Abstract
Fungi play a key role in the functioning of soil in terrestrial ecosystems, and in particular in the remediation of degraded soils. The contribution of fungi to carbon and nutrient cycles, along with their capability to mobilise soil trace elements, is well-known. However, the importance of life history strategy for these functions has not yet been thoroughly studied. This study explored the soil-fungi relationship of two wild edible fungi, the ectomycorrhizal Laccaria laccata and the saprotroph Volvopluteus gloiocephalus. Fruiting bodies and surrounding soils in a mine-spill contaminated area were analysed. Isotope analyses revealed Laccaria laccata fruiting bodies were 15N-enriched when compared to Volvopluteus gloiocephalus, likely due to the transfer of 15N-depleted compounds to their host plant. Moreover, Laccaria laccata fruiting bodies δ13C values were closer to host plant values than surrounding soil, while Volvopluteus gloiocephalus matched the δ13C composition to that of the soil. Fungal species presented high bioaccumulation and concentrations of Cd and Cu in their fruiting bodies. Human consumption of these fruiting bodies may represent a toxicological risk due to their elevated Cd concentrations.
Similar content being viewed by others
Introduction
Fungi play a key role in the functioning of terrestrial ecosystems due to their diverse capabilities and relevance in nutrient cycling. Fungal functionality is directly related to their wide life history strategies, being classified as saprobes, parasites, pathogens and/or symbionts. Saprobes (or saprotrophs) are free-living fungi that obtain nutrients mostly from decomposing soil organic matter and, due to this ability, are considered as the most important decomposers in terrestrial ecosystems. Symbiotic mycorrhizal fungi also play a key role in nutrient cycling1,2. Mycorrhizal fungi are able to take up nutrients and water, and transfer them to the host plant, in exchange for photosynthates3,4. Ectomycorrhizal fungi may also mobilise soil trace elements enhancing their transfer from soil to plant and/or fungal tissues5. Saprotrophic fungi dominate the litter layer in soils and acquire C and nutrients in the form of organic substances by enzymatic decomposition6. Symbiotic ectomycorrhizal fungi are normally found in deeper soil layers (well-degraded litter and humus) with a high nutrient content but low C content, as they are not C-limited7,8. Ectomycorrhizal fungi are highly relevant in forests, including Mediterranean ones, as they associate with numerous tree and shrub species9. Previous studies found independent roles between ectomycorrhizal and saprotrophic fungi, with ectomycorrhizal fungi being able to degrade recalcitrant N-rich compounds, while saprotrophic fungi being able to degrade labile C-rich biopolymers10. Although ectomycorrhizal fungi have a lower ability to decompose organic matter than saprobes, they can obtain resources along the biotrophy–saprotrophy continuum11,12. For example, limited supply of plant photosynthate may increase enzyme production for obtaining labile carbohydrates from soil organic matter by ectomycorrizal fungi9,13. Ecological consequences may arise depending on the situation of the fungus within the continuum as, for example, ectomycorrhizal fungi fructification may fail if separated from plant roots11,14. Therefore, the spectrum of life history strategies implies that fungi differ in their roles and in their effects on ecosystem functioning.
Different mechanisms of resource acquisition, loss and cycling in fungi can be inferred by studying stable isotopes in fungal fruiting bodies (mushrooms)15,16,17. Previous studies found that ectomycorrhizal fungal sporocarps present higher δ15N values and lower δ13C values than saprotrophic ones1,17,18,19. Ectomycorrhizal fungi compete for N against saprotrophic fungi and bacteria, and they discriminate strongly against 15N20. Ectomycorrhizal fungi are known for their N isotope fractionation in the plant-soil system, as fungi retain 15N-enriched N while 15N-depleted N is transferred to the host plant. Therefore, analysing and comparing δ13C and δ15N values in fruiting bodies and underlying soils could help us to understand differences between saprotrophic and mycorrhizal strategies for resource acquisition1.
Fungi have been extensively studied in trace element contaminated areas as they are able to develop mechanisms to tolerate high trace element concentrations (see Bellion et al. 2006 for ectomycorrhizal fungi21). Fungi may reduce the entry of trace element concentrations into cells by a range of extracellular mechanisms, such as chelation by excreted ligands, cell-wall binding and enhanced efflux. Despite this, some amount of trace elements may still enter cells, but their toxicity can be reduced by other intracellular mechanisms, such as chelation by peptides and subcellular compartmentation22. In basidiomycetes, the mechanisms of response to trace element contamination are diverse and species dependent22,23. For example, the response of Agaricus to copper (Cu) is to produce metallothionein, while the response to cadmium (Cd) is to induce the production of mycophosphatin22.
Due to these protective mechanisms of fungi, many studies have focused on analysing the accumulation of trace elements into aboveground fungal biomass, as human consumption of edible mushrooms causes concern. In some trace element contaminated soils, a high accumulation of trace elements has been found in edible mushrooms growing there. In a silver-mining area of Czech Republic, high concentrations of trace elements were found in edible mushrooms: 149 mg kg−1 dry matter of Cd in Agaricus silvaticus, 12.9 mg kg−1 dry matter of mercury (Hg) in Lepista nuda and 16.2 mg kg−1 dry matter of lead (Pb) in Lycoperdon perlatum24. In another study, in a smelter area of Austria, the collected mushrooms had elevated concentrations of zinc (Zn) (up to 777 mg kg−1) and Cd (up to 127 mg kg−1)25.
Besides the numerous studies published, the relationships between trace element concentrations in soils and in fungal fruiting bodies are not always consistent and remain unclear. Some studies in the literature have found that fungal species may behave as bioindicators of trace elements, as concentrations in fungal fruiting bodies and their substrates correlated26,27,28. On the other hand, separate studies reported that elemental concentrations in fungi and soil did not correlate; they could have the ability to exclude trace elements or, at the contrary, to accumulate them into their biomass to levels well above (but not necessarily correlated to) the corresponding concentrations in soil25,29. This controversy is also affected by the soil extraction method selected to determine trace element concentration30. Most of the fungal studies used soil total fractions (including residual unavailable soil fractions) instead of readily or potentially available fractions31,32,33. However, the use of soil “available” fractions to compare with sporocarp concentrations is recommended25,34.
In order to improve our understanding of the soil-fungi relationship, the selection and analysis of the multiple factors that may explain the mushroom trace element accumulation patterns is required. First, in multi-element contaminated substrates, interactions among trace elements in soil and fungal biomass may further complicate our ability to foresee bioaccumulation patterns. In conditions of high concentrations of soil trace elements there may be a competitive interaction between some elements, for example between Cd and Zn. Some ectomycorrhizal species have a preferential uptake of Zn (essential element), through a competitive inhibition of Cd uptake (toxic element)35,36. Second, there are many environmental factors related to fungal accumulation of trace elements37. Soil variables, such as soil organic matter, pH and iron (Fe), manganese (Mn) and aluminium (Al) oxides and oxyhydroxides (among others) may explain fungal trace element accumulation, as they have a profound influence on trace element availability in the soil solution. Third, trophic differences among saprotrophs and ectomycorrhizal fungi can be related with variations in trace elements concentrations in both fruiting bodies and underlying soils, due to their different roles in soil organic matter decomposition and C and nutrient cycles10. Therefore, the concentration of trace elements in the soil is not the only determinant on their accumulation pattern in fungal fruiting bodies, as the soil-fungi relationship is very complex. The Bioconcentration Factor (BCF) (concentration in sporocarp divided by concentration in soil) is commonly calculated to elucidate this intricate translocation of trace elements from surrounding soil to fruiting bodies37,38. This BCF is therefore a very useful ratio which allows us to understand how different fungal species, fructifying in similar environmental conditions, may differ in trace element uptake and accumulation.
