Abstract
Green tea supplementation has beneficial health effects. However, its underlying mechanisms, such as effects on modulating the intestinal microbiome and endogenous metabolome, particularly following short-term supplementation, are largely unclear. We conducted an integrative metabolomics study to evaluate the effects of short-term (7-day) supplementation of green tea extract (GTE) or its components, epigallocatechin gallate, caffeine, and theanine, on the caecum microbiota and caecum/skin metabolome in mice. Further, we established an integrative metabolome-microbiome model for correlating gut and skin findings. The effects of short-term supplementation with dietary compounds were evaluated with respect to UV stress response, with GTE showing the most remarkable effects. Biplot analysis revealed that Bifidobacteria and Lactobacillus spp. were considerably influenced by short-term GTE supplementation, while Clostridium butyricum was significantly increased by UV stress without supplementation. GTE supplementation helped the skin metabolome defend against UV stress. Interestingly, a significant positive correlation was observed between caecum bacteria (Bifidobacteria, Lactobacillus spp.) and metabolites including skin barrier function-related skin metabolites, caecal fatty acids, and caecal amino acids. Overall, 7-day GTE supplementation was sufficient to alter the gut microbiota and endogenous caecum/skin metabolome, with positive effects on UV stress response, providing insight into the mechanism of the prebiotic effects of GTE supplementation.
Similar content being viewed by others
Introduction
Dietary polyphenol compounds are important substrates for the intestinal microbiota and contribute to intestinal health by modulating intestinal microbial community composition1. Dietary polyphenol compounds may play important roles in the growth or survival of beneficial intestinal bacteria such as Lactobacillus and Bifidobacteria spp., and thus exert prebiotic actions and inhibit the growth of pathogenic bacteria species1,2. Green tea consumption has recently been shown to influence intestinal microbiome composition. Many studies showed that green tea consumption not only alters microbial diversity and core microbiota in healthy human faecal microbiota3, but also increases the proportion of Bifidobacteria species in human faecal microbiota2. Additionally, green tea consumption has shown beneficial and disease-improving effects in previous studies of high-fat diet-induced obesity, adipocyte hypertrophy, and hepatic steatosis. These effects are highly related to the modulation of the intestinal microbiota and metabolic pathways4,5. Dietary polyphenol compounds also show photo-protective properties and enhance endogenous photo-protection by scavenging reactive oxygen species and modulating cellular responses or stress-dependent signaling6. Numerous studies have reported the photo-protective effects of green tea administration7,8.
In biological systems such as cells, tissues, and organs, metabolomic approaches study various small molecules. Small molecules are the final products of metabolic responses in living systems, and can be used as biomarker candidates for various disease states9,10. Integrated analyses of metabolomics and microbial communities have recently increased in popularity11,12. Merging metabolomics and microbial community analyses can provide valuable information regarding how the microbiome functions in various environments such as the gut, which may be explained by modulation of the microbial community and metabolome. Particularly, recent studies examined the interrelationship between gut and skin conditions13,14. Additionally, we showed that prolonged green tea supplementation influences the large intestinal microbiota and exo/endogenous metabolome in ultraviolet (UV) B-exposed mice15. In addition, studies on the effects of short-term green tea intake on the body have also been carried out, showing that green tea extract (GTE) can increase fat oxidation and can improve insulin sensitivity and glucose tolerance during moderate-intensity exercise in healthy young men 24 h after intake16. Hodgson et al.17 reported acute effects of GTE intake for 7 days in endogenous metabolites in human plasma. Based on the results of these studies, we devised the experimental scheme of this study, and thus hypothesized that short-term (7-day) supplementation of GTE modulates the intestinal microbiota and endogenous metabolome in mice, mitigating responses to UV stress. Furthermore, we also examined the effects of short-term supplementation with single compounds including epigallocatechin gallate (EGCG), caffeine, and theanine in mice compared to supplementation with GTE.
Results
Study design and caecum microbial community analysis
Female Skh:HR-1 mice were subjected to comprehensive metabolomics analysis to evaluate the effects of supplementing with GTE or its components EGCG, caffeine, and theanine for 7 days prior to UV stress on the caecum microbiota and endogenous metabolome changes. Further, a correlation model was established to examine skin metabolome changes and erythema formation. To investigate the effects of short-term supplementation of GTE or its components on the caecum environment prior to UV stress in mice, the caecum microbiota of bacterial taxa derived from gut low-density array (GULDA) were quantitatively compared. The ratio of Firmicutes to Bacteroidetes, calculated as the relative abundance of the 16S rRNA gene with a specific bacterial primer, was significantly increased by UV stress without prior supplementation. In contrast, 7-day supplementation with GTE, EGCG, caffeine, or theanine significantly inhibited this increase in the ratio of Firmicutes to Bacteroidetes (Fig. 1). The fold changes in the relative abundance of bacterial taxa in the caecum from each group to the CON group are shown as bar charts (Fig. 2). Additionally, to determine the major bacterial taxa affected by supplementation with dietary compounds, we constructed principal component analysis (PCA) biplots (Fig. 2). Using bar charts and PCA biplots, we compared the effects of supplementing with dietary compounds prior to UV stress on the caecum microbial community. According to the PCA biplot of the CON and UV groups, Clostridium butyricum was highly correlated with the UV group, and also significantly increased in the UV group compared to the CON. Prior supplementation of dietary compounds modulated the microbial community, altering influential bacteria in each group from that in the CON group. The bacteria that differed the most in the GU group from that in the CON group were Bifidobacteria and Lactobacillus. The CU group showed a more diverse distribution of bacteria taxa including Prevotella, Bacteroides, Desulfovibrio, Clostridium, Firmicutes, and C. butyricum from the CON group. The EU and TU groups were not clearly discriminated from the CON group. These results indicate that short-term supplementation of GTE and caffeine modulate the caecum microbial community and that these changes remained even after UV stress. Short-term supplementation of EGCG and theanine also influenced the caecal microbial community, which inhibited modulations resulting from UV stress.
