Abstract
Anecdotic citations of food wasting have been described for parrots, but we lack a comprehensive knowledge about the extent of this behaviour, and its ecological and evolutionary implications. Here, we combine experimental and observational approaches to evaluate the spatial, temporal, typological and taxonomic extent of food wasting by parrots, to identify the ecological and evolutionary factors driving food wasting, and to assess the incidence of two ecological functions derived from food wasting, such as food facilitation to other animal species and secondary seed dispersal. We found that food wasting is a widespread behaviour found in all the studied parrot species. However, the proportion of food wasted differed among species and throughout the year. Parrots wasted more food during the non-breeding season, when they relied on exotic plants and on unripe fruits or seeds. We also recorded 86 animal species feeding on the food wasted by parrots, 27 of which potentially acted as secondary seed dispersers. Overall, our study emphasizes the universality of food wasting among parrots, and the important implications that this behaviour may have for the species involved (i.e., the parrot, the plant, the other species feeding on wasted food), and for the functioning of the whole ecosystem.
Similar content being viewed by others
Introduction
The way species interact with each other may have overarching implications for community organization and ecosystem functioning1,2,3. For example, several studies have documented that mutualistic animal-plant interactions, such as seed dispersal or pollination, drive important coevolutionary forces shaping the structure of both animal and plant communities4,5. Likewise, animal species may use each other as indicators of habitat quality, affecting the final distribution of the species in the communities6. Therefore, it is important to identify how species interact with each other and to understand the mechanisms shaping these interactions.
An interesting understudied behaviour related to interacting frugivorous animals and fruit-producing plants is food wasting (hereafter, waste). Howe7 found that monkeys discarded almost two-thirds of the seeds they handled, while Bosch & Wedde8 described how some parrots ate fruits only partially, discarding the rest. Anecdotic citations of waste by parrots can also be found in other dietary studies, both in places where animal species are native9,10,11,12 or exotic13. However, we lack a comprehensive understanding the ecological and evolutionary implications of this behaviour, as well as its implications.
Food wasting is illogical under classical ecological theories of resource use, where optimal foragers move between alternative patches14 or diet items (e.g.15) to prevent the total depletion of a particular resource. Predators may also maintain prey consumption within sustainable limits to ensure the viability of prey species16. Then, why do animals waste food? Some authors propose that waste may be an accidental behaviour. Frugivorous and granivorous species may unintentionally drop fruits and seeds while handling them during the foraging process17,18, larger fruits being involuntarily dropped with a higher frequency than small ones17. Other unexplored factors that may affect accidental dropping are conspecific disturbance or the coevolutionary history of the animal and the plant species. Conspecific density may affect waste, as individuals foraging in larger groups may drop more food than those in small ones. Besides, animals may drop more fruits from exotic than from native plants because they have had shorter evolutionary times together to get used to them. However, frugivores may also deliberately drop low-quality fruits such as those parasitized11, unripe, or those with low energetic and nutritional content19. In these cases, if waste is not accidental, individuals may be able to adjust this behaviour according to food availability, as dropping food when it is abundant does not have a large energetic cost.
Despite the uncertainty about why parrots waste food, several studies have suggested that this behaviour may benefit other species9,10,13. For example, dropped fruits or seeds become available to ground dwelling animals that otherwise cannot benefit from them9,10,13. Besides eating them, these species can also act as secondary dispersers9,10,11,12,13, increasing the number of different dispersal modes for the plant involved and, thus, the chances of effective dispersal. However, we do not know to what extent wasted food is consumed or secondarily dispersed by other species20.
Parrots (Psittaciformes order) are an evolutionarily old and diversified animal group21 where waste has been anecdotally observed, but never quantified. Therefore, we combined experimental approaches with fieldwork conducted in five continents to understand the importance of this behaviour across parrot species, its drivers and the consequences for other species. Table 1 summarizes the hypothesis tested about waste by parrots, as well as the associated predictions. Briefly, we evaluate the spatial, temporal, typological and taxonomic extent of waste by parrots. If waste is an anecdotal behaviour, we expect to find it only in few parrot species. Otherwise, if waste is a widespread behaviour, we would find it in most parrot species, independently of their evolutionary history and their region of origin.