This study is of interest within the overall context of soil remediation processes and arises from the necessity of monitoring trace element concentrations in edible mushrooms growing in a contaminated site, as well as to elucidate the differential response of saprotrophic and ectomycorrhizal fungi to soil contamination by trace elements. We analysed the dynamics of nutrients and trace elements in two wild edible fungi species: Laccaria laccata (Scop.) Cooke, which was associated with stone pine and grey-leaved cistus by ectomycorrhizal symbiosis in the study area, and Volvopluteus gloiocephalus (DC.) Vizzini, Contu & Justo, saprotrophic fungi, fructifying in grassland soils. In a trace element contaminated and remediated ecosystem, we selected these fungal species as both were edible and fructified in the study area in the same period; in addition, they have contrasting life history strategy.
The specific objectives of this study were: (1) Determining fungal nutritional sources using C and N isotope composition; (2) Determining the accumulation capacity of trace elements in fungal fruiting bodies and the relations with soil properties; (3) Evaluating the potential toxicological risk of fruiting bodies (mushrooms) for human consumption.
Results
Soil properties
Soil properties in the study area were relatively homogenous for total C and N, available P, total Mg and some trace elements (Tables 1 and 2). However, there were significant differences between soils underneath fruiting bodies of ectomycorrhizal Laccaria laccata and saprotrophic Volvopluteus gloiocephalus fungi for other soil variables. Despite the fact that soil pH was acidic in the study area, Laccaria laccata underlying soils had lower pH (mean difference of 1.47 units) than Volvopluteus gloiocephalus soils (Table 1). Available K and total Ca were significantly different in the soils underneath both species, with higher concentrations in Volvopluteus gloiocephalus underlying soils (Table 1).
Soil pseudo-total arsenic (As), Cu, Fe, nickel (Ni), Pb and sulphur (S) were relatively homogeneous in the study area. However, pseudo-total Cd, cobalt (Co), Mn and Zn differed between fungal species habitats, being significantly higher the concentrations in Volvopluteus gloiocephalus underlying soils (Table 2). Soil “available” (CaCl2-extracted) concentrations of trace elements presented a different trend than pseudo-total values. Soil underneath Laccaria laccata sporocarps had significantly higher CaCl2-extracted concentrations for almost all trace elements (Cd, Co, Cu, Mn, Ni and Zn), except for Fe, Pb and S concentrations that were similar in both soils (Table 2).
Carbon and nitrogen isotopes in soil and fungal sporocarps
Soil C content was similar for both fungal species, however C isotope composition of these soils was different; Laccaria laccata surrounding soils were 13C-enriched compared to Volvopluteus gloiocephalus 13C-depleted surrounding soils (Table 1 and Fig. 1A). Ectomycorrhizal Laccaria laccata fungal sporocarps were 13C-depleted relative to saprotrophic Volvopluteus gloiocephalus fungi (Fig. 1A). Laccaria laccata fruiting bodies were 13C-depleted compared to their soil (t = 4.24; p < 0.001), whereas Volvopluteus gloiocephalus fruiting bodies were not significantly different to their soil (t = −1.31; p = 0.208) (Fig. 1A).
(A) Carbon and (B) Nitrogen isotope composition in soil and sporocarps of Laccaria laccata and Volvopluteus gloiocephalus fungal species. Bars represent mean values and error bars represent standard error for N = 10. P values resulting from Student’s t-test are indicated (ns: non-significant; p < 0.05*; p < 0.01**; p < 0.001***).
Soil N content was similar for both fungal species as well as their N isotope composition (Fig. 1B). In contrast to δ13C values, ectomycorrhizal Laccaria laccata sporocarps were 15N-enriched in comparison to saprotrophic Volvopluteus gloiocephalus sporocarps. Ectomycorrhizal Laccaria laccata sporocarps were 15N-enriched compared to the surrounding soil (t = −2.99; p = 0.009), whereas saprotrophic Volvopluteus gloiocephalus fungi were 15N-depleted (t = 6.51; p < 0.001) (Fig. 1B).
Chemical composition of fungal sporocarps
There were differences in nutrient concentrations between the fungal species. Volvopluteus gloiocephalus fruiting bodies presented significantly higher contents of N, P, K and Mg compared to Laccaria laccata sporocarps, whilst this tendency was the opposite for Ca, with higher concentrations in Laccaria laccata fruiting bodies. There was no significant difference between fungal species regarding C content (Fig. 2).
The two fungal species had different patterns of accumulation for most of the trace elements and S (Fig. 3), with higher concentrations of As, Co, Cu, Fe, Ni, Pb and Zn in Laccaria laccata fruiting bodies, and of Cd and S in Volvopluteus gloiocephalus fruiting bodies. No significant differences were found for Mn among fungal sporocarps of these species.
Trace elements and S in Laccaria laccata and Volvopluteus gloiocephalus sporocarps. Bars represent mean values and error bars represent standard error for N = 10. Student’s t-test statistic and p value are indicated (ns: non-significant; p < 0.05*; p < 0.01**; p < 0.001***). α Data transformation for normality of the residuals. β Non-parametric Mann-Whitney test (U statistic).
Bioconcentration factors of trace elements
Cadmium BCF was high in both fungal species (means of 5.09–8.49; Table 3). Mean BCF values of Co, Cu and Zn were close to one (and maximum values exceeded the unity) in the mycorrhizal Laccaria laccata, but not in the saprotrophic Volvopluteus gloiocephalus. The rest of the trace elements (As, Fe, Mn, Ni, Pb) and S had low BCF values for both species.
According to the NMDS ordination, the soil variables that best explained BCF variability were pH, total Ca and available K (Fig. 4). Volvopluteus gloiocephalus sporocarps fructified in soils with higher pH and higher Ca and K concentrations compared to Laccaria laccata sporocarps. Two defined clusters separated the fungal species with a higher BCF of Cd and S in Volvopluteus gloiocephalus, while Laccaria laccata sporocarps presented higher BCF for the rest of the elements (Fig. 4).
Nonmetric multidimensional scaling (NMDS) ordination of trace element bioconcentration factors (BCF) of Laccaria laccata (orange squares) and Volvopluteus gloiocephalus (white squares) fungal species. Black dots represent BCF for each trace element and S. Brown arrows represent soil variables with a significant correlation with BCF (p = 0.001 for pH and K, p = 0.003 for Ca).