Ratio of Firmicutes to Bacteroidetes in each experimental group calculated using relative abundance of target 16S rRNA gene with a specific bacterial primer. CON (control), UV (exposure to single UV stress without supplementation), GU (7-day green tea extract supplementation followed by single UV stress), EU (7-day EGCG supplementation followed by single UV stress), CU (7-day caffeine supplementation followed by single UV stress), TU (7-day theanine supplementation followed by single UV stress). *p < 0.01 compared to CON group, #p < 0.01 compared to the UV group.
Principal component analysis (PCA) biplot analysis for caecum microbiota analysed by gut low-density array (GULDA). Each PCA biplot was generated using two groups. Black filled circle―CON (control), red filled circle―UV (exposure to single UV stress without supplementation), green filled circle―GU (7-day green tea extract supplementation followed by single UV stress), blue filled circle―EU (7-day EGCG supplementation followed by single UV stress), brown filled circle―CU (7-day caffeine supplementation followed by single UV stress), purple filled circle―TU (7-day theanine supplementation followed by single UV stress). Quantitative caecum microbial community comparison of bacterial taxa derived from GULDA. Ratio (log2) of the relative abundance of selected 16S rRNA gene targets in caecum from each group to that of the CON group.
Alterations in the caecum endogenous metabolome mediated by short-term dietary compound supplementation
To investigate the changes in the caecal metabolome associated with changes to the microbial community, endogenous caecum metabolite profiling was carried out by gas chromatography time-of-flight mass spectrometry (GC-TOF-MS) and ultraperformance-liquid chromatography quadrupole time-of-flight mass spectrometry (UPLC-Q-TOF-MS). Multivariate analysis was also performed to compare metabolite differences among each group using the partial least squares discriminant analysis (PLS-DA). According to PLS-DA score plots, the CON, UV, GU, EU, CU, and TU groups were clearly discriminated from each other (Fig. 3A,B). To select the major variables among the experimental groups, variable importance in projection (VIP) values (>1.0) of PLS-DA were used. Thirty-five caecum metabolites including amino acids, organic compounds, carbohydrates, nucleobases, lysophospholipids, and bile acids were selected and identified as significantly discriminant metabolites (Table S1). The relative levels of metabolites were normalized to CON group values and visualized using a heatmap (Fig. 3C). The heatmap showed increased levels of several amino acids, carbohydrates, nucleobases, and bile acids and decreasing patterns of fatty acids and lysophospholipids in the UV group. However, few metabolites were significantly modulated, including cysteine, oxalic acid, and adonitol, indicating that UV stress without supplementation caused mild changes in the endogenous caecum metabolome. However, short-term supplementation of GTE, EGCG, caffeine, or theanine prior to UV stress induced diverse metabolite changes in the caecum. Particularly, GTE and EGCG supplementation altered similar metabolites. The following metabolites showed remarkably higher variation in each experimental group than in the UV group: GU-glutamine, 5-oxoproline, myo-inositol, glucose, glycerol, oleic acid, palmitic acid, and stearic acid; EU-glutamine, tryptophan, 5-oxoproline, 2-hydroxyglutaric acid, glucose, uracil, thymine, and oxodeoxycholic acid; CU-leucine, isoleucine, 5-oxoproline, malic acid, 2-hydroxyglutaric acid, adonitol, thymine, inosine, lysoPC(20:2), and oxodeoxycholic acid; TU-phenylalanine, 5-oxoproline, aspartic acid, myo-inositol, glucose, glycerol, arabinose, valeric acid, arachidonic acid, and myristic acid.
Metabolite profiling of caecum extracts based on GC-TOF-MS and UPLC-Q-TOF-MS analysis. (A) Three-dimensional PLS-DA score plot of GC-TOF-MS analysis. (B) Three-dimensional PLS-DA score plot of UPLC-Q-TOF-MS analysis. (C) Heatmap of significantly different metabolites (VIP > 1, p-value < 0.05) derived from PLS-DA (A,B). Each data point shown on the heatmap was normalized to the values of the CON group. Black filled circle―CON (control), red filled circle―UV (exposure to single UV stress without supplementation), green filled circle―GU (7-day green tea extract supplementation followed by single UV stress), blue filled circle―EU (7-day EGCG supplementation followed by single UV stress), brown filled circle―CU (7-day caffeine supplementation followed by single UV stress), purple filled circle―TU (7-day theanine supplementation followed by single UV stress).
Biochemical observation of mouse dorsal skin and endogenous skin metabolome changes mediated by short-term dietary compound supplementation
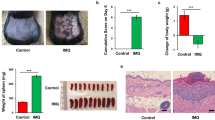
Erythema formation in the dorsal skin of mice was measured after 24 h of UV stress. Erythema formation in the UV groups was considerably higher than that in the CON group and slightly lower in the GU group (Fig. 4A). Differences from the UV group were unremarkable in the short-term GTE component-supplemented groups, EU, CU, and TU. Skin redness was measured with a colour reader to determine the a-value on the dorsal skin surface (Fig. 1B). Skin redness was 4.6 ± 0.58, 6.41 ± 0.94*, 4.71 ± 0.30#, 5.59 ± 0.30, 5.32 ± 0.93, and 5.4 ± 1.10 in the CON, UV, GU, EU, CU, and TU groups, respectively (*p < 0.01 compared to CON, #p < 0.01 compared to UV). Skin malondialdehyde (MDA) and tumour necrosis factor-α (TNF- α) levels did not significantly differ among groups (Fig. S1). These data indicate that 7-day supplementation with GTE significantly prevented UV stress-induced erythema formation, while EGCG, caffeine, and theanine showed no significant preventative effects.
