Abstract
Some symbiont species are highly host-specific, inhabiting only one or a very few host species, and typically have limited dispersal abilities. When they do occur on multiple host species, populations of such symbionts are expected to become genetically structured across these different host species, and this may eventually lead to new symbiont species over evolutionary timescales. However, a low number of dispersal events of symbionts between host species across time might be enough to prevent population structure and species divergence. Overall, processes of evolutionary divergence and the species status of most putative multi-host symbiont systems are yet to be investigated. Here, we used DNA metabarcoding data of 6,023 feather mites (a total of 2,225 OTU representative sequences) from 147 infracommunities (i.e., the assemblage consisting of all mites of different species collected from the same bird host individual) to investigate patterns of population genetic structure and species status of three different putative multi-host feather mite species Proctophyllodes macedo Vitzthum, 1922, Proctophyllodes motacillae Gaud, 1953, and Trouessartia jedliczkai (Zimmerman, 1894), each of which inhabits a variable number of different closely related wagtail host species (genus Motacilla). We show that mite populations from different host species represent a single species. This pattern was found in all the mite species, suggesting that each of these species is a multi-host species in which dispersal of mites among host species prevents species divergence. Also, we found evidence of limited evolutionary divergence manifested by a low but significant level of population genetic structure among symbiont populations inhabiting different host species. Our study agrees with previous studies showing a higher than expected colonization opportunities in host-specific symbionts. Indeed, our results support that these dispersal events would allow the persistence of multi-host species even in symbionts with limited dispersal capabilities, though additional factors such as the geographical structure of some bird populations may also play a role.
Similar content being viewed by others
Introduction
Symbiont organisms tend to be highly adapted to live on their native hosts1,2,3,4. One might expect that the level of specialisation of symbionts would drive symbiont species to be highly host-specific (i.e., to inhabit a single host species, in the most extreme scenario)5,6,7,8,9,10. However, symbiont species inhabiting more than a single host species are relatively frequent across different groups of symbionts1,2.
In general, populations of symbionts inhabiting different host species are expected to become genetically structured (or sorted) by host species and to lead to new symbiont races and species over evolutionary timescales8,11,12,13,14,15,16. Among the agents contributing to the divergence process, the symbiont mode of transmission (i.e., vertical vs horizontal) is one of the main factors contributing to symbionts population structure and divergence2,15,17,18. Vertical transmission, in which symbionts are transmitted from parents to offspring, is only possible between symbiont populations inhabiting a particular host species (though it may be possible if a hybridisation event occurs19). Thus, populations of symbionts whose main mode of transmission is vertical are expected to become quickly structured by host species and eventually speciate on that host species1,2,17,20,21,22,23. Alternatively, in horizontally transmitted symbionts, dispersal goes from one host species to another and tends to erode the population genetic structure among-host populations1,2,17,20,21,22,23. Thus, populations of symbionts with an elevated rate of horizontal transmission are expected to exhibit lower among-host population structure, not necessarily speciating on each host species1,2,17,20,21,22,23.
Highly host-specific symbionts, in which vertical transmission is generally the main mode of dispersal, are however known to contain multi-host species (e.g., Doña et al.24). One explanation for this pattern is that despite vertical transmission being the main mode of dispersal, dispersal events (either periodic or episodic) among populations on different host species might be enough to prevent populations from becoming genetically structured and diverging. Thus, these dispersal events would sustain multi-host species in host-specific symbionts2. Indeed, some recent studies have indicated that colonisation opportunities may be underestimated for some of these symbionts with limited transmission capabilities, and clade-limited host-switching may occur frequently25,26,27,28. Alternatively, multi-host symbiont species from relatively host-specific symbionts could represent undiscovered cryptic species29,30,31,32,33. Indeed, cryptic species might be common in small-bodied symbionts inhabiting closely related hosts, because these hosts can offer a similar habitat, and therefore, there might be strong selective pressure for highly similar symbiont phenotypes34. Overall, the population structure and species status of multi-host species of most host-specific symbiont systems are yet to be well understood.