The transfer ratio of trace elements from soil (CaCl2-extracted fraction) to fungal sporocarps was very high for Cd and Cu, moderate for Zn and Fe, low for Co, Mn and Ni, and negligible (<1) for S (Fig. S1). The transfer ratios from soil to fungus were higher in Volvopluteus gloiocephalus, in particular for Cd (x12), Zn (x6) and Mn (x5), with the exception of Fe which was higher (x2) in Laccaria laccata.
In general, Pearson´s correlation coefficients between trace element concentrations in fungal sporocarps and their underlying soils (CaCl2-extracted values) were low (Table S1). Laccaria laccata had a significant correlation (p < 0.05) with Co, and marginally significant correlations (p < 0.10) with As and Mn, and Volvopluteus gloiocephalus with Fe.
Toxicity risk
Calculations of the minimum daily intake of the studied fruiting bodies to reach maximum tolerable intake limits for toxicity in humans provided weights over 1 kg of fresh weight of both species for As, Cu, Fe, Pb and Zn. However, the consumption of 0.069 kg fresh weight of Volvopluteus gloiocephalus or 0.132 kg fresh weight of Laccaria laccata would be enough to reach the daily limit for Cd (Table S2). (All the calculations were based on a person of 70 kg bodyweight).
Discussion
Soil microorganisms play a key role in nutrient cycling, plant symbioses, organic matter decomposition, and other ecosystem processes39. Trace element contaminated soils are of concern due to their toxic effects on soil microbes40. In these soils, fungal communities provide many ecosystem services of regulation; in general, improving soil and water quality, nutrient cycling, soil fertility and carbon sequestration41. In particular, the “mycoremediation” potential to stabilise trace elements in fungal tissues can be considered as another ecosystem service. Thus, fungal mycelium secretes extracellular enzymes and acids to break down organic contaminants and has a high metal-binding capacity playing a promising role in remediation of trace elements42. However, from fungal mycelium, trace elements could be channelled to fruiting bodies implying a nutritional potential hazard due to mushroom consumption42. Fruiting bodies (mushrooms) of more than 1100 fungal species are used worldwide for food and medicines, thus delivering provision and cultural services43. With exception of poisonous species or edible fungi with high concentration of potentially toxic trace elements, which are a hazard for human health, and then represent a cultural “disservice”41.
The large-scale phytoremediation plan within the contaminated area considered in this study included planting of native trees and shrubs44. With time, planted trees modified the properties of underlying soil, for example, pine trees induced soil acidification and, consequently, higher concentrations of soluble fractions of trace elements44,45. Trees also influenced surrounding soil microbial activities, compared with adjacent treeless patches46. In this heterogeneous landscape mosaic the two studied fungal species fructified in contrasting habitats and soils: Laccaria laccata was associated with their host pine trees while saprotrophic Volvopluteus gloiocephalus was abundant in the grasslands without trees. Soils surrounding Volvopluteus gloiocephalus sporocarps presented higher pseudo-total concentrations of some elements (Cd, Co, Mn and Zn) than Laccaria laccata soils (under pine trees). These differences may be caused by the acidification process which increases losses of trace elements from soil due to enhanced leaching and take up by vegetation and fauna47. However, elements with low mobility, such as As and Pb, had similar concentrations between soil types. The opposite pattern was found for CaCl2-extracted trace element values; most of the trace elements were found in higher concentrations in soils surrounding Laccaria laccata sporocarps under pine trees probably due to the pine-induced soil acidification and subsequent trace element mobilisation, as already mentioned.
The differences in habitat and in life history strategy influence the mineral nutrition of both fungal species. Laccaria laccata sporocarps were found in soils close to their hosts Pinus pinea and Cistus albidus, with which they develop symbiotic relationships. The lower nutrient concentration of the mycorrhizal sporocarps, compared with those of Volvopluteus gloiocephalus, could be explained by the symbiotic association as pine and cistus may receive part of the nutrients taken up by this fungi, such as N and P10,19. In contrast, the saprotrophic Volvopluteus gloiocephalus, found in grassland areas dominated by herbaceous species, completely relies on soil sources for nutrition without loss by transfer to other organisms. The nutritional quality of both fungal sporocarps was within the usual content range for wild growing mushrooms, except for Mg that was lower in both species48.
Contrasting life history strategy of the studied fungal species influences C and N cycles. Despite similar soil C content, the observed differences in soil δ13C values may reflect a different isotope composition or different turnover rates, and the preferential use of 12C compared to 13C in biological processes49. Carbon isotope composition in Volvopluteus gloiocephalus fruiting bodies matched the ratios of the C soil source, according to its saprotrophic nature as litter decay fungi. In contrast, Laccaria laccata fruiting bodies were 13C-depleted (−28.14‰) compared to soil (−26.49‰), reflecting the isotope composition of its host photosynthate, which could be the main C source1,50. The differences in N isotope composition between both fungal species were even more conspicuous, where ectomycorrhizal Laccaria laccata fruiting bodies were 15N-enriched in comparison to the surrounding soil; potentially because of the transfer of 15N-depleted compounds to the symbiotic plants1,18,51,52,53,54,55. Whereas, saprotrophic Volvopluteus gloiocephalus fruiting bodies had much lower 15N than Laccaria laccata, and were 15N-depleted in comparison to the soil, despite having similar N isotope composition in both soil types.
A previous study in Sweden found fungi were able to take up and accumulate trace elements such as Cd, Cu and Zn in both sporocarps and mycelium (BCF from 1.9 to 8.8) in respect of bulk soil concentrations56. In this study, we found that the fungal uptake of trace elements is dependent on both the chemical element and the fungal species5. Low selectivity of fungal transporters for essential elements, such as Ca and Zn, favoured the transport of toxic ions with similar chemical properties, such as Cd22. In the study area, both Volvopluteus gloiocephalus and Laccaria laccata sporocarps mainly bioconcentrated Cd. Previous studies in the same area had found that roots of seven tree species also concentrated Cd (BCF > 1), making them suitable for the phytostabilisation of this potentially toxic element44. Although Cd was accumulated in fruiting bodies of both fungal species, the patterns and relations with soil differed; Volvopluteus gloiocephalus sporocarps accumulated much higher levels of Cd than Laccaria laccata. However, accumulation of Ca and Zn were opposite, with higher concentrations in Laccaria laccata sporocarps. The correlation between Cd levels in fruiting bodies and soils was positive for the saprotrophic species but negative for the ectomycorrhizal one; a possible explanation is that Cd-Zn-Ca competition influence elements accumulation in the studied sporocarps57. A higher Ca and Zn content was registered in Laccaria laccata sporocarps, besides Volvopluteus gloiocephalus surrounding soils recorded a higher Ca and Zn concentrations. Therefore, a competition among Cd-Zn-Ca could be responsible for the preference of translocation of Ca and Zn over Cd in Laccaria laccata fruiting bodies. On the other hand, the presence of Cd could induce the production of intracellular binding compounds such as metallothioneins, cadystin, and phytochelatins, and cellular compartmentalization in this species22,58,59. Besides the antagonism or interaction between chemical elements, the bioconcentration of trace elements in fungal fruiting bodies is influenced by soil properties60. In this study, soil pH, Ca and K were relevant variables explaining the BCF of trace elements in fruiting bodies, and these variables were positively correlated among them (p < 0.001). In acidic soils, the release of most trace elements increases, being more available in the soil solution and, therefore, more accessible to microorganisms61,62. In comparison with other studies, As concentrations in Laccaria laccata sporocarps are broad, depending on the location and soil conditions, with a range from 0.66 to 147 mg kg−1 in caps and stipes63.
