Abstract
In achondroplasia, lumbar spinal stenosis arises from congenital dysplasia and acquired degenerative changes. We here aimed to describe the changes of the lumbar spinal canal and intervertebral disc in adults. We included 18 adults (age ≥ 18 years) with achondroplasia and lumbar spinal stenosis. Radiographs were used to analyze spinal-pelvic angles. Antero-posterior diameter of the spinal canal and the grade of disc degeneration were measured by MRI. Antero-posterior diameters of the spinal canal differed by spinal level (P < 0.05), with lower values observed at T12-L1, L1-2 and L2-3. Degrees of disc degeneration differed by intervertebral level, with higher degrees observed at L1-2, L2-3 and L3-4. A significant correlation was found between disc degeneration and thoraco-lumbar kyphosis at L2-3, between antero-posterior diameter of the spinal canal and lumbar lordosis at T12-L1 and L2-3, and between antero-posterior diameter of the spinal canal and thoraco-lumbar kyphosis at L1-2. Unlike the general population, spinal stenosis and disc degeneration involve the upper part of the lumbar spine in adults with achondroplasia, associated with thoraco-lumbar kyphosis and loss of lumbar lordosis.
Similar content being viewed by others
Introduction
Achondroplasia is the most common form of skeletal dysplasia with micromelia, with an estimated prevalence is 1/16,000–25,000 live births1,2. The disease is due to a gain-of-function mutation in the fibroblast growth factor receptor 3, that affects growth through chondrocyte dysfunction and cartilage dysplasia3,4. The clinical consequence is a disproportionate short stature with short limbs and cranial and spine abnormalities1,2. Spinal disorders are major contributors to disability in adults with achondroplasia5. They include cervico-medullary compression and spinal stenosis, this being the most prevalent spinal feature of this condition. Approximatively 40% of adults with achondroplasia have chronic back problems. Half undergo surgery because of lumbar spinal stenosis6.
The clinical expression of lumbar spinal stenosis ranges from low back pain with neurogenic claudication to paraplegia with bladder and bowel dysfunction7. Achondroplasia-associated lumbar spinal stenosis results from a combination of congenital dysplasia and acquired degenerative changes8. Spinal dysplasia in achondroplasia is due to disordered endochondral ossification resulting in early fusion of the pedicles to the vertebral body9. Hence the cross-sectional area of the spinal canal is narrowed by the shortened pedicles and the decreased interpedicular distance. The dimensions of the spinal canal are further reduced by age-related degenerative changes, which eventually compromise the space available for the neural elements.
Lumbar spinal stenosis is also found in individuals without dysplasia10,11,12. The occurrence of the stenosis increases with aging. Acquired degenerative changes involved in lumbar spinal stenosis have largely been described in general populations, but less so in adults with achondroplasia8,13. The degenerative process of lumbar spinal stenosis leads to disc narrowing and bulging, infolding and thickening of the ligamentum flavum as well as facet osteoarthritis10,11,12. With advanced alterations, disc, ligamentum flavum, and facet joints protrude into the spinal canal. Increased stresses associated with disc degeneration and narrowing in turn induce in turn facet-joint and ligamentum-flavum degeneration. Thus degenerative disc changes may be a critical step in the cascade of events leading to lumbar spinal stenosis with its clinical manifestations.
Degenerative changes of the intervertebral disc have not been characterized in adults with achondroplasia. The identification of current changes is important to assess the long term impact of future therapies. Therefore, we conducted a retrospective study of achondroplasia patients with lumbar spinal stenosis in a single center to characterize degenerative changes of the intervertebral disc in this population and to identify factors associated with the process.
Results
Patient characteristics and measures
Eighteen adults with achondroplasia were referred to the reference center for constitutional bone diseases between January 2004 and February 2017, fulfilled the inclusion criteria and were included in the study. All had back and leg symptoms attributed to lumbar spinal stenosis. Clinical characteristics of included patients are in Table 1. Two patients had previously undergone lumbar laminectomy, with five (T12-L5) and three (L2-L5) operated levels. Each of the eight operated levels was excluded from the imaging analysis of antero-posterior diameter and disc degeneration. A total of 14 patients including one with previous spinal surgery, underwent spine radiographies and spino-pelvic angle assessment. Reliability of measurements was good to very good. Intra- and inter-observer ICCs were 0.93 (95% CI 0.86–0.96) and 0.92 (0.84 0.96) respectively for measuring antero-posterior spinal canal diameter. Intra- and inter-reliability kappa values were 0.87 (95%CI 0.73–1) and 0.78 (0.61–0.95) for measuring disc degeneration with the Pfirmann grading system.
