Abstract
The Jeju horse, indigenous to the Jeju Island in Korea may have originated from Mongolian horses. Adaptations to the local harsh environment have conferred Jeju horse with unique traits such as small-sized body, stocky head, and shorter limbs. These characteristics have not been studied previously at the genomic level. Therefore, we sequenced and compared the genome of 41 horses belonging to 6 breeds. We identified numerous breed-specific non-synonymous SNPs and loss-of-function mutants. Demographic and admixture analyses showed that, though Jeju horse is genetically the closest to the Mongolian breeds, its genetic ancestry is independent of that of the Mongolian breeds. Genome wide selection signature analysis revealed that genes such as LCORL, MSTN, HMGA2, ZFAT, LASP1, PDK4, and ACTN2, were positively selected in the Jeju horse. RNAseq analysis showed that several of these genes were also differentially expressed in Jeju horse compared to Thoroughbred horse. Comparative muscle fiber analysis showed that, the type I muscle fibre content was substantially higher in Jeju horse compared to Thoroughbred horse. Our results provide insights about the selection of complex phenotypic traits in the small-sized Jeju horse and the novel SNPs identified will aid in designing high-density SNP chip for studying other native horse breeds.
Similar content being viewed by others
Introduction
Jeju is the largest island in the Korean peninsula1 and has a climate that is distinct from that of the Korean mainland. The Jeju island is home to several indigenous livestock species, such as chickens, pigs, dogs, and horses, which have been bred as closed flock2. Among the indigenous species, the Jeju horse (JH) is one of the most important animals from an historical, cultural, and economical perspective3. Horses were domesticated in the Eurasian steppe over 6,000 years ago4 and have had a lasting impact on human civilization and lifestyle by influencing mobility, trade, food, and warfare5. The JH is a landrace with extensive history; it is the only Korean native horse3 and is registered with the Domestic Animal Diversity Information System of the Food and Agriculture Organization (FAO)6. Starting from the early 1970s, the population of the JH declined rapidly and was close to extinction by early 1980s3. This occurred because the demand for horses declined owing to industrialization and development of modern means of transportation and agricultural machinery7. Realizing the historical and cultural importance of JH and the need for its conservation, the Korean government designated the JH as a Korean National Treasure (No. 347) in 1986, and started a pedigree registry3,7. In 2000, a JH registration agency was established, and currently, approximately 2,500 JH are being raised at local farms7,8.
Several studies have examined the origin of the JH based on historical9, archaeological10, and molecular evidence11,12,13,14; however, the origin of these horses remains unclear. Based on historical records, Nam (1969) suggested that in 1276 AD, 160 horses were introduced to the Jeju Island by the Mongolian Yuan dynasty of China, and that these horses were bred for their warfare capabilities9. However, based on archaeological evidence10,12, horses seem to have inhabited the Jeju Island 2500 years prior to the Mongolian invasion. Moreover, the results of several studies have suggested multiple origins for JH11,13,14,15. Based on restricted fragment length polymorphism (RFLP) analysis of mtDNA, Oh et al. suggested in 1994 that JH are closer to Mongolian wild horses (E. przewalskii)12. However, the genetic origins of the JH are still incompletely understood, because these studies used a limited number of samples and did not conduct whole genome comparative analyses. In this study, we performed a whole genome comparative analysis of the JH and 5 other breeds, including Thoroughbred horses (TB), 3 Mongolian breeds (MH) (Mongolian Galshar (MG), Mongolian Domestic Horse (MD), Mongolian Jarglant (MJ)) and Przewalski’s (PZ) horses. The JH, which is found only in the Jeju island of Korea, is a small-sized hardy horse (Supplemental Figure S1). On average, it has a wither height of ~122 cm, which is shorter than those of the average MH (~140 cm), PZ (~148 cm), and TB horses (~154 cm)6,7,11,16,17,18. JH are gentle, hardy, and of a healthy constitution, and are resistant to diseases and stress derived from the harsh environment they live in3. The JH shows remarkable endurance and stamina and are raised for meat, farm labour, and riding19, however they are not intensively selected for any one purpose20,21. While TB have been intensively selected for speed, stamina, and agility22, PZ is the only species of wild horse surviving in the world today and is native to Mongolia23,24. The combined effect of environmental changes and human activities has drastically reduced the PZ population, with only 12 individuals surviving in the middle of the last century24,25,26,27. Currently, the number of PZ has increased to approximately 1,000–2,000; these horses are found in the wild and in zoos across the world24,26. In comparison, the population of MH, which includes breeds such as Galshar, Darkhad, Tes, Myangad, and Jargalant28, is large, with abundant genetic diversity, as they have been an integral part of nomadic pastoralist culture in North Asia. Comparative genomic analysis of these breeds can provide insights about the genetic origin of the JH, and can confirm whether the JH originated from a MH breed. Natural selection, domestication, and adaptation to local environments can cause phenotypic changes via mutations29, while robust selection of beneficial alleles leads to selective sweep29. These genomic footprints, which are left by selection, are known as selection signatures, and can be used to identify loci that were subjected to selection. Although JH was not subjected to artificial selection, the genetic consequences of naturally occurring positive selection are essentially the same as those of artificial selection30,31,32,33. Selective sweep analysis can identify loci that were selected in response to local adaptations. Similarly, comparative transcriptome analysis of JH and TB horses can provide molecular clues and help identify genes that influence traits such as stamina, endurance, hardiness, and small wither height and the poor athletic ability observed in JH.
The aims of this study were to: 1) examine the genetic relationship of the JH and Mongolian horses; 2) understand the demographic history of the JH; and 3) identify the genetic basis of selective and adaptive functional differences between the JH and TB. For this, we conducted whole-genome re-sequencing analysis of 41 horses belonging to 6 breeds including JH, TB, and 3 Mongolian horse breeds (Galshar, Jarglant, and Mongolian domestic horse), and performed RNA sequencing-based comparative transcriptome analysis between TB and JH using the lung, rump muscle, thigh muscle, liver, and heart tissues. We believe the results of this study will provide cues towards the genetic origin of the JH and also identify candidate genes that play a role in the small size and poor athletic performance of JH.
Results and Discussion
Resequencing, and SNP detection in 41 s horses
A total of 16.5 billion initial reads were generated by the Illumina HiSeq 4000 Sequencer, amounting to an average of 3.8 Gbp per library (average of 16.37X fold coverage across the genome, individual fold coverage ranged between 12.26X~23.28X) (Table 1, Supplementary File 1). The highest average fold coverage was obtained for PZ (20.72X), while the lowest average fold coverage was obtained for MG (13.9X). Potential polymerase chain reaction duplications were removed from the aligned reads, and the reads were further modified using Picard toolkit before SNP calling. Approximately, 16.15 billion reads, with an average alignment rate of 97.15%, were mapped against the reference genome, with the highest average alignment rate for TB (98.72%), and the lowest for MJ (96.23%).
