Abstract
The aim of the present study was to investigate the relationship of sleep quality and time with basal metabolic rate (BMR) and fuel oxidation in basal conditions and during exercise in sedentary middle-aged adults. We also studied the mediation role of dietary intake and adherence to the traditional Mediterranean Diet in the relationship between sleep parameters and energy metabolism parameters.A secondary analysis of the FIT-AGEING study was undertaken. 70 middle-aged sedentary adults (40–65 years old) participated in the present study. Sleep quality was assessed using the Pittsburgh Sleep Quality Index (PSQI) and wrist accelerometers (ActiSleep, Actigraph, Pensacola, Florida, USA) for 7 consecutive days. BMR was measured with indirect calorimetry and fuel oxidation was estimated through stoichiometric equations. Maximal fat oxidation was determined by a walking graded exercise test and dietary intake with 24 h recalls. Adherence to the traditional Mediterranean diet was assessed through the PREDIMED questionnaire. PSQI global score (poor sleep quality) was associated with lower basal fat oxidation (BFox), both expressed in g/min and as a percentage of BMR, independently of confounders. We did not find any association between other sleep and energy metabolism parameters. No mediating role of the dietary intake or PREDIMED global score was observed in the association of PSQI and BFox. In conclusion, our study showed that a subjective poor sleep quality was associated with lower BFox, which is not mediated by dietary intake in sedentary adults.
Similar content being viewed by others
Introduction
Cardiometabolic diseases and obesity are the leading causes of death in developed countries, becoming an epidemic in the last years1,2. Unhealthy diets represent one of the top risk factors for cardiometabolic diseases and obesity, developing a positive energy balance3. Simultaneously, a low basal metabolic rate (BMR), an impaired meal-induced thermogenesis and low physical activity levels could result in a reduced total energy expenditure4. This low total energy expenditure coupled to high energy intake could produce a gradual weight gain and visceral adipose tissue deposition, increasing the risk of cardiometabolic diseases and obesity5.
The ability to oxidize fat as a fuel is considered an important metabolic health parameter6. An impaired ability to oxidize fat is associated with an increased risk of obesity, type 2 diabetes mellitus, cardiovascular disease, metabolic syndrome, cancer and systemic inflammation7. Therefore, fat oxidation in basal conditions (BFox) and maximal fat oxidation during exercise (MFO) are considered markers of metabolic health8,9,10.
Sleep pattern variations, including a decrease in the quality and quantity of sleep, have been shown to be also a risk factor for the development of obesity and cardiometabolic diseases11. These changes in sleep quality and quantity disrupt the circadian rhythms and may have deleterious consequences on people health12. Previous studies have provided a causal link between short sleep duration and poor sleep quality with pathological metabolic consequences due to the disruption in the circadian rhythms and increasing levels of adiposity11,13. Metabolic regulation is not an output function of the circadian system12. However, nutrient, energy and redox levels signal back to cellular clocks to reinforce circadian rhythms and to adapt physiology (i.e. hormones, body temperature, nervous system) to temporal tissue-specific requirements11,13. In this sense, previous studies have demonstrated that poor sleep quality and quantity may decrease BMR and BFox14, and sleep deprivation may not affect MFO in young adults15. However, there are no studies testing the associations between sleep quality and time (both subjective and objective) with BMR and fuel oxidation in basal conditions and during exercise.
One of the possible causes of the relationship between sleep parameters with BMR and fuel oxidation could be the dietary modifications. In this sense, unhealthy sleep patterns could increase food consumption and consequently energy intake through several previously-explained potential mechanisms16. Previous studies have shown that dietary intake could influence BFox and MFO8,17,18. Concretely, a high-fat, low-carbohydrate intake could increase BFox and MFO6,19. As well as, the lack of sleep can increase the consumption of high fat energy-dense foods16, which theoretically may modify BFox and MFO. However, there is a lack of evidence investigating the mediating role of dietary intake in the relationship of sleep outcomes with energy metabolism parameters.
Therefore, the aim of the present study was to investigate the relationship of subjective and objective sleep quality and time with BMR and fuel oxidation in basal conditions and during exercise in sedentary middle-aged adults. We also aimed to study the mediation role of dietary intake and adherence to the traditional Mediterranean Diet between sleep parameters and energy metabolism parameters in sedentary middle-aged adults. The present study undertakes a secondary analysis of the FIT-AGEING study.
Materials and Methods
Participants and design
A total of 70 (36 women) middle-aged sedentary adults (40–65 years old) participated in this cross-sectional study. The participants were enrolled in the FIT-AGEING study20, an exercise-based randomized controlled trial (clinicaltrial.gov: ID: NCT03334357). Data for these subjects were collected at baseline data collection in the FIT-AGEING study. All of them declared: (i) to be non-physically active (<20 min of moderate-intensity physical activity on 3 days/week), (ii) to have a stable weight (weight changes <5 kg) over the last 5 months, (iii) to be healthy and (iv) to be free of medication (medication for thyroid, betablockers, benzodiazepines, glucose lowering medication, or cardiovascular medication) during the last 5 months. All participants gave their oral and written informed consent before the beginning of the intervention. The study was approved by the Ethics Committee on Human Research of the University of Granada and the Andalusian Health Service (SAS) (CEI-Granada) (0838-N-2017). The study protocols and experimental design were applied according to the last revised ethical guidelines of the Declaration of Helsinki. All assessments were made at the Sport and Health University Research Institute (iMUDS, Granada, Spain) during September and October 2016 and September and October 2017.
