Abstract
Populations in Mongolia from the late second millennium B.C.E. through the Mongol Empire are traditionally assumed, by archaeologists and historians, to have maintained a highly specialized horse-facilitated form of mobile pastoralism. Until recently, a dearth of direct evidence for prehistoric human diet and subsistence economies in Mongolia has rendered systematic testing of this view impossible. Here, we present stable carbon and nitrogen isotope measurements of human bone collagen, and stable carbon isotope analysis of human enamel bioapatite, from 137 well-dated ancient Mongolian individuals spanning the period c. 4400 B.C.E. to 1300 C.E. Our results demonstrate an increase in consumption of C4 plants beginning at c. 800 B.C.E., almost certainly indicative of millet consumption, an interpretation supported by archaeological evidence. The escalating scale of millet consumption on the eastern Eurasian steppe over time, and an expansion of isotopic niche widths, indicate that historic Mongolian empires were supported by a diversification of economic strategies rather than uniform, specialized pastoralism.
Similar content being viewed by others
Introduction
Mongolian empires, such as the Xiongnu and Mongols, are some of the most renowned imperial entities in public and academic thought. This is, in part, due to their historical portrayal as highly mobile, predatory horseback polities with a specialized dairy and meat-based economy1,2,3,4, an image that is perpetuated in cinema, novels, and documentaries alike. While such stereotypes likely arose from the hyperbolized accounts of neighboring adversaries, starting with the Han, who fought against the Xiongnu5, they have persisted and now pervade academic evaluations of the economic basis of these ancient peoples. The modern economic focus on pastoralism in rural areas of Mongolia today is frequently viewed as a relic of the past and has been drawn upon to interpret the often-fragmentary archaeological record of this region6,7, although ethnoarchaeological approaches often ignore the role of urban markets and motorized transport in modern mobile pastoralism. The view of uniformly specialized pastoral economies has also furthered the scholarly fascination with historical Mongolian populations, resurrecting the long-standing question of whether an empire can meet the costs and challenges of long-term political and economic organization in the absence of grain surpluses6,7.
Empires are, however, inherently complex and, by definition, extend their control over multiple societies, cultures, and economies, as well as heterogeneous landscapes8,9,10. Crop surplus has traditionally been viewed as an essential component of stable political entities and complex imperial food production and procurement systems are often over-simplified by historians and archaeologists, leading to their characterization as single-resource systems (such as maize for the pre-Columbian empires of South America). Refined analyses generally reveal diverse and dynamic economies supporting imperial expansions, which draw together a variety of food sources11. As a consequence, it is perhaps unsurprising that archaeological, archaeobotanical, and historical records are beginning to strongly hint at the possibility that historical Mongolian empires were not solely reliant on dairy pastoralism, but also featured agriculture, as well as craft specialization, and participation in trade systems that spanned thousands of kilometers12,13,14.
Of particular interest in this context has been the growing archaeobotanical evidence from across Central Asia that demonstrates an influx of millet, both broomcorn (Panicum miliaceum L.) and foxtail (Setaria italica L.), and other domesticated grains in the surrounding steppe lands of Siberia, Kazakhstan, and northwestern China during the second and first millennia B.C.E.15,16,17,18,19,20,21,22,23,24. There are some archaeobotanical data suggesting the use of crops in Mongolia starting around c. 100 B.C.E. – 200 CE25,26, though these have been dismissed as reflective of trade rather than local production6,27. Overall, due to issues of wind deflation and a lack of sampling during excavation, archaeobotanical evidence from Mongolia is severely lacking. Moreover, where present, it is difficult to determine the degree to which an archaeobotanical assemblage represents overall dietary reliance. To date, there have been no systematic, direct analyses of the consumption of domesticated crops among peoples in this region over the past three millennia, leaving the economic basis for some of the world’s most famous empires unresolved.
Stable isotope analysis of archaeological human and associated faunal remains has emerged as an increasingly powerful methodology for tracking palaeodietary and subsistence change in Central and East Asia23,28,29,30,31. The distinction in stable carbon isotope ratios (δ13C) between C3 plants on the one hand – including crops such as rice (Oryza sativa L.), wheat (Triticum spp. L.) and barley (Hordeum vulgare L.) – and C4 plants on the other – including millets and maize (Zea mays L.) – can be tracked through the bone collagen and bioapatite of humans relying on these resources and/or the animals feeding off of them32. While bone collagen δ13C is primarily derived from the protein portion of the diet, tooth enamel bioapatite δ13C reflects the whole diet33. Stable nitrogen isotope (δ15N) analysis provides additional insights into the trophic level position of an individual, orienting them within the local food chain34.
It is our aim to discover when Eastern Steppe populations began utilizing cultivated C4 resources (i.e. millet and millet-based foods). We are especially interested in dietary trends during the Xiongnu and Mongol imperial periods, as there has long been a dominant assumption that these empires wholly depended on dairy pastoralism. We use stable carbon isotope analysis of human tissues to directly test whether, in line with some previously published archaeobotanical and historic evidence, the Xiongnu and Mongol empires in fact relied quite significantly on millet-based agricultural systems. We present δ13C and δ15N analysis of human bone collagen and δ13C and δ18O analysis of human tooth enamel bioapatite from 137 previously-excavated individuals from across Mongolia dated to between c. 4400 B.C.E. and 1375 C.E. in order to directly assess changing diets through the region’s key imperial transitions.
Results
Preservation of samples
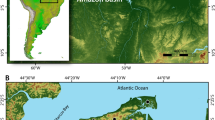
We analyzed 80 bone collagen and 108 dental enamel samples from 137 individuals from 60 archaeological sites (Tables 1–4; Fig. 1). Samples were separated into four chronological periods based on relative and absolute dating (Early [Neolithic - Bronze Age], Early Iron, Xiongnu, and Mongol; see Supplementary Table S4 for AMS dates). As there is only a single individual from the Neolithic period (c. 4400–3000 B.C.E.), this sample was combined with Bronze Age individuals dating to prior to 800 B.C.E. (n = 23; collagen n = 14, enamel n = 16) to create a single period labelled as ‘Early’. The Iron Age samples were split into two chronological periods, corresponding to the pre-imperial Early Iron Age (c. 800–200 B.C.E.) and the Xiongnu (c. 200 B.C.E. – 250 C.E.). The Early Iron Age samples include 16 individuals (collagen n = 7, enamel n = 16) from one site. From the subsequent Xiongnu, we analyzed 59 individuals (collagen n = 23, enamel n = 54) from 28 sites. Individuals from the later Mongol Empire (c. 1200–1375 C.E.) are grouped together and consist of 28 individuals (collagen n = 28, enamel n = 21) from 19 sites.