Then, we assessed if waste is a random process, independent of parrot life traits. To so do, we looked for differences in waste occurrence and amount of food wasted among species to identify the ecological factors driving waste. We hypothesized that this behaviour can be either accidental (i.e. plant parts fall because of difficulties during handling) or deliberate (i.e. parrots decide to waste food). Accidental waste may happen with a higher frequency when individuals forage in large groups, because of conspecifics disturbance, or with exotic plants, because parrots are less used to handle them. Contrarily, if waste is deliberate, this behaviour should happen more frequently with low-quality fruits and seeds (e.g. parasitized and unripe), and when energetic requirements are softer (e.g. outside the reproductive season, or when food availability is high). We also predict that smaller species will waste less food because they have a faster metabolism than large species (e.g.22) and need to optimize food intake. Lastly, we hypothesized that wasted food can benefit other species, so we assessed the extent and incidence of two possible ecological functions derived from waste: food facilitation to other animal species and seed dispersal. If wasted food is beneficial for the plant and for other species, we expect that the number of seeds underneath the trees where parrots have wasted food will be large and will attract many species. We also expect that waste will benefit a large number of different species with different potential dispersal modes. Finally, we discuss some possible ecological functions of waste and their ecological and evolutionary consequences.
Results
Food wasting extent and randomness
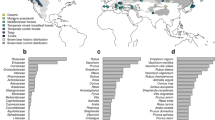
Waste by parrots is a widespread behaviour in many aspects (P1, Fig. 1). It was found in all the study sites visited (35 biomes, 17 countries and 5 continents), in native and introduced parrot ranges. Waste was observed throughout the year, in both the breeding and non-breeding season. Parrots wasted mainly fruits and seeds, but also flowers, leaves, twigs, stems, sprouts, parasites and bark. We observed 103 parrot species (40 species in the experimental approach, 75 in the field and 12 in both the experiment and the field) from 38 genera wasting food from 336 plant species belonging to 80 families (see Supplementary Tables S1 and S2 for complete lists of parrots wasting food and plants where waste occurred).
The spatial, temporal, typological and taxonomic extent of food wasting by parrots: Spatial: Each circle shows a surveyed area. Blue circles represent areas where parrots are exotic species, while red circles are areas included in their native distribution. Temporal: The graph shows the total number of waste observations per month by all parrot species (light green) and, specifically, by Psittacula kramerii (dark green). Typological: Number of observations for different plant parts wasted by parrots. Others include invertebrates, stems, sprouts, resin and tree bark. Taxonomic: Number of bird species found wasting food and plant species involved. Pictures: Poicephalus meyerii (left) wasting Terminalia sericea fruits and seeds (right), pictures by J.L. Tella. Icons authors: Georgiana Ionescu (fruit), Shawn Erdely (seed), Mansion@design (flower), Noël Rasendrason (twig), Myly (leave) and Lisa Staudinger (asterisk), all from thenounproject.com.
Following our expectations (P2), waste was independent of the evolutionary history of the species, as it did not show a significant phylogenetic signal using any of the 100 phylogenies used (mean K: 0.104, range: 0.007–0.165, all P > 0.409). Besides, we found that the percentage of waste by parrots was large (P3), as it averaged 21.2% (SD = 13.0) of the total food provided to individuals during an in-captivity experiment (range: 0.4–72.7%). In the wild, we found that parrots wasted 11.8% (SD = 25.1) of the fruits and 14.6% (SD = 20.3) of the seeds that they handled, ranging from lack of waste to a waste of all the fruits or up to 80% of the seeds.
Moreover, we found strong among-species differences in waste occurrence in the wild (δAIC with null model 2882) and in the proportion of food wasted during the experiment (δAIC with null model 12, Fig. 2), suggesting that food wasting is not a random process (P4).
Factors driving food wasting
Using a large database of field observations about waste occurrence we identified some factors affecting the occurrence of this behaviour. Fruit and seed wasting were more frequent during the non-breeding season (P10), in exotic plant species (P6) and on unripe fruits or seeds (P9) for all the species. We also repeated the analyses considering only data from P. krameri, which is the parrot species with more detailed information, and we observed the same pattern, both including all observations collected across the world and only those observed in Seville, the population with more information (Table 2, Fig. 3). This last analysis corroborates results obtained using the whole data. However, the analyses for all species but excluding P. krameri indicate that the season was the only factor driving waste occurrence.
Representation of the food wasting occurrence (food wasted vs. food not wasted) in relation to the ripening stage of the fruit/seed (unripe/ripe), the log-flock size, the season when the observation was taken (breeding/non-breeding), and the origin of the plant (native/exotic) where the observation was taken.
Results obtained using experimental data showed that species size (i.e. average body mass, P7), food availability (i.e. fasting, P8) and conspecifics disturbance (i.e. number of individuals in the cage, P5) did not affect the proportion of food wasted (all p-values > 0.14, Supplementary Table S3). Finally, we also found that the total proportion of wasted fruits that were parasitized was very low (<4%, n = 176, P11).
Ecological functions of food wasting
By checking the amount of food wasted under the trees where parrots had been wasting food, we found that this was large in many cases, as expected (P12). We found an average (±SD) of 53.4 ± 52.2 wasted fruits and 42.0 ± 54.9 wasted seeds under the trees (Supplementary Table S4). The maximum number of seeds found under a single tree (239) was larger than the number of fruits (164).
