Abstract
The impact of chemotherapy on tumor-immune system interaction can be either beneficial or harmful, which is represented by the immunogenic cell death (ICD) paradigm or overexpression of the immunosuppressive protein – programmed death ligand 1 (PD-L1). In this study we explore the impact of steroid receptor coactivator inhibitor, other targeted anti-cancer compounds and traditional chemotherapeutic agents on the expression of PD-L1 in four breast cancer (BC) cell lines. Our results show that these agents induce PD-L1 expression, yet the magnitude of this induction varies substantially across the different compounds. In addition, we utilized the E0771 ER + BC cells as a model to examine in greater detail the relationship between pharmacological pressure, cell stress and the induction of PD-L1. Our results imply that drug induced PD-L1 expression occurs in the broader context of cell-stress, without conferring acquired drug-resistance. Furthermore, a balance between BC cytotoxicity, induction of cell-stress and the overexpression of PD-L1 can be achieved through the selection of appropriate combinations of anti-cancer compounds. Therefore, we propose that drug combination can be employed not only for increasing the direct kill of cancer cells, but also as a strategy to minimize the activation of immunosuppressive and cancer cell pro-survival program responses during drug treatment.
Similar content being viewed by others
Introduction
Chemotherapy is one of the three standard treatment modalities for cancer treatment, along with surgery and radiation. In the last two decades, the development of new anti-cancer drugs has been focused on therapeutic agents that target oncogenic proteins that are specifically mutated or overexpressed in cancer cells. Examples include targeted therapeutics that are directed to block a specific oncogenic pathway (e.g. imatinib)1,2,3 or target (e.g. trastuzumab)4,5 in malignant cells. Included in this focus is the small molecule inhibitor - SI-2 - developed in our laboratory to target steroid receptor coactivator (SRC) members such as SRC-3 (SRC-3/NCOA3/AIB1/RAC3) and SRC-1/2 (SRC-1/NCOA1 and SRC-2/NCOA2/TIF2)6 All members of this oncogenic family of coactivators are overexpressed in a broad array of cancer subtypes7.
An emerging area for cancer therapy has been focused on the use of immune check point inhibitors to boost immune system for the destruction of tumors8,9. Check point blockade is a systemic approach which mostly targets the patient’s immune system, rather than the cancer cells themselves, in order to unleash the immune response and reactivate exhausted T cells to act against cancer cells10. Immune check-point blockade has emerged as a game changing approach in clinical oncology for the treatment of selected types of malignancies9. Of note are the encouraging survival rates and durability of response in tough-to-cure types of cancers such as in advanced metastatic melanomas11,12,13. Meanwhile, in breast and certain other cancers, the response rate for immune checkpoint inhibitors has not been as favorable14 in spite of the often significant interaction between breast tumors and the immune system15,16,17. Therefore, a strong impetus exists to design enhancements for the response of breast cancer patients to immunotherapy, possibly by combining it with either standard chemotherapy or targeted cancer drugs. Balancing the direct effects of chemotherapy on the breast tumor and their impact on the anti-cancer activity of the immune system is complex and can possess both beneficial and harmful effects18,19.
The beneficiary ‘side effects’ of chemotherapy on the anti-cancer immunity is modeled on an ICD paradigm, which is associated with specific chemotherapeutics and is predicated on the release of certain damage-associated molecular content from dying cancer cells20,21. In contrast, harmful effects of chemotherapy on anti-cancer immunity have been associated with the induction of PD-L1 - a central immunoregulatory protein that is expressed in both normal and cancer cells. PD-L1 engages its ligand – programmed death-1 (PD-1) - on activated immune effector cells, and signals the termination of effector cell proliferation and blocks pro-survival cytokine production, resulting in effector T cell death22,23,24,25. Two predicted recent studies have shown that PD-L1 expression on BC cells is induced following chemotherapeutic treatment26,27. In this context, it would be of interest to further assess the impact of different chemotherapeutics on the immunogenicity of BC cells representing different molecular subtypes.
In the present study we exploit a panel of four BC cell lines, representing triple negative breast cancer (TNBC) and ER + types, from both human and mouse species and apply a broad panel of BC small molecule therapeutics to measure the expression of PD-L1 as a result of drug exposure. We demonstrate that the majority of chemotherapeutic agents induce strong expression of PD-L1 as well as other pro-survival genes that are associated with cell stress. We show that a significant decrease in PD-L1 and cell-stress gene expression can be achieved by employing certain combinations of two different agents, which suggests that combinational drug treatment could be beneficial not only for their enhanced potential to directly kill cancer cells, but also as a strategy to effect breast cancer cell killing in a way that evades the immunosuppressive effects of elevated PD-L1 expression and activation of cancer cell pro-survival programs.
