Abstract
Thyroid dysfunction is frequently associated with functional disturbances of the brain. We performed a meta-analysis of previous positron emission tomography and single-photon emission computed tomography studies using a coordinate-based technique of activation-likelihood estimation (ALE) to investigate the potential background of neuropsychiatric complications in patients with hypo- and hyperthyroidism. We performed a systematic search of MEDLINE and EMBASE for English-language publications using the keywords of “positron emission tomography”, “single-photon emission computed tomography”, and “thyroid”. The software GingerALE ver 2.3.6 was used to transform all reported coordinates into stereotactic Montreal Neurological Institute space. A threshold of uncorrected p < 0.001 (minimum volume of 200 mm3) was applied to the resulting ALE map using cerebral metabolic rate of glucose (CMRglu), and cerebral blood flow (CBF). Six studies were eligible for inclusion in the study; 4 studies of cerebral metabolic rate of CMRglu, and 2 studies of CBF. In hypothyroidism, significant decreases in CMRglu were identified in 3 clusters including left anterior cingulate, right inferior occipital gyrus, and right cuneus. In hyperthyroidism, a significant decrease in CMRglu was identified in right superior frontal gyrus. In hypothyroidism, a significant decrease in CBF was observed in left postcentral gyrus. In conclusion, several brain regions showed altered CMRglu and CBF in patients with thyroid dysfunction compared with euthyroid controls. These findings might account for underlying mechanisms of thyroid hormones on psychological and physiological effects on brain.
Similar content being viewed by others
Introduction
Thyroid dysfunction is frequently associated with functional disturbances of the brain such as cognitive impairment1, neurodegenerative disorders2, dementia3, depression, and anxiety4. Transient thyroid dysfunction may also induce neuropsychiatric changes5. These disorders accompanying thyroid dysfunction are generally reversible after return to the euthyroid status6,7. Therefore, it seems that mood and cognitive impairments are often associated with putative disturbance of thyroid metabolism in the brain8. In spite of advances in the understanding of the metabolism and action of thyroid hormone in the human brain, the relationships between these neuropsychiatric disorders and brain metabolic function are poorly understood. Therefore, it is important to increase our understanding of the pathophysiology of neuropsychiatric disorders in patients with hypo- and hyperthyroidism and to translate these findings into more effective approaches for prevention and treatment.
Although there is no direct method to measure brain function, functional neuroimaging techniques of positron emission tomography (PET) and single-photon emission computed tomography (SPECT) have provided some promising insights into the underlying mechanisms of action of thyroid hormone on cerebral metabolic rate of glucose (CMRglu) and cerebral blood flow (CBF)8. Previously, patients with hypothyroidism have shown a global decrease in CMRglu and CBF9,10,11, especially in posterior regions10,11 or in the parietal lobe9. Diffuse metabolic and perfusion deficits have also been observed in patient with hyperthyroidism12,13,14. However, neuroimaging studies of thyroid dysfunction have yielded inconsistent findings9,10,11,12,13,14.
Hence, we performed a meta-analysis of previous PET and SPECT studies using a coordinate-based technique of activation-likelihood estimation (ALE) to investigate the potential background of neuropsychiatric complications in patients with hypo- and hyperthyroidism.
Materials and Methods
Data search, study selection, and data extraction
We performed a systematic search of MEDLINE (from inception to 26th December 2018) and EMBASE (from inception to 26th December 2018) for English-language publications using the keywords of “positron emission tomography”, “single-photon emission computed tomography”, and “thyroid”. The inclusion criteria were original research articles that reported the difference in cerebral glucose metabolism or cerebral blood flow in subjects with hyper/hypothyroidism. Results that reported as coordinates in a normalized standard stereotactic space (Talairach or Montreal Neurological Institute space) were included. Studies based on regions-of-interest were excluded. Data were extracted from the publications independently by two reviewers, and the following information was recorded: year of publication, country of affiliations of corresponding authors, name of journal, number of studies included, database search, radiopharmaceuticals, coordinates, number of subjects included. Data sharing is not applicable to this article as no new data were created or analyzed in this study.