Feather mites are host-specific symbionts with limited dispersal capabilities, but also with some clade-limited host-switching25,26,27,35,36,37,38,39. Previous studies have documented multi-host species35,36,37,40 and cases of morphologically-cryptic but genetically different mite species35,36,37,40. Also, genetic structure has been observed (1) among populations of mites at the level of infrapopulations41, (2) among feather mite populations inhabiting different bird host species35,36,37,40 and (3) among different populations of the same passerine bird species41. Most putative multi-host bird-mite systems remain, however, unexplored. Large scale studies, which might allow precise assessments of population genetic structure and species status41, are nonexistent.
Here, we investigated the process of evolutionary divergence and species status of populations of feather mite multi-host species in the genera Proctophyllodes and Trouessartia; each of which inhabits different closely related species of European wagtails hosts (genus Motacilla). Specifically, we used DNA metabarcoding data from 6,023 individual mites (a total of 2,225 OTU representative sequences) representing 146 infracommunities of Proctophyllodes motacillae, Trouessartia jedliczkai, and Proctophyllodes macedo inhabiting the white wagtail Motacilla alba Linnaeus, 1758, western yellow wagtail Motacilla flava Linnaeus 1758, and grey wagtail Motacilla cinerea Tunstall, 1771.
Results
Final mitochondrial COI sequence alignments contained 516 sequences of 388 bp for P. macedo, 680 sequences of 390 bp for P. motacillae, and 1,029 sequences of 388 bp for T. jedliczkai.
For P. motacillae, P. macedo, and T. jedliczkai, species delimitation analyses each supported the existence of a single mite species despite inhabiting different number of host species. First, when evaluating whether mite populations on different host species could be different species, we consistently found an overlap between within host - and among host infrapopulations genetic distances (Fig. 1). Accordingly, we did not find any suitable threshold for “species” delimitation nor high percentages of successful identifications when using the best close match method (38%, 32%, and 32% for P. motacillae, P. macedo, and T. jedliczkai, respectively; Fig. S1). Second, for each mite species studied, we found a single partition (i.e., a single hypothetical species) in the ABGD analyses.
Boxplots showing within and among host infrapopulation genetic distances of the different mite species. Dashed grey line shows a reference interspecific threshold for feather mites35.
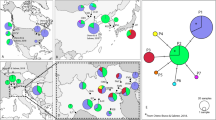
Population genetic analyses revealed variable but overall low levels of genetic structure among symbiont populations inhabiting different host species. AMOVA and PCA analyses showed that most of the variation was due to the intragroup variation (70%, 73%, and 52% for P. motacillae, P. macedo, and T. jedliczkai, respectively; P < 0.01 in all cases; Fig. 2, Table 1). Also, most of the major haplotypes were confined to a single host species (Fig. S2). In all mite species, we found a low, but still substantial, number of haplotypes shared between mite infrapopulations on different host species. Specifically, we found ten P. motacillae haplotypes shared between M. alba and M. flava, five T. jedliczkai haplotypes shared between M. alba and M. flava, three P. macedo haplotypes shared between M. alba and M. flava, and one P. motacillae haplotype shared between M. flava and M. cinerea. Lastly, in all mite species except T. jedliczkai, we did not find haplotypes significantly clustered by host species in haplotype networks (Fig. S2).
Nucleotide and haplotype diversity values were high for most species and populations but differed between mite populations of the same mite species inhabiting different hosts (Table 2). These differences, however, did not follow an apparent host-driven pattern across mite species (Table 2, Fig. S2). Also, in most mite populations on different host species, we found signatures of population expansion evidenced by small, negative Tajima’s D values, small, positive R2 values, and star-shaped haplotype networks (Table 2, Fig. S2).