The number of species that benefited from the food wasted by parrots was also large and widely distributed, supporting P13, including 86 species of birds, mammals, reptiles, fishes, and ants (Table 3) that widely ranged in body size (0.002–750 kg). Also, we detected 28 different species from 26 genera potentially acting as secondary dispersers of the fruits and seeds wasted, including ants, birds, mammals and reptiles. As predicted (P14), these species ranged from very small to very large body sizes, and their average dispersal distances were mainly large (>30 m), but also very large (>100 m) for two species.
Discussion
In this paper, we show that waste is not an anecdotic behaviour among parrots, but a widespread habit present in at least the 103 studied species. Waste is mainly focused on fruits and seeds, although it also involves other plant parts, and occurs throughout the year and through all the regions where parrots have native or introduced populations. We also show that waste has implications for other species that can forage or secondarily disperse wasted seeds and fruits. Thus, we can consider that parrots are interacting with plants along mutualism-antagonism continuums, as they can both prey upon several plant parts but also act as seed-dispersers, pollinators or plant healers12,23.
Besides waste being a widespread behaviour in parrots, we found that the proportion of food wasted changed between species, suggesting that some ecological or evolutionary factor may be modulating this behaviour. However, our tests to disentangle the accidental or deliberate nature of waste were not conclusive. We found that parrots waste more food from non-native plant species than from native ones, maybe because they had a shorter co-evolutionary history to adjust their handling techniques to the fruits and seeds of these novel species. Also, exotic plant species are often located closer to human infrastructures and thus, waste may be a consequence of human disturbance (i.e. parrots suddenly flying away and dropping food when humans get close to them). We also found that waste is more frequent during the non-breeding than during the breeding season, which is probably related to the higher nutritional requirements that individuals have while raising chicks24. If individuals need to gather more food for themselves and their chicks during the same time, they may choose to reduce food waste to increase foraging efficiency, suggesting that waste may also be deliberate. Also, parrots seem to waste with a higher frequency low quality (i.e. unripe) fruits, showing some type of selectivity. However, we did not find differences in food waste in relation to species body size or food availability (i.e. fasting), suggesting that metabolic differences among species and food availability are not driving this behaviour. Finally, some studies had suggested that parrots might preferentially waste or consume parasitized fruits, modulating the effect of parasites on plant fitness11,25. However, our observations did not show any preferential consumption of parasitized fruits.
Food wasted by parrots benefits many animal species, as seeds and fruits become more readily available and during a longer temporal window after being thrown from the tree by parrots. Also, wasted food is less often rotten or dry, as happens when food naturally falls from the trees. Parrots waste plenty of half-eaten fruits, reducing the original size of the fruit and breaking hard fruit parts, thus allowing their consumption by smaller species. For example, Douglas et al.26 found that the quantity of parrot frugivory increased habitat quality for Bananaquits (Coereba flaveola), because of parrot ability to open hard fruits and leave them partially consumed and thus available for other species. Besides, fruits become available for ground-dwelling species that otherwise cannot access the resource (as already suggested in9,10,13).
Fruits and seeds wasted by parrots can also be subject to secondary dispersal by many animals. This may be vital for some plant species, as many plants rely on secondary dispersal in their reproductive cycle27,28. It is important to underline that even if many of the wasted fruits and seeds are unripe and partially consumed, a large proportion of them can ripen and germinate after dispersal20,29,30. Also, having seed dispersers with different ecological characteristics (e.g. home ranges, places to defecate, dispersing methods) may increase the chances of effective seed dispersal28. The list of species either consuming food discarded by parrots or acting as secondary dispersers is very large, and includes many species from different taxonomic groups and with very different body sizes and dispersal distances. It is worth mentioning that our list is very conservative, as many of the species that we observed consuming the wasted fruits and seeds can also potentially disperse them. Thus, the impact of this wasteful behaviour on a large list of animal and plant species may be large given its universality.
Besides food facilitation and secondary seed dispersal, waste may have other ecological functions. If this behaviour has been maintained over the evolutionarily history of this old and diversified animal group21, it may be because it has some beneficial consequences for parrots12. One suggestion is that waste may benefit parrots by increasing the availability of high quality (e.g. large-sized, sugar-rich and nutrient-rich) fruits. In horticulture, it is widely known that such large high-quality fruits can be obtained through fruit and flower pruning31,32. Interestingly, pruning needs to be directed to unripe fruits to affect the quality of the non-wasted fruits33,34,35, coinciding with our result that waste occurs with a higher frequency in unripe fruits. Moreover, fruit and flower pruning are also known to reduce gaps between fructifications36 or reduce biennial bearing32,37,38. Thus, parrots may also be extending the fruiting period of the trees and increasing their predictability (i.e. shorter gaps between fructifications and transition from biennial to annual bearing). For any of these hypotheses to be possible, parrots need to be able to make intertemporal choices (i.e. sacrifice short-term satisfaction to obtain a higher reward in the future), which have already been detected for this bird group39. Overall, waste may have longer-time effects on wasted plants than expected, but further studies are needed to validate this hypothesis.