Anthropometric and body composition measurements
Anthropometric variables were measured by a certified anthropometrist [the International Society for the Advancement of Kinanthropometry (ISAK)] following the ISAK guidelines21. Both body weight and height were assessed using an electronic scale and stadiometer (Seca model 799, Hamburg, Germany), and the body mass index (BMI) was calculated as (weight [kg]/ height2 [m]). A dual-energy X-ray absorptiometry scanner (Discovery Wi, Hologic, Inc., Bedford, MA, USA) was used to measure body composition following the manufacturer’s recommendations. We conducted the quality controls, the positioning of the participants and the analyses of the results following the manufacturer’s recommendations. An automatic delineation of the anatomic regions was performed by the software APEX 4.0.2. We acquired spine phantom quality control scans on each study day. The results displayed lean mass and fat mass and the lean mass index (LMI) and the fat mass index (FMI), which were calculated as (lean mass [kg]/ height2 [m]) and (fat mass [kg]/ height2 [m]) respectively.
Sleep quality and time assessment
The Pittsburgh Sleep Quality Index (PSQI) is a self-report tool which consists of 19-item scale that provides 7 component scores (ranges 0–3): (i) subjective sleep quality (very good to very bad), (ii) sleep latency (≤15 minutes to >60 minutes), (iii) sleep duration (≥7 hours to <5 hours), (iv) sleep efficiency (≥85% to <65% hours sleep/hours in bed), (v) sleep disturbances (not during the past month to ≥ 3 times per week), (vi) use of sleeping medications (none to ≥ 3 times a week), and (vii) daytime dysfunction (not a problem to a very big problem), with a total global score ranging from 0 to 2122. A PSQI global score higher than 5 indicates poor sleep quality22.
Objective characteristics of sleep-wake cycles were monitored with a wrist-worn accelerometer (ActiSleep, Actigraph Pensacola, Florida, USA) for 7 consecutive days (24 hours/day)20. Participants received detailed information on how to wear the accelerometer and were asked to remove it only for water activities. It was also recorded the times in which the participants went to bed every night, woke up every morning and removed the device every day. The accelerometers used an epoch length of 5 seconds and a frequency rate of 100 Hz to store raw accelerations23. The raw accelerations were exported in “.csv” format using ActiLife v. 6.13.3 software (ActiGraph, Pensacola, FL, US) and processed using the GGIR package (v. 1.6–0, https://cran.r-project.org/web/packages/GGIR/index.html)24 in R (v. 3.1.2, https://www.cran.r-project.org/). We derived the Euclidean Norm Minus One G (ENMO) as √(x^2 + y^2 + z^2) −1G (where 1 G ~ 9.8 m/s2) with the accelerometer’s z angle to describe sleep patterns. We used a previously published algorithm combining data from the accelerometers and diary reports to detect sleep period time25,26. According to this algorithm, sleep was defined as any period of sustained inactivity, in which there were minimal changes in the arm angle (i.e., as much 5 degrees for 5 minutes periods) during a period recorded as sleep by the participant in their diary reports25. The following variables were analyzed: total sleep time (minutes slept between bedtime and wake time), sleep efficiency (percentage of time asleep while in bed) and wake after sleep onset (minutes awake between sleep onset and wake time). It is to note that only the participants wearing the accelerometers for ≥16 hours/day for at least 4 days (including at least 1 weekend day) were included in the final analyses (i.e. a total of 4 participants did not meet these conditions)23. The mean accelerometer wear-time for the 70 participants included in the final analyses were 6.7 days (4.3% of non-wear time).
Basal metabolic rate and fuel oxidation in basal conditions
Subjects were told to arrive at the laboratory in fasting condition of at least 8 h in a motor vehicle and to avoid any moderate/vigorous physical activity in the previous 24 h/48 h respectively; all were required to confirm that they had met these conditions. The evening meal consumed by the subjects prior to fasting was previously standardized: an egg omelet with fried tomato and boiled rice.
BMR and fuel oxidation in basal conditions were measured through indirect calorimetry (IC) following the current scientific consensus27. All tests were conducted in the same quiet room with controlled room temperature (i.e. 22–24 °C) and humidity (i.e. 35–45%). IC measurements were performed during 30-minute periods with a CPX Ultima CardiO2 system (Medical Graphics Corp, St Paul, MN, USA) employing a neoprene face-mask with no external ventilation27.
The Ultima CardiO2 metabolic cart device assessed oxygen consumption (VO2) using a galvanic fuel cell, and carbon dioxide production (VCO2) via non-dispersive infrared analysis using a breath-by-breath system28. A gas calibration using 2 standard gas concentrations and a flow calibration using a 3-L calibration syringe were performed following the manufacturer’s recommendations. Prior to the start of the BMR assessment, the subjects reclined on a bed for ~30 min in a comfortable supine position covered by a sheet29,30. During the assessment, participants laid on a bed in a supine position and were instructed to breathe normally and not to talk, fidget or sleep.