Results
Chemotherapeutic agents and targeted small molecule agents induce PD-L1 expression in breast cancer cell lines
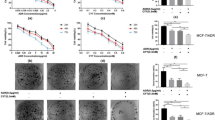
Recent studies have shown that PD-L1 expression in a variety of cancers is upregulated following exposure to diverse chemotherapeutics with distinct mechanisms of action26,28,29,30,31. In order to better understand the impact of anti-cancer drug treatment on cancer cell-autonomous expression of PD-L1 in breast/mammary gland cancer, four breast cancer (BC) cell lines - representing both TNBC and ER + – were used; MDA-MB-231 and 4T1 represent TNBC in humans and mice, and MCF-7 and E0771 represent ER + BC in humans and mice. The cells have been treated with a panel of six drugs/drug candidates with distinct mechanisms of inhibitory activity: doxorubicin (DOX), paclitaxel (PTX), Abemaciclib (ABE), Topotecan (TPTCN), BEZ235 and SI-2 representing respectively a topoisomerase-2 inhibitor, microtubulin inhibitor, CDK (cyclin dependent kinase)4/6 inhibitor, topoisomerase-1 inhibitor, PI3K-mTOR dual inhibitor and SRC-3 inhibitor6. Following exposure to a cytotoxic dose of each molecule – which was set at ~50% growth inhibition (GI) concentration (Fig. S1) – PD-L1 mRNA induction was observed in an overwhelming majority of cases (Fig. 1A). Because it can be used as an aggressive and ER + immunocompetent in vivo tumor model – E0771 cells were tested with additional molecules: cis-platin (cisPt), Palbociclib, Niraparib and methotrexate (MTX). Among the cell lines that we tested, E0771 is the most responsive model in terms of induced PD-L1 expression as a result of drug treatment. Among the tested molecules, DOX and TPTCN brought about the highest amounts of PD-L1 expression in E0771 cells. SI-2 was found to be the next strongest inducer of PD-L1 mRNA expression. Treatment of E0771 cells with SI-2 resulted in a time- and dose-dependent PD-L1 mRNA induction (Fig. 1B), which correlates with cell surface expression of PD-L1 (Fig. 1C). Collectively this data shows that PD-L1 is induced in E0771 breast cancer cells proportionally to the intensity of drug pressure.
Standard chemotherapeutics, targeted agents and an SRC inhibitor induce PD-L1 expression. (A) Cell lines representing either TNBC or ER+ subtypes of BC/mouse mammary carcinoma were treated with cytotoxic concentrations of the mentioned compounds (~IC50) and the expression of PD-L1 was determined by qPCR. (B) SI-2 induced PD-L1 mRNA expression in E0771 cells in a time and dose dependent manner. Cells were exposed to SI-2 at a concentration of 20 nM and PD-L1 mRNA was quantitated at 48 and 72 hr. (C) E0771 cells were treated with SI-2 for 72 hr with the indicated doses of SI-2 and then PD-L1 protein levels on the cell membrane were assessed by flow cytometry. n.s., not significant, ***P < 0.005, **P < 0.05, two-tailed Student’s t-test.
Drug induced PD-L1 expression takes place through a cellular stress response pathway
It has been reported that PD-L1 functions not only as a key regulator of T-cell exhaustion, but that it also has a cancer cell autonomous pro-survival function that can promote tumor metastasis and drug resistance32,33,34,35. To evaluate whether the overexpression of PD-L1 is taking place within the broader context of a cell stress survival program, we compared the quasi-physiological mode of PD-L1 induction with interferon-γ (IFN-γ) with drug-related induction. Under physiological conditions, IFN-γ signaling is reported to act through the JAK-STAT1-IRF1 axis to exert robust PD-L1 induction, whereas loss-of-function mutilations in JAK1/2 are correlated with loss of PD-L1 expression36,37. In our experimental model, IFN-γ-induced PD-L1 expression also was found to take place through the conventional JAK-STAT1-IRF1 pathway (Fig. 2A,B). However, during the induction of PD-L1 with anti-cancer compounds, upstream markers of the JAK-STAT1-IRF pathway were absent (Fig. 2B), suggesting that under stressful conditions there is an alternative pathway for PD-L1 expression, similar to an alternative cytokine signaling independent inflammation38.This form of cell stress-related, but not IFN-γ signaling context PD-L1 expression, could be associated with the emerging role of PD-L1 as a cell autonomous factor that is responsible for cancer cell survival and resistance to drug treatment32,39,40. This assumption is supported by the observed drug-induced upregulation of cell stress markers (Fig. 2C) which are known to be involved in oncogenic, cancer cell pro-survival processes35,41,42.