Spinal stenosis and disc alterations affect mainly the upper lumbar spine
Considering the study population, antero-posterior diameters of the spinal canal differed by vertebral or intervertebral disc level (Fig. 1), with lower values observed at T12-L1, L1-2 and L2-3 intervertebral levels. Lumbar spinal stenosis was maximal at L2-3. In addition, degrees of disc degeneration differed by intervertebral disc level (Tables 2 and 3), with higher degrees observed at L1-2, L2-3 and L3-4. The most advanced degenerative changes were again observed at L2-3. The involvement of the upper lumbar spine is illustrated in Fig. 2.
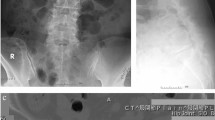
Representative lumbar spine imaging in adults with achondroplasia. (a) X-ray of a 58 year-old women revealing a thoraco-lumbar kyphosis. (b) T2-weighted MRI of a 25 year-old women showing an involvement of the upper part of the lumbar spine including T12-L2 antero-posterior stenoses (thick arrows) and T12-L4 disc degeneration (dark discs, dashed arrows).
Correlation analysis of disc degeneration and clinical parameters indicated a significant correlation with only age at the L2-3 (rs=0.50), L3-4 (rs=0.58), L4-5 (rs=0.78) and L5-S1 (rs=0.69, p < 0.05), and with height (rs = −0.55, p < 0.05) at L4-5. Correlation analysis of antero-posterior diameter of the spinal canal and clinical parameters indicated no significant correlation from T12-L1 to L5-S1.
For the radiographic spino-pelvic angles investigated, we found a significant correlation between disc degeneration and thoraco-lumbar kyphosis at L2-3 (Table 4), between antero-posterior diameter of the spinal canal and lumbar lordosis at T12-L1 and L2-L3, and between antero-posterior diameter of the spinal canal and thoraco-lumbar kyphosis at L1-2 level (Table 5).
We also did a secondary analysis excluding the patient with previous spinal surgery. All correlations persisted, with P value <0.05 for correlation between disc degeneration and thoraco-lumbar kyphosis at L2-3 as for correlation between antero-posterior diameter of the spinal canal and lumbar lordosis at T12-L1 and L2-L3; and with P value = 0.05 for correlation between antero-posterior diameter of the spinal canal and thoraco-lumbar kyphosis at L1-2 level.
Discussion
Herein, we describe degenerative changes of the intervertebral disc in adults with achondroplasia. In this single-center series, spinal stenosis and disc degeneration mainly involved the upper part of the lumbar spine, with maximal changes at L2-3. The degree of spinal stenosis was not correlated with age, sex, height, weight or BMI. Disc degeneration was correlated with age and height. However, this relationship was more pronounced at lower lumbar intervertebral disc levels. In contrast to clinical factors, radiographic spino-pelvic angles were correlated with spinal canal diameters and disc changes at the upper, but not lower part of the lumbar spine. Thoraco-lumbar kyphosis was positively correlated with L2-3 disc degeneration, and both thoraco-lumbar kyphosis and loss of lumbar lordosis were correlated with spinal stenosis from T12-L1 to L2-3.
Distribution of spinal stenosis among intervertebral thoraco-lumbar levels in our achondroplasia cohort differs from the preferential lower lumbar-spine location in the general population, with maximal shrinking at L4-5 level14. Preferential involvement of stenosis in the upper lumbar spine in achondroplasia was previously reported8,15. The lowest values of the antero-posterior canal diameter were found at L2-38. Furthermore, surgery for lumbar spinal stenosis in achondroplasia concerns L1-2, L2-3 and L3-4 in more than 50% of cases and the L2-3 in as much as 97% of cases16. Location of the highest degrees of disc degeneration at L1-2, L2-3 and L3-4 in our patients also contrasts with the highest prevalence at the lower lumbar level, especially L4-5, in the general population17. To our knowledge, the distribution of disc degeneration in adults with achondroplasia has not previously been reported.