Meta-analysis algorithm
The software GingerALE ver 2.3.6 (Research Imaging Institute, University of Texas Health Science Center at San Antonio, TX, USA) was used to transform all reported coordinates into stereotactic Montreal Neurological Institute space. The method used in this study is a variation of an original ALE by Turkeltaub et al.15, and later modified by Eickhoff et al.16,17. For each experiment, the modeled activation map is calculated by finding the maximum across each focus’s Gaussian. On the basis of empiric estimates of between-subject variability from the number of subjects in each study, the width of the Gaussian probability distribution is determined individually for each experiment. For each voxel, ALE value is calculated form the union of the modeled activation map. ALE values were combined across studies and tested against a null hypothesis of random distribution of ALE values, which are higher than could be expected by chance. A threshold of uncorrected p < 0.001 (minimum volume of 200 mm3) was applied to the resulting ALE map. ALE results were overlaid onto an anatomical template using Mango ver 4.0.1 (Research Imaging Institute, University of Texas Health Science Center at San Antonio, TX, USA).
Results
Literature search and study characteristics
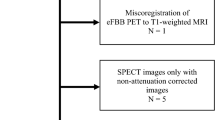
The electronic search identified 1,380 articles. 1,348 studies that did not meet the inclusion criteria based on their title and abstract were excluded (duplicates 60, non-human studies 754, conference abstract 201, non-English studies 61). After reviewing abstracts of 364 studies, 332 records were excluded. After reviewing the full-text of 32 articles, 6 studies were eligible for inclusion in the study. Four studies of CMRglu5,13,14,18, and 2 studies of CBF10,19 were included. The detailed procedure is shown in Fig. 1. In 4 studies10,13,14,18, subjects of hypothyroidism/hyperthyroidism were compared with controls of euthyroid state. In a study by Jeong et al.5, 20 patients with thyroid carcinoma who underwent total thyroidectomy and discontinued levothyroxine therapy before radioactive iodine ablation (hypothyroid state) were compared with 20 patients who underwent total thyroidectomy and continued levothyroxine therapy (euthyroid state). In a study by Schraml et al.19, 9 patients with thyroid carcinoma who underwent total thyroidectomy and discontinued levothyroxine therapy for radioactive iodine ablation (hypothyroid state) were compared with each patient after thyroid hormone replacement. Study characteristics are summarized in Table 1.
Anatomical likelihood estimate analysis
Cerebral glucose metabolism
In hypothyroidism, significant decreases in CMRglu were identified in 3 clusters including left anterior cingulate (cluster size 296 mm3, x −8; y 44; z −6, maximum ALE value of 0.0088), right inferior occipital gyrus (cluster size 216 mm3, x 26; y −100; z −6, maximum ALE value of 0.0096), and right cuneus (cluster size 216 mm3, x 6; y −72; z 14, maximum ALE value of 0.0096). In hyperthyroidism, a significant decrease in CMRglu was identified in 1 cluster of right superior frontal gyrus (cluster size 200 mm3, x 16.9; y 15.6; z 60.7, maximum ALE value of 0.0085) (Fig. 2 & Table 2).
Cerebral blood flow
In hypothyroidism, a significant decrease in CBF was observed in 1 cluster of left postcentral gyrus (cluster size 368 mm3, x −53; y −15; z 41, maximum ALE value of 0.0087) (Fig. 3 & Table 3).
Discussion
In this meta-analysis, we identified several brain regions in which patients with thyroid dysfunction reliably exhibited metabolic or perfusion decrease compared with euthyroid controls. The CMRglu in left anterior cingulate, right inferior occipital gyrus, and right cuneus was affected in hypothyroid patients. In addition, in hypothyroid status, CBF was also significantly decreased in left postcentral gyrus. On the other hand, in patients with hyperthyroidism, a significant decrease in CMRglu was identified in right superior frontal gyrus. These results suggest that thyroid hormone regulates regional CMRglu and CBF in the mature brain. These findings also demonstrate that the mechanism of neuropsychiatric disturbances in patients with hypothyroidism differs from those with hyperthyroidism.
