Abstract
The purpose of this study was to evaluate the effects of Spirulina Platensis supplementation on selected blood markers of oxidative stress, muscle damage, inflammation, and performance in trained rats. Rats (250 g - 300 g) were submitted to a strength training program (eight weeks), divided into four groups: control (GT) (trained without supplementation), trained with daily-supplementation of 50 mg/kg (GT50), 150 mg/kg (GT150) and 500 mg/kg (GT500). Training consisted of a jump protocol in PVC-cylinder containing water, with increasing load over experimental weeks. We evaluated the markers of oxidative stress (malondialdehyde - MDA and antioxidant capacity) and inflammation (C-reactive protein) at the end of the training. Among groups submitted to strength training, concentration of C-reactive protein decreased after 8 weeks of intervention in the trained group and GT500. Strength training enhanced plasma MDA concentration of malondialdehyde with supplementation of S. platensis in GT150 and GT500. In plasma analysis, strength training enhanced the percentage of oxidation inhibition, with spirulina supplementation in rates of 150 and 500 mg/kg. Spirulina supplementation for 8 weeks (in a dose-effect manner) improved antioxidant capacity as well as attenuated exercise-induced increases in ROS and inflammation. As a practical application, the use as high doses did not cause a reduction in positive physiological adaptations to exercise training. Additional studies are necessary to test the application of Spirulina Platensis in other contexts, as collective sports (basketball, football, soccer).
Similar content being viewed by others
Introduction
Spirulina platensis is a microalga with biological activity as antioxidant, immunomodulatory, and anti-inflammatory and nowadays is used to produce nutritional supplements1,2,3. S. platensis is composed of protein (55%–70%)4, carbohydrates (15%–20%)5, lipids (approximately 7%)5, fiber, ash, and water including various minerals, vitamins, γ-linolenic acid, chlorophyll, carotenoids, and phycocyanin2,6. Recently, some researchers have reported that the latter played a crucial role in the antioxidative action of S. platensis2.
In this way, many animal and human studies have reported possible beneficial effects of S. platensis under several diseases such as diabetes7, dyslipidemia8, and chronic obstructive pulmonary diseases9. In most cases, the decrease in the oxidative stress and the inflammatory process reportedly had beneficial effects2.
In addition to clinical settings, S. platensis has demonstrated promising results in an exercise context10. In India, athletes have been eating S. platensis while training for track and field events11. Chinese and Cuban Olympic teams are also known to eat S. platensis daily during their training12. It suggests some strength or exercise performance related to the effects of S. platensis supplementation in humans11. Concomitantly, we know that high volume and intensity of physical training imposes a challenge for athletes to modulate their immune system, as well as promotes an increase of reactive oxygen species (ROS) production11,13, that are associated with early fatigue. Due to its nutritional, immunosuppressive, and antioxidant properties, Spirulina platensis can protect against early fatigue onset.
In recent years, several studies have reported ergogenic effects in athletes using raw or unprocessed foods14. Evidence suggests that many phytochemicals (usually not yet fully known) present in these foods can act synergistically, increasing the effectiveness of physiological outcomes, and decreasing possible adverse effects, such as toxicity, and positive adaptations to training15. The latter decreased with supplementation (vitamin C, E, and Resveratrol)16,17,18. Thus, we can infer that S. platensis (that contains several compounds with bioactive properties and different mechanisms of action) can promote ergogenic action in the context of physical exercise with minimized negative effects.
To date, only aerobic exercises were used to analyze S. platensis supplementation on markers of oxidative stress, inflammation, muscle damage, and performance19,20. Previous studies could not answer whether the ergogenic effect of S. platensis is replicable in strength exercises. Therefore, this study aimed to investigate S. platensis supplementation on oxidative stress and inflammation and whether possible improvements in these variables would result in better performance in trained rats.