Biochemical observation of dorsal skin tissue. Erythema was measured after 24 h of UV stress. (A) Photograph, (B) skin redness (a-value). CON (control), UV (exposure to single UV stress without supplementation), GU (7-day green tea extract supplementation followed by single UV stress), EU (7-day EGCG supplementation followed by single UV stress), CU (7-day caffeine supplementation followed by single UV stress), TU (7-day theanine supplemented followed by single UV stress).*p < 0.01 compared to CON group, #p < 0.01 compared to UV group.
To determine the effect of short-term supplementation with GTE or its components prior to UV stress on modulating the endogenous skin metabolome, comprehensive metabolite profiling was performed. Three-dimensional PLS-DA score plots in the CON, UV, GU, EU, CU, and TU groups were clearly discriminated from each other (Fig. 5A,B), showing similar distribution patterns to those observed from caecum metabolite profiling. Forty-four discriminant skin endogenous metabolites among experimental groups were selected based on their VIP values (>1.0) and p value (<0.05), and tentatively identified. Those of discriminant metabolites included 10 amino acids, 10 organic compounds, 5 carbohydrates, 3 nucleobases, 4 fatty acids, and 12 lipids. Relative metabolite levels were expressed as the fold-change ratio by normalization with the CON group and a heatmap was constructed (Fig. 5C). Further information is summarized in Supplementary Table 2. According to the heatmap, UV stress without prior dietary compound supplementation increased the levels of most amino acids, organic compounds, nucelobases, and lysophospholipids and decreased levels of carbohydrates and fatty acids (Fig. 2C). Short-term supplementation of GTE, EGCG, caffeine, or theanine resulted in different effects on the skin metabolome. In the GU group, the opposite metabolic change patterns were observed to those in the UV group including several amino acids, organic compounds, and nucleobases, as well as most fatty acids and lysophospholipids. Particularly, histidine, tyrosine, trans-urocanic acid, histamine, adenosine, oleic acid, stearic acid, heptadecanoic acid, arachidonic acid, cholesterol, lysoPE(20:0), lysoPE(16:0), lysoPC(20:2), lysoPE(18:2), lysoPC(20:4), lysoPC(18:2), and monoolein showed the most significant changes. Slight skin metabolome differences were observed in the EU, CU, and TU groups compared to the UV group. Levels of several amino acids including alanine, aspartic acid, isoleucine, and threonine were significantly decreased and levels of nucleobases (inosine and uridine) were increased in the EU group, while the CU and TU groups showed very similar metabolic distribution patterns as those of the UV groups.
Metabolite profiling of skin tissue extracts based on GC-TOF-MS and UPLC-Q-TOF-MS analysis. (A) Three-dimensional PLS-DA score plot of GC-TOF-MS analysis. (B) Three-dimensional PLS-DA score plot of UPLC-Q-TOF-MS analysis. (C) Heatmap of significantly different metabolites (VIP > 1, p-value < 0.05) derived from PLS-DA (A,B). Each data point shown on the heatmap was normalized to the values of the CON group. Black filled circle―CON (control), red filled circle―UV (exposure to single UV stress without supplementation), green filled circle―GU (7-day green tea extract supplementation followed by single UV stress), blue filled circle―EU (7-day EGCG supplementation followed by single UV stress), brown filled circle―CU (7-day caffeine supplementation followed by single UV stress), purple filled circle―TU (7-day theanine supplementation followed by single UV stress).
Correlations between modulation of the caecum microbial community and endogenous metabolome
Correlation analysis of the caecum microbiota and endogenous metabolites in the caecum/skin was performed to determine the relationship between changes in the microbiota and metabolome in accordance with the effects of short-term dietary compound supplementation in mice (Fig. 6). Depending on the bacteria, the correlation map showed different correlation patterns in caecum and skin metabolites. Particularly, Lactobacillus spp. and Bifidobacteria were more distinct than the others. Lactobacillus spp. showed positive correlations with caecum fatty acids, amino acids, skin fatty acids, amino acids, trans-urocanic acid, and lipids. Furthermore, Bifidobacteria showed positive correlations with caecum amino acids and skin lysophospholipids. Clostridium cluster IV, Firmicutes, C. butyricum, Desulfovibrio, Bacteroides, and Prevotella showed positive correlations with various caecum metabolites including malic acid, thymine, taurodeoxycholic acid, adonitol, inosine, and lysoPC(20:2).
Discussion
Based on previous studies, we used an animal model to examine whether supplementation with GTE or its components EGCG, caffeine, and theanine modulate the intestinal microbiota and endogenous metabolome in mice. We focused on the effects of short-term (7-day) supplementation on modulating the caecum microbiota and caecum/skin endogenous metabolome, their relationships, and skin erythema-preventing effects. Green tea has a complex chemical composition containing proteins (15–20%), carbohydrates (including cellulose and pectins, 7–25%), amino acids (including theanine, 1–4%), minerals (5%), flavonoids (including catechins, 25–35%), xanthic bases (including caffeine, 3.5%), pigments (0.5–2%), and phenolic acids (including chlorogenic acid and caffeic acid)18,19. Therefore, in this study, we used GTEs with very high catechin contents (50% total catechin) to minimize the effects of these minor components. In addition, single components were supplemented at higher levels to mice than those present in the green tea extracts such as 50% EGCG, 10% caffeine, and 2% theanine. When we designed the animal experiment, we expected clear effects of each component treatment. For these, we choose higher concentrations than actual concentrations in green tea. The amount of EGCG was selected based on the same amount as the total catechin content. The amount of caffeine and theanine was increased by a similar ratio to that of EGCG. However, several studies reported antioxidant activities of green tea minor components such as trace elements, phenolic acids, and pigments18,20,21. Thus, the effect of the green tea extract in vivo might be due to the synergistic effect of the major components in green tea, or might be due to the combined effect of the minor and the major components.
