Abstract
An ossified or ‘fused’ mandibular symphysis characterizes the origins of the Anthropoidea, a primate suborder that includes humans. Longstanding debate about the adaptive significance of variation in this jaw joint centers on whether a bony symphysis is stronger than an unfused one spanned by cartilage and ligaments. To provide essential information regarding mechanical performance, intact adult symphyses from representative primates and scandentians were loaded ex vivo to simulate stresses during biting and chewing – dorsoventral (DV) shear and lateral transverse bending (‘wishboning’). The anthropoid symphysis requires significantly more force to induce structural failure vs. strepsirrhines and scandentians with unfused joints. In wishboning, symphyseal breakage always occurs at the midline in taxa with unfused conditions, further indicating that an ossified symphysis is stronger than an unfused joint. Greater non-midline fractures among anthropoids suggest that fusion imposes unique constraints on masticatory function elsewhere along the mandible, a phenomenon likely to characterize the evolution of fusion and jaw form throughout Mammalia.
Similar content being viewed by others
Introduction
The mandibular symphysis represents the third jaw joint of the mammalian masticatory system. It is one of the most interesting and morphologically complex joints in the body, varying from the primitive condition of smooth, opposing dentaries loosely connected by fibrous tissue, ligaments, a fibrocartilage pad and neurovascular bundles (amphiarthrosis) to a more tightly bound joint with greater sutural complexity consisting of variably interlocking rugosities and variably calcified ligaments (synarthrosis) to the derived condition of a wholly ossified or fused joint (synostosis) (Fig. 1)1,2,3,4,5,6,7. This remarkable evolutionary diversity in symphyseal character states is witnessed during the origin of Anthropoidea, where stem groups exhibit an unfused joint while crown clades such as Catarrhini and Platyrrhini evolved the synapomorphic condition of early ontogenetic fusion8,9,10,11,12,13,14,15. Symphyseal fusion has likewise characterized the evolution of diverse mammals, including extinct strepsirrhine primates as well as marsupials, megachiropterans, carnivorans, proboscideans, hyracoids, sirenians, artiodactyls and perissodactyls2,3,4,5,6,7,16,17,18,19,20,21,22.
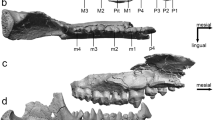
Variation in symphyseal character states across adult primates. Superior views of the symphysis in primitive (a: unfused), intermediate (b: partially fused) and derived (c: fused) conditions. The former two conditions are observed in extant strepsirrhines (Table 1), while modern anthropoids are characterized by only complete fusion of the mandibular symphysis.
Despite considerable experimental and comparative research, the functional significance of symphyseal fusion remains disputed, being linked either to joint strength or joint stiffness. This relates to the dual potential benefits and consequences of a joint formed by cortical bone. On one hand, a simple, unfused symphysis can experience considerable independent movement between mandibular halves – rotation about three axes and translation in at least two directions4,23,24. Taxa with intermediate levels of fusion experience reduced independent jaw movements depending on the orientation, number and mechanical properties of the cruciate and transverse ligaments as well as the degree of interdigitation among bony rugosities (Fig. 1)2,3,4,25. The apomorphic condition of a fused joint facilitates synchronous movements of the left and right jaws due to jaw-muscle activity on a single side26,27,28. It has been argued that ossification stiffens the symphysis for the effective transfer of transversely-oriented balancing-side (BS) jaw-adductor muscle forces during unilateral postcanine chewing and biting29,30 or vertically-oriented BS forces during off-midline incisal biting31,32. Such ‘Stiffness Models’ posit there is no necessary correspondence between symphyseal fusion and joint strength, with an unfused configuration being just as strong as an ossified one.
