Abstract
To assess the safety and clinical outcomes of multi-probe stereotactic radiofrequency ablation (SRFA) for very large (≥8 cm) primary and metastatic liver tumors with curative treatment intent. A retrospective, single center study carried out between 01.2005 and 06.2018. 34 consecutive patients had a total of 41 primary and metastatic liver tumors with a median size of 9.0 cm (8.0–18.0 cm) at initial SRFA. Patients were treated under CT guidance using a 3D navigation system. Endpoints consisted of (i) technical efficacy; primary - requiring one treatment, and secondary – requiring two treatments (ii) complication and mortality rates (iii) local and distant recurrence (LR), (iv) disease free survival (DFS), (v) overall survival (OS). 33/41 tumors were successfully ablated at initial SRFA (80.5% primary technical efficacy rate (PTE)). Four tumors required repeat ablation, resulting in a secondary technical efficacy (STE) rate of 90.2%. Local tumor recurrence (LR) developed in 4 of 41 tumors (9.8%). The 30-day perioperative mortality was 2.3% (1/ 44 ablations). The total major complication rate was 20.5% (9 of 44 ablations). Three of nine (33.3%) major complications, such as pleural effusion, pneumothoraces or perihepatic hemorrhages were relatively easy to treat. The overall survival (OS) rates at 1-, 3-, and 5- years from the date of the first SRFA were 87.1%, 71.8%, and 62.8% for patients with hepatocellular carcinoma (HCC) and 87.5%, 70.0% and 70.0% for patients with intrahepatic cholangiocarcinoma (ICC) respectively. Patients with metastatic disease had OS rates of 77.8% and 22.2% at 1- and 3- years. The clinical results of SRFA in this study are encouraging and warrant a prospective multicenter study. SRFA may become one of the best therapeutic choices for a growing number of patients with primary and metastatic liver cancer.
Similar content being viewed by others
Introduction
Radiofrequency (RF) ablation has been increasingly accepted as a curative and cost-effective treatment for small liver tumors1,2. Whilst hepatic resection (HR) is still the preferred treatment in patients with preserved liver function, in clinical practice less than one-third of the patients are eligible for HR at diagnosis3,4. Historically, the reported local recurrence rates after conventional RF ablation are relatively high and vary from 10% to 39.1% by 5 years, depending on tumor size and number5,6. It has been shown that an ablation zone ‘safety margin’ is required around tumors by at least 5 mm to achieve local control and good clinical outcome7. However, achieving adequate treatment margins may be challenging in large and irregularly shaped tumors or in situations where the target tumor is either difficult to visualize, awkward to access, or adjacent to vulnerable structures. Similar considerations apply to HR, with larger tumors more likely to be unresectable due to small future liver remnant and close relationship with the major vessels or hepatic hilum8.
Transarterial chemoembolization (TACE) is currently the recommended treatment method in patients with impaired liver function and large (>5 cm) hepatocellular carcinoma (HCC)9,10 and whilst a survival benefit has been demonstrated10,11, it is considered a palliative treatment12.
To overcome limitations in RF ablation zone size, other thermal ablation strategies have been developed including use of microwave (MW) energy and placement of multiple RF electrodes13,14,15,16. However, conventional multi-probe RF ablation using ultrasound or CT-fluoroscopy guidance lacks reliability for accurate three-dimensional RF probe placement and thus complete tumor coverage. Stereotaxy has proven useful for planning and executing complex or challenging ablations where access routes can be specifically facilitated and more precise coverage of the target tumor and safety margins can be accomplished17,18.
We have previously shown that SRFA is a viable treatment option in HCC19, although its role in very large lesions remains undefined.
The purpose of the current study was to assess the safety and clinical outcomes of multi-probe SRFA for very large (≥8 cm) primary and metastatic liver tumors, treated with curative intent. Specifically, our endpoints are primary (requiring one treatment) and secondary (requiring two treatments) technical efficacy, complication rates, recurrence, disease free and overall survival.
Materials and Methods
Selection criteria
The Institutional Review Board of the Medical University of Innsbruck approved this single-center, retrospective study. All participants gave their informed consent to collect their data. All treatment plans were consensually agreed in multidisciplinary tumor board meetings.
Between 01.2005 and 06.2018, 943 consecutive patients were treated by SRFA. Seventy-seven patients with portal venous invasion, extensive tumor with subsequent palliative intention to treat, or benign liver tumors were excluded (Fig. 1). Thirty-four patients with tumors ≥8 cm at initial SRFA were included. Baseline characteristics of included patients are shown in Table 1.
