Abstract
The homology and evolution of the archosaur ankle is a controversial topic that has been deeply studied using evidence from both extinct and extant taxa. In early stem archosaurs, the astragalus and calcaneum form the ancestral proximal tarsus and a single ossification composes the centrale series. In more recent stem archosaurs, the centrale is incorporated to the proximal row of tarsals laterally contacting the astragalus. This bone is subsequently lost as an independent ossification before the last common ancestor of birds and crocodilians, but the evolutionary fate of this element remains mostly unexplored. Here, we integrate embryological and palaeontological data with morphogeometric analyses to test the hypothesis of loss of the centrale or, alternatively, its incorporation into the archosaurian astragalus. Our results support the latter hypothesis, indicating that the astragalus developed ancestrally from two ossification centres in stem archosaurs and that the supposed tibiale of bird embryos represents a centrale. This conclusion agrees with previous embryological studies that concluded that the tibiale never develops in diapsids.
Similar content being viewed by others
Introduction
The tarsus of archosaurs – crocodilians, birds, non-avian dinosaurs and several other extinct clades – and their most immediate precursors has been one of the most deeply studied anatomical regions of this clade because of its strong phylogenetic signal and morphofunctional importance1,2,3,4,5. In particular, the homology of the primordial cartilages and their subsequent ossifications that form the archosaur tarsus has been a widely discussed topic that integrates palaeontological and embryological studies4,6,7,8,9,10,11,12. The proximal tarsus of archosaurs is ancestrally composed of a medial astragalus that articulates proximally with the tibia and fibula and a lateral calcaneum that articulates proximally with the fibula3,13. Separate astragalar and calcaneal ossifications have been retained in Crocodylia, but both bones fuse into a single proximal tarsal in Aves7. During the embryological development of extant crocodilians, the chondrogenic proximal tarsus possesses three condensations – the intermedium, centrale and fibulare – that subsequently ossify into two elements – the astragalus and calcaneum –10,11.
A single ossification originating from the four elements that are proposed to compose the centrale series of amniotes (e.g. juveniles of captorhinids, such as Moradisaurus grandis14) is retained in stem archosaurs5. This single centrale is located immediately mediodistal to the astragalus in the earliest archosauromorphs (e.g. Protorosaurus speneri15) but is incorporated to the proximal row of tarsals, laterally contacting the astragalus, in more crownward stem archosaurs16. As a result, in the latter taxa, the centrale, together with the astragalus, forms part of the articular facet for the distal end of tibia. In archosauromorphs more closely related to Archosauria, the centrale is lost as an independent ossification5,16. The absence of a centrale has long been recognized as a synapomorphy within stem Archosauria4,5,16, but the evolutionary fate of this element has received relatively poor attention. Some authors have proposed that the centrale fuses with the astragalus to form a single medial proximal tarsal12,17. However, this hypothesis remains untested. Here, we integrate embryological and palaeontological data and quantitative methodologies to test the hypothesis of fusion between the centrale and astragalus, or the alternative hypothesis of a complete loss of this element. The potential recognition of the centrale as a component of the proximal tarsus of archosaurs has interesting evolutionary implications in the discussion of the homology of the primordial cartilages that form the astragalus in amniotes and the homology of the cartilaginous condensations of bird embryos.
Results
Embryological development of the tarsus of Caiman
In the earliest-sampled embryonic growth stages of Caiman yacare [CY] and Caiman latirostris [CL] (MLP-R.6491 CL-17/18 and MLP-R.6490 CY-17/18, see Materials and Methods for nomenclature of growth stages) there are two cartilaginous condensations that articulate proximally and proximolaterally with the distal end of tibia (Fig. 1a–c), resembling the condition in bird embryos18. A single thin layer of cartilage extends over the proximal surface of both condensations and contributes to the tibial articulation (MLP-R.6490 CY-17/18-1). We interpret these condensations as the intermedium laterally and the centrale medially. The proximal surface of the intermedium articulates with the lateral and medial halves of the distal end of tibia and fibula, respectively. The lateral surface of the intermedium articulates with the fibulare and there is a thin layer of cartilage that will participates in the future crurotarsal joint. This same layer extends distally to form the articulation between the fibulare and a large distal tarsal 4. The distal tarsal 3 is present medial to the distal tarsal 4 and proximal to the metatarsal III, and immediately proximal to metatarsal II there is a small, spherical condensation that we identify as distal tarsal 2. In the later embryonic stage (MLP-R.6490 CY-19) there is no differentiation between an intermedium and centrale, which creates a single centre of ossification, the astragalus, in more advanced embryos. In all sampled stages of both species we have not observed a segmentation or a condensation adjacent to the distal end of the tibia that may indicate the development of a tibiale, in agreement with the absence of this element in at least several diapsids – including turtles19 – (e.g. refs. 11,12,20).
