Abstract
Crustacean eggs are rare in the fossil record. Here we report the exquisite preservation of a fossil polychelidan embedded within an unbroken nodule from the Middle Jurassic La Voulte-sur-Rhône Lagerstätte (France) and found with hundreds of eggs attached to the pleon. This specimen belongs to a new species, Palaeopolycheles nantosueltae sp. nov. and offers unique clues to discuss the evolution of brooding behaviour in polychelidan lobsters. In contrast to their development, which now relies on a long-lived planktic larval stage that probably did not exist in the early evolutionary steps of the group, the brood size of polychelidan lobsters seems to have remained unchanged and comparatively small since the Jurassic. This finding is at odds with reproductive strategies in other lobster groups, in which a long-lived planktic larval stage is associated with a large brood size.
Similar content being viewed by others
Introduction
Pleocyemata (including crabs, lobsters, crayfishes, caridean shrimps, polychelidan lobsters and others) is a group of decapod crustaceans characterised by a specific reproductive strategy, in which females brood their eggs on their abdominal appendages, instead of releasing them directly into the ocean.
The development of modern polychelidan lobsters (Polychelida), a group of deep-sea dwelling Pleocyemata1,2,3,4 involves a long-lived, giant, balloon-like larval stage, the eryoneicus, which feeds within the plankton. In contrast to other Pleocyemata with a long-lived larval stage5, polychelidan lobsters have a low number (hundreds) of small eggs (Figs. 1 and 2). Although polychelidan lobsters have a long fossil record dating back to at least the Triassic6, we lack key information on the evolution of their reproductive modes. There is no evidence of eryoneicus larvae in the fossil record probably until at least the Late Cretaceous7. In contrast, there are multiple reports of immature adult-looking specimens from the Jurassic3,8,9 (Fig. S1), suggesting that the development of polychelidan lobsters did not rely on a long-lived planktic stage in Jurassic waters.
Comparative brood and development strategies in some extant lobsters (Decapoda). (A) Achelata Scholtz & Richer, 1995, eggs, phyllosoma (by C. Jauvion after Haeckel 1899), adult (by D. Audo); (B) Astacidea Latreille, 1802, eggs, third zoea (by C. Jauvion after Herrick 1911), adult (by D. Audo); (C) Polychelida Scholtz & Richer, 1995, eggs, eryoneicus (by C. Jauvion after Bouvier 1917), adult (by C. Jauvion after Hickson 1893).
Modern ovigerous polychelidan lobsters; (A,B) Stereomastis auriculata (Bate, 1878), MNHN-IU-2016-9911; (A) dorsal view; (B) ventral view, 553 distinguishable eggs in this photograph; (C,D) Polycheles enthrix (Bate, 1878), MNHN-IU-2018-4209: (C) dorsal view; (D) ventral view, 389 distinguishable eggs in this photograph. Scale bars: 20 mm. Photographs: L. Cazes.
Although a few fossils of arthropods with preserved egg clutches have been described10,11,12,13,14,15,16,17, no fossil of a decapod crustacean with its eggs preserved has been reported so far, despite the discovery of several tens of well-preserved fossil specimens18. As a result, the number and size of eggs of Jurassic polychelidan lobsters was unknown, making it difficult to evaluate if the emergence of the eryoneicus larvae was accompanied by a change in fecundity.
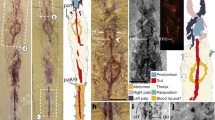
Here, we report the discovery of a unique specimen of polychelidan exquisitely preserved with its eggs within an unbroken nodule from the Middle Jurassic La Voulte-sur-Rhône Lagerstätte, France (Fig. 3).
Palaeopolycheles nantosueltae, MNHN.F.A58254; (A) unbroken nodule showing arms of brittle stars; (B–F) 3D model; (B) nodule with the specimen and associated brittle stars visible; (C) dorsal view; (D) ventral view; (E) left lateral view; (F) left lateral view, with the shield removed. a1, antennula; a2, antenna; cs, cardiac stomach; eg, eggs; en, uropodal endopod; ex, uropodal exopod; hp, hepatopancreas; o, eye; P1-5, pereiopods 1–5 (thoracopods 4–8); pl, pleon; pl1-5, pleopods 1–5; ps, pyloric stomach; sc, scaphocerite; sh, shield; t, telson. Scale bars: 10 mm. Photograph: P. Massicard.