Methods
Preparation and administration of Spirulina platensis
Spirulina platensis in powder form was obtained from Bio-Engineering Dongtai Top Co. Ltd laboratory (Nanjing, China) (Lot n°. 20130320). The extract was certificated by Pharma Nostra Quality Control Laboratory (Anápolis-GO, Brazil) (Lot No. 1308771 A); and preparing lyophilized by Dilecta Manipulation (João Pessoa-PB, Brazil) (lot n°. 20121025). The preparation of each dose of S. platensis was daily dissolved in saline in the proportions of 0.005, 0.015 and 0.05 g/mL for the preparation of the doses of 50, 150 and 500 mg/kg, respectively, which were administered to animals at the end of the preparation. We delivered the supplements over a period of eight weeks for all doses (50, 150 and 500 mg/kg/day) (BD-12, Insight, Ribeirão Preto, SP, Brazil) adapted from (Juarez-Oropeza et al., 2007)21. The oral administration was done from 12 to 14 hours, using stainless steel gavage needles (BD-12, Insight, Ribeirão Preto, SP) and 10 mL disposable syringes with 0.2 mL accuracy (BD, HIGILAB, João Pessoa, PB). At the beginning of each week, the animals in each group were weighed in order to consider the average weight of each group to properly calculate the gavage volume for each dose, considering the average animal weight. For groups undergoing strength training, supplementation was administered 30 minutes before the exercise session.
Animals, ethical and experimental protocol
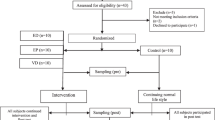
Wistar rats (Rattus norvegicus), weighing between 250 and 300 g, were obtained from Prof. Thomas George’s vivarium of the Center for Biotechnology (CBiotec/UFPB). The animals were kept under a balanced diet to feed base (Labina®) with free access to water. All experiments occurred from 08:00 h to 20:00 h, following guidelines for ethical animal use22, previously approved by the Ethics Committee of CBiotec (n° 0511/13). Forty animals were divided into groups subjected to the same strength training protocol and supplemented with S. platensis or saline (0.9% NaCl): trained group (GT; control group, n = 10), supplemented with saline, and trained group 50 (GT50, n = 10), (GT150, n = 10) and (GT500, n = 10), supplemented with S. platensis.
Strength training program
Animals from the strength training group underwent a specific jump protocol in a PVC cylinder with 30 cm in diameter and 70 cm length, containing water. Water depth was 50 cm, close to twice the rat height, to avoid the animals doing alternative climbing and clinging to the cylinder’s edge, not to perform the exercises. The water was at a temperature of around 32 °C23. Based on Marqueti et al. (2006)23, the protocol of strength training through jumps in the water consists of progressively increasing the load as it adjusts to body weight. Overload was applied to the animals’ chest through a special vest that did not disconnect or restrict movement during jumps. Two-hour sessions, from 12:00 to 14:00 h, took place three times a week on alternate days. During each exercise series, the analysis of the time taken by the animal to perform the exercise evaluated the effectiveness of exercise on muscle performance.
Strength training and overload adjustments were developed as follows (Table 1):
- 1.
Adaptation Week - performed with 50% of overload corresponding to animal body weight (1st day: 2 sets x 5 jumps; day 2: 4 sets x 5 jumps and day 3: 4 sets x 9 jumps) and a 30-second rest between sets
- 2.
1st and 2nd Weeks - 4 set of 10 jumps with and an overload corresponding to 50% and a 30-second rest between sets
- 3.
3rd and 4th weeks - 4 sets of 10 jumps, with a 30-second rest between sets and an overload corresponding to 60%.
- 4.
5th and 6th weeks - 4 sets of 10 jumps, with an overload corresponding to 80% and a 30-second rest between sets.
- 5.
7th and 8th weeks - 4 sets of 12 jumps with an overload of 80% and a 30-second rest between sets.
To evaluate exercise effectiveness on muscle strength, the runtime that the animal spent performing the same movement was assessed during each exercise. After 48 hours from the last training session and supplementing, animals were euthanized by cervical dislocation followed by cervical vessels section24.