Highly diverse bacteria comprise the intestinal microbiota and produce various microbial metabolites with important roles in modulating the microbiome modulation. These modulations are highly affected by environmental conditions in the intestine and influence the host metabolome and health22. We observed that UV stress without prior supplementation notably altered the caecum microbial community, as shown by the significant increase in C. butyricum and decrease in Bacteriodetes, Bacteroides, Prevotella, Bifidobacteria, and Desulfovibrio spp. (Fig. 2). Cutaneous UV exposure disturbs the immune system of the skin through UV-induced inflammation and damages the skin, skin-associated cells, and tissues23. Additionally, UV exposure affects bacterial cells. A previous study showed that the viability of Lactobacillus spp. decreased immediately upon UV exposure, but was restored over time and enhanced cholesterol removal ability24. We previously showed that prolonged UVB exposure induced marked changes in the microbiota of the large intestine15. Thus, UV stress appears to influence the intestinal bacteria. In our study, C. butyricum was significantly increased in the UV group. Few studies have examined the relationship between UV stress and C. butyricum levels. C. butyricum is a gram-positive, spore-forming, butyric acid-producing, and obligate anaerobic species found in faeces25. Indigenous spore-forming bacteria have various cell morphologies in the intestine such as resting spores, germinated spores, vegetative cells, and endotrophic cells25. Bacterial spores are even more resistant to chemicals, heat, and irradiation than their respective vegetative cells26. In addition, studies have shown that C. butyricum is resistant to various antibiotics and UV rays27. Particularly, Clostridium spp. show higher d-values, representing the decimal reduction time required at a given condition to kill 90% of the exposed microorganisms, than gram-negative bacteria28. Among the bacterial groups we analysed, Bacteroidetes, Bacteroides, Prevotella, Enterobacteriaceae, E. coli, and Desulfovibrio spp. are gram-negative. Among gram-positive bacteria, Clostridium spp. are the only spore-forming bacteria. Thus, C. butyricum might be relatively more resistant to single UVB irradiation than other bacteria, which leads to relatively high bacterial counts. Some studies also showed that C. butyricum has various uses as an antibacterial agent, in gut epithelial cell proliferation, and in ulcerative colitis therapy29,30,31. Unlike in the UV group, short-term supplementation with GTE showed different caecum microbiota modulation patterns including significantly decreased Clostridium cluster IV and Desulfovibrio spp. According to the PCA biplot, the most influential bacteria distinguishing the GU group from the CON group were Lactobacillus spp. and Bifidobacteria (Fig. 2). In agreement with previous studies, we observed increased levels of Lactobacillus spp. and Bifidobacterium spp. in human faecal samples upon green tea consumption1,2. Additionally, oral administration of probiotics such as Lactobacillus and Bifidobacterium species was previously shown to have photo-protective effects on UV-exposed skin32,33,34,35. Furthermore, profound correlations were observed between other skin diseases such as atopic dermatitis and intestinal probiotics36,37. Particularly, colonization of probiotics in the intestine was associated with a decrease in the disease state36,38. The PCA biplot of the CU and CON groups also showed clear discriminant patterns; levels of Lactobacillus spp. and Bifidobacteria were considerably lower in the CU group than levels in the CON group. Overall, these results and results of previous studies suggest that 7-day supplementation with GTE modulates sustained changes in the caecum microbial community, particularly Lactobacillus spp. and Bifidobacteria, and that these changes might prevented UV stress-induced modulation of the microbial community.
In metabolomics studies, sample preparation is extremely important because the metabolome composition could be affected by numerous factors, such as time, method of sampling, environment, and how the sample is stored. Before metabolite extraction, accurate sampling and quenching procedures are required to provide a snapshot of the metabolome and determine the repeatability of the experiment39. To provide exact metabolomics data, samples must be accurately prepared. We observed diverse skin metabolome changes following exposure to UV stress. In the GU group, the opposite metabolic change patterns were observed from those in the UV group, particularly for histidine, tyrosine, trans-urocanic acid, histamine, adenosine, oleic acid, stearic acid, heptadecanoic acid, arachidonic acid, cholesterol, lysoPE(20:0), lysoPE(16:0), lysoPC(20:2), lysoPE(18:2), lysoPC(20:4), lysoPC(18:2), and monoolein (Fig. 5C). These amino acids, urocanic acid, fatty acids, and lipids are closely related to skin barrier functions. Urocanic acid, which is found in the stratum corneum of the epidermis, plays an important role in protecting the epidermis from UVB-induced damage40. Free fatty acids are very actively synthesized in the epidermis, but are decreased by acute UV irradiation accompanied by decreased expression of lipid synthesis-related genes41,42. Cholesterol synthesis is also highly related to the recovery of the permeability barrier function of the skin42. Lysophospholipids are among the most important factors in skin barrier function, skin hydration, and skin inflammation43,44. We also observed significant preventive effects on erythema formation in only the GTE-supplemented group (Fig. 4). These differences in preventing erythema formation may be related to the intake level or characteristics of the supplements. According to other reports, both oral administration and topical application of green tea can reduce UVB irradiation-induced erythema in skin i.e., consuming green tea polyphenols of human (1402 mg/day, 6 weeks), oral administration of green tea extracts in mice (250 mg/kg. day, 12 weeks), treatment of green tea extracts on human skin (10 mg, 30 min before UV