On the other hand, anthropoids differ from strepsirrhines with unfused joints in exhibiting higher levels of BS jaw-adductor muscle activity during unilateral mastication, which results in elevated peak strains along the BS mandible and increased dorsoventral (DV) shear and lateral transverse bending (‘wishboning’) of the symphysis (Fig. 2)6,26,27,28,33,34,35,36,37,38,39,40. Mammals with partial or intermediate fusion possess cruciate ligaments spanning the symphysis oriented primarily to resist DV shear; such ligaments are more calcified and attached to variably interlocking bony rugosities (Fig. 1 and 2)2,3,4. A subset of taxa with partial fusion also exhibit transversely-oriented ligaments spanning the posterior joint that appear well-designed to counter moderate levels of symphyseal wishboning (Fig. 2)2,3,6. It follows from this in vivo and comparative research that variation in joint form and fusion is continuous and potentially proportional to the amount of stress experienced along the symphysis during biting and chewing2,3,5,6,13,17,26,27,28,33,34,35,36,37,38,39. Consequently, such ‘Strength Models’ argue that increased fusion strengthens the symphysis against elevated stresses because the cortical bone in a synostosis is stronger than the ligaments and fibrocartilage spanning a similarly sized amphiarthrosis. In this scenario, increased joint stiffness is a by-product of selection for the greater strength conferred by a bony symphysis.
Symphyseal loading patterns during unilateral mastication. Superior views (a,b): Wishboning in the transverse plane is due to a laterally-directed component of bite force (Fb) on the working side and an oppositely-directed jaw-adductor force (Fm) on the balancing side at terminal phase I of the masticatory power stroke. In 2a, the red shaded area indicates the stress distribution across the joint whereby wishboning results in tension (t) along the lingual side and compression (c) along the labial side. Note that the neutral axis where the stresses shift between compressive and tensile is located closer to the lingual border, which results in high stress concentrations lingually due to the steep strain gradient related to the bending of curved beams. (b) Depicts an unfused joint with transverse ligaments (two horizontal lines) posterior to the fibrocartilage (FC) pad oriented to resist limited wishboning where the symphyseal surfaces are pulled apart along the lingual aspect and the FC pad is compressed anteriorly. Frontal views (c,d): DV shear in the coronal plane is due to an inferiorly-directed component of bite force (Fb) along the working side and a superiorly-directed balancing-side jaw-adductor force (Fm) along the opposite jaw. In 2c, the dotted rectangle shows how a solid square is deformed under DV shear, where the maximum principal strain (maximum tension) is ɛ1 and the minimum principal strain (maximum compression) is ɛ2. (d) Illustrates an unfused joint where only half of the DV cruciate ligaments (dotted line) are oriented to resist DV shear during left-sided chewing. The other cruciate ligaments experience compression and have no role in resisting left-sided DV shear. When chewing is right-sided, directions of force resultants are reversed and the cruciate ligaments (oblique solid line) oriented in the opposite direction then experience tension due to DV shear. Increased stiffness of cruciate ligaments due to greater calcification allows such tissues to counter compression and effectively double the amount of material able to resist DV shear. DV shear also results in shear of the labially located FC pad.
Although morphological transformations in symphyseal fusion are well documented during anthropoid origins8,9,10,11,12,13,14,15, surprisingly little data exist to address whether these phylogenetic changes are functionally related to joint strength or joint stiffness. This is due, in part, to the paucity of information regarding the mechanical properties of cranial tissues, and because the symphysis is comprised of fibrocartilage and not hyaline cartilage as observed in the postcranium41. Indeed, many aspects of the biomechanics of syndesmoses (cranial sutures) and synovial joints (temporomandibular joint) are better documented than for arthroses like the mandibular symphysis42,43,44,45. To this end, we performed experiments to evaluate the mechanical strength and integrity of the mandibular symphysis in a diverse sample of adult primates. We compared anthropoids and strepsirrhines varying in the degree of symphyseal fusion via ex vivo macroscale tests of joint strength during simulated masticatory loads – DV shear and wishboning (Fig. 2; Table 1)6,26,27,28,33,34,35,36,37,38,39. In addition to furnishing novel data on symphyseal fusion and joint performance concerning a long-recognized synapomorphy of crown anthropoids, our analyses are highly relevant to investigations that require functional information about jaw form for inferring the behavior of fossil remains.