The first 5 minutes of each measurement were discarded and the most stable 5-min period that met steady state criteria (i.e. coefficient of variation <10% in VO2, CO2, minute ventilation, and coefficient of variation <10% in respiratory exchange ratio) was considered for further analyses following previous studies29,30,31,32. The Weir’s abbreviated equation33 was used to estimate the BMR expressed in kcal/day and also calculated with respect to the lean mass (BMRLM). The Frayn’s equation was used to estimate BFox and basal carbohydrate oxidation (BCHox) expressed in g/min34. The BFox and BCHox were also expressed as a percentage of the BMR.
Maximal fat oxidation during exercise assessment
MFO and the intensity that elicit MFO (FATmax) were assessed in a different day of the BMR and BFox/BCHox test (i.e. interval 3 to 15 days). Participants were asked to arrive at the laboratory in a fasted state of 6 hours and to avoid any physical activity both moderate (24 h) and vigorous intensity (48 h) before the measurement.
A walking graded test on a treadmill (H/P/cosmos pulsar, H/P/cosmos sports & medical GmbH, Nussdorf-Traunstein, Germany) was performed to calculate MFO and FATmax following a previously validated methodology35. Briefly, the protocol started assessing the maximal walking speed of each participant35,36,37. After ~3 minutes resting, the walking graded test started with a 3-minute warm up at 3.5 km/h. Subsequently, the treadmill speed was increased 1 km/h every 3 minutes until the maximal walking speed was reached. Thereafter, the treadmill gradient was increased 2% every 3 minutes until the respiratory exchange ratio was above 1.0. An automated gas analysis system (CPX Ultima CardiO2; Medical Graphics Corp, St Paul, MN) was used to record breath-by-breath gas exchange measurements. Participants wore an oronasal mask (model 7400, Hans Rudolph Inc, Kansas City, MO, USA) equipped with a prevent™ metabolic flow sensor (Medgraphics Corp, Minnesota, USA). Gas analysis systems were calibrated following the manufacturer’s recommendations. VO2 and VCO2 were averaged over and the last 60 seconds of each graded exercise protocol stage. Frayn’s equation was used to estimate fat oxidation rates34. These fat oxidation values were plotted against the relative-exercise intensity, expressed as the percentage of maximum oxygen uptake (VO2max); a third-degree polynomial curve was built to determine MFO and FATmax38. MFO was also expressed as MFOLM in order to relativize it to the lean mass. Maximal carbohydrate oxidation was not included in the analyses since it is not a key factor of energy metabolism during exercise39. Indeed, our recent systematic review has analyzed a total of 112 studies which included data about fuel oxidation during exercise37. None of those studies reported maximal carbohydrate oxidation during exercise.
Cardiorespiratory fitness assessment
VO2max was determined using a maximum treadmill (H/P/Cosmos Pulsar treadmill, H/P/Cosmos Sport & Medical GMBH, Germany) exercise test following the modified Balke protocol, which has been extensively validated40. In short, the warm up consisted in walking at 3 km/h for 1 minute followed by 2 minutes at 4 km/h. The incremental protocol started at a speed of 5.3 km/h (0% grade), which was kept constant with the gradient increasing by 1% every minute until the participants reached their volitional exhaustion. We used the same indirect calorimetry and software as in the MFO assessment.
Dietary intake assessment
Diet was assessed using three 24-hour recalls carried out on 3 separate days (2 weekdays and 1 weekend day) by a qualified and trained research dietitian. Dietary recalls were done on different days than the MFO and VO2 assessments.
In the face-to-face interviews, the participants were asked to recall all food consumed during the previous day. The interviews involved a detailed assessment and description of the food consumption using colored photographs of different-size portions of food to improve the participants’ accuracy of food quantification41. These data were introduced by two independent qualified and trained dietitians in the EvalFINUT® software. Energy, macronutrient, fiber, lipid profile and ethanol intake data were obtained by EvalFINUT®, which is based on the USDA (United States Department of Agriculture) and BEDCA (“Base de Datos Española de Composición de Alimentos”) databases.
Dietary energy density was calculated by dividing the energy contained in food and beverages (excluding water) by the total weight of daily food and beverages (expressed as kcal/g)42. Energy and weight data of daily food and beverages were obtained from the 24-hours recalls.
The traditional Mediterranean diet is associated with a lower prevalence of chronic diseases (i.e. obesity, metabolic syndrome, cardiovascular diseases, cancer) and mortality43. The adherence to the traditional Mediterranean Diet (MedDiet) was estimated by using the 14-point questionnaire of adherence to the MedDiet used and validated in the PREDIMED trial44. The PREDIMED questionnaire includes 12 questions related to frequency intake of key foods and 2 questions related to specific dietary habits of the MedDiet. Each question scores 0 or 1 point. The global score ranges from 0 to 14, being 0 points null adherence and 14 points complete adherence to the MedDiet. The PREDIMED questionnaire proved to be very useful in a large Spanish cohort for a quick adherence estimation to the traditional MedDiet44.