Altogether, across the genome of the different breeds, we identified a total of 21,651,273 SNPs (Table 2, Supplemental File S1). The number of SNPs in each breed varied between 9.4 million (TB) and 16.5 million (MD, MJ and MG) (Table 2 and Fig. 1a). Additionally, 5,185,635 (23.95%) of the identified SNPs were novel, when compared against the dbSNP database build 151; this was expected, because the reference genome (EquCab 2.0) was based on a female TB horse34. Amongst the identified SNPs, the highest percentage of novel SNPs was in the Mongolian breeds (~16.07%), while the lowest percentage of novel SNPs was in TB (6.16%) (Fig. 1a); 15.93% and 11.75% of SNPs identified in PZ and JH were novel (Fig. 1a). However, there was considerable within-population variation in MH; this was reflected by the higher mean autosomal nucleotide diversity among the Mongolian breeds (0.1549%, 0.1539%, and 0.1507% for MD, MG, and MJ, respectively) (Fig. 1b) when compared with those of PZ (0.1507%), JH (0.1334%), and TB (0.1209%). Nucleotide diversity is the per site average nucleotide difference between any two randomly chosen DNA sequences within a population35. The reduced nucleotide diversity in TB is likely due to generations of intensive artificial selection36. JH may have undergone a certain level of inbreeding owing to its long-term isolation and small population size6. Moreover, the population of JH was drastically reduced in the 1970s3. The low nucleotide diversity indicates that carefully planned breeding and conservation strategies are needed to maintain the genetic diversity of the JH. Presently, all the PZ horses trace their origin to 12 members of the founding population24,25,26,27; however, PZ horses show relatively high mean nucleotide diversity, similar to that reported by Goto et al.25. This may be due to the carryover of pre-existing elevated genetic diversity in the PZ horses, which occurred prior to the genetic bottleneck of the last century and before subsequent interbreeding of the PZ horses with domestic horses25,27. The quality of our SNP data was analyzed by calculating the transition to transversion ratio (Ts/Tv) (Table 2), which is used as an indicator of sequencing and SNP data quality in cattle, humans, pigs, and horses29,37,38,39,40. We recovered a Ts/Tv ratio of 1.97 across the genome, which is similar to the global Ts/Tv ratio of 1.92–2.2, reported in previous studies29,37,38,39.
Variant statistics, SNP annotation, and missense and loss of function (LOF) mutants detected in this study. (a) Top panel shows the number of homozygous and heterozygous SNPs (per million base-pairs) identified in this study; the number on top of the bar indicates the ratio of heterozygous to homozygous SNPs. Bottom panel shows the number of known and novel SNPs identified (per million base-pairs), with the percentage of novel SNPs provided at the top of the bars. (b) Nucleotide diversity ratios across the genome of the six breeds analyzed in this study. (c) Venn diagrams showing the total number of MS, fixed MS, LOF, and fixed LOF mutants in the JH and TB.
SNP annotation and function assessment of missense and loss-of-function SNPs
SNPs were functionally annotated using SnpEff41 (Fig. 1c and Table 2). Most of the SNPs (66%) were located in the intergenic regions. Among genic SNPs, intronic SNPs (70%) were the most common, followed by SNPs located upstream (14%) and downstream (13%) of the genes. Only 3% of the genic SNPs were located in the exonic region. We identified a large number (73,562) of missense mutations (Table 2), which were part of 15,427 genes; some of this mutation may be associated with traits of interest (Supplemental File S2). We also identified 36,033, 30,872, 53,362, 53,413, 53,358 and 34,1334 missense mutations, located in 11,764, 10,948, 13,825, 13,829, 13,822 and 11,613 genes in the JH, TB, MD, MG, MJ and PZ populations, respectively (Table 2 and Supplemental File S2). Among the identified missense mutations, we found 4,108 (in 2,733 genes), 2,459 (in 1,512 genes), 3,635 (in 2,362 genes), 3,749 (in 2,465 genes), 4,181 (in 2,643 genes) and 4,915 (in 3,085 genes) SNPs to be segregating (i.e. fixed) in JH, TB, MG, MJ, MD and PZ respectively, compared to the reference genome (Supplemental File S3). Comparative analysis of the missense mutations between JH and TB revealed that 21,661 missense SNPs, located in 8,988 genes, were found in both the breeds, however, 14,438 missense SNPs, located in 7,430 genes, and 9,211 missense SNPs, located in 5,525 genes, were unique to JH and TB respectively (Supplemental File S4). Among the fixed missense mutations, 2,105 (found in 1,655 genes) and 2,003 (found in 1,288 genes) SNPs were found only in JH and TB respectively, while 456 fixed missense SNPs (found in 359 genes) were found in both JH and TB (Supplemental File S4). Gene ontology (GO) and KEGG pathways analyses showed that in JH (Table 3), genes with fixed missense mutations were enriched in negative regulation of the Wnt signalling pathway, central nervous system development, metallopeptidase activity, ATPase activity, ECM receptor interactions, and peptide catabolic process. In TB, genes with missense mutations were enriched in cerebellar development, positive regulation of dendritic spine development, binding of protein kinase A, and negative regulation of canonical Wnt signalling pathway.
To identify SNPs underlying recessive traits, we grouped protein coding SNPs belonging to five high-impact severity types; including SNPs functionally classified as start loss, stop gain, splice acceptor, splice donor and stop loss, into loss of function (LOF) variants. In total, we detected 1,483 LOF SNPs (LOFs) across the populations (Table 2 and Supplemental File S5), with the MH breeds carrying the greatest number of LOF SNPs (MG -1,156, MJ - 1,154 and MD - 1,150). This may be due to larger number of variants identified in the MH breeds relative to the other breeds analyzed in this study. In the JH (824) and TB (737), 287 LOFs were unique to the JH (Table 4) and 198 LOF SNPs were unique to TB, while 496 LOF SNPs were found in both the JH and TB (Supplemental File 7), Out of this we identified 43 and 34 LOF (LOF) SNPs to be fixed only in JH and TB populations, while 107 fixed LOF were found in both JH and TB (Table 4, Supplemental File 7). Comparative analyses of LOF (LOF) variants among the breeds may uncover genes that, although not necessary for survival and reproduction, may have acted as a means for adaptive evolution against environmental changes, thereby contributing to the phenotypic differences between breeds42,43. There were 140 and 130 genes with LOF variants in JH and TB populations, respectively (Supplemental File S6). Our GO and KEGG pathway analyses of genes affected by LOF revealed that genes involved in voltage gated calcium channel complex, glucose transmembrane transporter activity, intracellular signal transduction, and apoptosis, were overrepresented in JH; in TB, the genes affected by LOF were enriched in the MAPK signalling pathway, calcium signalling pathway, and regulation of Notch signalling pathway (Table 4). Though determining the effect of each one of the identified MS and LOF variants on gene expression was beyond the scope of this study, several promising candidate SNPs, previously found to be associated with critical traits of interest, were identified in the JH (Table 5).
Population structure, admixture proportions, demography, and migration events
We used principal component analysis (PCA) and admixture analysis, based on genotypic data, to examine the genetic relationship among the sequenced horse populations. For PCA, we used only autosomal SNPs, and analysis was conducted using GCTA (version 1.25). The first and second principal components accounted for 16.3 and 10.7% of total variation (Fig. 2a). The individuals from the JH, MH (MJ, MG, and MD), TB, and PZ populations were grouped according to their origins using PCA (Fig. 2a). Our results indicated that the JH and MH were genetically closer, while TB and PZ horses were genetically distinct to the other breeds in the study. To estimate individual ancestry, admixture proportions were assessed without defined population information using ADMIXTURE (version 1.3.0)44 (Fig. 2b). The best K (number of ancestral population) was identified based on five-fold cross validation of the data. The individual population was grouped into separate clusters at K = 3 with the lowest cross validation error. The grouping of the JH into an independent unique cluster indicated that the ancestry of the JH was independent of that of MH, even though two individuals, JH 02 and JH 04, among the JH population showed 10 and 25% admixture with Mongolian genome, and this did not change even when K was increased to 4. This result indicates that within the JH population, some JH horses have Mongolian admixture across their genome, possibly because of the cross breeding of several native JH with MH in the past. This was reflected in the results of neighbour-joining (NJ) tree analysis, in which the two individuals (JH 02 and JH 04) were placed outside the main cluster (Fig. 2c).
Results of population genomic analysis. (a) PCA plot of populations of the six horse breeds; principal components 1 and 2 indicate 16.3 and 10.7% of the observed variance, respectively. (b) Population structure of the 41 horses in the six breeds; the length of each colored segment represents the proportion of the genome inferred from the ancestral population (K). (c) Neighbour joining tree inferred from identity by descent distance matrix. The clades are colored according to breeds.