Results and Discussion
Studies of musculoskeletal biomechanics routinely assume that phenotypic variation tracks differences in performance and, ultimately, fitness46,47. In terms of anthropoid origins, critical evidence has been lacking as to the mechanical performance of a fused mandibular symphysis, which hinders attempts to understand adaptive transformations in the fossil record48. The strength models predict that anthropoids will exhibit relatively and absolutely stronger symphyses that break at the midline to a lower extent than in strepsirrhines and scandentians. Thus, we investigated symphyseal strength and performance in several ways. Regression lines were compared for simulated ex vivo loads (wishboning, DV shear) necessary to induce structural failure of fused symphyses for anthropoids vs. strepsirrhines and scandentians with unfused or partially fused joints. To control for potential suborder variation in relative symphyseal size, fracture strengths were scaled to joint size and compared between anthropoids and strepsirrhines. To evaluate if the soft tissues of unfused joints are weaker in wishboning and DV shear, we compared symphyseal midline fracture frequencies during simulated loads between the anthropoid vs. strepsirrhine plus scandentian samples. To explore the relative importance of the two loading regimes on symphyseal strength, we then compared anthropoid regression lines describing the forces required to induce joint failure in wishboning vs. DV shear.
Suborder comparisons of the forces required to induce structural failure of the symphysis in wishboning (Fig. 3a) and DV shear (Fig. 3b) indicates that anthropoid allometric trajectories vs. mandibular length are significantly transposed above those for strepsirrhines (Table 2). Accompanying comparisons of symphyseal strength scaled to joint size confirms that anthropoids exhibit significantly higher relative strength in wishboning and DV shear than strepsirrhines (Table 3). In all such analyses, scandentians with unfused joints have similar relative levels of joint strength as strepsirrhines. Therefore, synostosis in anthropoids results in a significantly stronger symphysis, both absolutely and relatively, for countering masticatory stresses than the unfused joint of most strepsirrhines. These results are inconsistent with competing arguments that variation in fusion is unrelated to strengthening the mandibular symphysis29,30,31,32. Instead, our novel findings regarding symphyseal performance suggest that increased joint stiffness is a secondary consequence of increasing joint strength via synostosis.
Suborder comparison of symphyseal forces at joint failure during simulated: (a) Wishboning in 14 anthropoid, 20 strepsirrhine and 1 scandentian (Tupaia) species; and (b) DV shear in 7 anthropoid, 16 strepsirrhine and 1 scandentian (Tupaia) species. In both cases, the regression line for anthropoids (red) is significantly transposed above that for strepsirrhines and scandentians (blue), indicating that anthropoids have relatively stronger symphyses in lateral transverse bending and DV shear. Tupaia (*) exhibits an unfused joint like most strepsirrhines that also functions similarly. (Key:  = Anthropoidea;
= Anthropoidea;  = Strepsirrhini; *Tupaia).
= Strepsirrhini; *Tupaia).
Comparison of symphyseal failure locations between anthropoids and strepsirrhines indicate that the latter exhibit a significantly higher frequency of midline joint fractures during simulated wishboning loads (Fig. 4; Table 3). This pattern is predicted by the ‘Strength’ models, which posit that symphyseal soft tissues constitute the weakest component of an unfused joint, particularly with regard to high wishboning stress concentrations along the lingual surface of the symphysis where crack propagation is initiated49,50. In contrast, there are no suborder differences in the degree of midline joint fractures during DV shear (Table 3). Rather than providing support for the ‘Stiffness’ models, this discrepancy highlights the likelihood that wishboning is the singular functional determinant of symphyseal fusion in anthropoids. Since the anthropoid symphysis is equally strong in countering DV shear as it is in resisting wishboning (Fig. 5; Table 2), this suggests that the greater strength of the anthropoid symphysis in DV shear vs. strepsirrhines is largely due to being designed to resist elevated wishboning. Given that incision has been eliminated as a potential determinant of symphyseal fusion in primates5,13,17, our current findings provide unique empirical support for the central role of stresses during postcanine biting and chewing, specifically wishboning, as a determinant of symphyseal fusion in anthropoids.
Medial views of different symphyseal fracture patterns during simulated wishboning in primates: (a) right mandible and symphyseal surface in a longer-faced macaque with a midline joint fracture between the central incisors; and (b) left mandible and majority of the symphysis in a shorter-faced owl monkey with a joint fracture just lateral to the right canine.
Comparison of symphyseal forces at joint failure during simulated wishboning and DV shear in 14 and 7 anthropoid species, respectively. Regression lines for wishboning (red) and DV shear (blue) in anthropoids are coincidental, suggesting the symphysis is comparably designed for both jaw-loading regimes at a given size. (Key:  = wishboning;
= wishboning;  = DV shear).