Besides the apparent negative effect that fruit and flower wasting may have for the plant, it is widely known that many plant species naturally produce more flowers and fruits than they can set36. The overproduction of juvenile fruits may be evolutionarily adaptive as these can satiate pre-dispersal fruit and seed predators40,41 and plants may be able to produce larger yields than average in occasional years of plentiful resources42,43. Plants may also compensate this overproduction by aborting flowers or fruits during the growth process36. As wasted fruits are preferentially unripe, their loss can be partially compensated by reducing fruit or flower abortion, reducing the negative effect of fruit thinning for the plant.
Waste may also have important consequences for the soil, as large amounts of organic matter accumulate over short time periods under a single tree. This behaviour may also expedite nutrient cycling, as wasted plant parts may enter the decomposition phase faster than if they were to stay in the tree for longer time periods12. Overall, our study wants to emphasize the universality of waste in parrots and the important implications this behaviour may have for the species involved (i.e. plants and parrots), but also show how other species benefit from the wasted food and for the functioning of the ecosystem.
Two specifications about our study need to be done before concluding. First, it is important to notice that the experimental data cannot be directly compared with what happens in the natural environment as the food provided and the way parrots obtain is markedly different. However, the waste experiment was designed to control for several factors that would be very challenging to account for in the wild. For example, it is very difficult to test among-species differences on the wild because the species are never found under the same circumstances (e.g., environmental conditions, food species). Another important factor that cannot be tested in the field but was easily addressed in the experiment was food availability. Therefore, the experiment offers complementary information to that provided by the data gathered in the field. Also, in our predictions 7–11, we assume that an individual wasting less food has a higher foraging efficiency than an individual wasting more food. However, it may be more efficient to feed by partially eating and wasting various fruits than to handle the same fruit for a long time, which will become smaller and more difficult to eat. This does not invalidate our predictions, as about half of the fruits and seeds wasted are intact (see Supplementary Table S3).
Despite the several aspects that deserve more research, this study adds to the growing evidence that parrots have a much more important role for the conservation of the ecosystems than previously thought23,44. Some of the functions attributed to parrots, such as primary and secondary dispersal [12,45, this study], may have a very important role in the current scenario of global change where tropical forests are being fragmented at high rates and forest recovery may depend on the efficiency of seed dispersal. However, many parrot species are highly endangered, with several species functionally extinct and many of the remaining species under strong human pressure (e.g., 28% of extant species are classified as threatened under IUCN46). Thus, management efforts to conserve parrot species should considerate the ecological interactions of food waste in the conservation planning strategies of habitat protection and population recovery (e.g. selection of priority areas for conservation and selection of release sites).
Methods
Datasets used
To test our hypotheses about waste, we combined information gathered using different approaches and unified in five different datasets (All of them available as Supplementary Material).
Dataset 1
Experiment in captivity. We first quantified waste experimentally using captive individuals. We performed 362 experimental trials using 130 individuals from 40 parrot species belonging to 24 genera (body weight range: 33–550 g) to measure the proportion of food wasted by the species. Individuals were kept in groups of 1–4 individuals, simulating their typical aggregated foraging behaviour. As cages used for the experiment always have the same size, experiments done including a larger number of individuals simulate situations of higher conspecific disturbance. Parrots were acclimated to the cages and diet for 10 days prior to the experiment. In a third of the experiments, individuals were fasted 24 hours before the start of the experiment, to simulate low food availability. Each species was fed with the typical food used in captivity (e.g. mixtures of millet, birdseed, sunflowers, and peanuts). Food was weighted (±0.1 g) and provided ad libitum to the parrots. Cages were set so that wasted food could not be eaten by individuals after falling to the cage floor. Each group of birds was tested approximately five times, and each trial lasted 24 hours. Wasted food was weighted, including only those seeds that were intact (i.e. half consumed seeds were not included). Finally, we calculated the proportion of food wasted by each group of parrots in each trial. All experimental protocols are in accordance with the relevant guidelines and regulations. Birds were kept in captivity under permit SGYB/FOA/AFR from the Consejería de Medio Ambiente, Junta de Andalucía, in the authorized centre for experimental avian research SE/16/U (REGA ES410910008016).