Results
X-Ray microtomography allowed the discovery and detailed reconstruction of anatomical features without damaging the sample. This is the sole representative of a new species. The investigated specimen displays a low number (n = 459) of relatively small-sized eggs under its pleon (abdomen), attached to the pleopods (swimming legs; Figs. 3 and S2). In addition to its eggs preserved in biological position, this specimen also displays a spermatheca, a specific organ used for sperm storage (Fig. 4D) present in modern Decapoda. All the mouthparts, the hepatopancreas (digestive gland), and the digestive tube (e.g. cardiac and pyloric stomachs; Fig. 3) are also preserved.
Modern eggs and spermatheca from polychelidans (A,B) and comparison with Jurassic Palaeopolycheles nantosueltae (C,D); (A) modern polychelidan eggs, Stereomastis galil Ahyong & Brown, 2002, MNHN-IU-2016.991; (B) modern spermatheca, Polycheles coccifer Galil, 2000, MNHN-IU-2018–4214; (C) close-up of preserved eggs; (D) close-up of preserved spermatheca; (E) Egg size and shield length of modern Polychelida and of Palaeopolycheles nantosueltae. 10 eggs were measured per specimen, error bars represent standard deviation. The egg size is not statistically different from that of MNHN-IU-2008-10470 (Stereomastis helleri; Wilcoxon-Mann-Whitney test, U = 38, p = 0.38396, N = 10). Scale bars: 5 mm in A and C; 2 mm in B and D. Photographs: L. Cazes.
The position of eggs (under the pleon, carried by pleopods 1–5), their number (n = 459) and the size range (0.7–0.9 mm) are remarkably similar to those of modern polychelidan lobsters (Figs. 4 and S1). These clues suggest that Jurassic and extant polychelidan lobsters had a comparable reproduction mode and a relatively small brood size compared with other Pleocyemata. For comparative purposes, a new database of egg sizes in modern polychelidan lobsters was generated (Table S2).
Systematic palaeontology
Eucrustacea Kingsley, 1894
Decapoda Latreille, 1802
Pleocyemata Burkenroad, 1963
Polychelida Scholtz & Richter, 1995
Palaeopolycheles Knebel, 1907
Type species. – Eryon longipes Fraas, 1855, by monotypy (Late Jurassic, Kimmeridgian, Germany).
Palaeopolycheles nantosueltae sp. nov.
Type material. – Holotype by monotypy (MNHN.F.A58254) from the La Voulte-sur-Rhône Lagerstätte, France (Middle Jurassic, Callovian19). Only known specimen of this species.
Etymology. – The specific epithet is referring to Nantosuelta, a Celtic goddess associated with fertility, alluding to the ovigerous state of the holotype.
Palaeopolycheles nantosueltae is ascribed to Polychelida based on the presence of chelate pereiopods 2–4, a concave frontal margin, and a dorsoventrally flattened body (Fig. S3). More precisely, it can be ascribed to Palaeopolycheles by its long anterolateral angle forming a spine orientated forward, ocular incision opening laterally, small cervical and hepatic incisions, short posterolateral angle, pleonite 5 on which the posterior transverse groove intersects the median line, very rounded scaphocerite, and narrow third maxilliped ischium20. Palaeopolycheles nantosueltae differs from the only other known species, Palaeopolycheles longipes (Fraas, 1855), by its shorter antennular peduncle reaching less than half the length of the scaphocerite (almost as long as the scaphocerite in P. longipes) and less curved cervical groove (more curved in P. longipes). These differences, added to the stratigraphic gap (Callovian-Kimmeridgian) separating both species, lead us to consider P. nantosueltae as a distinct species, albeit closely allied to P. longipes.