The CaCl2-extracted fraction for most of the trace elements in the soil usually represents a small proportion of their total concentration (Table 2). Therefore, the transfer ratios of trace elements from soil (CaCl2-extracted pool) to fungal sporocarps are functionally more realistic than BCF values (calculated using pseudo-total soil concentrations) and worthwhile to consider. Thus, besides the important Cd accumulation already discussed above, we have detected a high transfer of Cu from soil to fungus (over 200 times). Other trace elements with remarkable soil to fungus transfer were Zn (78 times more in Volvopluteus gloiocephalus) and Fe (48 times more in Laccaria laccata). If we assume that sporocarps are reflecting mycelium chemical composition, we can infer that both fungal species are contributing potentially to the “mycostabilisation” of Cd, Cu, Zn and Fe in these contaminated and remediated soils37,42.
The well-known capacity of mushrooms to accumulate trace elements could make them useful organisms to bioindicate soil contamination64. However, in this study, trace element concentrations in fruiting bodies were not significantly correlated to soil CaCl2-extracted concentrations, with the exception of Co in Laccaria laccata. Many studies have also found that fruiting bodies are not correlated with soil contamination by trace elements; therefore concluding that they are not good bioindicators of soil trace element contamination32,65,66,67. On the contrary, there are authors supporting the use of fungal sporocarps as bioindicators26,27,28. In our study site, the concentration of trace elements in leaves of poplar trees and in soil were correlated for Cd, Cu, Mn and Zn68. The potential utility as bioindicator would then depend on the fungal species, the particular trace element, and the environmental conditions.
From the food quality perspective, the usually high accumulation of potentially toxic elements in fungal fruiting bodies is a relevant ecosystem disservice. Trace elements concentrations in fruiting bodies were similar to those measured in edible mushrooms from unpolluted areas67, except for the increased mean concentrations of Cd in Volvopluteus gloiocephalus (and maximum in Laccaria laccata) and mean Cu and maximum Fe, Mn and Zn concentrations in Laccaria laccata sporocarps (Table S3).
Food regulation to avoid toxicity risk includes concentration limits for several trace elements in wild edible mushrooms. Thus, the European Union regulation established a concentration limit of 0.3 mg Pb kg−1 fresh weight and 0.2 mg Cd kg−1 fresh weight for cultivated fungal species (Agaricus bisporus, Pleurotus ostreatus and Lentinula edodes). For other fungal species, this limit was established at 1.00 mg Cd kg−1 fresh weight and no regulation limit was established in other species for Pb69,70. The maximum Cd concentration registered in Volvopluteus gloiocephalus sporocarps was 0.84 mg Cd kg−1 fresh weight, which is well above the European Union regulation limit for cultivated mushrooms, although still below the limit for wild species. The maximum Pb concentration in our sporocarps was 0.25 mg Pb kg−1 fresh weight, which is also below the limit (for cultivated species only). Therefore, according to European Union regulation, neither Cd nor Pb concentrations reached these maximum levels (Table S3; dry weight).
The World Health Organization71 evaluated certain trace element contaminants in food to estimate the tolerable intake limits (Table S2; PMTDI). We calculated the minimum weight to reach tolerable limits, and Cd presented the lowest weight (Table S2). A daily intake of 0.069 kg fresh weight of Volvopluteus gloiocephalus or 0.132 kg fresh weight of Laccaria laccata (for a person with 70 kg bodyweight) would reach the maximal tolerable Cd daily limit, without taking into account other Cd daily sources. The consumption of these fungi could represent a toxicological risk due to elevated Cd content, especially Volvopluteus gloiocephalus species, as this element is of high toxicological importance for humans67,72. Laccaria laccata consumption would entail a higher toxicological risk than Volvopluteus gloiocephalus for the rest of trace elements (As, Cu, Fe, Pb and Zn), however the daily intake to reach toxicity (over 1 kg fresh weight) represents an unrealistically high level of consumption. In any case, for a complete evaluation of actual potential toxicity to humans the effect of mushroom processing during cooking on its metal concentrations should also be taken into account.
Conclusions
After a long-term restoration process to mitigate the effects of a mine-spill in SW Spain, Laccaria laccata and Volvopluteus gloiocephalus sporocarps and surrounding soil analyses demonstrated trace element persistence in the study area. Some soil trace element concentrations were above background values and soil acidification seemed to be responsible for the higher trace element concentrations in Laccaria laccata fruiting bodies. Analyses of δ15N and δ13C in fruiting bodies and surrounding soils allowed us to differentiate the saprotrophic Volvopluteus gloiocephalus fungi from the ectomycorrhizal Laccaria laccata fungi, proving this technique as adequate to differentiate fungal life history and to contribute to a better knowledge of the soil-fungi relations.
Bioconcentration factors demonstrated that both fungal species accumulated Cd, and that Cu was transferred from the soil “available” (CaCl2-extracted) pool to fungal sporocarps. Human consumption of Laccaria laccata and/ or Volvopluteus gloiocephalus sporocarps collected in the area may represent a toxicological risk due to the elevated concentrations of Cd. Therefore, strict control is required in the area to avoid human consumption of these fruiting bodies. Due to the ability of these species to accumulate some metals, monitoring on trace element concentrations in these fungi is thus highly recommended, even if mobility of metals in soils is assumed to be low.
Materials and methods
Study area
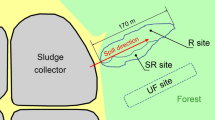
The study was conducted in the Guadiamar Green Corridor (Seville, SW Spain; 37°23.165′N, 6°13.668′W) which is a public area managed by the Spanish Regional Government. The study area is located 12 km downstream from the Aznalcóllar tailings dam failure, which affected about 4000 ha and a 60 km stretch along the Guadiamar River73,74. Aznalcóllar mine is located in the Iberian Pyrite Belt formed by a large concentration of polymetallic massive sulphides with high grade on silver (Ag), Pb and Zn74. The mine-spill covered these soils with 6 hm3 of acidic waters and sludge loaded with a mixture of trace elements, mainly As, Cd, Cu, Pb, thallium (Tl) and Zn75. Since the accident (in 1998) until 2001, a remediation plan was put in place which consisted in sludge and top soil removal by heavy machinery and application of amendments (sugar lime, clay and organic matter) to immobilise trace elements. Subsequently, afforestation of the area with native trees and shrubs was carried out44. Differences in soil type and remediation outcome created a heterogeneous Corridor, and 15 years after the remediation of the area some patches still presented high soil acidification and trace element content76. The study area is one of these degraded areas with high toxicity risks due to the naturally sandy and acidic soil, with a limited capacity of soils to buffer against acid drainage caused by the remnants of mining residues.
