Abstract
The corallivorous Crown-of-Thorns Starfish (CoTS, Acanthaster spp.) has been linked with the widespread loss of scleractinian coral cover on Indo-Pacific reefs during periodic population outbreaks. Here, we re-examine CoTS consumption by coral reef fish species by using new DNA technologies to detect Pacific Crown-of-Thorns Starfish (Acanthaster cf. solaris) in fish faecal and gut content samples. CoTS DNA was detected in samples from 18 different coral reef fish species collected on reefs at various stages of CoTS outbreaks in the Great Barrier Reef Marine Park, nine of which had not been previously reported to feed on CoTS. A comprehensive set of negative and positive control samples confirmed that our collection, processing and analysis procedures were robust, although food web transfer of CoTS DNA cannot be ruled out for some fish species. Our results, combined with the (i) presence of CoTS spines in some samples, (ii) reported predation on CoTS gametes, larvae and settled individuals, and (iii) known diet information for fish species examined, strongly indicate that direct fish predation on CoTS may well be more common than is currently appreciated. We provide recommendations for specific management approaches to enhance predation on CoTS by coral reef fishes, and to support the mitigation of CoTS outbreaks and reverse declines in hard coral cover.
Similar content being viewed by others
Introduction
Crown-of-Thorns Starfish (CoTS, Acanthaster spp.) are carnivorous starfish that feed on living tissue of scleractinian (i.e. hard) corals1. Across the Indo-Pacific, CoTS have been linked with the widespread loss of hard coral cover on reefs during periodic and recurrent population outbreaks2. For example, average hard coral cover halved on the Great Barrier Reef (GBR; Australia) from 1985 to 2012, with 42% of this decline attributed to coral predation by the Pacific Crown-of-Thorns Starfish (Acanthaster cf. solaris)3. Renewed outbreaks of CoTS are causing extensive coral loss on the GBR4, notably in areas that were mostly unaffected by recent mass bleaching events5,6. These high levels of CoTS predation on scleractinian corals have the potential to fundamentally alter the form and structure of coral reefs and their biological communities1.
Mitigating the detrimental impacts of CoTS predation, and reversing the declines in coral cover requires effective control and prevention of CoTS population outbreaks3,7. Current management interventions include manual control of individual starfish to reduce CoTS populations directly6, and reducing land-based run-off to lower recruitment rates of CoTS pelagic larvae into benthic settlement8,9,10. In addition, a variety of analyses have linked Marine Protected Areas (i.e. zoning) and CoTS outbreaks on the GBR, including (i) reefs closed to fishing having fewer CoTS than those open to fishing11,12, and (ii) the impacts of CoTS outbreaks being reduced at reefs closed to fishing13. These findings suggest an impact of zoning on CoTS population outbreaks13, with one potential pathway being a higher level of predation on one or more CoTS life stages on reefs closed to fishing14.
The release from predator pressure, i.e. the ‘predator removal’ hypothesis, has been posited as a potential contributor to CoTS population outbreaks15. Originally, a decrease in population densities of the Giant Triton (Charonia tritonis) was thought to underpin this release, being the only known predator of larger juvenile and adult CoTS at the time15. Since then, a total of 80 species of coral reef fishes and invertebrates have been identified as predators of planktonic and settled life stages of CoTS16. This would suggest that, rather than being influenced by just a single predator, the combined consumption of CoTS by a range of predators may mitigate the severity of CoTS population outbreaks15,16. Most of these records, however, comprise predation on injured and moribund or dead individuals under experimental conditions, with only a limited number of field observations of predation on early life stages and on healthy, un-injured adults16. Despite this lack of field observations of CoTS predation, several independent modelling studies have provided support for the ‘predator removal’ hypothesis17,18,19. Hence, a more comprehensive understanding of coral reef organisms that prey on live CoTS in the field, including on early life stages, is critical to inform active intervention in predation pressure to mitigate CoTS population outbreaks.
In this study, we re-examine CoTS consumption by coral reef fish species by using evidence of CoTS DNA detected in fish faecal and gut content samples. Specifically, we apply a new digital droplet PCR (ddPCR)-based method to detect DNA of the Pacific Crown-of-Thorns Starfish (A. cf. solaris)20 in samples from wild-caught fish. First, we conducted a literature review to target our field collections towards those coral reef fish species that are likely to consume the different life stages of CoTS, using Cowan et al.16 as a starting point. Second, we conducted two pilot studies to confirm our ability to detect CoTS DNA in fish faecal and gut content samples in both laboratory and field collected samples. Finally, we collected faecal and gut content samples from a total of 678 individual fish from 101 different coral reef fish species and 21 different families on reefs experiencing varying levels of CoTS outbreaks during and outside the CoTS spawning seasons1,21. Our results demonstrate that potential predation by coral reef fish on different life stages of CoTS is likely to be more widespread than is currently appreciated.
Results
Reports on Crown-of-Thorns Starfish predation by coral reef fish
To target our field collections towards those coral reef fish species that are likely to consume the different life stages of CoTS, a detailed literature review was conducted. A recent synthesis on known predators, reporting a total of 56 species of coral reef fish from 11 families feeding on CoTS (Cowan et al.16), was used as the starting point. Our review identified a total of 71 coral reef fish species from 16 families reported to have fed on Acanthaster spp. based on field and laboratory observations (Table 1). Fifty-two of these species fed on injured, moribund or dead CoTS with all but one feeding event observed under experimental conditions; 41 species were observed in field settings, 9 in both field and laboratory settings and 2 in laboratory settings. Most of these species (n = 42) have not been reported to consume living and un-injured CoTS. Field reports on predation on the early life stage of CoTS, including pelagic gametes and larvae and early post-settlement juveniles, are very limited16 at least in part due to the difficulties of observing such predation1. Indeed, such observations are restricted to three fish species, namely the Scissortail Sergeant (Abudefduf sexfasciatus) and Staghorn Damsel (Amblyglyphidodon curacao) consuming CoTS eggs15,22 and the Oriental Butterflyfish (Chaetodon auripes) eating CoTS sperm23 released during spawning. Recent laboratory feeding experiments, however, have reported an additional 11 damselfish feeding on spawned eggs and pelagic larvae, with several species readily taking to the food offered24,25,26. Field reports on predation on the settled life stage of CoTS are slightly more common16, having been reported for 12 fish species from the families Lethrinidae (emperor; 4 species), Balistidae (triggerfish; 2 species), Tetraodontidae (pufferfish; 2 species), Diodontidae (porcupinefish; 1 species), Labridae (wrasse; 1 species), Pomacanthidae (angelfish; 1 species) and Serranidae (grouper, 1 species) (Table 1). Remains of CoTS have been confirmed in gut contents of five of these fish species, namely Spotted Porcupinefish (Diodon hystrix)27, Humphead Maori Wrasse (Cheilinus undulatus)27,28,29, Redthroat Emperor (Lethrinus miniatus)27, Spangled Emperor (Lethrinus nebulosus)30 and Queensland Grouper (Epinephelus lanceolatus)31,32, and reported but not confirmed for Yellowmargin Triggerfish (Pseudobalistes flavimarginatus)33. Lethal predation by fish in the field on settled and apparent healthy CoTS has only been observed sporadically, namely by Stars-and-stripes Puffer (Arothron hispidus), Titan Triggerfish (Balistoides viridescens) and Spangled Emperor (L. nebulosus)27, with most studies reporting sublethal predation on CoTS arms34,35. Finally, Acanthaster planci DNA has been detected in gut contents using metabarcoding in a cardinal fish (Nectamia savayensis), Bigscale Soldierfish (Myripristis berndti) and Smallmouth Squirrelfish (Sargocentron microstoma)36.