Maps of sites used in this study. These maps were created for this study and were produced using QGIS 3.089 https://qgis.org/en/site and using the Natural Early Data maps from https://www.naturalearthdta.com/downloads/ by Shevan Wilkin and Michelle O’Reilly (Graphic Designer for the Max Planck Institute for the Science of Human History, Jena, Germany).
All of the human bone collagen samples included in this study had atomic C:N ratios between 3.1 and 3.5 and were thus within the accepted range for good collagen preservation35 (Supplementary Table S1). The collagen yields of these samples ranged between 6 and 30%, with none falling below 1%, a further check of data quality35. Furthermore, the majority of collagen samples have greater than 11% N and greater than 30% C, within acceptable ranges36. Each bone collagen sample was run in duplicate, and averages are presented in Supplementary Table S1 along with their standard deviation.
Bone collagen carbon and nitrogen stable isotope results
δ13C and δ15N results from human bone collagen are grouped into four chronological periods, as detailed above, for comparative analysis. The pre-Bronze and Bronze Age average values are the closest to those of the average faunal values, although the faunal values have a higher standard deviation. The widest range of carbon and nitrogen isotope values were found in the Xiongnu and Mongol Period populations. For all of the individual δ13C and δ15N values from each time period see Supplementary Table 1.
Dental enamel carbon stable isotope results
The data from δ13C values of human enamel bioapatite are divided into the same chronological periods as the bone collagen data. The Xiongnu population had the largest range of stable carbon isotope values, followed by the Mongol period and Early Iron Age (Table 2). The pre-Bronze/Bronze period had the lowest range of stable carbon isotope values when compared to the later populations. For all of the individual δ13C and δ18O values from each time period see Supplementary Table 2 (Samples with both collagen and enamel Supplementary Table 3).
Environmental differences
As stable carbon and nitrogen isotope values may vary in different environments (i.e. temperature and aridity), to adequately assess human δ13C and δ15N values from normal steppe (>200 mL of annual precipitation) and dry (<200 mL of annual precipitation) regions, we also determined the average values for each environmental type (Table 3). In these tables we have separated the previously published faunal stable carbon and nitrogen isotope values into the “steppe” or “dry” regions according to modern annual rainfall37,38.
Statistical tests
Boxplots of our results can be found in Fig. 2A–C, and statistical comparisons between the time periods can be found in Supplementary Table 5 (δ13C bone collagen), Supplementary Table 6 (δ13C enamel bioapatite), and Supplementary Table 7 (δ15N bone collagen). For the bone collagen data, both the Xiongnu and the Mongol average δ13C values were significantly higher than those of Early individuals (p < 0.05). The same trend was seen for tooth enamel δ13C, with Early Iron, Xiongnu, and Mongol samples having δ13C significantly higher than that of the Early group (for the overall p < 0.05, and the specific pairwise comparisons are available in Supplementary Table 6). There was no significant difference between average dental enamel values for the Early Iron, Xiongnu, and Mongol periods δ13C (p > 0.05). Bronze Age δ15N values were also significantly higher (p < 0.05) than those of the Early Iron, Xiongnu, and Mongol periods (Supplementary Table 7).
Boxplots showing the range of carbon values for all individuals from each period. Outliers are shown as individual data points. (A) Comparison of the bone collagen carbon values for humans and fauna. Faunal data derives from previously published data20,29,30,44, and all human data is from this study. (B) Difference in human enamel values between Early, Early Iron, Xiongnu, and Mongol periods. The Early Iron, Xiongnu, and Mongol period average values are significantly higher than the Early period average. (C) Boxplots showing the range of oxygen values from enamel samples. There are no significant differences between any of the time periods.
Isotopic temporal trends in Mongolia and environmental impacts
Higher δ13C values in individuals from the Early Iron Age, Xiongnu, and Mongol periods could be the product of the increased direct consumption of C4 crops or wild plants or animals consuming C4 plants. It should also be understood that both Mongolians and foreign travellers would have been moving within and outside of the imperial borders, and dietary intake likely varied greatly in different regions. In areas with environmentally-linked variation in wild C4 and C3 plant distributions, such as Mongolia, it is important to rule out a climatically driven change (see Supporting Information Text 1). Modern plant samples from Mongolia have yielded δ13C values ranging from −28.3 to −23.4‰ for C3 photosynthetic pathways and an average δ13C of −14.7‰ for plants following the C4 photosynthetic pathway39. Notably, wild C4 plants make up a much smaller proportion of Mongolian and other Central Asian environments than C3 plants40,41. Overall, contemporary studies suggest that leaf δ13C values decrease with increasing mean annual precipitation42, both as a product of reduced C4 plants in wetter landscapes and aridity-driven changes in δ13C among C3 plants (see Supplemental Text S1).
While C4 plants make up a relatively limited portion of the biotic community today, we established local isotopic baselines for Mongolia in the past using archaeological fauna in order to determine if shifts in δ13C values through time are the product of environmental variations or social and economic choices. Isotopic studies of modern and archaeological herd animals have shown differences in δ13C values between more and less arid regions43,44,45, and that there is variation in the availability of C3 and C4 plants across the country46,47. While there were no fauna associated with the human remains collected for this study, we were able to use previously published faunal stable isotope data from the Minusinsk Basin of Siberia (just north of Mongolia)(MNSK, AD, AM; n = 21)20,29, the Gobi (BGC; n = 14)30,48, Gobi-Altai (SBR; n = 5)30, and north central Mongolia (EG; n = 13)30 areas to show that regional herbivores generally consumed C3 plants, with some having higher stable carbon isotope values, indicative of C4 plant consumption, in the hyper-arid desert regions30,48.
Statistical tests further support this assessment, with humans having higher δ13C values than the available fauna in all periods, with the greatest difference occurring in the Xiongnu and Mongol periods (p ≤ 0.005)(Figs. 2A and 3A). For terrestrial faunal remains (Fig. 3A), there is a significant correlation between δ15N and δ13C bone collagen values (R2 = 0.64, p-value <0.001) which is a product of higher levels of aridity leading to a higher availability of C4 plants in the natural vegetation cover. However, no such correlation is observed in humans (Fig. 3A,B), either between δ15N and δ13C bone collagen values (R2 = 0.01, p-value = 0.15) or between δ15N bone collagen and δ13C enamel values (R2 = 0.05, p-value = 0.13). Given this, alongside the consistent elevation of human δ13C values over the available fauna δ13C values, this indicates that higher δ13C values in human bone collagen and enamel is a product of direct consumption of non-wild C4 plants.
