Abstract
The liver is a unique organ that has a phenomenal capacity to regenerate after injury. Different surgical procedures, including partial hepatectomy (PH), intraoperative portal vein ligation (PVL), and associated liver partition and portal vein ligation for staged hepatectomy (ALPPS) show clinically distinct recovery patterns and regeneration. The observable clinical differences likely mirror some underlying variations in the patterns of gene activation and regeneration pathways. In this study, we provided a comprehensive comparative transcriptomic analysis of gene regulation in regenerating rat livers temporally spaced at 24 h and 96 h after PH, PVL, and ALPPS. The time-dependent factors appear to be the most important determinant of post-injury alterations of gene expression in liver regeneration. Gene expression profile after ALPPS showed more similar expression pattern to the PH than the PVL at the early phase of the regeneration. Early transcriptomic changes and predicted upstream regulators that were found in all three procedures included cell cycle associated genes (E2F1, CCND1, FOXM1, TP53, and RB1), transcription factors (Myc, E2F1, TBX2, FOXM1), DNA replication regulators (CDKN1A, EZH2, RRM2), G1/S-transition regulators (CCNB1, CCND1, RABL6), cytokines and growth factors (CSF2, IL-6, TNF, HGF, VEGF, and EGF), ATM and p53 signaling pathways. The functional pathway, upstream, and network analyses revealed both unique and overlapping molecular mechanisms and pathways for each surgical procedure. Identification of molecular signatures and regenerative signaling pathways for each surgical procedure further our understanding of key regulators of liver regeneration as well as patient populations that are likely to benefit from each procedure.
Similar content being viewed by others
Introduction
While adult hepatocytes are normally quiescent, they show phenomenal replicative potential when parenchymal loss occurs. This has made the liver an excellent model for studying organ regeneration1,2,3,4. The regenerative process is typically divided into the priming, proliferating and termination phases, occurring approximately at first, third and seventh days after resection, respectively5. Clinically, the regenerative ability of livers provides the cornerstone of many surgical treatments for liver diseases; resection of diseased portions seems to improve survival6. However, extensive removal of liver parenchyma, which may be necessary in some cases, may overcome the functional reserve of the remaining hepatocyte population, leading to decompensation, failure and even mortality7. To avoid this complication, pre-operative iatrogenic induction of hepatocyte proliferation can be attempted to increase the future remnant tissue’s resilience against the anticipated surgical volume loss7.
Several options are available to induce the regenerative capacity of the liver. While cellular proliferation may be chemically induced8, surgical induction procedures provide additional advantages such as intraoperative assessment of the extent of disease, anatomically-targeted induction, and radical disease resection7. These procedures include: Partial hepatectomy (PH), intraoperative portal vein ligation (PVL), percutaneous portal vein embolization (PVE), and associated liver partition and portal vein ligation for staged hepatectomy (ALPPS). Rates at which the remaining tissue regenerates differ between procedures. For example, three to twelve weeks are needed after PVL to achieve adequate regenerative volume compared to 7–8 days in ALPPS9. Similarly, Schlegel et al. showed a significantly accelerated increase in future liver remnant volume after ALPPS than PVL (1 vs. 4 days, respectively)10.
Contrary to other organs where dedicated stem cells replace injured tissues11, functional hepatocytes appear to be responsible for liver regeneration12. Therefore, major shifts in gene activation and transcription patterns are needed for the transformation from fully-differentiated to highly-proliferative cells. Interestingly, while eventual replacement of lost tissue is a common outcome, contemporary evidence suggests that genomic and transcriptomic alterations may vary based on regeneration-driving factors8. Indeed, systematic analysis of genome-wide shifts can identify either common or specific signatures of different procedures, which may provide insights into the key regulators of such a complex process13. Additionally, the signatures may prove clinically useful in gauging patients’ treatment responses, planning further treatments, and providing prognostic information.
To date, several reports have investigated the effect of surgical procedures on gene expression patterns in regenerating rat livers after PH14, PVL15, and ALPPS when compared with controls10, as well as PVL vs. ALPPS10,16,17. However, to our knowledge, no study has explored the global gene expression patterns in regenerating livers after different regeneration-promoting surgical procedures and performed inter-procedural comparison. Using the power of next-generation RNA sequencing (RNAseq), we provided an overarching comparative analysis of differentially-regulated genes among three surgical groups (PH, PVL, ALPPS versus Sham) in regenerating rat livers, at early regenerating phase (i.e. 24 hours) and late-stage (i.e. 96 hours) post-operatively. Furthermore, we identified both unique and shared molecular mechanisms and signaling pathways for liver regeneration in the investigated surgical procedures.
Materials and Methods
Animals
Six to 8 weeks old Male Sprague–Dawley rats weighing 200–250 g were obtained from the Charles-River Laboratories UK Ltd. The animals were housed in an alternating light and dark room with controlled temperature and relative humidity (23 ± 2 °C, 50 ± 10%). They were given standard laboratory rodent chow and free water access, as detailed previously9. All the experiments were approved by the local ethics committee (University College London) and conducted according to Home Office guidelines under the UK Animals and Scientific Procedures Act 1986.
Experimental design and animal groups
Rats were divided into groups and anesthetized under isoflurane administration. A middle line incision was performed on the abdomen. (1) Sham group: Abdomen was closed after manipulation of the liver hilum; (2) PVL group: Portal vein ligation was performed on all branches except the branch to the right median lobe using a size 7–0 nylon thread; and (3) ALPPS group: selective portal vein ligation and liver parenchymal partitioning were performed (4) PH group: A 5–0 nylon thread was tied tightly around the median and left lateral lobes (~70% of the total liver mass) and both lobes were then resected, as described previously9. There were three rats for each group at each time point (24 h and 96 h). The detail of surgical operations and experimental grouping is provided in our previous study9.