To our knowledge, correlations between spinal stenosis or disc degeneration, and age, sex, weight, height or BMI have not been investigated in achondroplasia patients. We found a correlation between disc degeneration and age at L2-3, L3-4, L4-5 and height at L4-5. These findings agree with age-associated disc degeneration features in the general population. However, we found no association with other risk factors of disc degeneration identified in the general population, such as body weight and BMI18,19. Height was not related to disc degeneration in the general population. The correlation between disc degeneration and height in our patients should be viewed with caution. Its reasons appear difficult to describe. Conversely to age and disc degeneration, it involves L4-5 only and not higher levels which are more affected by disc degeneration; and no such association has previously been found in general population. The correlations between MRI findings and radiographic spino-pelvic parameters involved the upper part of the lumbar spine only and therefore are likely to be directly related to lumbar spinal stenosis. Even if not all statistically significant for each intervertebral disc level in the area of interest, our results suggest that the spino-pelvic angles, which characterize developmental spinal dysplasia, may have a role in the pathogenesis of disc degeneration and spinal stenosis at the upper lumbar spine in adults with achondroplasia. The results support the hypothesis that thoraco-lumbar kypkosis is a factor of disc degeneration and spinal stenosis. Arguments for this hypothesis are as follows: thoraco-lumbar kyphosis is associated with disc degeneration and spinal stenosis in our patients; previous biomechanical data in human suggest that kyphosis increases forces acting on discs which can contribute to disc degeneration and spinal stenosis20; clinical manifestations usually attributed to disc degeneration or spinal stenosis have been reported to be associated with thoraco-lumbar kyphosis in adults with achondroplasia21,22. We also found loss of lumbar lordosis related to thoraco-lumbar spinal stenosis. Loss of lumbar lordosis was previously found associated with pain in adults with achondroplasia14. However our results involving lumbar lordosis should be cautiously considered. Indeed, we assessed thoraco-lumbar kyphosis from T10 to L2, and lumbar lordosis from L1 to S1. Due to the overlap of the measure, the values of the thoraco-lumbar kyphosis may have influenced the correlations involving lumbar lordosis. Further investigations are therefore required to separately assess lordosis in upper and in lower levels of the lumbar spine. They may also be useful to clarify the association of thoraco-lumbar kyphosis with disc degeneration and spinal stenosis.
Our study provides some insight into associations among disc degeneration, lumbar spinal stenosis and radiographic spino-pelvic parameters in achondroplasia. However, it is limited by its small sample size, retrospective design, no quantitative assessment of pain and functional limitation, no pain-free control, and no follow-up. Furthermore, the radiographic spino-pelvic analysis did not involve the full spine. Data on lumbar spine in adults with achondroplasia are sparse and analysis of 18 patients is of interest in this context. The small sample we used was due to the rare disease and close to that of others8.
In conclusion, we describe lumbar spinal stenosis and disc degeneration in adults with achondroplasia. Disc degeneration was not previously described in this population. Our results suggest that spinal stenosis and disc degeneration are located at the upper part of the lumbar spine, with maximal changes at L2-L3. We also suggest their association with thoraco-lumbar kyphosis and loss of lumbar lordosis. Further investigations remain necessary to reinforce our results and hypotheses, and for better understanding of the condition.
Patients and methods
The study was a retrospective case series. Due to the rare disease, the sample size was not planned, but convenience sample. Adults (age ≥ 18 years) with achondroplasia referred to the reference center of constitutional bone diseases between January 2004 and February 2017 were considered for inclusion. Patients with available MR images of the lumbar spine were included in the study. The study involved review of clinical information by the treating clinicians. It does not require ethics approval. No patient expressed any opposition to the study.
Clinical and imaging parameters
Back and leg symptoms, age, sex, height and body mass index (BMI) were recorded from medical charts. Radiographs of the lumbar spine, thoraco-lumbar junction, and hip joints in standing position, were analyzed. Spino-pelvic angles measured on these radiographs included the sacral slope (SS), pelvic tilt (PT), pelvic incidence (PI), lumbar lordosis (LL) and thoraco-lombar kyphosis (TLK)21. The SS corresponded to the angle between the sacral endplate and the horizontal plane; the PT, the angle between the line joining the middle of the sacral endplate and the hip axis, and the vertical plane; the PI, the angle between a line perpendicular to the sacral endplate and a line joining the middle of the sacral plate and the hip axis; the LL, the angle between the upper endplate of L1 and the upper endplate of S1; and the TLK, the angle between the upper endplate of T10 and the lower endplate L2.