Carbon and nitrogen values from bone collagen with ellipses showing ranges at 95% confidence. (A) Individuals included in this study as well as humans and faunal values from previously published data31 (B) Humans included in this study showing the variation between those in the “Dry” and “Steppe” zones. “Dry” sites have less than 250 mm of annual precipitation, “Steppe” sites have over 250 mm of precipitation per year. (C) δ15N from bone collagen versus δ13C values from dental enamel demonstrating the shift from primarily C3 reliant diets in the Early period to a wider range of carbon and nitrogen values, indicating an increase in the diversity of diets in the later three periods. (D) δ18O from dental enamel versus δ15N from bone collagen showing the values in “Dry” and “Steppe” areas.
Mean bone collagen δ13C values for faunal remains from steppe regions are typically C3 (−19.3 ± 1.3‰), and the stable carbon isotopic offset between bone collagen of herbivores and carnivores is c. 1‰49. Thus, human bone collagen steppe samples dating to the Early period (prior to 800 B.C.E.) do not show δ13C values indicative of a millet dietary contribution (−18.3 ± 0.1‰). This same offset applies to human bone collagen samples from dry regions from all periods since these are elevated by up to 1‰ (Early −17.2 ± 0.7‰; Early Iron = −16.2 ± 0.9‰; Xiongnu −16.1 ± 1.1‰; Mongol −16.1 ± 1.4‰) when compared to the bone collagen mean for faunal samples from dry regions (−17.2 ± 1.5‰). However, for later time periods in steppe regions average human bone collagen δ13C values are elevated by c. 3‰ (Early Iron −16.3 ± 0.7‰; Xiongnu −16.1 ± 1.2‰; Mongol −16.8 ± 2.2‰) when compared to faunal values. Thus, the higher (c. 2‰) human bone collagen δ13C values observed for later periods in steppe regions when compared to the early period is indicative of a temporal increase in millet-based food consumption. Furthermore, given that bone collagen reflects primarily consumed protein, and that millet has a poor protein content, the dietary caloric contribution from millet was likely much higher than its protein contribution50. This is corroborated by human enamel δ13C values given that this isotopic proxy reflects the carbon dietary mix50. Steppe human enamel samples for the later periods show mean δ13C values higher by c. 3.5‰ when compared to the Early period. For dry areas, mean human enamel samples δ13C values are higher by c. 1‰ when compared to the Early period, which indicates a temporal increase in millet-based food consumption although considerably smaller than that observed in the steppe regions as shown also in the model estimates for millet caloric contributions (Fig. 4).
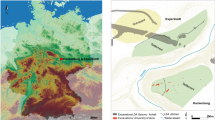
Sites in and around Mongolia with archaeological or archaeobotanical evidence for C3 (wheat and barley) and C4 (broomcorn and foxtail millet) grain cultivation during the Iron Age. This map was newly created for this study and produced using QGIS 3.089 https://qgis.org/en/site and using the Natural Early Data maps from https://www.naturalearthdta.com/downloads/ by Shevan Wilkin, Bryan K. Miller, and Michelle O’Reilly (Graphic Designer for the Max Planck Institute for the Science of Human History, Jena, Germany).
Further evidence for C4 plant consumption is offered by the distribution of isotopic values. For faunal remains there is a positive significant correlation (R2 = 0.67, p-value <0.05, correlation coefficient =1.126) between δ15N and δ13C bone collagen values which is expected given that an increase in aridity leads to a higher availability of C4 plants in the vegetation cover. A similar correlation, albeit with isotopic offsets, would be expected if humans relied exclusively on animal products. However, no clear environmentally-driven correlation is observed for the human groups. There is no significant correlation for the Early (R2 = 0.65, p-value <0.08, correlation coefficient = 0.65) and Xiongnu (R2 = 0.0, p-value <0.46, correlation coefficient = 0.11) periods, and although the correlation is significant for the Mongol period (R2 = 0.13, p-value <0.02, correlation coefficient = 0.39), it only explains 39% of the variability. For similar δ15N bone collagen values across the human individuals, there are wide ranges in δ13C collagen values. Whereas during the Xiongnu period one can observe a significant negative correlation (R2 = 0.0, p-value <0.46, correlation coefficient = 0.11), which implies the contribution from a food source with higher δ13C values but lower δ15N values when compared to animal food sources. These isotopic relationships are indicative of varying individual intake of a food with elevated δ13C values, such as millet, and having relatively uniform δ15N and δ13C values across regions with varying levels of aridity.
Bayesian spatial modelling of C4 plant caloric consumption
To further confirm that the increased δ13C values in human bone collagen and tooth enamel through time is a product of the consumption of crops rather than changing availabilities of baseline C4/C3 plant ratios or the availability of samples in different local environments, we developed a Bayesian model to produce a C4 dietscape, representing estimates of spatial distribution of C4 plants based on per capita caloric consumption (See SI for detailed discussion). Stable carbon isotope data of dental enamel was used, and individuals were separated into two periods, Early (Neolithic - Bronze Age) or Late (Xiongnu, and Mongol). The results for the two models show that during the Early period C4 caloric contributions were very low across Mongolia, likely including consumption of local plants and livestock consuming natural vegetation, with mean estimates varying between c. 2.5 and 5.0% of calories (interpolation 1-sigma uncertainty up to 0.5% calories) (Fig. 5A,B). During the later periods, the variability in millet-based food consumption increases considerably as shown by the range in the mean estimate (between 3 and 26% of per capita millet calories) and in the 1-sigma interpolation uncertainty for each location (between c. 3 and 6% of per capita millet calories) (Fig. 5C,D). The C4 plant dietscape for the late period also shows that millet consumption is concentrated in central northern Mongolia (reaching the highest mean value [26% per capita millet calories]), an area where environmental increase of carbon values would not be expected naturally (Figs. 4 and 5).
Dietscape representing average millet caloric consumption per capita. (A) Average millet caloric consumption for the Early period estimated through Bayesian modelling of dental bioapatite carbon stable isotope values. (B) Standard error of the mean for millet caloric consumption average during the Early period. (C) Average millet caloric consumption for the Late period estimated through Bayesian modelling of dental bioapatite carbon stable isotope values. (D) Standard error of the mean for millet caloric consumption average during the Late period.
Discussion
Isotopic indicators of diet through time in mongolia
Our results clearly demonstrate an increase of human consumption of C4 plants during the imperial periods in ancient and historic Mongolia (Figs. 2, 3, and 5). While high δ15N values in human bone collagen relative to the faunal data (Fig. 2A and 3A) supports evidence for human reliance on dairy and meat products throughout the periods under study, the change in C4 plant consumption represents the major dietary shift within this timeframe. The significant decrease in δ15N values in the later periods, in comparison to the Bronze Age, further supports this point, potentially indicating reduced consumption of meat and milk and increased consumption of grains. Moreover, comparisons with faunal datasets and environmental background data allows us to confidently state that this shift is a consequence of increasing rates of consumption of C4 resources. Stable carbon isotope values from individuals before the Early Iron Age show little indication of C4 plant consumption beyond the local natural baseline identified through the fauna from similar environments. Individuals from the cemetery of Chandman Mountain (c. 900–400 B.C.E.) analyzed in this study show the first visible evidence of C4 plant consumption as part of a mixed agro-pastoral diet. However, this particular site in northwest Mongolia is more a part of the Minusinsk Basin region of southern Siberia, an area where millet consumption was common by the Late Bronze Age, than the rest of Mongolia. Previously published contemporaneous human and faunal isotope analyses within this region are indicative of animals consuming primarily C3 plants20, with the human population postulated as having a diet additionally composed of C4 plants in concert with meat and milk.
