Abstract
A burgeoning number of studies are demonstrating aluminium in human brain tissue. While research has both quantified and imaged aluminium in human brain tissue in neurodegenerative and neurodevelopmental disease there are few similar data for brain tissue from non-neurologically impaired donors. We have used microwave assisted acid digestion and transversely heated graphite furnace atomic absorption spectrometry to measure aluminium in twenty brains from donors without recognisable neurodegenerative disease. The aluminium content of 191 tissue samples was invariably low with over 80% of tissues having an aluminium content below 1.0 μg/g dry weight of tissue. The data for these control tissues were compared with data (measured using identical procedures) for sporadic Alzheimer’s disease, familial Alzheimer’s disease, autism spectrum disorder and multiple sclerosis. Detailed statistical analyses showed that aluminium was significantly increased in each of these disease groups compared to control tissues. We have confirmed previous conclusions that the aluminium content of brain tissue in Alzheimer’s disease, autism spectrum disorder and multiple sclerosis is significantly elevated. Further research is required to understand the role played by high levels of aluminium in the aetiology of human neurodegenerative and neurodevelopmental disease.
Similar content being viewed by others
Introduction
Human exposure to aluminium is burgeoning and its entry into and presence within the human body is inevitable1,2,3. There is no ‘aluminium homeostasis’4. The bioinorganic chemistry of aluminium dictates that it will ‘piggy-back’ upon essential biochemistry and it is such adventitious interactions that determine its fate in the human body. The brain is a target tissue for accumulation of aluminium5,6. Long-lived neurones provide intracellular pools, such as citrate, ATP and glutamic acid, where aluminium can remain complexed without necessarily disrupting cellular biochemistry7. Aluminium is neurotoxic and is found in brain tissue in extracellular milieu associated with neuropathology including senile plaques and neurofibrillary tangles in Alzheimer’s disease8,9. While there is no longer any debate as to the presence of aluminium in human brain tissue, there remains the question of how much aluminium in brain tissue is too much10. A number of recent studies have provided data on aluminium content in brain tissue in Alzheimer’s disease11, multiple sclerosis12 and autism13. The quantitative data, supported by aluminium-specific imaging, are invariably reported as being high, higher than expected. However, data on the latter, true control data are extremely rare. Brain banks have themselves struggled with the concept of what constitutes a true control14. We asked one such brain bank to identify a set of donor brain tissues that could act as a control for brains affected and diagnosed with a neurodegenerative disease. The majority of control brains available through brain banks are from older donors and so most still show some signs of age-related degeneration. Herein we have measured the aluminium content of twenty control brains where in each case there was no overt neurodegeneration, no diagnosis of a neurodegenerative disease but some age-related changes in the older donors. We have then compared these data with data, measured under identical conditions, for donors having died with diagnoses of Alzheimer’s disease, multiple sclerosis and autism.
Results
Control brain tissues
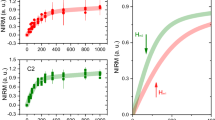
The aluminium content of all tissues ranged from 0.01 (the limit of quantitation) to 9.28 μg/g dry wt. (Table 1). The majority of tissues (150 out of 191) were below 1.00 μg/g dry wt. though 28, 6 and 7 tissues were in the range 1.00–1.99, 2.00–2.99 and ≥3.00 μg/g dry wt. respectively. The aluminium content of each lobe (mean and SD) were 1.03 (1.64), 1.02 (1.27), 0.95 (0.88), 0.77 (0.92) and 0.51 (0.51) μg/g dry wt. for frontal, temporal, parietal, occipital and cerebellum respectively. There were no statistically significant differences between aluminium content and age (p-value = 0.7656) or gender (p-value = 0.4005) and even though the cerebellum had the lowest content of aluminium, there were no statistically significant differences between any of the five brain regions (p-value = 0.2488; Table 2; Fig. 1).
Aluminium content, μg/g dry wt., of brain tissue for each brain region of the twenty donor controls, comprising 5 males and 15 females. Mean and one standard deviation are indicated. For primary data, please see Table 1.