Dataset 2
Field transects. We collected observations of waste in different fieldwork campaigns performed in 17 countries and 5 continents (Fig. 1a). We looked for groups of foraging parrots in pre-defined roadside and walking transects (see details about transects on47). Every time we detected a foraging group, we observed focal parrot groups or individuals for 5–10 minutes and annotated the occurrence of food wasting (1/0), the parrot species, the plant species consumed, the part of the plant that was wasted (flowers, fruits, seeds, bark, leaves, twigs, sprouts, stems, resin or invertebrates), flock size, the season when the observation was taken (breeding/non-breeding), the ripening stage of the fruits/seeds (unripe/ripe), date, site and the origin of the plant where the observation was taken (native/exotic). This dataset includes a total of 6253 observations of foraging parrots observed between 2011 and 2019 in 37,612 km of transects.
Dataset 3
Waste quantification. We quantified the proportion of food wasted in the field by observing the foraging behaviour of individuals detected handling fruits or seeds during the field transects. For this and the following datasets, and for the statistical analyses in this study, we focused on fruits or seeds and excluded other wasted plant parts because fruits and seeds are the main food types wasted by parrots (see Fig. 1). Foraging individuals were observed from a distance with binoculars or telescopes. We identified the bird and plant species, and we counted the flock size and the number of fruits/seeds each individual ate or wasted. We then calculated the proportion of food wasted as the number of wasted fruits/seeds divided by the total number of fruits/seeds handled. We compiled 412 observations of individual birds from 20 species in Bolivia, Costa Rica, Namibia, Brazil, Peru, Argentina and Spain, between 2014 and 2019. Data was collected during six different months in both the breeding and non-breeding season, and birds handled 1841 fruits and 934 seeds. As some studies suggest that parrots may be wasting parasitized fruits or seeds (e.g.11), we also counted the total number of wasted fruits with worms for 176 fruits under 7 different tree species during fieldwork in the Brazilian cerrado in 2017.
Dataset 4
Waste under tree. We estimated the number of fruits/seeds a group of parrots could waste per individual tree. To do so, we counted the total number of intact and wholly or partially eaten fruits/seeds under a tree after a group of parrots foraged on it. We also identified the plant species. When the number of fruits was very large or the area was hard to screen because of the dense vegetation, we counted half of the area under the tree and then doubled the number of fruits/seeds. We compiled information on 98 trees from 29 species in Australia, Peru, Ecuador, Bolivia and Brazil between 2013 and 2017.
Dataset 5
Camera traps and direct observations. We used 96 camera traps to monitor the animal species using fruits and seeds wasted by parrots. Cameras were located under the plant, in front of a bunch of fruits/seeds. They stayed activated 5–7 days during 24 hours. Data was gathered in Brazil and Bolivia, under four different plant species where waste had been observed: Attalea totai, A. barreirensis, A. speciosa and Mauritia flexuosa. From the pictures, we separated species that consumed the fruits/seeds and those that took entire fruits/seeds out of the camera, thus being possible secondary seed dispersions. We combined this information with 293 direct observations of food facilitation and secondary dispersal taken in Australia, Spain, Puerto Rico, South Africa, Argentina and Sri Lanka between 2012 and 2019.
Food wasting extent
To test our first prediction (P1) and describe the spatial extent of waste, we identified all the areas around the world where we had observed waste in the wild. The temporal extent of waste was described as the total number of waste events recorded per month. We also calculated the total number of waste events for ring-necked parakeets Psittacula krameri, the species with the largest number of waste events detected. For the temporal extent we used the Field transects dataset, as this is the largest compilation of waste events taken using a standardized method. This same dataset was used to identify the typological extent of waste by counting the total number of waste events found for each plant type (flower, fruit, seed, bark, leaves, twigs, sprouts, stems or invertebrates). Finally, the taxonomic extent of waste by parrots was quantified by identifying the total number of parrot species that was found wasting food and the total number of plant species that were subject to waste by parrots in any of our datasets, and in non-systematic observations performed during fieldwork.
Our second prediction (P2) that waste is independent of the evolutionary history of the species was explored using data from the Experiment. We assessed if there was a phylogenetic signal in the proportion of food experimentally wasted by the different species using the descriptive statistics K48. When K < 1, the relatives resemble each other less than expected under the Brownian motion evolution, while when K > 1 close relatives are more similar than expected under the Brownian evolution. We evaluated the statistical significance of the phylogenetic signal by comparing the observed variance of independent contrasts of the proportion of food experimentally wasted by the different parrot species to a null model of shuffling taxa labels across the tips of the phylogeny. We calculated K and the statistical significance of the phylogenetic signal for 100 bird phylogenies from Jetz et al.49 using the picante package50 in R version 3.5.351.
Finally, we evaluated the proportion of food wasted (P3) using two datasets: the Experiment and the Waste quantification. We used the average (±SD) proportion of food wasted by each group of parrots in each trial for the Experiment data and the average (±SD) number of wasted fruits/seeds by each individual parrot for the Waste quantification data.