RNA sequencing analysis
TRIzol (Invitrogen) was used for isolation of total RNA (15 µg) that was utilized for purifying the poly(A)-containing mRNA molecules, RNA amplification, and synthesis of double-stranded cDNAs according to Illumina’s TruSeq RNA Sample Prep guidelines (San Diego, CA). Multiplexed samples were sequenced at 43 bp length on Illumina-based Technology. Triplicate biological replicates were performed for each group. The paired-end reads of each sample were aligned to the rat genome (rn5) using the TopHat18. Transcript abundance was estimated by Cufflinks19. The quantification and normalization (DESeq method20) and further downstream analyses of identification of differentially expressed genes (DEGs) were done by using Strand NGS 2.7 (Strand Life Sciences, India) and PARTEK Genomics Suite (Partek Inc., St. Louis, MO, USA). Significantly regulated genes across different operation types (ALPPS, PVL, PH, and Sham) and two time points (24 h and 96 h) were determined using two-factor analysis of variance (ANOVA) by taking operation type, time and their interactions into the statistical model. Genes exhibiting false discovery rate (FDR)-adjusted P value<0.05 and the absolute fold changes (FC) > 2 were considered significant.
Functional pathway, upstream regulator, and network analyses
Functional, pathway, and gene ontology (GO) enrichment analysis were performed using Database for Annotation, Visualization and Integrated Discovery (DAVID)21, Protein Analysis Through Evolutionary Relationships (PANTHER™) classification systems and Ingenuity Pathways Analysis (IPA) (QIAGEN Inc., https://www.qiagenbioinformatics.com/products/ingenuity-pathway-analysis). We also performed upstream regulator, canonical pathways, and gene network analyses using IPA and Network Analyst22. The DEGs lists for each surgical procedure for different time points were mapped to its corresponding gene object in the Ingenuity pathway knowledge base and protein-protein interaction networks. A right-tailed Fisher’s exact test was used to calculate a p-value determining the probability that the biological function (or pathway) assigned to that data set is explained by chance alone. The IPA upstream regulator analysis predicts the upstream transcriptional regulators based on the Ingenuity® Knowledge Base by examining how many known targets of the upstream regulators are present in the differentially expressed gene list. An overlap p‐value, based on significant overlap between genes in the list and known targets regulated by the transcriptional regulator, and an activation z‐score are computed. The predicted activation state and activation z-score are based on the direction of fold change values that we observed in the gene expression data. The activation z‐score is to infer likely activation states, “activated” or “inhibited”, of upstream transcriptional regulators. It was considered significantly activated (or inhibited) with an overlap p-value ≤ 0.05 and an z-score ≥ 2.0 (or ≤ −2.0).
Results
Global gene expression changes in regenerating livers after ALPPS, PVL and PH
Global transcriptome changes associated with the surgical procedures at each time point were measured by RNAseq approach. The analysis generated 14,989,372 to 120,655,689 reads, which represented 88.5% of the genome (rn5), and 31,399 transcripts (Supplementary Table S1). There were 2,014 genes that showed a significant change in at least one surgical procedure (with respect to sham group) at either of the two time points (Fig. 1a). The variation in the data matrix was mostly due to the time effect, followed by the operation type (Fig. 1b). Principal components analyses (PCA) and unsupervised hierarchical clustering separated the samples according to two time points (24 h and 96 h) and surgical procedure types (Fig. 1c,d, respectively). Genes involved in cell cycle, mitosis, developmental process, and DNA replication were up-regulated in three surgical groups at 24 h with highest increase was observed in PH and ALPPS compared to the PVL group, which were later repressed at 96 h (Fig. 1d). Macrophage activation, cell adhesion, immune process, ion transport, and lipid metabolic process related genes had low level of expression at 24 h, but were up-regulated at 96 h post-operation (Fig. 1d).
(a) Venn diagrams representing the differentially expressed genes specific or common among ALPPS, PH and PVL at 24 h and 96 h, respectively. (b) Sources of variation in the data matrix. The x-axis shows the components of the 2-way ANOVA model and the y-axis shows the mean signal to noise ratio. (c) Unsupervised PCA analysis. Different colors indicate different surgical types and shapes indicate different time points. (d) Two-dimensional hierarchical clustering of genes, that are significant in at least one surgical procedure (with respect to sham group) at any of the two time points, and samples. The figure shows the most associated GO biological processes for each cluster of genes. Red and green denote highly and weakly expressed genes, respectively.
Inter-procedural similarities/differences at 24 h and 96 h post operations
Since the time had the greatest effect on the expression, we next compared the transcriptomes of the procedures at each time point separately. Differentially expressed genes (DEGs) that are specific or commonly dysregulated between the procedures at 24 h and 96 h were obtained using the Venn diagram approach (Fig. 1a). The PCA and hierarchical clustering analyses of the significant genes (FDR < 5% and FC > 2) separated the samples according to the surgical types at each time points (Supplementary Fig. S1).
At 24 h, the PH vs Sham produced the largest number of DEGs, followed by ALPPS vs Sham (Fig. 1a, Supplementary Table S2). There were 1242 DEGs for the PH (703↑ up-regulated, 539↓ down-regulated), 814 for the ALPPS group (536↑, 278↓), and 539 for the PVL (428↑, 111↓), and 411 DEGs (351↑, 60↓) were common to all surgical procedures. The arrow represents an increase (↑) or decrease (↓) in fold compared to Sham. Comparing different procedures, 123, 542, and 71 genes were specifically expressed in ALPPS, PH, and PVL, respectively (Fig. 1a). There were more DEGs commonly shared between the ALPPS and the PH than with the PVL, especially at 24 h. The list of DEGs shared and unique to each procedure is given in Supplementary Table S2. The ALPPS showed more similar expression pattern to the PH than the PVL especially at the early phase of the regeneration at 24 h post operation (Supplementary Fig. S1).