A platelet count of <50000/mm3 and prothrombin activity <50% were exclusion criteria for SRFA. Tumor diagnosis was confirmed by typical imaging appearances on multiphasic contrast MRI or CT, with histopathological confirmation before or during SRFA procedure.
Multi-probe stereotactic radiofrequency ablation - procedure
The basic principle of multi-probe RF ablation is the simultaneous usage of multiple RF probes to overcome ablation size limitation of single probe techniques by creating multiple overlapping ablation zones (Fig. 2). “Stereotaxy” derives from two Greek roots - “stereos” meaning solid, and “taksis” meaning arrangement. Stereotactic technique relates the position of targets and entrance points within the body to a Cartesian coordinate system.
The method of SRFA has previously been reported in detail20,21,22. In brief, the whole treatment is performed under general anesthesia with muscle paralysis. Patients are immobilized on the CT-table by a single (Bluebag, Medical Intelligence Schwabmünchen, Germany) or double vacuum fixation technique (BodyFix, Medical Intelligence Schwabmünchen, Germany). For image-to-patient registration, 10–15 fiducials, (X-SPOT, Beekley Corporation, Bristol, CT, USA) are broadly attached to the skin of the thorax and upper abdomen. A contrast-enhanced planning CT (Siemens SOMATOM Sensation Open, sliding gantry with 82 cm diameter, Siemens AG, Erlangen, Germany) is obtained with 3 mm slice thickness. To enable precise stereotactic conditions, the endotracheal tube (ETT) is temporarily disconnected during the planning CT, during each stereotactic needle placement and for the final control CT. The CT dataset is transferred to an optical-based navigation system (Stealth Station Treon plus, Medtronic Inc., Louisville, KY, USA). Multiple SRFA probe trajectories with an interprobe spacing of 1.5–2 cm are planned by using multiplanar and 3D reconstructed images.
After registration and a registration accuracy check, the probe of the navigation system is introduced into the ATLAS aiming device (Medical Intelligence Inc., Schwabmünchen, Germany). After alignment of the probe axis with the planned virtual trajectory by using the guidance software of the navigation system, 15G/17.2 cm coaxial needles (Bard Inc., Covington, GA, USA) are sequentially advanced through the adjusted targeting device without real-time imaging control. The depth from the aiming device to the target is automatically calculated by the navigation software. For verification of correct needle placement, a native control CT is performed and fused with the planning CT. In cases with lack of histological confirmation, a 16G coaxial biopsy sample is obtained. Thereafter, three 17G RF-electrodes (Cool-tip, Medtronic, Mansfield, MA, USA, 25 cm in length with 3 cm exposure) are introduced through the coaxial needles for serial tumor ablation. RF ablation is carried out using the unipolar Cool-tip RF generator (Cool-tip, Medtronic, Mansfield, MA, USA), including the Cool-tip_RF switching controller. The standard ablation time for three electrodes is 16 minutes. However, if a significant increase of impedance is observed (the so-called ‘roll-off effect’) the ablation process is finished. Track ablation is performed during every repositioning and final removal of the RF-electrodes to reduce bleeding and tumor seeding.
For verification of the ablation zone and for assessment of complications, a final contrast-enhanced CT scan is superimposed onto the planning CT.
Example images from multi-probe SRFA of very large liver tumors are shown in Figs. 3 and 4.
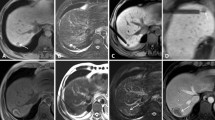
Case of a 40-year old female with a 9.0 cm breast cancer liver metastasis in segment IVa/II close to the heart. (A) Portal venous phase initial CT-scan with a hypo-enhancing large nodule in segment IVa/II (red dashed circle). (B) Maximum Intensity Projection (MIP) of the control CT with 15 coaxial needles in place (red arrowhead). (C,D) Fused CT-images of the contrast enhanced planning and final control CT showing a complete coverage of the tumor (left image) by the coagulation zone (right image). (E,F) The red dashed circle illustrates the progressively shrinking coagulation zone on MR images at 3 months (F) and 12 months (G) after SRFA with no evidence of local recurrence.