= DV shear).
Indeed, the onset of wishboning and symphyseal fusion imposes a number of unappreciated constraints on phenotypic diversity in the anthropoid masticatory system. Previous work indicates that symphyseal midline fractures are more frequent in marmosets with more curved symphyses than other South American monkeys49,50. In our more diverse sample, three prognathic cercopithecine taxa with correspondingly greater joint curvature exhibit 100% midline fractures vs. a mean of 22.6% for nine species of shorter-faced anthropoids (Fig. 4; Supplementary Table 1). Increased symphyseal curvature generates higher wishboning stress concentrations that can be effectively countered via added cortical bone at the joint midline33,34. That symphyseal curvature increases with positive allometry during ontogeny and interspecifically in cercopithecine monkeys34,38 further underscores the overarching role of wishboning stress on symphyseal function and patterns of mandibular cortical bone distribution during phyletic size change in living and extinct anthropoids. This phenomenon in primate jaws also represents an example of skeletal curvature and load predictability more typically observed in vertebrate limb elements51.
As wishboning results in fractures at off-axis locations in more orthognathic anthropoids with less joint curvature, this suggests the presence of structural tradeoffs elsewhere along the mandible that also may be novel for this primate suborder. Herein, non-midline cracks due to wishboning differentially propogated through tooth roots along the anterior portion of the jaw. Many of these cases involved the canine, which is particularly well developed in anthropoids14. Although camelids do not exhibit wishboning, the presence of relatively larger incisor roots and less cortical bone along the symphysis has been invoked to explain why this clade is singular among selenodont artiodactyls in having novel functional tradeoffs underlying the evolution of a fused symphysis21. Accordingly, the evolution of orthognathy in anthropoids such as hominids may be characterized by greater functional limits on cortical bone quantity and quality in more anterior mandibular sites where large tooth roots represent sources of potential weakness due to the relative reduction of surrounding cortical bone to resist wishboning stress52,53.
Additional craniodental features are associated with the evolution of a wishboning loading regime during anthropoid origins. Phase II molar facets are crushing surfaces that are loaded maximally during jaw movement in the latter portion of Phase I occlusion28,54. A relatively larger, more transverse component to Phase II molar occlusal facets is noted for early anthropoids55,56,57,58,59. In concert with an increased emphasis on postcanine crushing and trends toward isodonty and isognathy13,60, the duration of the anthropoid masticatory power stroke occupies a relatively higher percentage of the chewing cycle61. Because of these derived occlusal trends, jaw-adductor muscle and jaw-kinematic patterns underscore the greater importance of the transverse component of the masticatory power stroke in anthropoids; they are likely to be linked both functionally and phylogenetically to the evolution of a fused symphysis designed to counter increased wishboning stress.
In sum, our study suggests that the evolution of symphyseal fusion in primates imposes novel constraints on other aspects of masticatory form and function62. This is consistent with findings that scandentians with unfused joints exhibit similar levels of joint strength as strepsirrhines. As symphyseal fusion is an important synapomorphy of diverse mammalian clades and does not exhibit evolutionary reversals13,22, our data on macroscale joint properties uniquely inform symphyseal trait performance and contribute key insights into the biomechanics of character-state variation and covariation in the skull of living and fossil mammals46,47,48,62.