Biochemical measurements
After euthanasia of the animals, we collected 3 mL of blood through cardiac puncture25. From it, 1 mL of blood was collected immediately for determination of C-reactive protein (CRP)26. The other 2 mL was placed in test tubes containing (EDTA) to determine nitrite, malondialdehyde (MDA), and antioxidant activity26. Samples were centrifuged in a CENTRIBIO centrifuge, model 80-2B-15ML (Guarulhos- SP, Brazil). The supernatant or serum was transferred to Eppendorf tubes and refrigerated at -20 °C until analysis.
The CRP activities assay followed the method described by the International Federation of Clinical Chemistry and Laboratory Medicine26, using the commercial kit Labtest (Minas Gerais, Brazil). Absorbance was read in an automatic analyzer, LabMax 240 premium, at 340 nm for CK and LDH, and 540 nm for CRP, at ambient temperature.
Assessment of malondialdehyde (MDA) levels
Samples of liver and quadriceps muscle were quickly removed, cleaned with Krebs solution to remove remaining blood, placed in Eppendorf tubes and stored in a freezer at -80 °C until analysis. After these tissues were homogenized with a 10% KCl ratio of 1:1, both 250 µL of plasma and homogenate were removed separately and incubated in a water bath at 37 °C for 60 minutes. Later, samples from both preparations were precipitated by Ohkawa et al.27. The material was read in a spectrophotometer (Biospectro, SP-220 model/Brazil) at 532 nm wavelength. A standard curve determined the MDA concentration in each plasma sample or tissue10. In tissues, absorbance values obtained were normalized by dry weight from the given sample volume.
Evaluation of antioxidant activity
The basis of the procedure was the method described by Brand-Williams et al.28. The supernatant was necessary for the reading in a spectrophotometer at 515 nm (Biospectro, model SP-220/Brazil). The results were expressed as a percentage of oxidation inhibition, wherein: AOA = 100 − ([DPPH•R]T/[DPPH•R]B 100). Where [DPPH•R]T and [DPPH • R]B corresponding to DPPH concentration remaining after 30 minutes, measured in the sample (T) and blank (B) prepared with distilled water.
Statistical analysis
Results were expressed as mean and standard deviation (SD). These results were analyzed statistically using analysis of variance (ANOVA) two-way followed by Bonferroni post-test, where the differences between means were considered significant when p ≤ 0.05. All results were analyzed by GraphPadPrism® program version 5.01 (GraphPad Software Inc., San Diego CA, USA).
Results
Effect of strength training and S. platensis supplementation on systemic inflammation level in rats
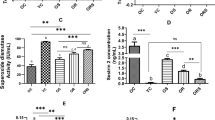
Among groups submitted to strength training, the concentration of C-reactive protein decreased after eight weeks of intervention in the trained group and supplemented with 500 mg/kg, but this decrease was significant when compared to the group submitted only to strength training (0.100 ± 0.011 vs. 0.154 ± 0.011 mg/dL, respectively). Trained group values supplemented with 50 mg/kg was pre (0.100 ± 0.011) vs. post 0.140 ± 0.03 mg/dL), and trained group values supplemented with 150 mg/kg was pre (0.100 ± 0.011) vs. post (0.140 ± 0.011 mg/dL). Thus, data reflect training the effectiveness as well as effectiveness of the most concentrated supplementation (Fig. 1).
C-reactive protein after eight weeks of exercise for the groups ( ) GT (
) GT ( ) and GT50 (
) and GT50 ( ), GT150 (
), GT150 ( ) and GT500 (
) and GT500 ( ). Vertical columns and bars represent mean and standard deviation, respectively (n = 8). “Two-way” ANOVA followed by Bonferroni post-test. *p ≤ 0.05 GT vs. GT500; #p ≤ 0.05 GT50 vs. GT500; †p ≤ 0.05 GT150 vs. GT500.
). Vertical columns and bars represent mean and standard deviation, respectively (n = 8). “Two-way” ANOVA followed by Bonferroni post-test. *p ≤ 0.05 GT vs. GT500; #p ≤ 0.05 GT50 vs. GT500; †p ≤ 0.05 GT150 vs. GT500.