In the rest of Mongolia during the tenure of the Xiongnu and Mongol empires human stable carbon values became more varied with increasing numbers of individuals displaying bone collagen and tooth enamel δ13C values suggestive of moderate to high C4 plant consumption, with the number of individuals with such values reaching their peak during these imperial periods. We also observe the largest range and diversity of δ13C and δ15N values during the imperial periods. This is likely due to diverse subsistence strategies being pursued across each empire, reflecting different environmental zones and levels of imperial support. This is result of the extensive range of each empire, and includes the knowledge that not everyone that died in Mongolia would have been “Mongolian”, but these individuals likely lived and died within the empires. Since the majority of the individuals analyzed were excavated from elite imperial tombs, and human remains representing other sectors of society are lacking at present, attributing all outliers to non-local outsiders would be to dismiss the agency of Mongolian populations and provide something of a ‘colonial’ narrative.
Individual bone collagen and tooth enamel δ13C values for the Xiongnu and Mongol empires range between those indicative of a pure C3 diet to those that suggest heavy C4 plant consumption. Interestingly, during this period, a few individuals had δ13C values lower than those of the Early period which, alongside lower δ15N values, indicates a staple intake of C3 plants, likely crops such as wheat and barley. Historical and archaeobotanical sources suggest that cereal crops were commonly cultivated or obtained through trade during the Mongol period13,51,52,53,54,55,56. In addition to grains, carbonized fruit and nut remains have been recovered from sediments at the Mongol capital of Kharakhorum (also used during the Mongol rule in the Yuan Dynasty) showing the diversity of imported plants through the presence of rice (Oryza sativa L.), over a dozen cultivated fruits, including grapes (Vitis vinifera L.), figs (Ficus carica L.), and jujube (Ziziphus jujube Mill.), as well as vegetable and oil-seed crops. There are also remains of spices – notably a few, such as black pepper (Piper nigrum L.) and caraway (Carum carvi I.), that were imported along the trade routes with South Asia, and would have involved transport across distances of up to 2000 kilometers12.
The resulting bone collagen δ13C and δ15N values have been plotted to show this increase of dietary diversity over time (Fig. 2A). From our data, alongside the growing corpus of biomolecular, archaeological, and historical data, it is evident that the Xiongnu and Mongol Empires had complex imperial structures that facilitated increasingly diverse subsistence economies. The combination of crop cultivation in tandem with dairy pastoralism would have allowed these empires to sustain a diverse economic surplus that defended against livestock depletion from harsh winters, crop loss, or volatile political episodes. Diverse dietary values likely also reflect an increasingly cosmopolitan society in which dietary heterogeneity within populations increased with growing migration, trade and interaction, and the emergence of increasingly elaborated elite statuses. The diversity could also reflect temporal political shifts within the time-span covered by our sampling groups, with trade routes to Karakorum decreasing in volume during the Mongol Period after the switch of the capital in 1260 and ending with the end of the Yuan Dynasty in 136812,57, for example.
Mongolian empires in context
Historical and ethnographic research indicates the importance of pastoralism on the historic and proto-historic eastern Eurasian Steppe. Recent proteomic research has demonstrated the clear importance of dairy-based pastoralism to Mongolian dietary practices from at least c. 1500 B.C.E.58. Nevertheless, our data clearly highlight that pastoral lifestyles did not preclude the inclusion, and later intensification, of crop use. Millet’s suitability to arid environments combined with its short growing period is compatible with the often peripatetic, mobile lifestyles of pastoralists22,26. Indeed, during the Xiongnu and Mongol empires, we see clear evidence for human dietary reliance on millet in a significant proportion of individuals. Although some scholars contend that all grains were either extorted or imported from China and other exterior polities6, we argue that our data, alongside existing archaeobotanical and archaeological findings25,59,60, provide clear evidence for imperial reliance on locally grown crops in the Xiongnu and Mongol heartlands, as well as the coordination of diverse economic connections and exchanges12. These discoveries bolster the notion of an economically diverse population across much of Mongolian history14,61,62.
Agricultural tools for plowing, hoeing, and grinding have been uncovered from permanent Xiongnu settlements in Mongolia, implying local plant cultivation and processing63, and charred remains of millet, barley, and wheat grains have been recovered through flotation at pit-house villages at Boroo64 and ephemeral campsites61. Studies in the Egiin Gol valley and at the large site complex of Ivolga have illustrated the presence of long-season cereal crops (wheat and barley) in the Iron Age, which represent more labor investment in farming practices than millets61,63. At Ivolga, this occurs alongside evidence of ploughshares at permanent settlements63, as well as written accounts of crops suitable for the northern steppe being managed by imperial Xiongnu administrators, such as the ‘Lord of Millet Distribution’, referred to in the 1st century C.E. Chinese accounts54. Millet grains, still articulated in their chaff, have been found within the graves of Xiongnu rulers at Gol Mod and Noyon Uul27 as well as of local elites throughout the steppe60. There are also uncharred grains found within Xiongnu pit-house villages, all of which were unprocessed (i.e. with palea and lemma) and thus most likely not transported long distances60 instead representing local production and consumption (Fig. 4).
Scholars working in Mongolia have extensively discussed the formation of hierarchical political systems and greater concentrations of population densities in the absence of farming, often describing imperial systems in Central Eurasia as unique due to their economic basis65,66,67,68,69. In other parts of Asia, farming is linked to demographic expansion and the congregation of greater population densities70. Notably, millet farming is linked to urbanization71,72 and imperial formation73 in East Asia. Boserupian economics suggest that increased investment in farming, along with a diversified economy and higher levels of cultural exchange, often lead to a demographic transition74,75,76. The data presented in this paper suggests that, while Mongolian empires have often been seen as outliers in global comparisons of imperial structures, they were in fact, like many others around the world, highly reliant on economic diversification, local adaptations to a diversity of environments, and the creation of reliable and stable subsistence resources and economic surpluses8,9,10.