Can CoTS DNA be detected in fish faecal and gut content samples?
The ability to detect DNA from Pacific Crown-of-Thorns Starfish (CoTS, A. cf. solaris) in fish faecal and gut content samples was confirmed in two pilot studies (Supplementary Text 1). First, CoTS DNA (mtCOI gene fragment) was detected in faecal samples collected from the Blackspotted Puffer (Arothron nigropunctatus) fed freshly-killed CoTS in controlled laboratory settings, with detection up to seven days post-feeding in two of the five pufferfish (Supplementary Fig. 1.1). In addition, faecal samples preserved in 100% EtOH and liquid nitrogen showed similar amplification for CoTS mtCOI fragments (both 919 bp and 126 bp), providing confidence in the use of 100% EtOH as a preservative during field trips in remote locations (Supplementary Fig. 1.2). Second, faecal and gut content samples were collected from a range of coral reef fish species in the field, with CoTS spines detected in faeces from wild-caught Spangled Emperor (L. nebulosus) and Starry Puffer (Arothron stellatus) (Supplementary Fig. 1.3). Learnings from these two pilot studies were applied during subsequent field collections, in particular around the collection and preservation (Supplementary Text 2) and preventing contamination (Supplementary Text 3) of faecal and gut content samples when CoTS DNA is potentially present in the environment20,21.
Collection of faeces and gut contents from coral reef fish species
A total of 678 individual fish from 101 different coral reef fish species and 21 different families were collected on reefs experiencing varying levels of CoTS population outbreaks in January 2018, July 2018 and July 2019 (Figure 1; Table 2; Supplementary Text 2; Supplementary Table 2.1). During the CoTS breeding season1,21 in January 2018, a total of 418 individuals from 59 different fish species were collected (range 1 to 21 individuals per fish species). Outside the CoTS breeding season1,21, in July 2018, a total of 173 individuals from 37 different fish species were collected (range 1 to 18 individuals per fish species). During both these trips, fish were kept overnight on the RV Cape Ferguson for collection of their faeces the next morning; faecal material was observed for most of these fish (for 405 and 138 individual fish, respectively). During the third field trip conducted in July 2019, speared fish were kept on ice and dissected on return to the RV Cape Ferguson for collection of their gut content. A total of 87 individuals from 19 different fish species were collected (range 1 to 10 individuals per fish species); gut content material was present in all fish.
Detection of CoTS DNA in faeces and gut contents from coral reef fish species
CoTS DNA was detected in faecal and gut content samples from a total of 30 individuals from 18 different coral reef fish species and eight different families (Table 3). During the CoTS spawning season (January 2018), CoTS DNA was detected in the faeces of seven individual fish from six different damselfish species (Table 3; Fig. 2). For each of these six species, this represented ≤50% of intraspecies samples collected at the reefs where positive CoTS DNA detections on faecal samples were made. One of these species, namely the Neon Damsel (Pomacentrus coelestis), has previously not been reported feeding on CoTS (Table 1). For all seven fish, faecal matter (albeit no distinct CoTS remains) was present in the holding bag or after filtration through the mesh sieve. The size range for these seven fish ranged from 20 to 52 mm Standard Length (SL), suggesting young of the year for all but two species namely the Banded Humbug (Dascyllus aruanus) and Neon Damsel (P. coelestis). All seven fish were collected from two reefs (Bramble, Rib) that experienced severe CoTS outbreak status at the time of collection (Table 2). CoTS DNA was detected in plankton samples collected at Bramble and Rib Reef indicating the presence of CoTS gametes and/or larvae (Supplementary Text 3; Supplementary Table 3.1)21. In contrast, no CoTS DNA was detected in plankton samples or faecal matter from fish collected at the two other reefs (Unnamed, Kelso), despite an established CoTS outbreak at Unnamed Reef and CoTS present at certain locations of Kelso Reef but overall being below outbreak threshold status (Table 2).
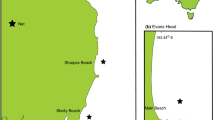
Sampling locations. Locations of coral reef fish collections at eight midshelf reefs in the central Great Barrier Reef World Heritage Area, Australia, conducted from the RV Cape Ferguson in 2018 and 2019. The status of CoTS population outbreaks varied from no outbreak to severe across the eight reefs at the time of fish collection (Table 2). Insert shows location of study area in Australia. The spatial layers to create the map were obtained from the Great Barrier Reef Marine Park Authority under a Creative Commons Attribution 4.0 licence (CC BY) (http://www.gbrmpa.gov.au/about-us/resources-and-publications/spatial-data-information-services).
Detection of CoTS DNA in fish faecal and gut content samples. Examples for positive and negative digital droplet PCR results for four different coral reef fish, namely Banded Humbug (Dascyllus aruanus; positive), Blackvent Damsel (Dischistodus melanotus; negative), Spangled Emperor (Lethrinus nebulosus; positive), and Common Coral Trout (Plectropomus leopardus; positive). Sample collection number for each individual fish are given. Examples of digital droplet PCR results for positive (one to two 8-day old Acanthaster cf. solaris larvae) and negative (blanks) controls are also provided.
Outside the CoTS spawning season (July 2018 and 2019), CoTS DNA was detected in faecal and gut content samples of 23 individual fish from 12 different fish species and seven families (Fig. 2; Table 3). For seven of these species, this represented ≥50% of intraspecies samples collected at the reefs where positive CoTS DNA detections on faecal and gut content samples were made. Eight of these species, namely the Painted Sweetlips (Diagramma pictum labiosum), Floral Maori Wrasse (Cheilinus chlorourus), Robinson’s Seabream (Gymnocranius grandoculis), Redspot Emperor (Lethrinus lentjan), Ornate Emperor (Lethrinus ornatus), Blackspot Snapper (Lutjanus fulviflama), Moses’ Snapper (Lutjanus russelli) and Purple Rockcod (Epinephelus cyanopodus) have previously not been reported feeding on CoTS (Table 1). For all but three fish, faecal matter (including distinct CoTS remains in three Spangled Emperor, L. nebulosus) was present in the holding crate or after filtration on the mesh sieves. The size range for these 23 fish ranged from 110 to 420 mm SL (Table 3), suggesting all individuals were 1+ year classes for all species. The 23 fish were collected from four reefs (Unnamed, Kelso, Rib, Lodestone) that experienced different levels of CoTS outbreaks (Table 2).
Preventing contamination of faecal and gut content samples
To prevent and examine potential contamination of fish faecal and gut content samples from CoTS DNA present in the environment20,21, a comprehensive set of negative and positive control measures were taken prior and during field trips (Supplementary Text 3). For negative control measures, this included confirming the removal of CoTS DNA from unfiltered seawater following treatment in our filtration system on-board the RV Cape Ferguson in July 2018 (Supplementary Table 2.3; Supplementary Fig. 2.1). In addition, no CoTS DNA was detected in faecal samples collected from all but one fish species assigned as negative controls, namely from six different herbivorous species (n = 12 individuals) from four different families, and from six different corallivorous species (n = 15 individuals) from two different families (Supplementary Table 3.1). CoTS DNA was detected in one out of 17 samples collected from adult Common Coral Trout (Plectropomus leopardus), an obligate piscivore when an adult37, suggesting that food web transfer of CoTS DNA cannot be ruled out for some of the fish species that tested positive for CoTS DNA. Moreover, CoTS DNA was absent in all faecal samples collected from 13 different fish species (n = 97 individuals) on reefs at Lizard Island without CoTS population outbreaks during the CoTS spawning season in January 2019 (Supplementary Table 3.2). Individuals of these 13 fish species (of similar sizes as those collected at Lizard Island) including adult Common Coral Trout, collected on reefs with CoTS outbreaks on the central GBR in January and July 2018, had tested positive for CoTS DNA (Table 3; Supplementary Table 3.1). Equally important, our processing and preserving procedures did not destroy CoTS DNA as exemplified by the positive detection of CoTS DNA in water samples collected from the CoTS aquarium in the National Sea Simulator, and in 100% EtoH preserved freshly collected CoTS spines from the CoTS aquarium at the Lizard Island Research Station (Supplementary Table 3.1).