General discussion
In extant lobsters, a long larval stage generally goes together with large brood (i.e., large number of smaller eggs per clutch), while species that hatch directly as adult-looking individuals generally produce a small number of relatively large eggs (Fig. 1). For instance, spiny and slipper lobsters (Achelata) produce in the range of tens of thousands to almost two million small eggs per clutch and live quite a long time as long-legged planktic larvae (phyllosoma; Table S1). In contrast, marine clawed lobsters and freshwater crayfishes (Astacidea) produce fewer (only a few hundred per clutch) but bigger eggs and display a shorter, more direct development5. Palaeontology and phylogenetics suggest that the ancestral state in decapod crustaceans is a relatively long and direct development pattern, without dramatic changes at each ontogenetic stage8,9,21.
Since the development of polychelidan lobsters incorporated a long-lived, giant planktic larval stage at the end of the Mesozoic7, it is surprising that their brood and egg sizes do not seem to have changed much in 165 Ma. Physiological or environmental constraints may have been at play. There is no data on the relationship between reproductive strategy and environmental conditions in extant polychelidan lobsters; however, the relationship between habitat and brood size has been reported in slipper lobsters. Species of slipper lobsters with a pelagic phyllosoma stage produce relatively small clutches of big eggs associated with a short-lived phyllosoma stage, while species with a pelagic phyllosoma stage produce very large clutches of small eggs associated with a longer-lived phyllosoma stage22. These specific behaviours are directly related to the larval survival rates in both environments, which is higher in coastal settings22, due to a lower dispersion and higher food availability in this environment compared to the open ocean.
The establishment of the long-lived planktic larva (eryoneicus), probably during the Cretaceous7 or after, likely offered the possibility to polychelidan lobsters to survive in the deep sea, while shallow water taxa went extinct1, possibly owing to the evolution of true crabs that likely competed for similar resources and habitats23,24. At the same time, the limited resources of this harsh environment may have been constraining brood size in polychelidan lobsters. Eryoneicus larvae develop higher in the water column than the deep sea, alleviating competition with their parents for food resources on the seafloor25. This giant larval stage thus likely increased the fitness of polychelidan lobsters and allowed their wide geographical dispersal25, despite a comparatively small brood size.
Material and Methods
Specimens
The fossil specimen is embedded within an unbroken nodule (MNHN.F.A58254, Fig. 3A,B) from the La Voulte-sur-Rhône Lagerstätte, Callovian, France, and is housed in the palaeontology collection of the Muséum national d’Histoire naturelle (acronym: MNHN.F). The La Voulte-sur-Rhône Lagerstätte is world renowned for the diversity and quality of its fauna, especially arthropods19,26. Fossiliferous concretions with exceptional preservation come from marls outcropping at the Ravin des Mines locality, which are topped by 15 m of iron carbonate deposits19.
Comparisons were made with extant specimens housed in the zoological collections of the Muséum national d’Histoire naturelle (acronym: MNHN-IU) and to fossil specimens of Palaeopolycheles longipes (Fraas, 1855; Fig. S4) housed in the Staatliches Museum für Naturkunde, Stuttgart (acronym: SMNS). The new database of egg size in modern polychelidan lobsters was generated for comparison from the MNHN collections. 10 egg diameters were measured per specimen (photograph) using ImageJ. Statistical difference between each extant specimen and MNHN.F.A58254 were tested using a Wilcoxon-Mann-Whitney test.
Microtomography
For microtomography, we used the same method as described in Jauvion et al.2. The unbroken nodule was imaged with a v│tome│× 240 L tomograph (GE Sensing & Inspection Technologies Phoenix ×│ray) equipped with a microfocus 240 kV/320 W tube delivering a current/voltage of 220 mA/120 kV. Microtomography was performed at the AST-RX technical platform of the MNHN, Paris. Data were processed to obtain a series of virtual slices with a voxel size (cubic voxel) of 31.4 mm. Virtual slices were saved as a series of image files in 16 bits greyscale indicating differences in absorption of X-ray within the nodule (darker for low absorption, brighter for high absorption). 1900 virtual slices with a resolution of 1340 × 1198 pixels were thus obtained. Outlines of the fossilized structure were segmented using Mimics 20.0 (Materialise) for 3D reconstruction. MeshLab was used for 3D rendering.