Case of a 62-year old female with a very large ICC IVa/II. (A) Fused CT-images of the ce-enhanced planning CT with a 9 cm HCC in segment IV/VIII (red dashed circle). (B) 3D volume rendering of the tumor (red arrowhead). (C) Fused images from the navigation system with 3D views from the contrast enhanced final control CT with complete coverage of the tumor. (D) 3D views from the navigation system with the coaxial needles in place fused with the predefined paths (colored lines). (E,F) The red dashed circle illustrates the progressively shrinking coagulation zone on CT images at 2 years (E) and 7 years (F) after SRFA with no evidence of local recurrence.
Study endpoints
The endpoints of the study consisted of (i) technical success and efficacy rates, (ii) complication and mortality rates, (iii) local and distant recurrence rates, (iv) DFS and (v) OS.
-
(I).
Technical success was defined as accurate electrode placement, with a deviation <1 cm at the needle tip, according to predefined plans. Primary technical efficacy, requiring one treatment with complete ablation, and secondary technical efficacy, requiring more treatments for complete ablation, were determined by follow-up contrast-enhanced CT or MR scans performed at 1- month. The imaging tests were evaluated by two board certified abdominal radiologists by consensus.
-
(II).
Considering the complexity of the procedures and for better comparison to surgery, the study endpoint major complications were defined according to the Clavien-Dindo classification (Grade III+)23. Mortality was defined as death within 30 days after SRFA treatment.
-
(III).
Local recurrence was specified as appearance of new enhancing nodules (CT of MR scans at 3–6 months intervals) within or directly adjacent to the initially tumor-free ablation zone. New enhancing nodules distant to the ablation zone and or to the initial tumor location were specified as distant tumor recurrence.
-
(IV).
(IV, V) Survival was calculated from the date of initial SRFA to the date of death attributable to malignancy or other causes (i.e., event) or to the most recent follow-up visit (i.e., censoring).
Statistical analysis
The statistical analysis was performed with IBM SPSS version 24 (IBM, Armonk, New York). The Kolmogorov-Smirnov-Test, tested normality of distribution. Data were expressed as total numbers, median and range. Survival data (OS and DFS) were analyzed with the Kaplan Meier method. The X² test was used to determine differences between categorical variables, and the Mann-Whitney U test between independent continuous variables. A two-tailed p-value of <0.05 was considered as statistically significant.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Consent for publication
Consent for publication was obtained for every individual person’s data included in the study.
Results
Patient characteristics
34 Patients, 7 females and 27 males, with a median age of 66 years (range 27–81) underwent 44 SRFA treatments of 41 very large primary or metastatic liver tumors Table 1. Diagnoses were 16 (47.1%) HCCs, 8 (23.5%) ICCs, and 10 (29.4%) metastatic tumors. The majority (4/10) of metastatic disease originated from colorectal cancer. The median size of the 41 nodules was 9.0 cm (8.0–18.0 cm). At treatment begin, 22 (64.7%) patients had a solitary liver tumor, 7 (20.6%) had two and 5 (14.7%) patients had more than two tumors. 12 (34.3%) patients had underlying cirrhosis (10 (22.2%) Child-Pugh A, 2 (4.4%) Child-Pugh B and 1 (0.2%) Child-Pugh C). 6 (17.6%) patients underwent chemotherapy, 2 (5.9%) patients underwent surgical resection, 4 (11.8%) patients underwent TACE and 2 (5.9%) patients conventional RF ablation prior to (not combined) SRFA. None of the patients received any additional loco(regional) therapy such as resection, TACE or SIRT.
The histopathological results from the biopsies obtained during SRFA (in 18 patients) were positive for malignancy in all cases.
Perioperative complications
Major perioperative complications are presented in Table 2. One death was attributed to bleeding after SRFA of an 8 cm ICC (mortality rate 2.3% (1/44 ablations)). The total major complication rate was 20.5% (9 of 44 ablations).
One patient developed acute liver failure after treatment of 3 HCCs (largest tumor 8 cm) requiring salvage liver transplantation. Thermal injuries of the bowel (1 patient) and gallbladder (1patient) had to be surgically treated. One liver abscess required CT guided drainage. Other complications included transient liver failure (1 patient) and pleural effusions (3 patients) requiring thoracoenteses respectively. Additionally, two perihepatic hemorrhages and one pneumothorax were treated within the same session by embolization and chest drain which did not influence the postoperative course. 3/9 (33%) major complications were successfully treated by the interventional radiologist within the next days by placing a pleural drainage in case of major pleural effusions (3 patients).