irradiation), and topical application of dermal gels containing green tea extracts (3 g/100 g gel)45,46,47,48. However, it remains controversial whether erythema formation is inhibited by EGCG administration. Jeon et al.49 reported that oral EGCG supplementation for 8 weeks significantly increased minimal erythema dose and attenuated UVB-induced skin damage, whereas Chow et al.50 found that administering a high daily bolus dose of EGCG for 4 weeks did not protect against UV-induced erythema. These differences may be related to the different conditions of the experiments such as supplementation amount and period. Reports substantiating the relationship between caffeine or theanine administration and anti-erythema forming effects are lacking. Instead, caffeine was shown to inhibit UV-induced carcinogenesis8,51, while theanine showed various bioactivities such as neuroprotective, antioxidant, anti-inflammatory effects, and improved immunity52. Thus, our results showed that 7-day prior supplementation of GTE, rather than single compounds (EGCG, caffeine, or theanine), significantly modulated various skin metabolites related to skin barrier function. This modulation inhibited skin metabolome changes according to UV stress, and the effects may help prevent skin erythema formation. On the other hand, no significant differences in MDA and TNF-α levels were observed among groups. MDA is a product of lipid peroxidation which is used as an oxidative stress indicator and TNF-α facilitates the migration of inflammatory cells. According to other animal studies that used similar experimental conditions as those used in our study, similar phenomena were observed in MDA and TNF-α measurements. Svobodova et al.53 measured MDA levels in hairless mice skin homogenates after exposure to a single dose of UVB (200 or 800 mJ/cm2) at a 45-cm distance. At both 4 h and 24 h after UVB (200 mJ/cm2) exposure, no significant differences in MDA levels were found compared to non-irradiated controls, but significantly decreased 24 h afterward with a higher UVB dose (800 mJ/cm2). Sharma et al.54 measured TNF-α mRNA levels in mice irradiated with UVB (100 mJ/cm2 day) for 5 days. At 3 h after the last UVB irradiation, levels of TNF-α mRNA significantly decreased, whereas no difference from non-irradiated controls was observed after 20 h. However, the MDA levels of mice skin that were exposed to a single dose of UVB (240 mJ/cm2) at a 7-cm distance showed significant increase53. Overall, results of our study and previous studies showed that the levels of MDA and TNF-α in mouse skin were induced by UVB irradiation, but results were highly influenced by irradiation conditions such as dose, distance from device, and the amount of time passed after exposure.
The relationship between the gut and skin was previously not well-understood. Microbial compounds including cell wall fragments, dead cells, and microbial metabolites may induce immune responses that improve skin health55. Endogenous caecum metabolite profiling was performed to evaluate the relationship between the caecum microbiota and endogenous skin metabolites. According to the heatmap of the caecum metabolome, a few metabolites were significantly altered by UV stress. However, short-term supplementation with GTE, EGCG, caffeine, or theanine induced more diverse metabolite modulations in the caecum (Fig. 3C). This may be because diet is a crucial factor affecting the composition of the intestinal microbiota, and thus each compound influenced the host in a different manner56. According to the heat map, bile acid levels were much higher in all treatment groups than in the CON group. Bile acids are well-known gut bacteria metabolites with various biological functions including absorbing lipid-soluble vitamins; facilitating lipid absorption; maintaining intestinal barrier function; and regulating triglyceride, cholesterol, glucose, and energy homeostasis57. Bacteria including Bacteroides, Clostridium, Lactobacillus, Bifidobacterium, Enterobacter, and Eubacterium are associated with bile acid metabolism22. These highly increased bile acids levels may be related to UV stress. In the GU group, levels of several metabolites such as glutamine, 5-oxoproline, myo-inositol, glucose, glycerol, oleic acid, palmitic acid, and stearic acid were altered. These metabolites showed a high correlation with the caecum probiotic bacteria Lactobacillus spp. and Bifidobacteria. Thus, modulation of the Lactobacillus spp. and Bifidobacteria populations and the effects of GTE supplementation on mice intestine appear to be closely related. These relationships may affect erythema formation in the skin.
In conclusion, we conducted comprehensive metabolome analysis to elucidate the effects of short-term (7-day) supplementation of dietary components on modulating the gut and endogenous metabolomes. Among the supplements examined, including GTE, EGCG, caffeine, and theanine, only short-term supplementation with GTE remarkably modulated the microbiome-metabolome accompanied with significant prevention of UV stress in mice. Particularly, probiotics including Bifidobacteria and Lactobacillus spp. were highly associated with GTE supplementation and bacteria levels were highly correlated with skin barrier function-related metabolites. Overall, 7-day supplementation of GTE effectively modulated the caecum microbiota and endogenous caecum/skin metabolome in mice; these modulations may act as indirect prebiotics and showed positive effects on preventing external UV stress.
Methods
Chemicals
Water, methanol, and acetonitrile were purchased from Fisher Scientific (Waltham, MA, USA). Pyridine, formic acid, N-methyl-N-(trimethylsilyl)-trifluoroacetamide, methoxyamine hydrochloride, EGCG, caffeine, and theanine were obtained from Sigma Chemical Co. (St. Louis, MO, USA). The analytical grade of chemicals were used. Green tea extract (GTE) was obtained from AmorePacific Corp. (Yongin, Korea) and contains catechins (49.9%), including epigallocatechin (EGC, 9.7%), epicatechin (EC, 5.4%), epigallocatechin-gallate (EGCG, 28.4%), and epicatechin-gallate (ECG, 6.4%), as well as caffeine (4.5%) and theanine (0.4%).