Stimulation of PD-L1 expression with IFN-γ results in activation of IFN-γ related signaling pathway components. (A,B) E0771 cells were treated with 100 ng/ml IFN-γ and Western analyses for IRF-1, STAT1, pSTAT1 (S727), PD-L1 and HSP-90 was performed indicated that IFN-γ stimulated canonical components of the IFN-γ signaling pathway. In contrast, treatment of E0771 cells with SI-2 induced PD-L1 without elevating pSTAT1 or IRF-1. (C) SI-2 induced the expression of cell stress response proteins ATF4, IRE1α, p-IRE1α and XBP1s. E0771 cells were treated for 24 hr with the indicated concentrations of SI-2. ***P < 0.005, two-tailed Student’s t-test. For each group of WB figures (A–C): all the blots were performed in the same experimental setup and processed in parallel. All gels were loaded with identical amount of protein sample. Dashed separating lines indicate that the blots were run in parallel but on separate gels. Full length blots are available in the supplementary information.
PD-L1 induction and cell stress are concomitantly regulated by drug combination
Our results shown above demonstrate that each small-molecule drug has a different impact on the magnitude of PD-L1 mRNA induction (Fig. 1A) with fold change values at the mRNA level in E0771 cells varying from ~10 fold (PTX and ABE) to ~102 fold (DOX and TPTCN). Intrigued by these differences, we tested the effects of a combination treatment with strong and weak PD-L1 inducers to explore which one of them possesses the dominant impact on PD-L1 induction. To this purpose, SI-2 (strong) and ABE (weak) were combined for the first test. Surprisingly, combined treatment with these two drugs did not produce an additive effect on PD-L1 induction but rather resulted in PD-L1 expression levels that resembled the weaker PD-L1 inducer - ABE (Fig. 3A top and 3A bottom right), without reducing cancer cell cytotoxicity (Fig. S2). Exposure of cells to ABE alone at a range of concentrations did not change PD-L1 levels -which remained similar to the untreated control cells (Fig. 3A bottom left). These observations indicate that by itself, ABE has a neutral to moderate-inducing effect on protein expression of PD-L1 but is able to suppress PD-L1 overexpression that would be otherwise induced by SI-2. Bearing in mind the possibility that under pharmacological pressure, PD-L1 expression might be associated with activation of stress-related survival programs, we tracked the expression of the stress marker activating transcription factor 4 (ATF4) under conditions of combinatorial ABE/SI-2 treatment and found that the addition of ABE - up to 625 nM - to SI-2 doesn’t result in further upregulation of ATF4 (Fig. 3B). Moreover, exposure of E0771 cells to lethal doses of ABE alone (up to 1.25 µM), didn’t upregulate the expression of ATF4 (Fig. 3C).
Combined treatment with the CDK4/6 inhibitor abemaciclib (ABE) and SI-2 can be strongly cytotoxic without inducing PD-L1 expression. (A) E0771 cells were treated with SI-2 or ABE or both for 72 hr at the indicated concentrations. Increased concentrations of ABE result in reduced PD-L1 expression compared to SI-2 alone, on protein (top) and mRNA (bottom right) levels. Compared to SI-2, cytotoxic concentrations of ABE alone do not bring about strong induction of PD-L1 mRNA (bottom right) or protein levels (top right, bottom left). (B) Addition of up to 625 nM ABE to SI-2 does not induce ATF4 expression compared with SI-2 alone. Western analysis of ATF-4 protein expression was determined 24 hr after treatment with the indicated concentrations of SI-2 and/or ABE. (C) Exposure of E0771 cells to cytotoxic concentrations of ABE for 24 hr only moderately induces the expression of ATF4. ***P < 0.005, **P < 0.05, two-tailed Student’s t-test. For each group of WB figures (A–C): all the blots were performed in the same experimental setup and processed in parallel. All gels were loaded with identical amount of protein sample. Dashed separating lines indicate that the blots were run in parallel but on separate gels. Full length blots are available in the supplementary information.