Discussion
For each of the three multi-host feather mite species distributed across three closely related host species, species delimitation analyses supported the existence of a single species inhabiting different host species. Our results agree with previous morphological assessments carried out on these mite species inhabiting these bird species (Doña et al.24 and references therein). Notably, however, in all the symbiont species studied, we found evidence of population divergence manifested by a low but significant level of population genetic structure among symbionts populations inhabiting different host species. Altogether, these results add to previous findings suggesting that feather mites can potentially disperse among host species more than expected based on their known biology, and suggest the presence of small scale gene flow between populations of feather mites on different host species24,25,27. Nevertheless, additional factors such as the geographical structure of some bird populations, introgression, or incomplete lineage sorting may be playing a role in explaining some of the patterns found here (e.g., P. macedo and T. jedliczkai haplotype distributions; Fig. S2)16,42.
The levels of genetic structuring found for these multi-host mite species are higher than those found in multi-host symbionts with higher dispersal capabilities, e.g., this study: Fst (Phist) = 0.26–0.48 vs 0.01–0.20 between populations of conspecific lice on different host species17,18. This result supports that differences in dispersal ability shape patterns of population structure and evolution17,18,19. In the same vein, the degree in which mite populations were structured by host species was variable across the three mite species, thus again supporting that symbiont-specific traits such as dispersal capabilities (e.g., phoretic vs non-phoretic or strict vertical vs horizontal dispersal) might influence genetic structuring patterns18,23,37,43,44,45. Specifically, some of the patterns of genetic structuring found here (see below), even though not directly comparable, are similar to those of previous studies on feather mite species from the same genera32. In particular, we found that Trouessartia populations were more structured than those of Proctophyllodes, as was previously found in two different species of the same genera, e.g., 50% vs 19% of the variation explained by the among-infrapopulation genetic structure component in Trouessartia bifurcata (Trouessart) vs Proctophyllodes sylviae32. Nonetheless, contrasting effective population sizes46 or processes such as hybridisation19 may distort genetic estimates from a single-locus such as the COI used in this study, and therefore, studies using multilocus population genomic approaches (e.g., SNPs15) are encouraged.
Different non-mutually exclusive scenarios can explain the dispersal events that have occurred or still do occur between host species, and that are behind the existence of single species of mites across these host species. First, mites may have recently colonised these host species, and hence they may not have diverged yet (i.e., a recent host switch without ongoing gene flow). These colonisations may have occurred at different times, and therefore, explain the differences in the degree of genetic structuring among mite species. However, they could have also colonised these different host species nearly simultaneously, and thus the differences in genetic structuring may be the consequence of mite-specific traits (e.g., a species-specific dispersal rate). Small populations are especially vulnerable to genetic drift47, and this applies particularly to mitochondrial markers because of their lower effective population size compared to diploid nuclear loci, as well as to symbionts colonising a new host species, because only a few haplotypes are likely found in new populations21,48,49,50. Therefore, under a recent colonisation scenario, we might expect low levels of genetic diversity, and multiple recent haplotypes to be descendant from a clustered pool of ancestral haplotypes32,47. Instead, we found that genetic diversity was relatively high for most within-host populations, and multiple haplotypes were shared all along the haplotype networks. Furthermore, mite species did not consistently exhibit higher or lower genetic diversity values on particular host species (Table 2), and some within-host populations (e.g., T. jedlizckai in M. flava) showed low levels of genetic diversity and a star-shaped haplotype network with multiple recent haplotypes descended from a single common ancestral haplotype (Table 2, Fig. S2).
Second, mites may have been associated only with one of these or another wagtail species24 and colonised different Motacilla species afterwards while maintaining gene flow with the source population (i.e., incomplete host-switching51). If an incomplete host switch has occurred, we would expect an increase in the population size of the colonising symbiont32. Interestingly, we have found evidence of population growth in all the populations studied (Table 1, Fig. S2). These population growth signatures may support a scenario of incomplete host-switching32, which may also be following recent studies on the dynamism of bird feather mites associations25,27,37,52.