Data availability
X-Ray microtomogaphy data (virtual slices) are available here27: https://doi.org/10.5281/zenodo.3624687.
References
Audo, D., Charbonnier, S. & Krobicki, M. Rare fossil polychelid lobsters in turbiditic palaeoenvironments. J. Syst. Palaeontol. 16, 1017–1036 (2018).
Jauvion, C., Audo, D., Charbonnier, S. & Vannier, J. Virtual dissection and lifestyle of a 165 million-year-old female polychelidan lobster. Arthropod Struct. Dev. 45, 122–132 (2016).
Audo, D., Schweigert, G., Saint Martin, J.-P. & Charbonnier, S. High biodiversity in Polychelida crustaceans from the Jurassic La Voulte-sur-Rhône Lagerstätte. Geodiversitas 36, 489–525 (2014).
Audo, D. et al. Palaeoecology of Voulteryon parvulus (Eucrustacea, Polychelida) from the Middle Jurassic of La Voulte-sur-Rhône Fossil-Lagerstätte (France). Sci. Rep. 9, 5332 (2019).
Pollock, D. E. Egg production and life-history strategies in some clawed and spiny lobster populations. Bull. Mar. Sci. 61, 97–109 (1997).
Audo, D., Williams, M., Charbonnier, S. & Schweigert, G. Gabaleryon, a new genus of widespread early Toarcian polychelidan lobsters. J. Syst. Palaeontol. 15, 205–222 (2017).
Haug, J. T. et al. Unique occurrence of polychelidan lobster larvae in the fossil record and its evolutionary implications. Gondwana Res. 28, 869–874 (2015).
Eiler, S. M. & Haug, J. T. Larval development of fossil polychelidan crustaceans, exemplified by the 150 million years old species Palaeopentacheles roettenbacheri. Neues Jahrb. für Geol. und Paläontologie - Abhandlungen 279, 295–310 (2016).
Bravi, S. et al. Middle Jurassic Monte Fallano Plattenkalk (Campania, southern Italy): first report on terrestrial plants, decapod crustaceans and fishes. Neues Jahrb. für Geol. und Paläontologie - Abhandlungen 272, 79–107 (2014).
Caron, J. B. & Vannier, J. Waptia and the Diversification of Brood Care in Early Arthropods. Curr. Biol. 26, 69–74 (2016).
Vannier, J., Thiéry, A. & Racheboeuf, P. R. Spinicaudatans and ostracods (Crustacea) from the Montceau lagerstätte (Late Carboniferous, France): Morphology and Palaeoenvironmental significance. Palaeontology 46, 999–1030 (2003).
Siveter, D. J. et al. Exceptionally preserved 450-million-year-old ordovician ostracods with brood care. Curr. Biol. 24, 801–806 (2014).
Siveter, D. J., Siveter, D. J., Sutton, M. D. & Briggs, D. E. G. Brood care in a Silurian ostracod. Proc. Biol. Sci. 274, 465–469 (2006).
Perrier, V. et al. Syncarid crustaceans from the Montceau lagerstatte (Upper Carboniferous; France). Palaeontology 49, 647–672 (2006).
Damotte, R., Durand, M. & Lethiers, F. Découverte d’une faune d’ostracodes dans la Formation d’Abbé (Permien basal, Vosges). Géologie la Fr. 2, 82–87 (1996).
Lethiers, F., Damotte, R. & Whatley, R. Evidence of brooding in Permian non-marine Ostracoda. Lethaia 29, 219–223 (1996).
Gueriau, P. et al. A 365-million-year-old freshwater community reveals morphological and ecological stasis in branchiopod crustaceans. Curr. Biol. 26, 383–390 (2016).
Charbonnier, S., Audo, D., Garassino, A. & Hyžný, M. Fossil Crustacea of Lebanon. (Publications Scientifiques du Muséum, 2017).