The median hospital stay after the ablation was 7 days, ranging from 3–21 days. The median number of inserted coaxial needles per tumor was 12 (6–31).
Technical Success
SRFA was successfully completed according to plan in all 41 tumors (technical success rate 100%) Table 3. 33/41 tumors were successfully ablated at initial SRFA (80.5% primary technical efficacy rate). Two very large tumors with 18 cm and 10 cm in diameter required three and two ablation sessions, respectively. All other tumors were treated in one session. After the first follow-up, 4 tumors were retreated, resulting in a secondary technical efficacy rate of 90.2%. 6–31 (median 12) RF electrodes were inserted in each tumor.
A sub analysis based on tumor type (HCC, ICC and Metastasis) showed the highest PTE rates for HCC tumors with 88.2% (15/17) and the lowest for ICC with 75% (9/12). However, the differences between groups were non-significant (p = 0.607).
Local recurrence rate and distant recurrence
During a median imaging follow-up of 9.4 months (range 1–133 months) local tumor recurrence developed in 4 of 41 tumors (9.8%, Table 4). Distant tumor recurrence in the liver was found in 24 patients (70.6%), and extrahepatic metastasis in 6 patients (17.6%), respectively. 19 (55.9%) patients received repeated SRFA for newly developed tumors. 15 patients (44.1%) developed untreatable tumor progression with multiple new nodules or extrahepatic spread.
A sub analysis based on tumor type (HCC, ICC and Metastasis) showed the lowest LR rates for HCC at 5.9% (1/16) and the highest for ICC at 16.7% (2/12). However, the differences between groups were non-significant (p = 0.600).
Overall and disease-free survival
For HCC, the median OS and OS rates (Fig. 5) at 1-, 3-, and 5- years from the date of the first SRFA were 95.4 months (95% CI 36.1–169.4), 87.1%, 71.8%, and 62.8%, and for ICC 87.5%, 70.0% and 70.0%, respectively (median OS N/A).
Among patients with metastatic disease, the OS rates at 1- and 3- years were 77.8% and 22.2% with a median OS of 22.8 months (95% CI 17.4–36.2).
There was a difference, however, non-significant between groups based on tumor entity (p = 0.15).
The disease-free (DFS) rates (Fig. 6) at 1-, 3-, and 5- years from the date of first SRFA were 43.8%, 29.2%, and 29.2%, 37.5%, 25.0% and 12.5% for patients with HCC or ICC with a median OS of 7.1 months (95% CI 0–15.5), and 4.35 months (95% CI 0–8.9), respectively. Among patients with metastatic disease, the DFS rates at 1- year were 25% with a median OS of 5.4 months (95% CI 0–16.8).
There was no significant difference between groups based on tumor entity (p = 0.72).
Discussion
The main finding of our study is that SRFA is a feasible and effective treatment strategy for tumors >8 cm. To the best of our knowledge, there are no reports available dealing with thermal ablation of this tumor size, partially because HR remains the recommended treatment in large tumors with preserved liver function. However, the minority of patients are suitable for resection3,4, meaning other curative treatment options are required.
Since RFA results in complete cure for small HCCs (<3 cm) and colorectal liver metastases (CRLM) in greater than 90% of cases24,25,26, it has been incorporated into international guidelines for small tumors12,27,28,29. However, early studies demonstrated that complete cure falls to 40–70%30,31 for medium HCCs (up to 5 cm) and to 23–45% for large HCCs31,32 respectively, which is very likely to reflect the size limitations of RF ablation zones and the difficulties in producing overlapping ablations. To overcome size limitations, two main strategies were implemented. Firstly, increasing energy transfer in the form of microwave energy, and secondly using multiple RF probes to create overlapping ablation zones. More recent studies have consequently shown better PTE rates up to 86–97% in medium and large HCCs up to 10 cm15,33,34.
Whilst MWA can produce larger ablation zones faster and with less ‘heat-sink’ effect (i.e., large vessels reduce local tissue heating due to cooling) than RFA35, this higher thermal energy might injure adjacent vulnerable structures or create unnecessarily large ablation zones36. Whilst the lower energy transferred in RFA may result in more predictable ablation zones, the greater number of probes required considerably increases the complexity and difficulty of the procedure.