Statistical analysis
The sample size and power calculations were made based on the data of a pilot study of the FIT-AGEING study20. This study aimed to compare the influence of different exercise programs on BMR, BFox and MFO in sedentary middle-aged adults. We based the sample size calculations on a minimum predicted change in MFO of 0.05 g/min between the intervention groups and the control group, and an SD for this change of 0.05 g/min. A sample size of 17 participants was predicted to provide a statistical power of 80%, considering a type I error of 0.05. Assuming a maximum loss of 25% at follow-up, we decided to recruit at least 20 participants for each group (N = ~80 individuals). The present study is based on a secondary analysis using baseline data from the FIT-AGEING study, and therefore a specific sample size calculation was not conducted.
We used the Shapiro–Wilk test, visual check of histograms, Q-Q and box plots to verify all variable distributions. The descriptive parameters were reported as mean and standard deviation. Given that we did not observe a sex interaction, we conducted the analysis including men and women together. Simple linear regressions were performed to examine the association between sleep time and quality (PSQI global score, total sleep time, sleep efficiency and wake after sleep onset) with BMR, BMRLM, BFox, BCHox, MFO, MFOLM and FATmax. We also conducted multiple linear regression models to test these associations after adjusting for sex (Model 1), sex and age (Model 2) and sex, age and FMI (Model 3).
Pearson correlation was performed to assess the association between sleep parameters and dietary outcomes. Effect modification analyses were conducted to test the joint effects of dietary intake (dietary intake outcome*sleep outcome) and sleep quality on energy metabolism. To quantify the mediating role of dietary intake (i.e. energy, macronutrient, fiber, ethanol and lipid profile intake, and PREDIMED total score) in the relationship between sleep parameters and BMR and fuel oxidation, we conducted mediation analyses45. We used the PROCESS macro version 3.3, model 4 with 5,000 bias-corrected bootstrap samples and 95% confidence intervals. Bootstrapping is a nonparametric resampling procedure that does not require the assumption of normality of the sampling distribution46 The mediation was estimated using the indirect effect, which indicates the change in the effect of the independent variable on the outcome that can be endorsed to the proposed mediator. Indirect effects (a × b paths) with confidence intervals not including zero are interpreted as statistically significant47 which could occur regardless of the significance of the total effect (c path, effect of the independent variable on the dependent variable) and the direct effect (c’ path, effect on the dependent variable when both the independent and the mediator variables are included as independent variables)45. To quantify how much of the total effect was due to the mediation, we calculated the percentage of mediation ([indirect effect / total effect] × 100) provided when the total effect was larger than the indirect effect with the same direction45.
All analyses were conducted using the Statistical Package for Social Sciences (SPSS, v. 25.0, IBM SPSS Statistics, IBM Corporation) and the level of significance was set at <0.05. Graphical presentations were prepared using GraphPad Prism 8 (GraphPad Software, San Diego, CA, USA).
Ethical standards
Ethical approval for the study was given by the Ethics Committee on Human Research at the University of Granada and Servicio Andaluz de Salud (CEI-Granada) (0838-N-2017). Written informed consent was obtained from all subjects. This study was in accordance with the last revised ethical guidelines of the Declaration of Helsinki.
Results
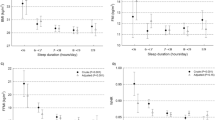
The characteristics of the study sample are shown in Table 1. We observed an inverse association between total sleep time and BMR (P < 0.001; Table 2), which disappeared after including sex, age and FMI in the model (all P > 0.105; Table 2). No association was found between the remaining subjective or objective sleep parameters with BMR and BMRLM (all P > 0.071; Table 2), neither when we accounted for confounders.
An inverse association was detected between PSQI global score with BFox (expressed in g/min, and as %BMR) (all P < 0.001; Table 3), which remained significant after including sex, age and FMI in the model (all P < 0.002; Table 3). We did not find any significant association between any objective sleep parameter with BFox (all P > 0.265; Table 3). PSQI was also positively associated with BCHox (expressed in g/min, and as %BMR) even after controlling for sex, age and FMI (all P < 0.002; Table S1).
We showed an inverse association between total sleep time with MFO (expressed in g/min; P = 0.008; Table 4), which disappeared when the model includes sex, age and FMI (all P > 0.651; Table 4). No association was found between the remaining sleep parameters with MFO and MFOLM (all P > 0.171; Table 4).
We repeated all previous associations controlling for menopausal status (pre- or post-menopausal) in order to avoid the possible cofounder of female hormones, and the results did not change (data not shown).
We observed only a negative association of fiber intake and PSQI global score and cholesterol intake negatively and positively associated with total sleep time and wake after sleep onset respectively (Table S5). However, we observed a modification effect of different dietary factors (i.e. fiber and ethanol intake; Table S5). Despite this modification effect of dietary factors and the several associations between dietary factors and sleep parameters, we did not find a mediating effect of energy, dietary energy density, fat, protein, carbohydrate, fiber intake, lipid profile intake, ethanol intake and PREDIMED total score on the association of the PSQI global score and BFox both expressed in g/min and in %BMR (Figs. S1–S4).
Discussion
The main finding of the present study is that a poor subjective sleep quality was associated with lower BFox independently of sex, age and body composition outcomes. No consistent association was observed between any sleep quality and time parameters with BMR, MFO and FATmax. Moreover, our results indicated that the association of PSQI global score with BFox was not mediated by dietary intake and MedDiet adherence.