Animal experiments
Six-week-old female Skh-1 mice were purchased from Orient Bio (Seongnam, Korea). Mice were housed under controlled conditions as follows: diet (Ain-93G), temperature (24 ± 2 °C), light (12-h light/dark cycle), and humidity (55 ± 10%). The mice were randomly divided into six experimental groups (n = 7, each group) after one week of acclimation: Control (CON), exposure to single UV stress without supplementation group (UV), 7-day GTE supplementation followed by single UV stress group (GU; 6.7 mg/day), 7-day EGCG supplementation followed by single UV stress group (EU; 3.35 mg/day), 7-day caffeine supplementation followed by single UV stress group (CU; 0.67 mg/day), and 7-day theanine supplementation followed by single UV stress group (TU; 0.134 mg/day). For the single exposure to UV stress, four fluorescent lamps (TL 20 W/12 RS SLV) were used with peak emission of 315 nm and wavelength of 290–390 nm (Philips, Amsterdam, Netherlands). Intensity was monitored using a UV meter (v.2.03, VARIOCONTROL; Waldmann, Villingen-Schwenningen, Germany). Mice were exposed to UVB irradiation after 7 days of supplementation with each compound by moving freely in the cage at 30 cm from the lamps (188 mJ/cm2). After 24 h of UV stress, the mice were sacrificed, and their dorsal skin and caecum tissues were taken and stored in a deep freezer (−80 °C). The protocols of animal experiments were approved by the Institutional Animal Care and Use Committee of Gyeonggi Institute of Science & Technology. The experimental methods were also performed in accordance with their guidelines and regulations (permit number: 2016–03–0007).
Microbial analysis of caecum
The caecum microbiota was analysed by real-time PCR-based gut low-density array (GULDA)58. Cellular DNA extraction was conducted using the QIAGEN Stool mini kit (Hilden, Germany) according to the manufacturer’s instructions (except inhibitor EX), including bead-beating steps (0.1, 1 mm zirconium). Relative quantification of 16 s rRNA gene target primers was conducted in the following bacterial groups: Firmicutes, Lactobacillus spp., Clostridium cluster IV, Clostridium butyricum, Bacteroidetes, Bacteroides, Prevotella, Bifidobacteria, Enterobacteriaceae, Escherichia coli, and Desulfovibrio spp.
Sample preparation and MS-based metabolite profiling
For metabolite extraction, finely chopped skin tissues (2 × 3 cm) and caecum samples stored in the deep freezer were used immediately. Samples were homogenized in 1 mL of methanol using an MM400 mixer mill (Retch, Haan, Germany) with a frequency of 30 s−1 for 10 min using zirconium beads. The homogenates were kept at 4 °C for 60 min, then centrifuged (12,578 × g, 4 °C, 10 min). The filtered (0.2-μm polytetrafluoroethylene filter) supernatants were dried under vacuum using a speed-vacuum concentrator (Biotron, Gyeonggi-Do, Korea). The chamber temperature of speed-vacuum concentrator was set at 30 °C. Dried extracts were further analysed by two different MS-based methods i.e., GC-TOF MS and UPLC-Q-TOF-MS. During MS analysis, samples were analysed randomly. The operational conditions for GC-TOF-MS analysis and UPLC-Q-TOF-MS were described in our previous study59.
Clinical observation of skin tissues
The dorsal skin of hairless mice was photographed before sacrifice. The erythema index of the dorsal skin of hairless mice was measured using a Mexameter 18 and Corneometer 825 before sacrifice.
Data processing and multivariate statistical analysis
The data processing procedure of MS-derived raw data, multivariate analysis method, and metabolite identifications were performed as described in our previous study60. A correlation map was constructed using the MeV software package (http://www.tm4.org/) based on correlation coefficient values calculated with PASW Statistics 18 software.
Data availability
All data generated or analysed during this study are included in this published article and its Supplementary Information Files.
References
Hervert-Hernandez, D. & Goñi, I. Dietary polyphenols and human gut microbiota: a review. Food Rev. Int. 27, 154–169 (2011).
Jin, J. S., Touyama, M., Hisada, T. & Benno, Y. Effects of green tea consumption on human fecal microbiota with special reference to Bifidobacterium species. Microbiol. Immunol. 56, 729–739 (2012).
Yuan, X. et al. Green tea liquid consumption alters the human intestinal and oral microbiome. Mol. Nutr. Food Res. 62, 1800178 (2018).
Zhang, X. et al. Metagenomics analysis of gut microbiota modulatory effect of green tea polyphenols by high fat diet-induced obesity mice model. J. Funct. Food. 46, 268–277 (2018).
Wang, L. et al. The effect of green tea polyphenols on gut microbial diversity and fat deposition in C57BL/6J HFA mice. Food Funct. 7, 4956–4966 (2016).
Stahl, W., Heinrich, U., Aust, O., Tronnier, H. & Sies, H. Lycopene-rich products and dietary photoprotection. Photochem. Photobiol. Sci. 5, 238–242 (2006).
Rhodes, L. E. et al. Oral green tea catechin metabolites are incorporated into human skin and protect against UV radiation-induced cutaneous inflammation in association with reduced production of pro-inflammatory eicosanoid 12-hydroxyeicosatetraenoic acid. Br. J. Nutr. 110, 891–900 (2013).
Lu, Y. P. et al. Stimulatory effect of oral administration of green tea or caffeine on ultraviolet light-induced increases in epidermal wild-type p53, p21 (WAF1/CIP1), and apoptotic sunburn cells in SKH-1 mice. Cancer Res. 60, 4785–4791 (2000).