We further performed an extensive comparison of functional enrichment and gene networks for the three surgical procedures using different bioinformatics tools to further investigate molecular similarities/differences among the procedures. Gene ontology enrichment analysis revealed that genes related to cell cycle, mitotic cell cycle, M phase, and DNA replication and repair were significantly enriched among the up-regulated genes at 24 h for all surgical procedures (Supplementary Fig. S1). However, genes related to oxidation-reduction, triglyceride, and steroid metabolic processes were the most critically enriched among the down-regulated genes in ALPPS and PH only (Fig. 2a, Table 1, and Supplementary Fig. S4).
At 96 h post-operatively, there were 657 (493↑, 164↓), 448 (387↑, 61↓), and 211 (179↑, 32↓) genes for ALPPS, PH and PVL, respectively, when compared with the Sham livers (FDR < 5% and FC > 2) (Fig. 1a, Supplementary Table S3). The ALPPS vs Sham produced the largest number of DEGs. Genes responsible for cell cycle, ion transport, mitosis, and organelle fission seemed to be highly enriched in the up-regulated genes in the ALPPS; genes responsible for cell activation, response to wounding, and immune response seemed to be the most significantly enriched in the PH. The down-regulated genes at 96 h, especially for the ALPPS and PH, were enriched for oxidation-reduction, metabolic process and inflammatory response (Fig. 2b). The cell adhesion, immune system process, cellular process and cell proliferation associated genes were significantly enriched after ALPPS and PH, and macrophage activation, system development, and cellular component and morphogenesis were significant after all the procedures at 96 h (Table 1).
Common gene signatures of liver regeneration at 24 h and 96 h
Among the all surgical procedures, 411 DEGs (351↑, 60↓) and 105 DEGs (99↑, 6↓) were dysregulated at 24 h and 96 h, respectively (FDR < 5% and absolute FC > 2) (Supplementary Fig. S2A, Tables S2 and S3, respectively). GO enrichment and functional analyses of DEGs at 24 h indicated involvement of genes related to cell cycle, DNA replication and repair, cell division, mitosis, and DNA metabolic process (Table 1, Fig. 3, and Supplementary Fig. S2B). Furthermore, the gene network, functional and pathway analyses revealed potentially important roles of Ccnb1, Cdk1, E2f1, Ccna2, Rrm2, Lcn2, NF-κB, and Vegf in liver regeneration and alterations in cell cycle and p53 pathways (Fig. 3a–c and Supplementary Fig. S3).
Gene interaction network, functional, and canonical pathway analyses (a–c, respectively) at 24h and (d–f, respectively) at 96h of common DEGs among ALPPS, PH and PVL. Red and green denote up- and down-regulated genes, respectively, and grey indicates direct interactors of the DEGs. The sizes of nodes (in a) are proportional to their betweenness centrality values22. Straight and dashed lines (in d) represent direct or indirect gene to gene interactions, respectively. The color intensity is correlated with fold change. X-axis (in bar graphs) indicates the significance (–log10(p-value)) of the functional/pathway association. The threshold line represents a P value of 0.05.The network (in d) and functional/pathway analyses were generated using IPA (QIAGEN Inc., https://www.qiagenbioinformatics.com/products/ingenuity-pathway-analysis).
At 96 h, the DEGs that are commonly regulated in all surgical groups were mostly up-regulated (94%) and mainly involved in cellular movement, system development, extracellular matrix organization, and immune response (Supplementary Fig. S2B). The gene ontology and network analyses indicated enrichment of genes related to cell death and survival, cellular growth and proliferation, lipid metabolism, and immune cell trafficking, including genes such as LGALS3, FCGR2A, SPARC, Integrin, and Collagens (p-value <0.01) (Fig. 3d–f), at 96 h post-operations.
Functional, pathway and gene network comparison of temporal changes after ALPPS, PH and PVL
We further performed an extensive comparison of functional enrichment and gene networks for the three surgical procedures using different bioinformatics tools to further investigate molecular similarities/differences of the procedures as well as to investigate temporal changes within each surgical procedure at two time points.
Genes related to cell cycle, mitotic cell cycle, M phase, and DNA replication and repair were significantly enriched among the up-regulated genes at 24 h for all surgical procedures (Fig. 2a, Table 2, and Supplementary Fig. S4). However, genes related to oxidation-reduction, triglyceride, and steroid metabolic processes were the most critically enriched among the down-regulated genes in ALPPS and PH only. On the other hand, at 96 h, genes responsible for cell cycle and organelle fission seemed to be highly enriched in the up-regulated genes in the ALPPS; genes responsible for cell activation, response to wounding, and immune response seemed to be the most significantly enriched in the PH. The down-regulated genes at 96 h, especially for the ALPPS and PH, were enriched for oxidation-reduction, metabolic process and inflammatory response (Fig. 2b).