(a–c) Photographs of embryos of (a) Caiman latirostris and (b,c) Caiman yacare showing the chondrifications of the tarsus during embryological development; and (d–f) astragali and centrale of the Triassic non-archosaurian archosauromorphs (d) Proterosuchus sp. (AMNH FR 2237), (e) Pamelaria dolichotrachela (ISIR 316) and (f) a Rhadinosuchinae indet. (CRILAR-Pv 492) in anterior views. Abbreviations: I–V, digits I–V; as, astragalus; ca, calcaneum; ce, centrale; ct, calcaneal tuber; dt2–4, distal tarsals 2–4; in, intermedium; f.ca, facet for calcaneum; f.fi, facet for fibula; f.ti, facet for tibia; fi, fibula; fib, fibulare; nag, non-articulating gap; p.as, precursor of the astragalus; pf, perforating foramen; su, suture; ti, tibia. Scale bars equal 0.2 mm in (a–c), 1 cm in (d) and 5 mm in (e,f). Photographs (b,d,f) reversed.
Evolution of the tarsus in early archosauromorphs
In the ancestral condition of the archosauromorph tarsus, the centrale is located ventral to the medial half of the astragalus and, as a result, it does not articulate with the tibia. In more crownward archosauromorphs (Crocopoda), the centrale occupies a proximal position in the tarsus and is located medial to the astragalus, participating in the articulation with the tibia4,5,12,16 (Fig. 1d,e). In addition, the centrale fuses with the astragalus in some individuals of allokotosaurs, rhynchosaurs and proterosuchids17,21. All known specimens of the azendohsaurid allokotosaurs Pamelaria dolichotrachela (ISIR 316/58), Azendohsaurus madagaskarensis17 and Shringasaurus indicus21 present such fusion, but in the trilophosaurid allokotosaur Trilophosaurus buettneri its presence is polymorphic and apparently not related to the size of the individuals17. The astragalus and centrale are separated from each other in Early and Middle Triassic rhynchosaurs22,23,24,25, but the co-ossification between both bones occurs in large-sized individuals of at least some hyperodapedontine rhynchosaurs17. Among the Proterosuchidae, a small-sized and probably immature individual of Proterosuchus fergusi (SAM-PK-K14026) possesses an astragalus unfused to the centrale (the latter bone is not preserved in this specimen). By contrast, the astragalus is co-ossified with the centrale in larger specimens of the genus (e.g. Proterosuchus sp.: AMNH FR 2237; Proterosuchus alexanderi: NMQR 1484). Thus, at least in proterosuchids, it is possible that the fusion between the centrale and astragalus occurs later in post-hatching ontogeny. In those crocopods, in which the centrale is fused to the astragalus, a line of suture or ventral cleft persists showing the original separation between both elements (Fig. 1d,e). In Erythrosuchidae and Eucrocopoda, the centrale is absent as a separate ossification and the tibial-tarsal facet retains a similar proportional size, although it is formed only by the astragalus (Fig. 1f).