T1-weighted and T2-weighetd sequences MR imaging sequences of the lumbar spine were obtained with a 1.5-T MR system. The antero-posterior diameters of the spinal canal were measured on mid-sagittal T2-weighted MR images. The antero-posterior diameter of the spinal canal was measured at each mid-height of the disc space from T12 to S1, and at each mid-vertebral level from T12 to L5. The anterior and posterior margins of the thecal sac were used as anterior and posterior landmarks of the spinal canal8. In case of previous lumbar spinal surgery, operated levels were excluded from the analyses. Measurements on radiographs and MR images involved using a Vue PACS workstation (v11.3, Carestream Health, Rochester, NY).
Mid-sagittal T2-weighted MR images were used for grading degenerative intervertebral disc changes. Degenerative intervertebral disc changes at each intervertebral disc level of the lumbar spine were assessed with the Pfirrmann grading system23. The Pfirrmann grading system involves five degrees from normal disc to advanced degeneration: grade 1, homogeneous disc with a bright high signal intensity, clear distinction between nucleus and annulus, and normal disc height; grade 2, inhomogeneous disc with a white signal intensity, clear distinction between nucleus and annulus, and normal height; grade 3, inhomogeneous disc with an intermediate gray signal intensity, unclear distinction between nucleus and annulus, and normal or slightly decreased disc height; grade 4, inhomogeneous disc with a dark low signal intensity, no possible distinction between nucleus and annulus, and normal or moderately decreased disc height; and grade 5, inhomogeneous disc with black low signal intensity, no possible distinction between nucleus and annulus, and complete disc narrowing. Each intervertebral disc was given a score from 1 to 5 according to the degrees of degeneration by the Pfirrmann grading system.
Statistical analysis
To ensure the study results, measurements of the antero-posterior diameter of the spinal canal and degenerative intervertebral disc changes, were assessed for reliability. In total, 30 measurements of the canal diameter and disc degeneration were tested in the study population. Two independent physicians specialized in spinal disorders participated in the testing. One (TH) measured imaging parameters twice at a 3-month interval. The other (JB) measured parameters once. Intra- and inter-observer reliability was expressed by the intra-class correlation coefficient (ICC) for canal diameter and kappa coefficient for disc degeneration. Reliability results were given with 95% confidence intervals (95%CIs). Reliability was considered poor with values <0.41, moderate ≥ 0.41, good ≥ 0.61, and very good ≥ 0.8124. All reported parameters in the study were further measured by the same investigator (TH). Quantitative variables are described with mean ± SD. Categorical variables are described with number (%). Comparisons involved the Kruskal-Wallis test. Correlation was assessed with the Spearman correlation coefficient. Statistical significance was set at P < 0.05. All statistical analyses involved using XLSTAT (Addinsoft, France).
Data availability
The authors state that the data will be available to readers for non-profit companies upon request.
References
Horton, W. A., Hall, J. G. & Hecht, J. T. Achondroplasia. Lancet 370, 162–72 (2007).
Baujat, G., Legeai-Mallet, L., Finidori, G., Cormier-Daire, V. & Le Merrer, M. Achondroplasia. Best Pract Res Clin Rheumatol. 22, 3–18 (2008).
Rousseau, F. et al. Mutations in the gene encoding fibroblast growth factor receptor-3 in achondroplasia. Nature. 371, 252–4 (1994).
Shiang, R. et al. Mutations in the transmembrane domain of FGFR3 cause the most common genetic form of dwarfism, achondroplasia. Cell. 78, 335–42 (1994).
Mahomed, N. N., Spellmann, M. & Goldberg, M. J. Functional health status of adults with achondroplasia. Am J Med Genet. 78, 30–5 (1998).
Hunter, A. G., Bankier, A., Rogers, J. G., Sillence, D. & Scott, C. I. Jr. Medical complications of achondroplasia: a multicentre patient review. J Med Genet. 35, 705–12 (1998).
Ain, M. C. et al. Progression of low back and lower extremity pain in a cohort of patients with achondroplasia. J Neurosurg Spine. 13, 335–40 (2010).
Jeong, S. T. et al. MRI study of the lumbar spine in achondroplasia. A morphometric analysis for the evaluation of stenosis of the canal. J Bone Joint Surg Br. 88, 1192–6 (2006).
Matsushita, T. et al. FGFR3 promotes synchondrosis closure and fusion of ossification centers through the MAPK pathway. Hum Mol Genet. 18, 227–40 (2009).