These observations indicate that cell stress pathway induction could be uncoupled from cancer cell cytotoxicity by choosing specific drug combinations. This suggests that it might be possible to better kill malignant cells through an appropriate combination of drugs and administration regimen that does not activate PD-L1 expression or cellular stress pathways. We refer to this fashion of cell death of the cancer cell a ‘silent death’ which might be desirable for killing cancer cells without enacting the immunosuppressive effects of PD-L1 expression. Further investigation of the mechanisms by which some anti-cancer agents induce cytotoxicity without inducing PD-L1 expression or cellular stress pathways is needed to understand this process fully at a molecular level.
PD-L1 induction is transient and not associated with cancer cell acquired resistance to drug pressure
Recent studies have demonstrated that there is a correlation between PD-L1 expression and cancer cell intrinsic drug resistance43. In order to investigate this correlation in greater detail in our BC cell line models, we induced PD-L1 overexpression in E0771 cells either pharmacologically (by SI-2 or DOX) or by mimicking physiological conditions (exposure to IFN-γ). Following treatment, the cells were sorted into PD-L1hi and PD-L1neg populations (Fig. 4A) and cultured for a recovery period of seven days after which both groups were measured for PD-L1 expression levels again. After the recovery period, PD-L1 levels in cells which initially had high PD-L1 expression return to the basal expression state (Fig. 4B). To test the possibility that transient PD-L1 induction is a marker for the activation of a stable, long-term pro-survival program, we performed viability assays, which indicated that PD-L1hi and PD-L1low cells respond similarly to drug treatment, either SI-2 or DOX, independently on their initial PD-L1 expression levels (Fig. 4C). These observations indicate that PD-L1 induction is more likely to take place and to remain highly elevated under ‘continuous’ stressful conditions.
Cell populations expressing elevated PD-L1 levels do not acquire short-term resistance to SI-2. (A) E0771 cells were treated with either IFN-γ (24 hr) or SI-2 or DOX (72 hr) at the indicated concentrations that then sorted into PD-L1 negative and PD-L1 positive populations. (B) After allowing cells to recover from compound exposure for seven days, PD-L1 levels were assessed again by flow cytometry, revealing that PD-L1 expression returned to basal levels of expression. (C) Cells from both the PD-L1 negative and positive populations treated with either SI-2 or IFN-γ were treated again with a dose range of SI-2 or doxorubicin (DOX) after the seven day recovery period and their viability was determined by MTS assay.
Discussion
Expression of the immunosuppressive PD-L1 protein on the surface of cancer cells has been established as having a major role in the ability of malignant cells to evade immune responses23,44,45. Moreover, several studies have shown that PD-L1 upregulation in cancerous and pre-malignant cells possesses non-immune functionalities associated with cancer-initiation, tumor cell-intrinsic cell survival and tumor progression32,46,47,48. In this context, the upregulation of PD-L1 by cancer cells in response to drug exposure implies that this is part of a pro-survival program. In agreement with previous studies26,27, here we show that PD-L1 is overexpressed in cancer cells as a result of treatment with a variety of anti-cancer agents; exposure of BC cell lines to various small-molecule therapeutics resulted in a general phenomenon of PD-L1 upregulation, through a non-canonical signaling pathway that does not involve IFN-γ. However, from our cell-based experiments, we found that drug-induced PD-L1 overexpression didn’t result in the acquisition of intrinsic cancer cell resistance to a second round of drug treatment. Indeed, after the removal of drug pressure, PD-L1 expressed returned to a low, basal level. This observation suggests that it might be possible to time the delivery of chemotherapeutic treatment with immune checkpoint inhibitors or other anti-cancer agents in a way that results in a more optimal outcome. We also show that it might be possible, through the pre-selection of appropriate cancer drug combinations, to achieve cancer cell growth inhibition in a way that does not trigger highly elevated PD-L1 expression. We speculate that many/most current standard-of-care therapies for BC are likely to elevate PD-L1 expression. Because the transient induction of this immunosuppressive protein only occurs in response to drug stress-pressure, pre-intervention analyses of BC biopsies fail to adequately account for the dynamic expression of PD-L1 during chemotherapy. Testing of chemotherapeutics, targeted therapeutics and checkpoint inhibitors in neoadjuvant window-of-opportunity studies may be useful to find ways to promote BC tumor cell cytotoxicity without inducing PD-L1 or other pro-survival stress related pathways.