We then estimated past effective population size (Ne) of each breed in order to understand the demographic history of each breed (Fig. 3a). Considering their evolutionary history of extinction in the wild before successful captive breeding24,25,26,27, PZ experienced the greatest decline in population when compared with populations of other horse breeds examined in this study. The demographic history of the JH resembles that of PZ horses, which is compatible with the recent severe population reduction of JH3. MH was distinct from the other breeds, showing less fluctuation in population size over time, which reflects the abundant genetic diversity of MH26. Considering that the actual population of JH is closer to 2500 and they underwent severe reduction in population size recently3, the effective population size estimated was considerably higher (Fig. 3a), this is possibly due to the limitation of PopSizeABC method, which can overestimate effective population size when there is a large decline in population within the last few hundred years45, so only the trend in historical effective population size can be confidently estimated by this method. JH, TB, and PZ horses showed a common trend in population decline, starting approximately 4,500–10,000 years before present time; this was a likely consequence of horse domestication4.
Historical demography and gene flow analysis. (a) Estimated effective population size of MG, TB, JH, and PZ populations. Inset shows the 95% confidence interval for the estimates. (b) Pattern of population splits and gene flow between the breeds. The drift parameter is proportional to effective population size (Ne). The migration edge is coloured according to the percentage of ancestry received from the donor. Scale bar shows the average standard error (10 times) of the entry in the sample co-variance matrix.
To understand the historical relationship among the populations, we generated a maximum likelihood (ML) tree and calculated the residual covariance matrix of MH, TB, JH, and PZ using Treemix46 (Fig. 3b and Supplemental Fig. 2a–c). We sequentially added the migration events to the ML tree and inferred that the tree with one migration edge had the smallest residual error, therefore the best fit for our data. This tree indicated a gene flow from PZ to MH (Supplemental Fig. 2d–f). However there was no significant migration edge between the JH and MH. Archeological evidence suggests that horses were present in the Jeju island prior to the introduction of the Mongolian horses in the 12th century10, and there are reports of size similarity between the JH and horses in southern japan47, and phylogenetic analysis based on mitochondrial genome sequence had placed the JH with European and Middle eastern breeds3 rather than the Mongolian breeds, these results along with our findings, leads us to speculate that the JH has an ancestry independent of the MH, however since 2 of the 12 JH samples used in this study showed some admixture with MH, a future study with a larger number of JH samples must be carried out, to verify if there is a population substructure within the JH, one with no admixture with Mongolian horse and another with admixture.
Signature of selection in the genome of the Jeju horse
The characteristic features of the JH are its small body size, stocky head and shorter limbs, they also have poor athletic ability, in comparison, the TB horse are tall, slender and have been intensively artificially selected for their athletic ability, and candidate genes underlying body confirmation athletic performance traits are very well characterized17,18,20,48,49,50. Therefore, we performed a cross-population extended haplotype homozygosity (XP-EHH)51 between the JH and TB genome to identify selection signatures for body confirmation and performance traits in JH, and also looked for signatures for environmental adaptation within the JH genome by examining heterozygosity and extreme haplotype homozygosity using Z transformations of pooled heterozygosity (ZHp)52. First, we scanned the whole genome to detect regions with high degrees of fixation, which are indicative of selection signatures53. We applied 50% overlapping sliding windows, which were 150 Kb in size, along all the 31 autosomes. ZHp scores were calculated for each of the 29,840 windows analyzed. The ZHp scores ranged from −6.50 to 3.63 in the JH (Fig. 4a and Supplemental file S8). Because an extremely low ZHp score indicates putative selective sweep due to excess homozygosity, we focused on ZHp scores in the extreme lower end of the distribution. We observed 103 windows with ZHp scores of less than −3.5 in the JH. GO analysis of the candidate selection signature genes showed significant enrichment (P < 0.05) for adaptive immune response, muscle cell development, regulation of aerobic respiration, zinc ion binding, and cell adhesion molecules (Fig. 4c).
Selection signature analysis. (a,b) Manhattan plot showing the distribution of P values inferred from ZHp (a) and XP-EHH (b) analyses. The XP-EHH analysis was performed between JH and TB horses, while ZHp was performed only with JH. The total number of windows found to be significant is provided at the top of the panel. c - d) Enriched gene ontology and KEGG pathway for genes in the candidate selection signature regions in ZHp (c) and XP-EHH (d). (e) Venn diagram showing the total number of genes found within the significant candidate selection signature regions.
Next, we divided the whole genome into non-overlapping windows of 50 Kb. A total of 43,061 windows were used to calculate the XP-EHH in each window. We observed 222 windows with significant XP-EHH (P ≤ 0.005) in the JH (Fig. 4b). These windows were considered candidate regions under selection and used for further analyses. As shown by the distribution of raw ZHp and XP-EHH values in Fig. 4a,b, we observed 121 and 112 genes in the candidate regions for ZHp and XP-EHH, respectively. GO analysis showed significant enrichment (P < 0.05) for ATP binding, p53 signalling pathway, and protein kinase activity (Fig. 4d). Among the candidate regions evaluated using these two approaches only 30 genes overlapped, they were enriched for apoptotic processes, mitochondrial outer membrane permeabilization, signal transduction in response to DNA damage, and mitotic nuclear division (Table 6 and Fig. 4e). The overlapping of low number of genes may be due to differences in the statistical methods used for data analysis. Haplotype-based selection signature analysis, such as XP-EHH, has a greater power, to detect ongoing selection, while analyses based on allele frequency patterns, such as ZHp, can detect recent fixation of alleles54. The candidate selection signature genes, indicates selection for environmental adaptation, athletic performance, muscle cell development and body confirmation traits.
Genes underlying physical conformation traits in the Jeju horse
Among the regions under selection, we identified several genes associated with equine body confirmation traits such as body size; wither height, and dwarfism (Table 5). These included LCORL, HMGA2, NCAPG, PROP1, and ZFAT. The ligand-dependent nuclear receptor co-repressor like protein (LCORL) is associated with body size in several horse breeds18,55,56,57. Moreover, a recent study has shown that a T homozygous genotype on rs68603064 at ECA 3 near LCORL is significantly associated with lower wither height in Brazilian pony, with average wither height less than 158 cm58. In this study, we observed the same SNP to be fixed in the JH, which shows an average wither height of 122 cm. This observation was consistent with the previous results of Schroder et al. (2010), who found that a QTL on ECA 3 in the region of LCORL influences body conformation traits such as head, neck, and frame development57. Notably, the JH also shows a heavy head, thick neck, and short and thick limbs59.
A large-scale genome-wide analysis, examining 65 horse breeds, identified 4 loci on ECA 3, 6, 9, and 11 near LCORL/NCAPG, HMGA2, ZFAT, and LASP1 to account for the 83% variation in body size in horses56. These four genes (LCORL, HMGA2, NCAPG, and ZFAT) were present in the candidate selection signature regions in JH. Zinc finger and AT hook domain containing gene (ZFAT) on ECA 9 may play an important role in haematopoiesis60 and may be associated with height in horses55,56. The horse ZFAT gene is orthologous to human ZFAT, which influences height61,62,63. The four SNPs (rs68748127, rs68750453, rs68748129, rs68748130) in the ZFAT gene were fixed in the JH (Table 5). The high mobility group AT-hook 2 gene (HMGA2) is an architectural transcription factor that regulates and directs cellular growth, proliferation, and differentiation64. Moreover, HMGA2 is amongst the first genes associated with human height61,65. HMGA2 plays a role in the control of body size in dogs66,67. Prophet of the Pit-1 gene (PROP1) is a homeodomain transcription factor associated with pituitary development68. Mutations in this gene are implicated in deficiency of pituitary hormone in humans69. PROP1 causes dwarfism in Ames dwarf mice70 and may be associated with dwarfism in Friesian horses71. The presence of these genes in candidate selection signature regions indicates that these genes may robustly drive selection for body confirmation traits and small body size in JH. These genes are also good candidates for further targeted studies to find the causative SNPs associated with physical traits and small body size in horses.