We observed an inverse association between total sleep time and BMR which disappeared after controlling for confounders. The energy expenditure is lowest during sleep, therefore a high total sleep time is related with a prolonged period of the lowest energy expenditure48. Sleep deprivation could increase energy expenditure since energy expenditure is reduced during sleep48. Sharma et al. proposed that these reduction in energy expenditure could be influenced by circadian rhythm, body temperature and muscle temperature48. However, the results should be interpreted with caution because this association disappeared after controlling for sex, age and FMI. Several physiological mechanisms could explain the relationship between sleep quality and BFox. Sleep restriction is associated with insulin resistance characterized by a decreased insulin-mediated glucose uptake49, which could develop metabolic inflexibility characterized by an impaired BFox50. Short sleep duration and sleep fragmentation arealso related to low leptin levels or leptin resistance51 which are associated with an impaired fatty acid oxidation52. Sleep disruption (discontinuity of sleep) can lead to the disruption of circadian rhythms13, which orchestrate crucial physiological and behavioral functions, being one of these the regulation of carbohydrate and fatty acid metabolism12. Higher sleep duration and quality are associated with a healthier gut microbiome53, which could suppress insulin signaling, increase β-oxidation and inhibit fat oxidation derived from the production of short-chain fatty acids54. Furthermore, sleep disruption (discontinuity of sleep) could decrease melatonin production13, which has important metabolic functions, such as lipolysis, regulating the energy flow55. An increase in the production of pro-inflammatory cytokines and reactive oxygen species is observed in impaired sleep patterns13. Both inflammation and oxidative stress could modulate metabolic flexibility, specifically fat oxidation56,57. Therefore, based on the above-mentioned mechanisms, a healthy sleep pattern could improve metabolic health via the increment of BFox and viceversa.
In addition, an impaired sleep pattern, determined by a low sleep duration could increase energy intake through several potential mechanisms: increment of time and opportunities for eating, psychological distress, sensitivity to food reward, energy needed to sustain wakefulness, hunger hormones and decrease dietary restraint16. A lack of sleep or low sleep quality could increase the intake of high energy-dense foods, high fat and sugary snacks, which are low in fiber16. In this sense, although we did not find any association between energy and macronutrient intake, we observed that fiber intake was negatively associated with PSQI global score. Fiber intake could have different metabolic effects (i.e. insulin sensitivity and glycemia improvement)58, that could have a potential role in the regulation of fat oxidation. However, we did not find any mediating role of dietary intake (i.e. fiber intake) between the association of PSQI with BFox. The lack of a mediating role may be due to specific issues: (i) since dietary outcomes were assessed in a specific time point, it could be that the dietary intake was insufficiently maintained over time to modify BFox; (ii) the possible lower and upper threshold for when dietary intake (i.e. fat intake) could modify fat oxidation59; (iii) the inter-individual variability, body composition and metabolic status influence on fat oxidation8; (iv) a sleep patterns insufficiently maintained over time.
The lack of association between any sleep outcomes with BMR and MFO could be explained by different factors. Sleep is a complex phenomenon influenced by behavioral and physiological mechanisms (i.e. homeostatic, circadian and metabolic control) under the participant’s natural sleep environment that we have not investigated60. These factors could influence the relationship between sleep parameters and BMR and MFO.
We also observed an inverse association between total sleep time with MFO. A previous study of Konishi et al. observed that a night of sleep deprivation did not affect MFO in healthy young men15. It has been reported several detrimental effects of long sleep for optimal health61. In addition, long sleep could increase fatigue, physiological deprivation, which could influence insulin resistance and hormonal imbalance62. Although the mechanisms are not clear, the above-mentioned mechanisms could have influenced this relationship. However, the results should be interpreted with caution because this association disappeared after controlling for sex, age and FMI.
Surprisingly, different results were observed when the association between sleep quality and energy metabolism was performed considering subjective instead of objective measures of sleep quality. It has been previously reported that PSQI and accelerometer records measure different attributes of sleep, highlighting the bias of accelerometry to register wakefulness, thus lying in bed awake but motionless is likely to be coded as sleep63. Therefore, it is recommended to use both methods to obtain complementary information additionally to the body movements64. These differences in measurement of sleep attributes could explain the different results of the associations between sleep quality and energy metabolism.
Despite accelerometer records and subjective measurements are a valid and extensively used measure of sleep quality26,65 they cannot differentiate between rapid eye movement sleep (REM) and non-rapid eye movement sleep (NREM), restricting the detailed assessment of the real biologic process of sleep. REM and NREM phases are metabolically different66. In REM sleep glucose uptake is increased, leading to anaerobic glucose metabolism67,68, therefore sleep quality in each phase could be differently associated with energy metabolism. Future studies that examine the relationship between REM and NREM sleep using polysomnography records with BMR and fuel oxidation in basal conditions and during exercise are needed.