The DEGs for each procedure at each time point were mapped to gene interaction networks in order to obtain deeper insights into the interactions of these genes among various pathways. The functional, pathway and gene network analyses highlighted the potentially critical genes, biological processes, and signaling pathways for the temporal changes within ALLPS, PVL, and PH groups (Table 2, Supplementary Fig. S4). Cell cycle, mitosis, and DNA replication, process-related genes, such as Cdk1, E2f1, Ezh2, Ccnb1, Cdkn1a, Myc, and p53 pathways were significantly regulated at 24 h in all procedures. The networks for ALLPS and PH were more similar than PVL at early phase of the regeneration, corroborating with the clustering results in Fig. 1 and Supplementary Fig. S1. At 96 h, cell proliferation, cell cycle, mitosis and cell division remained active in the ALPPS when compared to the PH and PVL. Genes related to macrophage activation, anion transport, immune response, lipid metabolic process were also significantly regulated in the ALPPS (Table 1, Supplementary Fig. S4). Significantly altered pathways included the FXR/RXR activation, cell cycle and integrin signaling pathways in the ALPPS group at 96 h. On the other hand, cellular movement, immune cell trafficking, and signal transduction related genes and T-cell activation and Ras pathways were significantly altered in the PH group at 96 h. Top five significant molecular and cellular functions, canonical pathways, and predicted upstream regulators (activated/inhibited) for each surgical procedure at two time points are given in Table 2. Furthermore, the complete list of significant PANTHER and canonical pathways are given in Table 3 and Supplementary Table S4, respectively.
Temporal pattern of upstream transcriptional regulators after ALPPS, PH and PVL
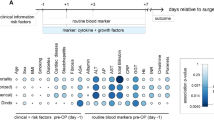
We identified the transcriptional regulators likely to be involved in the regenerating liver after each surgical procedure at two time points (Fig. 4). The IPA upstream regulator analytic identifies the upstream transcriptional regulators and mechanistic networks that can explain the gene expression changes observed after each procedure. There were 184, 158, and 206 upstream regulators predicted to be activated or inhibited after ALPPS, PH, and PVL, respectively at 24 hr (Fig. 4a). There was a noteworthy overlap of transcriptional regulators between different procedures at 24 h. The inter-procedural variations in ALPPS, PVL and PH are displayed at 24 h and 96 h (in Fig. 4b,c, respectively) for the top 15 activated upstream regulators. The union of top 15 activated upstream regulators in ALPPS, PVL and PH at 24 h and 96 h is used for the heatmap. Similarly, the temporal variations in each procedure are also investigated for ALPPS, PH and PVL, in Fig. 4d–f, respectively. Figure 4 also displays the heatmap of the top 50 activated/inhibited upstream regulators across all samples to investigate the temporal changes for the three procedures (complete list is given in Supplementary Table S5) (Fig. 4g).
The upstream regulator analyses. (a) Venn diagrams representing the overlap of predicted upstream regulators that are unique or common among ALPPS, PH and PVL at 24 h (top) and 96 h (bottom). (b,c) Inter-procedural variations at 24 h and 96 h, respectively. (d–f) Temporal variations after ALPPS, PH and PVL, respectively. Heatmaps for union of top 15 activated upstream regulators in ALPPS, PVL and PH at 24 h and 96 h are used. Color scale shows significance by –log10(p-value). (g) Heatmap that displays surgical procedure as well as temporal changes for the top 50 activated/inhibited upstream regulators. The upstream regulators are ordered according to significance in ALPPS procedure. The color-key is for the z-score. Data were analyzed through the use of IPA (QIAGEN Inc., https://www.qiagenbioinformatics.com/products/ingenuitypathway-analysis).
Activated regulators, such as E2F1, CCND1, TBX2, RABL6, MYC, EP400, Vegf, FOXM1, HGF, and IL6, and inhibited regulators, including CDKN1A, NUPR1, Rb, HNF4A, and TP53, may play important roles for early liver regeneration (Fig. 4). TGFB, KRAS, IL1, ERK1/2 appears to be activated in ALLPS and PH (Fig. 4g and Supplementary Table S5). On the other hand, at 96 h, CSF2, Cg, TGFB1, IFNG, IL5, and F2 were predicted to be activated in all procedures. FOXM1, PTGER2, E2F3, TBX2, CCND1, E2F1, and Notch were activated significantly in the ALPPS; TNF, TP53, STAT3, PDGF, and IL1A in the PH; JUN and POU5F1 in the PVL. Vegf, HGF, IL6, IL1B, NFKB, IFNA, and MITF were activated both in ALPPS and PH (Fig. 4 and complete list in Supplementary Table S5). There were 101 and 23 shared regulators among the procedures at 24 h and 96 h, respectively (Fig. 4a and Supplementary Table S5). The upstream regulator analysis also revealed several activated mechanistic networks at early and late-stage of liver regeneration, including E2F1, HGF and TP53 at 24 h and TGFB1, Vegf, and TP53 at 96 h (Fig. 5).
Discussion
We performed next-generation RNAseq approach to comprehensively delineate liver regeneration induced by ALPPS, PVL, and PH approaches at two time points (24 h and 96 h). Recently, we have replicated ALPPS in a rat model and presented its quick rejuvenation mode17. In this study, we identified pertinent common transcriptomic signatures at critical time points (i.e. early proliferation and late-proliferation phase) of liver regeneration and revealed the inter-procedural and temporal variations in gene expression patterns.
In response to injury, liver regeneration is achieved by the activation of otherwise functional, fully-differentiated hepatocytes as a result of the autocrine and paracrine signaling (e.g., cytokines and growth factors)12,17. Thus, major underlying changes in gene networks are expected to be seen at the cellular level. Depending on the initial injurious stimulus (or surgical procedure), such networks may have distinct elements as well as shared factors that are critical for regeneration. Therefore, delineation of the gene networks in liver regeneration is of particular interest. Moreover, understanding of signaling cascades and gene networks of the ALPPS, PVL and PH at 24 hours is vital as these may be critical factors for the initiation of regeneration or resulting in poor recovery, incomplete regeneration, and ultimately, liver failure.