Genes under selection for athletic performance and muscle fibre composition
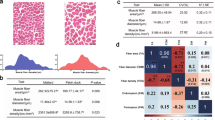
The JH is an indigenous native horse breed predominantly used for draught purpose in the nomadic habitual environment, while TB has been intensively selected for superior athletic performance. Because these two breeds are intended for different purposes, comparison of the genomes revealed numerous genes associated with athletic performance and muscle composition. Muscle fibres are classified into Type I (slow twitch), and Type IIA, IIB, and IIC (fast twitch)72. Horses with higher content of slow-twitch oxidative muscle fibres develop strength and power, and are well-suited for draught work and meat production. The muscles of horses selected for racing, high speed running, and other athletic purposes consist of fast-twitch glycolytic fibres73. Comparative histochemical analysis of gluteus medialis (rump muscle) and vastus lateralis (thigh muscle) in the JH and TB showed that the content of Type I fibres in the JH exceeded that in TB by ~15%–20% (Fig. 5C, Supplemental Figure S3).
In the candidate selection signature regions, we identified five genes that are associated with athletic performance, namely, ACTN250, COX4I150, COX4I249, CKM49, and PDK450. Creatine kinase muscle (CKM), which is a muscle type isozyme of creatine kinase, and COX4I2, which is a subunit of cytochrome c oxidase (COX), increase the efficiency of cellular respiration and are associated with athletic performance in TB49. Similarly, pyruvate dehydrogenase kinase isozyme 4 (PKD4) plays a key role in oxidation of fatty acids for ATP generation49. Three SNPs on PKD4 (g.39020227, g.39017959, and g.39024151) are significantly associated with winning performance in elite TB horses50. In particular, the A alleles of both g. 39017959 C > A and g. 39024151 G > A were found to be the favourable allele at both loci and were strongly associated with athletic performance TB, the C and G alleles were found to be fixed at the respective loci in the JH. Similarly, g.16079732 A > G (CKM) and g.23314524 C > T (COX4I2) were also associated with athletic performance in TB, with the A and T being the favourable alleles49. However, the C allele of g. 23314524 > T was fixed in JH, and the G allele of g. 16079732 A > G was the predominant allele (0.90) in JH (Table 5). The two genes ACTN3 and MSTN, which function in muscle development and muscle fibre composition, were also identified in the selection signature regions. ACTN3 is a sarcomeric protein involved in structure, muscle metabolism, and calcineurin signalling74,75. Mutations in ACTN3 change muscle fibre phenotype from fast-twitch fibres to slow-twitch fibres75. Moreover, rs1144978872 in the 5′UTR of ACTN3 leads to g.30226577 G > A and may affect athletic performance74,75. Interestingly, the A allele of g.30226577 G > A was identified as being detrimental to sprint performance, while endowing the carrier with strength and power suitable for draught work. The A allele (0.89) and Type I fibres predominated in the JH (Table 5).Myostatin, encoded by the MSTN gene, limits skeletal muscle mass by controlling and regulating the growth and number of muscle fibres76,77,78. Variations in MSTN are implicated in muscle fibre composition in horses73,79,80,81. The presence of a short interspersed nuclear element (SINE; 227 bp) in the promoter region and C allele in intron 1 (Chr 18: 66608679; rs397152648) are significantly associated with a greater proportion of Type 2B fibres and reduction in Type I fibres in TB and Quarterback horse populations80,81. The promoter SINE was absent, while the variant T allele at g.66608679was highly predominant (0.9) in the JH. Moreover, the g. 66608679 C > T on MSTN is also associated with sprinting ability and distance racing, and is known to be a gene associated with speed48. The C allele may be better suited for fast and short-distance races, while the T allele is suited for long-distance slower-speed racing82. Hence, the predominant T allele (0.95) in the JH may explain the ability of this breed to run long distances and its strong endurance in the nomadic habitual environment.
The majority of identified candidate selection signature genes for body confirmation, athletic performance and muscle compositions were identified in comparison with the TB horse genome (XP-EHH method). Since the TB horse are artificially selected for these traits, they could be representative of this selection, therefore the functional variants and genes identified as selection signatures in JH must be validated by further genetic studies such as genome-wide association studies.
Transcriptomic landscape of the JH compared with that of TB
To understand the genetic basis underlying the morphological and physiological differences in body confirmation and running abilities of JH relative to TB horses, we compared mRNA expression levels between JH and TB, in five tissues including the rump muscle, thigh muscle, liver, heart, and lung. These five tissues were chosen for their critical role in energy metabolism, aerobic capacity and athletic ability of horse. The RNAseq analysis showed that relative to TB, 5,462 genes were found to be differentially expressed (DE) in JH, including 3,498 genes in heart, 1,101 genes in the liver, 1,417 genes in the lung, 139 genes in rump muscle, and 193 genes in thigh muscle (Supplemental file S9). GO analysis (Fig. 5a) and KEGG pathway analysis (Fig. 5b), conducted using DEGs, indicated significant enrichment in the following terms: transmembrane receptor kinase signalling pathway; positive regulation of smooth muscle cell proliferation; immune response; angiogenesis and cell adhesion molecules (CAM) pathway; Toll-like receptor signalling pathway and tight junction pathways in heart; skeletal muscle cell differentiation; cellular response to extracellular stimulus and CAM pathway in the rump muscle; fatty acid homeostasis; muscle contraction and calcium signalling pathway in the thigh muscle; brown fat cell differentiation; cellular calcium ion homeostasis; oxidative phosphorylation pathway and biosynthesis of antibiotics pathway in the liver; fatty acid beta oxidation; cholesterol homeostasis; response to oxidative stress; cAMP signalling pathway; fat digestion and absorption pathway; and calcium signalling pathway in the lung.
Among the DE genes, 71 genes were located in the candidate selection signature regions (Supplemental file S10) identified in the JH. The majority of these genes were in the heart (39 genes), followed by liver (16 genes), lung (16 genes), rump (3 genes), and 1 gene in the thigh muscle. Five of these genes (LCORL, snoR442, GRIA2, PRRT1, and U6atac) were up-regulated in all these tissues; 4 genes (U6, U4, SNORD112, and 7SK) were up-regulated in the liver and down-regulated in the heart; and the rest of the genes were down-regulated in all these tissues. Interestingly LCORL, a gene associated with the body size of horse was differentially expressed. A previous study has shown that in hanoverian wamblood horses the expression level of LCORL was substantially higher in the TT horse (rs68603064), which has a smaller body size, than in CT and CC horses18. We found the T allele to be fixed in the JH at the rs68603064 locus. Our result also showed the expression of LCORL was significantly higher in the JH (FDR < 0.05) than in the TB horse. Several other important genes responsible for muscle fibre composition (MSTN) and athletic performance (ACTN2, COX4I1, COX4I2, and CKM) were downregulated in JH tissues. Long-term athletic endurance requires high oxygen carrying capacity (aerobic capacity; Vo2max), high density of mitochondria in skeletal muscle, and a large lung volume83,84. The abundance of mitochondrial COX4 (COX4 I1 and COX4I2) is directly related to mitochondrial density85. The expression of COX4I1 and COX4I2 can also affect velocity at maximum heart rate (VHRmax). VHRmax is a predictor for overall athletic ability and peak post-exercise plasma lactate concentration; consequently VHRmax is generally used as an essential predictor for overall anaerobic capacity of TB83,84. In TB that are bred for superior athletic ability, COX4I1 levels are significantly increased after exercise, leading to a positive correlation with VHRmax86. Therefore, the significant downregulation of COX4I1 and COX4I2 expression observed in the JH may indicate a lower oxygen carrying capacity in the JH, resulting in its reduced anaerobic capacity and ability for athletic racing. However, this requires further study, including measuring and comparing lung capacity, VHRmax, and Vo2max between JH and TB horses.