Mongolian empires have traditionally conjured up exotic ideas of mobile pastoral specialists who roamed the Asian steppes attacking more sedentary communities1,2,3,4. While prominent in the public sphere, such preconceptions have also directed the type of questions academics have asked. For example, comparative analysis of Mongolian empires with others around the world has been limited, with ‘Steppe Empires’ often portrayed as deficient or somehow doomed to failure in the absence of reliable crop-based surplus6,77. As in other parts of Central Asia, where occupation sites have been hard to come by78,79, simplistic projections of ethnographic and ethnohistoric datasets into the past have been common in Mongolian archaeology. We hope to have demonstrated how multidisciplinary approaches, built on datasets from different parts of Mongolian imperial networks, can begin to provide novel insights into their economic systems and, perhaps most importantly, their geographic and temporal variability. While there is no doubt that the Xiongnu and Mongol empires were unique, they were also built upon many of the same tenants of economic diversity, stability, and reliability that have characterized imperial structures throughout prehistory and history, demonstrating the importance of a core set of underlying variables in both enabling and driving the formation of empires.
Methods and Materials
Sites and materials analyzed
All bone and tooth samples included in this study were collected from the National University of Mongolia’s Department of Archaeology during the winter of 2016. Bone collagen was analyzed for carbon (δ13C) and nitrogen (δ15N) stable isotopes, and the carbonate of dental enamel bioapatite was measured for carbon stable isotopes (δ13C), with some individuals analyzed for both bone collagen and bioapatite (see Supplementary Table 3). Time periods for samples ranged from the mid-fifth millennium B.C.E. to the Mongol Empire, as dated by AMS radiocarbon methods where possible (see below).
Samples of bones and teeth were collected from archaeological sites across the country of Mongolia, through varying environmental and topographical zones. Where possible, we collected a tooth and long bone fragment from individuals from each time period. While we aimed to assemble an equal number of samples from all time periods, the collection was dominated by individuals from the imperial periods, resulting in fewer individuals prior to the Iron Age. Bone collagen was preferably extracted from rib bones, but occasionally other bone fragments were employed (clavicle, femur, crania). δ13C and δ15N stable isotope measurements of human bone collagen inform primarily on protein source80, and the bones sampled (i.e. ribs) represent a period of diet of approximately the last 20 years of life80.
By contrast, tooth enamel δ13C values are indicative of the whole dietary carbon (carbon mix of protein, lipids, and carbohydrates) consumed during enamel formation33,49. First molars mineralize before an individual is 3 years old, second molars are fully formed around age 8, and third molars, if present, are completely mineralized between the age of 7 and 1681. To avoid tooth samples that might show a breastfeeding isotopic signal contribution in older children and adolescents we preferentially selected second and third molars. First molars were chosen only when both the M2 and M3 were unavailable.
Stable isotope analysis methods
Bone collagen
We selected ribs for bone collagen analysis as representative of the last c. 20 years of life80. Collagen was extracted from each rib sample following standard procedures35. Approximately 1 gram of pre-cleaned bone was demineralized in 10 ml aliquots of 0.5 M HCL at 4 °C, with changes of acid until CO2 stopped evolving. The residue was then rinsed three times in deionized water before being gelatinized in pH 3 HCl at 75 °C for 48 hours. The resulting solution was filtered, with the supernatant then being lyophilized over a period of 24 hours.
After calculating the collagen yield, all purified collagen samples (~1 mg) were located in tin capsules to be analyzed in duplicate at the Department of Archaeology, Max Planck Institute for the Science of Human History by the elemental analyzer/continuous flow isotope ratio mass spectrometry (EA-IRMS) using a ThermoFisher Elemental Analyzer coupled to a ThermoFisher Delta V Advantage Mass Spectrometer via a ConFloIV system. δ13C and δ15N values were compared and calibrated against International Standards (USGS40 (δ13Craw = −26.4 ± 0.1‰, δ13Ctrue = −26.4 ± 0.0‰, δ15Nraw = −4.4 ± 0.1‰, δ15Ntrue = −4.5 ± 0.2‰), IAEA N2 (δ15Nraw = +20.2 ± 0.1‰, δ15Ntrue = +20.3 ± 0.2‰), IAEA C6 (δ13Craw = −10.9 ± 0.1‰, 13Ctrue = −10.8 ± 0.0‰) Replicate analysis of an in-house fish gelatin standard suggests that machine measurement error is c. 0.1‰ for δ13C and 0.3‰ for δ15N.
Tooth enamel
Teeth or tooth fragments were cleaned using air-abrasion to remove any adhering external material. 8 mg of enamel powder was obtained using gentle abrasion with a diamond-tipped drill along the full length of the buccal surface or fragment in order to maximize the period of formation represented by the resulting isotopic analysis for bulk samples. Enamel powder was pre-treated using a protocol to remove any organic or secondary carbonate contaminates21. This consisted of a series of washes in 1.5% sodium hypochlorite for 60 minutes, followed by three rinses in purified H2O and centrifuging, before 0.1 M acetic acid was added for 10 minutes, followed by another three rinses in purified H2O (as per35).
Following reaction with 100% phosphoric acid, gases evolved from the samples were analyzed to stable carbon and oxygen isotopic composition using a Thermo Gas Bench II connected to a Thermo Delta V Advantage Mass Spectrometer at the Max Planck Institute for the Science of Human History, Jena (MPI-SHH). Carbon and oxygen isotope values were compared against an International Atomic Energy Agency (NBS 19) and in-house standard (MERCK). Replicate analysis of internal bovid enamel standards suggests that machine measurement error is c. ± 0.2‰ for δ13C and ± 0.2‰. Using a Thermo Gas Bench 2 in tandem with a Thermo Delta V Advantage Mass Spectrometer at MPI-SHH, gases produced from a reaction with 100% phosphoric acid were analyzed for stable carbon and oxygen isotopic composition. We compared the resulting values against International Standards (IAEA-603 (δ13C = 2.5; δ18O = −2.4); IAEA-CO-8 (δ13C = −5.8; δ18O = −22.7); USGS44 (δ13C = −42.2)); as well as an in-house standard of (MERCK (δ13C = −41.3; δ18O = −14.4)). The data from these standards suggest that the machine measurement error is c. ± 0.1‰ for δ13C and ± 0.2‰ for δ18O. We increased the precision of our analyzed samples by measuring repeats of extracts using a tooth enamel bovid standard (n = 20, ± 0.2‰ for δ13C and ± 0.3‰).
Statistical tests
To determine whether the differences in human δ13C between each period were significant, we performed a Wilcoxon rank sum test, with multiple test correction using the Benjamini-Hochberg procedure. All tests were performed using the free R statistical software82.