Finally, it is possible that these mites were inherited from the common ancestor of all these wagtail species and maintained gene flow among diverging host species as the wagtail species were diverging from each other. Indeed, this scenario could be supported by the fact that all these wagtail hosts diverged very recently from each other53. Furthermore, even low levels of gene flow54,55,56 among populations of mites on different host species could be enough to prevent species divergence (i.e., failure to speciate20). In a failure to speciate scenario, the symbiont population size is expected to remain constant. Hence, the signatures of population growth that were observed in all species of mites do not support this scenario32. However, over long time periods, severe bottlenecks are expected given the population dynamics of these symbionts, and population growth after bottlenecks may also explain the signatures found in the demographic tests37,52.
Materials and Methods
We followed the sampling protocol and the DNA metabarcoding pipeline of Vizcaíno et al.57 and Doña et al.27. In brief, we collected feather mites from live individuals of M. alba, M. flava, and M. cinerea in different localities in Spain (Table S1). A total of 6,023 mites from 146 feather mite infracommunities were collected from the host species M. alba (n = 2,660), M. flava (n = 3,255), and M. cinerea (n = 108) (Table S1). Mites were sampled with a flattened preparation needle or with an ethanol-impregnated cotton swab. Afterward, we extracted DNA from all the collected mites of each individual bird (i.e., infracommunity) using the HotSHOT method58. The ethanol was first evaporated, then a solution of NaOH 1-M was added to the dry wells, incubated at 95 °C and finally neutralized with equivalent amounts of Tris-Cl. For each infracommunity, we constructed the DNA amplicon libraries by amplifying a region of the mitochondrial COI gene in a two-step PCR (see Vizcaino et al.57 for details). We followed the Illumina protocol for bacterial 16S DNA metabarcoding. The 146 libraries were pooled together (i.e., multiplexed) and sequenced in one MiSeq PE300 run (MiSeq Reagent Kit v3).
After sequence pre-processing, we ended up with 1,394,138 sequences (statistics per sample, i.e., infracommunity: mean = 9,548; min = 109; max = 174,412) which were assigned to five different mite species (Table S1). Proctophyllodes motacillae was the most prevalent species [Number of infrapopulations (N) = 95; 65%], followed by T. jedliczkai (N = 46; 31%), and P. macedo (N = 37; 25%) (Table 1). Pteronyssoides motacillae Mironov, 1985 (N = 1; 0.7%) and Proctophyllodes sylviae Gaud, 1957 (N = 2; 1.4%) were also found but not included in further analyses because of their extremely low prevalence (Table S1).
The fastq reads were quality-checked with FastQC59. Then, we imported the reads into Geneious v.8.1.760 for quality-trimming analyses. We trimmed a region of 36 and 120 bp from R1 and R2 reads, respectively, according to the average Phred score (minimum quality score of 28). The R1 and R2 files were then exported in FASTA format and were concatenated using the fuse.sh script available from the BBmap package v.37.0061. We only saved the concatenated sequences with the maximum possible length. To label the sequences with the sample identifier and merge them into a single file, we used the split_libraries.py script included in the pipeline of QIIME v.1.9.062. Then, we selected the Operational Taxonomic Units (OTUs) with a 100% similarity threshold using a de novo clustering method and the UCLUST algorithm63. We also used a filter to eliminate or minimize mistagging events (e.g., only OTUs with more than 100 identical reads were kept), and after this, we selected the most abundant sequence of each OTU as the representative sequence of that OTU. We made the taxonomic assignment of each representative sequence using the assign_taxonomy.py script of QIIME. The assignment was done with the RDP classifier64 and a minimum confidence score of 96.6% against a reference database (Doña et al.35). Lastly, we aligned the representative sequences of each mite species using MAFFT v7.38865 with default parameters and refined the alignments with trimAL v1.266 using the “gappyout” method.
Unless otherwise specified, all the analyses were done in R v3.567. We conducted species delimitation analyses for the mite species P. macedo, P. motacillae, and T. jedlizcai. First, we used the local minima function of the package SPIDER v.1.368 to estimate an optimal threshold for delimiting species. We also used the web version of ABGD69 to review our species hypotheses. We use the default settings on a set of prior minimum genetic distances ranging from 0.001 to 0.1. Lastly, we evaluated the accuracy in cataloguing mite sequences to putatively different host-specific species using the genetic distance-based method BCM70. We used the bestCloseMatch function of SPIDER and the genetic threshold previously calculated (see above).