Analyses of the morphogeometric configurations
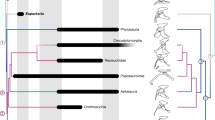
The optimization of the two alternative morphogeometric configurations (sampling only the astragalus or the astragalus + centrale) in the phylogenetic supertrees (see Materials and Methods) recovers the transformation of an astragalus + centrale into the astragalus as more parsimonious than the alternative hypothesis of loss of the centrale (taking into account the length of the branch along which the loss of a separate centrale is optimized) (Fig. 2; Table 1). The main landmark transformation in this branch (i.e. Erythrosuchidae + Eucrocopoda) corresponds to a lateral displacement of the ventral margin of the calcaneal facet (Landmark 8) in both trees (Supplementary Information 1, Supplementary Table S3). This change is related to a shallower notch that receives the ventral portion of the astragalar facet of the calcaneum, which in turn probably corresponds to the loss of the foramen for the passage of the perforating artery in the Erythrosuchidae + Eucrocopoda branch16. In the trees optimizing the astragalus + centrale, other changes in landmark and semilandmark positions are considerably lower and the transformations of the landmarks that describe the shape of the medial margin of the structure (i.e. Landmark 2 and semilandmark 2) are very minor. By contrast, in the other configuration, there is a distinct ventromedial displacement of Landmark 2 and a lower, but conspicuous, dorsolateral displacement of semilandmark 2. These changes in the latter optimization show that a medial expansion of the astragalus is the most parsimonious transformation in this branch. However, in the other configuration (astragalus + centrale), the medial expansion of the astragalus is not necessary because this space is occupied by the centrale in more basal branches.
Optimization of both morphogeometric configurations shown in a reduced phylogeny of early archosauromorphs (after Sengupta et al.22). Colours in the branches indicate the difference in the number of steps between both configurations for the landmarks that sample the medial margin of the astragalus and/or centrale (landmarks 2, 6 and 9, and semi-landmarks 1 and 2). Recovered hypothetical ancestral configurations (red lines) are shown above (only astragalus) and below (astragalus + centrale) each branch. Dotted grey lines show the configuration ancestral to that node and the blue lines indicate the displacement of the landmarks in the ancestor-descendant transformation.
In the Principal Component Analysis, the early archosauromorphs that retain a distinct centrale (i.e. allokotosaurs and proterosuchids) occur at the bottom of the right side of the morphospace (Supplementary Fig. 4). In this quadrant, the early archosauromorphs that are more distant to the region occupied by members of the Erythrosuchidae + Eucrocopoda clade are those for which the centrale was not sampled in the morphogeometric configuration. By contrast, when both astragalus and centrale are sampled together, these taxa occupy a position closer to more derived archosauromorphs and the centroid of the morphospace. In agreement, the Sum of Variances (SoV) of the combined dataset found a lower disparity (SoV = 0.03135668 versus 0.03382903) when the astragalus of erythrosuchids + eucrocopods incorporates the centrale of more basal archosauromorphs.
Discussion
The evolutionary fate of the archosauromorph centrale
Some previous authors have interpreted that the medial proximal tarsal of Archosauria is a result of the fusion between the centrale and astragalus that occur as separate ossifications in post-hatching individuals ancestrally in Archosauromorpha (e.g. refs. 5,12,17). The results recovered from the optimization of the morphogeometric configurations in the phylogenies and Principal Component Analyses are the first quantitative evidences that support the hypothesis that the centrale has actually fused to the astragalus to form a single ossification in the Erythrosuchidae + Eucrocopoda clade rather than that the centrale failed to ossify. The hypothesis of fusion between the astragalus and centrale agrees with the retention of a suture line in the co-ossified elements that form the medial proximal tarsal of allokotosaurs, some hyperodapedontine rhynchosaurs and medium- to large-sized proterosuchid specimens. The astragalus-centrale fusion also matches the topological relationship and subsequent fusion between the two chondrogenic condensations during the embryological development of the medial proximal tarsal in crocodilians (intermedium and centrale; e.g. Caiman yacare and Caiman latirostris: this study; Alligator mississippiensis11; Melanosuchus niger27) and birds (intermedium and tibiale, e.g. ref. 18; but see below). The fusion between the astragalus and centrale occurs during the post-hatching ontogeny in proterosuchid archosauriforms, but the merging of the structures that we consider as equivalent (i.e. intermedium and centrale) takes place during the stage of cartilaginous condensation in the tarsus of crocodilian and bird embryos. This results in a single ossification centre that forms the definitive astragalus of extant archosaurs. As a consequence, we interpret that a peramorphic heterochronic event in the timing of fusion between the components of the medial proximal tarsal should have occurred in the branch leading to the Erythrosuchidae + Eucrocopoda clade.