Study fungal species
The two selected species in this study were Laccaria laccata, which form ectomycorrhizae, and Volvopluteus gloiocephalus, which is a saprobe (Fig. 5). Both species belong to the Basidiomycota division and Agaricales order, but are classified in different families. Laccaria laccata belongs to the Hydnangiaceae family and Volvopluteus gloiocephalus to the Pluteaceae family. Laccaria laccata is distributed broadly in the Northern Hemisphere and has small, red-orange fruiting bodies. This fungal species has a wide ectomycorrhizal host potential, a low specificity and it is characteristic of young stands of trees77. This species has been identified as component of the fungal ectomycorrhizal communities associated with Quercus ilex in the study area5,78. Volvopluteus gloiocephalus is distributed in Europe and North America and has bigger fruiting bodies than Laccaria laccata with white or grey-brown colours79.
The specimens of fruiting bodies collected for this work were registered in the University of Seville Herbarium to the codes SEV-C 51 for Laccaria laccata and SEV-C 50 for Volvopluteus gloiocephalus. Molecular analyses were carried out to confirm the species morphological determination and to identify potential ecotypes. Sequences are available in GenBank (https://www.ncbi.nlm.nih.gov/genbank/) under accession numbers MK616245 - MK616246 for Laccaria laccata and MK616344 - MK616347 for Volvopluteus gloiocephalus.
Sampling design
In December 2016, a field sampling was conducted in order to evaluate the wild growing edible fungal fruiting bodies that fructified in the remediated soils of this area. Fruiting bodies were sampled in the locations where they fructified, naturally occurring in separated habitats. Laccaria laccata sporocarps were found close to the ectomycorrhizal tree, Pinus pinea, and the shrub, Cistus albidus. In contrast, Volvopluteus gloiocephalus sporocarps were found in adjacent treeless areas covered by different herbaceous species. Therefore, sampling locations were dependent on the distribution of these habitats in the study area (Fig. S2). For each species, ten points and at least three sporocarps per point were sampled. Soil surrounding each sporocarp was taken (top 0–5 cm depth) with an auger from at least five locations within each point to make a composite sample per point. Sporocarps and soils were kept refrigerated until being processed the day after sampling.
Soil analyses
Soil samples (top 0–5 cm depth; 100 g fresh weight) were air-dried and sieved to <2 mm for chemical analysis, where soil pH was measured in a 1:2.5 soil-water suspension after 30 min of shaking. Bray 1 method was selected to determine soil P in these acidic soils80. Available K was extracted with 1 M ammonium acetate and determined by multiparametric Bran-Luebbe autoanalyser. Pseudo-total concentrations of trace elements (As, Cd, Co, Cu, Fe, Mn, Ni, Pb and Zn), S, Ca and Mg were extracted by digestion of soils, ground to <60 µm, with aqua regia (1:3 v/v conc. HNO3/ HCl) in a Digiprep MS block digester (SPS Science) for 2 h at 110 °C. Trace elements (As, Cd, Co, Cu, Fe, Mn, Ni, Pb and Zn) and S were extracted from <2 mm sieved soils with a 0.01 M CaCl2 solution to estimate their “bioavailability”81. Extracts after digestion with aqua regia or CaCl2, were determined by inductively coupled plasma optical emission spectrophotometry (ICP-OES) with Varian ICP 720-ES. The quality of the analyses was assessed using the reference sample ERM-CC141 (loam soil) and recoveries from 93.8% to 100.4% were obtained. Arsenic extracted by CaCl2 presented concentrations below detections limits (0.005 mg kg-1), and its “availability” is underestimated by this method.
Chemical composition of fungal fruiting bodies
Fruiting bodies were dry cleaned with a brush and fresh weighed, with some biomass kept at −80 °C for further species identification by molecular analysis. Sporocarps were dried at 70 °C for 48 h and stored until analysis. The whole fruiting body (cap, hymenophore and stalk) was analysed as one single sample, as all the parts are usually ingested when consumed by humans. Fruiting bodies macronutrients (Ca, K, Mg, S and P) and trace elements were extracted by digestion with HNO3 in a Digiprep MS block digester (SPS Science) for 2 h at 110 °C and determined by ICP-OES with Varian ICP 720-ES. Arsenic was determined by ICP-OES with Ion Exchange Hydride Generation (IE-HG-ICP-OES) methodology82. The quality of the analysis was assessed using the reference sample ID 124 Type Lucerne (Medicago sativum) (WEPAL; IPE) for As, with recoveries of 95.9%, and the reference sample INCT-OBTL-5 (tobacco leaves) for the rest of the trace elements, with recoveries from 79.9% to 112.2%.
Isotopic analysis
Total C and N and their isotope composition (δ13C and δ15N, respectively) in soil and sporocarps were determined by elemental analysis and isotope ratio mass spectrometry (EA/IRMS) system by means of Flash HT Plus elemental analyser coupled to a Delta-V Advantage isotope ratio mass spectrometer via a CONFLO IV interface (Thermo Fisher Scientific, Bremen, DE). The analytical measurement errors were ±0.1‰ for δ13C and ±0.2‰ for δ15N. Total C was organic C due to the absence of carbonates in the study area76.
Data analysis
Mean and standard error (SE) of datasets from Laccaria laccata and Volvopluteus gloiocephalus sporocarps and respective soils were calculated. The chemical composition of soils and sporocarps of the studied species were compared through a parametric Student’s t-tests. Normality assumptions were verified with Lilliefors-corrected Kolmogorov-Smirnov test for normality and Levene test for homogeneity of variance. If the assumptions were not met, the data was Box-Cox transformed. If this transformation did not meet the assumptions, a non-parametric Mann-Whitney test was subsequently performed.
To evaluate the level of contamination, the values of soil pseudo-total concentration of trace elements were compared with background values (BV) for the South Portuguese Zone83. The reference level (percentile 90), which is the maximum value accepted for non-contaminated soils, was used.
Bioconcentration factors of trace elements were calculated as concentration in fruiting bodies divided by pseudo-total concentration in soil underneath (both in mg kg−1)37,38. To interpret the results, a BCF > 1 suggests that fungi have some transporters (active or passive) that can carry trace elements into fungal biomass resulting in accumulation, while a BCF < 1 may indicate the existence of fungal mechanisms against take up of trace elements30. In addition, the ratio between trace elements accumulated in the fungal fruiting bodies and those “bioavailable” in the soil (estimated by CaCl2 extraction)was calculated as a functional transfer ratio34.
To explore relationships among sporocarps BCF and soil variables, a nonmetric multidimensional scaling (NMDS) was performed84. The BCF data was standardised with a Hellinger transformation and selected Euclidean distance metrics, where the number of dimensions was k = 2 with an adequate level of stress of 0.010. Soil variables effects were calculated using a permutation model (with 999 permutations) and fitted using the envfit function85. In this plot, the length of arrows represents the strength of the correlation and its direction indicates the trend of change in the variable.