We estimated the population genetic structure of these above-mentioned mite species among mite populations on different host species. First, we conducted analyses of molecular variance (AMOVA) using the poppr.amova function from POPPR v.2.871,72, and we tested for statistical significance using the randtest function of ADE4 v.2.173,74,75,76. We further investigated the genetic structuring among samples, based on genetic distances, through principal components analyses (PCA) with the dudi.pca function of ADE4.
Additionally, we estimated population genetic parameters using PEGAS v.0.177. We used the nuc.div function to calculate nucleotide diversity (π, hereafter genetic diversity78). Also, we calculated parameters to detect population expansions. We calculated Tajima’s D79, using the tajima.test function; and estimated statistical significance assuming that D follows a normal distribution with mean zero and variance one. We also calculated R2 using the R2.test function and evaluated its statistical significance through 1,000 coalescent simulations. Finally, we estimated the haplotype diversity (Hd) using the hap.div function, and used the haplotype and haplonet functions to build haplotype networks and study the mites haplotype distribution.
Data availability
The raw data and the representative sequences have been deposited in Figshare (https://doi.org/10.6084/m9.figshare.7857647; https://figshare.com/s/38abf165a4e5c87ec26f).
References
Poulin, R. Evolutionary ecology of parasites. (Princeton university press, 2011).
Clayton, D. H., Bush, S. E. & Johnson, K. P. Coevolution of life on hosts: integrating ecology and history. (University of Chicago Press, 2015).
Fry, J. D. Trade-offs in fitness on different hosts: evidence from a selection experiment with a phytophagous mite. The american naturalist 136, 569–580 (1990).
Lajeunesse Marc, J. & Forbes Mark, R. Host range and local parasite adaptation. Proceedings of the royal society of london. Series b: biological sciences 269, 703–710 (2002).
Combes, C. Evolution of parasite life cycles. In Toft CA, Aeschlimann A, Bolis L (eds) Parasite-Host Associations: Coexistence or Conflict? 62–82 (Oxford University Press, 1991).
Thompson, J. N. The coevolutionary process. (University of Chicago Press, 1994).
Norton, D. A. & Carpenter, M. A. Mistletoes as parasites: host specificity and speciation. Trends in ecology and evolution 13, 101–105 (1998).
Mccoy, K. D., Boulinier, T., Tirard, C. & Michalakis, Y. Host specificity of a generalist parasite: genetic evidence of sympatric host races in the seabird tick Ixodes uriae. Journal of evolutionary biology 14, 395–405 (2001).
Giorgi, M. S. et al. Causal mechanisms underlying host specificity in bat ectoparasites. Oecologia 138, 648 (2004).
Antonovics, J. et al. The Origin of Specificity by Means of Natural Selection: Evolved and Nonhost Resistance in Host–Pathogen Interactions. Evolution 67, 1–9 (2013).
McCoy, K. D., Boulinier, T., Tirard, C. & Michalakis, Y. Host-dependent genetic structure of parasite populations: differential dispersal of seabird tick host races. Evolution 57, 288–296 (2003).
McCoy, K. D. et al. Recurrent evolution of host-specialized races in a globally distributed parasite. Proceedings of the royal society of london. Series b: biological sciences 272, 2389–2395 (2005).
Malenke, J. R., Johnson, K. P. & Clayton, D. H. Host specialization differentiates cryptic species of feather-feeding lice. Evolution 63, 1427–1438 (2009).
Rivera‐Parra, J. L., Levin, I. I., Johnson, K. P. & Parker, P. G. Lineage sorting in multihost parasites: Eidmanniella albescens and Fregatiella aurifasciata on seabirds from the Galapagos Islands. Ecology and evolution 5, 3264–3271 (2015).
Sweet, A. D. et al. Integrating phylogenomic and population genomic patterns in avian lice provides a more complete picture of parasite evolution. Evolution 72, 95–112 (2017).