Our data revealed that time-dependent factors appear to be the major source of variation in post-injury alterations of gene expression in liver regeneration. Early transcriptomic changes and the upstream regulators after all the procedures included cell cycle associated genes (E2F1, CCND1, FOXM1, TP53, and RB1), transcription factors (Myc, E2F1, TBX2, FOXM1)23,24,25,26,27,28, DNA replication regulators (CDKN1A, EZH2, RRM2)29, G1/S-transition regulators (CCNB1, CCND1, RABL6)30, cytokines and growth factors (CSF2, IL-6, TNF, HGF, VEGF, and EGF)9,16,31,32. At the cellular level, this corresponds to the transition from the quiescent G0 phase to active mitosis. Liver regeneration is governed by numerous growth factors, including HGF and EGF that are responsible priming the parenchymal and nonparenchymal liver cells and boosting their access into the cell cycle to proliferate to restore the original liver size after the surgical procedures9,16,31,32. Growth factors and signaling pathways activates cyclin-dependent kinases (CDKs), and upregulates the expression of CCND1 that encodes cyclin D1. CDK1 is essential for DNA replication and downstream formation of replication-initiation complexes in hepatocytes and shown to play a critical role in DNA replication control during rat liver regeneration following PH33. It is also required for the activity of CCNB1, a protein from cyclin regulatory proteins family that is essential for cell cycle control during G2/M (mitosis) transition.
Upstream events that can induce the quiescence-to-mitosis transition may potentially be important in liver regeneration, such as activation of E2F1, CCND1, FOXM1, and inhibition of TP53, and RB1. Notably, enhancer of zeste homolog 2 (EZH2) was among the early significantly up-regulated genes. As the functional subunit of Polycomb Repressive Complex 2 (PRC2), which normally regulates development and differentiation in healthy embryonic tissue, it is potentially involved in early de-differentiation that hepatocytes undergo to become highly proliferative34. Additionally, its up-regulation is consistent with the decreased lipid metabolism identified by our functional network analysis35. As a specialized function of hepatocytes, the decrease in lipid metabolism may well be an indication of dedifferentiation. Besides the critical inducers of dedifferentiation and G0-to-G1 transition, other molecules seem to be critical in sustaining a robust intracellular environment. One example is p400 E1A-associated protein (EP400), which was predicted to be an early upstream regulator. EP400 is essential for the control of reactive oxygen species (ROS) intracellularly, maintaining an oxidative-stress free environment without which DNA damage, senescence, and apoptosis may ensue36. Another example is Lipocalin 2 (LCN2), an innate-immunity molecule with iron-sequestering properties. It has a wide expression in various tissues but has been particularly used as an early biomarker for kidney injury37. Recently, the increased expression level of LCN2 has been demonstrated in acute liver injury as well38,39,40. Indeed, protein and mRNA levels of Lcn2 is significantly increased after partial hepatectomy41.
Immune mediators of liver regeneration that showed a significant increase in expression immediately after the operations were IL-6 and VEGF17,42,43,44. These molecules are known mediators of acute and chronic inflammatory response, carrying out essential functions in repairing tissue injury in all parts of the human body. IL-6, secreted by macrophages, is a known pyrogen and a potent stimulator of the acute phase reactants following infection or trauma. VEGF is secreted by endothelial cells to promote vasculogenesis and angiogenesis. In liver injury, VEGF mediates the proliferation and mobilization of liver sinusoidal endothelial cell progenitor cells form the bone marrow to allow for the formation of new sinusoids in the regenerating, highly-vascular liver tissue45.
The network analysis of up-regulated genes at 96 h post-operatively revealed an expression profile predominantly in mediating tissue reconstruction, including cellular movement, system development, extracellular matrix organization and restoring specialized hepatocytic functions (e.g., carbohydrate transport and morphogenesis). Of note, this phase seems to be orchestrated by the macrophages (known as Kuppfer cells in the liver). Transforming growth factor-β (TGF-β), a potent inhibitor of inflammation and a stimulator of healing and tissue repair that is secreted by macrophages, was significantly up-regulated at 96 h. Furthermore, the analysis also highlighted genes such as LGALS3, FCGR2A, SPARC, integrin, and collagens, that may play significant roles in tissue regeneration and repair46,47,48. These genes closely coordinate their function with TGF-β in tissue organization, matrix structure and cell-to-cell interactions. The knockout mice of secreted protein acidic and rich in cysteine (SPARC) showed a reduction of expression of TGF-β1 and collagen in hepatic tissue49.
Among the three examined procedures, as noted in the clustering and upstream regular analyses, ALPPS and PH shared many significantly regulated genes whose expression were not otherwise significantly changed in PVL, especially at the early phase of regeneration. One possible explanation lies in the differences of the type of injury caused by each procedure. Specifically, ALPPS and PH both require the removal of hepatic parenchyma. On the other hand, the main stimulant of regeneration in PVL is ischemia and oxygen deprivation. Clinically, PVL is the least invasive and has the lowest rate of morbidity and mortality but also has a slower rate of regeneration than the other procedures50. Compared to PVL, the upstream regulator analysis revealed the activation of TGF-β, KRAS, ERK1/2, IL1, and INS at early phase and Vegf, HGF, Interferan alpha, IL6, IL1B, and NFKB at the later regeneration phase after ALPPS and PH. On the other hand, compared to PH and PVL, the cell cycle associated genes, cytokines, and transcription regulators, such as E2F1, TBX2, FOXM1, and EP400, are still activated after ALPPS at later regeneration phase. Finally, the Notch signaling, which is a complex signaling pathway that is crucial for the development of multiple organs, was also seemed to be activated in ALPPS. In the liver, it controls the hepatic cell differentiation into, and the formation of, the biliary system51. In addition, notch proteins have an extracellular membrane component with epidermal growth-like factor (EGF) repeats, which may be responsible for the high growth rate seen clinically in ALPPS52.