Bayesian dietary modelling
Caloric estimates of millet intakes were obtained using the Bayesian mixing model FRUITS having as input data individual tooth enamel δ13C values and local food isotopic values adjusted for spatial variations due to varying environmental conditions80. To achieve the latter, we grouped site locations into the categories of “steppe” and “dry” depending on modern day annual precipitation. Steppe sites have a range from 250–350 mm in precipitation per annum and arid sites have below 250 mm of yearly rainfall. It was assumed that the enamel δ13C signal is defined by the dietary carbon mix50. To extrapolate the spatial distribution of per capita millet caloric intakes (dietscape) a Bayesian additive mixed model with error-in variables83,84,85 available as an online app via the Pandora & IsoMemo initiatives was employed86. Dietscapes were generated for two main periods corresponding to a temporal divide defined by the intensification of millet consumption as observed from the interpretation of raw isotopic data, into Early (Bronze Age) and Late (combining the Early Iron Age; Xiongnu; Mongol periods). Modelling at a higher chronological resolution was not possible given a lack of data for shorter time periods. Further details on dietscape modelling are available in Supplementary Text S3.
Radiocarbon and archaeologically classified dates
AMS radiocarbon dates were conducted at the Oxford Radiocarbon Accelerator Unit (ORAU), Oxford, England, UK (n = 14; bone collagen and dentine)87 and at the University of Groningen, Faculty of Science and Engineering, Groningen, The Netherlands (n = 25; bone collagen and dentine)88. All pre-Xiongnu samples and 30% of the Xiongnu samples were radiocarbon dated to solidify the dating of individuals from early time periods. Most Xiongnu and Mongol samples were separated into periods based on archaeological materials and burial styles as assessed by excavators and curators from the National University of Mongolia. See Supplementary Text S2 for additional details.
Data availability and ethical approval statement
All of the data included in the study have been made available in Tables 1 and 2, the Supplementary Information. Samples analyzed for this study (AT- denoted codes) are currently curated at the Max Planck Institute for the Science of Human History, Jena, Germany. Samples were exported to the Max Planck Institute for the Science of Human History under permission from the Ministry of Culture, Education, Science and Sports (Export number 10/413 (7b/52) which was received on 2nd February, 2017 #A0109258, MN DE 7 643)
References
Myadar, O. Imaginary nomads: Deconstructing the representation of Mongolia as a land of nomads. Inner Asia 335–362 (2011).
Golden, P. B. Central Asia in World History. Oxford: Oxford University Press. (2011).
Liu, X. The Silk Road in World History. Oxford: Oxford University Press. (2010).
Beckwith, C. I. Empires of the Silk Road: A History of Central Eurasia from the Bronze Age to the Present. Princeton: Princeton University Press. (2009).
Di Cosmo, N. Ancient China and its Enemies: The Rise of Nomadic Power in East Asian History. Cambridge: Cambridge University Press. (2002).
Barfield, T. J. The shadow empires: Imperial state formation along the Chinese-nomad frontier. Empires: perspectives from archaeology and history 122, 10 (2001).
Honeychurch, W. Alternative Complexities: The Archaeology of Pastoral Nomadic States. J Archaeol. Res. 22, 277–326 (2014).
Sinopoli, C. M. The Archaeology of Empires. Annu. Rev. Anthropol. 23, 159–180 (1994).
Pomper, P. The History and Theory of Empires. Hist. Theory 44, 1–27 (2005).
Morris, I. & Scheidel, W. The Dynamics of Ancient Empires: State Power from Assyria to Byzantium. Oxford University Press. (2009).
Hastorf, C. A. & Johannessen, S. Pre-Hispanic political change and the role of maize in the Central Andes of Peru. Am. Anthropol. 95, 115–138 (1993).
Rösch, M., Fischer, E. & Märkle, T. Human diet and land use in the time of the Khans—Archaeobotanical research in the capital of the Mongolian Empire, Qara Qorum, Mongolia. Veg. Hist. Archaeobot. 14, 485–492 (2005).
Kim, H. & 金浩東 The Unity of the Mongol Empire and Continental Exchanges over Eurasia. Journal of Central Eurasian Studies 1, 15–42 (2009).
Honeychurch, W. & Makarewicz, C. A. The Archaeology of Pastoral Nomadism. Annu. Rev. Anthropol. 45, 341–359 (2016).
Hunt, H. V. et al. Millets across Eurasia: chronology and context of early records of the genera Panicum and Setaria from archaeological sites in the Old World. Veg. Hist. Archaeobot. 17, 5–18 (2008).
Hunt, H. V. et al. Genetic diversity and phylogeography of broomcorn millet (Panicum miliaceum L.) across Eurasia. Mol. Ecol. 20, 4756–4771 (2011).
Lightfoot, E., Liu, X. & Jones, M. K. Why move starchy cereals? A review of the isotopic evidence for prehistoric millet consumption across Eurasia. World. Archaeology 45, 574–623 (2013).
Spengler, R. N. Agriculture in the Central Asian Bronze Age. J. World Prehist. 28, 215–253 (2015).
Wang, T. et al. Tianshanbeilu and the Isotopic Millet Road: reviewing the late Neolithic/Bronze Age radiation of human millet consumption from north China to Europe. Natl. Sci. Rev. https://doi.org/10.1093/nsr/nwx015 (2017).
Svyatko, S. V., Schulting, R. J., Mallory, J. & Murphy, E. M. Stable isotope dietary analysis of prehistoric populations from the Minusinsk Basin, Southern Siberia, Russia: a new chronological framework for the introduction of millet to the eastern Eurasian steppe. J. of Arch. Sci. (2013).
Yang, Y. et al. Economic Change in the Prehistoric Hexi Corridor (4800-2200 bp), North-West China. Archaeometry. https://doi.org/10.1111/arcm.12464 (2019).
Motuzaite Matuzeviciute, G. et al. The extent of cereal cultivation among the Bronze Age to Turkic period societies of Kazakhstan determined using stable isotope analysis of bone collagen. J. Archaeol. Sci. 59, 23–34 (2015).
Ventresca Miller, A. R. & Makarewicz, C. A. Intensification in pastoralist cereal use coincides with the expansion of trans-regional networks in the Eurasian Steppe. Scientific Reports 9, (2019).
Spengler, R. N. Fruit from the Sands: Artifacts of the Silk Road on Your Dinner Table. University of California Press. (2019).
Di Cosmo, N. Ancient Inner Asian Nomads: Their Economic Basis and Its Significance in Chinese History. J. Asian Stud. 53, 1092–1126 (1994).
Spengler, R. N., Ryabogina, N., Tarasov, P. E. & Wagner, M. The spread of agriculture into northern Central Asia: Timing, pathways, and environmental feedbacks. Holocene 26, 1527–1540 (2016).
Korolyuk, E. A. & Polosmak, N. V. Plant remains from Noin Ula Burial Mounds 20 and 31 (Northern Mongolia). Archaeology, Ethnology and Anthropology of Eurasia 38, 57–63 (2010).
Hermes, T. R. et al. Urban and nomadic isotopic niches reveal dietary connectivities along Central Asia’s Silk Roads. Scientific Reports vol. 8 (2018).