Effect of strength training and Spirulina platensis supplementation on the production of malondialdehyde
Strength training enhanced the decreased plasma MDA concentration of malondialdehyde with S. platensis supplementation at rates of 150 and 500 mg/kg. Values decreased from 7.1 ± 1.0 (GT50) and 6.9 ± 2.0 (GT50) to 5.6 ± 0.4 (GT150) and 3.9 ± 0.4 (GT500) nmol/L, yet less than GT500 and GT150 (Fig. 3A). Unlike plasma, the liver, when the trained group was supplemented with 500 mg/kg, presented lower MDA production compared to sedentary group exposed to the same condition (8 ± 2 vs 18 ± 2 uM/g) (Fig. 3B). Similarly, in the quadriceps, GT500 showed a lower MDA production regarding WG (4 ± 2 vs 10 ± 2 uM/g) (Fig. 2).
Malondialdehyde concentration in blood plasma (A), liver (B) and quadriceps (C) obtained after eight weeks of treatment groups GT( ), GT50 (
), GT50 ( ), GT150 (
), GT150 ( ) and GT500 (
) and GT500 ( ). Vertical columns and bars represent mean and standard deviation, respectively (n = 8). *p≤ GT vs. GT150; GT vs. GT500; #p ≤ 0.05; GT50 vs. GT150; GT50 vs. GT500; †p < 0.01; GT150 vs. GT500.
). Vertical columns and bars represent mean and standard deviation, respectively (n = 8). *p≤ GT vs. GT150; GT vs. GT500; #p ≤ 0.05; GT50 vs. GT150; GT50 vs. GT500; †p < 0.01; GT150 vs. GT500.
Effect of strength training and Spirulina platensis supplementation in antioxidant activity determination
In plasma analysis, strength training enhanced the percentage of antioxidant activity with S. platensis supplementation in rates of 150 and 500 mg/kg. Values increased from 65 ± 2 (GT) and 67 ± 2 (GT50) to 82 ± 2 (GT150) and 93 ± 2 (GT500), though greater than GT500 and GT150. In the liver, the trained group supplemented with 150 mg/kg (68 ± 2%) and 500 mg/kg (78 ± 3%) showed a higher percentage of antioxidant activity compared to GT and GT50 (45 ± 2; 50 ± 2 uM/g). And in the quadriceps muscle, GT500 had a higher antioxidant activity percentage compared to GT, GT150, and GT150 (40 ± 3 vs. 18 ± 2, 20 ± 3 and 23 ± 2%) (Fig. 3).
Antioxidant activity percentage in blood plasma (A), liver (B) and quadriceps (C) obtained after eight weeks of treatment GT ( ) and GT50 (
) and GT50 ( ), GT150 (
), GT150 ( ) and GT500 (
) and GT500 ( ). Vertical columns and bars represent mean and standard deviation, respectively (n = 8).*p ≤ 0.05; GT vs. GT150; GT vs. GT500; #p ≤ 0.05; GT50 vs. GT150; GT50 vs. GT500; †p < 0.01; GT150 vs. GT500.
). Vertical columns and bars represent mean and standard deviation, respectively (n = 8).*p ≤ 0.05; GT vs. GT150; GT vs. GT500; #p ≤ 0.05; GT50 vs. GT150; GT50 vs. GT500; †p < 0.01; GT150 vs. GT500.
Discussion
To our knowledge, this is the first study to examine the effects of chronic Spirulina supplementation on markers of oxidative stress and inflammation related to resistance training. Results showed that Spirulina supplementation for eight weeks (in a dose-effect manner) induced a significant improvement in the antioxidant capacity (AC) as well as attenuated exercise-induced increases in ROS (measured by MDA) and inflammation (CRP). Our research group recently demonstrated that Spirulina supplementation for eight weeks induced significant improvement in exercise performance (time of execution) and decrease of muscle damage with the reduction of creatine kinase (CK) and lactated dehydrogenase (LDH) (Brito et al.)10.