In summary, we identified differential expression of numerous genes within the candidate selective sweep regions, indicating that the extensive adaptation of the JH conferred it with physiological and morphological variation relative to that of TB. These results helps in understanding the unique adaptations found in the JH.
There are limitations in this study due to the use of only TB (a highly artificially selected breed for athletic performance) as a reference for compassion with JH for identifying selection signature and gene expression difference. Further studies involving other extreme sized horse breeds such as small sized: shetland ponies, highland ponies, and large sized: shire horses, clydesdales and belgian draft horses could help in identifying regions under selection and genes that are differentially expressed in JH for stature, hardiness, longevity, muscle composition and strength.
Conclusions
In this study, we generated whole-genome sequence data on 41 horses that included three MH breeds (MG, MJ, and MD), as well as PH, TB, and JH. We identified 5.1 million (~24%) novel SNPs among the ~21 million observed SNPs. Additionally, we identified breed-specific nsSNPs and LOF mutants in the JH and TB, respectively. Analyses performed using data on population genetics, admixture, and demography indicated that the JH did not genetically originate from MH breeds, even though the JH is closer to MH genetically than to other horse breeds examined in this study. We also found that nucleotide diversity in the PZ, which was close to extinction in the last century, is much higher than that in the JH, indicating lower genetic diversity within JH and the need to design conservation strategies to increase and maintain genetic diversity within the JH population. Our selection-signature analyses, based on allele frequency homozygosity and haplotype fixation, revealed numerous candidate genes involved in the environmental adaptation, muscle composition, and unique body confirmation traits of the JH. Finally, we performed RNAseq analysis using five types of tissues (heart, lung, liver, rump muscle, and thigh muscle) acquired from the JH and TB, and found numerous important DE genes within the selective sweep regions. The data generated in this study will serve as a valuable resource for researchers studying the evolution and domestication of Equus caballus; these data can also be used for the study of other small-sized or non-bred horse breeds. The large number of novel SNPs identified in this study will aid in designing a genome-wide high-density SNP chip. This will help to design conservation strategies for various native horse breeds across the globe.
Methods
Sampling and whole-genome re-sequencing
All the experimental procedures were verified and approved by the Institutional Animal Care and Use committee of the National Institute of Animal Science (NIAS2015–775), and all methods were performed in accordance with the relevant guidelines and regulations.
Whole-genome re-sequencing data were generated for Mongolian Galshar (N = 6, MG), Mongolian Jarglant (N = 5, MJ), Mongolian Domestic Horses (N = 4, MD), Przewalski’s Horses (N = 4, PZ), Jeju horse (N = 12, JH), and Thoroughbred horse (N = 10, TB) (Table 1). Samples of JH and TB tissues were obtained from the National Institute of Animal Science (NIAS, Jeju Island, Korea). Samples of MH tissues were obtained from Mongolian University of Life Sciences (Ulaanbaatar, Mongolia). PZ horse samples were obtained from Seoul Zoo (Seoul, Republic of Korea). Indexed shotgun paired-end libraries with average insert size of 500 bp were generated using TruSeq Nano DNA Library Prep Kit (Illumina, San Diego, CA, USA) following the standard Illumina sample-preparation protocol. Briefly, 200 ng of gDNAs were fragmented with Covaris M220 (Woburn, MA, USA) to obtain median fragment size of ~500 bp. These fragmented DNAs were end-repaired, followed by A-tailing and ligation to the indexed adapter (~125-bp adapter). Gel-based selection was performed to select sizes of 550 to 650 bp. Eight cycles of PCR amplification was performed on GeneAmp PCR system 2700 thermal cycler (Applied Biosystems®, Foster City, CA). Size-selected libraries were then analysed with Agilent Bioanalyzer 2100 (Agilent, Santa Clara, CA, USA) to determine size distribution and adapter contamination status. The resulting libraries without adaptor contamination were sequenced on Illumina HiSeq. 4000 sequencing platforms for 2 × 125 bp paired-end sequencing.
Sequence mapping, SNP calling, and annotation
After quality-control assessment and trimming, the sequences were aligned to Equine reference genome assembly (EquCab 2.034) using Bowtie2 v2.2.487 at default parameters. Mapped reads were converted, sorted, and indexed using SAM-tools v1.3.188. Removal of duplicate reads and generation of quality matrices for mapping were performed using Picard tools v2.1.0 (http://broadinstitute.github.io/picard). Local recalibration and realignment were conducted using Genome Analysis Toolkit (GATK; v3.3). A multi-sample SNP-calling procedure was performed to discover SNPs using UnifiedGenotyper in the GATK package89. Finally, a filtering step, based on GATK best practice guidelines was used as follows: QD < 5.0, MQ < 40.0, FS > 200.0, and QUAL < 30.089. SNPs were also filtered for call rate < 0.9 and MAF (minor allele frequency) ≤ 0.01.The filtered SNPs were then annotated to 12 functional categories (Table 2) using SnpEff version 4.141. SnpSift version 4.1 was used for filtering loss-of-function (LOF) and non-synonymous (NS) mutations. We also determined which SNPs were fixed in the JH and TB, and identified breed-specific fixed NS mutants. Non-reference genotypes that were homozygous for the entire population were deemed fixed SNPs90.
Remapping of the identified SNPs to EquCab3.0 Coordinates
Following the release of the new reference genome EquCab3.091 we remapped the coordinates of the SNPs identified using the EquCab 2.0 (Assembly SeqID GCF_000002305.2) reference genome to the corresponding EquCab 3.0 (Assembly SeqID GCF_002863925.1) coordinates using the NCBI Remap tool (May 2018 release), which facilitates the remapping of genomic features from one assembly to another. All options were set to default. Only EquCab 3.0 annotated SNPs are reported and discussed herein. And all the genomic coordinates given are based on EquCab 3.0.
Population genetics analysis
VCFtools v4.092 was used to estimate mean autosomal nucleotide diversity using windows of 10 kb. PCA analysis was performed using genome-wide complex trait analysis (GCTA)93. Genotype data for all the samples were used to estimate the eigenvectors. To refine the quantification of different ancestry proportions, we performed a model-based unsupervised hierarchical clustering of the individuals using Admixture 1.22 software44. Admixture provides a likelihood estimate in which ancestral populations in an unsupervised analysis are clustered based on allele-frequency similarities. We analysed breed proportions using K = 2 to 6 assumed ancestral populations. Recent demographic history was inferred by assessing changes in the trend of effective population size (Ne) using PopSizeABC94 with parameters set as described previously35. An IBS-based distance matrix was calculated using SNP genotypes in PLINK v1.995, and a neighbour-joining tree was constructed with FigTree v1.4.4. We then inferred population-level phylogeny based on maximum likelihood (ML) statistics implemented in TreeMix46. A linkage disequilibrium (-k) size of 1000 and ‘-global’ options were used to generate the ML tree, and migration events (-m) were sequentially added to the tree.
Selective sweep analysis
We used Z transformation of pooled heterozygosity (ZHp) and XP-EHH to detect putative selective sweeps (positive selection) in the JH population. All the high-quality SNPs, derived from the JH, were used to identify pooled heterozygosity (ZHp). The numbers of major and minor alleles were counted; then, SNP positions with a minor allele frequency less than 0.05 were removed. Subsequently, we applied 50% overlapping sliding windows that were 150 kb is size; ZHp was eventually calculated for each window after removing windows with fewer than 10 SNPs following the method by Rubin et al.52,96. A threshold of 3 was used for identifying significant signals. We then used xpehh software (http://hgdp.uchicago.edu/Software/) to perform a cross-population extended haplotype homozygosity (XP-EHH) analysis to detect selection signatures in the JH population relative to those in the thoroughbred horse population51. This analysis detects haplotypes that show increased frequency to the point of complete fixation in one of the populations. We initially split the genome into non-overlapping segments of 50 kb and used the maximum (positive) XP-EHH score of all SNPs within a window as a summary statistic for that window. To assess variations in SNP density, we binned genomic windows according to their numbers of SNPs in increments of 500 SNPs (combining all windows ≥ 1000 SNPs into one bin). Within each bin, for each window i, the fraction of windows with a value of the statistic greater than in i was defined as the empirical P value. Regions with P value less than 0.005 (0.5%) were considered statistically significant signals. Regions that were found to be significant using either one of the methods were considered as candidate regions under selection (CRS).