To evaluate relationships of trace elements in the soil (CaCl2-extracted fraction) with sporocarps we performed a Pearson´s correlation test, adjusted with Benjamini-Hochberg correction86.
All data analyses were performed utilising the R software package v3.5.187 using ggplot288, lawstat89, MASS90, nortest91, psych92 and vegan85 packages.
Evaluation of toxicity risk
Fruiting bodies concentrations of potentially toxic elements (Cd and Pb) in the two fungal species were compared with the regulation limits (in fresh weight) for edible mushrooms of the European Union69,70. The fruiting bodies concentrations of Cd and Pb were transformed from dry weight to fresh weight by dividing the limits (Cd limited to 1 mg kg−1 fresh weight; Pb limited to 0.3 mg kg−1 fresh weight) by the mean fresh weight (Laccaria laccata 7.13 g fresh weight; Volvopluteus gloiocephalus 5.77 g fresh weight) (Table S3).
Fungi minimum daily intake to reach the upper level of the tolerable intake threshold, established by the Joint FAO/WHO Expert Committee on Food Additives93, were calculated to those trace elements reported in that guideline: As, Cd, Cu, Fe, Pb and Zn. The guideline limits were transformed into provisional maximum tolerable daily intake (PMTDI) and the weights were expressed as kg of fresh weight for a person with 70 kg bodyweight (as proposed by the European Food Safety Authority) (Table S2).
References
Hobbie, E. A., Macko, S. A. & Shugart, H. H. Insights into nitrogen and carbon dynamics of ectomycorrhizal and saprotrophic fungi from isotopic evidence. Oecologia 118, 353 (1999).
Phillips, R. P., Brzostek, E. & Midgley, M. G. The mycorrhizal-associated nutrient economy: a new framework for predicting carbon-nutrient couplings in temperate forests. New Phytol. 199, 41–51 (2013).
Lindahl, B. D. & Tunlid, A. Ectomycorrhizal fungi - potential organic matter decomposers, yet not saprotrophs. New Phytol. 205, 1443–1447 (2015).
Smith, S. & Read, D. Mycorrhizal Symbiosis, https://doi.org/10.1016/B978-0-12-370526-6.X5001-6 (Academic Press, 2008).
Gil-Martínez, M. et al. Ectomycorrhizal fungal communities and their functional traits mediate plant–soil interactions in trace element contaminated soils. Front. Plant Sci. 9, 1682 (2018).
Sinsabaugh, R. L. et al. Stoichiometry of soil enzyme activity at global scale. Ecol. Lett. 11, 1252–1264 (2008).
Lindahl, B. D. et al. Spatial separation of litter decomposition and mycorrhizal nitrogen uptake in a boreal forest. New Phytol. 173, 611–620 (2007).
McGuire, K. L., Allison, S. D., Fierer, N. & Treseder, K. K. Ectomycorrhizal-dominated boreal and tropical forests have distinct fungal communities, but analogous spatial patterns across soil horizons. PLoS One 8, 1–9 (2013).
Courty, P. E. et al. The role of ectomycorrhizal communities in forest ecosystem processes: new perspectives and emerging concepts. Soil Biol. Biochem. 42, 679–698 (2010).
Talbot, J. M. et al. Independent roles of ectomycorrhizal and saprotrophic communities in soil organic matter decomposition. Soil Biol. Biochem. 57, 282–291 (2013).
Koide, R. T., Sharda, J. N., Herr, J. R. & Malcolm, G. M. Ectomycorrhizal fungi and the biotrophy–saprotrophy continuum. New Phytol. 178, 230–233 (2008).
Read, D. J., Leake, J. R. & Perez-Moreno, J. Mycorrhizal fungi as drivers of ecosystem processes in heathland and boreal forest biomes. Can. J. Bot. 83, 1243–1263 (2004).
Courty, P. E., Bréda, N. & Garbaye, J. Relation between oak tree phenology and the secretion of organic matter degrading enzymes by Lactarius quietus ectomycorrhizas before and during bud break. Soil Biol. Biochem. 39, 1655–1663 (2007).
Ishida, T. A., Nara, K., Tanaka, M., Kinoshita, A. & Hogetsu, T. Germination and infectivity of ectomycorrhizal fungal spores in relation to their ecological traits during primary succession. New Phytol. 180, 491–500 (2008).
Agerer, R., Christan, J., Mayr, C. & Hobbie, E. Isotopic signatures and trophic status of Ramaria. Mycol. Prog. 11, 47–59 (2012).
Hobbie, E. A., Sánchez, F. S. & Rygiewicz, P. T. Carbon use, nitrogen use, and isotopic fractionation of ectomycorrhizal and saprotrophic fungi in natural abundance and 13C-labelled cultures. Mycol. Res. 108, 725–736 (2004).
Hobbie, E. A. & Horton, T. R. Evidence that saprotrophic fungi mobilise carbon and mycorrhizal fungi mobilise nitrogen during litter decomposition. New Phytol. 173, 447–449 (2007).
Hobbie, E. A., Weber, N. S. & Trappe, J. M. Mycorrhizal vs saprotrophic status of fungi: the isotopic evidence. New Phytol. 150, 601–610 (2001).
Mayor, J. R., Schuur, E. A. G. & Henkel, T. W. Elucidating the nutritional dynamics of fungi using stable isotopes. Ecol. Lett. 12, 171–183 (2009).
Hobbie, E. & Högberg, P. Nitrogen isotopes link mycorrhizal fungi and plants to nitrogen dynamics. New Phytol. 196, 367–382 (2012).
Bellion, M., Courbot, M., Jacob, C., Blaudez, D. & Chalot, M. Extracellular and cellular mechanisms sustaining metal tolerance in ectomycorrhizal fungi. FEMS Microbiol. Lett. 254, 173–181 (2006).
Baldrian, P. Effect of heavy metals on saprotrophic soil fungi. in Soil Heavy Metals (eds. I., Sherameti & A., Varma) 263–279 (Springer-Verlag, 2010).
Godbold, D. L., Jentschke, G., Winter, S. & Marschner, P. Ectomycorrhizas and amelioration of metal stress in forest trees. Chemosphere 36, 757–762 (1998).
Svoboda, L., Havlíčková, B. & Kalač, P. Contents of cadmium, mercury and lead in edible mushrooms growing in a historical silver-mining area. Food Chem. 96, 580–585 (2006).
Krpata, D., Fitz, W., Peintner, U., Langer, I. & Schweiger, P. Bioconcentration of zinc and cadmium in ectomycorrhizal fungi and associated aspen trees as affected by level of pollution. Environ. Pollut. 157, 280–286 (2009).
Malinowska, E., Szefer, P. & Falandysz, J. Metals bioaccumulation by bay bolete, Xerocomus badius, from selected sites in Poland. Food Chem. 84, 405–416 (2004).
Alonso, J., García, M. A., Pérez-López, M. & Melgar, M. J. The concentrations and bioconcentration factors of copper and zinc in edible mushrooms. Arch. Environ. Contam. Toxicol. 44, 180–188 (2003).