The upstream analyses also indicated the predicted inhibition of NUPR1, CDKN2A, Rb, PAX6, and TP53, at initial phase of liver regeneration53,54. However, at the later stage of liver regeneration, TP53 is activated, especially after PH. Furthermore, the mechanistic network of upstream regulators and their predicted relationship based on the observed gene expression changes in our data reveals the working mechanism of TP53 at early and late stage of the liver regeneration, as demonstrated in Fig. 5. p53 regulates liver homeostasis, and initiation of cell proliferation through proliferative signaling and disruption of p53 signaling lead to faster recovery53,54.
The limitation of the study is that we examined the liver proliferation at two time points, early phase (i.e. 24 h) and late-stage (i.e. 96 h). Our earlier study of ALPPS and PVL indicated higher proliferation index (PI) at 24 h and 48 hr comparing to 96 h, and there was no significant difference between the two time-points (i.e. 24 h vs. 48 h)17. The future remnant liver volume (FRLV) ratio was significantly higher in ALPPS comparing to PVL at 24 hr and 96 hr time points, but not significantly different at 48 h or 1 week. Higher FRL ratio is critical factor in improving surgical outcomes and liver regeneration. In addition, the ALPPS model has significantly more inflammatory cells infiltration at 24 hr comparing to 48 hr; higher infiltration of inflammatory might promoted earlier liver regeneration in the ALPPS model. It also indicated higher portal pressure in ALPPS group at 24 hr comparing to other time points; having higher portal pressure might act as physical stressor that contribute to ignite the regeneration process. Xu et al. studied the expressed genes in regenerating rat liver after PH, also reported that temporal patterns of gene expression were similar at 48 h and 96 h after PH55. Nevertheless, more time points should worth to be assessed to fully investigate the liver proliferation process. While recognizing this limitation, we believe we have largely achieved our aim, as the major objective of the study is to fish out early phase (i.e. 24 h) and late-stage (i.e. 96 h) liver regeneration molecular markers and examining molecular differences between these phases in the ALPSS, PVL and PH models that have provided unique molecular signatures.
In summary, our study presents a comprehensive transcriptomic profiling of three surgical procedures that are commonly used in clinical practice and identified the inter-procedural and temporal variations in gene expression patterns in each surgical procedure. Identification of molecular signatures and signaling pathways specific to each surgical procedure further our understanding of key regulators of liver regeneration as well as patient populations that are likely to benefit from each procedure.
Data availability
The datasets generated and analyzed during the current study is available at this link (https://www.dropbox.com/s/hgewpv07qce6fed/RNASeqTopHataligned_Normdata_sample29.txt?dl=0).
Change history
05 May 2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Kimura, N. et al. Gene expression of ATP-binding cassette transporters during liver regeneration after 90% hepatectomy in rats. International journal of molecular medicine 30, 28–34, https://doi.org/10.3892/ijmm.2012.972 (2012).
Fukuhara, Y. et al. Gene expression profile in the regenerating rat liver after partial hepatectomy. Journal of hepatology 38, 784–792 (2003).
Colak, D. et al. Integrative and comparative genomics analysis of early hepatocellular carcinoma differentiated from liver regeneration in young and old. Mol Cancer 9, 146, https://doi.org/10.1186/1476-4598-9-146 (2010).
Lu, X. et al. Integrated analysis of microRNA and mRNA expression profiles highlights the complex and dynamic behavior of toosendanin-induced liver injury in mice. Sci Rep 6, 34225, https://doi.org/10.1038/srep34225 (2016).
Chen, X. G., Xu, C. S. & Liu, Y. M. Involvement of ERK1/2 signaling in proliferation of eight liver cell types during hepatic regeneration in rats. Genetics and molecular research: GMR 12, 665–677, https://doi.org/10.4238/2013.March.11.14 (2013).
Hadden, W. J. et al. Resection of colorectal liver metastases and extra-hepatic disease: a systematic review and proportional meta-analysis of survival outcomes. HPB: The Official Journal of the International Hepato Pancreato Biliary Association 18, 209–220, https://doi.org/10.1016/j.hpb.2015.12.004 (2016).
Schnitzbauer, A. A. et al. Right portal vein ligation combined with in situ splitting induces rapid left lateral liver lobe hypertrophy enabling 2-staged extended right hepatic resection in small-for-size settings. Annals of surgery 255, 405–414, https://doi.org/10.1097/SLA.0b013e31824856f5 (2012).
Locker, J. et al. A common set of immediate-early response genes in liver regeneration and hyperplasia. Hepatology (Baltimore, Md.) 38, 314–325, https://doi.org/10.1053/jhep.2003.50299 (2003).
Dhar, D. K., Mohammad, G. H., Vyas, S., Broering, D. C. & Malago, M. A novel rat model of liver regeneration: possible role of cytokine induced neutrophil chemoattractant-1 in augmented liver regeneration. Annals of Surgical Innovation and Research 9, 1–10, https://doi.org/10.1186/s13022-015-0020-3 (2015).
Schlegel, A. et al. ALPPS: from human to mice highlighting accelerated and novel mechanisms of liver regeneration. Annals of surgery 260, 839–846; discussion 846–837, https://doi.org/10.1097/sla.0000000000000949 (2014).
Blanpain, C. & Fuchs, E. Plasticity of epithelial stem cells in tissue regeneration. Science 344, https://doi.org/10.1126/science.1242281 (2014).
Font-Burgada, J. et al. Hybrid Periportal Hepatocytes Regenerate the Injured Liver without Giving Rise to Cancer. Cell 162, 766–779, https://doi.org/10.1016/j.cell.2015.07.026 (2015).
Bhate, A. et al. ESRP2 controls an adult splicing programme in hepatocytes to support postnatal liver maturation. Nature communications 6, 8768, https://doi.org/10.1038/ncomms9768 (2015).