Comparison with disease groups
We compared control brain data with each of four treatment groups, namely sporadic Alzheimer’s disease (sAD), familial Alzheimer’s disease (fAD), autism spectrum disorder (ASD) and multiple sclerosis (MS). The descriptive statistics for each of these groups are shown in Table 3. The sAD group is actually composed of both control donors and donors diagnosed with sporadic Alzheimer’s disease, an approximate 50/50 split, see later for a more detailed explanation of this. In addition this group has been analysed according to how negative values in the original data set are dealt with, for example; sAD- sporadic Alzheimer’s disease (including negative values); sAD+- sporadic Alzheimer’s disease (negative values adjusted to LOQ); sAD– - sporadic Alzheimer’s disease (negative values excluded).
Treatment group (sAD; sAD–; sAD + ; fAD; ASD; MS) was the only factor that consistently affected the aluminium content of donor brain tissue. This was the case for all statistical analyses carried out including; when units were unweighted observations or means of individuals; for both adjustments sAD+ and sAD–; for truncation of outliers; parametric or non-parametric procedures (Table 4). The aluminium content of brain tissue in the control group was significantly lower than sAD (P = 0.0006), fAD (P = 0.0020), ASD (P = 0.0123) and MS (P < 0.0001) (Fig. 2).
Statistical comparisons, using both mean of observations and mean of individuals, between aluminium content, μg/g dry wt., of brain tissue in control and disease groups. Mean and 95% confidence intervals are indicated. For descriptive statistics, please see Table 3.
Discussion
We present the first comprehensive data set for the aluminium content of brain tissue in donors without a diagnosis of neurodegenerative disease. All donors fulfilled recently revised criteria for control brain tissues14. Approximately 80% of measured tissues have an aluminium content below 1.0 μg/g dry wt. (Table 1). There are some anomalies, 6 out of 191 tissues have an aluminium content ≥3.00 μg/g dry wt., and these are worth future investigation to identify possible neuropathology. There was no statistically significant relationship between brain aluminium content and age of donor and this observation is contrary to a previous investigation of brain aluminium in a neurologically normal population15. An explanation may be that herein only two out of twenty donors were below 66 years old. The data do support a conclusion that a high content of brain aluminium is not an inevitability of ageing.
When we compared the new control data set with data produced in an identical manner in donors dying with diagnoses of sporadic Alzheimer’s disease (sAD)16, familial Alzheimer’s disease (fAD)11, autism spectrum disorder (ASD)13 and multiple sclerosis (MS)12 all of these disease groups had significantly higher brain aluminium content. The differences were always highly significant regardless of the method of statistical analysis (Table 4). The largest disease group, designated as sAD, was actually composed of approximately equal numbers of donors previously described by a brain bank as controls and donors diagnosed with sAD. Unfortunately, information discriminating between control and sAD donors was not made available to us17. However, the observation that the aluminium content of brain tissue in this group as a whole was significantly higher than the similarly aged control group emphasised the likelihood that brain aluminium content is increased in sAD. The data for the control group demonstrate that high content of brain aluminium is not an inevitability of living in the aluminium age. All disease groups had significantly higher brain aluminium content than the control group in spite of low numbers of donor brains, for example only 5 in ASD, and high variability within measured tissues. The disease groups, sAD, fAD, ASD and MS shared the characteristics of significant focal deposits of aluminium throughout all main lobes of the brain and associated neuropathology and neurodegeneration. Quantitative data, even when complemented with high quality aluminium-specific fluorescence microscopy18, do not directly confirm a role for aluminium in each of these diseases. However, since there is no debate as to the neurotoxicity of aluminium in humans19,20,21, such data do implicate aluminium in disease aetiology. Animal models of aluminium intoxication reproduce the neuropathologies and neurodevelopmental effects of human neurodegenerative disease, if not the diseases per se22,23. Cell models and in vitro studies demonstrate mechanisms of aluminium toxicity known to be involved in human neurodegenerative disease24,25. Perhaps the information that is still missing from our understanding of aluminium’s role in each of the diseases compared herein is how much aluminium is too much in human brain tissue10. The comparison we have made herein between control brain tissue showing no signs of neurodegenerative disease and the disease groups sAD, fAD, ASD and MS is beginning to answer this question. Only further measurements on more donor brains will enable a definitive conclusion to be reached on the role played by aluminium in human neurodegenerative disease.