The present study should be interpreted with caution; the study has a cross-sectional design that does not allow to establish causal relationship. Therefore, experimental studies should manipulate BMR and fuel oxidation and/or sleep (e.g. sleep deprivation) under well-controlled lab conditions in order to establish causal relationship. Furthermore, sleep and dietary parameters were assessed only in a specific timepoint, which do not allow us to extrapolate our results to chronic sleep or dietary patterns. Our study only included middle-aged sedentary adults and, consequently, we cannot extrapolate our results to older, younger, and/or physically active individuals. The difficulty of an accurate dietary evaluation with possible underreporting or misclassification should be considered, as in all cross-sectional studies. Lastly, the narrow PSQI global score range should be taken into account when interpreting our results.
Conclusions
In conclusion, our study showed that a subjective poor sleep quality was associated with lower BFox. No association was found between the remaining sleep parameters with BMR and fuel oxidation in basal conditions and during exercise. Moreover, our findings indicated that the association of PSQI global score with BFox was not mediated by dietary intake and MedDiet adherence. Further studies are needed to better understand the physiological mechanisms of sleep regulation and how it could influence the BMR and fuel oxidation in basal conditions and during exercise.
References
WHO. Noncommunicable Diseases Country Profiles 2018. World Health Organization 369 (2018).
Abarca-Gómez, L. et al. Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: a pooled analysis of 2416 population-based measurement studies in 128·9 million children, adolescents, and adults. Lancet 390, 2627–2642 (2017).
Mozaffarian, D. Foods, obesity, and diabetes-are all calories created equal? Nutr. Rev. 75, 19–31 (2017).
St-Onge, M.-P. & Gallagher, D. Body composition changes with aging: The cause or the result of alterations in metabolic rate and macronutrient oxidation? Nutrition 26, 152–155 (2010).
Tchernof, A. & Després, J. Pathophysiology of human visceral obesity: an update aspects of regional body fat distribution. Physiol. Rep. 93, 359–404 (2013).
Fletcher, G. et al. Dietary intake is independently associated with the maximal capacity for fat oxidation during exercise. Am. J. Clin. Nutr. 105, 864–872 (2017).
Smith, R. L., Soeters, M. R., Wüst, R. C. I. & Houtkooper, R. H. Metabolic flexibility as an adaptation to energy resources and requirements in health and disease. Endocr. Rev. 39, 489–517 (2018).
Galgani, J. E., Moro, C. & Ravussin, E. Metabolic flexibility and insulin resistance. Am. J. Physiol. Metab. 295, E1009–E1017 (2008).
Maunder, E., Plews, D. J. & Kilding, A. E. Contextualising maximal fat oxidation during exercise: Determinants and normative values. Front. Physiol. 9, 1–13 (2018).
Goodpaster, B. H., Sparks, L. M. & Hospital, F. Metabolic flexibility in health and disease. Cell Metab. 25, 1027–1036 (2017).
Cappuccio, F. P. & Miller, M. A. Sleep and cardio-metabolic disease. Curr Cardiol Rep 19, 67–79 (2018).
Reinke, H. & Asher, G. Crosstalk between metabolism and circadian clocks. Nat. Rev. Mol. Cell Biol. 20, 227–241 (2019).
Medic, G. & Wille, M. H. M. E. Short- and long-term health consequences of sleep disruption. Nat. Sci. Sleep 9, 151–161 (2017).
Penev, P. D. Update on energy homeostasis and insufficient sleep. J. Clin. Endocrinol. Metab. 97, 1792–1801 (2012).
Konishi, M. et al. Effect of one night of sleep deprivation on maximal fat oxidation during graded exercise. J. Phys. Fit. Sport. Med. 2, 121–126 (2013).
Chaput, J. P. Sleep patterns, diet quality and energy balance. Physiol. Behav. 134, 86–91 (2014).
Kahlhöfer, J. et al. Carbohydrate intake and glycemic index affect substrate oxidation during a controlled weight cycle in healthy men. Eur. J. Clin. Nutr. 68, 1060–1066 (2014).
Carstens, M. T. et al. Fasting substrate oxidation in relation to habitual dietary fat intake and insulin resistance in non-diabetic women: A case for metabolic flexibility? Nutr. Metab. 10, 1–8 (2013).
Labayen, I., Forga, L. & Martínez, J. A. Nutrient oxidation and metabolic rate as affected by meals containing different proportions of carbohydrate and fat, in healthy young women. Eur. J. Nutr. 38, 158–166 (1999).
Amaro-Gahete, F. J. et al. Exercise training as S-Klotho protein stimulator in sedentary healthy adults: rationale, design, and methodology. Contemp. Clin. Trials Commun. 11, 10–19 (2018).
Marfell-Jones, M., Olds, T. & Stewart, A. International standards for anthropometric assessment. International Society for the Advancement of Kinanthropometry. Potchefstroom, South Africa ISAK (2011).
Buysse, D. J., Reynolds, C. F., Monk, T. H., Berman, S. R. & Kupfer, D. J. The Pittsburgh sleep quality index: A new instrument for psychiatric practice and research. Psychiatry Res. 28, 193–213 (1989).