RNAseq analysis
Total RNA from five types of tissues (heart, lung, liver, rump muscle, and thigh muscle) was isolated from JH (n = 3) and TB (n = 4) using Trizol reagent (Invitrogen, Carlsbad, CA, USA) following the manufactures protocol. The quality of isolated RNA was assessed using Bioanalyzer 2100 (Agilent, Santa Clara, CA, USA). Only RNA with an RNA integrity value (RIN) greater than 8 was used for library preparation. The sequencing libraries were prepared using a TruSeq RNA kit (Illumina; San Diego, CA), and Paired End sequencing was carried out on an Illumina HiSeq-2000 (Illumina; San Diego, CA), using individual lanes. The generated reads were trimmed to remove adapter sequences using Trimmomatic 0.3697; the reads were assessed for quality using FastQC version 0.11.698. The reads were then aligned to the Equine reference genome assembly (EquCab2.1) using Hisat2 version 2.0599. The aligned reads were counted using featureCounts in the Subread package version 1.6.0100. The count data were analysed for differential gene expression (DEG) using EdgeR package in R101. Genes with a false discovery rate corrected Q value < 0.05 were considered to be significantly differentially expressed and were used for downstream analysis.
Functional enrichment analysis
Functional enrichment analysis was performed on genes in the CRS, genes with LOF and NS mutations, and differentially expressed genes (DE). The enriched GO terms were obtained using the web program DAVID (Databank for Annotation, Visualization and Integrated Discovery)102; REVIGO103 and Clusterprofiler R package104 were used for summarizing the GO terms.
Histochemical analysis
Histochemical analysis was performed to examine muscle fibre characteristics in the gluteus medialis (rump) and vastus lateralis (thigh) muscles of JH and TB horses. The analysis was performed as previously described by Lee et al.,105. Briefly, tissue samples were collected from two horses per breed. These samples were cut into 0.5 cm * 0.5 cm * 1.0 cm blocks, frozen in isopentane cooled by liquid nitrogen, and stored at −80 ◦C until further use. The muscle tissues were consequently sectioned at 10μm thickness using a cryostat microtome (CM 1850, Leica, Heidelberger, Germany) at 20◦C, and the activity of myosin adenosine triphosphatase (ATP) was detected after acid pre-incubation (pH 4.7) as described previously106. The predominance of Type I or Type II muscle fibres in the two breeds was then evaluated using an optical microscope, equipped with a charge-coupled device (CCD) colour camera (IK-642K, Toshiba, Tokyo, Japan), and image analysis was carried out using Image-Pro plus software (Media,Cybernetics, Silver Springs, USA).
Data availability
All the data generated in this study are freely available for download at the National Agricultural Biotechnology Information Center (NABIC) website (www.nabic.rda.go.kr). The accession numbers for whole genome data are JH (NN-5490-000001 ~ NN-5490-000012), TB(NN-5491-000001 ~ NN-5491-000010), MH and PZ (NN-5492-000001 ~ NN-5492-000017), and for RNAseq data are JH (NN-5494-000001 ~ NN-5494-000015), TB (NN-5495-000001 ~ NN-5495-000019).
Change history
21 October 2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Jo, Y.-S., Kim, T.-W., Choi, B.-J. & Oh, H.-S. Current status of terrestrial mammals on Jeju Island. Journal of Species Research 1, 249–256 (2012).
Kim, B.-W. et al. Characterization of the European type of maternal lineage evident in extant Jeju native pigs. Genes & Genomics 33, 111 (2011).
Yoon, S. H. et al. Complete mitochondrial genome sequences of Korean native horse from Jeju Island: uncovering the spatio-temporal dynamics. Molecular biology reports 44, 233–242 (2017).
Levine, M. Investigating the origins of horse domestication. Equine Veterinary Journal 31, 6–14 (1999).
Vilà, C. et al. Widespread origins of domestic horse lineages. Science 291, 474–477 (2001).
Kim, N. Y. et al. Genome-wide analyses of the Jeju, Thoroughbred, and Jeju crossbred horse populations using the high density SNP array. Genes & genomics, 1–10 (2018).
Do, K.-T., Lee, J.-H., Lee, H.-K., Kim, J. & Park, K.-D. Estimation of effective population size using single-nucleotide polymorphism (SNP) data in Jeju horse. Journal of animal science and technology 56, 28 (2014).
Lee, J.-H., Song, K.-D., Kim, J.-M., Leem, H.-K. & Park, K.-D. Identification of genes with nonsynonymous SNP in Jeju horse by whole-genome resequencing reveals a functional role for immune response. Journal of animal science 94, 895–901 (2016).
Nam, D. Horse production in Cheju during Lee dynasty. Studies on Korean History 4, 131–131 (1969).
Shin, T. et al. An anatomy study of animal bones excavated in the Kwakji archaeological site in Cheju Island. Go-Moon-Wha 40, 31–42 (1992).
Kim, K. I. et al. Phylogenetic relationships of Cheju horses to other horse breeds as determined by mtDNA D‐loop sequence polymorphism. Animal Genetics 30, 102–108 (1999).
Oh, M. et al. Phylogenetic relationship of Cheju native horses by mitochondrial DNA analysis. Molecules and Cells (Korea Republic) (1994).
Xu, S. et al. High altitude adaptation and phylogenetic analysis of Tibetan horse based on the mitochondrial genome. Journal of Genetics and Genomics 34, 720–729 (2007).
Jung, Y.-H., Han, S.-H., Shin, T. & Oh, M.-Y. Genetic characterization of horse bone excavated from the Kwakji archaeological site, Jeju, Korea. Molecules and cells 14, 224–230 (2002).
Yang, Y., Kim, K., Cothran, E. & Flannery, A. Genetic diversity of Cheju horses (Equus caballus) determined by using mitochondrial DNA D-loop polymorphism. Biochemical genetics 40, 175–186 (2002).
Kim, N. Y. et al. Estimation of genetic parameters for temperament in Jeju crossbred horses. Asian-Australasian journal of animal sciences 31, 1098 (2018).
Brown-Douglas, C. G. & Pagan, J. D. Body weight, wither height and growth rates in Thoroughbreds raised in America, England, Australia, New Zealand and India. Advances in Equine Nutrition IV, 213 (2009).
Metzger, J., Schrimpf, R., Philipp, U. & Distl, O. Expression levels of LCORL are associated with body size in horses. PLoS One 8, e56497 (2013).
Lee, J.-R. et al. Genome-wide analysis of DNA methylation patterns in horse. BMC genomics 15, 598 (2014).
Lee, W. et al. Analysis of cross-population differentiation between Thoroughbred and Jeju horses. Asian-Australasian journal of animal sciences 31, 1110 (2018).
Choi, S.-K., Cho, C.-Y., Yeon, S.-H., Cho, B.-W. & Cho, G.-J. Genetic characterization and polymorphisms for parentage testing of the Jeju horse using 20 microsatellite loci. Journal of Veterinary Medical Science 70, 1111–1115 (2008).
Gim, J.-A. et al. HEpD: A database describing epigenetic differences between Thoroughbred and Jeju horses. Gene 560, 83–88 (2015).
Schubert, M. et al. Prehistoric genomes reveal the genetic foundation and cost of horse domestication. Proceedings of the National Academy of Sciences 111, E5661–E5669 (2014).