Conclusions
Strong induction of PD-L1 expression was observed in a panel of BC cells exposed to various small molecule anti-cancer drugs. Drug-induced PD-L1 expression was taking place in the context of cell stress, implying that its activation is part of a pro-survival program in response to drug pressure. We found however, that the activation of the cell-intrinsic pro-survival programs, which manifested through cell-autonomous induction of PD-L1 and upregulation of cell stress pathways, could be avoided with specific drug-combinations. We demonstrated the concept of such as drug-combination by utilizing the SRC-3 inhibitor SI-2 alongside the CDK4/6 inhibitor abemaciclib. This combination resulted in a cancer-cell killing effect that was accompanied by only moderate upregulation of PD-L1 and cell-stress markers. The ability of ABE to suppress PD-L1 expression levels might be attributed to its ability to induce cell cycle arrest at G149. We refer to this effect as a ‘silent death’ to point out that it is possible to achieve potent BC cytotoxicity while also preventing BC tumor cells from activating immunosuppressive and pro-survival programs which could latter lead to the expansion of drug-resistant populations of cancer cells. We also found that the induction of PD-L1 that arose from the anti-cancer agents that we tested occurred outside of the established IFN-γ pathway and is not specific to inhibition of the steroid receptor coactivator signaling.
Materials and Methods
Cell culture
E0771, 4T1, MCF-7 and MDAMB-231 cell lines were obtained from American type culture collection (ATCC). E0771, MCF-7 and MDA-MB-231 cells were maintained in Dulbecco’s modified eagle media (DMEM). 4T1 cells were maintained in Roswell park memorial institute (RPMI) medium. All media were supplemented with 10% fetal bovine serum (FBS), 1% Glutamax (ThermoFisher 35050061) and 1% penicillin/streptomycin. Cells were grown at 37 °C under 5% CO2 atmosphere. Stimulation with Interferon gamma (IFN-γ) was performed by treating cells for 24 hr with 100 ng/ml recombinant human (Biolegend #570206) or mouse (Biolegend #575306) IFN-γ for human or mouse cell lines, respectively.
Cell viability assay
Cells were seeded on 96 well plates (E0771 and MDAMB-231 at 3,000 cells/well, 4T1 at 6,000 cells/well and MCF-7 at 10,000 cells/well) and allowed to adhere overnight. Media was then removed and cells were provided with fresh compound(s)-containing media. After indicated periods of treatment, drug-containing media was replaced with fresh media containing MTS ([3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium, inner salt]) reagent (Promega cat no. G3582) and then the cells were incubated for an additional 1–4 hr. MTS absorbance measurements were then performed with a Multiskan FC Microplate Photometer plate reader (ThermoFisher) at 490 nm. After blank (media only) normalization, cell viability was calculated relative to vehicle-treated cells. Each point reflects at least four replicates.
All commercial small molecule drugs were obtained from MCE. SI-2 was synthesized as previously described6.
Immunoblotting
Whole cell lysates were obtained using RIPA Lysis and extraction buffer (ThermoFisher, cat no. 89900). Following ice-cold incubation with extraction buffer for 30 min, the lysates were centrifuged at 4 °C, 15000 rpm for 15 min after which the supernatants were collected and protein concentrations were calculated using the Pierce BCA (bicinchoninic acid assay) protein assay kit (ThermoFisher, cat no. 23225). The supernatants were proportionally mixed with 4x Laemmli sample buffer (BioRad, cat no. 1610747) containing 10% 2-mercaptoethanol and boiled for five min at 95 °C. Equal protein amounts were brought to equal volumes with sample buffer and run on 4–20% precast polyacrylamide gels (BioRad Mini-PROTEAN TGX Precast Protein Gels). The protein samples were transferred to nitrocellulose membranes using an iBlot Gel Transfer Device (ThermoFisher), blocked with PBS (phosphate-buffered saline)-Tween buffer, containing 5% nonfat milk dissolved powder for 60 min. After this, relevant primary antibodies were added and the membranes were incubated over-night at 4 °C (Table S1). After thorough washing with PBS, the membranes were incubated at room temperature for 60 min in PBS-Tween buffer containing 5% nonfat milk supplied with a relevant HRP (horseradish peroxidase)-conjugated secondary antibody. Membranes were then washed three times with PBS and protein bands were detected with either enhanced chemiluminescence (ECL) substrate (ThermoFisher, cat no. 32106) or Pico PLUS Chemiluminescent substrate (ThermoFisher, cat no. 34580).