Charbonnier, S., Audo, D., Caze, B. & Biot, V. The La Voulte-sur-Rhône Lagerstätte (Middle Jurassic, France). Comptes Rendus - Palevol 13, 369–381 (2014).
Audo, D. et al. Diversity and palaeoecology of the enigmatic genus Knebelia (Eucrustacea, Decapoda, Eryonidae) from Upper Jurassic plattenkalks in southern Germany. Palaeontology 57, 397–416 (2014).
Haug, J. T. & Haug, C. An unusual fossil larva, the ontogeny of achelatan lobsters, and the evolution of metamorphosis. Bull. Geosci. 88, 195–206 (2012).
Booth, J. D., Webber, W. R., Sekiguchi, H. & Coutures, E. Review: Diverse larval recruitment strategies within the Scyllaridae. New Zeal. J. Mar. Freshw. Res. 39, 581–592 (2005).
Luque, J. The oldest higher true crabs (Crustacea: Decapoda: Brachyura): Insights from the Early Cretaceous of the Americas. Palaeontology 58, 251–263 (2015).
Klompmaker, A. A., Schweitzer, C. E., Feldmann, R. M. & Kowalewski, M. The influence of reefs on the rise of Mesozoic marine crustaceans. Geology 41, 1179–1182 (2013).
Galil, B. S. Crustacea Decapoda: review of the genera and species of the family Polychelidae Wood-Mason, 1874. in In: Crosnier, A. (Ed.) (2000). Results of the MUSORSTOM expeditions: 21. Mémoires du Muséum National d’Histoire Naturelle (Paris), A (Zoologie) vol. 184, 285–387 (2000).
Jauvion, C. et al. Exceptional preservation requires fast biodegradation: thylacocephalan specimens from La Voulte-sur-Rhône (Callovian, Jurassic, France). Palaeontology 1–19, https://doi.org/10.1111/pala.12456 (2019).
Jauvion, C. et al. Data from: A new polychelidan lobster preserved with its eggs in a 165 Ma nodule. https://doi.org/10.5281/zenodo.3624687#.XihfBcw1OpQ.mendeley, 10.5281/ZENODO.3624687 (2020).
Acknowledgements
The authors would like to acknowledge Maïté Adam and Patricia Wils (AST-RX, plateau d’Accès Scientifique à la Tomographie à Rayons X du MNHN, UMS 2700 Outils et méthodes de la systématique intégrative CNRS-MNHN, Paris) for X-Ray acquisition and pre-treatment and Florent Goussard (MNHN, UMR 7207 CR2P) for technical assistance with data reconstruction. We thank Günter Schweigert (SMNS) for access to specimens. Lilian Cazes (MNHN) and Peter Massicard (projet e-recolnat, MNHN, Investissements d’avenir, ANR-11-INBS-0004-RECOLNAT) provided photographs of specimens. DA research was supported by the China Postdoctoral Science Foundation, Yunnan Province Postdoctoral Science Foundation, NSFC grant 41861134032, and Yunnan Provincial Research Grants 2018FA025 and 2018IA073. CJ, SB and SC benefitted from two MNHN grants: ATM Crevoulte (MNHN) and PerSysT (Origin and Evolution Department). We thank Carrie E. Schweitzer and an anonymous reviewer for their constructive reviews.
Author information
Authors and Affiliations
Contributions
C.J. managed the specimen scanning procedure and 3D reconstructions, visited the zoological collections, measured egg sizes, composed the figures and drafted the manuscript. D.A. managed the systematic palaeontology, drafted the corresponding part, provided pictures of Palaeopolycheles longipes and critically revised the manuscript. S.B. and S.C. coordinated the study and critically revised the manuscript. A.C.D. and J.V. critically revised the manuscript. All authors gave final approval for publication and agree to be held accountable for the work performed therein.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jauvion, C., Audo, D., Bernard, S. et al. A new polychelidan lobster preserved with its eggs in a 165 Ma nodule. Sci Rep 10, 3574 (2020). https://doi.org/10.1038/s41598-020-60282-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-60282-1
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.