Martinů, J., Hypša, V. & Štefka, J. Host specificity driving genetic structure and diversity in ectoparasite populations: Coevolutionary patterns in Apodemus mice and their lice. Ecology and evolution 8, 10008–10022 (2018).
Johnson, K. P., Williams, B. L., Drown, D. M., Adams, R. J. & Clayton, D. H. The population genetics of host specificity: genetic differentiation in dove lice (Insecta: Phthiraptera). Molecular ecology 11, 25–38 (2002).
Sweet, A. D. & Johnson, K. P. The role of parasite dispersal in shaping a host–parasite system at multiple evolutionary scales. Molecular ecology 27, 5104–5119 (2018).
Doña, J., Sweet, A. & Johnson, K. Parasite dispersal influences introgression rate. bioRxiv 527226, https://doi.org/10.1101/527226 (2019).
Johnson, K. P., Adams, R. J., Page, R. D. & Clayton, D. H. When do parasites fail to speciate in response to host speciation? Systematic biology 52, 37–47 (2003).
Huyse, T., Poulin, R. & Theron, A. Speciation in parasites: a population genetics approach. Trends in parasitology 21, 469–475 (2005).
Criscione, C. D., Poulin, R. & Blouin, M. S. Molecular ecology of parasites: elucidating ecological and microevolutionary processes. Molecular ecology 14, 2247–2257 (2005).
Archie, E. A. & Ezenwa, V. O. Population genetic structure and history of a generalist parasite infecting multiple sympatric host species. International journal for parasitology 41, 89–98 (2011).
Doña, J., Proctor, H., Mironov, S. V., Serrano, D. & Jovani, R. Global associations between birds and vane‐dwelling feather mites. Ecology 97, 3242–3242 (2016).
Doña, J. et al. Cophylogenetic analyses reveal extensive host-shift speciation in a highly specialized and host-specific symbiont system. Molecular phylogenetics and evolution 115, 190–196 (2017).
Doña, J., Proctor, H., Mironov, S., Serrano, D. & Jovani, R. Host specificity, infrequent major host switching and the diversification of highly host-specific symbionts: The case of vane-dwelling feather mites. Global ecology and biogeography 27, 188–198 (2018).
Doña, J., Serrano, D., Mironov, S., Montesinos-Navarro, A. & Jovani, R. Unexpected bird–feather mite associations revealed by DNA metabarcoding uncovers a dynamic ecoevolutionary scenario. Molecular ecology 28, 379–390 (2019).
Bourguignon, T. et al. Rampant host switching shaped the termite gut microbiome. Current biology 28, 649–654 (2018).
Smith, M. A., Wood, D. M., Janzen, D. H., Hallwachs, W. & Hebert, P. D. DNA barcodes affirm that 16 species of apparently generalist tropical parasitoid flies (Diptera, Tachinidae) are not all generalists. Proceedings of the national academy of sciences 104, 4967–4972 (2007).
Poulin, R. & Keeney, D. B. Host specificity under molecular and experimental scrutiny. Trends in parasitology 24, 24–28 (2008).
de León, G. P.-P. & Nadler, S. A. What we don’t recognize can hurt us: a plea for awareness about cryptic species. Journal of parasitology 96, 453–464 (2010).
Banks, J. C. & Paterson, A. M. Multi-host parasite species in cophylogenetic studies. International journal for parasitology 35, 741–746 (2005).
de Rojas, M. et al. Evidence of cryptic species in the genus Tinaminyssus (Acari: Rhinonyssidae) based on morphometrical and molecular data. Experimental and applied acarology 75, 355–368 (2018).
Struck, T. H. et al. Finding evolutionary processes hidden in cryptic species. Trends in ecology and evolution 33, 153–163 (2018).
Doña, J. et al. DNA barcoding and minibarcoding as a powerful tool for feather mite studies. Molecular ecology resources 15, 1216–1225 (2015).
Stefan, L. M., Gómez-Díaz, E., Mironov, S. V., González-Solís, J. & McCoy, K. D. “More Than Meets the Eye”: Cryptic Diversity and Contrasting Patterns of Host-Specificity in Feather Mites Inhabiting Seabirds. Frontiers in ecology and evolution 6, 97 (2018).