Discussion
To inform active intervention in predation pressure to mitigate CoTS population outbreaks, a more comprehensive understanding of coral reef organisms that feed on live CoTS in the field, including on early life stages, is critical16. Here, we applied a ddPCR-based method to detect DNA of the Pacific Crown-of-Thorns Starfish (A. cf. solaris)20 in faecal and gut content samples from fish caught at reefs with varying levels of CoTS outbreaks. CoTS DNA was detected in samples from a total of 30 individuals from 18 different coral reef fish species and eight different families. Comparing these results with our literature review on CoTS predation by coral reef fish, nine of these fish species had not previously been reported feeding on CoTS. Our comprehensive set of negative and positive control measures were found to be robust, confirming that these positive detections were extremely unlikely to have resulted from contamination38 including with CoTS DNA present in the environment20,21. Food web transfer of CoTS DNA, however, cannot be ruled out for some of the fish species, based on one positive detection in adult Common Coral Trout (P. leopardus), an obligate piscivore species when adult37. Nonetheless based on known diet information for the coral reef fish species examined, the presence of CoTS spines in some samples, combined with reported lethal predation on CoTS gametes15,22,23 and larvae24,26 and lethal27 and sublethal34,35 predation on settled individuals, our results strongly indicate that direct fish predation on CoTS may well be more common than is currently appreciated.
Traditional gut content analysis has been largely ineffective in identifying putative CoTS predators and this method often fails to find remains of Acanthaster spp.29,30,39. Indeed, CoTS remains have only been confirmed in five coral reef fish species27,28,29,30,31,32, and reported but not confirmed for one other33. This is at least partly a result of fish being collected in areas with low29 or unknown30 abundance of adult Acanthaster spp., or of small sample sizes collected near areas with high CoTS densities39. In addition, identification of CoTS remains in the guts can be challenging and may easily be mistaken for other echinoderms27,33,40. Our application of a ddPCR-based method to detect A. cf. solaris DNA20 in faecal and gut content samples corroborated potential CoTS predation by known fish predators16 and identified previously unknown fish predators. Similarly, A. planci DNA has previously been detected in gut contents using metabarcoding in three other coral fish species36. These findings demonstrate that these methods can be applied to screen faecal or gut content samples of presumed CoTS predators, and validate field predation by fish and invertebrate species that have been reported to consume CoTS in experimental settings41,42,43,44. Importantly our study shows this can be achieved using non-invasive and non-lethal methods, a critical consideration for the ethical use of animals as well as for examining threatened species. Moreover, our ddPCR-based method detected CoTS DNA in a proportionally higher number of individuals in two emperor species (L. miniatus, L. nebulosus) compared to gut-content studies on the same species27,30,39. Similarly, CoTS remains were observed in only three of the 30 samples testing positive for CoTS DNA highlighting the superior sensitivity of this method versus gut content analyses. Increased detection of predation using qPCR versus visual gut content analysis has been reported for other fish species45. Hence, the ddPCR method would be a superior approach to identify additional CoTS predators on coral reefs across the Indo-Pacific2, and ascertain the frequency of CoTS consumption by known predators16.
Our findings strongly support the notion that coral reef fish feed on both the pelagic and benthic phases of CoTS in the field. Various behaviours by CoTS further corroborate that predation by visual predators is highly likely, including suggested spawning times during outgoing tides late afternoon and at night23, and highly cryptic and nocturnal behaviour by settled CoTS, particularly the smaller ones46. Consumption of the pelagic life stages of CoTS, i.e. gametes and larvae, has rarely been observed in the field15,22,23. Under controlled laboratory conditions, a wide range of damselfish species readily feed on CoTS eggs and larvae, including in the presence and in preference of Blue Seastar (Linckia laevigata) larvae24,26. This includes three of the six damselfish species that tested positive for CoTS DNA in our study. The confirmed presence of CoTS gametes and/or larvae at reef locations where the seven positive damselfish were collected, in combination with reported zooplankton diets for at least five of these six damselfish species47, indicate that feeding on pelagic CoTS larvae in the field is probable. Furthermore, the absence of CoTS DNA in faecal samples from obligate corallivorous and herbivorous fish species corroborates that these damselfish detections were not a result of feeding on coral mucus or filamentous algae contaminated with CoTS DNA. The relatively low incidence of CoTS DNA detection within individual damselfish species may well be related to a sporadic spatio-temporal distribution of the pelagic CoTS phase within a complex reef matrix21,48. Nevertheless, planktivorous fishes could still markedly reduce the number of CoTS larvae under such conditions, based on estimated CoTS consumption rates for damselfish in laboratory settings24,26 and high damselfish abundances in the field49.
Predation on the benthic phase of CoTS, i.e. juveniles and adults, has been observed slightly more often in the field16, with reports for at least eleven fish species. Our results strongly suggest that predation by coral reef fish on settled CoTS is more common, based on (i) detection of CoTS DNA in samples from nine wild-caught fish species previously not reported feeding on CoTS; (ii) detection of CoTS remains in wild-caught Spangled Emperor (L. nebulosus) and Starry Puffer (A. stellatus); and (iii) laboratory observations on Blackspotted Puffer (A. nigropunctatus) (previously not reported feeding on live CoTS) readily consuming freshly-killed CoTS, and biting off arms from an apparent healthy juvenile CoTS (Kroon, unpublished). Benthic CoTS were present, including at outbreak levels, at all reef locations where these 23 positive fish were collected during the non-breeding season. For all 12 species identified, reported diet information indicate that predation on benthic CoTS in the field is likely. However, the presence of CoTS DNA in a sample from one adult P. leopardus, used as a negative control in our study given that adults are obligate piscivores37, reveals that food web transfer of CoTS remains or CoTS DNA cannot be ruled out for at least some fish species. Similarly, the presence of CoTS DNA in field-collected samples cannot distinguish between consumption of a live, moribund or dead benthic CoTS, although inferences from known dietary habits and field observations can inform this. Combining this information and our ddPCR results for fish species in the emperor (Lethrinidae) and tropical snapper (Lutjanidae) families, particularly for Spangled and Redthroat Emperor (L. nebulosus and L. miniatus), strongly indicates that they may well be critical predators on the benthic phase of CoTS.