Flow cytometry
Cells were harvested, washed with PBS and the cell pellet was obtained by brief centrifugation. Cell pellets was then rinsed in 100–200 μL staining buffer (BioLegend, cat no. 420201) and PE-conjugated CD274 antibody was added (BioLegend, cat no. 124308, 1/100). Following 45–60 min incubation on ice, cells were washed with PBS and resuspended in 0.5–1 ml staining buffer. Before subjecting cells to FACS analysis, all the samples were passed through a 0.40 μm filter and additionally stained with DAPI (4′,6-diamidino-2-phenylindole, Invitrogen, cat no. R37606), except for auto-fluorescence control samples. For all samples a ‘full minus one’ (FMO) control was performed50. Samples were run on a LSRII cell analyzer (Becton and Dickinson) and sorted with a FACSArial cell sorter (Becton and Dickinson) using the Diva software package (Becton and Dickinson). Analyses were performed with either Diva or FlowJo_v10 software. Normalized MFI (mean fluorescent intensity) was calculated for all samples relatively to the untreated control group. Graphs were generated with Prism 7 software (GraphPad Software, Inc.).
Real time quantitative polymerase chain reaction (RT qPCR)
RNA was extracted from cells using TRIzol reagent (Invitrogen). Reverse transcription cDNA synthesis was performed with the VILO SuperScript cDNA Synthesis Kit (Invitrogen) using 2 μg of total RNA. Probe-based real time quantitative PCR was performed using Universal Probe Library probes (Roche) and TaqMan Universal Master Mix (Applied Biosystems) on a StepOnePlus Real-Time PCR machine (Applied Biosystems). Primers were designed using the Roche ‘universal probe library assay design center system’ (Table S2). Relative mRNA expression of CD274 was calculated by the ΔΔCT method with normalization to GAPDH or ACTB. Each result is represented by SD of at least three technical replicates.
Change history
05 March 2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Druker, B. J. et al. Five-Year Follow-up of Patients Receiving Imatinib for Chronic Myeloid Leukemia. N Engl J Med 355, 2408–2417 (2006).
Roskoski, R. A historical overview of protein kinases and their targeted small molecule inhibitors. Pharmacological Research 100, 1–23 (2015).
Zhang, J., Yang, P. L. & Gray, N. S. Targeting cancer with small molecule kinase inhibitors. Nature Reviews Cancer 9, 28 (2009).
Greenberg, P. A. et al. Long-term follow-up of patients with complete remission following combination chemotherapy for metastatic breast cancer. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 14, 2197–2205 (1996).
Slamon, D. J. et al. Use of Chemotherapy plus a Monoclonal Antibody against HER2 for Metastatic Breast Cancer That Overexpresses HER2. N Engl J Med 344, 783–792 (2001).
Song, X. et al. Development of potent small-molecule inhibitors to drug the undruggable steroid receptor coactivator-3. Proc Natl Acad Sci USA 113, 4970–4975 (2016).
Lonard, D. M. & O’Malley, B. W. Molecular Pathways: Targeting Steroid Receptor Coactivators in Cancer. Clin Cancer Res 22, 5403–5407 (2016).
Callahan, M. K., Postow, M. A. & Wolchok, J. D. Targeting T Cell Co-receptors for Cancer Therapy. Immunity 44, 1069–1078 (2016).
Ribas, A. & Wolchok, J. D. Cancer immunotherapy using checkpoint blockade. Science 359, 1350–1355 (2018).
Wei, S. C., Duffy, C. R. & Allison, J. P. Fundamental Mechanisms of Immune Checkpoint Blockade Therapy. Cancer Discov 8, 1069–1086 (2018).
Wolchok, J. D. et al. Guidelines for the evaluation of immune therapy activity in solid tumors: immune-related response criteria. Clin Cancer Res 15, 7412–7420 (2009).
Schadendorf, D. et al. Pooled Analysis of Long-Term Survival Data From Phase II and Phase III Trials of Ipilimumab in Unresectable or Metastatic Melanoma. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 33, 1889–1894 (2015).
Ott, P. A., Hodi, F. S. & Robert, C. CTLA-4 and PD-1/PD-L1 blockade: new immunotherapeutic modalities with durable clinical benefit in melanoma patients. Clin Cancer Res 19, 5300–5309 (2013).
Polk, A., Svane, I. M., Andersson, M. & Nielsen, D. Checkpoint inhibitors in breast cancer - Current status. Cancer Treat Rev 63, 122–134 (2018).
Ali, H. R., Chlon, L., Pharoah, P. D., Markowetz, F. & Caldas, C. Patterns of Immune Infiltration in Breast Cancer and Their Clinical Implications: A Gene-Expression-Based Retrospective Study. PLoS Med 13, e1002194 (2016).