Aluminium is not a member of the human metallome4. However, its omnipresence in human tissue and especially the brain cannot be without consequence. It is only inimical to life, there is no homeostasis, and it is always a burden to life’s processes. Every atom of aluminium in human brain tissue must be accommodated as aluminium as Al3+(aq), is highly biologically reactive. Life is robust and some aluminium in human brain tissue is tolerated without overt effects. We need to define such limits in the terms of both quantity and location and we need to be more fully aware of human exposure to aluminium. We may then live healthily in the aluminium age (https://www.hippocraticpost.com/mens-health/the-aluminium-age/).
Methods
Tissues
Brain tissues were obtained from the London Neurodegenerative Diseases Brain Bank following ethical approval (NRES Approval No. 08/MRE09/38+5). Donor brains were chosen on our behalf by the consultant neuropathologist at the brain bank. All had a clinical diagnosis of ‘control’ while some had a pathological diagnosis that included age-related changes in tissue. There were five male and fifteen female donors. They were aged between 47 and 105 years old. Tissues were obtained from frontal, occipital, parietal and temporal lobes and cerebellum from all donors.
Quantitative measurements
The aluminium content of tissues was measured by an established and fully validated method16 that herein is described only briefly. Samples of cortex, approximately 1 g in weight, were thawed at room temperature and cut using a stainless steel blade into sections approximately 0.5 g in weight. Tissues were dried for 48 h to a constant weight in an incubator at 37 °C. Dry and thereafter weighed tissues were digested in a microwave (MARS Xpress CEM Microwave Technology Ltd.) in a mixture of 1 mL 15.8 M HNO3 (Fisher Analytical Grade) and 1 mL 30% w/v H2O2 (BDH Aristar). Resulting digests were clear with no fatty residues and, upon cooling, were made up to 5 mL volume using ultrapure water (cond. <0.067μS/cm). Total aluminium was measured in each sample by transversely heated graphite furnace atomic absorption spectrometry (TH GFAAS) using matrix-matched standards and an established analytical programme alongside previously validated quality assurance data. The latter included method blanks, detailed descriptions of which have been published recently10,16.
Statistical analyses
Comparisons between brain lobes were analysed using control group data
The control group dataset was well balanced with repeated measurements. Analyses were performed using the SAS general linear models (GLM) procedure, including gender, age and lobes as factors. Observations (OB) was the unit of analysis. The programme and raw data are available upon request. We considered a p-value smaller than 0.05 to be statistically significant using the model shown below.
Analysis by disease group
The distribution of aluminium content data is heavily skewed in the treatment groups. Data are not balanced with the number of observations and their respective lobe varying considerably between treatment groups. There is large variability in repeated measurements taken from the same donor. Analyses were performed for both the unweighted observations, and means across all lobes for each individual.
The assumption that the data across all groups are normally distributed, an assumption that underlies any ANOVA model, is questionable at best. A non-parametric approach was used to avoid this assumption. For the non-parametric tests, the SAS NPAR1WAY procedure was used, with the Dwass, Steel, Critchlow-Fligner Method for two-way comparisons between the different disease categories.
Null and Alternative hypothesis
H0:: µi = µj for all i≠j (No differences between means of control and disease groups)
H1: µi≠µj for some i≠j (At least one difference between means of control and disease groups)
Change history
10 June 2020
The HTML version of this Article previously published contained corrupted equations. This has now been corrected in the HTML version of the Article; the PDF version was correct at time of publication.
References
Exley, C. Human exposure to aluminium. Environ. Sci: Processes Impacts 15, 1807–1816 (2013).
Klotz, K. et al. The health effects of aluminium exposure. Deutsches Arzteblatt Int. 114, 653 (2017).
Stahl, T., Falk, S., Taschan, H., Boschek, B. & Brunn, H. Evaluation of human exposure to aluminium from food and food contact materials. Eur. Food Res. Technol. 244, 2077–2084 (2018).
Exley, C. Darwin, natural selection and the biological essentiality of aluminium and silicon. Trends Biochem. Sci. 34, 589–593 (2009).
Bondy, S. C. Low levels of aluminium can lead to behavioral and morphological changes associated with Alzheimer’s disease and age-related neurodegeneration. Neurotoxicology 15, 222–229 (2016).
Exley, C. Why industry propaganda and political interference cannot disguise the inevitable role played by human exposure to aluminium in neurodegenerative diseases including Alzheimer’s disease. Front. Neurol. 5, 212 (2014).