Many of the coral reef fish species that have been reported to feed on Acanthaster sp, including those testing positive for CoTS DNA in our study, are subject to various fisheries across the Indo-Pacific14. In the GBR Marine Park, the commercial Marine Aquarium Fish Fishery targets over 600 different coral reef fish species50, including damselfish, pufferfish and triggerfish51. This commercial fishery dates back to the 1970s52 and collected between 130 000 and 260 000 coral reef fish annually between 1998 and 200850. The Coral Reef Fin Fish Fishery, comprising commercial, recreational (including charters) and Indigenous fishers, targets a range of fish species that are known or likely CoTS predators including emperors (Lethrinidae) and tropical snappers (Lutjanidae)53. Species in these two families have been fished commercially and recreationally on the GBR back to at least the 1950s54. Currently, Redthroat Emperor (L. miniatus) is one of the two primary target species in this fishery, with commercial harvests ranging from 137 tonnes to 271 tonnes per year comprising 10% to 17% of the commercial component of this fisheries’ total harvest from 2009/10 to 2017/1855. Spangled Emperor (L. nebulosus) is the other emperor of fisheries’ significance, with commercial harvests averaging 56 tonnes per year in the fisheries56. The harvest estimates for the charter, recreational and indigenous fisheries for these two species are less reliable but of a similar order of magnitude57,58. Whether the individual or combined take of these various fisheries, and associated release from predator pressure15, has been playing a role in influencing CoTS population outbreaks remains unclear. Fishing does reduce densities of planktivorous damselfish and ‘secondary target’ species, principally lethrinids and lutjanids, being lower on offshore reefs open to fishing than those closed to fishing59. This strongly supports the notion that a higher level of predation on one or more CoTS life stages on reefs closed to fishing may underpin the reported impact of zoning on CoTS population outbreaks13. Hence, further examination of potential spatio-temporal relationship(s) between coral reef fish presence and abundance, coral reef fish harvest by various fisheries and CoTS population outbreaks on Indo-Pacific coral reefs, including the GBR Marine Park, is warranted.
Potential mitigation of CoTS population outbreaks by coral reef fish is most likely to occur through lethal predation on the pelagic phase15,22,23,24,25,26 regulating the settlement of CoTS larvae, and lethal and sub-lethal predation on the settled phase influencing growth, reproduction35 and mortality27, in particular during low, non-outbreak, densities1. In the context of the GBR Marine Park, this would suggest that enhanced and targeted management of coral reef fish to mitigate CoTS outbreaks would be most effective in the initiation zone, i.e. the midshelf reefs between Lizard Island and Cairns1,15,60,61,62 during pre-outbreak conditions. The progressive southward spread of these outbreaks could also potentially be mitigated by management of coral reef fish, in particular on reefs that are identified as key nodes in COTS outbreak and spread processes63,64. Such management approaches may involve (i) reduced fisheries take of single or a suite of coral reef fish species known to consume CoTS; (ii) augmentative strategies to increase the abundance of single or a suite of coral reef fish species known to consume CoTS; (iii) temporal closures of reefs to fishing when environmental conditions conducive to outbreaks are predicted; and/or (iv) establishing new marine reserves (i.e. no-take areas) in the outbreak initiation zone and on highly connected reefs further south. These approaches would enhance fisheries management reforms currently being implemented, including delivery of sustainable fisheries catch limits to ensure 60% of the biomass remains to deliver more resilient marine ecosystems65. They would also complement current management interventions such as direct manual control6 and improving water quality in land-based run-off8,9,10. Given that CoTS population outbreaks are a major driver of the reported and projected decline in hard coral cover on the GBR3,7, current fisheries management reforms need to be fully implemented to contribute positively to reversing this decline. These additional proposed management approaches to mitigate CoTS outbreaks by enhancing coral reef fish predation should be seriously considered to improve hard coral cover now and into the future, both on the GBR and on Indo-Pacific reefs more broadly.
Methods
Study area
The GBR extends for 2,000 km along Australia’s north-eastern coast (Fig. 1) and contains a variety of tropical marine ecosystems including ~20 000 km2 of coral reefs, ~43 000 km2 of seagrass meadows and extensive mangrove forests (Great Barrier Reef Marine Park Authority, 2014). The 344,400 km2 GBR Marine Park was established under the Federal Great Barrier Reef Marine Park Act 197566, followed with the listing of the 348,000 km2 GBR World Heritage Area (WHA) by UNESCO in 198167. Since 1975, the GBR has been managed as a multiple-use area by the Australian Federal and Queensland State Governments68. The main commercial and non-commercial uses are commercial marine tourism, defence activities, fishing, ports, recreation (not including fishing), research and educational activities, shipping, and traditional use of marine resources68. Since 2004, these multiple uses have been managed under the Zoning Plan for the GBR Marine Park outlining activities allowed, prohibited or requiring a permit in the seven different zones69,70. The three major zone types are ‘General Use’ (open and fished; 33.8% of the GBR Marine Park), ‘Habitat Protection’ (open and fished except for trawling; 28.2%), and ‘Marine National Park’ (no-take; 33.3%)71.
Crown-of-Thorns Starfish outbreaks on the GBR
The first and second CoTS population outbreaks on the GBR were noticed at Green Island and nearby reefs in 196215,60 and in 197961, respectively (Fig. 1). The first stages of the third CoTS population outbreak were detected in 1993, with outbreaks recorded on midshelf reefs between Lizard Island and Cairns in 1994-9562 (Fig. 1). A fourth outbreak is now in progress in the central and southern GBR72 which again was first detected on midshelf reefs between Lizard Island and Cairns in 20101.
The four recorded CoTS population outbreaks on the central GBR seem to have followed a similar pattern of initiation and spread1. The first detection (or at least reporting) of high CoTS densities on midshelf reefs in the north-central section of the GBR suggests that outbreaks initiate roughly between Lizard Island (14.6°S) and Cairns (17°S)15,60,61,62,73 (the ‘initiation box’). The limited spatial and temporal resolution of reef monitoring in this area precludes the identification of single or multiple reefs as the source of primary outbreaks73,74. Following initiation, each of the four outbreaks shows a progressive southward spread at a rate of 1° of latitude every three years1,75,76,77. This is consistent with southward dispersal of CoTS larvae spawned at the outbreak front75. After approximately 15 years, CoTS population outbreaks appear to die off on midshelf reefs in the Pompeys’ section (21.0°–22.0° S) of the southern GBR1. Northward spread of CoTS population outbreaks from the initiation zone has also been reported77, although the pattern of spread is less clear due to fewer surveys having been conducted in the northern GBR.
Life history of Crown-of-Thorns Starfish
The life history of Acanthaster spp. consists of a relatively short planktonic stage (i.e. days to weeks) and a longer settled stage (i.e. years) (all information from1, and references therein, unless otherwise noted). The planktonic stage begins with the release of female and male gametes which can include up to 65 million eggs per individual female. On the GBR, spawning generally occurs at the start of the summer wet season from November to February21. Following fertilization the larval phase, ranging in size from 0.5 to 1.5 mm long, can last from 9 to 42 days depending on temperature and food availability. Larvae settle onto reef habitat, showing a strong association with crustose coralline algae, and subsequently metamorphosize from a planktonic larva into a benthic juvenile seastar (0.5 mm diameter) over a period of two days. For the next six months, the juvenile seastar (1–10 mm diameter) will feed on crustose coralline algae before a permanent shift in diet to polyps of reef-building corals. The coral-feeding juvenile and sub-adult stages (10–200 mm diameter) last approximately two years, after which they sexually mature into a coral-feeding adult stage (200–350 mm diameter) which lasts two to five years. After five years, senile adult CoTS (>350 mm in diameter) generally show a decline and subsequent cessation of gametogenesis. It is likely that at all stages of their life history CoTS are exposed to predation by a variety of coral reef organisms, including by coral reef fishes16.