The combination of phytochemicals can exhibit complementary and overlapping mechanisms of action, including antioxidant activity, and scavenging free radicals; modulation of enzymes activity in detoxification, oxidation, and reduction; and stimulation of the immune system and regulation of hormone metabolism29,30. Besides, undesirable effects such as induction of pro-oxidant states and decrease of mitochondrial biogenesis and myofibrillar hypertrophy have been recently demonstrated with dietary supplementation (isolated: vitamins C, E, and Resveratrol)31,32,33,34. By presenting various phytochemicals, especially phycocyanin, S. platensis shows great potential for its employment in sports, with possible deleterious effects minimized.
Dose analysis of dietary intervention is of most importance. In this context, only35 attempted to identify S. platensis dose-response. Our study confirms their findings since we observed that 500 mg/kg showed as more effective. Adverse effects have been observed with high doses of fish oil - inflammatory aspects36 and bulbine natalensis – hormonal aspects37. Thus, it appears that the use of S. platensis is a viable supplementation alternative regarding adverse events (with high doses). Further dose–response studies are required.
Supplements antioxidants may benefit exercise performance, directly by a muscle fatigue decrease and indirectly by a decrease of physiological stressors or improvement to recover from training33. Results found in human studies conducted on S. platensis chronic consumption demonstrated there were moderate performance improvements in quadriceps 1 and in one rat study34. It is necessary to recognize that these studies were carried out with aerobic exercises, while our study contemplated strength training protocol. Thus, our data demonstrate, in an unprecedented manner (as shown by execution time), that S. platensis supplementation may benefit strength athletes (Brito et al.)10.
Regarding muscle damage, our study showed that higher doses (500 mg/kg) decreased CK and LDH. Similarly, Lu et al38. demonstrated positive changes in plasma concentrations of MDA, SOD, LDH, and GPx after Spirulina supplementation for three weeks in untrained individuals and a single exercise session. On the other hand, Franca et al19. did not show any improvements in muscle damage biomarkers (in well trained cyclists). About that, some considerations are mandatory. Biomarkers are frequently thought in terms of muscle damage. However, they differ in time, course of appearance, and disappearance after exercise. So, a study design concerning the timeline for blood sampling must take into account the exercise type, duration, and intensity, diet, and if the subjects recruited are trained or untrained32.
The parameters evaluated in our study were inflammation and oxidative stress. In this context, the investigations showed that systemic inflammation could be one of the physiological stress markers in athletes12,39 because this process is required to promote training adaptations. Our study shows that both parameters improved with S. platensis supplementation. Therefore, other markers as cytokines and CRP were used as inflammatory markers for manuscripts previously to us. Although CRP has a fast response (1 to 2 days), it remains increased for short periods (5 to 6 days)40. In our study, we evaluated CRP and saw a reduction in this inflammatory marker. Regarding oxidative stress, we evaluated lipid peroxidation markers, the thiobarbituric acid reactive substances (TBARS). We observed a decrease in them, demonstrating an improvement in oxidative stress. That observation suggests that supplementation with Spirulina in athletes can promote a decrease in systemic inflammation.
Although not regarded in this study, the physiological alterations and mechanisms by which Spirulina promotes performance improvement need to be further investigated due to the lack of published studies. Some other possible mechanisms included: increased supply of cysteine (cysteine is generally the limiting amino acid for GSHx synthesis in humans and other animals), inhibition of NF-Kb, COX-2, and NADPH oxidase41,42,43. Possible mechanism of S. platensis in the improvement of oxidative stress has been demonstrated by McCarty 200743, where he showed that S. platensis inhibits NADPH oxidase, which is an enzyme that catalyzes the production of the superoxide free radical. Meanwhile, the reduction in the inflammatory process, as demonstrated in our study, was also seen in other studies, they showed that S. platensis reduces the production of proinflammatory cytokines such as TNF-α, IL-1β, and IL-6 by NF-κB pathway inhibition42,43.