Xu, C. S. et al. Gene expression differences of regenerating rat liver in a short interval successive partial hepatectomy. World journal of gastroenterology 10, 2680–2689 (2004).
Nagano, Y. et al. Gene expression profile analysis of regenerating liver after portal vein ligation in rats by a cDNA microarray system. Liver international: official journal of the International Association for the Study of the Liver 24, 253–258, https://doi.org/10.1111/j.1478-3231.2004.0912.x (2004).
Garcia-Perez, R. et al. Associated Liver Partition and Portal Vein Ligation (ALPPS) vs Selective Portal Vein Ligation (PVL) for Staged Hepatectomy in a Rat Model. Similar Regenerative Response? PloS one 10, e0144096, https://doi.org/10.1371/journal.pone.0144096 (2015).
Dhar, D. K., Mohammad, G. H., Vyas, S., Broering, D. C. & Malago, M. A novel rat model of liver regeneration: possible role of cytokine induced neutrophil chemoattractant-1 in augmented liver regeneration. Annals of surgical innovation and research 9, 11, https://doi.org/10.1186/s13022-015-0020-3 (2015).
Langmead, B., Trapnell, C., Pop, M. & Salzberg, S. L. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome biology 10, R25, https://doi.org/10.1186/gb-2009-10-3-r25 (2009).
Trapnell, C. et al. Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nature protocols 7, 562–578, https://doi.org/10.1038/nprot.2012.016 (2012).
Anders, S. & Huber, W. Differential expression analysis for sequence count data. Genome biology 11, R106, https://doi.org/10.1186/gb-2010-11-10-r106 (2010).
Dennis, G. Jr. et al. DAVID: Database for Annotation, Visualization, and Integrated Discovery. Genome biology 4, P3 (2003).
Xia, J., Gill, E. E. & Hancock, R. E. NetworkAnalyst for statistical, visual and network-based meta-analysis of gene expression data. Nature protocols 10, 823–844, https://doi.org/10.1038/nprot.2015.052 (2015).
Farra, R. et al. Impairment of the Pin1/E2F1 axis in the anti-proliferative effect of bortezomib in hepatocellular carcinoma cells. Biochimie 112, 85–95, https://doi.org/10.1016/j.biochi.2015.02.015 (2015).
Ella, E. et al. Specific genomic and transcriptomic aberrations in tumors induced by partial hepatectomy of a chronically inflamed murine liver. Oncotarget 5, 10318–10331, https://doi.org/10.18632/oncotarget.2515 (2014).
Wang, H. B. et al. Myc and ChREBP transcription factors cooperatively regulate normal and neoplastic hepatocyte proliferation in mice. J Biol Chem 293, 14740–14757, https://doi.org/10.1074/jbc.RA118.004099 (2018).
Berthet, B., DiCostanzo, J., Di Costanzo, V., Frigerio, J. M. & Dagorn, J. C. Expression of genes associated with liver regeneration following partial hepatectomy in the rat. Influence of Epidermal Growth Factor and Interleukin 6. Ann Gastroent Hepato 34, 201–210 (1998).
Yin, L., Wang, Y. H., Guo, X. Q., Xu, C. S. & Yu, G. Y. Comparison of gene expression in liver regeneration and hepatocellular carcinoma formation. Cancer Manag Res 10, 5691–5708, https://doi.org/10.2147/Cmar.S172945 (2018).
Chishti, M. A. et al. Induction of cell proliferation in old rat liver can reset certain gene expression levels characteristic of old liver to those associated with young liver. Age 35, 719–732, https://doi.org/10.1007/s11357-012-9404-z (2013).
Yin, L., Wang, Y. Y., Lin, Y. Z., Yu, G. Y. & Xia, Q. F. Explorative analysis of the gene expression profile during liver regeneration of mouse: a microarray-based study. Artif Cell Nanomed B 47, 1113–1121, https://doi.org/10.1080/21691401.2019.1593851 (2019).
Shizu, R. et al. PXR stimulates growth factor-mediated hepatocyte proliferation by cross-talk with the FOXO transcription factor. Biochem J 473, 257–266, https://doi.org/10.1042/Bj20150734 (2016).
Shi, H. et al. A preliminary study of ALPPS procedure in a rat model. Scientific reports 5, 17567, https://doi.org/10.1038/srep17567 (2015).
Yao, L. B. et al. In situ splitting after selective partial portal vein ligation or simultaneous hepatic artery ligation promotes liver regeneration. Sci Rep 8, 8699, https://doi.org/10.1038/s41598-018-26742-5 (2018).
Garnier, D., Loyer, P., Ribault, C., Guguen-Guillouzo, C. & Corlu, A. Cyclin-dependent kinase 1 plays a critical role in DNA replication control during rat liver regeneration. Hepatology (Baltimore, Md.) 50, 1946–1956, https://doi.org/10.1002/hep.23225 (2009).
Bae, W. K. et al. The methyltransferases enhancer of zeste homolog (EZH) 1 and EZH2 control hepatocyte homeostasis and regeneration. FASEB journal: official publication of the Federation of American Societies for Experimental Biology 29, 1653–1662, https://doi.org/10.1096/fj.14-261537 (2015).
Vella, S. et al. EZH2 down-regulation exacerbates lipid accumulation and inflammation in in vitro and in vivo NAFLD. International journal of molecular sciences 14, 24154–24168, https://doi.org/10.3390/ijms141224154 (2013).
Mattera, L. et al. The E1A-associated p400 protein modulates cell fate decisions by the regulation of ROS homeostasis. PLoS genetics 6, e1000983, https://doi.org/10.1371/journal.pgen.1000983 (2010).
Bolignano, D. et al. Neutrophil gelatinase-associated lipocalin (NGAL) as a marker of kidney damage. American journal of kidney diseases: the official journal of the National Kidney Foundation 52, 595–605, https://doi.org/10.1053/j.ajkd.2008.01.020 (2008).