Collection of faeces and gut contents from coral reef fish species
Field and laboratory work on fish described in this study was conducted in accordance with relevant guidelines and regulations, under permits from the Great Barrier Reef Marine Park Authority, the Queensland Department of Agriculture and Fisheries and the James Cook University Animal Ethics Committee.
To examine fish faecal and gut content samples for CoTS DNA, coral reef fish were collected at midshelf reefs experiencing varying levels of CoTS population outbreaks during three field trips in 2018 and 2019 (Table 2). To target our field collections towards those coral reef fish species that are likely to consume the different life stages of CoTS, we first conducted a literature review using Cowan et al.16 as a starting point. Specific information was sourced from the primary sources, including on (i) fish species (common and scientific name; family; Codes for Australian Aquatic Biota (CAAB) number), (ii) status of CoTS being consumed (pelagic or benthic stage; injured, moribund or dead), (ii) whether predation was observed in the field or laboratory, and (iv) location of predation.
In parallel with this literature review, we conducted two pilot studies to examine the feasibility to detect DNA from the Pacific Crown-of-Thorns Starfish (CoTS; A. cf. solaris) in fish faecal samples. The first pilot study examined whether CoTS DNA could be detected in fish faecal samples collected from dog faced pufferfish (A. nigropunctatus) fed freshly-killed CoTS in controlled laboratory settings (Supplementary Text 1; Supplementary Figs. 1.1; 1.2). The second pilot study examined whether faecal samples, potentially containing CoTS DNA, could be collected using non-lethal and non-invasive methods from coral reef fish in the field (Supplementary Text 1; Supplementary Fig. 1.3).
Based on the findings of the review and two pilot studies, a list of fish species was developed to target for collection during three field trips. To further refine our collections towards those fish species likely to consume different life stages of CoTS, we collated and reviewed information on diet and food items for each of these species from FishBase78 and associated primary sources, and from several reports by the Food and Agriculture Organization of the United Nations on fish and fisheries species in the Indo-Pacific region (Supplementary Table 2.2). For some species no information on diet, food items or primary sources was presented on Fishbase; in these cases we also searched the Web of Science. Based on dietary information we included certain fish species as ‘negative controls’, i.e. species that were highly unlikely to consume CoTS such as obligate herbivores, corallivores and piscivores (Supplementary Text 3; Supplementary Table 2.2). For all fish species, detailed procedures were implemented for the collection and preservation of their faecal and gut content samples (Supplementary Text 2). The first field trip, conducted during the CoTS spawning season1,21 in January 2018, targeted fish species that may consume early life history stages of CoTS (gametes, planktonic larvae and newly settled juveniles) (Supplementary Text 2). The second and third field trip, conducted outside the CoTS spawning season1,21 in July 2018 and July 2019, targeted fish species that may consume settled life history stages of CoTS (juveniles, sub-adults and adults) (Supplementary Text 2). The third and final field trip targeted specific coral reef fish species that proved difficult or impossible to capture using non-lethal methods during the July 2018 trip.
Based on the findings of the two pilot studies, a comprehensive set of negative and positive control measures were taken prior to and during these field trips (Supplementary Text 3), to prevent and examine potential contamination of fish faecal and gut content samples38 including with CoTS DNA present in the environment20,21. One key measure involved minimising the probability of false positives associated with fish predation on moribound and dead CoTS resulting from the CoTS control program run by the Great Barrier Reef Marine Park Authority (GBRMPA)79. All field collection trips on the central GBR were conducted on reefs that were experiencing CoTS outbreaks (Table 2), with some of these reefs being visited by the CoTS control program. This control program kills individual CoTS by using a single small volume injection of oxbile41; the dead CoTS are left on the reef to decompose. Four days after injection, little evidence of dead CoTS remain except for small piles of spines and skeletal elements41. Similar disintegration rates have been reported for moribund and dead CoTS with few spines and ossicles remaining after 4 to 8 days80,81. Fish predation on these moribund and dead CoTS could greatly increase the probability of detecting CoTS DNA in fish faeces and gut contents when no predation on life CoTS had occurred (i.e. false positive). Further, results from a preliminary study suggests that CoTS DNA decays exponentially in seawater to being undetectable after 8 days (Doyle, unpublished). To minimise the probability of false positives, we closely consulted with GBRMPA and only visited reefs that had either not experienced CoTS culling at all, or had not experienced CoTS culling for at least four weeks prior to the respective field trip. For the latter reefs, this would have allowed ample time for complete disintegration of culled CoTS (4–8 days) and complete decay of associated CoTS DNA in surrounding waters (8 days). For the January 2018 field trip, CoTS had never been culled (18-025) or had not been culled in the five weeks prior (Bramble, Kelso and Rib reefs). Similarly, for the July 2018 field trip, the final CoTS culling on Bramble, Rib, Kelso, Lodestone and 18-025 reefs took place at least five weeks prior (i.e. before or on 29 May 2018) and ceased until after this trip was finished in consultation with GBRMPA. Finally, for the July 2019 field trip, CoTS had not been culled (Kelso, Little and Big Broadhurst reefs) or had not been culled in the four weeks prior (Keeper).
Processing faecal and gut content samples for detection of CoTS DNA
Fish faecal and gut content samples were preserved in 100% EtOH in sample vials ranging from 5 ml to 500 ml (Supplementary Text 2). Subsequent sample homogenisation, extraction and ddPCR, including assessing the ddPCR outcomes, were conducted by investigators who were unaware of the sample allocation (i.e. fish species). Preserved samples were homogenised in different ways depending on the sample volume. Faecal and gut content samples in 5 ml or 50 ml vials were homogenised in a bead-beater with stainless steel beads (BioSpec) for two minutes. For faecal samples captured over a filter disc (Supplementary Text 2), the disc was opened carefully into a cylindrical shape inside their sample vial and beads placed into the centre before being homogenised in a bead-beater (BioSpec) for two min. Gut content samples in 120 ml or 500 ml vials were homogenised using a commercial ‘paint shaker’ following the addition of stainless steel beads (BioSpec) for five min.
DNA extraction was conducted on a sub-sample of each homogenised sample. These sub-samples were taken after inverting the homogenised sample several times to ensure thorough mixing. For samples preserved and homogenised in 5 ml vials, a sub-sample of 1 ml was transferred to a 2 ml screw top tube. Following removal of EtOH using a rotary vacuum concentrator (Savant), DNA was extracted using slightly modified versions of the Qiagen DNeasy Blood and Tissue extraction kit. Briefly, a lysis solution of Qiagen buffer ATL (360 μl) and proteinase K (10 mg ml-1, 40 μl) was added followed by an overnight incubation (56 °C) with constant rotation. From each sample, 200 µl was then transferred into a new 2 ml microtube, which was placed in the sample rack within a Qiacube robot for automated DNA extraction using the following protocol. After addition of Qiagen buffer AL (200 µl), the ATL/proteinase K/AL solution incubated (56 °C) with agitation for 30 min. Ethanol (200 µl) was added to and mixed with the incubated solution. The ATL/proteinase K/AL/EtOH solution (600 µl) was loaded on to a Qiagen spin column and centrifuged at 10,000 × g for 1 min. Qiagen buffer AW1 (500 µl) was applied to the spin column followed by centrifugation at 10,000 × g for 1 min. Qiagen buffer AW2 (500 µl) was applied to the spin column followed by centrifugation at 20,000 × g for 3 min. Elution of DNA from the spin column was performed in 3 ×50 µl TE(0.1). After each addition of 50 µl TE(0.1), the spin column was incubated at room temperature for 1 min followed by a centrifugation at 10,000 g for 1 min.