Waste differences among species
If food wasting is a not a random process (P4) waste occurrence and quantity should differ among species. We used the Field transects data to compare waste occurrence (1/0) among species and the Experiment dataset to compare the proportion of food wasted among species. To test if waste occurrence differed between species, we fitted a Generalized Linear Model (GLM) in R with species as a predictor variable and occurrence as a response variable, using a binomial distribution. We then compared the proportion of food wasted (response variable) among species (predictor variable) in the experiment by means of Generalized Linear Mixed Models (GLMM) using a beta distribution with the glmmADMB library52. Because the same individuals were used for different trials, we included individual (or group of individuals) as a random factor in all the models. In both cases, we compared the model including species as predictor variable with a null one. Models with a difference in AIC smaller than 10 were considered equally supported.
Factors driving food wasting
Waste may be driven by different factors, depending on its accidental or deliberate behaviour (predictions 5–11, Table 1). Because the different factors affecting waste may be related, we fitted multivariate models including several predictor variables at the same time. We ran one model for waste occurrence (1/0) (Field transects dataset) and one for the proportion of wasted food (Experiment dataset). Experiment data were used to relate the number of individuals in the cage during the trial (P5), parrot body size (mean weight in g, P7) and the reduction in food availability (simulated by a fasting period, P8) (predictor variables) with the proportion of food wasted (dependent variable) by means of GLMMs using a beta distribution. We included individual (or group of individuals) as a random factor in all the models, nested within species. The weight of the birds was standardized before modelling (i.e. transformed to have a mean of 0 and standard deviation of 1).
Then, we evaluated factors affecting waste occurrence (1/0) in relation with the season when the observation was taken (breeding/non-breeding, P10), the ripening stage of the fruit/seed (unripe/ripe, P9) and the origin of the plant where the observation was taken (native/exotic, P6). We also included the number of individuals in the flock as a covariate in the models to control for the potential effect of larger flocks having a higher chance of showing waste. To test the consistency of the results, we performed the analyses for all the species, but also for the species with the largest number of observations (P. krameri); additionally, only for P. krameri in the study site with a larger number of observations (Seville, Spain, where the species is introduced) and finally, for all species excluding P. krameri. We fitted Generalized Linear Models (GLM) in R using a binomial error distribution for the models for P. krameri and GLMMs with species as a random term for the model with all the species and for the model with all the species, but excluding P. krameri.
As some studies suggest that parrots may be wasting parasitized fruits or seeds (P11), we calculated the proportion of wasted fruits or seeds that were parasitized for 176 fruits found underneath seven different tree species.
Ecological functions of food wasting
To test if waste is large under the tree (P12), we used the Waste under tree dataset. We calculated the mean (±SD) number of intact and partially eaten fruits/seeds under a tree. We also identified the maximum number of intact and partially eaten fruits and seeds found.
We finally evaluated two possible ecological functions of waste by parrots, facilitation of food to other species and secondary seed dispersal (P13 & P14), using the Camera traps and direct observations dataset. For each species benefiting from wasted food, we identified its taxonomic group (i.e. ant, bird, mammal, reptile or fish) and estimated its body size (i.e. very large [>10 kg], large [>1 kg], medium [>0.1 kg], small [>0.01 kg], very small [<= 0.01 kg]) using published studies (see reference list in Dataset). For those species detected acting as secondary dispersers, we also identified the mean dispersal distance from the literature (i.e. very large [>100 m], large [>30 m], medium [>10 m], small [<= 10 m]).
Data availability
All data generated or analysed during this study are included in this published article (and its Supplementary Information Files).
References
Bascompte, J. & Jordano, P. Plant-animal mutualistic networks: The architecture of biodiversity. Annu. Rev. Ecol. Syst. 38, 567–593 (2007).
Bastolla, U. et al. The architecture of mutualistic networks minimizes competition and increases biodiversity. Nature 458, 1018–1020 (2009).
Sebastián-González, E. et al. Nested species-rich networks of scavenging vertebrates support high levels of interspecific competition. Ecology 97, 95–105 (2016).
Guimarães, P. R., Jordano, P. & Thompson, J. N. Evolution and coevolution in mutualistic networks. Ecol. Lett. 14, 877–885 (2011).
Guimarães, P. R., Pires, M. M., Jordano, P., Bascompte, J. & Thompson, J. N. Indirect effects drive coevolution in mutualistic networks. Nature 550, 511–514 (2017).
Sebastián-González, E., Sánchez-Zapata, J. A., Botella, F. & Ovaskainen, O. Testing the heterospecific attaction hypothesis with time-series data on species co-occurrence. Proc. Royal Soc. B 277, 2983–2990 (2010).
Howe, H. F. Monkey dispersal and waste in a neotropical fruit. Ecology 61, 944–959 (1980).