Conclusions section
This study is the first to demonstrate the use of Spirulina platensis in the context of physical training (high-intensity exercise) since it is effective in improving the performance and reducing muscle damage, oxidative stress, and inflammation. The chronic nature of the study (8 weeks) differs from other studies in the literature that only verified the acute responses to an exercise session. Therefore, proving it to be a dietary strategy of high applicability in sport. It is noteworthy that the use of high doses did not cause a reduction of positive physiological adaptations to exercise training, a fact already seen with some supplements (vitamins C, E, and Resveratrol). Additional studies are necessary to test the application of Spirulina platensis in other contexts, such as collective sports (such as basketball, football, and soccer) and during low-calorie diets (bodybuilding athletes, martial arts, and gymnastics).
References
Kalafati, M. et al. Ergogenic and antioxidant effects of spirulina supplementation in humans. Med. Sci. Sports. Exerc. 42, 142–151 (2010).
Wu, Q., Liu, L., Klímová, B., Wan, D. & Kuca, K. The antioxidant, immunomodulatory, and anti-inflammatory activities of Spirulina: an overview. Arch. Toxicol. 90, 1817–1840 (2016).
Hoseini, S. M., Khosravi-Darani, K. & Mozafari, M. R. Nutritional and medical applications of spirulina microalgae. Mini. Rev. Med. Chem. 13, 1231–1237 (2013).
Phang, S. M. & Chu, W. L. University of Malaya Algae Culture Collection (UMACC). Catalogue of strains. Institute of Postgraduate Studies and Research. University of Malaya, Kuala Lumpur. (1999).
Mathur, M. Bioactive Molecules of Spirulina: A Food Supplement. In: Mérillon, J. M. & Ramawat, K. (eds) Bioactive Molecules in Food. Reference Series in Phytochemistry. Springer, Cham. (2018).
Soheili, M. & Khosravi-Darani, K. The potential health benefits of algae and micro algae in medicine: a review on Spirulina platensis. Curr. Nutr. Food. Sci 27, 279–285 (2011).
El-Sheekh, M. M., Daboor, S. M., Swelim, M. A. & Mohamed, S. Production and characterization of antimicrobial active substance from Spirulina platensis. Iran. J. Microbiol. 6, 112–119 (2014).
Mazokopakis, E. E., Starakis, I. K., Papadomanolaki, M. G., Mavroeidi, N. G. & Ganotakis, E. S. The hypolipidaemic effects of Spirulina (Arthrospira platensis) supplementation in a Cretan population: a prospective study. J. Sci. Food. Agric. 94, 432–437 (2014).
Ismail, M., Hossain, M. F., Tanu, A. R. & Shekhar, H. U. Effect of Spirulina Intervention on Oxidative Stress, Antioxidant Status, and Lipid Profile in Chronic Obstructive Pulmonary Disease Patients. Biomed. Res. Int. 2015, 1–7 (2015).
Brito, A. F. et al. Aortic Response to Strength Training and Spirulina platensis Dependent on Nitric Oxide and Antioxidants. Front. Physiol. 31, 1–14 (2018).
Sandhu, J. S., Dheera, B. & Shweta, S. Efficacy of Spirulina Supplementation on Isometric Strength and Iso- metric Endurance of Quadriceps in Trained and Untrained Individuals – a comparative study. Ibnosina. Journal of Medicine and Biomedical Sciences 2, 79–86 (2010).
Matuszczak, Y. et al. Effects of N-acetylcysteine on glutathione oxidation and fatigue during handgrip exercise. Muscle Nerve. 32, 633–638 (2005).
Micucci, M. et al. Cardiac and Vascular Synergic Protective Effect of Olea europea L. Leaves and Hibiscus sabdariffa L. Flower Extracts. Oxid. Med. Cell. Longev. 2015, 1–14 (2015).
Maughan, R. J. et al. IOC consensus statement: dietary supplements and the high-performance athlete. Br. J. Sports. Med. 52, 439–455 (2018).
Liu, R. H. Potential synergy of phytochemicals in cancer prevention: mechanism of action. J. Nutr. 134, 3479S–3485S (2004).