Exley, C. & House, E. Aluminium in the human brain. Monat. Chem. – Chem. Monthly 142, 357–363 (2011).
Bondy, S. C. The neurotoxicity of environmental aluminium is still an issue. Neurotoxicology 31, 575–81 (2010).
Exley, C. What is the risk of aluminium as a neurotoxin? Expert Rev. Neurother. 14, 589–591 (2014).
Exley, C. & Mold M. J. Aluminium in human brain tissue: How much is too much? J. Biol. Inorg. Chem., https://doi.org/10.1007/s00775-019-01710-0 (2019)
Mirza, A., King, A., Troakes, C. & Exley, C. Aluminium in brain tissue in familial Alzheimer’s disease. J. Trace Elem. Med. Biol. 40, 30–36 (2017).
Mold, M. et al. Aluminium in brain tissue in multiple sclerosis. Int. J. Environ. Res. Public Health 15, 1777 (2018).
Mold, M., Umar, D., King, A. & Exley, C. Aluminium in brain tissue in autism. J. Trace Elem. Med. Biol. 46, 76–82 (2018).
Nolan, M., Troakes, C., King, A., Bodi, I. & Al-Sarraj, S. Control tissue in brain banking: the importance of thorough neuropathological assessment. J. Neural. Transm. 122, 949–956 (2015).
Roider, G. & Drasch, G. Concentration of aluminium in human tissue – investigations on an occupationally non-exposed population in southern Bavaria, Germany. Trace Elem. Electrolytes 16, 77–86 (1999).
House, E., Esiri, M., Forster, G., Ince, P. G. & Exley, C. Aluminium, iron and copper in human brain tissues donated to the medical research council’s cognitive function and ageing study. Metallomics 4, 56–65 (2012).
Exley, C., House, E., Polwart, A. & Esiri, M. M. Brain burdens of aluminium, iron and copper and their relationships with amyloid-β pathology in 60 human brains. J. Alzh. Dis. 31, 725–730 (2012).
Mirza, A., King, A., Troakes, C. & Exley, C. The identification of aluminium in human brain tissue using lumogallion and fluorescence microscopy. J. Alzh. Dis. 54, 1333–1338 (2016).
Polizzi, S. et al. Neurotoxic effects of aluminium among foundry workers and Alzheimer’s disease. Neurotoxicology 23, 761–774 (2002).
Meyer-Baron, M., Schaper, M., Knapp, G. & van Thiel, C. Occupational aluminium exposure: evidence in support of its neurobehavioural impact. Neurotoxicology 28, 1068–1078 (2007).
Exley, C. The toxicity of aluminium in humans. Morphologie 100, 51–55 (2016).
Pratico, D. et al. Aluminum modulates brain amyloidosis through oxidative stress in APP transgenic mice. FASEB J. 16, 1138–1140 (2002).
Oshima, E. et al. Accelerated tau aggregation, apoptosis and neurological dysfunction caused by chronic oral administration of aluminium in a mouse model of tauopathies. Brain Pathol. 23, 633–644 (2013).
Strong, M. J., Garruto, R. M., Joshi, J. G., Mundy, W. R. & Shafer, T. J. Can the mechanisms of aluminium neurotoxicity be integrated into a unified scheme? J. Tox. Environ. Health 48, 599–613 (1996).
Exley, C. The pro-oxidant activity of aluminium. Free Radic. Biol. Med. 36, 380–387 (2004).
Acknowledgements
There were no direct funding sources involved in this research. CE is thankful for multiple donations to his research group from private individuals enabling the purchase of brain tissues and their processing and measurement. CE would like to thank A. King, consultant neuropathologist at The London Neurodegenerative Diseases Brain Bank for selecting the donor brain tissues for this study.
Author information
Authors and Affiliations
Contributions
C.E. conceived the study and carried out the measurements of aluminium in brain tissue. E.C. carried out all statistical analyses. C.E. and E.C. wrote and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Exley, C., Clarkson, E. Aluminium in human brain tissue from donors without neurodegenerative disease: A comparison with Alzheimer’s disease, multiple sclerosis and autism. Sci Rep 10, 7770 (2020). https://doi.org/10.1038/s41598-020-64734-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-64734-6
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.