Varn, F. S., Mullins, D. W., Arias-Pulido, H., Fiering, S. & Cheng, C. Adaptive immunity programmes in breast cancer. Immunology 150, 25–34 (2017).
Binnewies, M. et al. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nature Medicine 24, 541–550 (2018).
Garg, A. D. & Agostinis, P. Cell death and immunity in cancer: From danger signals to mimicry of pathogen defense responses. Immunol Rev 280, 126–148 (2017).
Galluzzi, L., Buque, A., Kepp, O., Zitvogel, L. & Kroemer, G. Immunological Effects of Conventional Chemotherapy and Targeted Anticancer Agents. Cancer Cell 28, 690–714 (2015).
Krysko, D. V. et al. Immunogenic cell death and DAMPs in cancer therapy. Nat Rev Cancer 12, 860–875 (2012).
Kroemer, G., Galluzzi, L., Kepp, O. & Zitvogel, L. Immunogenic cell death in cancer therapy. Annu Rev Immunol 31, 51–72 (2013).
Boussiotis, V. A. Molecular and Biochemical Aspects of the PD-1 Checkpoint Pathway. N Engl J Med 375, 1767–1778 (2016).
Sun, C., Mezzadra, R. & Schumacher, T. N. Regulation and Function of the PD-L1 Checkpoint. Immunity 48, 434–452 (2018).
Francisco, L. M., Sage, P. T. & Sharpe, A. H. The PD-1 pathway in tolerance and autoimmunity. Immunol Rev 236, 219–242 (2010).
Keir, M. E., Butte, M. J., Freeman, G. J. & Sharpe, A. H. PD-1 and its ligands in tolerance and immunity. Annu Rev Immunol 26, 677–704 (2008).
Samanta, D. et al. Chemotherapy induces enrichment of CD47(+)/CD73(+)/PDL1(+) immune evasive triple-negative breast cancer cells. Proc Natl Acad Sci USA 115, E1239–e1248 (2018).
Zhang, P., Su, D. M., Liang, M. & Fu, J. Chemopreventive agents induce programmed death-1-ligand 1 (PD-L1) surface expression in breast cancer cells and promote PD-L1-mediated T cell apoptosis. Mol Immunol 45, 1470–1476 (2008).
Peng, J. et al. Chemotherapy Induces Programmed Cell Death-Ligand 1 Overexpression via the Nuclear Factor-κB to Foster an Immunosuppressive Tumor Microenvironment in Ovarian Cancer. Cancer Res, (2015).
Jiao, S. et al. PARP Inhibitor Upregulates PD-L1 Expression and Enhances Cancer-Associated Immunosuppression. Clin Cancer Res, (2017).
Leduc, C. et al. TPF induction chemotherapy increases PD-L1 expression in tumour cells and immune cells in head and neck squamous cell carcinoma. ESMO Open 3, (2018).
Wargo, J. A., Reuben, A., Cooper, Z. A., Oh, K. S. & Sullivan, R. J. Immune Effects of Chemotherapy, Radiation, and Targeted Therapy and Opportunities for Combination With Immunotherapy. Semin Oncol 42, 601–616 (2015).
Clark, C. A. et al. Tumor-Intrinsic PD-L1 Signals Regulate Cell Growth, Pathogenesis, and Autophagy in Ovarian Cancer and Melanoma. Cancer Res 76, 6964–6974 (2016).
Dong, P., Xiong, Y., Yue, J., Hanley, S. J. B. & Watari, H. Tumor-Intrinsic PD-L1 Signaling in Cancer Initiation, Development and Treatment: Beyond Immune Evasion. Front Oncol 8, 386 (2018).
Hsu, J. M. et al. STT3-dependent PD-L1 accumulation on cancer stem cells promotes immune evasion. Nat Commun 9, 1908 (2018).
Avril, T., Vauleon, E. & Chevet, E. Endoplasmic reticulum stress signaling and chemotherapy resistance in solid cancers. Oncogenesis 6, e373 (2017).
Shin, D. S. et al. Primary Resistance to PD-1 Blockade Mediated by JAK1/2 Mutations. Cancer Discov 7, 188–201 (2017).
Garcia-Diaz, A. et al. Interferon Receptor Signaling Pathways Regulating PD-L1 and PD-L2 Expression. Cell Rep 19, 1189–1201 (2017).