Draeger, C. L. et al. Controversies of antioxidant vitamins supplementation in exercise: ergogenic or ergolytic effects in humans? J. Int. Soc. Sports. Nutr. 11, 1–4 (2014).
Gliemann, L. et al. Resveratrol blunts the positive effects of exercise training on cardiovascular health in aged men. J. Physiol 591, 5047–59 (2015).
Paulsen, G. et al. Vitamin C and E supplementation hampers cellular adaptation to endurance training in humans: a double-blind, randomised, controlled trial. J. Physiol. 592, 1887–901 (2014b).
Franca, G. A. M. et al. Spirulina does not decrease muscle damage nor oxdidative stress in cycling athletes with adequate nutritional status. Biol. Sport. 27, 249–253 (2010).
Liping, L., Li-an, Q., Yiquan, W. & Guorong, Y. Spirulina platensis extract supplementation attenuates oxidative stress in acute exhaustive exercise: A pilot study. I. Int. J. Phys. Sci. 6, 2901–2906 (2011).
Juárez-Oropeza, M. A., Mascher, D., Torres-Durán, P. V., Farias, J. M. & Paredes-Carbajal, M. C. Effects of dietary Spirulina on vascular reactivity. J. Med. Food. 12, 15–20 (2009).
Sherwin, C. M. et al. Guidelines for the ethical use of animals in applied animal behaviour research. J. Appl. Toxicol. 81, 291–305 (2012).
Marqueti, R., Parizotto, N., Chriguer, R., Perez, S. & Selistre-de-Araujo, H. Androgenic-anabolic steroids associated with mechanical loading inhibit matrix metallopeptidase activity and affect the remodeling of the achilles tendon in rats. Am. J. Sports. Med. 34, 1274–1280 (2006).
Moura, L. P. et al. Spirulina, exercise and serum glucose control in diabetic rats. Arq. Bras. Endocrinol. Metabol. 56, 25–32 (2012).
Okafor, O. et al. Modulatory effect of pineapple peel extract on lipid peroxidation, catalase activity and hepatic biomarker levels in blood plasma of alcohol-induced oxidative stressed rats. Asian. Pac. J. Trop. Biomed. 1, 12–14 (2011).
Siekmann, L. et al. International Federation of Clinical Chemistry and Laboratory Medicine. Certification of four reference materials for the determination of enzymatic activity of gamma-glutamyltransferase, lactate dehydrogenase, alanine aminotransferase and creatine kinase accord. Clin. Chem. Lab. Med. 40, 739–745 (2002).
Ohkawa, H., Ohishi, N. & Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 95, 351–358 (1979).
Brand-Williams, W., Cuvelier, M. E. & Berset, C. Use of a free radical method to evaluate antioxidant activity. Lebenson Wiss Technol 28, 25–30 (1995).
Chu, Y. F., Sun, J., Wu, X. & Liu, R. H. Antioxidant and antiproliferative activities of common vegetables. J. Agric. Food. Chem. 50, 6910–6916 (2002).
Paulsen, G. et al. Can supplementation with vitamin C and E alter physiological adaptations to strength training? BMC Sports. Sci. Med. Rehabil. 6, 28 (2014a).
Gliemann, L. et al. Resveratrol blunts the positive effects of exercise training on cardiovascular health in aged men. J. Physiol 15, 5047–59 (2013).
Draeger, C. L. et al. Controversies of antioxidant vitamins supplementation in exercise: ergogenic or ergolytic effects in humans? J. Int. Soc. Sports. Nutr. 11, 4 (2014).
Myburgh, K. H. Polyphenol supplementation: benefits for exercise performance or oxidative stress? Sports. Med. 44, S57–70 (2014).
Paulsen, G. et al. Vitamin C and E supplementation hampers cellular adaptation to endurance training in humans: a double-blind, randomised, controlled trial. J. Physiol 592, 1887–901 (2014b).
Liping, L., Li-an, Q., Yiquan, W. & Guorong, Y. Spirulina platensis extract supplementation attenuates oxidative stress in acute exhaustive exercise: A pilot study. International Journal of Physical Sciences 6, 2901–2906 (2011).