Brain is the main target organ of thyroid hormones, and adult-onset thyroid dysfunction can have a significant impact on neuropsychiatric disturbances1,2,3,4,5. In patients with thyroid dysfunction, there may be behavioural abnormalities that mimic depression, mania, and dementia due to hormonal excess and deficit8. The thyroxine is a prohormone and converted to triiodothyronine (T3) within cells via deiodinase enzymes20. Regulation of T3 production by expression of brain-region specific deiodinase, thyroid hormone transporters and receptors are believed to maintain thyroid hormone homeostasis in the brain21,22,23. Therefore, current laboratory tests forthyroid dysfunction may not accurately measure thyroid hormone status in the brain8,21,22,23. Functional neuroimaging studies suggest a direct association between thyroid and brain activity, therefore, these can provide some clues of underlying mechanisms of thyroid hormones on psychological and physiological effects on brain.
The anterior cingulate cortex and the parieto-occipital area including cuneus and postcentral gyrus showed decreased CMRglu and CBF in patients with hypothyroidism. The anterior cingulate cortex plays an important role in affective and cognitive regulation, involving attention, problem solving, motivation, error detection, decision making, and social behaviors24,25. In a study of 13 hypothyroid patients and 10 euthyroid controls, hypothyroid patients exhibited lower CMRglu in anterior cingulate cortex18. Thyroid hormone replacement therapy has caused a reduction in the somatic complaints and depressive symptoms associated with a restoration of metabolic activity in the brain18. Another study identified reversible hypoperfusion in the anterior and posterior cingulate cortex, amygdala and hippocampus in previously untreated hypothyroidism26. Similarly, decreased pattern of brain metabolism in parieto-occipital areas was consistent with most previous neuroimaging studies performed on hypothyroid patients9,10,19,27. Metabolic and perfusion deficits in these area can affect working memory and attention, written word recognition, transient memory retrieval, awareness and imagery of visuospatial input, and priming processes, often compromised in patients with hypothyroidism28. Therefore, it is noteworthy that the anterior cingulate cortex and parieto-occipital area were identified as regions of decreased brain metabolism in this ALE meta-analysis.
In patients with hyperthyroidism, a significant decrease in CMRglu was identified in superior frontal gyrus of the right hemisphere. Miao et al.13 showed a decreased CMRglu in the left parahippocampal, fusiform, and right superior frontal gyrus, when comparing hyperthyroid patients with euthyroid controls. And, treatment with methimazole specifically increased regional activity in these regions and these changes significantly correlated with the anxiety and depressive symptoms13. In another included study, the CMRglu in the limbic system is affected in hyperthyroid subjects with significantly correlated with both anxiety and depressive symptoms14. The superior frontal gyrus is located at the superior part of the prefrontal cortex and several studies have found a broader role of this region in anatomical and functional connectivity29. The functional analysis demonstrates that the superior frontal gyrus has positive association with cingulate cortex, middle frontal gyrus, caudate, and thalamus and negative association with inferior frontal and precentral gyrus30. Further research on the value of superior frontal gyrus in mood changes caused by hyperthyroidism is needed to support our findings.
This study has some limitations. The small number of studies included in this meta-analysis meant that we had relatively limited power to detect brain regions with significance. However, we have minimized selection bias by excluding the studies based on the region-of-interest. The region-of-interest approach might result in a targeted but biased search for metabolic abnormalities. Also, we could not perform subgroup analysis based on the sex difference due to insufficient data. In addition, this study cannot demonstrate the specific relationship between altered CMRglu or CBF and clinical symptoms. Nevertheless, this meta-analysis is the first to evaluate the potential background of neuropsychiatric complications in patients with hypo- and hyperthyroidism. In addition, there was no previous study that reported no significant difference of CMRglu and CBF in hyperthyroidism and hypothyroidism. However, publication bias may exist in these previous studies.
In conclusion, brain regions with altered CMRglu and CBF were observed in patients with thyroid dysfunction. These particular regions differ in patients with hypothyroidism from those with hyperthyroidism. These findings might account for underlying mechanisms of thyroid hormones on psychological and physiological effects on brain. Further study is needed to valid the relationship between change of CMRglu and/or CBF in these regions and neuropsychiatric disturbances in the future.
Change history
19 March 2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Rieben, C. et al. Subclinical Thyroid Dysfunction and the Risk of Cognitive Decline: a Meta-Analysis of Prospective Cohort Studies. The Journal of clinical endocrinology and metabolism 101, 4945–4954, https://doi.org/10.1210/jc.2016-2129 (2016).
