is upregulated in primary Sjogren’s syndrome and triggers the secretion of pro-inflammatory cytokines in vitro. Clin Exp Rheumatol 35, 129–136 (2017).
Acknowledgements
The authors would like to thank Marcella Mori and her team from the Belgian Federal Research Institute for Public Health (Sciensano) for kindly providing the human field strain, Benoit Muylkens, Eric Muraille and the ethical commission of the University of Namur for fruitful discussions, input and feedback. Moreover, we would like to thank Benoit Balau and the PFT Morph-IM laboratory as well as Etienne Hanon and Gael Bertrand for excellent technical support in data acquisition, analyses and visualization. Our study was supported by the German Federal Institute for Risk Assessment (grant number 1329–555).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: S.A.D., A.D., W.J., A.R. Analysed the data: S.A.D., J.G., W.J., C.N., A.R. Contributed reagents/materials/analysis tools: X.D., A.D., W.J., C.N., A.R. Wrote the manuscript: W.J., Revised the manuscript: S.A.D., X.D., A.D., J.G., W.J., C.N., A.R.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jansen, W., Demars, A., Nicaise, C. et al. Shedding of Brucella melitensis happens through milk macrophages in the murine model of infection. Sci Rep 10, 9421 (2020). https://doi.org/10.1038/s41598-020-65760-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-65760-0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.