Murphy, E. M. et al. Iron Age pastoral nomadism and agriculture in the eastern Eurasian steppe: implications from dental palaeopathology and stable carbon and nitrogen isotopes. J. Archaeol. Sci. 40, 2547–2560 (2013).
Machicek, M. L. Reconstructing Diet, Health and Activity Patterns in Early Nomadic Pastoralist Communities of Inner Asia. University of Sheffield. (2011).
Ventresca Miller, A. et al. Subsistence and social change in central Eurasia: stable isotope analysis of populations spanning the Bronze Age transition. J. Archaeol. Sci. 42, 525–538 (2014).
van der Merwe, N. J. & Vogel, J. C. 13C content of human collagen as a measure of prehistoric diet in woodland North America. Nature 276, 815–816 (1978).
Ambrose, S. H. & Norr, L. Experimental Evidence for the Relationship of the Carbon Isotope Ratios of Whole Diet and Dietary Protein to Those of Bone Collagen and Carbonate. Prehistoric Human Bone 1–37 (1993).
Schoeninger, M. J. & DeNiro, M. J. Nitrogen and carbon isotopic composition of bone collagen from marine and terrestrial animals. Geochim. Cosmochim. Acta 48, 625–639 (1984).
Ambrose, S. H. Preparation and characterization of bone and tooth collagen for isotopic analysis. J. Archaeol. Sci. 17, 431–451 (1990).
van Klinken, G. J. Bone Collagen Quality Indicators for Palaeodietary and Radiocarbon Measurements. J. Archaeol. Sci. 26, 687–695 (1999).
An, C.-B., Chen, F.-H. & Barton, L. Holocene environmental changes in Mongolia: A review. Glob. Planet. Change 63, 283–289 (2008).
Schwanghart, W., Schütt, B. & Walther, M. Holocene climate evolution of the Ugii Nuur basin, Mongolia. Adv. Atmos. Sci. 25, 986–998 (2008).
Stacy, E. M. Stable Isotopic Analysis of Equid (Horse) Teeth from Mongolia. University of Pittsburgh, (2009).
Toderich, K. et al. C3/C4 plants in the vegetation of Central Asia, geographical distribution and environmental adaptation in relation to climate. Climate Change and Terrestrial Carbon Sequestration in Central Asia 33–63 (2007).
Ventresca Miller, A. R. et al. Pastoralist Mobility in Bronze Age Landscapes of Northern Kazakhstan: 87Sr/86Sr and δ18O Analyses of Human Dentition from Bestamak and Lisakovsk. Environ. Archaeol. 23, 352–366 (2018).
Wang, C. et al. Variations in leaf carbon isotope composition along an arid and semi-arid grassland transect in northern China. J. Plant Ecol. 9, 576–585 (2016).
Auerswald, K., Max, H. O., Tungalag, R., Bai, Y. & Schnyder, H. Sheep Wool δ13C Reveals No Effect of Grazing on the C3/C4 Ratio of Vegetation in the Inner Mongolia–Mongolia Border Region Grasslands. PLoS One 7, e45552 (2012).
Makarewicz, C. A. Winter is coming: seasonality of ancient pastoral nomadic practices revealed in the carbon (δ13C) and nitrogen (δ15N) isotopic record of Xiongnu caprines. Archaeol. Anthropol. Sci. 9, 405–418 (2017).
Burnik Šturm, M., Ganbaatar, O., Voigt, C. C. & Kaczensky, P. Sequential stable isotope analysis reveals differences in multi-year dietary history of three sympatric equid species in SW Mongolia. J. Appl. Ecol. 54, 1110–1119 (2017).
Pyankov, V. I., Gunin, P. D., Tsoog, S. & Black, C. C. C4 plants in the vegetation of Mongolia: their natural occurrence and geographical distribution in relation to climate. Oecologia 123, 15–31 (2000).
Davie, H., Murdoch, J. D., Lini, A., Ankhbayar, L. & Batdorj, S. Carbon and Nitrogen Stable Isotope Values for Plants and Mammals in a Semi-Desert Region of Mongolia. Mong. J. Biol. Sci. 12, 33–43 (2014).
Makarewicz, C. & Tuross, N. Foddering by Mongolian pastoralists is recorded in the stable carbon (δ13C) and nitrogen (δ15N) isotopes of caprine dentinal collagen. J. Archaeol. Sci. 33, 862–870 (2006).
Lee-Thorp, J. A., Sealy, J. C. & van der Merwe, N. J. Stable carbon isotope ratio differences between bone collagen and bone apatite, and their relationship to diet. J. Archaeol. Sci. 16, 585–599 (1989).
Fernandes, R., Nadeau, M.-J. & Grootes, P. M. Macronutrient-based model for dietary carbon routing in bone collagen and bioapatite. Archaeol. Anthropol. Sci. 4, 291–301 (2012).
Fenner, J. N., Tumen, D. & Khatanbaatar, D. Food fit for a Khan: stable isotope analysis of the elite Mongol Empire cemetery at Tavan Tolgoi, Mongolia. J. Archaeol. Sci. 46, 231–244 (2014).
de Rachewiltz, I. The Secret History of the Mongols. The Mongolia Society Bulletin 9, 55–69 (1970).
(of Rubruck.), W. The Mission of Friar William of Rubruck: His Journey to the Court of the Great Khan Mongke, 1253–1255. (Hakluyt Society, 1990).
Ban, G. Han shu. vol. 5 (Ding wen shu ju, 1962).
Sima, Q. Shiji (Records of the grand historian). Qin Dynasty Volume (transl. Wtson, B.)(Columbia University Press, New York, 1961) (1959).
Song, L. & Others. Yuanshi [History of the Yuan Dynasty]. Beijing: Zhong Hua Book Company (1976).
Bemmann, J., Höllmann, T. O., Ahrens, B., Kaiser, T. & Müller, S. A Stone Quarry in the Hinterland of Karakorum, Mongolia, with Evidence of Chinese Stonemasons. Journal of Inner Asian Art and Archaeology 6, 101–136 (2011).
Jeong, C. et al. Bronze Age population dynamics and the rise of dairy pastoralism on the eastern Eurasian steppe. Proc. Natl. Acad. Sci. USA 115, E11248–E11255 (2018).
Spengler, R. N., de Nigris, I., Cerasetti, B., Carra, M. & Rouse, L. M. The breadth of dietary economy in Bronze Age Central Asia: Case study from Adji Kui 1 in the Murghab region of Turkmenistan. Journal of Archaeological Science: Reports. https://doi.org/10.1016/j.jasrep.2016.03.029 (2016).
Amartüvshin, N. Tariany khar budaag (panicum miliaceum L.) Mongol orond nutagshuulsan tüükhees. in Syan’bi, Zhuzhany üeiin tüükh, soyolyn sudalgaa (ed. Ts Odbaatar And Ts) 152–158 (2018).