For samples preserved and homogenised in 50 ml, 120 ml or 500 ml vials, the amount of sub-sample taken for DNA extraction was based on the original amount of faecal or gut content preserved. Specifically, these samples were grouped into three relative biomass categories namely ‘small’, ‘medium’ and ‘large’, and a sub-sample of 16 ml (small), 8 ml (medium) and 4 ml (large) was transferred into new 50 ml sample vials. Following removal of EtOH using a rotary vacuum concentrator (Savant), DNA was extracted as described above with the following modifications. The initial lysis solution added to the sub-sample consisted of Qiagen buffer ATL (1.8 ml) and proteinase K (0.2 ml, 10 mg ml-1). From each sample, 600 µl was transferred into a new 2 ml microtube, and the DNA extraction process completed on the Qiacube robot with a 600 µl volume used where a 200 µl was described above. In addition, the entire ATL/proteinase K/AL/EtOH solution (1800 µl) was loaded on to a Qiagen spin column in 3 ×600 µl batches. All other steps remained the same.
Digital droplet PCR on the smaller 126 bp CoTS mtCOI fragment, including positive and negative extraction controls as well as positive and negative PCR controls, was conducted following methods described in20. Blank extractions consisted of 5 ml or 50 ml tubes filled with 100% EtOH, and 1 ml or 4 ml sub-samples, respectively, processed as described above. Positive controls consisted of one to two 8-day old CoTS larvae added to 1 ml and 4 ml sub-samples and processed as a sample. Extraction controls (total: positive, n = 21; negative, n = 33) and PCR controls (total: positive, n = 38; negative, n = 40) were run for each batch of extractions.
To prevent potential contamination of fish faecal and gut content samples during laboratory processing and analyses38, the following measures were implemented. All pre- and post-PCR activities were kept physically separate by having a dedicated PCR room and equipment. For each batch of extractions, work-space and equipment used for sample processing such as pipettes were DNA cleaned (LookOut DNA Erase, Sigma). Implements used for sample processing such as tweezers and forceps were exposed to 100% bleach for at least 30 min, washed in Milli Q and dried in 100% EtOH prior to subsequent use on individual samples.
Data availability
All data needed to evaluate the conclusions in the paper are present in the paper and/or the Supplementary Information. Additional data related to this paper may be requested from the authors.
Change history
28 October 2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Pratchett, M., Caballes, C. F., Rivera-Posada, J. A. & Sweatman, H. P. A. Limits to our understanding and managing outbreaks of Crown-of-Thorn Starfish (Acanthaster spp.). Oceanogr. Mar. Biol. Annu. Rev. 52, 133–200 (2014).
Bruno, J. F. & Selig, E. R. Regional decline of coral cover in the Indo-Pacific: Timing, extent, and subregional comparisons. Plos one 2, e711 (2007).
De’ath, G., Fabricius, K., Sweatman, H. & Puotinen, M. The 27–year decline of coral cover on the Great Barrier Reef and its causes. Proc. Natl Acad. Sci. USA 109, 17995–17999 (2012).
Emslie, M. Mixed bill of health for the Great Barrier Reef. 11 (Australian Institute of Marine Science, Townsville, Australia, 2019).
Hughes, T. P. et al. Global warming and recurrent mass bleaching of corals. Nature 543, 373–377 (2017).
Westcott, D. A. et al. Coral cover increases following manual control of Crown-of-Thorns Starfish outbreaks on Australia’s Great Barrier Reef. Sci Rep (In review).
Condie, S. A. et al. Intervention on the Great Barrier Reef can delay decline. Nat Clim (In review).
Birkeland, C. Terrestrial runoff as a cause of outbreaks of Acanthaster planci (Echinodermata: Asteroidea). Mar. Biol. 69, 175–185 (1982).
Fabricius, K. E., Okaji, K. & De’ath, G. Three lines of evidence to link outbreaks of the crown-of-thorns seastar Acanthaster planci to the release of larval food limitation. Coral Reefs 29, 593-593-605 (2010).
Wolfe, K., Graba-Landry, A., Dworjanyn, S. A. & Byrne, M. Larval starvation to satiation: Influence of nutrient regime on the success of Acanthaster planci. PloS one 10 (2015).
Sweatman, H. No-take reserves protect coral reefs from predatory starfish. Curr. Biol. 18, R598–R599 (2008).
Vanhatalo J, Hosack GR, Sweatman H. Spatiotemporal modelling of crown-of-thorns starfish outbreaks on the Great Barrier Reef to inform control strategies. J Appl Ecol, https://doi.org/10.1111/1365-2664.12710 (2016).
Mellin C, MacNeil AM, Cheal AJ, Emslie MJ, Caley JM. Marine protected areas increase resilience among coral reef communities. Ecol Lett, https://doi.org/10.1111/ele.12598 (2016).
Dulvy, N. K., Freckleton, R. P. & Polunin, N. V. C. Coral reef cascades and the indirect effects of predator removal by exploitation. Ecol. Lett. 7, 410–416 (2004).
Endean R. Report on investigations made into aspects of the current Acanthaster planci (Crown of Thorns) infestations of certain reefs of the Great Barrier Reef. 35 (Queensland Department of Primary Industries (Fisheries Branch), Brisbane, Australia, 1969).
Cowan ZL, Pratchett MS, Messmer V, Ling S. Known predators of crown-of-thorns starfish (Acanthaster spp.) and their role in mitigating, if not preventing, population outbreaks. Diversity (2017).
McCallum, H. I. Predator regulation of Acanthaster planci. J. Theor. Biol. 127, 207–220 (1987).
Antonelli P, Bradbury R, Hammond L, Ormond R & Reichelt R. The Acanthaster phenomenon: A modelling approach rapporteurs’ report. In: Acanthaster and the Coral Reef: A Theoretical Perspective (ed Bradbury R). Vol. 88 Lecture Notes in Biomathematics (ed Roger Bradbury) Ch. 20, 329–338 (Springer Berlin Heidelberg, 1990).
Condie, S. A., Plaganyi, E. E., Morello, E. B., Hock, K. & Beeden, R. Great Barrier Reef recovery through multiple interventions. Conserv. Biol. 32, 1356–1367 (2018).
Uthicke, S., Lamare, M. & Doyle, J. R. eDNA detection of corallivorous seastar (Acanthaster cf. solaris) outbreaks on the Great Barrier Reef using digital droplet CR. Coral Reefs 37, 1229–1239 (2018).
Uthicke, S. et al. Spawning time of Acanthaster cf. solaris on the Great Barrier Reef inferred using qPCR quantification of embryos and larvae: do they know it’s Christmas? Mar. Biol. 166, 1–10 (2019).
Pearson, R. G. & Endean, R. A preliminary study of the coral predator Acanthaster planci (L.) (Asteroidea) on the Great Barrier Reef. Fish. Notes 3, 27–68 (1969).
Keesing, J. K. & Halford, A. R. Importance of postsettlement processes for the population dynamics of Acanthaster planci (L.). Austr J. Mar. Freshw. Res. 43, 635–651 (1992).
Cowan ZL, Dworjanyn SA, Caballes CF, Pratchett MS. Predation on crown-of-thorns starfish larvae by damselfishes. Coral Reefs, 1–10 (2016).
Cowan, Z. L., Ling, S. D., Dworjanyn, S. A., Caballes, C. F. & Pratchett, M. S. Interspecific variation in potential importance of planktivorous damselfishes as predators of Acanthaster sp eggs. Coral Reefs 36, 653–661 (2017).