The centrale and tibiale as components of the astragalus in archosauromorphs
Multiple embryological studies have shown that there are two chondrogenic condensations that form the single ossification centre that constitutes the astragalus of crocodilians and birds. In particular, Ossa-Fuentes et al.18 have recently found compelling evidence that the intermedium forms the ascending process of the astragalus (a structure absent in crocodilians and synapomorphic for Dinosauriformes28) and another chondrogenic condensation forms the astragalar body during the embryological development of birds. These authors identified this latter condensation as a tibiale following previous embryological studies of birds, although they acknowledge that other authors have proposed the absence of a tibiale in reptiles and that this cartilage was alternatively interpreted as a proximal centrale10,11. If a tibiale homologous to the ossified tibiale of early tetrapods is present in bird embryos it would contradict the hypothesis supported here of the incorporation of the centrale to the astragalus –to form a single centre of ossification– in early archosaurs and their immediate precursors (e.g. Euparkeria, proterochampsids).
The tibiale develops from a transverse segmentation of the distal end of the tibia in non-amniote tetrapods9,10,11 and embryological studies on reptile limb development have not observed such segmentation and, as a result, proposed that the tibiale never forms in diapsids9,10,11,12,20. Instead, the element that chondrifies in the equivalent position as the tibiale in diapsid embryos lacks a connection with the distal end of tibia, but it contacts the intermedium and has been therefore interpreted as a centrale10. As a consequence, the medialmost chondrification in the proximal tarsus of bird embryos should not be homologous to a tibiale. The evidence presented here for an archosaur astragalus composed of the ancestral centrale and astragalus of diapsids reinforces the interpretation that the two chondrogenic condensations that form the astragalus in birds should be identified as the intermedium and the centrale, as occurs in lepidosauromorph, turtle and crocodilian embryos (e.g. refs. 11,12,27,29).
Our interpretations also have implications for the homology of the single centrale ossification retained in archosauromorphs. The separate chondrogenic condensation of the most preaxial centrale (centrale 1) originates from a segmentation of the tibiale in non-amniote tetrapods (e.g. Ambystoma10). Thus, the absence of a tibiale in Diapsida implies the absence of such preaxial centrale. Previous authors have interpreted that the intermedium and centrale 4 are integrated to form the amniote astragalus (the three-centre model of Peabody8). An alternative hypothesis, the four-centre model, also includes the centrale 3 as part of the amniote astragalus14. As a consequence, following these evolutionary hypotheses that the centrale 4, and possibly the centrale 3 as well, form part of the astragalus, the single independent centrale ossification retained in archosauromorphs should be homologous to the centrale 2 (given the four-centre model) or centrale 2 + 3 (given the three-centre model) of non-amniote tetrapods. This interpretation contradicts the traditional hypothesis that the centrale that chondrifies in the tarsus of diapsid embryos is homologous to the centrale 4, a view often followed by embryological studies (e.g. refs. 27,29). In addition, the centrale of crocodilian embryos and early archosauromorphs shows a topological relationship that matches the position of centrale 2 in early tetrapods14.
The evolution of the developmental pathway of the archosauromorph astragalus
The results obtained here allow hypothesizing about the developmental pathways of the archosauromorph astragalus. The centrale of early archosauromorphs forms the medial one-third of the proximal tarsus. As a result, we infer that the centrale should also have been medially restricted in the embryos of these extinct species (Fig. 3). By contrast, the homologous chondrogenic condensations in crocodilian embryos (centrale and intermedium) form each approximately half of the medial proximal tarsal. Thus, it could be inferred that during the evolution towards extant crocodilian species a modification of the developmental pathway occurred in which the centrale increased its proportional participation in the composition of the ossification centre that results in the astragalus. Unfortunately, we are not aware of any fossil pseudosuchian embryo or juvenile that preserves evidence of the size of the separate condensations that constitute the astragalus and, as a consequence, we lack information of how this modification occurred through phylogeny.