Cappola, A. R. et al. Thyroid function in the euthyroid range and adverse outcomes in older adults. The Journal of clinical endocrinology and metabolism 100, 1088–1096, https://doi.org/10.1210/jc.2014-3586 (2015).
Chaker, L. et al. Thyroid function and the risk of dementia: The Rotterdam Study. Neurology 87, 1688–1695, https://doi.org/10.1212/wnl.0000000000003227 (2016).
Ittermann, T., Volzke, H., Baumeister, S. E., Appel, K. & Grabe, H. J. Diagnosed thyroid disorders are associated with depression and anxiety. Social psychiatry and psychiatric epidemiology 50, 1417–1425, https://doi.org/10.1007/s00127-015-1043-0 (2015).
Jeong, H. S. et al. Differences in Brain Glucose Metabolism During Preparation for (131)I Ablation in Thyroid Cancer Patients: Thyroid Hormone Withdrawal Versus Recombinant Human Thyrotropin. Thyroid: official journal of the American Thyroid Association 27, 23–28, https://doi.org/10.1089/thy.2016.0293 (2017).
Haupt, M. & Kurz, A. [Reversibility of dementia in hypothyroidism]. Zeitschrift fur die gesamte innere Medizin und ihre Grenzgebiete 48, 609–613 (1993).
Davis, J. D. & Tremont, G. Neuropsychiatric aspects of hypothyroidism and treatment reversibility. Minerva endocrinologica 32, 49–65 (2007).
Bauer, M., Goetz, T., Glenn, T. & Whybrow, P. C. The thyroid-brain interaction in thyroid disorders and mood disorders. Journal of neuroendocrinology 20, 1101–1114, https://doi.org/10.1111/j.1365-2826.2008.01774.x (2008).
Constant, E. L. et al. Cerebral blood flow and glucose metabolism in hypothyroidism: a positron emission tomography study. The Journal of clinical endocrinology and metabolism 86, 3864–3870, https://doi.org/10.1210/jcem.86.8.7749 (2001).
Krausz, Y. et al. Regional cerebral blood flow in patients with mild hypothyroidism. Journal of nuclear medicine: official publication, Society of Nuclear Medicine 45, 1712–1715 (2004).
Krausz, Y. et al. Brain SPECT study of common ground between hypothyroidism and depression. The international journal of neuropsychopharmacology 10, 99–106, https://doi.org/10.1017/s1461145706006481 (2007).
Seo, S. W. et al. Thyrotoxic autoimmune encephalopathy: a repeat positron emission tomography study. Journal of neurology, neurosurgery, and psychiatry 74, 504–506 (2003).
Miao, Q. et al. Reversible changes in brain glucose metabolism following thyroid function normalization in hyperthyroidism. AJNR. American journal of neuroradiology 32, 1034–1042, https://doi.org/10.3174/ajnr.A2449 (2011).
Schreckenberger, M. F. et al. Positron emission tomography reveals correlations between brain metabolism and mood changes in hyperthyroidism. The Journal of clinical endocrinology and metabolism 91, 4786–4791, https://doi.org/10.1210/jc.2006-0573 (2006).
Turkeltaub, P. E., Eden, G. F., Jones, K. M. & Zeffiro, T. A. Meta-analysis of the functional neuroanatomy of single-word reading: method and validation. NeuroImage 16, 765–780 (2002).
Eickhoff, S. B., Bzdok, D., Laird, A. R., Kurth, F. & Fox, P. T. Activation likelihood estimation meta-analysis revisited. NeuroImage 59, 2349–2361, https://doi.org/10.1016/j.neuroimage.2011.09.017 (2012).
Eickhoff, S. B. et al. Coordinate-based activation likelihood estimation meta-analysis of neuroimaging data: a random-effects approach based on empirical estimates of spatial uncertainty. Human brain mapping 30, 2907–2926, https://doi.org/10.1002/hbm.20718 (2009).
Bauer, M. et al. Brain glucose metabolism in hypothyroidism: a positron emission tomography study before and after thyroid hormone replacement therapy. The Journal of clinical endocrinology and metabolism 94, 2922–2929, https://doi.org/10.1210/jc.2008-2235 (2009).