Cowan, Z. L., Ling, S. D., Caballes, C. F., Dworjanyn, S. A. & Pratchett, M. S. Crown-of-thorns starfish larvae are vulnerable to predation even in the presence of alternative prey. Coral Reefs 39, 293–303 (2020).
Marine Bio Logic. Survey of marine scientists and other experts for anecdotal observations of Crown of Thorns predation. 56 (Great Barrier Reef Marine Park Authority, Townsville, Australia, 1991).
Randall, J. E., Head, S. M. & Sanders, A. P. L. Food habits of the giant humphead wrasse, Cheilinus undulatus (Labridae). Env. Biol. Fishes 3, 235–238 (1978).
Ormond RF, Campbell AC. Formation and breakdown of Acanthaster planci aggregations in the Red Sea. In: Proceedings of the 2nd International Coral Reef Symposium, Vol. 1 (eds. Cameron, A. M. et al.) 595–620 (The Great Barrier Reef Committee, Brisbane, Australia, 1974).
Birdsey R. Large reef fishes as potential predators of Acanthaster planci: A pilot study by alimentary tract analysis of predatory fishes from reefs subject to Acanthaster feeding. 37 (Great Barrier Marine Park Authority, Townsville, Australia, 1988).
Endean, R. Destruction and recovery of coral reef communities. In: Biology and geology of coral reefs (eds OA Jones & R Endean) 215-254 (Academic Press, New York, USA, 1976).
Endean R. Acanthaster planci infestations of reefs of the Great Barrier Reef. In: Proceedings of Third International Coral Reef Symposium, Vol. 1 (ed. Taylor, D. L.) 185–191 (Miami, Florida, USA, 1977).
Owens, D. Acanthaster planci starfish in Fiji: survey of incidence and biological studies. Fiji Agr. J. 33, 15–23 (1971).
Wilmes, J. C., Hoey, A. S., Messmer, V. & Pratchett, M. S. Incidence and severity of injuries among juvenile crown-of-thorns starfish on Australia’s Great Barrier Reef. Coral Reefs 38, 1187–1195 (2019).
Budden, C. et al. Effect of sublethal predation on reproductive output of the crown-of-thorns starfish Acanthaster sp., with an overview of arm damage. Mar. Ecol. Prog. Ser. 629, 103–116 (2019).
Leray, M. et al. A new versatile primer set targeting a short fragment of the mitochondrial COI region for metabarcoding metazoan diversity: application for characterizing coral reef fish gut contents. Front. Zool. 10, 34 (2013).
St John, J. Ontogenetic changes in the diet of the coral reef grouper Plectropomus leopardus (Serranidae): patterns in taxa, size and habitat of prey. Mar. Ecol. Prog. Ser. 180, 233–246 (1999).
Thomsen, P. F. & Willerslev, E. Environmental DNA – An emerging tool in conservation for monitoring past and present biodiversity. Biol. Conserv. 183, 4–18 (2015).
Sweatman H. Commercial fishes as predators of adult Acanthaster planci. In Proceedings of the 8th International Coral Reef Symposium, Vol. 1 (eds HA Lessios & IG Macintyre) 617–620 (Panama, 1997).
Mendonça, V. M. et al. Persistent and expanding population outbreaks of the corallivorous starfish Acanthaster planci in the northwestern Indian Ocean: are they really a consequence of unsustainable starfish predator removal through overfishing in coral reefs, or a response to a changing environment? Zool. Stud. 49, 108–123 (2010).
Rivera-Posada, J., Pratchett, M. S., Aguilar, C., Grand, A. & Caballes, C. F. Bile salts and the single-shot lethal injection method for killing crown-of-thorns sea stars (Acanthaster planci). Ocean. Coast. Manage. 102, 383–390 (2014).
Rivera-Posada J, Caballes CF, Pratchett MS. Size-related variation in arm damage frequency in the crown-of-thorns sea star, Acanthaster planci. J Coast Life Med, https://doi.org/10.12980/jclm.2.2014j52 (2014).
Boström-Einarsson, L. & Rivera-Posada, J. Controlling outbreaks of the coral-eating crown-of-thorns starfish using a single injection of common household vinegar. Coral Reefs 35, 223–228 (2016).
Boström-Einarsson, L., Bonin, M. C., Moon, S. & Firth, S. Environmental impact monitoring of household vinegar-injections to cull crown-of-thorns starfish, Acanthaster spp. Ocean. Coast. Manage 155, 83–89 (2018).
Jarrett, K. W., Bell, E., Wilson, E. A., Dudley, T. & Geraghty, C. M. Using eDNA to validate predation on native Oncorhynchus mykiss by invasive Sacramento pikeminnow (Ptychocheilus grandis). Calif. Fish. Game 105, 177–187 (2019).
De’ath, G. & Moran, P. J. Factors affecting the behaviour of crown-of-thorns starfish (Acanthaster planci L.) on the Great Barrier Reef: 1: Patterns of activity. J. Exp. Mar. Biol. Ecol. 220, 83–106 (1998).
Sano M, Shimizu M, Nose Y. Food habits of teleostean reef fishes in Okinawa Island, southern Japan. In: University of Tokyo Bulletin, no. 25. University of Tokyo Press, Tokyo, Japan, http://umdb.um.u-tokyo.ac.jp/DKankoub/Bulletin/no25/no25000.html (1984).
Doyle, J. R., McKinnon, A. D. & Uthicke, S. Quantifying larvae of the coralivorous seastar Acanthaster cf. solaris on the Great Barrier Reef using qPCR. Mar. Biol. 164, 176 (2017).
Cheal, A. J., Wilson, S. K., Emslie, M. J., Dolman, A. M. & Sweatman, H. Responses of reef fish communities to coral declines on the Great Barrier Reef. Mar. Ecol. Prog. Ser. 372, 211–223 (2008).
Department of Primary Industries and Fisheries. A guide to the Queensland Marine Aquarium Fish Fishery and the Queensland Coral Fishery. 16 (Queensland Department of Employment, Economic Development and Innovation, Brisbane, Australia, 2009).
Ryan S & Clarke K. Ecological assessment of the Queensland Marine Aquarium Fish Fishery. A report to the Australian Government Department of Environment and Heritage on the ecologically sustainable management of the Queensland marine aquarium harvest fishery. 78 (Queenland Government, Brisbane, Australia, 2005).
Department of the Environment and Heritage. Assessment of the Queensland Marine Aquarium Fish Fishery. 25 (Australian Department of the Environment and Heritage, Canberra, Australia, 2005).
Department of the Environment and Energy. Assessment of the Queensland Coral Reef Fin Fish Fishery. 39 (Australian Department of the Environment and Energy, Canberra, Australia, 2017).
Steven A. An analysis of fishing activities on possible predators of crown of thorn starfish (Acanthaster planci) on the Great Barrier Reef. Unpublished report to Great Barrier Reef Marine Park Authority. (Project 329: 1986-88). 131 (Great Barrier Reef Marine Park Authority, Townsville, Australia, 1988).
Department of Agriculture and Fisheries. Queensland Fisheries Summary. 62 (Queensland Department of Agriculture and Fisheries, Brisbane, Australia, 2018).
Newman, S. et al. Spangled Emperor (2018), http://www.fish.gov.au/report/228-Spangled-Emperor-2018 (Fisheries Research and Development Corporation, Canberra, Australia, 2018).
Webley J, McInnes K, Teixeira D, Lawson A, Quinn R. Statewide Recreational Fishing Survey 2013–14. 145 (Queensland Department of Agriculture and Fisheries, Brisbane, Australia, 2015).