Simplified cladogram of Archosauromorpha (after Ezcurra17) showing the evolution of the proximal tarsus hypothesized here. Photographs of post-hatching specimens in anterior view (right) and outline drawings of cartilaginous condensations in extant species as continuous lines (Crocodylia and Aves) or hypothetical condensations as dotted lines for fossil species (left). In all cases lateral is to the right of the figure. Abbreviations: I–V, metatarsals I–V; as, astragalus; asp, ascending process; ca, calcaneum; ce, centrale; d4, distal tarsal 4; fi, fibulare; in, intermedium; tbt, tibiotarsus; tmt; tarsometatarsus. Collection numbers: Macrocnemus bassanii (PIMUZ T4355); Proterosuchus alexanderi (NMQR 1484); Rhadinosuchinae indet. (CRILAR-Pv 492); Lewisuchus admixtus (MACN-Pv 18954); Pandroravenator fernandezorum (MPEF-PV 1733-4).
It is interesting to note that, at least in the early embryological development of some bird embryos, the intermedium and centrale contribute in an approximately equal amount to the medial proximal tarsal, but this condition changes subsequently in later embryological stages as the centrale comes to form most of the astragalus and the intermedium becomes restricted to the ascending process18. Therefore, it could be hypothesized that the ancestral archosaur had a sub-equally developed intermedium and centrale because this is the condition observed in the embryos of both extant birds and crocodilians. The fossil record of theropod dinosaurs preserves evidence of the modifications in the developmental pathway of the astragalus. The early tetanuran Pandoravenator fernandezorum and some other basal averostrans possess a well-defined, mostly transverse groove on the anterior surface of the astragalus30. This groove subdivides the bone into a dorsomedial portion that includes the ascending process and a ventrolateral one that covers most of the astragalar body30. Thus, this condition seems to represent an intermediate stage in which the intermedium was dorsomedially restricted in the astragalus and the centrale was extended substantially laterally and ventrally to the intermedium, from the inferred ancestral archosaur condition, to form most of the astragalar body. A condition closer to that present in late bird embryos is found in some fossil coelurosaurian theropod specimens, in which the subdivision of the astragalus is placed more dorsally than in early averostrans and suggests a more proximally restricted intermedium19,30.
Materials and Methods
Embryological analysis
Pre-hatching ontogenetic series of 37 embryos of Caiman latirostris and 34 of C. yacare were studied. Specimens are housed in the herpetological collection of the Museo de La Plata [MLP] (Buenos Aires, Argentina) under a collection number for each ontogenetic series, MLP-R.6490 for C. yacare and MLP-R.6491 for C. latirostris (Supplementary Table 1). In addition, the growth stage in each ontogenetic series is indicated as CL (Caiman latirostris) and CY (Caiman yacare) followed by the number of the embryonic stage sensu Iungman et al.31. The material was collected during three field trips to the Chaco Province in 2012, 2015 and 2018. All the eggs belong to two nests per species removed from nature and were artificially incubated with constant conditions of relative humidity (95%) and temperature (30 °C ± 1). Embryos of both species were collected every day until hatching, fixed and kept in a 5% formaldehyde solution with calcium carbonate. Embryonic stages 20 to 27–28 and 17/18 to 25 (sensu Iungman et al.31) were sampled in C. latirostris and C. yacare, respectively. Embryos were prepared for the observation of cartilage and bone according to the double staining and diaphanization technique of Taylor & Van Dyke32. Fixed embryos were immersed in baths of successively increasing alcohol concentration, embedded in Alcian Blue, submerged in 1% KOH, inserted in Alizarin red, and finally submerged again in 1% KOH. They were left in KOH solution until the material was totally cleared and the cartilages and bones were visible. Each specimen is conserved in glycerol and was studied using a Zeiss stereomicroscope and a NIKON Stereo Microscope SMZ745/SMZ745T magnifier with a NIKON NI-150 Illuminator illumination. Photographs were taken using a Nikon D40 camera.