Wright, J., Honeychurch, W. & Amartuvshin, C. The Xiongnu settlements of Egiin Gol, Mongolia. Antiquity 83, 372–387 (2009).
Wright, J., Honeychurch, W. & Amartuvshin, C. Initial findings of the Baga Gazaryn Chuluu archaeological survey (2003–2006). Antiquity 81, 313 (2007).
Davydova, A. Ivolginskii arkheologicheskii kompleks: Ivolginskoe gorodishche. Saint Petersburg: AziatlKA (1995).
Pousaz, N., et al. L’Habitat Xiongnu de Boroo Gol. Gollion (2013).
Alcock, S. E., John H D’Arms Collegiate Professor of Classical Archaeology and Classics and Arthur F Thurnau Professor Susan E Alcock, D’Altroy, T. N., Morrison, K. D. & Sinopoli, C. M. Empires: Perspectives from Archaeology and History. Cambridge: Cambridge University Press. (2001).
Chase-Dunn, C. A. T. D. H. Cross-World-System Comparisons: Similarities and Differences. in World-Systems and Civilizations (ed. Sanderson, S.) (1996).
Khazanov, A. M. & Wink, A. Nomads in the sedentary world. London: Routledge. (2001).
Grousset, R. The Empire of the Steppes: A History of Central Asia. (Rutgers University Press, 1970).
Yu, Y. Nomads and Han China. Expanding Empires: Cultural Interactions and Exchange in World Societies from Ancient to Early Modern Times. Wilmington, DE: A Scholarly Resource Inc 133–141 (2002).
Bocquet-Appel, J.-P. The Neolithic demographic transition, population pressure and cultural change. Comparative Civilizations Review 58, 6 (2008).
Zhao, Z. New Archaeobotanic Data for the Study of the Origins of Agriculture in China. Curr. Anthropol. 52, S295–S306 (2011).
Barton, L. et al. Agricultural origins and the isotopic identity of domestication in northern China. Proc. Natl. Acad. Sci. USA 106, 5523–5528 (2009).
Hosner, D., Wagner, M., Tarasov, P. E., Chen, X. & Leipe, C. Spatiotemporal distribution patterns of archaeological sites in China during the Neolithic and Bronze. Age: An overview. The Holocene. 26, 1576–1593 (2016).
Boserup, E. Agricultural Growth and Population Change. In The World of Economics (eds. Eatwell, J., Milgate, M. & Newman, P.) p. 1–14 Palgrave Macmillan UK. (1991).
Spengler, R. N., Miller, N. F., Neef, R., Tourtellotte, P. A. & Chang, C. Linking agriculture and exchange to social developments of the Central Asian Iron Age. Journal of Anthropological Archaeology 48, 295–308 (2017).
Boserup, E. The impact of population growth on agricultural output. Q. J. Econ. 257–270 (1975).
Kradin, N. N. Nomadism, Evolution and World-Systems: Pastoral Societies in Theories of Historical Development. Journal of World-Systems Research 8, 368–388 (2002).
Johnson, J. A. & Hanks, B. Society, demography and community: reassessing Bronze Age Sintashta populations in the southern Urals, Russia (2100–1700 BC). in Beyond Elites: Alternatives to Hierarchical Systems in Modelling Social Formations vol. 215 355–367 Teil 2. Aus dem Institut für Archäologische Wissenschaften der Universität. (2012).
Hanks, B. Archaeology of the Eurasian Steppes and Mongolia. Annu. Rev. Anthropol. 39, 469–486 (2010).
Hedges, R. E. M., Clement, J. G., Thomas, C. D. L. & O’Connell, T. C. Collagen turnover in the adult femoral mid-shaft: Modeled from anthropogenic radiocarbon tracer measurements. American Journal of Physical Anthropology 133, 808–816 (2007).
Hillson, S. Dental Anthropology. Cambridge: Cambridge University Press. (1996).
R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria http://www.R-project.org/ (2013).
Groß, M. Modeling body height in prehistory using a spatio-temporal Bayesian errors-in variables model. AStA Adv. Stat. Anal. 100, 289–311 (2016).
Rosenstock, E. et al. Human Stature in the Near East and Europe ca. 10 000 – 1000 BC: its spatio-temporal development in a Bayesian errors-in-variables model. Archaeol. Anthropol. Sci. (2019).
Fernandes, R., Millard, A. R., Brabec, M., Nadeau, M.-J. & Grootes, P. Food reconstruction using isotopic transferred signals (FRUITS): a Bayesian model for diet reconstruction. PLoS One 9, e87436 (2014).
Fernandes, R. et al. IsoMemo: a Big isotopic Data initiative for archaeology, ecology, and environmental & life sciences. IsoMemo, http://www.isomemo.com (2019).
Taylor, W. et al. Radiocarbon dating and cultural dynamics across the early pastoral transition in eastern Eurasia. PLoS One (2019).
Brock, F., Higham, T., Ditchfield, P. & Ramsey, C. B. Current Pretreatment Methods for AMS Radiocarbon Dating at the Oxford Radiocarbon Accelerator Unit (Orau). Radiocarbon 52, 103–112 (2010).
QGIS Development Team. QGIS Geographic Information System. Open Source Geospatial Foundation Project. http://qgis.osgeo.org (2019).
Acknowledgements
This research was funded by the Max Planck Society. We owe thanks to the National University of Mongolia, the Mongolian Institute of Archaeology, and Dr. Mark Horton for introduction and access to the collections used in this study. We also thank Michelle O’Reilly and Hans Sell for their work on the figures created for this paper.
Author information
Authors and Affiliations
Contributions
S.W., P.R. and N.B. designed the experiments. S.W., M.B., S.U. and E.M. collected the samples. S.W. and J.Z. completed laboratory analyses. S.W., A.V.M., R.F., R.H., J.Z. and P.R. analyzed the data. S.W., P.R., A.V.M., B.K.M., R.F., W.T., R.N.S., S.U. and E.M. contextualised the data and wrote the article with input from all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wilkin, S., Ventresca Miller, A., Miller, B.K. et al. Economic Diversification Supported the Growth of Mongolia’s Nomadic Empires. Sci Rep 10, 3916 (2020). https://doi.org/10.1038/s41598-020-60194-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-60194-0
This article is cited by
-
Presenting the Compendium Isotoporum Medii Aevi, a Multi-Isotope Database for Medieval Europe
Scientific Data (2022)
-
Early emergence and development of pastoralism in Gan-Qing region from the perspective of isotopes
Archaeological and Anthropological Sciences (2021)
-
Prehistoric agriculture and social structure in the southwestern Tarim Basin: multiproxy analyses at Wupaer
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.