Accurate three-dimensional probe alignment to create sufficiently overlapping ablation zones demands a high level of operator experience using conventional US- and CT- guidance. However, SRFA offers three-dimensional ablation planning to achieve an optimal configuration of RF probes and create multiple overlapping coagulation volumes. Usage of an aiming device and triggering of respiratory motion37 facilitate precise path needle and probe placement17. Immediate post ablation contrast-enhanced CT fusion with the planning CT allows for rapid, reliable intraoperative judgment of the ablation results with the option for re-ablation within the same session. In a recent histopathological validation study in explanted livers after bridging therapy with SRFA, we found no viable tumor in 183 of 188 treated lesions (97.3%) and in 50 of 52 lesions >3 cm (96.2%).
Despite our inclusion of tumors up to 18 cm in diameter in the present study, LR developed in only 4 of 41 tumors (9.8%). In contrast, best results from the US- or CT- guided multiprobe thermal ablation literature with ‘freehand’ targeting quote LR rates of 15.9–19.7% after thermal ablation of large HCCs (largest tumors 10 cm, mean 3–5.7 cm) and other large liver tumors up to 8 cm (mean tumor size 2.8 cm)33,34,38.
OS rates in our study at 1-, 3-, and 5- years from first SRFA were 87.1%, 71.8%, 62.8%, and 87.5%, 70.0%, 70.0% for patients with HCC and ICC, respectively whereas patients with metastatic disease had OS rates of 77.8% and 22.2% at 1- and 3-years. Data are sparse regarding RF ablation of tumors >5 cm with Ma et al.34 reporting lower OS rates than in our study - specifically 65.5%, 36.3% and 21% at 1-, 3- and 5- years, respectively. Several retrospective studies compared HR and TACE in solitary HCC >5 cm, and reported 5-year OS of 43–65% and 17.5–49.6% for HR and TACE respectively39,40,41. Two studies regarding HR in HCC >5 cm by Zhao et al.42 and Hwang et al.43 reported OS of 75%, 59% and 44% and 78.4%, 58.8% and 49.3% at 1-, 3- and 5- years, respectively. For HCC (>10 cm), Zhou et al.44 showed OS of 60.7%, 34% and 28.6% at 1-, 3- and 5- years respectively. Data are sparse for HR of large CRLM with Nomit et al.45 reporting OS at 1-, 3- and 5- years of 87.2%, 56.1% and 35.4% respectively after laparoscopic HR.
In the present study, distant tumor recurrence was found in 70.6% of patients and median DFS rates were 4.4–7.1 months. These unfavourable prognostic indicators despite high levels of effective local tumor control (90.8%) might be explained by large lesions having a greater chance of vascular invasion46 and micrometastases47, and are line with the surgical literature at 57.1–82.4%43,44.
Our major complication rate of 20.5% is higher than in the study of Ma et al.34 and Laeseke et al.32 who reported a major complication rate of 16% following RFA for HCC >5 cm. One of the main reasons for that could be the high number of needles used (median 12 per tumor). However, our mortality rate compares favorably to Filmann et al.48 who reported a mortality of 9.1–25.5% after major HR in large outcome analysis of 110 332 procedures. Zhou et al.44 also published a systematic review after HR of large HCC (>10) a median perioperative morbidity of 29.2% (13.6–72%) and mortality of 3.5% (0–18.2%) and Nomit et al.45 reported a major complication rate of 50.0% after laparoscopic HR of large CRLM.
Limitations
Study limitations include the retrospective design, heterogeneity of previous unsuccessful loco(regional) treatments (TACE or HR), and single treatment center bias. Comparisons with previous related studies are limited as stereotactic navigation systems were not employed in prior reports.
In summary, although all patients in this study were considered to be inoperable, we report lower mortality rates, lower complication rates and similar survival to the curative literature meaning SRFA may represent an important tool in the treatment of very large liver tumors.
References
EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. J. Hepatol. 56, 908-943, https://doi.org/10.1016/j.jhep.2011.12.001 (2012).
Ahmed, M. et al. Image-guided tumor ablation: standardization of terminology and reporting criteria–a 10-year update. Radiology 273, 241–260, https://doi.org/10.1148/radiol.14132958 (2014).
Abdalla, E. K. et al. Recurrence and outcomes following hepatic resection, radiofrequency ablation, and combined resection/ablation for colorectal liver metastases. Ann. Surg. 239, 818–825 (2004).