We complied with all relevant ethical regulations for animal testing and research. All experimental protocols concerning embryonic manipulation were developed during the PhD research of MVFB and approved by the Facultad de Ciencias Naturales y Museo-Universidad de La Plata, accordingly with the governmental regulations of the Chaco Province.
Geometric morphometric analysis
The two-dimensional geometric morphometric analysis was conducted on a sample of five astragali and centrale and 37 astragali (in taxa lacking a separate centrale) of Triassic archosauromorphs. The landmarks were placed on photographs of the astragali and centrale in anterior view taken at first hand by one of the authors (MDE), and some other photographs were taken from the literature. The sampled archosauromorph species are housed in different repositories worldwide and are listed in Supplementary Table 2. The shape sample was focused on the outline of the dorsal, ventral and medial margins of the bone and not on the calcaneal articular region. Right tarsals were mirrored in Photoshop CS6 version 13.0 in order to always analyze the same side (left). We selected a series of nine type II and III landmarks33 and two semilandmarks (Supplementary Table 3). We used two different morphogeometric configurations of the 42 taxa in order to test the hypothesis about the fate of the centrale bone. The first configuration samples the astragalus and centrale as a single unit to test the hypothesis that the centrale is incorporated to the astragalus to form a unique undivided structure in erythrosuchids and eucrocopods. On the other hand, the second configuration samples only the astragalus to test the alternative hypothesis (Supplementary Fig. 1).
The landmarks and semilandmarks were digitized using the programs tpsUtil 1.7634 and tpsDig2 2.3135. A Generalized Procrustes analysis was conducted on both configurations, sliding the semilandmarks along their tangent directions using the Procrustes distance criterion, with the function gpagen of the package geomorph 3.0.136 in the software environment R37. A Principal Component Analysis was conducted on all the aligned coordinates and the Sum of Variances was calculated for both groups, respectively, as a descriptor of the morphospace. The aligned coordinates of each configuration were exported to TNT 1.538 and they were used as a single morphogeometric continuous character to test each hypothesis. The configurations were re-aligned in TNT applying the minimum distances criterion and optimized using maximum parsimony on two alternative phylogenetic topologies (Supplementary Information 1). In both topologies, the tree was rooted with a clade composed of three allokotosaurian species (Pamelaria dolichotrachela, Shringasaurus indicus and Azendohsaurus madagaskarensis). Thus, the re-alignment of the data set in TNT was conducted using Pamelaria dolichotrachela as reference taxon because it is the earliest branching of the sampled allokotosaurs.
The hypotheses of incorporation of the centrale to the astragalus or complete loss of the former bone in archosauromorphs were tested comparing the length of the branch in which the centrale is absent as a different ossification (i.e. the Erythrosuchidae + Eucrocopoda clade) between the different topologies. In this way, the configuration that requires the highest number of steps in that branch will be the least parsimonious and its associated hypothesis will be rejected over the other. We used a scale in millimetres for the scaling of each morphogeometric configuration and, as a result, the differences in branch lengths and the total number of steps for each tree are expected to be relatively low (i.e. a magnitude of decimals). However, this optimization is based on maximum parsimony and any difference, regardless of how small it is (contrasting with probabilistic methods), is enough to prefer one hypothesis over others. We did not conduct a probabilistic Templeton test to explore statistical differences between optimizations because this analysis evaluates differences between alternative topologies, but not differences in character optimizations using the same topology.
Data availability
All the data analysed in this study are available in the Supplementary Data Files.
References
Cruickshank, A. R. I. The ankle joint in some early archosaurs. S. Afr. J. Sci. 75, 168–178 (1979).
Chatterjee, S. Phylogeny and classification of thecodontian reptiles. Nature 295, 317–320 (1982).
Sereno, P. C. Basal archosaurs: phylogenetic relationships and functional implications. Soc. Vertebr. Paleontol. Mem. 2, 1–53 (1991).