Katz, J. N. & Harris, M. B. Clinical practice. Lumbar spinal stenosis N Engl J Med. 358, 818–25 (2008).
Lee, S. Y., Kim, T. H., Oh, J. K., Lee, S. J. & Park, M. S. Lumbar Stenosis: A Recent Update by Review of Literature. Asian. Spine J. 9, 818–28 (2015).
Lurie, J. & Tomkins-Lane, C. Management of lumbar spinal stenosis. BMJ. 352, h6234 (2016).
Modi, H. N., Suh, S. W., Song, H. R. & Yang, J. H. Lumbar nerve root occupancy in the foramen in achondroplasia: a morphometric analysis. Clin Orthop Relat Res. 466, 907–13 (2008).
Epstein, N. E., Maldonado, V. C. & Cusick, J. F. Symptomatic lumbar spinal stenosis. Surg Neurol. 50, 3–10 (1998).
Srikumaran, U. et al. Pedicle and spinal canal parameters of the lower thoracic and lumbar vertebrae in the achondroplast population. Spine. 32, 2423–31 (2007).
Thomeer, R. T. & van Dijk, J. M. Surgical treatment of lumbar stenosis in achondroplasia. J Neurosurg. 96, 292–7 (2002).
Teraguchi, M. et al. Prevalence and distribution of intervertebral disc degeneration over the entire spine in a population-based cohort: the Wakayama Spine Study. Osteoarthritis Cartilage. 22, 104–10 (2014).
like, M. et al. Solovieva S, Lamminen A, Luoma K, Leino-Arjas P, Luukkonen R, Riihimäki H (2005) Disc degeneration of the lumbar spine in relation to overweight. Int J Obes. 29, 903–8 (2005).
Samartzis, D., Karppinen, J., Chan, D., Luk, K. D. & Cheung, K. M. The association of lumbar intervertebral disc degeneration on magnetic resonance imaging with body mass index in overweight and obese adults: a population-based study. Arthritis Rheum. 64, 1488–96 (2012).
Roussouly, P. & Pinheiro-Franco, J. L. Biomechanical analysis of the spino-pelvic organization and adaptation in pathology. Eur Spine J. 20, 609–18 (2011).
Hong, J. Y., Suh, S. W., Modi, H. N., Park, J. W. & Park, J. H. Analysis of sagittal spinopelvic parameters in achondroplasia. Spine. 36, E1233–9 (2011).
Kahanovitz, N., Rimoin, D. L. & Sillence, D. O. The clinical spectrum of lumbar spine disease in achondroplasia. Spine. 7, 137–40 (1982).
Pfirrmann, C. W., Metzdorf, A., Zanetti, M., Hodler, J. & Boos, N. Magnetic resonance classification of lumbar intervertebral disc degeneration. Spine. 26, 1873–8 (2001).
Landis, J. R. & Koch, G. G. The measurement of observer agreement for categorical data. Biometrics. 33, 159–74 (1977).
Acknowledgements
The study has been performed within the French Rare Bone Diseases Network “Oscar”.
Author information
Authors and Affiliations
Contributions
Conception, analysis and interpretation of data: T.H., J.D.L., M.C.S. and J.B. Reading the MRI: T.H., J.D.L., J.B. Drafting the manuscript: T.H., M.C.S., C.C. and J.B. Providing intellectual content of critical importance: J.D.L., C.C., G.B., V.C.D., A.Y., P.O. and J.B. Revising the manuscript: all authors. All gave the final approval of the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Huet, T., Cohen-Solal, M., Laredo, JD. et al. Lumbar spinal stenosis and disc alterations affect the upper lumbar spine in adults with achondroplasia. Sci Rep 10, 4699 (2020). https://doi.org/10.1038/s41598-020-61704-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-61704-w
This article is cited by
-
European Achondroplasia Forum Practical Considerations for Following Adults with Achondroplasia
Advances in Therapy (2024)
-
Fat infiltration in the thigh muscles is associated with symptomatic spinal stenosis and reduced physical functioning in adults with achondroplasia
Orphanet Journal of Rare Diseases (2023)
-
Recommendations for neuroradiological examinations in children living with achondroplasia: a European Society of Pediatric Radiology and European Society of Neuroradiology opinion paper
Pediatric Radiology (2023)
-
High prevalence of symptomatic spinal stenosis in Norwegian adults with achondroplasia: a population-based study
Orphanet Journal of Rare Diseases (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.