Dabert, M. et al. Differences in speciation progress in feather mites (Analgoidea) inhabiting the same host: the case of Zachvatkinia and Alloptes living on arctic and long-tailed skuas. Experimental and applied acarology 65, 163–179 (2015).
Doña, J. et al. Vertical transmission in feather mites: insights into its adaptive value. Ecological entomology 42, 492–499 (2017).
Proctor, H. C. Feather mites (Acari: Astigmata): ecology, behavior, and evolution. Annual review of entomology 48, 185–209 (2003).
Dabert, M., Solarczyk, P., Badek, A. & Dabert, J. Taxonomic status of the oligoxenous feather mite species: are we dealing with species in statu nascendi? Phytophaga 14, 425–431 (2005).
Fernández-González, S., Pérez-Rodríguez, A., Proctor, H. C., De la Hera, I. & Pérez-Tris, J. High diversity and low genetic structure of feather mites associated with a phenotypically variable bird host. Parasitology 1–8 (2018).
Lack, J. B., Pfau, R. S. & Wilson, G. M. Demographic history and incomplete lineage sorting obscure population genetic structure of the Texas mouse (Peromyscus attwateri). Journal of mammalogy 91, 314–325 (2010).
Appelgren, A. S. C., Saladin, V., Richner, H., Doligez, B. & McCoy, K. D. Gene flow and adaptive potential in a generalist ectoparasite. BMC Evolutionary Biology 18, 99 (2018).
Bourguignon, T. et al. Rampant Host Switching Shaped the Termite Gut Microbiome. Current biology 28, 649–654.e2 (2018).
Fernández-González, S., Pérez-Rodríguez, A., Proctor, H. C., Hera, I. Dla & Pérez-Tris, J. High diversity and low genetic structure of feather mites associated with a phenotypically variable bird host. Parasitology 145, 1243–1250 (2018).
Klimov, P. B., Skoracki, M. & Bochkov, A. V. Cox1 barcoding versus multilocus species delimitation: validation of two mite species with contrasting effective population sizes. Parasites & vectors 12, 8 (2019).
Mayr, E. Systematics and the origin of species. (Columbia University Press, 1942).
Ebert, D. Experimental Evolution of Parasites. Science 282, 1432–1436 (1998).
Avise, J. C. et al. Intraspecific phylogeography: the mitochondrial DNA bridge between population genetics and systematics. Annual review of ecology and systematics 18, 489–522 (1987).
Zink, R. M. & Barrowclough, G. F. Mitochondrial DNA under siege in avian phylogeography. Molecular ecology 17, 2107–2121 (2008).
Clayton, D., Al-Tamimi, S. & Johnson, K. P. The ecological basis of coevolutionary history. In Tangled Trees. Phylogeny, Cospeciation, and Coevolution 310–341 (University of Chicago Press).
Doña, J., Moreno‐García, M., Criscione, C. D., Serrano, D. & Jovani, R. Species mtDNA genetic diversity explained by infrapopulation size in a host-symbiont system. Ecology and evolution 5, 5801–5809 (2015).
Harris, R. B., Alström, P., Ödeen, A. & Leaché, A. D. Discordance between genomic divergence and phenotypic variation in a rapidly evolving avian genus (Motacilla). Molecular phylogenetics and evolution 120, 183–195 (2018).
Spieth, P. T. Gene flow and genetic differentiation. Genetics 78, 961–965 (1974).
Kimura, M. & Ohta, T. Theoretical aspects of population genetics. (Princeton University Press, 1971).
Lewontin, R. C. The genetic basis of evolutionary change. vol. 560 (Columbia University Press New York, 1974).
Vizcaíno, A. et al. Enabling large-scale feather mite studies: an Illumina DNA metabarcoding pipeline. Experimental and applied acarology 76, 81–97 (2018).