Do, K.-T. et al. Genomic characterization of the Przewalski׳ s horse inhabiting Mongolian steppe by whole genome re-sequencing. Livestock Science 167, 86–91 (2014).
Goto, H. et al. A massively parallel sequencing approach uncovers ancient origins and high genetic variability of endangered Przewalski’s horses. Genome biology and evolution 3, 1096–1106 (2011).
Huang, J. et al. Analysis of horse genomes provides insight into the diversification and adaptive evolution of karyotype. Scientific reports 4, 4958 (2014).
Volf, J., Kus, E. & Prokopova, L. General studbook of the Przewalski horse. Zoological Garden Prague, Prague (1991).
Minjigrorj, N. & Austbo, D. Production of mare’s milk in Mongolia. Billige M., Liu W., Rina W., Wang L., Sun T., Wang J., Li H., & Zhang H. Evaluation of potential probiotics properties of the screened Lactobacilli isolated from home-made koumiss in Mongolia. Annals of Microbiology 59, 493–498 (2009).
Zhang, C. et al. Detecting the Population Structure and Scanning for Signatures of Selection in Horses (Equus caballus) From Whole-Genome Sequencing Data. Evolutionary Bioinformatics 14, 1176934318775106 (2018).
Gouveia, J. Jd. S., Silva, M. V. G. Bd, Paiva, S. R. & Oliveira, S. M. Pd Identification of selection signatures in livestock species. Genetics and molecular biology 37, 330–342 (2014).
Avise, J. C. & Ayala, F. J. In the light of evolution III: Two centuries of Darwin. Proceedings of the National Academy of Sciences 106, 9933–9938 (2009).
Driscoll, C. A., Macdonald, D. W. & O’Brien, S. J. From wild animals to domestic pets, an evolutionary view of domestication. Proceedings of the National Academy of Sciences 106, 9971–9978 (2009).
Gregory, T. R. Artificial selection and domestication: modern lessons from Darwin’s enduring analogy. Evolution: Education and Outreach 2, 5 (2009).
Wade, C. et al. Genome sequence, comparative analysis, and population genetics of the domestic horse. Science 326, 865–867 (2009).
Kim, J. et al. The genome landscape of indigenous African cattle. Genome biology 18, 34 (2017).
Moon, S. et al. A genome-wide scan for selective sweeps in racing horses. Asian-Australasian journal of animal sciences 28, 1525 (2015).
Choi, J.-W. et al. Whole-genome analyses of Korean native and Holstein cattle breeds by massively parallel sequencing. PloS one 9, e101127 (2014).
Consortium, G. P. An integrated map of genetic variation from 1,092 human genomes. Nature 491, 56 (2012).
Choi, J.-W. et al. Whole-genome resequencing analyses of five pig breeds, including Korean wild and native, and three European origin breeds. DNA Research 22, 259–267 (2015).
Wang, J., Raskin, L., Samuels, D. C., Shyr, Y. & Guo, Y. Genome measures used for quality control are dependent on gene function and ancestry. Bioinformatics 31, 318–323 (2014).
Cingolani, P. et al. A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly 6, 80–92 (2012).
Kaiser, V. B. et al. Homozygous loss-of-function variants in European cosmopolitan and isolate populations. Human molecular genetics 24, 5464–5474 (2015).
Oh, H. J., Choi, D., Goh, C. J. & Hahn, Y. Loss of gene function and evolution of human phenotypes. BMB reports 48, 373 (2015).
Alexander, D. H., Novembre, J. & Lange, K. Fast model-based estimation of ancestry in unrelated individuals. Genome research 19, 1655–1664 (2009).
Boitard, S., Rodriguez, W., Jay, F., Mona, S. & Austerlitz, F. Inferring population size history from large samples of genome-wide molecular data-an approximate Bayesian computation approach. PLoS genetics 12, e1005877 (2016).
Pickrell, J. K. & Pritchard, J. K. Inference of population splits and mixtures from genome-wide allele frequency data. PLoS genetics 8, e1002967 (2012).
Nozawa, K. & Kondo, K. Gene constitution of Cheju native horse and its phylogenetic relationships with Japanese native horses. SABRAO Newsletter 2, 7–18 (1970).
Hill, E. W. et al. Correction: A sequence polymorphism in MSTN predicts sprinting ability and racing stamina in thoroughbred horses. PloS one, 5 (2010).
Gu, J. et al. Association of sequence variants in CKM (creatine kinase, muscle) and COX4I2 (cytochrome c oxidase, subunit 4, isoform 2) genes with racing performance in Thoroughbred horses. Equine Veterinary Journal 42, 569–575 (2010).
Hill, E., Gu, J., McGivney, B. & MacHugh, D. Targets of selection in the Thoroughbred genome contain exercise‐relevant gene SNPs associated with elite racecourse performance. Animal genetics 41, 56–63 (2010).
Sabeti, P. C. et al. Genome-wide detection and characterization of positive selection in human populations. Nature 449, 913 (2007).
Rubin, C.-J. et al. Strong signatures of selection in the domestic pig genome. Proceedings of the National Academy of Sciences 109, 19529–19536 (2012).
Qanbari, S. et al. A high resolution genome-wide scan for significant selective sweeps: an application to pooled sequence data in laying chickens. PloS one 7, e49525 (2012).
Qanbari, S. & Simianer, H. Mapping signatures of positive selection in the genome of livestock. Livestock Science 166, 133–143 (2014).
Signer-Hasler, H. et al. A genome-wide association study reveals loci influencing height and other conformation traits in horses. PloS one 7, e37282 (2012).
Makvandi-Nejad, S. et al. Four loci explain 83% of size variation in the horse. PLoS One 7, e39929 (2012).
Schröder, W. Athletic performance and conformation in Hanoverian warmblood horses-population genetic and genome-wide association analyses. cumulative thesis. Hannover: University of Veterinary Medicine (2010).
Junior, A. B. et al. Polymorphisms in the LASP1 gene allow selection for smaller stature in ponies. Livestock Science 216, 160–164 (2018).
Kang, M.-s Grazing behaviour of Jeju Native Horses. Journal of integrated field science 1, 33–34 (2004).
Tsunoda, T. et al. Immune-related zinc finger gene ZFAT is an essential transcriptional regulator for hematopoietic differentiation in blood islands. Proceedings of the National Academy of Sciences 107, 14199–14204 (2010).
Allen, H. L. et al. Hundreds of variants clustered in genomic loci and biological pathways affect human height. Nature 467, 832 (2010).
Takeuchi, F. et al. Evaluation of genetic loci influencing adult height in the Japanese population. Journal of human genetics 54, 749 (2009).
N’Diaye, A. et al. Identification, replication, and fine-mapping of Loci associated with adult height in individuals of african ancestry. PLoS genetics 7, e1002298 (2011).
Cleynen, I. & Van de Ven, W. J. The HMGA proteins: a myriad of functions. International journal of oncology 32, 289–305 (2008).
Weedon, M. N. et al. A common variant of HMGA2 is associated with adult and childhood height in the general population. Nature genetics 39, 1245 (2007).
Boyko, A. R. et al. A simple genetic architecture underlies morphological variation in dogs. PLoS biology 8, e1000451 (2010).
Jones, P. et al. Single-nucleotide-polymorphism-based association mapping of dog stereotypes. Genetics 179, 1033–1044 (2008).
Davis, S. et al. Molecular mechanisms of pituitary organogenesis: in search of novel regulatory genes. Molecular and cellular endocrinology 323, 4–19 (2010).
Deladoëy, J. et al. “Hot spot” in the PROP1 gene responsible for combined pituitary hormone deficiency. The Journal of Clinical Endocrinology & Metabolism 84, 1645-–1650 (1999).
Sornson, M. W. et al. Pituitary lineage determination by the Prophet of Pit-1 homeodomain factor defective in Ames dwarfism. Nature 384, 327 (1996).