Bosch, K. & Wedde, U. Amazonen – Freileben, Haltung, Zucht, Arten (Horst Müller-Verlag Walsrode, Bomlitz 1981).
Galetti, M. Diet of the Scaly-headed Parrot (Pionus maximiliani) in a semideciduous forest in southeastern Brazil. Biotropica 25, 419–425 (1993).
Galetti, M. & Rodrigues, M. Comparative seed predation on pods by parrots in Brazil. Biotropica 24, 222–224 (1992).
Symes, C. T. & Perrin, M. R. Feeding biology of the Grey headed Parrot Poicephalus fiscicollis suahelicus (Reichenow) in Northern Province, South Africa. Emu 103, 49–58 (2003).
Blanco, G., Hiraldo, F. & Tella, J. L. Ecological functions of parrots: an integrative perspective from plant life cycle to ecosystem functioning. Emu 118, 36–49 (2018).
Martens, J., Hoppe, D. & Woog, F. Diet and feeding behaviour of naturalized Amazon Parrots in a European city. Ardea 101, 71–76 (2013).
Charnov, E. L. Optimal foraging: the marginal value theorem. Theoret. Pop. Biol. 9, 129–136 (1976).
Tinker, M. T., Bentall, G. B. & Estes, J. A. Food limitation leads to behavioural diversification and dietary specialization in sea otters. Proc. Nat. Acad. Sci. (USA) 105, 560–565 (2008).
Slobodkin, L. B. How to be a predator. Am. Zool. 8, 43–51 (1968).
Avery, M. L., Goocher, K. J. & Cone, M. A. Handling efficiency and berry size preferences of cedar waxwings. Wilson Bul. 105, 604–611 (1993).
Rey, P. J., Gutiérrez, J. E., Alcántara, J. & Valera, F. Fruit size in wild olives: implications for avian seed dispersal. Funct. Ecol. 11, 611–618 (1997).
Foster, M. S. Ecological and Nutritional Effects of Food Scarcity on a Tropical Frugivorous Bird and its Fruit. Ecology 58, 73–85 (1977).
Barnett, A. A. et al. Primary seed dispersal by three Neotropical seed-predating primates (Cacajao melanocephalus ouakary, Chiropotes chiropotes and Chiropotes albinasus). J. Trop. Ecol. 28, 543–555 (2012).
Toft, C. A. & Wright, T. F. Parrots of the Wild: A Natural History of the World’s Most Captivating Birds (University of California Press, 2015).
Lovegrove, B. G., Perrin, M. R. & Brown, M. The allometry of parrot BMR: seasonal data for the greater vasa parrot, Coracopsis vasa, from Madagascar. J Comp Physiol B 181, 1075–1087 (2011).
Montesinos-Navarro, A., Hiraldo, F., Tella, J. L. & Blanco, G. Network structure embracing mutualism-antagonism continuums increases community robustness. Nature Ecol. Evol. 1, 1661 (2017).
Martin, T. E. Food as a limit on breeding birds: a life-history perspective. Annu. Rev. Ecol. Syst. 18, 453–487 (1987).
Selman, R. G., Hunter, M. L. & Perrin, M. R. The feeding ecology of Rüppell’s Parrot Poicephalus rueppellii in Namibia. Ostrich 73, 127–134 (2002).
Douglas, L. R., Winkel, G. & Sherry, T. W. Does the Bananaquit Benefit Commensally from Parrot Frugivory? An Assessment Using Habitat Quality. Biotropica 45, 457–464 (2013).
Chambers, J. C. & MacMahon, J. A. A day in the life of a seed: movements and fates of seeds and their implications for natural and managed systems. Annu. Rev. Ecol. Syst. 25, 263–292 (1994).
Vander Wall, S. B. & Longland, W. S. Diplochory: are two seed dispersers better than one? Trends Ecol. Evol. 19, 297–314 (2004).
Tella, J. L. et al. Endangered plant-parrot mutualisms: seed tolerance to predation makes parrots pervasive dispersers of the Parana pine. Sci. Rep. 6, 31709 (2016).
Tella, J. L., Blanco, G., Dénes, F. V. & Hiraldo, F. Overlooked parrot seed dispersal in Australia and South America: insights on the evolution of dispersal syndromes and seed size in Araucaria trees. Front. Ecol. Evol. 7, 82 (2019).
Link, H. Significance of flower and fruit thinning on fruit quality. Plant Growth Regul. 31, 17–26 (2000).
Hehnen, D., Hanrahan, I., Lewis, K., McFerson, J. & Blanke, M. Mechanical flower thinning improves fruit quality of apples and promotes consistent bearing. Sci. Hortic. 134, 241–244 (2012).