Proskura, N., Skopicz-Radkiewicz, L. & Podlasińska, J. Chemical composition and bioaccumulation ability of Boletus badius (Fr.) Fr. collected in western Poland. Chemosphere 168, 106–111 (2017).
Colpaert, J. V., Wevers, J. H. L., Krznaric, E. & Adriaensen, K. How metal-tolerant ecotypes of ectomycorrhizal fungi protect plants from heavy metal pollution. Ann. For. Sci. 68, 17–24 (2011).
Buscaroli, A. An overview of indexes to evaluate terrestrial plants for phytoremediation purposes (Review). Ecol. Indic. 82, 367–380 (2017).
Mleczek, M. et al. Accumulation of elements by edible mushroom species: Part I. Problem of trace element toxicity in mushrooms. J. Environ. Sci. Heal. - Part B Pestic. Food Contam. Agric. Wastes 48, 69–81 (2013).
Melgar, M. J., Alonso, J. & García, M. A. Cadmium in edible mushrooms from NW Spain: Bioconcentration factors and consumer health implications. Food Chem. Toxicol. 88, 13–20 (2016).
Falandysz, J. et al. Mercury in forest mushrooms and topsoil from the Yunnan highlands and the subalpine region of the Minya Konka summit in the Eastern Tibetan Plateau. Environ. Sci. Pollut. Res. 23730–23741 (2016) https://doi.org/10.1007/s11356-016-7580-6.
Komárek, M., Chrastný, V. & Štíchová, J. Metal/metalloid contamination and isotopic composition of lead in edible mushrooms and forest soils originating from a smelting area. Environ. Int. 33, 677–684 (2007).
Hartley, J., Cairney, J. W. G., Sanders, F. E. & Meharg, A. A. Toxic interactions of metal ions (Cd2+, Pb2+, Zn2+ and Sb3-) on in vitro biomass production of ectomycorrhizal fungi. New Phytol. 137, 551–562 (1997).
Colpaert, J. V. & Van Assche, J. A. The effects of cadmium and the cadmium-zinc interaction on the axenic growth of ectomycorrhizal fungi. Plant Soil 145, 237–243 (1992).
Ali, A. et al. Mycoremediation of potentially toxic trace elements—a biological tool for soil cleanup: a review. Pedosphere 27, 205–222 (2017).
Tyler, G. Metal accumulation by wood-decaying fungi. Chemosphere 11, 1141–1146 (1982).
Nannipieri, P. et al. Microbial diversity and soil functions. Eur. J. Soil Sci. 68, 12–26 (2017).
Giller, K. E., Witter, E. & Mcgrath, S. P. Toxicity of heavy metals to microorganisms and microbial processes in agricultural soils: A review. Soil Biol. Biochem. 30, 1389–1414 (1998).
Bakker, M. R. et al. Belowground biodiversity relates positively to ecosystem services of European forests. Front. For. Glob. Chang. 2, 1–23 (2019).
Purohit, J., Chattopadhyay, A., Biswas, M. K. & Singh, N. K. Mycoremediation of agricultural soil: bioprospection for sustainable development. In Mycoremediation and Environmental Sustainability (eds. R., Prasad) vol. 2 39–60 (Springer International Publishing AG, 2018).
Boa, E. Wild Edible Fungi: A global overview of their use and importance to people. Non-wood forest products (Food and Agriculture Organization of the United Nations, 2004).
Madejón, P. et al. Evaluation of amendment addition and tree planting as measures to remediate contaminated soils: the Guadiamar case study (SW Spain). Catena 166, 34–43 (2018).
Marañón, T., Navarro-Fernández, C. M., Domínguez, M. T., Madejón, P. & Murillo, J. M. How the soil chemical composition is affected by seven tree species planted at a contaminated and remediated site. Web Ecol. 15, 45–48 (2015).
Gil-Martínez, M. et al. Long-term effects of trace element contamination and tree species on soil microbial biomass and enzyme activities. Proc. 12th Int. Conf. Mine Clos. 633–644 (2018).
Bolan, N. et al. Remediation of heavy metal(loid)s contaminated soils - to mobilize or to immobilize? J. Hazard. Mater. 266, 141–166 (2014).
Kalač, P. Chemical composition and nutritional value of European species of wild growing mushrooms: a review. Food Chem. 113, 9–16 (2009).
Dawson, T. E., Mambelli, S., Plamboeck, A. H., Templer, P. H. & Tu, K. P. Stable isotopes in plant ecology. Annu. Rev. Ecol. Syst. 33, 507–559 (2002).
Rosling, A., Lindahl, B. D. & Finlay, R. D. Carbon allocation to ectomycorrhizal roots and mycelium colonising different mineral substrates. New Phytol. 162, 795–802 (2004).
Hou, W., Lian, B., Dong, H., Jiang, H. & Wu, X. Distinguishing ectomycorrhizal and saprophytic fungi using carbon and nitrogen isotopic compositions. Geosci. Front. 3, 351–356 (2012).
Kohzu, A. et al. Natural 13C and 15N abundance of field-collected fungi and their ecological implications. New Phytol. 144, 323–330 (1999).
Gebauer, G. & Taylor, A. F. S. 15N natural abundance in fruit bodies of different functional groups of fungi in relation to substrate utilization. New Phytol. 143, 93–101 (1999).
Hobbie, E. A., Macko, S. A. & Shugart, H. H. Interpretation of nitrogen isotope signatures using the NIFTE model. Oecologia 120, 405–415 (1999).
Kohzu, A., Tateishi, T., Yamada, A., Koba, K. & Wada, E. Nitrogen isotope fractionation during nitrogen transport from ectomycorrhizal fungi, Suillus granulatus, to the host plant, Pinus densiflora. Soil Sci. Plant Nutr. 46, 733–739 (2000).
Vinichuk, M. M. Copper, zinc, and cadmium in various fractions of soil and fungi in a Swedish forest. J. Environ. Sci. Heal. - Part A Toxic/Hazardous Subst. Environ. Eng. 48, 980–987 (2013).
Hartley, J., Cairney, J. W. G. & Meharg, A. A. Do ectomycorrhizal fungi exhibit adaptive tolerance to potentially toxic metals in the environment? Plant Soil 189, 303–319 (1997).
Blaudez, D., Botton, B. & Chalot, M. Cadmium uptake and subcellular compartmentation in the ectomycorrhizal fungus Paxillus involutus. Microbiology 146, 1109–1117 (2000).
Guerinot, M. & Lou. The ZIP family of metal transporters. Biochim. Biophys. Acta 1465, 190–198 (2000).
Elekes, C. C., Busuioc, G. & Ionita, G. The bioaccumulation of some heavy metals in the fruiting body of wild growing mushrooms. Not. Bot. Horti Agrobot. Cluj-Napoca 38, 147–151 (2010).
Kabata-Pendias, A. & Pendias, H. Trace elements in soils and plants. (CRC Press LLC, 2001).
Ledin, M., Krantz-Rülcker, C. & Allard, B. Microorganisms as metal sorbents: Comparison with other soil constituents in multi-compartment systems. Soil Biol. Biochem. 31, 1639–1648 (1999).