Clancy, C., Burke, J. P., Barry, M., Kalady, M. F. & Calvin Coffey, J. A meta-analysis to determine the effect of primary tumor resection for stage IV colorectal cancer with unresectable metastases on patient survival. Ann. Surg. Oncol. 21, 3900–3908, https://doi.org/10.1245/s10434-014-3805-4 (2014).
Lencioni, R. et al. Early-stage hepatocellular carcinoma in patients with cirrhosis: long-term results of percutaneous image-guided radiofrequency ablation. Radiology 234, 961–967, https://doi.org/10.1148/radiol.2343040350 (2005).
Nakazawa, T. et al. Radiofrequency ablation of hepatocellular carcinoma: correlation between local tumor progression after ablation and ablative margin. Ajr 188, 480–488, https://doi.org/10.2214/AJR.05.2079 (2007).
Kim, Y. S. et al. The minimal ablative margin of radiofrequency ablation of hepatocellular carcinoma (>2 and <5 cm) needed to prevent local tumor progression: 3D quantitative assessment using CT image fusion. Ajr 195, 758–765, https://doi.org/10.2214/AJR.09.2954 (2010).
Fan, S. T. Liver functional reserve estimation: state of the art and relevance for local treatments: the Eastern perspective. J. Hepatobiliary Pancreat. Sci. 17, 380–384, https://doi.org/10.1007/s00534-009-0229-9 (2010).
Llovet, J. M. et al. Arterial embolisation or chemoembolisation versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: a randomised controlled trial. Lancet 359, 1734–1739, https://doi.org/10.1016/S0140-6736(02)08649-X (2002).
Lo, C. M. et al. Randomized controlled trial of transarterial lipiodol chemoembolization for unresectable hepatocellular carcinoma. Hepatology 35, 1164–1171, https://doi.org/10.1053/jhep.2002.33156 (2002).
Llovet, J. M. & Bruix, J. Systematic review of randomized trials for unresectable hepatocellular carcinoma: Chemoembolization improves survival. Hepatology 37, 429–442, https://doi.org/10.1053/jhep.2003.50047 S0270913902141619 [pii] (2003).
de Lope, C. R., Tremosini, S., Forner, A., Reig, M. & Bruix, J. Management of HCC. J. Hepatol. 56(Suppl 1), 75–87, https://doi.org/10.1016/S0168-8278(12)60009-9 (2012).
Kodama, H. et al. Radiofrequency Ablation Using a Multiple-Electrode Switching System for Lung Tumors with 2.0-5.0-cm Maximum Diameter: Phase II Clinical Study. Radiology 277, 895–902, https://doi.org/10.1148/radiol.2015141153 (2015).
Yoon, J. H. et al. Switching bipolar hepatic radiofrequency ablation using internally cooled wet electrodes: comparison with consecutive monopolar and switching monopolar modes. Br. J. Radiol. 88, 20140468, https://doi.org/10.1259/bjr.20140468 (2015).
Lee, J. et al. Percutaneous radiofrequency ablation with multiple electrodes for medium-sized hepatocellular carcinomas. Korean J. Radiol. 13, 34–43, https://doi.org/10.3348/kjr.2012.13.1.34 (2012).
Haemmerich, D. & Lee, F. T. Jr. Multiple applicator approaches for radiofrequency and microwave ablation. Int. J. Hyperth. 21, 93–106, https://doi.org/10.1080/02656730412331286894 (2005).
Widmann, G., Schullian, P., Haidu, M., Fasser, M. & Bale, R. Targeting accuracy of CT-guided stereotaxy for radiofrequency ablation of liver tumours. Minim. Invasive Ther. Allied Technol. 20, 218–225, https://doi.org/10.3109/13645706.2010.533923 (2011).
Perrodin, S. et al. Percutaneous stereotactic image-guided microwave ablation for malignant liver lesions. Sci. Rep. 9, 13836, https://doi.org/10.1038/s41598-019-50159-3 (2019).
Bale, R. et al. Stereotactic Radiofrequency Ablation of Hepatocellular Carcinoma: a Histopathological Study in Explanted Livers. Hepatology 70, 840–850, https://doi.org/10.1002/hep.30406 (2019).
Bale, R., Widmann, G. & Haidu, M. Stereotactic radiofrequency ablation. Cardiovasc. Intervent. Radiol. 34, 852–856, https://doi.org/10.1007/s00270-010-9966-z (2011).