Salim, C. G. et al. Dietary Docosahexaenoic Acid and Eicosapentaenoic Acid Influence Liver Triacylglycerol and Insulin Resistance in Rats Fed a High-Fructose. Diet. Mar. Drugs. 13, 1864–1881 (2015).
Yakubu, M. T. & Afolayan, A. J. Reproductive toxicologic evaluations of Bulbine natalensis Baker stem extract in albino rats. Theriogenology 72, 322–332 (2009).
Lu, H. K., Hsieh, C. C., Hsu, J. J., Yang, Y. K. & Chou, H. N. Preventive effects of Spirulina platensis on skeletal muscle damage under exercise-induced oxidative stress. Eur. J. Appl. Physiol. 98, 220–226 (2006).
Gholamnezhad, Z., Boskabady, M. H., Hosseini, M., Sankian, M. & Khajavi, R. A. Evaluation of immune response after moderate and overtraining exercise in wistar rat. Iran. J. Basic. Med. Sci. 17, 1–8 (2014).
Garza, C. A1-Acid glycoprotein, hepcidin, C-reactive protein, and serum ferritin are correlated in anemic schoolchildren with Schistosoma haematobium. Am. J. Clin. Nutr 91, 1784–1790 (2010).
McCarty, M. F. Clinical potential of Spirulina as a source of phycocyanobilin. J. Med. Food 10, 566–570 (2007).
Reddy, C. M. et al. Selective inhibition of cyclooxygenase-2 by C-phycocyanin, a biliprotein from Spirulina platensis. Biochem. Biophys. Res. Commun. 277, 599–603 (2000).
Ku, C. S. et al. Edible blue-green algae reduce the production of pro-inflammatory cytokines by inhibiting NF-KB pathway in macrophages and splenocytes. Biochim. Biophys. Acta. 1830, 2981–2988 (2013).
Acknowledgements
The authors thank the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Fundação de Apoio à Pesquisa do Estado da Paraíba (FAPESQ-PB) for their financial support and Federal University of Paraíba for logistical support.
Author information
Authors and Affiliations
Contributions
A.F.B., A.S.S. and B.A.S. developed the hypothesis and experimental design. A.F.B., I.L.L.S., C.V.O.C. and L.C.C.A. analyzed the data and wrote the manuscript. A.F.B., I.L.L.S. and P.B.F. performed the experimental work. R.S.S. contributed to the in vitro work. R.L.T., A.A.S., G.S.F., R.A.P., M.M.N. and L.C.C.A. contributed to the in vivo work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Brito, A.d.F., Silva, A.S., de Oliveira, C.V.C. et al. Spirulina platensis prevents oxidative stress and inflammation promoted by strength training in rats: dose-response relation study. Sci Rep 10, 6382 (2020). https://doi.org/10.1038/s41598-020-63272-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-63272-5
This article is cited by
-
Sulfonated magnetic spirulina nanobiomaterial as a novel and environmentally friendly catalyst for the synthesis of dihydroquinazolin-4(1H)-ones in aqueous medium
Scientific Reports (2024)
-
RETRACTED ARTICLE: Evaluating the neuroprotective effect of Spirulina platensis–loaded niosomes against Alzheimer’s disease induced in rats
Drug Delivery and Translational Research (2023)
-
Repro-protective activity of amygdalin and spirulina platensis in niosomes and conventional forms against aluminum chloride–induced testicular challenge in adult rats: role of CYP11A1, StAR, and HSD-3B expressions
Naunyn-Schmiedeberg's Archives of Pharmacology (2023)
-
The mitigating effect of Spirulina (Arthrospira platensis) on the hemotoxicity of gibberellic acid on juvenile tilapia (Oreochromis niloticus)
Environmental Science and Pollution Research (2022)
-
The effect of spirulina on type 2 diabetes: a systematic review and meta-analysis
Journal of Diabetes & Metabolic Disorders (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.