Migueles, J. H., Ulf, C. C. & Nystro, C. D. Accelerometer Data Collection and Processing Criteria to Assess Physical Activity and Other Outcomes: A Systematic Review and Practical Considerations. Sport. Med. 47, 1821–1845 (2017).
van Hees, V. T. et al. Separating Movement and Gravity Components in an Acceleration Signal and Implications for the Assessment of Human Daily Physical Activity. PLoS One 8, e61691 (2013).
van Hees, V. T. et al. A Novel, Open Access Method to Assess Sleep Duration Using a Wrist-Worn Accelerometer. PLoS One 10, e0142533 (2015).
Sadeh, A. The role and validity of actigraphy in sleep medicine: An update. Sleep Med. Rev. 15, 259–267 (2011).
Fullmer, S. et al. Evidence Analysis Library Review of Best Practices for Performing Indirect Calorimetry in Healthy and Non-Critically Ill Individuals. J. Acad. Nutr. Diet. 115, 1417–1446.e2 (2015).
Sundström, M., Tjäder, I., Rooyackers, O. & Wernerman, J. Indirect calorimetry in mechanically ventilated patients. A systematic comparison of three instruments. Clin. Nutr. 32, 118–21 (2013).
Amaro-Gahete, F. J. et al. Congruent Validity of Resting Energy Expenditure Predictive Equations in Young Adults. Nutrients 11, 1–13 (2019).
Amaro-Gahete, F. et al. Accuracy and Validity of Resting Energy Expenditure Predictive Equations in Middle-Aged Adults. Nutrients 10, 1635 (2018).
Sanchez-Delgado, G. et al. Reliability of resting metabolic rate measurements in young adults: Impact of methods for data analysis. Clin. Nutr., https://doi.org/10.1016/j.clnu.2017.07.026 (2017).
Alcantara, J. M. A. et al. Congruent validity and inter-day reliability of two breath by breath metabolic carts to measure resting metabolic rate in young adults. Nutr. Metab. Cardiovasc. Dis. 28, 929–936 (2018).
Weir, J. New methods for calculating metabolic rate with special reference to protein metabolism. J. Physiol. 109, 1–9 (1949).
Frayn, K. N. Calculation of substrate oxidation rates in vivo from gaseous exchange. J. Appl. Physiol. 55, 628–34 (1983).
Amaro-Gahete, F. J. et al. Diurnal Variation of Maximal Fat Oxidation Rate in Trained Male Athletes. Int. J. Sports Physiol. Perform. 2, 1–20 (2019).
Amaro-Gahete, F. J. & Ruiz, J. R. Methodological issues related to maximal fat oxidation rate during exercise: Comment on: Change in maximal fat oxidation in response to different regimes of periodized high-intensity interval training (HIIT). Eur. J. Appl. Physiol. 118, 2029–2031 (2018).
Amaro-Gahete, F. J. et al. Assessment of maximal fat oxidation during exercise: A systematic review. Scand. J. Med. Sci. Sport. 29, 910–921 (2019).
Amaro-Gahete, F. J. et al. Impact of data analysis methods for maximal fat oxidation estimation during exercise in sedentary adults. Eur. J. Sport Sci. 19, 1230–1239 (2019).
Goodpaster, B. H. & Sparks, L. M. Metabolic Flexibility in Health and Disease. Cell Metab. 25, 1027–1036 (2017).
Balke, B. & Ware, R. W. An experimental study of physical fitness of Air Force personnel. US Armed Forces Med. J. 10, 675–88 (1959).
López, M. D. R., Martín-Lagos, R. A. Guía para estudios dietéticos: álbum fotográfico de alimentos. (Editorial Universidad de Granada, 2010).
Ledikwe, J. H. et al. Dietary Energy Density Determined by Eight Calculation Methods in a Nationally Representative United States Population. J. Nutr. 135, 273–278 (2005).
Zaragoza-Martí, A., Cabañero-Martínez, M. J., Hurtado-Sánchez, J. A., Laguna-Pérez, A. & Ferrer-Cascales, R. Evaluation of Mediterranean diet adherence scores: A systematic review. BMJ Open 8, 1–8 (2018).
Schroder, H. et al. A Short Screener Is Valid for Assessing Mediterranean Diet Adherence among Older Spanish Men and Women. J. Nutr. 141, 1140–1145 (2011).
Hayes, A. Introduction to mediation, moderation, and conditional process analysis: A regression-based approach. (Guilford Publications., 2017).
Preacher, K. J. & Hayes, A. F. Asymptotic and resampling strategies for assessing and comparing indirect effects in multiple mediator models. Behav. Res. Methods 40, 879–891 (2008).
Hayes, A. F. Beyond Baron and Kenny: Statistical mediation analysis in the new millennium. Commun. Monogr. 76, 408–420 (2009).
Sharma, S. & Kavuru, M. Sleep and metabolism: An overview. Int. J. Endocrinol. 2010, (2010).
Rao, M. N. et al. Subchronic sleep restriction causes tissue-specific insulin resistance. J. Clin. Endocrinol. Metab. 100, 1664–1671 (2015).
Galgani, J. E., Moro, C. & Ravussin, E. Metabolic flexibility and insulin resistance. AJP Endocrinol. Metab. 295, E1009–E1017 (2008).
Pan, W. & Kastin, A. J. Leptin: A biomarker for sleep disorders? Sleep Med. Rev. 18, 183–290 (2014).