Methods
Sample
Adult primates with unfused (amphiarthrosis), partially fused (synarthrosis) and fully fused (synostosis) symphyses were obtained and intact joints were fixed for mechanical tests of joint strength (Table 1). As an initial test of whether joint form and function is independent of phylogeny, we included a tree shrew species (Scandentia) as an outgroup with an unfused symphysis with the strepsirrhine sample of taxa with unfused and partially fused joints. All primate, tree shrew and rat (below) specimens were obtained postmortem with permission from personal collections, those of colleagues, and the Duke Lemur Center (DLC). Mandibles were used only if the adult dentition was fully erupted and there was a full complement of teeth in functional occlusion. To prevent degradation of soft tissues, joints were fixed in 10% buffered formalin. This facilitated the use of specimens obtained opportunistically due to death from natural causes that needed to be stored prior to experiments as well as frozen specimens from the DLC that uniquely sampled a broad range of rare strepsirrhines. Although fixation increases cross-linking among proteins that may differentially strengthen the cartilage and ligaments of an unfused joint, the mechanical properties of bone and some soft tissues are minimally affected63,64,65. Thus, while the disparity in joint performance among fusion cohorts might be greater in comparisons of ‘fresh’ specimens, the use of fixed specimens greatly increased our total samples. Comparisons of joint strength during wishboning for five adult rat jaws frozen for one month and then fixed vs. five adult rat jaws fixed without freezing indicate no difference between groups (means of 15.4 N vs. 15.8 N, Mann-Whitney U test, p > 0.05). This suggests that variation in sample preparation (frozen then fixed [DLC] vs. fixed only [all other samples]) does not significantly influence variation in the strength of an unfused symphysis vis-à-vis wishboning. If a species sample was comprised of more than one intact symphysis, about half the sample from each taxon was utilized in macroscale tests of joint performance during simulated DV shear. In such cases, the other half was used for macroscale tests of symphysis strength during simulated wishboning. However, due to small sample sizes in a number of taxa, there are differences in species composition for each suborder in the macroscale tests.
Mechanical tests of joint performance
We determined the strength (i.e., load at failure in Newtons, N) of formalin-fixed, articulated mandibular symphyses from cadavers in 14 anthropoid, 20 strepsirrhine and one scandentian species in either simulated wishboning or DV shear (Table 1; Supplementary Tables 1 and 2). The harvested symphyses were attached to a Universal Testing Machine (Instron, Norwood, MA) and loaded to structural failure in either loading regime at a constant rate of 2.54 cm/min49,50. Depending on specimen size, either a 250 N or 5,000 N load cell was used to measure load on each symphysis. To simulate wishboning, we fixed wires or metal posts to both sides of the jaw just posterior to the symphysis (to eliminate/minimize the bending moment arm) and attached the wires/posts to either the load cell or stationary grip. Each symphyseal half was loaded in lateral transverse bending to structural failure. To simulate DV shear, we potted each hemimandible in epoxy and, once hardened, a hole was drilled through the epoxy immediately lateral to the symphysis and orthogonal to the jaw’s long axis. Metal posts were inserted through the hole on each side and oriented in opposite directions prior to being fastened and attached to either the load cell or stationary grip. Subsequently, each symphyseal half was loaded to structural failure in simulated DV shear. For all macroscale tests, we recorded the force required for joint failure in N. While mammals do not load their symphyses to structural failure in vivo, predictions regarding ex vivo analyses are based on the previously determined correspondence in cortical bone between ultimate failure vs. failure in cyclical loading66. For all wishboning tests, fracture location was recorded to determine if ossified joints failed at different locations than in species with unfused joints. Due to a very limited number of taxa with partial fusion in our sample, we combined these force values (N) with those for species with unfused joints.
Statistics
Least-squares and reduced major axis bivariate regressions (p < 0.05) of species means were performed in each suborder between natural logs of measures of symphyseal strength (N) and mandibular length (0.1 mm). The latter tracks variation in masticatory size and estimates a masticatory load arm. Comparison of least-squares scaling trajectories between suborders employed ANCOVA (p < 0.05); similar analyses were utilized to compare allometric patterns within anthropoids for joint strength in wishboning vs. DV shear. To control for the effect of suborder variation in relative joint size on symphyseal strength, failure stress per joint area (N/mm2) was compared between suborders for each loading regime (ANOVA, p < 0.05). In each species, the frequency of specimens where the location of joint failure occurred at the symphyseal midline was calculated. These values were used for comparisons of suborder means performed via ANOVA (p < 0.05). Data used for the bivariate and univariate analyses are located in Supplementary Tables 1 and 2.
References
Rigler, L. & Mlinsek, B. Die Symphyse der Mandibula beim Rinde. Ein Beitrag zur Kenntnis ihrer Struktur und Funktion. Anat. Anz. 122, 293–314 (1968).
Beecher, R. M. Function and fusion at the mandibular symphysis. Am. J. Phys. Anthropol. 47, 325–336 (1977).
Beecher, R. M. Functional significance of the mandibular symphysis. J. Morphol. 159, 117–130 (1979).
Scapino, R. P. Morphological investigation into functions of the jaw symphysis in carnivorans. J. Morphol. 167, 339–375 (1981).