Garg, A. D. et al. ER stress-induced inflammation: does it aid or impede disease progression? Trends in molecular medicine 18, 589–598 (2012).
Zhang, P. et al. Upregulation of programmed cell death ligand 1 promotes resistance response in non-small-cell lung cancer patients treated with neo-adjuvant chemotherapy. Cancer Sci 107, 1563–1571 (2016).
Black, M. et al. Activation of the PD-1/PD-L1 immune checkpoint confers tumor cell chemoresistance associated with increased metastasis. Oncotarget 7, 10557–10567 (2016).
Wang, M. & Kaufman, R. J. The impact of the endoplasmic reticulum protein-folding environment on cancer development. Nat Rev Cancer 14, 581–597 (2014).
Chevet, E., Hetz, C. & Samali, A. Endoplasmic reticulum stress-activated cell reprogramming in oncogenesis. Cancer Discov 5, 586–597 (2015).
Chen, J., Jiang, C. C., Jin, L. & Zhang, X. D. Regulation of PD-L1: a novel role of pro-survival signalling in cancer. Ann Oncol 27, 409–416 (2016).
Noguchi, T. et al. Temporally Distinct PD-L1 Expression by Tumor and Host Cells Contributes to Immune Escape. Cancer Immunol Res 5, 106–117 (2017).
Juneja, V. R. et al. PD-L1 on tumor cells is sufficient for immune evasion in immunogenic tumors and inhibits CD8 T cell cytotoxicity. J Exp Med 214, 895–904 (2017).
Dong, P., Xiong, Y., Yue, J., Hanley, S. J. B. & Watari, H. Tumor-Intrinsic PD-L1 Signaling in Cancer Initiation, Development and Treatment: Beyond Immune Evasion. Frontiers in oncology 8, 386–386 (2018).
Gupta, H. B. et al. Tumor cell-intrinsic PD-L1 promotes tumor-initiating cell generation and functions in melanoma and ovarian cancer. Signal Transduct Target Ther 1, (2016).
Escors, D. et al. The intracellular signalosome of PD-L1 in cancer cells. Signal Transduction and Targeted Therapy 3, 26 (2018).
Ingham, M. & Schwartz, G. K. Cell-Cycle Therapeutics Come of Age. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 35, 2949–2959 (2017).
Hulspas, R., O’Gorman, M. R. G., Wood, B. L., Gratama, J. W. & Sutherland, D. R. Considerations for the control of background fluorescence in clinical flow cytometry. Cytometry Part B: Clinical Cytometry 76B, 355–364 (2009).
Acknowledgements
Cell sorting was supported by the Cytometry and Cell Sorting Core (CCSC) at Baylor College of Medicine with funding from the CPRIT Core Facility Support Award (CPRIT-RP180672), P30 Cancer Center Support Grant (NCI-CA125123), NIH-RR024574, and NIH S10 OD025251 (Union BioMetrica BioSorter).
Author information
Authors and Affiliations
Contributions
Y.G., Y.E. S.J.H., B.W.O., and D.M.L. contributed to the conception and design of the study and interpretation of the results. Y.G. performed all the experiments, collected, analyzed and visualized the data. Y.Y. participated in cell based growth inhibition assays. Y.E. participated in data analysis and visualization. Y.Y. was responsible for administrative support. Y.G., B.W.O. and D.M.L. wrote the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gilad, Y., Eliaz, Y., Yu, Y. et al. Drug-induced PD-L1 expression and cell stress response in breast cancer cells can be balanced by drug combination. Sci Rep 9, 15099 (2019). https://doi.org/10.1038/s41598-019-51537-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-51537-7
This article is cited by
-
Bio-orthogonal click chemistry strategy for PD-L1-targeted imaging and pyroptosis-mediated chemo-immunotherapy of triple-negative breast cancer
Journal of Nanobiotechnology (2024)
-
Boron Derivatives Inhibit the Proliferation of Breast Cancer Cells and Affect Tumor-Specific T Cell Activity In Vitro by Distinct Mechanisms
Biological Trace Element Research (2023)
-
Steroid receptor coactivator-3 inhibition generates breast cancer antitumor immune microenvironment
Breast Cancer Research (2022)
-
PD-L1 induction via the MEK-JNK-AP1 axis by a neddylation inhibitor promotes cancer-associated immunosuppression
Cell Death & Disease (2022)
-
Anti-PD-L1 mediating tumor-targeted codelivery of liposomal irinotecan/JQ1 for chemo-immunotherapy
Acta Pharmacologica Sinica (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.