Zhang, J., Li, T., Yang, Y. L., Liu, H. G. & Wang, Y. Z. Arsenic concentrations and associated health risks in Laccaria mushrooms from Yunnan (SW China). Biol. Trace Elem. Res. 164, 261–266 (2015).
Markert, B. A., Breure, A. M. & Zechmeister, H. G. Trace metals and other contaminants in the environment. Volume 6 - Bioindicators and biomonitors. (Pergamon, 2003).
Petrini, O., Cocchi, L., Vescovi, L. & Petrini, L. Chemical elements in mushrooms: their potential taxonomic significance. Mycol. Prog. 8, 171–180 (2009).
Cocchi, L., Vescovi, L., Petrini, L. E. & Petrini, O. Heavy metals in edible mushrooms in Italy. Food Chem. 98, 277–284 (2006).
Kalač, P. Trace element contents in European species of wild growing edible mushrooms: A review for the period 2000–2009. Food Chem. 122, 2–15 (2010).
Madejón, P., Marañón, T., Murillo, J. M. & Robinson, B. White poplar (Populus alba) as a biomonitor of trace elements in contaminated riparian forests. Environ. Pollut. 132, 145–155 (2004).
European Commission. Commission Regulation (EU) 2015/1005 of 25 June 2015 amending Regulation (EC) No 1881/2006 as regards maximum levels of lead in certain foodstuffs. Official Journal of the European Union vol. 161 (2015).
European Commission. Commission Regulation (EU) No 488/2014 of 12 May 2014 amending Regulation (EC) No 1881/2006 as regards maximum levels of cadmium in foodstuffs. Official Journal of the European Union vol. 138 (2014).
WHO. Summary Report of the 72nd Meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), http://www.fao.org/ag/agn/agns/jecfa_index_en.asp%0Ahttp://www.fao.org/ag/agn/agns/jecfa_index_en.asp (2010).
Kalač, P. & Svoboda, L. A review of trace element concentrations in edible mushrooms. Food Chem. 69, 273–281 (2000).
Domínguez, M. T., Marañón, T., Murillo, J. M., Schulin, R. & Robinson, B. H. Trace element accumulation in woody plants of the Guadiamar Valley, SW Spain: A large-scale phytomanagement case study. Environ. Pollut. 152, 50–59 (2008).
Grimalt, J. O., Ferrer, M. & MacPherson, E. The mine tailing accident in Aznalcollar. Sci. Total Environ. 242, 3–11 (1999).
Cabrera, F., Clemente, L., Díaz Barrientos, E., López, R. & Murillo, J. M. Heavy metal pollution of soils affected by the Guadiamar toxic flood. Sci. Total Environ. 242, 117–129 (1999).
Domínguez, M. T. et al. River banks and channels as hotspots of soil pollution after large-scale remediation of a river basin. Geoderma 261, 133–140 (2016).
Dix, N. J. & Webster, J. Terrestrial Macrofungi. in Fungal Ecology 341–397, https://doi.org/10.1007/978-94-011-0693-1_13 (Springer Netherlands, 1995).
López-García, Á. et al. Functional diversity of ectomycorrhizal fungal communities is reduced by trace element contamination. Soil Biol. Biochem. 121, 202–211 (2018).
Justo, A. et al. Species recognition in Pluteus and Volvopluteus (Pluteaceae, Agaricales): Morphology, geography and phylogeny. Mycol. Prog. 10, 453–479 (2011).
Kuo, S. Phosphorus. In Methods of Soil Analysis: Part 3- Chemical Methods (ed. D. L., Sparks) 869–919 https://doi.org/10.1111/j.1469-7610.2007.01847.x (SSSA, 1996).
Houba, V. J. G., Temminghoff, E. J. M., Gaikhorst, G. A. & van Vark, W. Soil analysis procedures using 0.01 M calcium chloride as extraction reagent. Commun. Soil Sci. Plant Anal. 31, 1299–1396 (2000).
European Committee for Standardization. UNE-EN 15517:2008. Foodstuffs - Determination of trace elements - Determination of inorganic arsenic in seaweed by hydride generation atomic absorption spectrometry (HGAAS) after acid extraction. 1–17 (2008).
Galán, E. & Romero, A. Contaminación de suelos por metales pesados. in Sociedad Española de Mineralogía vol. 10 48–60 (2008).
Kruskal, J. B. Multidimensional scaling by optimizing goodness of fit to a nonmetric hypothesis. Psychometrika 29, 1–27 (1964).
Oksanen, J. et al. vegan: Community Ecology Package, https://cran.r-project.org/package=vegan (2019).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B 57, 289–300 (1995).
R Core Team. R: a language and environment for statistical computing, https://www.r-project.org (2018).
Wickham, H. ggplot2: elegant graphics for data analysis. (Springer-Verlag, 2016).
Gastwirth, J. L. et al. lawstat: tools for biostatistics, public policy, and law, https://cran.r-project.org/package=lawstat (2019).
Venables, W. N. & Ripley, B. D. Modern Applied Statistics with S. (Springer, 2002).
Gross, J. & Ligges, U. nortest: tests for normality, https://cran.r-project.org/package=nortest (2015).
Revelle, W. psych: procedures for psychological, psychometric, and personality research. https://cran.r-project.org/package=psych (2019).
WHO. Evaluations of the Joint FAO/WHO Expert Committee on Food Additives (JECFA). 84th JECFA, http://apps.who.int/food-additives-contaminants-jecfa-database/search.aspx (2017).
Acknowledgements
We thank Álvaro López-García and “Herbario de la Universidad de Sevilla” for fungal sequencing. We thank LIE-IRNAS for isotopic analysis and IRNAS Analytical Service for trace elements determination in soil and mushrooms. We thank Thomas Ashley for English proofreading. This work was supported by European Union Seventh Framework Programme (FP7/2007–2013) [grant number 603498 - RECARE], and Spanish Ministry of Science, Innovation and Universities [grants number CGL2014–52858-R – RESTECO and CGL2017–82254-R - INTARSU]. MGM was supported by Spanish Ministry of Economy and Competitiveness [grant number BES-2015–073882], and MTD by a postdoctoral grant by Universidad de Sevilla (V Plan Propio de Investigación).
Author information
Authors and Affiliations
Contributions
Marta Gil-Martínez: Conceptualisation, Methodology, Software, Formal Analysis, Investigation, Writing- Original draft, Writing- Reviewing and Editing. Carmen M. Navarro-Fernández: Methodology, Investigation, Writing- Reviewing and Editing. José M. Murillo: Conceptualisation, Methodology, Investigation, Writing- Reviewing and Editing. María T. Domínguez: Methodology, Writing- Reviewing and Editing. Teodoro Marañón: Conceptualisation, Methodology, Investigation, Writing- Reviewing and Editing, Supervision, Funding Acquisition.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gil-Martínez, M., Navarro-Fernández, C.M., Murillo, J.M. et al. Trace elements and C and N isotope composition in two mushroom species from a mine-spill contaminated site. Sci Rep 10, 6434 (2020). https://doi.org/10.1038/s41598-020-63194-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-63194-2
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.