Truett, G. E. et al. Preparation of PCR-quality mouse genomic DNA with hot sodium hydroxide and tris (HotSHOT). BioTechniques 29(52), 54 (2000).
Andrews, S. FastQC: a quality control tool for high throughput sequence data. (Babraham Bioinformatics, Babraham Institute, Cambridge, United Kingdom, 2010).
Kearse, M. et al. Geneious Basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28, 1647–1649 (2012).
Bushnell, B. BBMap: a fast, accurate, splice-aware aligner (2014).
Caporaso, J. G. et al. QIIME allows analysis of high-throughput community sequencing data. Nature methods 7, 335 (2010).
Edgar, R. C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26, 2460–2461 (2010).
Wang, Q., Garrity, G. M., Tiedje, J. M. & Cole, J. R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Applied and environmental microbiology 73, 5261–5267 (2007).
Katoh, K. & Standley, D. M. MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Molecular biology and evolution 30, 772–780 (2013).
Capella-Gutiérrez, S., Silla-Martínez, J. M. & Gabaldón, T. trimAl: a tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinformatics 25, 1972–1973 (2009).
RCore Team. R: A language and environment for statistical computing (2013).
Brown, S. D. J. et al. Spider: An R package for the analysis of species identity and evolution, with particular reference to DNA barcoding. Molecular ecology resources 12, 562–565 (2012).
Puillandre, N., Lambert, A., Brouillet, S. & Achaz, G. ABGD, Automatic Barcode Gap Discovery for primary species delimitation. Molecular ecology 21, 1864–1877 (2012).
Meier, R., Shiyang, K., Vaidya, G. & Ng, P. K. L. DNA Barcoding and Taxonomy in Diptera: A Tale of High Intraspecific Variability and Low Identification Success. Systematic biology 55, 715–728 (2006).
Kamvar, Z. N., Tabima, J. F. & Grünwald, N. J. Poppr: an R package for genetic analysis of populations with clonal, partially clonal, and/or sexual reproduction. PeerJ 2, e281 (2014).
Kamvar, Z. N., Brooks, J. C. & Grünwald, N. J. Novel R tools for analysis of genome-wide population genetic data with emphasis on clonality. Frontiers in genetics 6, 208 (2015).
Bougeard, S. & Dray, S. Supervised multiblock analysis in R with the ade4 package. Journal of statistical software 86, 1–17 (2018).
Dray, S. & Dufour, A.-B. The ade4 package: implementing the duality diagram for ecologists. Journal of statistical software 22, 1–20 (2007).
Chessel, D., Dufour, A. B. & Thioulouse, J. The ade4 package-I-One-table methods. R news 4, 5–10 (2004).
Dray, S., Dufour, A. B. & Chessel, D. The ade4 package-II: Two-table and K-table methods. R news 7, 47–52 (2007).
Paradis, E. pegas: an R package for population genetics with an integrated–modular approach. Bioinformatics 26, 419–420 (2010).
Nei, M. Molecular evolutionary genetics. (Columbia university press, 1987).
Tajima, F. Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 123, 585–595 (1989).
Acknowledgements
The authors especially thank José Luis Arroyo, Francisco Jiménez-Cazalla, and José Luis Garzón Pérez for help in collecting samples. RA acknowledges the Parc Natural del Delta de l’Ebre for supporting the long-term study on the migration of the yellow wagtail. This work was supported by the MINECO CGL2011-24466 to RJ and CGL2015-69650-P to RJ and DS.
Author information
Authors and Affiliations
Contributions
J.D., C.O.M., K.P.J., D.S. and R.J., conceived the study. J.D., C.O.M., D.S., R.A. and R.J., collected the samples. J.D. and C.O.M analyzed the data and wrote the manuscript with the help of all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Doña, J., Osuna-Mascaró, C., Johnson, K.P. et al. Persistence of single species of symbionts across multiple closely-related host species. Sci Rep 9, 17442 (2019). https://doi.org/10.1038/s41598-019-54015-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-54015-2
This article is cited by
-
Horizontal transmission maintains host specificity and codiversification of symbionts in a brood parasitic host
Communications Biology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.