Bale, R. et al. Percutaneous stereotactic radiofrequency ablation of colorectal liver metastases. Eur. Radiol. 22, 930–937, https://doi.org/10.1007/s00330-011-2314-0 (2012).
Schullian, P. et al. Stereotactic radiofrequency ablation of subcardiac hepatocellular carcinoma: a case-control study. Int. J. Hyperth. 36, 876–885, https://doi.org/10.1080/02656736.2019.1648886 (2019).
Dindo, D., Demartines, N. & Clavien, P. A. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann. Surg. 240, 205–213 (2004).
Lencioni, R. A. et al. Small hepatocellular carcinoma in cirrhosis: randomized comparison of radio-frequency thermal ablation versus percutaneous ethanol injection. Radiology 228, 235–240, https://doi.org/10.1148/radiol.2281020718 (2003).
Shiina, S. et al. A randomized controlled trial of radiofrequency ablation with ethanol injection for small hepatocellular carcinoma. Gastroenterology 129, 122–130, https://doi.org/10.1053/j.gastro.2005.04.009 (2005).
Sotirchos, V. S. et al. Colorectal Cancer Liver Metastases: Biopsy of the Ablation Zone and Margins Can Be Used to Predict Oncologic Outcome. Radiology 280, 949–959, https://doi.org/10.1148/radiol.2016151005 (2016).
Aube, C. et al. EASL and AASLD recommendations for the diagnosis of HCC to the test of daily practice. Liver Int. 37, 1515–1525, https://doi.org/10.1111/liv.13429 (2017).
Llovet, J. M., Bru, C. & Bruix, J. Prognosis of hepatocellular carcinoma: the BCLC staging classification. Semin. Liver Dis. 19, 329–338, https://doi.org/10.1055/s-2007-1007122 (1999).
Van Cutsem, E. J., Oliveira, J., Kataja, V. V. & Force, E. G. T. ESMO Minimum Clinical Recommendations for diagnosis, treatment and follow-up of advanced colorectal cancer. Ann. Oncol. 16(Suppl 1), 18–19, https://doi.org/10.1093/annonc/mdi803 (2005).
Sala, M. et al. Initial response to percutaneous ablation predicts survival in patients with hepatocellular carcinoma. Hepatology 40, 1352–1360, https://doi.org/10.1002/hep.20465 (2004).
Livraghi, T. et al. Hepatocellular carcinoma: radio-frequency ablation of medium and large lesions. Radiology 214, 761–768, https://doi.org/10.1148/radiology.214.3.r00mr02761 (2000).
Laeseke, P. F. et al. Multiple-electrode radiofrequency ablation creates confluent areas of necrosis: in vivo porcine liver results. Radiology 241, 116–124, https://doi.org/10.1148/radiol.2411051271 (2006).
Lin, C. C., Cheng, Y. T., Chen, M. W. & Lin, S. M. The Effectiveness of Multiple Electrode Radiofrequency Ablation in Patients with Hepatocellular Carcinoma with Lesions More than 3 cm in Size and Barcelona Clinic Liver Cancer Stage A to B2. Liver Cancer 5, 8–20, https://doi.org/10.1159/000367755 (2016).
Ma, S. et al. Ultrasound-guided percutaneous microwave ablation for hepatocellular carcinoma: clinical outcomes and prognostic factors. J. Cancer Res. Clin. Oncol. 143, 131–142, https://doi.org/10.1007/s00432-016-2266-5 (2017).
Pillai, K. et al. Heat sink effect on tumor ablation characteristics as observed in monopolar radiofrequency, bipolar radiofrequency, and microwave, using ex vivo calf liver model. Med. 94, e580, https://doi.org/10.1097/MD.0000000000000580 (2015).
Poulou, L. S., Botsa, E., Thanou, I., Ziakas, P. D. & Thanos, L. Percutaneous microwave ablation vs radiofrequency ablation in the treatment of hepatocellular carcinoma. World J. Hepatol. 7, 1054–1063, https://doi.org/10.4254/wjh.v7.i8.1054 (2015).
Widmann, G., Schullian, P., Haidu, M., Wiedermann, F. J. & Bale, R. Respiratory motion control for stereotactic and robotic liver interventions. Int. J. Med. Robot. 6, 343–349, https://doi.org/10.1002/rcs.343 (2010).