Gower, D. J. The tarsus of erythrosuchid archosaurs, and implications for early diapsid phylogeny. Zool. J. Linn. Soc. 116, 347–375 (1996).
Nesbitt, S. J. The early evolution of archosaurs: relationships and the origin of major clades. B. Am. Mus. Nat. Hist. 352, 1–292 (2011).
Gegenbaur, C. Untersuchungen zur vergleichenden Anatomie der Wirbelthiere. Erstes Heft. Carpus und Tarsus. (Wilhelm Engelmann, Leipzig, 1864).
Romer, A. S. Osteology of the reptiles. (The University of Chicago Press, Chicago & London, 1956).
Peabody, F. E. The origin of the astragalus of reptiles. Evolution 5, 339–344 (1951).
Burke, A. C. & Alberch, P. The development and homology of the chelonian carpus and tarsus. J. Morphol. 186, 119–131 (1985).
Shubin, N. H. & Alberch, P. In Evolutionary Biology (eds. Hecht, M. K., Wallace, B. & Prance, G. T.) 319–387 (Springer, 1986).
Müller, G. B. & Alberch, P. Ontogeny of the limb skeleton in Alligator mississippiensis: developmental invariance and change in the evolution of archosaur limbs. J. Morphol. 203, 151–164 (1990).
Rieppel, O. Studies on Skeleton Formation in Reptiles. IV. The Homology of the Reptilian (Amniote) Astragalus Revisited. J. Vertebr. Paleontol. 13, 31–47 (1993).
Cruickshank, A. R. I. The pes of Erythrosuchus africanus Broom. Zool. J. Linn. Soc. 62, 161–177 (1978).
O’Keefe, F. R., Sidor, C. A., Larsson, H. C., Maga, A. & Ide, O. Evolution and homology of the astragalus in early amniotes: new fossils, new perspectives. J. Morphol. 267, 415–425 (2006).
Gottmann-Quesada, A. & Sander, P. M. A redescription of the early archosauromorph Protorosaurus spenseri (Meyer, 1832) and its phylogenetic relationships. Palaeontogr. Abt. A 287, 123–220 (2009).
Ezcurra, M. D. The phylogenetic relationships of basal archosauromorphs, with an emphasis on the systematics of proterosuchian archosauriforms. PeerJ 4, e1778 (2016).
Nesbitt, S. J. et al. Postcranial anatomy and relationships of Azendohsaurus madagaskarensis. B. Am. Mus. Nat. Hist. 398, 1–126 (2015).
Ossa-Fuentes, L., Mpodozis, J. & Vargas, A. O. Bird embryos uncover homology and evolution of the dinosaur ankle. Nat. Commun. 6, 8902 (2015).
Bever, G. S., Lyson, T. R., Field, D. J. & Bhullar, B. A. S. Evolutionary origin of the turtle skull. Nature 525, 239–242 (2015).
Schaeffer, B. The morphological and functional evolution of the tarsus in amphibians and reptiles. B. Am. Mus. Nat. Hist. 78, 395–472 (1941).
Sengupta, S., Ezcurra, M. D. & Bandyopadhyay, S. A new horned and long-necked herbivorous stem-archosaur from the Middle Triassic of India. Sci. Rep. 7, 8366 (2017).
Carroll, R. L. Noteosuchus-the oldest known rhynchosaur. Ann. S. Afr. Mus. 72, 37–57 (1976).
Dilkes, D. W. The rhynchosaur Howesia browni from the Lower Triassic of South Africa. Palaeontology 38, 665–685 (1995).
Dilkes, D. W. The Early Triassic rhynchosaur Mesosuchus browni and the interrelationships of basal archosauromorph reptiles. Philos. T. Roy. Soc. B 353, 501–541 (1998).
Huene, F. von. Die fossilen Reptilien des südamerikanischen Gondwanalandes. Neues Jahrb. Min. Geol. Paläontol. Abteil. B 1938, 142–151 (1938).