Henry GW, Lyle JM. The national recreational and indigenous fishing survey. 190 (Australian Government Department of Agriculture, Fisheries and Forestry, 2003).
Emslie, M. J. et al. Expectations and outcomes of reserve network performance following re-zoning of the Great Barrier Reef Marine Park. Curr. Biol. 25, 983–992 (2015).
Barnes, J. H. The crown-of-thorns starfish as a destroyer of coral. Aust. Nat. Hist. 15, 257–261 (1966).
Endean, R. Crown-of-thorns starfish on the Great Barrier Reef. Endeavour 6, 10–14 (1982).
Wachenfeld DR, Oliver JK, Morrissey JI. State of the Great Barrier Reef World Heritage Area. 139 (Great Barrier Reef Marine Park Authority, Townsville, Australia, 1998).
Hock K. et al. Controlling range expansion in habitat networks by adaptively targeting source populations. Conserv Biol, https://doi.org/10.1111/cobi.12665 (2015).
Hock, K. et al. Connectivity and systemic resilience of the Great Barrier Reef. Plos Biol. 15, e2003355 (2017).
Department of Agriculture and Fisheries. Queensland Sustainable Fisheries Strategy 2017–2027. 32 (Queensland Government, Brisbane, Australia, 2017).
Department of the Environment. Great Barrier Reef Marine Park Act 1975. 210 (Australian Department of the Environment, Canberra, Australia, 2015).
UNESCO. World Heritage Committee Fifth Session. 1-14 (UNESCO World Heritage Committee, Paris, France, 1981).
GBRMPA. Great Barrier Reef Outlook Report 2019. 374 (Great Barrier Reef Marine Park Authority, Townsville, Australia, 2019).
GBRMPA. Great Barrier Reef Marine Park zoning plan 2003. 220 (Great Barrier Reef Marine Park Authority, Townsville, Australia, 2004).
Queensland Government. Marine Parks (Great Barrier Reef Coast) Zoning Plan 2004. Current as at 1 September 2017. 148 (Queensland Government, Brisbane, Australia, 2004).
McCook, L. J. et al. Adaptive management of the Great Barrier Reef: A globally significant demonstration of the benefits of networks of marine reserves. Proc. Natl Acad. Sci. USA 107, 18278–18285 (2010).
AIMS. Long-term Reef Monitoring Program - Annual Summary Report on coral reef condition for 2019. 11 (Australian Institute of Marine Science, Townsville, Australia, 2019).
Sweatman, H. et al. Long-term monitoring of the Great Barrier Reef. Status report number 3. (Australian Institute of Marine Science, Townsville, Australia, 1998).
Pratchett, M. S. Dynamics of an outbreak population of Acanthaster planci at Lizard Island, northern Great Barrier Reef (1995–1999). Coral Reefs 24, 453–462 (2005).
Kenchington, R. A. Growth and recruitment of Acanthaster planci (L.) on the Great Barrier Reef. Biol. Conserv. 11, 103–118 (1977).
Reichelt, R. E., Bradbury, R. H. & Moran, P. J. Distribution of Acanthaster planci outbreaks on the Great Barrier Reef between 1966 and 1989. Coral Reefs 9, 97–103 (1990).
Moran, P. J. et al. Pattern of outbreaks of crown-of-thorns starfish (Acanthaster planci L.) along the Great Barrier Reef since 1966. Aust. J. Mar. Freshw. Res. 43, 555 (1992).
Froese R, Pauly D. FishBase 2000: concepts, design and data sources. 344 (Los Baños, Laguna, Philippines, 2000).
GBRMPA. Crown-of-thorns starfish control program. Great Barrier Reef Marine Park Authority, Townsville, Australia, http://www.gbrmpa.gov.au/our-work/our-programs-and-projects/crown-of-thorns-starfish-control-program (2019).
Glynn, P. W. An amphinomid worm predator of the crown-of-thorns sea star and general predation on asteroids in eastern and western Pacific coral reefs. Bull. Mar. Sci. 35, 54–71 (1984).
Moran, P. J. Preliminary observations of the decomposition of crown-of-thorns starfish, Acanthaster planci (L.). Coral Reefs 11, 115–118 (1992).
Ormond, R. F. G. et al. Formation and breakdown of aggregations of the Crown-of-Thorns Starfish, Acanthaster planci (L.). Nature 246, 167–169 (1973).
Sweatman, H. P. A. A field study of fish predation on juvenile crown-of-thorns starfish. Coral Reefs 14, 47–53 (1995).
Ormond, R. et al. Test of a model of regulation of Crown-of-Thorns starfish by fish predators. In Acanthaster and the Coral Reef: A Theoretical Perspective Vol. 88 Lecture Notes in Biomathematics (ed. Bradbury, R.) Ch. 12, 189-207 (Springer Berlin Heidelberg, Germany,1990).
Lucas J. S. Environmental influences on the early development of Acanthaster planci (L.). In Crown-of-Thorns Starfish Seminar Proceedings. 109-121 (Australian Government Publishing Service, 1975).
Wilson, B. R. & Marsh, L. M. B. H. Puffer fish predator of crown of thorn starfish in Australia. Search 5, 601–602 (1974).
De’ath, G. Analyses of crown-of-thorns starfish data from the fine-scale surveys and the long-term monitoring program manta tow surveys. 75 (CRC Reef Research Centre Ltd, Townsville, Australia, 2003).
Acknowledgements
We appreciate the excellent facilities and support provided by the RV Cape Ferguson, the National Sea Simulator, and the Australian Museum’s Lizard Island Research Station. We acknowledge GBRMPA’s Eye on the Reef for data on CoTS presence, abundance and culling. This study was conducted with permission from the GBRMPA, Queensland Department of Agriculture and Fisheries and James Cook University Animal Ethics Committee. This study and its publication were supported with funding from the Ian Potter Foundation 50th Anniversary Commemorative Grants Scheme; the Lizard Island Reef Research Foundation; and the Australian Government’s National Environmental Science Program’s Tropical Water Quality Hub.
Author information
Authors and Affiliations
Contributions
F.J.K. conceived, designed and led the study, and wrote the paper. C.D.L. and F.J.K. conducted the two literature reviews. C.D.L. and C.L.J. organised collection and animal ethics permits, and field logistics. F.J.K., C.D.L., J.R.D., G.M., A.S., M.K., P.T.H. and D.A.W. collected coral reef fish. G.M., A.S., P.T.H. and J.R.D. developed, tested and designed the filtration skid. J.R.D., F.P., C.L.J. and S.S. extracted and analysed samples for CoTS DNA. M.B. and D.S.C. contributed information on the status of CoTS outbreaks and the CoTS control program. All authors contributed to the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kroon, F.J., Lefèvre, C.D., Doyle, J.R. et al. DNA-based identification of predators of the corallivorous Crown-of-Thorns Starfish (Acanthaster cf. solaris) from fish faeces and gut contents. Sci Rep 10, 8184 (2020). https://doi.org/10.1038/s41598-020-65136-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-65136-4
This article is cited by
-
Novel rubble-dwelling predators of herbivorous juvenile crown-of-thorns starfish (Acanthaster sp.)
Coral Reefs (2023)
-
Fish predators control outbreaks of Crown-of-Thorns Starfish
Nature Communications (2021)
-
Relative efficacy of three approaches to mitigate Crown-of-Thorns Starfish outbreaks on Australia’s Great Barrier Reef
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.