Borkham-Kamphorst, E. et al. Protective effects of lipocalin-2 (LCN2) in acute liver injury suggest a novel function in liver homeostasis. Biochimica et biophysica acta 1832, 660–673, https://doi.org/10.1016/j.bbadis.2013.01.014 (2013).
Cheng, Q. et al. The roles of lipocalin-2 in small-for-size fatty liver graft injury. Annals of surgery 260, 1062–1072, https://doi.org/10.1097/SLA.0000000000000427 (2014).
Roudkenar, M. H. et al. Gene expression profiles in mouse liver cells after exposure to different types of radiation. J Radiat Res 49, 29–40 (2008).
Lai, H. S., Wu, Y. M., Lai, S. L. & Lin, W. H. Lipocalin-2 gene expression during liver regeneration after partial hepatectomy in rats. International journal of surgery 11, 314–318, https://doi.org/10.1016/j.ijsu.2013.02.008 (2013).
Chinnici, C. M. et al. Mesenchymal stromal cells isolated from human fetal liver release soluble factors with a potential role in liver tissue repair. Differentiation 105, 14–26, https://doi.org/10.1016/j.diff.2018.12.001 (2019).
Gu, K. et al. Hepatic Regeneration after Sublethal Partial Liver Irradiation in Cirrhotic Rats. J Radiat Res 52, 582–591, https://doi.org/10.1269/jrr.11002 (2011).
Jia, C. K. Advances in the regulation of liver regeneration. Expert Rev Gastroent 5, 105–121, https://doi.org/10.1586/Egh.10.87 (2011).
Wang, L. et al. Hepatic vascular endothelial growth factor regulates recruitment of rat liver sinusoidal endothelial cell progenitor cells. Gastroenterology 143, 1555–1563.e1552, https://doi.org/10.1053/j.gastro.2012.08.008 (2012).
Li, X. W. et al. Proteomics analysis of plasma membrane from liver sinusoidal endothelial cells after partial hepatectomy by an improved two-dimensional electrophoresis. Mol Cell Biochem 344, 137–150, https://doi.org/10.1007/s11010-010-0537-z (2010).
Issa, R. et al. Mutation in collagen-I that confers resistance to the action of collagenase results in failure of recovery from CCl4-induced liver fibrosis, persistence of activated hepatic stellate cells, and diminished hepatocyte regeneration. Faseb Journal 16, 47−+, https://doi.org/10.1096/fj.02-0494fje (2002).
Li, J. et al. Human Hepatic Progenitor Cells Express Hematopoietic Cell Markers CD45 and CD109. Int J Med Sci 11, 65–79, https://doi.org/10.7150/ijms.7426 (2014).
Atorrasagasti, C. et al. Lack of the Matricellular Protein SPARC (Secreted Protein, Acidic and Rich in Cysteine) Attenuates Liver Fibrogenesis in Mice. PloS one 8, e54962, https://doi.org/10.1371/journal.pone.0054962 (2013).
Treska, V. Methods to Increase Future Liver Remnant Volume in Patients with Primarily Unresectable Colorectal Liver Metastases: Current State and Future Perspectives. Anticancer research 36, 2065–2071 (2016).
Morell, C. M. & Strazzabosco, M. Notch signaling and new therapeutic options in liver disease. Journal of hepatology 60, 885–890, https://doi.org/10.1016/j.jhep.2013.11.028 (2014).
Kopan, R. & Ilagan, M. X. G. The Canonical Notch Signaling Pathway: Unfolding the Activation Mechanism. Cell 137, 216–233, https://doi.org/10.1016/j.cell.2009.03.045.
Krstic, J., Galhuber, M., Schulz, T. J., Schupp, M. & Prokesch, A. p53 as a Dichotomous Regulator of Liver Disease: The Dose Makes the Medicine. International journal of molecular sciences 19, https://doi.org/10.3390/ijms19030921 (2018).
Borude, P. et al. Pleiotropic Role of p53 in Injury and Liver Regeneration after Acetaminophen Overdose. The American journal of pathology 188, 1406–1418, https://doi.org/10.1016/j.ajpath.2018.03.006 (2018).
Xu, C. S. et al. Expressed genes in regenerating rat liver after partial hepatectomy. World journal of gastroenterology 11, 2932–2940, https://doi.org/10.3748/wjg.v11.i19.2932 (2005).
Acknowledgements
Authors would like to thank King Faisal Specialist Hospital and Research Centre (KFSH&RC), Research Advisory Council (grant numbers 2110006 and 2180030 to Dilek Colak). We would like to thank Dr. Namik Kaya, Genetics Department, KFSH&RC for much helpful discussion and valuable remarks. We also would like to thank Mr. Ibrahim Hamza Kaya, Alfaisal University, College of Medicine for editing the manuscript and Ms. Sukina Qanbar for administrative assistance.
Author information
Authors and Affiliations
Contributions
Conceptualization (D.K.D., D.C. and D.C.B.); Performed experiments: D.K.D.; Analysed data: D.C., O.A.; Editing and critical reading of manuscript: F.M., A.M.A. and D.C.B.; Prepared manuscript: D.C., O.M.M. and O.A.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Colak, D., Al-Harazi, O., Mustafa, O.M. et al. RNA-Seq transcriptome profiling in three liver regeneration models in rats: comparative analysis of partial hepatectomy, ALLPS, and PVL. Sci Rep 10, 5213 (2020). https://doi.org/10.1038/s41598-020-61826-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-61826-1
This article is cited by
-
Effects of Sodium Chromate Exposure on Gene Expression Profiles of Primary Rat Hepatocytes (In Vitro)
Biological Trace Element Research (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.