Arakaki, A. K., Skolnick, J. & McDonald, J. F. Marker metabolites can be therapeutic targets as well. Nature 456, 443 (2008).
Zhang, A., Sun, H., Wang, P., Han, Y. & Wang, X. Recent and potential developments of biofluid analyses in metabolomics. J. Proteomics 75, 1079–1088 (2012).
Turnbaugh, P. J. & Gordon, J. I. An invitation to the marriage of metagenomics and metabolomics. Cell 134, 708–713 (2008).
Lu, K. et al. Arsenic exposure perturbs the gut microbiome and its metabolic profile in mice: an integrated metagenomics and metabolomics analysis. Environ. Health Perspect. 122, 284 (2014).
Abrahamsson, T. R. et al. Low diversity of the gut microbiota in infants with atopic eczema. J. Allergy Clin. Immunol. 129, 434–440 (2012).
Arck, P. et al. Is there a ‘gut–brain–skin axis’? Exp. Dermatol. 19, 401–405 (2010).
Jung, E. S. et al. The green tea modulates large intestinal microbiome and exo/endogenous metabolome altered through chronic UVB-exposure. PLoS One 12, e0187154 (2017).
Venables, M. C., Hulston, C. J., Cox, H. R. & Jeukendrup, A. E. Green tea extract ingestion, fat oxidation, and glucose tolerance in healthy humans. Am. J. Clin. Nutr. 87, 778–784 (2008).
Hodgson, A. B. et al. Acute effects of green tea extract intake on exogenous and endogenous metabolites in human plasma. J. Agric. Food Chem. 62, 1198–1208 (2014).
Cabrera, C., Artacho, R. & Giménez, R. Beneficial effects of green tea—a review. Am. Coll. Nutr. 25, 79–99 (2006).
Graham, H. N. Green tea composition, consumption, and polyphenol chemistry. Prev. Med. 21, 334–350 (1992).
Xu, J. et al. The influence of selenium on the antioxidant activity of green tea. J. Sci. Food Agric. 83, 451–455 (2003).
Higashi‐Okai, K., Taniguchi, M. & Okai, Y. Potent antioxidative activity of non‐polyphenolic fraction of green tea (Camellia sinensis)–association with pheophytins a and b. J. Sci. Food Agric. 80, 117–120 (2000).
Vernocchi, P., Del Chierico, F. & Putignani, L. Gut microbiota profiling: metabolomics based approach to unravel compounds affecting human health. Front. Microbiol. 7, 1144 (2016).
Clydesdale, G. J., Dandie, G. W. & Muller, H. K. Ultraviolet light induced injury: immunological and inflammatory effects. Immunol. Cell. Biol. 79, 547–568 (2001).
Lye, H. S., Alias, A. K., Rusul, G. & Liong, M. T. Enhanced cholesterol removal ability of lactobacilli via alteration of membrane permeability upon ultraviolet radiation. Ann. Microbiol. 62, 1709–1721 (2012).
Sato, R. & Tanaka, M. Intestinal distribution and intraluminal localization of orally administered Clostridium butyricum in rats. Microbiol. Immunol. 41, 665–671 (1997).
Brown, K. L. Control of bacterial spores. Br. Med. Bull. 56, 158–171 (2000).
Ferraris, L., Butel, M. J. & Aires, J. Antimicrobial susceptibility and resistance determinants of Clostridium butyricum isolates from preterm infants. Int. J. Antimicrob. Agents 36(5), 420–423 (2010).
Jay, J. M., Loessner, M. J., & Golden, D. A. Radiation protection of foods, and nature of microbial radiation resistance. (ed. Jay J. M.) Modern Food Microbiology, Springer, Boston, MA, 371–394 (2005).
Takahashi, M. et al. The effect of probiotic treatment with Clostridium butyricum on enterohemorrhagic Escherichia coli O157: H7 infection in mice. FEMS Immunol. Med. Microbiol. 41, 219–226 (2004).
Ichikawa, H. et al. Probiotic bacteria stimulate gut epithelial cell proliferation in rat. Dig. Dis. Sci. 44, 2119–2123 (1999).
Zhang, H. Q. et al. Therapeutic effects of Clostridium butyricum on experimental colitis induced by oxazolone in rats. World J. Gastroenterol. 15, 1821–1828 (2009).
Bouilly‐Gauthier, D. et al. Clinical evidence of benefits of a dietary supplement containing probiotic and carotenoids on ultraviolet‐induced skin damage. Br. J. Dermatol. 163, 536–543 (2010).
Lee, D. E. et al. Clinical evidence of effects of Lactobacillus plantarum HY7714 on skin aging: a randomized, double blind, placebo-controlled study. J. Microbiol. Biotechnol. 25, 2160–2168 (2015).
Sugimoto, S. et al. Photoprotective effects of Bifidobacterium breve supplementation against skin damage induced by ultraviolet irradiation in hairless mice. Photodermatol. Photoimmunol. Photomed. 28, 312–319 (2012).
Ishii, Y. et al. Oral administration of Bifidobacterium breve attenuates UV-induced barrier perturbation and oxidative stress in hairless mice skin. Arch. Dermatol. Res. 306, 467–473 (2014).
Nermes, M., Kantele, J. M., Atosuo, T. J., Salminen, S. & Isolauri, E. Interaction of orally administered Lactobacillus rhamnosus GG with skin and gut microbiota and humoral immunity in infants with atopic dermatitis. Clin. Exp. Allergy 41, 370–377 (2011).
Rather, I. A. et al. Park, Probiotics and atopic dermatitis: an overview. Front. Microbiol. 7, 507 (2016).
Clemente, J. C., Ursell, L. K., Parfrey, L. W. & Knight, R. The impact of the gut microbiota on human health: an integrative view. Cell. 148, 1258–1270 (2012).