Ravosa, M. J. & Hylander, W. L. Function and fusion of the mandibular symphysis in primates: Stiffness or strength? In (eds. Fleagle, J. G. & Kay, R. F.) Anthropoid Origins 447–468 (Plenum Press, New York), (1994).
Ravosa, M. J. & Hogue, A. S. Function and fusion of the mandibular symphysis in mammals: A comparative and experimental perspective. In (eds. Ross, C. F. & Kay, R. F.) Anthropoid Evolution. New Visions 413–462 (Springer/Kluwer Publishers, New York), (2004).
Scott, J. E., Hogue, A. S. & Ravosa, M. J. The adaptive significance of mandibular symphyseal fusion in mammals. J. Evol. Biol. 25, 661–673 (2012).
Simons, E. L. Description of two genera and species of Late Eocene Anthropoidea from Egypt. Proc. Natl. Acad. Sci. USA 86, 9956–9960 (1989).
Simons, E. L. Diversity in the early Tertiary anthropoidean radiation in Africa. Proc. Natl. Acad. Sci. USA 89, 10743–10747 (1992).
Simons, E. L. Skulls and anterior teeth of Catopithecus (Primates: Anthropoidea) from the Eocene and anthropoid origins. Science 268, 1885–1888 (1995).
Beard, K. C., Qi, T., Dawson, M. R., Wang, B. & Li, C. A diverse new primate fauna from Middle Eocene fissure-fillings in Southeastern China. Nature 368, 604–609 (1994).
Beard, K. C., Tong, Y., Dawson, M. R., Wang, J. & Huang, J. Earliest complete dentition of an anthropoid primate from the late Middle Eocene of Shanxi Province, China. Science 272, 82–85 (1996).
Ravosa, M. J. Anthropoid origins and the modern symphysis. Folia Primatol. 70, 65–78 (1999).
Williams, B. A., Kay, R. F. & Kirk, E. C. New perspectives on anthropoid origins. Proc. Natl. Acad. Sci. USA 107, 4797–4804 (2010).
Ni, X. et al. The oldest known primate skeleton and early haplorhine evolution. Nature 498, 60–64 (2013).
Gingerich, P. D. & Sahni, A. Dentition of Sivaladapis nagrii (Adapidae) from the late Miocene of India. Int. J. Primatol. 5, 63–79 (1984).
Ravosa, M. J. Structural allometry of the mandibular corpus and symphysis in prosimian primates. J. Human Evol. 20, 3–20 (1991).
Ravosa, M. J. Mandibular form and function in North American and European Adapidae and Omomyidae. J. Morphol. 229, 171–190 (1996).
Simons, E. L., Rasmussen, D. T. & Gingerich, P. D. New cercamoniine adapid from Fayum, Egypt. J. Human Evol. 29, 577–589 (1995).
Hogue, A. S. & Ravosa, M. J. Transverse masticatory movements, occlusal orientation and symphyseal fusion in selenodont artiodactyls. J. Morphol. 249, 221–241 (2001).
Williams, S. H., Wall, C. E., Vinyard, C. J. & Hylander, W. L. Symphyseal fusion in selenodont artiodactyls: New insights from in vivo and comparative data. In (eds. Vinyard, C. J., Ravosa, M. J. & Wall, C. E.) Primate Craniofacial Function and Biology 39–61 (Springer Academic Publishers, New York), (2008).
Scott, J. E., Lack, J. B. & Ravosa, M. J. On the irreversibility of mandibular symphyseal fusion. Evol. 66, 2940–2952 (2012).
Crompton, A. W. & Hiiemäe, K. M. Molar occlusion and mandibular movements during occlusion in the American opossum, Didelphis marsupialis L. Zool. J. Linn. Soc. 49, 21–47 (1970).
Crompton, A. W., Thexton, A. J., Parker, P. & Hiiemäe, K. The activity of the jaw and hyoid musculature in the Virginian opossum, Didelphis virginiana. In (eds. Gilmore, D. & Robinson, B.) The Biology of Marsupials. 2: Biology and Environment 287–305 (Macmillan, London, 1977).
Ravosa, M. J. & Simons, E. L. Mandibular growth and function in. Archaeolemur. Am. J. Phys. Anthropol. 95, 63–76 (1994).