Ezcurra, M. D. & Butler, R. J. Taxonomy of the proterosuchid archosauriforms (Diapsida: Archosauromorpha) from the earliest Triassic of South Africa, and implications for the early archosauriform radiation. Palaeontology 58, 141–170 (2015).
Vieira, L. G. Desenvolvimento embrionário de Melanosuchus niger (Crocodylia, Alligatoridae): descrição de estágios e ontogenia do esqueleto. PhD, Universidade de Brasilia, Brasilia, 2011).
Novas, F. E. Dinosaur monophyly. J. Vertebr. Paleontol. 16, 723–741 (1996).
Leal, F., Tarazona, O. A. & Ramírez‐Pinilla, M. P. Limb development in the gekkonid lizard Gonatodes albogularis: a reconsideration of homology in the lizard carpus and tarsus. J. Morphol. 271, 1328–1341 (2010).
Rauhut, O. W. & Pol, D. A theropod dinosaur from the Late Jurassic Cañadón Calcáreo formation of central Patagonia, and the evolution of the theropod tarsus. Ameghiniana 54, 539–567 (2017).
Iungman, J., Piña, C. I. & Siroski, P. Embryological development of Caiman latirostris (Crocodylia: Alligatoridae). Genesis 46, 401–417 (2008).
Taylor, W. R. & Van Dyke, G. C. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium 9, 107–119 (1985).
Bookstein, F. L. Morphometric tools for landmark data: geometry and biology. (Cambridge University Press, New York, 1991).
Rohlf, F. J. tpsUtil, Version 1.76. (State University of New York at Stony Brook, New York, 2018).
Rohlf, F. J. TpsDig2, version 2.31. (State University of New York at Stony Brook, New York, 2017).
Adams, D. C. & Otárola-Castillo, E. geomorph: an R package for the collection and analysis of geometric morphometric shape data. Methods Ecol. Evol. 4, 393–399 (2013).
R Development Core Team. R: A language and environment for statistical computing, software. (R Foundation for Statistical Computing, Vienna, 2017).
Goloboff, P. A. & Catalano, S. A. TNT version 1.5, including a full implementation of phylogenetic morphometrics. Cladistics 32, 221–238 (2016).
Acknowledgements
We thank the following curators, researchers and collection managers that provided access to specimens: Carl Mehling (AMNH), Saswati Bandyopadhyay and Dhurjati Sengupta (ISI), Daniela Schwarz (MB), Claudia Malabarba (MCP), Leandro Alcalde and Jorge Williams (MLP), Elize Butler and Jennifer Botha-Brink (NMQR), Jaime Powell and Pablo Ortiz (PVL), Ricardo Martínez (PVSJ), Rainer Schoch (SMNS), Kevin Padian, Pat Holroyd and Randall Irmis (UCMP), Mathew Lowe (UMZC), Michael Brett-Surman and Hans-Dieter Sues (UMZC), and Mateusz Talanda and Tomasz Sulej (ZPAL). We also thank Eduardo Bolaños and Oscar Gómez from “El Cachapé” farm, El Chaco Province, Argentina, for the access to caiman’s nests and embryos. Access to the free version of TNT 1.5 was possible due to the Willi Henning Society. This study was partially funded by the ANPCyT PICT No: 2016-0159 and CONICET PIP 112201301-00733 to P.B. MDE thanks the Emmy Noether Programme of the Deutsche Forschungsgemeinschaft (BU 2587/3-1 to Richard Butler), a Marie Curie Career Integration Grant (PCIG14-GA-2013-630123 to Richard Butler), and a National Geographic Young Explorers Grant (#9467-14 to M.D.E.) for financial support during collection visits.
Author information
Authors and Affiliations
Contributions
M.V.F.B., M.D.E. and P.B. conceived the research. The data set was compiled and analyzed by M.V.F.B. and M.D.E. All authors discussed results, prepared figures and contributed to writing the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Blanco, M.V.F., Ezcurra, M.D. & Bona, P. New embryological and palaeontological evidence sheds light on the evolution of the archosauromorph ankle. Sci Rep 10, 5150 (2020). https://doi.org/10.1038/s41598-020-62033-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-62033-8
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.