Schraml, F. V., Beason-Held, L. L., Fletcher, D. W. & Brown, B. P. Cerebral accumulation of Tc-99m ethyl cysteinate dimer (ECD) in severe, transient hypothyroidism. Journal of cerebral blood flow and metabolism: official journal of the International Society of Cerebral Blood Flow and Metabolism 26, 321–329, https://doi.org/10.1038/sj.jcbfm.9600191 (2006).
Bianco, A. C., Salvatore, D., Gereben, B., Berry, M. J. & Larsen, P. R. Biochemistry, cellular and molecular biology, and physiological roles of the iodothyronine selenodeiodinases. Endocrine reviews 23, 38–89, https://doi.org/10.1210/edrv.23.1.0455 (2002).
Hernandez, A., Quignodon, L., Martinez, M. E., Flamant, F. & St Germain, D. L. Type 3 deiodinase deficiency causes spatial and temporal alterations in brain T3 signaling that are dissociated from serum thyroid hormone levels. Endocrinology 151, 5550–5558, https://doi.org/10.1210/en.2010-0450 (2010).
Shukla, P. K. et al. Prenatal thyroxine treatment disparately affects peripheral and amygdala thyroid hormone levels. Psychoneuroendocrinology 35, 791–797, https://doi.org/10.1016/j.psyneuen.2009.10.019 (2010).
Sittig, L. J., Herzing, L. B., Shukla, P. K. & Redei, E. E. Parent-of-origin allelic contributions to deiodinase-3 expression elicit localized hyperthyroid milieu in the hippocampus. Molecular psychiatry 16, 786–787, https://doi.org/10.1038/mp.2011.19 (2011).
Bush, G., Luu, P. & Posner, M. I. Cognitive and emotional influences in anterior cingulate cortex. Trends in cognitive sciences 4, 215–222 (2000).
Rushworth, M. F., Behrens, T. E., Rudebeck, P. H. & Walton, M. E. Contrasting roles for cingulate and orbitofrontal cortex in decisions and social behaviour. Trends in cognitive sciences 11, 168–176, https://doi.org/10.1016/j.tics.2007.01.004 (2007).
Zhu, D. F. et al. fMRI revealed neural substrate for reversible working memory dysfunction in subclinical hypothyroidism. Brain: a journal of neurology 129, 2923–2930, https://doi.org/10.1093/brain/awl215 (2006).
Nagamachi, S. et al. Cerebral blood flow abnormalities induced by transient hypothyroidism after thyroidectomy–analysis by tc-99m-HMPAO and SPM96. Annals of nuclear medicine 18, 469–477 (2004).
Cabeza, R. & Nyberg, L. Imaging cognition II: An empirical review of 275 PET and fMRI studies. Journal of cognitive neuroscience 12, 1–47 (2000).
Li, W. et al. Subregions of the human superior frontal gyrus and their connections. NeuroImage 78, 46–58, https://doi.org/10.1016/j.neuroimage.2013.04.011 (2013).
Hu, S., Ide, J. S., Zhang, S. & Li, C. R. The Right Superior Frontal Gyrus and Individual Variation in Proactive Control of Impulsive Response. The Journal of neuroscience: the official journal of the Society for Neuroscience 36, 12688–12696, https://doi.org/10.1523/jneurosci.1175-16.2016 (2016).
Acknowledgements
This work was supported by a Research Grant of BioMedical Research Institute for Convergence of Biomedical Science and Technology of Pusan National University Yangsan Hospital (Grant No; 2017-024).
Author information
Authors and Affiliations
Contributions
Kyoungjune Pak, Mijin Kim; study design and writing of the manuscript. Keunyoung Kim; study design. Bo Hyun Kim; image analysis. Seong-Jang Kim, In Joo Kim; data analysis
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pak, K., Kim, M., Kim, K. et al. Cerebral glucose metabolism and Cerebral blood flow in thyroid dysfunction: An Activation Likelihood Estimation Meta-analysis. Sci Rep 10, 1335 (2020). https://doi.org/10.1038/s41598-020-58255-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-58255-5
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.