Hylander, W. L. Mandibular function in Galago crassicaudatus and Macaca fascicularis: An in vivo approach to stress analysis of the mandible. J. Morphol. 159, 253–296 (1979).
Hylander, W. L. The functional significance of primate mandibular form. J. Morphol. 160, 223–240 (1979).
Hylander, W. L., Johnson, K. R. & Crompton, A. W. Loading patterns and jaw movements during mastication in Macaca fascicularis: A bone-strain, electromyographic and cineradiographic analysis. Am. J. Phys. Anthropol. 72, 287–314 (1987).
Dessem, D. Interactions between jaw-muscle recruitment and jaw-joint forces in Canis familiaris. J. Anat. 164, 101–121 (1989).
Lieberman, D. E. & Crompton, A. W. Why fuse the mandibular symphysis? A comparative analysis. Am. J. Phys. Anthropol. 112, 517–540 (2000).
Greaves, W. S. A functional consequence of an ossified mandibular symphysis. Am. J. Phys. Anthropol. 77, 53–56 (1988).
Greaves, W. S. A reply to Ravosa and Hylander. Am. J. Phys. Anthropol. 90, 513–514 (1993).
Hylander, W. L. Stress and strain in the mandibular symphysis of primates: A test of competing hypotheses. Am. J. Phys. Anthropol. 64, 1–46 (1984).
Hylander, W. L. Mandibular function and biomechanical stress and scaling. Am. Zool. 25, 315–330 (1985).
Hylander, W. L., Ravosa, M. J., Ross, C. F. & Johnson, K. R. Mandibular corpus strain in primates: Further evidence for a functional link between symphyseal fusion and jaw-adductor muscle force. Am. J. Phys. Anthropol. 107, 257–271 (1998).
Hylander, W. L., Ravosa, M. J., Ross, C. F., Wall, C. E. & Johnson, K. R. Symphyseal fusion and jaw-adductor muscle force: An EMG study. Am. J. Phys. Anthropol. 112, 469–492 (2000).
Hylander, W. L. et al. Temporalis function in anthropoids and strepsirrhines: An EMG Study. Am. J. Phys. Anthropol. 128, 35–56 (2005).
Vinyard, C. J. & Ravosa, M. J. Ontogeny, function, and scaling of the mandibular symphysis in papionin primates. J. Morphol. 235, 157–175 (1998).
Vinyard, C. J. et al. Jaw muscle function and the origin of primates. In (eds. Ravosa, M. J. & Dagosto, M.) Primate Origins: Adaptations and Evolution 179–231 (Springer Academic Publishers, New York), (2006).
Vinyard, C. J., Wall, C. E., Williams, S. H. & Hylander, W. L. Patterns of variation across primates in jaw-muscle electromyography during mastication. Int. Comp. Biol. 48, 294–311 (2008).
Herring, S. W. & Ochareon, P. Bone – Special problems of the craniofacial region. Orthod. Craniofac. Res. 8, 174–182 (2005).
Mao, J. J., Wang, X. & Kopher, R. A. Biomechanics of craniofacial sutures: Orthopedic implications. Angle Orthod. 73, 128–135 (2003).
Byron, C. D. et al. Effects of increased muscle mass and mouse sagittal suture morphology and mechanics. Anat. Rec. 279A, 676–684 (2004).
Sun, Z., Lee, E. & Herring, S. W. Cranial sutures and bones: Growth and fusion in relation to masticatory strain. Anat. Rec. 276A, 150–161 (2004).
Ravosa, M. J. & Kane, R. J. Dietary variation and mechanical properties of articular cartilage in the temporomandibular joint: Implications for the role of plasticity in mechanobiology and pathobiology. Zool. 124, 42–50 (2017).
Arnold, S. J. Morphology, performance and fitness. Am. Zool. 23, 347–361 (1983).
Wainwright, P. C. Ecomorphology: Experimental functional anatomy for ecological problems. Am. Zool. 31, 680–693 (1991).
Lauder, G. V. On the inference of function from structure. In (ed. Thomason, J. J.) Functional Morphology in Vertebrate Paleontology 1–18 (Cambridge University Press, Cambridge), (1995).
Hogg, R., Ravosa, M. J., Ryan, T. M. & Vinyard, C. J. The functional morphology of the anterior masticatory apparatus in tree-gouging marmosets (Cebidae, Platyrrhini). J. Morphol. 272, 833–849 (2011).