Orr, N. et al. Genome‐wide SNP association–based localization of a dwarfism gene in Friesian dwarf horses. Animal Genetics 41, 2–7 (2010).
Rivero, J., Serrano, A. L., Henckel, P. & Aguera, E. Muscle fiber type composition and fiber size in successfully and unsuccessfully endurance-raced horses. Journal of Applied Physiology 75, 1758–1766 (1993).
Dall’Olio, S. et al. Analysis of horse myostatin gene and identification of single nucleotide polymorphisms in breeds of different morphological types. BioMed Research International, 2010 (2010).
Thomas, K., Hamilton, N., North, K. & Houweling, P. Sequence analysis of the equine ACTN3 gene in Australian horse breeds. Gene 538, 88–93 (2014).
Wang, J. et al. Analysis of Equine ACTN3 Gene Polymorphisms in Yili Horses. Journal of Equine Veterinary Science 70, 101–106 (2018).
McPherron, A. C., Lawler, A. M. & Lee, S.-J. Regulation of skeletal muscle mass in mice by a new TGF-p superfamily member. Nature 387, 83 (1997).
McPherron, A. C. & Lee, S.-J. Double muscling in cattle due to mutations in the myostatin gene. Proceedings of the National Academy of Sciences 94, 12457–12461 (1997).
Miyata, H. et al. Effect of Myostatin SNP on muscle fiber properties in male Thoroughbred horses during training period. The Journal of Physiological Sciences 68, 639–646 (2018).
Rooney, M. F., Porter, R. K., Katz, L. M. & Hill, E. W. Skeletal muscle mitochondrial bioenergetics and associations with myostatin genotypes in the Thoroughbred horse. PloS one 12, e0186247 (2017).
Petersen, J. L., Valberg, S. J., Mickelson, J. R. & McCue, M. E. Haplotype diversity in the equine myostatin gene with focus on variants associated with race distance propensity and muscle fiber type proportions. Animal genetics 45, 827–835 (2014).
Petersen, J. L. et al. Genome-wide analysis reveals selection for important traits in domestic horse breeds. PLoS genetics 9, e1003211 (2013).
McGivney, B. A. et al. MSTN genotypes in T horoughbred horses influence skeletal muscle gene expression and racetrack performance. Animal genetics 43, 810–812 (2012).
Constantinopol, M. et al. Oxygen transport during exercise in large mammals. II. Oxygen uptake by the pulmonary gas exchanger. Journal of Applied Physiology 67, 871–878 (1989).
Erickson, B. et al. Mechanism of reduction in alveolar-arterial PO2 difference by helium breathing in the exercising horse. Journal of Applied Physiology 76, 2794–2801 (1994).
Flück, M. Functional, structural and molecular plasticity of mammalian skeletal muscle in response to exercise stimuli. Journal of Experimental Biology 209, 2239–2248 (2006).
Eivers, S. S. et al. Alterations in oxidative gene expression in equine skeletal muscle following exercise and training. Physiological genomics 40, 83–93 (2009).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nature methods 9, 357 (2012).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079, https://doi.org/10.1093/bioinformatics/btp352 (2009).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome research 20, 1297–1303, https://doi.org/10.1101/gr.107524.110 (2010).
Aslam, M. L. et al. Whole genome SNP discovery and analysis of genetic diversity in Turkey (Meleagris gallopavo). BMC genomics 13, 391 (2012).
Kalbfleisch, T. S. et al. EquCab3, an Updated Reference Genome for the Domestic Horse. BioRxiv, 306928 (2018).
Danecek, P. et al. The variant call format and VCFtools. Bioinformatics 27, 2156–2158 (2011).
Yang, J., Lee, S. H., Goddard, M. E. & Visscher, P. M. GCTA: a tool for genome-wide complex trait analysis. The American Journal of Human Genetics 88, 76–82 (2011).
Rodriguez, W., Jay, F., Mona, S. & Austerlitz, F. Inferring population size history from large samples of genome-wide molecular data-an approximate bayesian computation approach. Plos Genetics, 3(12), 1–36 (2016) (2016).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. The American Journal of Human Genetics 81, 559–575 (2007).
Rubin, C.-J. et al. Whole-genome resequencing reveals loci under selection during chicken domestication. Nature 464, 587 (2010).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–2120 (2014).
Andrews, S. FastQC: a quality control tool for high throughput sequence data. (2010).
Kim, D., Langmead, B. & Salzberg, S. (2016).
Liao, Y., Smyth, G. K. & Shi, W. The Subread aligner: fast, accurate and scalable read mapping by seed-and-vote. Nucleic acids research 41, e108–e108 (2013).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Huang, D. W., Sherman, B. T. & Lempicki, R. A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nature protocols 4, 44 (2008).
Supek, F., Bošnjak, M., Škunca, N. & Šmuc, T. REVIGO summarizes and visualizes long lists of gene ontology terms. PloS one 6, e21800 (2011).
Yu, G., Wang, L.-G., Han, Y. & He, Q.-Y. clusterProfiler: an R package for comparing biological themes among gene clusters. Omics: a journal of integrative biology 16, 284–287 (2012).
Lee, S. H., Kim, J.-M., Ryu, Y. C. & Ko, K. S. Effects of Morphological Characteristics of Muscle Fibers on Porcine Growth Performance and Pork Quality. Korean journal for food science of animal resources 36, 583 (2016).
Brooke, M. H. & Kaiser, K. K. Muscle fiber types: how many and what kind? Archives of neurology 23, 369–379 (1970).
Hansen, M., Knorr, C., Hall, A., Broad, T. & Brenig, B. Sequence analysis of the equine SLC26A2 gene locus on chromosome 14q15→ q21. Cytogenetic and genome research 118, 55–62 (2007).
Acknowledgements
The authors would like to thank Rural Development Administration and National Institute of Animal Science for providing the funding to carry out this research. We also thank the Mongolian University of Life Sciences (Baatartsogt Oyungerel) for providing tissue samples from Mongolian horses, Mi-Hyun Yu at the Seoul Zoo for providing tissue samples from Przewalski’s horses, and Korean Horse Racing Authority (KRA) for providing tissue samples of Thoroughbred horses. This study was carried out with the support of “Cooperative Research Program for Agriculture Science & Technology Development” (Project No. PJ01040602, PJ01045302), Rural Development Administration (RDA), Republic of Korea. KS was supported by a 2019 RDA Fellowship Program of National Institute of Animal Science, Rural Development Administration, Republic of Korea. This work was supported by the Ministry of Education of Republic of Korea and National Research Foundation of Korea (grant number NRF-2019R1A6A1A03025159).
Author information
Authors and Affiliations
Contributions
DL, JMK, KTL, and NYK conceived and designed the study. DL, HHC, GWJ and JEP acquired the funding. KS, WCP, YCR, JK (Jae-Min Kim), KDK, HK, JWC, JWN, JHS and JMK performed the experiments and analyzed the data. KS, NYK, JMK and WCP prepared the figures. KS, JMK and JK drafted the manuscript. DL and HHC edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Srikanth, K., Kim, NY., Park, W. et al. Comprehensive genome and transcriptome analyses reveal genetic relationship, selection signature, and transcriptome landscape of small-sized Korean native Jeju horse. Sci Rep 9, 16672 (2019). https://doi.org/10.1038/s41598-019-53102-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-53102-8
This article is cited by
-
Biogeographic origin and genetic characteristics of the peopling of Jeju Island based on lineage markers
Genes & Genomics (2023)
-
Whole-genome sequence analysis reveals selection signatures for important economic traits in Xiang pigs
Scientific Reports (2022)
-
Selection signatures for heat tolerance in Brazilian horse breeds
Molecular Genetics and Genomics (2022)
-
An overview of remote monitoring methods in biodiversity conservation
Environmental Science and Pollution Research (2022)
-
Genome-wide scans for signatures of selection in Mangalarga Marchador horses using high-throughput SNP genotyping
BMC Genomics (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.