Dunn, W. B. & Ellis, D. I. Metabolomics: current analytical platforms and methodologies. Trac-Trends. Anal. Chem. 24, 285–294 (2005).
Barresi, C. et al. Increased sensitivity of histidinemic mice to UVB radiation suggests a crucial role of endogenous urocanic acid in photoprotection. J. Invest. Dermatol. 131, 188–194 (2011).
Kim, E. J. et al. UV decreases the synthesis of free fatty acids and triglycerides in the epidermis of human skin in vivo, contributing to development of skin photoaging. J. Dermatol. Sci. 57, 19–26 (2010).
Feingold, K. R. The outer frontier: the importance of lipid metabolism in the skin. J. Lipid Res. 50(Supplement), S417–S422 (2009).
Ryborg, A. K., Grøn, B. & Kragballe, K. Increased lysophosphatidylcholine content in lesional psoriatic skin. Br. J. Dermatol. 133, 398–402 (1995).
Yahagi, S., Koike, M., Okano, Y. & Masaki, H. Lysophospholipids improve skin moisturization by modulating of calcium‐dependent cell differentiation pathway. Int. J. Cosmetic Sci. 33, 251–256 (2011).
Heinrich, U., Moore, C. E., De Spirt, S., Tronnier, H. & Stahl, W. Green tea polyphenols provide photoprotection, increase microcirculation, and modulate skin properties of women. J. Nutr. 141, 1202–1208 (2011).
Jung, E. S. et al. A metabolomics approach shows that catechin-enriched green tea attenuates ultraviolet B-induced skin metabolite alterations in mice. Metabolomics 11, 861–871 (2015).
Elmets, C. A. et al. Cutaneous photoprotection from ultraviolet injury by green tea polyphenols. J. Am. Acad. Dermatol. 44, 425–432 (2001).
Türkoğlu, M., Uğurlu, T., Gedik, G., Yılmaz, A. M. & Yalçin, A. S. In vivo evaluation of black and green tea dermal products against UV radiation. Drug Discov. Ther. 4, 362–367 (2010).
Jeon, H. Y., Kim, J. K., Kim, W. G. & Lee, S. J. Effects of oral epigallocatechin gallate supplementation on the minimal erythema dose and UV-induced skin damage. Skin Pharmacol. Physiol. 22, 137–141 (2009).
Chow, H. H. S. et al. Pharmacokinetics and safety of green tea polyphenols after multiple-dose administration of epigallocatechin gallate and polyphenon E in healthy individuals. Clin. Cancer Res. 9, 3312–3319 (2003).
Camouse, M. M., Hanneman, K. K., Conrad, E. P. & Baron, E. D. Protective effects of tea polyphenols and caffeine. Expert Rev. Anticancer Ther. 5, 1061–1068 (2005).
Saeed, M. et al. Green tea (Camellia sinensis) and l-theanine: Medicinal values and beneficial applications in humans—A comprehensive review. Biomed. Pharmacother. 95, 1260–1275 (2017).
Svobodova, A. R. et al. Acute exposure to solar simulated ultraviolet radiation affects oxidative stress-related biomarkers in skin, liver and blood of hairless mice. Biol. Pharm. Bull. 34, 471–479 (2011).
Sharma, M. R., Werth, B. & Werth, V. P. Animal models of acute photodamage: comparisons of anatomic, cellular and molecular responses in C57BL/6J, SKH1 and Balb/c mice. Photochem. Photobiol. 87, 690–698 (2011).
Lew, L. C. & Liong, M. T. Bioactives from probiotics for dermal health: functions and benefits. J. Appl. Microbiol. 114, 1241–1253 (2013).
Hullar, M. A. & Fu, B. C. Diet, the gut microbiome, and epigenetics. Cancer J. 20, 170–175 (2014).
Nicholson, J. K. et al. Host-gut microbiota metabolic interactions. Science, 1223813 (2012).
Bergström, A. et al. Introducing Gut Low-Density Array (GULDA) – a validated approach for qPCR-based intestinal microbial community analysis. FEMS microbial. Lett. 337, 38–47 (2012).
Suh, D. H. et al. In vivo metabolomics interpretation of the anti-obesity effects of hyacinth bean (Dolichos lablab L.) administration in high-fat diet mice. Mol. Nutr. Food Res. 61, 1600895 (2017).
Jung, E. S. et al. Integrative metabolomic analysis reveals diet supplementation with green tea alleviates UVB-damaged mouse skin correlated with ascorbate metabolism and urea cycle. Metabolomics 13, 82 (2017).
Acknowledgements
This research was supported by the Bio & Medical Technology Development Program of the National Research Foundation (NRF) funded by the Korean government (MSIT) (No. 2016M3A9A5923160), and by the Bio & Medical Technology Development Program of the National Research Foundation (NRF) funded by the Ministry of Science & ICT(NRF-2018M3A9F3021968).
Author information
Authors and Affiliations
Contributions
E.S.J. performed experiments, analysed metabolomics data, and wrote the manuscript. J.i.P. performed animal experiments. H.P. and W.H. performed microbial community analysis. J.S.H. and C.H.L. supervised and took complete responsibility for this project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jung, E.S., Park, J.i., Park, H. et al. Seven-day Green Tea Supplementation Revamps Gut Microbiome and Caecum/Skin Metabolome in Mice from Stress. Sci Rep 9, 18418 (2019). https://doi.org/10.1038/s41598-019-54808-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-54808-5
This article is cited by
-
Phenolic Compounds Promote Diversity of Gut Microbiota and Maintain Colonic Health
Digestive Diseases and Sciences (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.