Vinyard, C. J. et al. Are we looking for loads in all the right places? New research directions for studying the masticatory apparatus of New World monkeys. Anat. Rec. 294A, 2140–2157 (2011).
Bertram, J. E. & Biewener, A. A. Bone curvature: Sacrificing strength for load predictability? J. Theor. Biol. 131, 75–92 (1988).
Wood, B. A., Abbott, S. A. & Uytterschaut, H. Analysis of the dental morphology of Plio-Pleistocene hominids IV. Mandibular postcanine root morphology. J. Anat. 156, 107–139 (1988).
Haile-Selassie, Y. et al. New species from Ethiopia further expands Middle Pliocene hominin diversity. Nature 521, 483–488 (2015).
Teaford, M. F. Molar microwear and diet in the genus. Cebus. Am. J. Phys. Anthropol. 66, 363–370 (1985).
Hiiemäe, K. M. & Kay, R. F. Trends in the evolution of primate mastication. Nature 240, 486–487 (1972).
Hiiemäe, K. M. & Kay, R. F. Evolutionary trends in the dynamics of primate mastication. In (ed. Zingeser, M. R.) Symposia of the Fourth International Congress of Primatology, Volume 3: Craniofacial Biology of Primates 28–64 (S. Karger, Basel), (1973).
Kay, R. F. & Hiiemäe, K. M. Jaw movement and tooth use in recent and fossil primates. Am. J. Phys. Anthropol. 40, 227–256 (1974).
Kay, R. F. The evolution of molar occlusion in the Cercopithecidae and early catarrhines. Am. J. Phys. Anthropol. 46, 327–352 (1977).
Rasmussen, D. T. & Simons, E. L. Paleobiology of the oligopithecines, the earliest known anthropoid primates. Int. J. Primatol. 13, 477–508 (1992).
Ravosa, M. J., Vinyard, C. J., Gagnon, M. & Islam, S. A. Evolution of anthropoid jaw loading and kinematic patterns. Am. J. Phys. Anthropol. 112, 493–516 (2000).
Hiiemäe, K. M. Mammalian mastication: A review of the activity of the jaw muscles and the movements they produce in chewing. In (eds. Butler, P. M. & Joysey, K. A.) Development, Function and Evolution of Teeth 359–398 (Academic Press, New York), (1978).
Cheverud, J. M. Phenotypic, genetic, and environmental morphological integration in the cranium. Evol. 36, 499–516 (1982).
Bauer, A. M., Russell, A. P. & Shadwick, R. E. Mechanical properties and morphological correlates of fragile skin in gekkonid lizards. J. Exp. Biol. 145, 79–102 (1989).
Currey, J. D., Brear, K., Zioupos, P. & Reilly, G. C. Effect of formaldehyde fixation on some mechanical properties of bovine bone. Biomat. 16, 1267–1271 (1995).
Wilke, H.-J., Krischak, S. & Claes, L. E. Formalin fixation strongly influences biomechanical properties of the spine. J. Biomech. 29, 1629–1631 (1996).
Currey, J. D. The Mechanical Adaptations of Bones (Princeton University Press, Princeton), (1984).
Acknowledgements
The following are thanked for assistance with various aspects of the study: W. Hylander, E. Lautenschlager, S. Khanna, D. Brewer, E. Ehmke, S. Zehr, A. Taylor, M. Teaford, J. Scott, S. Williams, C. Wall and T. Smith. S. Lad, E. Franks, E. Nett, S. Cortese, B. Anderson and two anonymous reviewers offered helpful comments on this manuscript. The Duke Lemur Center (DLC) provided access to many of the strepsirrhine specimens used herein. This research was funded by NSF grant BCS-0924592/1214766.
Author information
Authors and Affiliations
Contributions
M.J.R. and C.J.V. designed the study. C.J.V. and M.J.R. collected the data. C.J.V. and M.J.R. performed the research. M.J.R and C.J.V. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ravosa, M.J., Vinyard, C.J. Masticatory Loading and Ossification of the Mandibular Symphysis during Anthropoid Origins. Sci Rep 10, 5950 (2020). https://doi.org/10.1038/s41598-020-62025-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-62025-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.