Stern, J. H., Rutkowski, J. M. & Scherer, P. E. Adiponectin, leptin, and fatty acids in the maintenance of metabolic homeostasis through adipose tissue crosstalk. Cell Metab. 23, 770–784 (2016).
Hagen, E. W., Holzhausen, E. A., Sethi, A. K., Malecki, K. M. & Nasia Safdar, P. E. P. 0106 Sleep Duration and Quality and Diversity of the Gut Microbiome in a General Population Sample of Adults. Sleep 42, A43–A44 (2019).
Serrano, J., Cassanye, A., Martín-Gari, M., Granado-Serrano, A. & Portero-Otín, M. Effect of Dietary Bioactive Compounds on Mitochondrial and Metabolic Flexibility. Diseases 4, 14 (2016).
Cipolla-Neto, J., Amaral, F. G., Afeche, S. C., Tan, D. X. & Reiter, R. J. Melatonin, energy metabolism, and obesity: a review. J. Pineal Res. 56, 371–381 (2014).
Sala, C. et al. The role of low-grade inflammation and metabolic flexibility in aging and nutritional modulation thereof: A systems biology approach. Mech. Ageing Dev. 136–137, 138–147 (2014).
Hotamisligil, G. S. Inflammation, metaflammation and immunometabolic disorders. Nature 542, 177–185 (2017).
Canfora, E. E., Jocken, J. W. & Blaak, E. E. Short-chain fatty acids in control of body weight and insulin sensitivity. Nat. Rev. Endocrinol. 11, 577–591 (2015).
Whelan, M. E., Wright, O. R. L. & Hickman, I. J. A Review of the Effect of Dietary Composition on Fasting Substrate Oxidation in Healthy and Overweight Subjects. Crit. Rev. Food Sci. Nutr. 56, 146–151 (2016).
Acosta F. M. et al. Sleep duration and quality are not associated with brown adipose tissue volume or activity - as determined by 18F-FDG uptake, in young, sedentary adults Author. Sleep 1–27 (2019).
Grandner, M. A. & Drummond, S. P. A. Who are the long sleepers? Towards an understanding of the mortality relationship. Sleep Med. Rev. 11, 341–360 (2007).
Kim, C. E. et al. Association between sleep duration and metabolic syndrome: A cross-sectional study. BMC Public Health 18, 1–8 (2018).
Berger, I., Obeid, J., Timmons, B. W. & Dematteo, C. Exploring Accelerometer Versus Self-Report Sleep Assessment in Youth With Concussion. Glob. Pediatr. Heal. 4, 1–7 (2017).
Jurado-Fasoli, L. et al. Association between Sleep Quality and Body Composition in Sedentary Middle-Aged Adults. Medicina (Kaunas). 54 (2018).
Martin, J. L. & Hakim, A. D. Wrist actigraphy. Chest 139, 1514–1527 (2011).
Copinschi, G., Leproult, R. & Spiegel, K. The important role of sleep in metabolism. How Gut Brain Control Metab. 42, 59–72 (2014).
Nedeltcheva, A. V., Program, M. C. & Disorders, C. Metabolic effects of sleep disruption, links to obesity and diabetes. Curr Opin Endocrinol Diabetes Obes 21, 293–298 (2014).
Parmeggiani, P. L. & Velluti, R. A. The physiologic nature of sleep. (World Scientific., 2005).
Acknowledgements
The authors would like to thank all the participants that took part of the study for their time and effort. This study is part of a Ph.D. Thesis conducted in the Biomedicine Doctoral Studies of the University of Granada, Spain. We are grateful to Dr. Ángel Gutiérrez and Alejandro De la O for all their support in the study. We are grateful to Ms. Ana Yara Postigo-Fuentes for her assistance with the English language. The study was supported by the Spanish Ministry of Education (FPU14/04172 and FPU15/03960), by the University of Granada UGR Research and Knowledge Transfer Fund (PPIT) 2016 (Excellence Actions Programme. Units of Scientific Excellence: Scientific Unit of Excellence on Excercise and Health [UCEES]) and Plan Propio de Investigación 2019 - Programa Contratos-Puente, by the Regional Government of Andalusia, Regional Ministry of Economy, Knowledge, Entreprises and University, by the European Regional Development Fund (ERDF), ref. SOMM17/6107/UGR and by Redes Temáticas de Investigación Cooperativa RETIC grant Red SAMID RD16/0022.
Author information
Authors and Affiliations
Contributions
conceptualization, L.J.-F., S.M.-B., M.J.C. and F.J.A.-G; methodology, L.J.-F., S.M.-B., M.J.C. and F.J.A.-G.; formal analysis, L.J.-F.; investigation, L.J.-F., S.M.-B., M.J.C. and F.J.A.-G.; writing—original draft preparation, L.J.-F.; writing—review and editing, L.J.-F., S.M.-B., M.J.C. and F.J.A.-G.; supervision, M.J.C. and F.J.A.-G.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jurado-Fasoli, L., Mochon-Benguigui, S., Castillo, M.J. et al. Association between sleep quality and time with energy metabolism in sedentary adults. Sci Rep 10, 4598 (2020). https://doi.org/10.1038/s41598-020-61493-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-61493-2
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.