Yu, J. et al. Local tumour progression after ultrasound-guided microwave ablation of liver malignancies: risk factors analysis of 2529 tumours. Eur. Radiol. 25, 1119–1126, https://doi.org/10.1007/s00330-014-3483-4 (2015).
Jin, Y. J. et al. Surgery versus transarterial chemoembolization for solitary large hepatocellular carcinoma of BCLC stage A. J. Gastrointest. Surg. 18, 555–561, https://doi.org/10.1007/s11605-013-2440-x (2014).
Lee, Y. B. et al. Comparison of transarterial chemoembolization and hepatic resection for large solitary hepatocellular carcinoma: a propensity score analysis. J. Vasc. Interv. Radiol. 26, 651–659, https://doi.org/10.1016/j.jvir.2015.02.004 (2015).
Zhu, S. L. et al. Comparison of long-term survival of patients with solitary large hepatocellular carcinoma of BCLC stage A after liver resection or transarterial chemoembolization: a propensity score analysis. PLoS One 9, e115834, https://doi.org/10.1371/journal.pone.0115834 (2014).
Zhao, H. C. et al. A retrospective analysis of long term outcomes in patients undergoing hepatic resection for large (>5 cm) hepatocellular carcinoma. HPB 18, 943–949, https://doi.org/10.1016/j.hpb.2016.08.005 (2016).
Hwang, S. et al. Prognostic Prediction Models for Resection of Large Hepatocellular Carcinoma: A Korean Multicenter Study. World J. Surg. 42, 2579–2591, https://doi.org/10.1007/s00268-018-4468-2 (2018).
Zhou, Y. M., Li, B., Xu, D. H. & Yang, J. M. Safety and efficacy of partial hepatectomy for huge (>/=10 cm) hepatocellular carcinoma: a systematic review. Med. Sci. Monit. 17, RA76–83, https://doi.org/10.12659/msm.881443 (2011).
Nomi, T., Fuks, D., Louvet, C., Nakajima, Y. & Gayet, B. Outcomes of Laparoscopic Liver Resection for Patients with Large Colorectal Liver Metastases: A Case-Matched Analysis. World J. Surg. 40, 1702–1708, https://doi.org/10.1007/s00268-016-3467-4 (2016).
Pawlik, T. M. et al. Tumor size predicts vascular invasion and histologic grade: Implications for selection of surgical treatment for hepatocellular carcinoma. Liver Transpl. 11, 1086–1092, https://doi.org/10.1002/lt.20472 (2005).
Hosch, S. B., Steffani, K. D., Scheunemann, P. & Izbicki, J. R. Micrometastases from HBP malignancies and metastatic cancer. J. Hepatobiliary Pancreat. Surg. 9, 583–591, https://doi.org/10.1007/s005340200078 (2002).
Filmann, N. et al. Mortality after liver surgery in Germany. Br. J. Surg., 106(11):1523–1529, https://doi.org/10.1002/bjs.11236 (2019).
Author information
Authors and Affiliations
Contributions
P.S. contributed to study concepts, study design, data acquisition, data analysis, statistical analysis and manuscript preparation and editing. E.J., D.P., G.E. and G.L. contributed to data acquisition and manuscript review. B.R. contributed to study concepts, study design, data analysis and manuscript review.
Corresponding author
Ethics declarations
Competing interests
Reto Bale is a consultant for CAScination. All other authors have no conflicts of interest to disclose.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schullian, P., Johnston, E.W., Putzer, D. et al. Safety and efficacy of stereotactic radiofrequency ablation for very large (≥8 cm) primary and metastatic liver tumors. Sci Rep 10, 1618 (2020). https://doi.org/10.1038/s41598-020-58383-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-58383-y
This article is cited by
-
Optimization of laser dosimetry based on patient-specific anatomical models for the ablation of pancreatic ductal adenocarcinoma tumor
Scientific Reports (2023)
-
High-Level Standardization of Ablative Procedures: The Way to Go
CardioVascular and Interventional Radiology (2023)
-
Stereotactic Radiofrequency Ablation of Breast Cancer Liver Metastases: Short- and Long-Term Results with Predicting Factors for Survival
CardioVascular and Interventional Radiology (2021)
-
Volumetric assessment of the periablational safety margin after thermal ablation of colorectal liver metastases
European Radiology (2021)
-
Triple-Antenna Microwave Ablation with Repositioning for the Creation of a Reliable 6-cm Ablation Zone in the Liver
CardioVascular and Interventional Radiology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.