Havis, L. Effects of time of fruit thinning of Redhaven peach. Proc. Amer. Soc. Hort. Sci. 80, 172–176 (1962).
Tukey, H. B. & Einset, O. Effect of fruit thinning on size, color, and yield of peaches and on growth and blossoming of the tree. Proc. Amer. Soc. Hort. Sci. 36, 314–319 (1938).
Dennis, F. J. The history of fruit thinning. Plant Growth Regul. 31, 1 (2000).
Stephenson, A. G. Flower and fruit abortion: Proximate causes and ultimate functions. Ann. Rev. Ecol. Syst. 12, 253–279 (1981).
Dag, A., Bustan, A., Avni, A., Lavee, S. & Riov, J. Fruit thinning using NAA shows potential for reducing biennial bearing of ‘Barnea’ and ‘Picual’ oil olive trees. Crop Pasture Sci. 60, 1124–1130 (2009).
Seehuber, C., Damerow, L. & Blanke, M. Regulation of source: sink relationship, fruit set, fruit growth and fruit quality in European plum (Prunus domestica L.)—using thinning for crop load management. Plant Growth Regul 65, 335–341 (2011).
Krasheninnikova, A., Höner, F., O’Neil, L., Penna, E. & von Bayern, A. M. P. Economic decision-making in parrots. Sci. Rep. 8, 12537 (2018).
Janzen, D. H. Seed predation by animals. Ann. Rev. Ecol Syst. 2, 465–92 69 (1971).
Janzen, D. H. Escape of Cassia grandis L. beans from predators in time and space. Ecology 52, 964–79 (1971).
Udovic, D. Determinants of fruit set in Yuccaw hipplei. Reproductive expenditure vs. pollinator availability. Oecologia 48, 389 (1981).
Wilson, M. F. & Price, P. W. The evolution of in florescence size in Asclepias (Asclepiadaceae). Evolution 1, 495–511 (1977).
Blanco, G., Hiraldo, F., Rojas, A., Dénes, F. V. & Tella, J. L. Parrots as key multilinkers in ecosystem structure and functioning. Ecol. Evol. 18, 4141–4160 (2015).
Tella, J. L. et al. Parrots as overlooked seed dispersers. Front. Ecol. Environ. 13, 338–339 (2015).
Olah, G. et al. Ecological and socio-economic factors affecting extinction risk in parrots. Biodivers. Conserv. 25, 205–223 (2016).
Tella, J. L., Rojas, A., Carrete, M. & Hiraldo, F. Simple assessments of age and spatial population structure can aid conservation of poorly known species. Biol. Conserv. 167, 425–434 (2013).
Blomberg, S. P., Garland, T. Jr. & Ives, A. R. Testing for phylogenetic signal in comparative data: behavioural traits are more labile. Evolution 57, 717–745 (2003).
Jetz, W. et al. The global diversity of birds in space and time. Nature 491, 444–448 (2012).
Kembel, S. W. et al. Picante: R tools for integrating phylogenies and ecology. Bioinformatics 26, 1463–1464 (2010).
R Development Core Team. R: A language and environment for statistical computing (R Foundation for Statistical Computing, 2019).
Skaug, H., Fournier, D., Nielsen, A., Magnusson, A & Bolker, B. Generalized Linear Mixed Models using AD Model Builder. R package version 0.7.5 (2013).
Acknowledgements
We thank L. Lima, M. de la Riva, A. Montesinos-Navarro for their help in the fieldwork. ESG was supported by Generalitat Valenciana (SEJI/2018/024) and Spanish Government (IJCI-2015-24947). ECP has PhD fellowship of CAPES-Brazil. JADL thanks to Centro de Biodiversidad y Genética (UMSS) for supporting the work as Institución Científica Acreditada, and Dirección General de Biodiversidad (MMyMA) for the permits (MMAyA/VMABCCGDF/UVSAPN°0122/2016). Field work was funded by Fundación Repsol, Fundación Biodiversidad, Severo Ochoa grant to EBD, World Parrot Trust, FPCILB and Loro Parque Fundación. Logistic and technical support for fieldwork were provided by Doñana ICTS-RBD.
Author information
Authors and Affiliations
Contributions
E.S.G. organized data, carried out statistical analyses and wrote the manuscript. F.H., G.B. and J.L.T. conceived and designed the study, and helped draft the manuscript. F.H., G.B., D.H.B., P.R.V., E.G.L., E.C.P., J.A.D.L., M.C., F.V.D. and J.L.T. collected field data and critically revised the manuscript. All authors gave final approval for publication and agree to be held accountable for the work performed therein.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sebastián-González, E., Hiraldo, F., Blanco, G. et al. The extent, frequency and ecological functions of food wasting by parrots. Sci Rep 9, 15280 (2019). https://doi.org/10.1038/s41598-019-51430-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-51430-3
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.